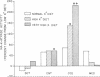
Abstract
Potassium secretion and sodium-potassium adenosine triphosphatase (Na-K-ATPase) activity in the distal nephron segments are known to be influenced by the dietary intake of K+. This has been attributed to a change in the plasma aldosterone level, which also influences K+ secretion and Na-K-ATPase activity in the distal nephron. To investigate whether or not dietary K+ can modulate Na-K-ATPase activity in the distal nephron independently of aldosterone, we determined Na-K-ATPase activity in four distinct nephron segments of adrenalectomized (adx) rabbits given four specific diets for 1 wk before experimentation. Na-K-ATPase activity was determined by a fluorometric microassay in which ATP hydrolysis is coupled to NADH oxidation. The nephron segments examined were the distal convoluted tubule (DCT), the connecting tubule (CNT), the cortical collecting duct (CCD), and the outer medullary collecting duct (MCD). All diets were similar in composition except for their K+ contents, which were 100, 300, 500, and 700 meq/kg in groups 1-4, respectively. In these adx animals, Na-K-ATPase activity increased greater than 200% in the CCD as the dietary intake of K+ increased. There was a linear relationship between K+ excretion and the enzyme activity in this segment. There was a 50% increase in Na-K-ATPase activity in the CNT as the dietary intake of K+ increased in adx animals. However, there were no significant differences in Na-K-ATPase activities in the DCT and MCD among the four treatment groups. It is concluded that dietary K+ intake can influence Na-K-ATPase activity in the CCD and CNT independently of plasma aldosterone levels.
Full text
PDF
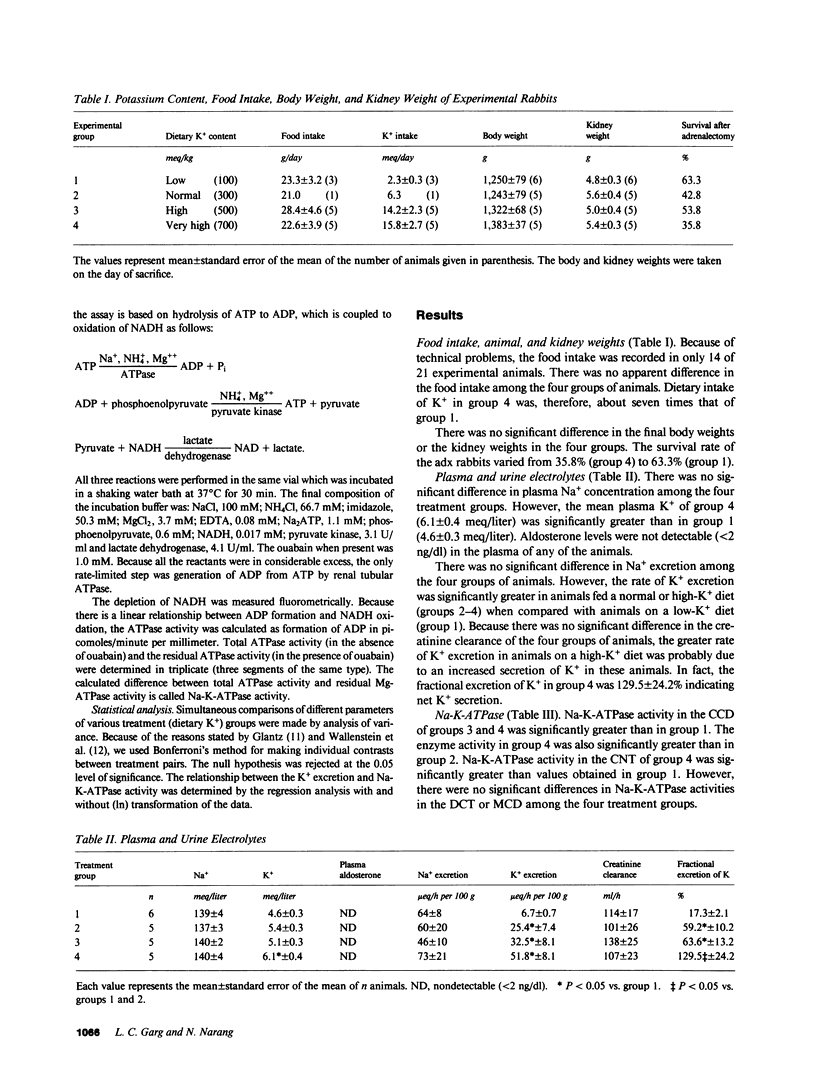
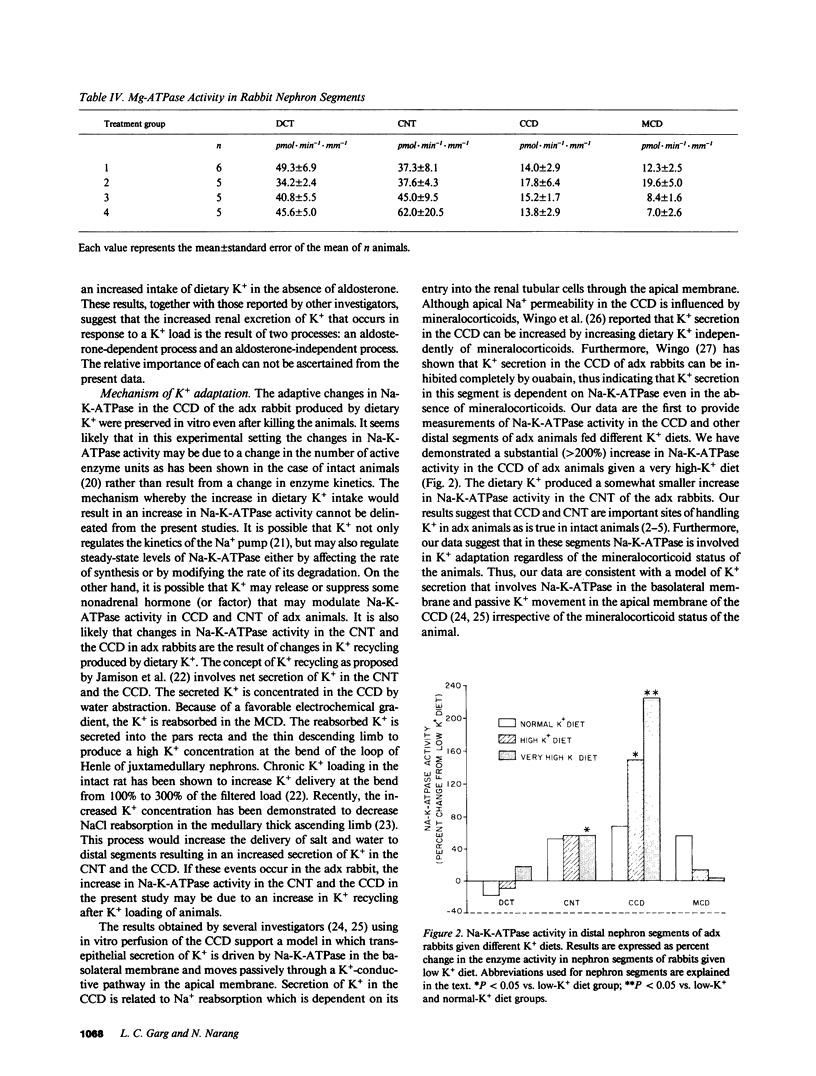


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander E. A., Levinsky N. G. An extrarenal mechanism of potassium adaptation. J Clin Invest. 1968 Apr;47(4):740–748. doi: 10.1172/JCI105769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bia M. J., DeFronzo R. A. Extrarenal potassium homeostasis. Am J Physiol. 1981 Apr;240(4):F257–F268. doi: 10.1152/ajprenal.1981.240.4.F257. [DOI] [PubMed] [Google Scholar]
- Boyd J. E., Mulrow P. J. Further studies of the influence of potassium upon aldosterone production in the rat. Endocrinology. 1972 Jan;90(1):299–301. doi: 10.1210/endo-90-1-299. [DOI] [PubMed] [Google Scholar]
