Abstract
Objective(s):
Obesity considered being a low-grade inflammatory disease. The objective of this study was to examine the association between inflammatory markers (IM) including C-reactive protein (hs-CRP), Interleukin-6 (IL-6), and homocystein (Hcy) and obesity-related factors (e.g. BMI, waist, hip) in adult participants of Tehran lipid and glucose study (TLGS).
Materials and Methods:
In this cross-sectional study, 352 individuals (132 men and 220 women), age ≥19 years, were randomly recruited from participants of TLGS population. The serum levels of hs-CRP, IL-6, Hcy were determined using the enzyme linked immunosorbent assay (ELISA) method. Variables were compared by sample t-test. Bivariate linear correlation was estimated using Pearson's correlation coefficient. Linear regression analysis was applied to investigate the association between IMs and anthropometric and biochemical variables.
Results:
The mean age of participants was 46.1±16.1 years. abdominal obesity was present in 199(56.5%) individuals. levels of hs-CRP and IL-6 increased in the abdominally obese group (1507±3.3 vs. 577.8±4.3 ng/ml P<0.001) (3.6±3.3 vs. 1.9±3.8 pg/ml P< 0.001), and in the same group, the best predictors for hs-CRP, IL-6 and Hcy were waist (WC), waist to height ratio (WHtR) and wrist respectively; hip and WHtR were the best predictors for Hcy and hs-CRP in the normal group. A linear augmentation in hs-CRP and IL-6 levels was observed in association with obesity categorizes.
Conclusion:
This study provides evidence that abdominally obese individuals had higher levels of IMs. Wrist, waist and WHtR were the best predictors for Hcy, hs-CRP and IL-6 respectively in this group.
Keywords: Hcy, hs-CRP, IL-6, Inflammatory markers, Obesity, TLGS
Introduction
Obesity as a factor associated with numerous chronic disease conditions such as diabetes, hypertension, and cardiovascular diseases (1), is a major public health problem worldwide, affecting people in both the developed and developing countries (2). It is now evident that adipose tissue not only stores excess triglycerols, but also functions as an endocrine organ by releasing adipokines, which play important roles in regulation of appetite, glucose and lipid metabolism, inflammation, and insulin resistance (1, 3). Serum concentrations of a number of inflammatory markers (IM) including C-reactive protein (hs-CRP), tumor necrosis factor-α (TNF-α), and interleukin-6 (IL-6) elevated in overweight and obese individuals, therefore, obesity especially visceral obesity is now viewed as a low-grade inflammatory disease (4-6). Pro inflammatory cytokine such as IL-6 has been found to be independently related to cardiovascular risk factors such as increased blood pressure and body mass index (BMI), and decreased high density lipoprotein (HDL-C) (7). During obesity, IL-6 is released by the visceral adipose tissue into the portal circulation; hs-CRP is generated in the liver in response to IL-6 and TNF-α (8), and has an important effect on amplifying the inflammatory response (9); hs-CRP is used as a marker of obesity-related inflammatory factor and is a predictor of cardiovascular events and diabetes (10). High levels of homocysteine (Hcy) are linked with obesity and early development of heart and blood vessel disease, and could be considered as an independent risk factor for heart disease; one possible risk factor for increasing the level of Hcy could be BMI (11, 12). The relationship between obesity and inflammatory markers has been investigated extensively in several populations but not explicitly in Iranians, whom the prevalence of obesity and associated co-morbidities is increasing. In this study, we assessed the inflammatory marker levels in subjects with and without abdominal obesity, and their association with obesity-related factors (e.g. BMI, waist, hip etc).
Materials and Methods
Study population
Participants for this cross-sectional study were randomly recruited from among participants of the Tehran lipid and glucose study (TLGS), which is a population, based study being conducted to determine the prevalence of non-communicable diseases among Tehran’s urban population and to develop population-based measures to decrease the prevalence or prevent the rising trend of diabetes mellitus and dyslipidemia. A multistage stratified cluster random sampling technique was used to select 15005 people, aged 3 years and over, from district 13 of Tehran, the capital of Iran. The district is located in the center of Tehran, and the age distribution of this population is representative of the overall population of Tehran (13). For the present study, a total of 352 individuals including 132 Men and 220 Women, age ≥19 years, were recruited during phase III of TLGS. The study protocol was approved by the Research Ethics Committee of the Research Institute for Endocrine Sciences affiliated to Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Anthropometric and biochemical measures
Anthropometric measurements including height, weight, WC, hip, and wrist were taken for the subjects as described previously (14, 15). BMI was calculated by dividing weight (Kg) by the square of height (meter2); waist to hip ratio (WHR), and waist to height ratio (WHtR) were calculated. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were recorded using a standard sphygmomanometer after at least 10 min of rest. Abdominal obesity was defined using cutoff of waist circumference > 89 cm for men and > 91 cm for women (16); based on these criteria, subjects were grouped as normal weight and abdominally obese subjects. For categorize the obesity, we divided subjects based on having general and/or abdominal obesity as follows: 1) The normal group with normal range of BMI (18<BMI≤25 kg/m2) and waist circumference (WC≤ 89 cm for men and ≤ 91 cm for women); 2) The group with high BMI or WC and 3) The group with both high BMI and WC.
