Abstract
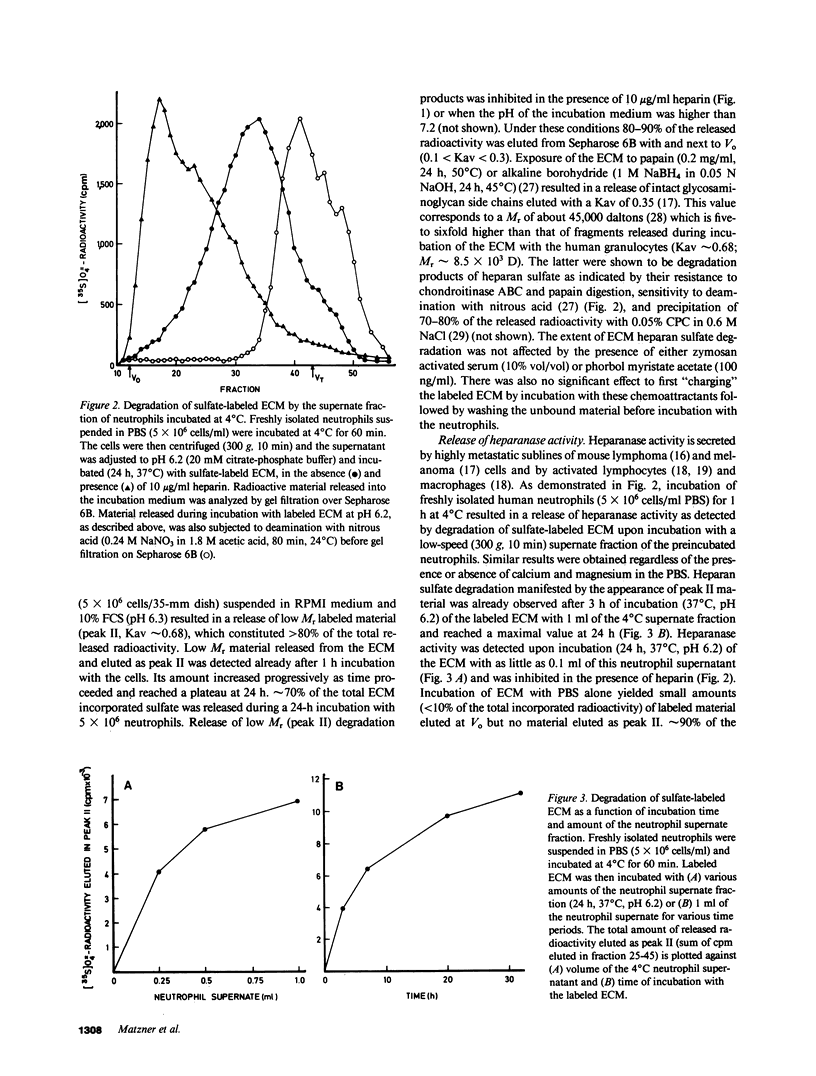
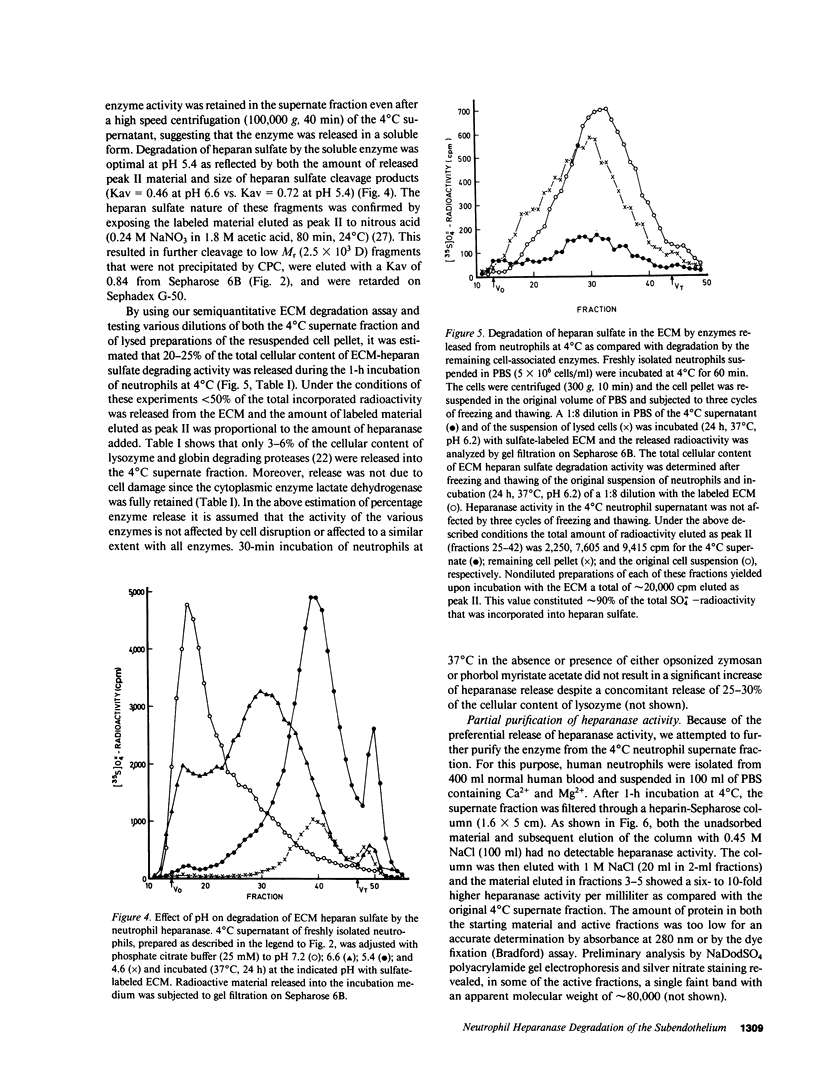
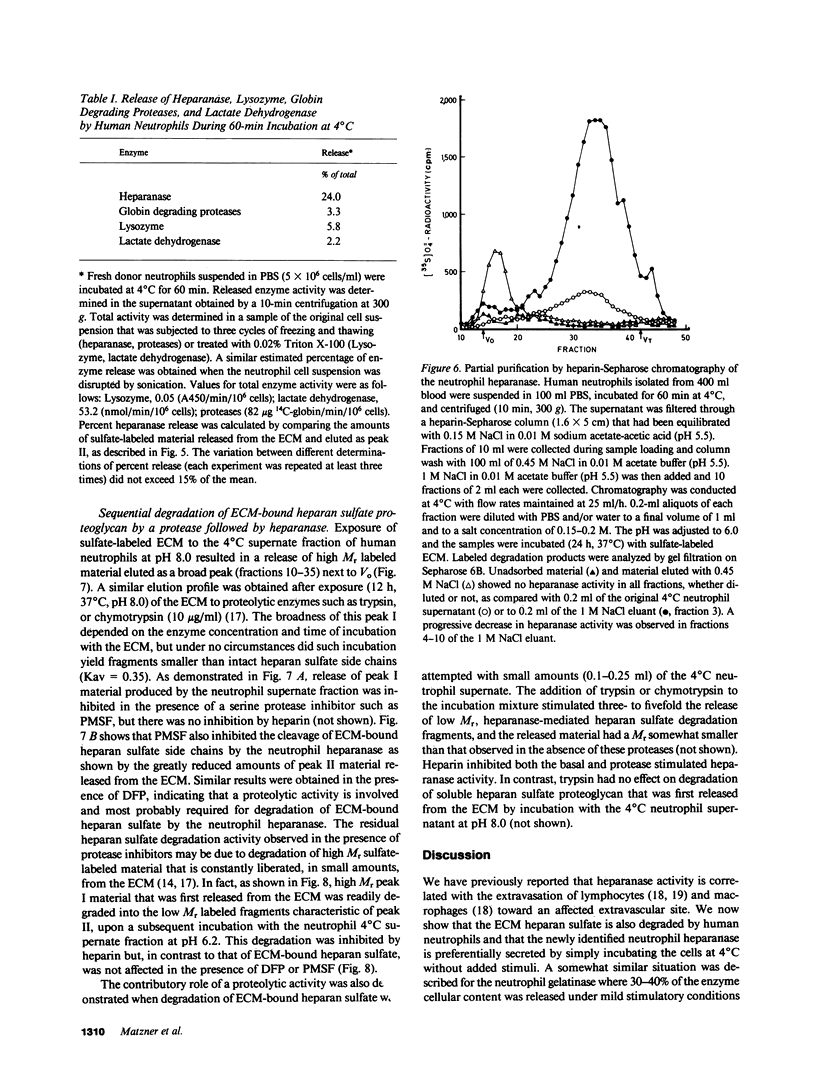
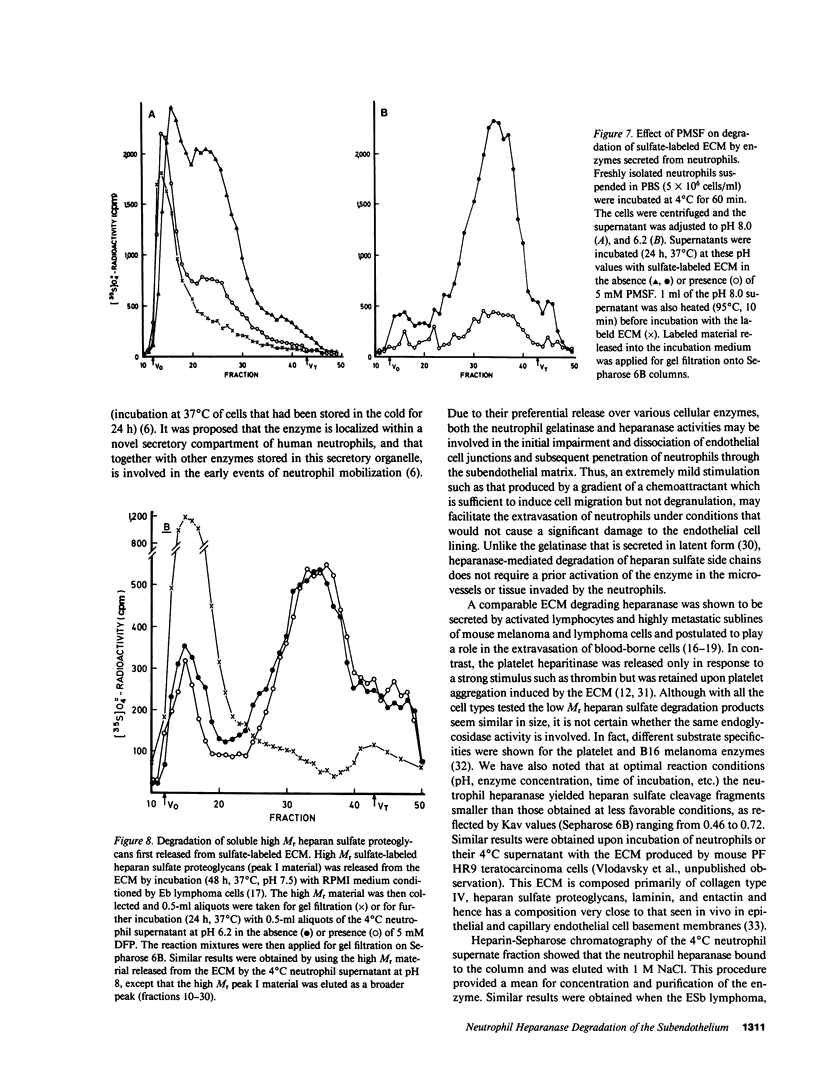
Freshly isolated human neutrophils were investigated for their ability to degrade heparan sulfate proteoglycans in the subendothelial extracellular matrix (ECM) produced by cultured corneal and vascular endothelial cells. The ECM was metabolically labeled with Na2(35S)O4 and labeled degradation products were analyzed by gel filtration over Sepharose 6B. More than 90% of the released radioactivity consisted of heparan sulfate fragments 5-6 times smaller than intact heparan sulfate side chains released from the ECM by either papain or alkaline borohydride. These fragments were sensitive to deamination with nitrous acid and were not produced in the presence of either heparin or serine protease inhibitors. In contrast, degradation of soluble high molecular weight heparan sulfate proteoglycan, which was first released from the ECM, was inhibited by heparin but there was no effect of protease inhibitors. These results indicate that interaction of human neutrophils with the subendothelial ECM is associated with degradation of its heparan sulfate by means of a specific, newly identified, heparanase activity and that this degradation is facilitated to a large extent by serine proteases. The neutrophil heparanase was readily and preferentially released (15-25% of the cellular content in 60 min) by simply incubating the cells at 4 degrees C in the absence of added stimuli. Under these conditions, less than 5% of the cellular content of lactate dehydrogenase, lysozyme, and globin degrading proteases was released. Further purification of the neutrophil heparanase was achieved by its binding to heparin-Sepharose and elution at 1 M NaCl. It is suggested that heparanase activity is involved in the early events of extravasation and diapedesis of neutrophils in response to a threshold signal from an extravascular inflamed organ.