- Boyd J. E., Palmore W. P., Mulrow P. J. Role of potassium in the control of aldosterone secretion in the rat. Endocrinology. 1971 Mar;88(3):556–565. doi: 10.1210/endo-88-3-556. [DOI] [PubMed] [Google Scholar]
- Doucet A., Katz A. I. Renal potassium adaptation: Na-K-ATPase activity along the nephron after chronic potassium loading. Am J Physiol. 1980 May;238(5):F380–F386. doi: 10.1152/ajprenal.1980.238.5.F380. [DOI] [PubMed] [Google Scholar]
- El Mernissi G., Doucet A. Short-term effects of aldosterone and dexamethasone on Na-K-ATPase along the rabbit nephron. Pflugers Arch. 1983 Oct;399(2):147–151. doi: 10.1007/BF00663911. [DOI] [PubMed] [Google Scholar]
- Garg L. C., Knepper M. A., Burg M. B. Mineralocorticoid effects on Na-K-ATPase in individual nephron segments. Am J Physiol. 1981 Jun;240(6):F536–F544. doi: 10.1152/ajprenal.1981.240.6.F536. [DOI] [PubMed] [Google Scholar]
- Garg L. C., Mackie S., Tisher C. C. Effect of low potassium-diet on Na-K-ATPase in rat nephron segments. Pflugers Arch. 1982 Aug;394(2):113–117. doi: 10.1007/BF00582911. [DOI] [PubMed] [Google Scholar]
- Garg L. C. The absence of renal bicarbonate reabsorption maxima during carbonic anhydrase inhibition. J Pharmacol Exp Ther. 1975 Jul;194(1):96–102. [PubMed] [Google Scholar]
- Glantz S. A. Biostatistics: how to detect, correct and prevent errors in the medical literature. Circulation. 1980 Jan;61(1):1–7. doi: 10.1161/01.cir.61.1.1. [DOI] [PubMed] [Google Scholar]
- Hayslett J. P., Binder H. J. Mechanism of potassium adaptation. Am J Physiol. 1982 Aug;243(2):F103–F112. doi: 10.1152/ajprenal.1982.243.2.F103. [DOI] [PubMed] [Google Scholar]
- Jamison R. L., Work J., Schafer J. A. New pathways for potassium transport in the kidney. Am J Physiol. 1982 Apr;242(4):F297–F312. doi: 10.1152/ajprenal.1982.242.4.F297. [DOI] [PubMed] [Google Scholar]
- Jorgensen P. L. Regulation of the (Na+ + K+)-activated ATP hydrolyzing enzyme system in rat kidney. I. The effect of adrenalectomy and the supply of sodium on the enzyme system. Biochim Biophys Acta. 1968 Jan 8;151(1):212–224. doi: 10.1016/0005-2744(68)90176-9. [DOI] [PubMed] [Google Scholar]
- Koeppen B. M., Biagi B. A., Giebisch G. H. Intracellular microelectrode characterization of the rabbit cortical collecting duct. Am J Physiol. 1983 Jan;244(1):F35–F47. doi: 10.1152/ajprenal.1983.244.1.F35. [DOI] [PubMed] [Google Scholar]