All assays including fasting blood sugar (FBS), triglycerides (TG), total cholesterol (TC) and high density lipoprotein- cholesterol (HDL-C) were done on the day of sampling. FBS level was measured by the glucose oxidase method (Glucose kit; Pars Azmun, Tehran, Iran). TG and TC levels were measured using enzymatic calorimetric tests with glycerol phosphate oxidase and cholesterol esterase and cholesterol oxidase respectively. HDL-C levels were determined after precipitation of apolipoprotein B containing lipoprotein with phosphotungistic acid; LDL-C was calculated using the Friedewald formula (17); participants with TG≥ 400 mg/dl were excluded. Plasma C-reactive protein (hs-CRP), homocyctein (Hcy) and interleukin-6 (IL-6) levels were measured using enzyme linked immunosorbent assay (ELISA) methods (dbc co. Canada; Diazyme co. USA; Diaclone Co. French) respectively. All samples were analyzed when internal quality control met the acceptable gold standard. Inter-and intra-assay coefficients of variations were 2.1% for FBS, 1.9% for TG and TC, 2.8% for HDL-C, 5.8% for hc-CRP, 6.2% for Hcy and 6.5% for IL-6 respectively. TG to HDL-C ratio (TG/HDL-C), TC to HDL-C ratio (TC/HDL-C), and LDL-C to HDL-C ratio (LDL-C/HDL-C) were calculated.
Statistical analysis
The Kolmogorov-Smirnov goodness-of-fit test was used to assess normal distribution of continuous data; All of which data are expressed as mean ± standard deviation. Logarithmic transformation was performed to normalize the distribution of DBP, TG, Hcy, hs-CRP, and IL-6. The means were compared by a two-tailed unpaired sample t-test. Bivariate linear correlation was estimated using Pearson’s correlation coefficient. A stepwise linear regression analysis was applied to investigate the association of Hcy, hs-CRP, and IL-6, as dependent variables, with anthropometric and biochemical variables, as independent ones. Statistical analysis was carried out using SPSS software (version 15). All statistical tests were two sided and differences with probability values <0.05 were considered statistically significant.
Results
The mean age of the whole study population (352 subjects) was 46.1±16.1 years, and 199 (56.5%) individuals had abdominal obesity. Anthropometric and biochemical characteristics of participants, based on the presence or absence of abdominal obesity, are shown in Table 1. As expected, the mean levels of obesity-related factors, lipid profiles, and inflammatory markers, except for FBS and Hcy, were markedly higher and HDL-C level was lower in the abdominally obese group, compared to the normal one.
Table 1.
Demographic, anthropometric and biochemical variables of participants with abdominal obesity status
| Variables | Total (n=352) | nAB (n= 153) | AB (n= 199) |
|---|---|---|---|
|
| |||
| Age (years) | 46.1±16.1 | 39.2±15.5 | 51.4±14.5* |
| Height (cm) | 161.3±9.8 | 159.7±8.9 | 162.6±10.4* |
| Weight (kg) | 71.5±12.4 | 62.9±9.4 | 78.2±10.1* |
| BMI (kg/m2) | 27.6±4.7 | 24.7±3.6 | 29.7±4.2* |
| WC (cm) | 90.8±12.6 | 79.2±7.8 | 99.7±7.4* |
| Hip (cm) | 101.6±8.8 | 97.8±6.8 | 104.5±8.9* |
| Wrist (cm) | 16.6±1.4 | 15.7±1.1 | 17.3±1.2* |
| WHR | 0.98±0.09 | 0.81±0.07 | 0.95±0.06* |
| WHtR | 0.56±0.08 | 0.49±0.06 | 0.62±0.06* |