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bar-Ner M., Kramer M. D., Schirrmacher V., Ishai-Michaeli R., Fuks Z., Vlodavsky I. Sequential degradation of heparan sulfate in the subendothelial extracellular matrix by highly metastatic lymphoma cells. Int J Cancer. 1985 Apr 15;35(4):483–491. doi: 10.1002/ijc.2910350411. [DOI] [PubMed] [Google Scholar]
- Barrett A. J. Leukocyte elastase. Methods Enzymol. 1981;80(Pt 100):581–588. doi: 10.1016/s0076-6879(81)80046-8. [DOI] [PubMed] [Google Scholar]
- Becker R. P., De Bruyn P. P. The transmural passage of blood cells into myeloid sinusoids and the entry of platelets into the sinusoidal circulation; a scanning electron microscopic investigation. Am J Anat. 1976 Feb;145(2):183–205. doi: 10.1002/aja.1001450204. [DOI] [PubMed] [Google Scholar]
- Boxer L. A., Allen J. M., Baehner R. L. Diminished polymorphonuclear leukocyte adherence. Function dependent on release of cyclic AMP by endothelial cells after stimulation of beta-receptors by epinephrine. J Clin Invest. 1980 Aug;66(2):268–274. doi: 10.1172/JCI109853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bøyum A. Isolation of lymphocytes, granulocytes and macrophages. Scand J Immunol. 1976 Jun;Suppl 5:9–15. [PubMed] [Google Scholar]
- Castellot J. J., Jr, Favreau L. V., Karnovsky M. J., Rosenberg R. D. Inhibition of vascular smooth muscle cell growth by endothelial cell-derived heparin. Possible role of a platelet endoglycosidase. J Biol Chem. 1982 Oct 10;257(19):11256–11260. [PubMed] [Google Scholar]
- Culp L. A., Murray B. A., Rollins B. J. Fibronectin and proteoglycans as determinants of cell-substratum adhesion. J Supramol Struct. 1979;11(3):401–427. doi: 10.1002/jss.400110314. [DOI] [PubMed] [Google Scholar]