- Petty K. J., Kokko J. P., Marver D. Secondary effect of aldosterone on Na-KATPase activity in the rabbit cortical collecting tubule. J Clin Invest. 1981 Dec;68(6):1514–1521. doi: 10.1172/JCI110405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silva P., Hayslett J. P., Epstein F. H. The role of Na-K-activated adenosine triphosphatase in potassium adaptation. Stimulation of enzymatic activity by potassium loading. J Clin Invest. 1973 Nov;52(11):2665–2671. doi: 10.1172/JCI107460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stokes J. B. Consequences of potassium recycling in the renal medulla. Effects of ion transport by the medullary thick ascending limb of Henle's loop. J Clin Invest. 1982 Aug;70(2):219–229. doi: 10.1172/JCI110609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stokes J. B. Na and K transport across the cortical and outer medullary collecting tubule of the rabbit: evidence for diffusion across the outer medullary portion. Am J Physiol. 1982 May;242(5):F514–F520. doi: 10.1152/ajprenal.1982.242.5.F514. [DOI] [PubMed] [Google Scholar]
- Stokes J. B. Potassium secretion by cortical collecting tubule: relation to sodium absorption, luminal sodium concentration, and transepithelial voltage. Am J Physiol. 1981 Oct;241(4):F395–F402. doi: 10.1152/ajprenal.1981.241.4.F395. [DOI] [PubMed] [Google Scholar]
- Talbott J. H., Pecora L. J., Melville R. S., Consolazio W. V. RENAL FUNCTION IN PATIENTS WITH ADDISON'S DISEASE AND IN PATIENTS WITH ADRENAL INSUFFICIENCY SECONDARY TO PITUITARY PAN-HYPOFUNCTION. J Clin Invest. 1942 Jan;21(1):107–119. doi: 10.1172/JCI101272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallenstein S., Zucker C. L., Fleiss J. L. Some statistical methods useful in circulation research. Circ Res. 1980 Jul;47(1):1–9. doi: 10.1161/01.res.47.1.1. [DOI] [PubMed] [Google Scholar]
- Wingo C. S. Effect of ouabain on K secretion in cortical collecting tubules from adrenalectomized rabbits. Am J Physiol. 1984 Oct;247(4 Pt 2):F588–F595. doi: 10.1152/ajprenal.1984.247.4.F588. [DOI] [PubMed] [Google Scholar]
- Wingo C. S., Seldin D. W., Kokko J. P., Jacobson H. R. Dietary modulation of active potassium secretion in the cortical collecting tubule of adrenalectomized rabbits. J Clin Invest. 1982 Sep;70(3):579–586. doi: 10.1172/JCI110650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright F. S. Sites and mechanisms of potassium transport along the renal tubule. Kidney Int. 1977 Jun;11(6):415–432. doi: 10.1038/ki.1977.60. [DOI] [PubMed] [Google Scholar]