| FBS (mg/dl) | 99.9±57.3 | 96.8±78.7 | 102.3±32.4 |
| TG (mg/dl)† | 130.7±1.7 | 104.7±1.6 | 154.9±1.6* |
| TC(mg/dl) | 190.1±39.1 | 178.8±39.1 | 198.8±36.9* |
| HDL-C (mg/dl) | 41.7±9.7 | 43.7±9.4 | 40.2±9.6* |
| LDL-C (mg/dl) | 118.6±32.9 | 111.7±33.5 | 123.9±31.6* |
| TG/HDL-C | 0.12±0.03 | 0.11±0.03 | 0.13±0.04* |
| TC/HDL-C | 4.7±1.3 | 4.2±1.2 | 5.1±1.3* |
| LDL-C/HDL-C | 2.9±0.9 | 2.7±0.9 | 3.2±0.9* |
| SBP (mmHg) | 115.9±17.5 | 107.4±13.1 | 122.5±17.7* |
| DBP (mmHg)† | 71.8±1.1 | 68.4±1.1 | 74.5±1.1* |
| Hcy (µmol/l)† | 12.4±2.12 | 12.3±1.9 | 12.5±2.2 |
| Hs-CRP (ng/ml)† | 993.8±4.1 | 577.8±4.3 | 1507±3.3* |
| IL-6 (pg/ml)† | 2.8±3.7 | 1.9±3.8 | 3.6±3.3* |
AB: With abdominal obesity; nAB: Without abdominal obesity; BMI: Body mass index; WC: Waist circumference; WHR: Waist to hip ratio; WHtR: Waist to height ratio; FBS: Fasting blood sugar; TG: triglyceride; TC: Total Cholesterol; HDL-C: High density lipoprotein cholesterol; LDL-C: Low density lipoprotein cholesterol; SBP: Systolic blood pressure; DBP: Diastolic blood pressure; Hcy: Homocystein; hs-CRP:C-reactive protein; IL-6:Interleukin-6;
P<0.05
Logarithmic transformation
To identify potential confounders prior to inclusion of the variables in a linear regression model, a bivariate Pearson correlation analysis between the levels of hs-CRP, IL-6, Hcy and selected variables was performed. In the abdominally obese group, there were statistically significant positive correlations for Hcy with height and wrist and negative ones with HDL-C; the positive correlations between hs-CRP and BMI, WC, hip, WHtR, TC, LDL-C, FBS, IL-6 and negative ones with height were significant; IL-6 was positively related to WHtR and hs-CRP levels. On the other hand, in the normal group, Hcy and hs-CRP levels were negatively correlated with hip and height respectively; there was a significant positive correlation between hs-CRP levels and age, BMI, WC, hip, WHR, WHtR, TG, TC, LDL-C, TC/HDL-C, LDL-C/HDL-C, SBP and DBP (Table 2). The association between obesity and lipid related variables and IMs, which was evaluated by stepwise linear regression analysis, revealed that the best predictors for Hcy, hs-CRP, IL-6 in the abdominally obese group, were wrist, WC, and WHtR respectively. In normal group, hip and WHtR were the best predictors for Hcy and hs-CRP; in the same group, there was no variable significantly correlated with IL-6, therefore it was not possible to consider an independent predictor for IL-6 (Table 3).
Table 2.
Correlation between inflammatory markers and anthropometric/ biochemical variables among subjects with and without abdominal obesity
| Hcy | Hs-CRP | IL-6 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||||
| Variables | AB | n AB | AB | n AB | AB | n AB | ||||||||
|
| ||||||||||||||
| r | P | r | P | r | P | r | P | r | P | r | P | |||
|
| ||||||||||||||
| Sex | -0.211 | 0.003* | -0.227 | 0.005* | 0.168 | 0.018* | 0.112 | 0.169 | 0.087 | 0.221 | 0.092 | 0.258 | ||
| Age | -0.007 | 0.926 | 0.148 | 0.068 | 0.070 | 0.329 | 0.346 | 0.000* | 0.105 | 0.140 | 0.018 | 0.827 | ||
| Height | 0.200 | 0.005* | -0.111 | 0.177 | -0.181 | 0.010* | -0.209 | 0.010* | -0.139 | 0.050 | -0.035 | 0.665 | ||