- David G., Bernfield M. R. Collagen reduces glycosaminoglycan degradation by cultured mammary epithelial cells: possible mechanism for basal lamina formation. Proc Natl Acad Sci U S A. 1979 Feb;76(2):786–790. doi: 10.1073/pnas.76.2.786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dewald B., Bretz U., Baggiolini M. Release of gelatinase from a novel secretory compartment of human neutrophils. J Clin Invest. 1982 Sep;70(3):518–525. doi: 10.1172/JCI110643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gospodarowicz D., Bialecki H., Greenburg G. Purification of the fibroblast growth factor activity from bovine brain. J Biol Chem. 1978 May 25;253(10):3736–3743. [PubMed] [Google Scholar]
- Gospodarowicz D., Greenburg G., Foidart J. M., Savion N. The production and localization of laminin in cultured vascular and corneal endothelial cells. J Cell Physiol. 1981 May;107(2):171–183. doi: 10.1002/jcp.1041070203. [DOI] [PubMed] [Google Scholar]
- Gospodarowicz D., Mescher A. L., Birdwell C. R. Stimulation of corneal endothelial cell proliferations in vitro by fibroblast and epidermal growth factors. Exp Eye Res. 1977 Jul;25(1):75–89. doi: 10.1016/0014-4835(77)90248-2. [DOI] [PubMed] [Google Scholar]
- Gospodarowicz D., Moran J., Braun D., Birdwell C. Clonal growth of bovine vascular endothelial cells: fibroblast growth factor as a survival agent. Proc Natl Acad Sci U S A. 1976 Nov;73(11):4120–4124. doi: 10.1073/pnas.73.11.4120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gospodarowicz D., Vlodavsky I., Savion N. The extracellular matrix and the control of proliferation of vascular endothelial and vascular smooth muscle cells. J Supramol Struct. 1980;13(3):339–372. doi: 10.1002/jss.400130307. [DOI] [PubMed] [Google Scholar]
- Huybrechts-Godin G., Hauser P., Vaes G. Macrophage-fibroblast interactions in collagenase production and cartilage degradation. Biochem J. 1979 Dec 15;184(3):643–650. doi: 10.1042/bj1840643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones P. A., De Clerck Y. A. Extracellular matrix destruction by invasive tumor cells. Cancer Metastasis Rev. 1982;1(4):289–317. doi: 10.1007/BF00124214. [DOI] [PubMed] [Google Scholar]
- Jones P. A., DeClerck Y. A. Destruction of extracellular matrices containing glycoproteins, elastin, and collagen by metastatic human tumor cells. Cancer Res. 1980 Sep;40(9):3222–3227. [PubMed] [Google Scholar]
- Kidron M., Nachshon I., Mayer M., Fibach E. Plasminogen activator activity in differentiating leukemia cells. FEBS Lett. 1984 Nov 5;177(1):66–70. doi: 10.1016/0014-5793(84)80982-5. [DOI] [PubMed] [Google Scholar]
- Kramer R. H., Vogel K. G., Nicolson G. L. Solubilization and degradation of subendothelial matrix glycoproteins and proteoglycans by metastatic tumor cells. J Biol Chem. 1982 Mar 10;257(5):2678–2686. [PubMed] [Google Scholar]
- Leivo I., Alitalo K., Risteli L., Vaheri A., Timpl R., Wartiovaara J. Basal lamina glycoproteins laminin and type IV collagen are assembled into a fine-fibered matrix in cultures of a teratocarcinoma-derived endodermal cell line. Exp Cell Res. 1982 Jan;137(1):15–23. doi: 10.1016/0014-4827(82)90002-7. [DOI] [PubMed] [Google Scholar]
- Lindahl U., Bäckström G., Jansson L., Hallén A. Biosynthesis of heparin. II. Formation of sulfamino groups. J Biol Chem. 1973 Oct 25;248(20):7234–7241. [PubMed] [Google Scholar]
- Liotta L. A., Thorgeirsson U. P., Garbisa S. Role of collagenases in tumor cell invasion. Cancer Metastasis Rev. 1982;1(4):277–288. doi: 10.1007/BF00124213. [DOI] [PubMed] [Google Scholar]
- Matzner Y., Brass L. M., McMurrich B. J., Peters W. A., André-Schwartz J., Babior B. M. Expression of a chronic granulomatous disease-like defect by fluoride-exhausted neutrophils. Blood. 1982 Oct;60(4):822–826. [PubMed] [Google Scholar]
- Milks L. C., Brontoli M. J., Cramer E. B. Epithelial permeability and the transepithelial migration of human neutrophils. J Cell Biol. 1983 May;96(5):1241–1247. doi: 10.1083/jcb.96.5.1241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakajima M., Irimura T., Di Ferrante D., Di Ferrante N., Nicolson G. L. Heparan sulfate degradation: relation to tumor invasive and metastatic properties of mouse B16 melanoma sublines. Science. 1983 May 6;220(4597):611–613. doi: 10.1126/science.6220468. [DOI] [PubMed] [Google Scholar]
- Nakajima M., Irimura T., Di Ferrante N., Nicolson G. L. Metastatic melanoma cell heparanase. Characterization of heparan sulfate degradation fragments produced by B16 melanoma endoglucuronidase. J Biol Chem. 1984 Feb 25;259(4):2283–2290. [PubMed] [Google Scholar]
- Naparstek Y., Cohen I. R., Fuks Z., Vlodavsky I. Activated T lymphocytes produce a matrix-degrading heparan sulphate endoglycosidase. Nature. 1984 Jul 19;310(5974):241–244. doi: 10.1038/310241a0. [DOI] [PubMed] [Google Scholar]
- Oosta G. M., Favreau L. V., Beeler D. L., Rosenberg R. D. Purification and properties of human platelet heparitinase. J Biol Chem. 1982 Oct 10;257(19):11249–11255. [PubMed] [Google Scholar]
- Pauli B. U., Schwartz D. E., Thonar E. J., Kuettner K. E. Tumor invasion and host extracellular matrix. Cancer Metastasis Rev. 1983;2(2):129–152. doi: 10.1007/BF00048966. [DOI] [PubMed] [Google Scholar]
- Rosenshein M. S., Price T. H., Dale D. C. Neutropenia, inflammation, and the kinetics of transfused neutrophils in rabbits. J Clin Invest. 1979 Aug;64(2):580–585. doi: 10.1172/JCI109496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savion N., Vlodavsky I., Fuks Z. Interaction of T lymphocytes and macrophages with cultured vascular endothelial cells: attachment, invasion, and subsequent degradation of the subendothelial extracellular matrix. J Cell Physiol. 1984 Feb;118(2):169–178. doi: 10.1002/jcp.1041180209. [DOI] [PubMed] [Google Scholar]
- Sherwin R. P., Richters A. Pathobiologic nature of lymphocyte interactions with human breast cancer. J Natl Cancer Inst. 1972 Apr;48(4):1111–1115. [PubMed] [Google Scholar]
- Sopata I., Wize J. A latent gelatin specific proteinase of human leucocytes and its activation. Biochim Biophys Acta. 1979 Dec 7;571(2):305–312. doi: 10.1016/0005-2744(79)90100-1. [DOI] [PubMed] [Google Scholar]
- Vlodavsky I., Fuks Z., Bar-Ner M., Ariav Y., Schirrmacher V. Lymphoma cell-mediated degradation of sulfated proteoglycans in the subendothelial extracellular matrix: relationship to tumor cell metastasis. Cancer Res. 1983 Jun;43(6):2704–2711. [PubMed] [Google Scholar]
- Vlodavsky I., Lui G. M., Gospodarowicz D. Morphological appearance, growth behavior and migratory activity of human tumor cells maintained on extracellular matrix versus plastic. Cell. 1980 Mar;19(3):607–616. doi: 10.1016/s0092-8674(80)80037-7. [DOI] [PubMed] [Google Scholar]
- Wasteson A. A method for the determination of the molecular weight and molecular-weight distribution of chondroitin sulphate. J Chromatogr. 1971 Jul 8;59(1):87–97. doi: 10.1016/s0021-9673(01)80009-1. [DOI] [PubMed] [Google Scholar]
- Weiss S. J., Regiani S. Neutrophils degrade subendothelial matrices in the presence of alpha-1-proteinase inhibitor. Cooperative use of lysosomal proteinases and oxygen metabolites. J Clin Invest. 1984 May;73(5):1297–1303. doi: 10.1172/JCI111332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weissmann G., Smolen J. E., Korchak H. M. Release of inflammatory mediators from stimulated neutrophils. N Engl J Med. 1980 Jul 3;303(1):27–34. doi: 10.1056/NEJM198007033030109. [DOI] [PubMed] [Google Scholar]
- Werb Z., Banda M. J., Jones P. A. Degradation of connective tissue matrices by macrophages. I. Proteolysis of elastin, glycoproteins, and collagen by proteinases isolated from macrophages. J Exp Med. 1980 Nov 1;152(5):1340–1357. doi: 10.1084/jem.152.5.1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yahalom J., Eldor A., Fuks Z., Vlodavsky I. Degradation of sulfated proteoglycans in the subendothelial extracellular matrix by human platelet heparitinase. J Clin Invest. 1984 Nov;74(5):1842–1849. doi: 10.1172/JCI111603. [DOI] [PMC free article] [PubMed] [Google Scholar]