| Weight | 0.081 | 0.256 | -0.020 | 0.806 | 0.480 | 0.505 | 0.112 | 0.169 | -0.010 | 0.890 | -0.097 | 0.232 | ||
| BMI | -0.107 | 0.133 | -0.117 | 0.149 | 0.204 | 0.004* | 0.288 | 0.000* | 0.137 | 0.053 | -0.061 | 0.452 | ||
| WC | -0.007 | 0.918 | -0.015 | 0.849 | 0.175 | 0.014* | 0.312 | 0.000* | 0.088 | 0.214 | 0.056 | 0.493 | ||
| Hip | -0.072 | 0.310 | -0.209 | 0.010* | 0.163 | 0.022* | 0.239 | 0.003* | 0.111 | 0.120 | 0.054 | 0.511 | ||
| Wrist | 0.225 | 0.001* | 0.159 | 0.490 | -0.139 | 0.050 | 0.094 | 0.247 | -0.065 | 0.358 | -0.126 | 0.121 | ||
| WHR | 0.010 | 0.161 | 0.152 | 0.060 | 0.004 | 0.959 | 0.166 | 0.040* | -0.043 | 0.545 | 0.020 | 0.803 | ||
| WHtR | -0.123 | 0.085 | -0.063 | 0.442 | 0.233 | 0.001* | 0.366 | 0.000* | 0.154 | 0.030* | 0.068 | 0.406 | ||
| FBS | -0.114 | 0.109 | 0.105 | 0.198 | 0.174 | 0.014* | 0.069 | 0.396 | 0.123 | 0.084 | -0.146 | 0.071 | ||
| TG† | -0.103 | 0.149 | 0.068 | 0.436 | 0.051 | 0.473 | 0.284 | 0.000* | -0.054 | 0.452 | -0.034 | 0.673 | ||
| TC | 0.099 | 0.163 | 0.085 | 0.297 | 0.150 | 0.035* | 0.403 | 0.000* | 0.002 | 0.980 | -0.032 | 0.694 | ||
| HDL-C | -0.155 | 0.028* | -0.033 | 0.683 | -0.009 | 0.897 | 0.109 | 0.181 | 0.064 | 0.372 | 0.078 | 0.335 | ||
| LDL-C | -0.006 | 0.930 | 0.750 | 0.359 | 0.156 | 0.029* | 0.345 | 0.000* | 0.013 | 0.859 | -0.056 | 0.489 | ||
| TG/HDL-C | 0.080 | 0.260 | 0.069 | 0.395 | 0.004 | 0.952 | -0.004 | 0.962 | -0.048 | 0.497 | -0.082 | 0.316 | ||
| TC/HDL-C | 0.049 | 0.491 | 0.109 | 0.182 | 0.098 | 0.167 | 0.229 | 0.004* | -0.037 | 0.605 | -0.094 | 0.247 | ||
| LDL-C/HDL-C | 0.118 | 0.099 | 0.098 | 0.226 | 0.115 | 0.110 | 0.220 | 0.006* | -0.028 | 0.696 | -0.105 | 0.197 | ||
| SBP | 0.002 | 0.975 | 0.091 | 0.270 | -0.004 | 0.958 | 0.190 | 0.020* | 0.123 | 0.085 | 0.007 | 0.932 | ||
| DBP† | 0.005 | 0.947 | 0.470 | 0.566 | -0.056 | 0.433 | 0.297 | 0.001* | 0.081 | 0.262 | -0.099 | 0.227 | ||
| Hcy† | - | - | - | - | 0.158 | 0.414 | 0.021 | 0.797 | -0.007 | 0.921 | -0.103 | 0.207 | ||
| hsCRP† | 0.058 | 0.414 | 0.210 | 0.797 | - | - | - | - | 0.157 | 0.026* | 0.014 | 0.859 | ||
| IL6† | -0.007 | 0.921 | -0.103 | 0.207 | 0.157 | 0.026* | 0.014 | 0.859 | - | - | - | - | ||
Hcy: Homocystein; hs-CRP: C-reactive protein; IL-6: Interleukin-6; AB: Abdominal obesity; nAB: without abdominal obesity; BMI: Body mass index; WC: Waist circumference; WHR: Waist to hip ratio; WHtR: Waist to height ratio; FBS: Fasting blood sugar; TG: Triglyceride; TC: Total Cholesterol; HDL-C: High density lipoprotein cholesterol; LDL-C: Low density lipoprotein cholesterol; SBP: Systolic blood pressure; DBP: Diastolic blood pressure;
P<0.05
Logarithmic transformation;
r: pearson correlation
Table 3.
Results of the multiple linear regression model including all of the variables
| Criterion | Groups | Predictors | β-coefficient | P | Adjusted R2 model (%) |
|---|---|---|---|---|---|
|
| |||||
| Hcy | AB | Wrist | 0.95 | <0.001 | 91.1 |
| nAB | Hip | 0.96 | <0.001 | 92.2 | |
| Hs-CRP | AB | WC | 0.98 | <0.001 | 97.3 |
| nAB | WHtR | 0.98 | <0.001 | 95.7 | |
| IL-6 | AB | WHtR | 0.27 | <0.001 | 7 |
| nAB | - | - | - | - | |
Hcy: Homocystein; hs-CRP: C-reactive protein; IL-6: Interleukin-6; AB: with abdominal obesity; nAB: Without abdominal obesity; WC: Waist circumference
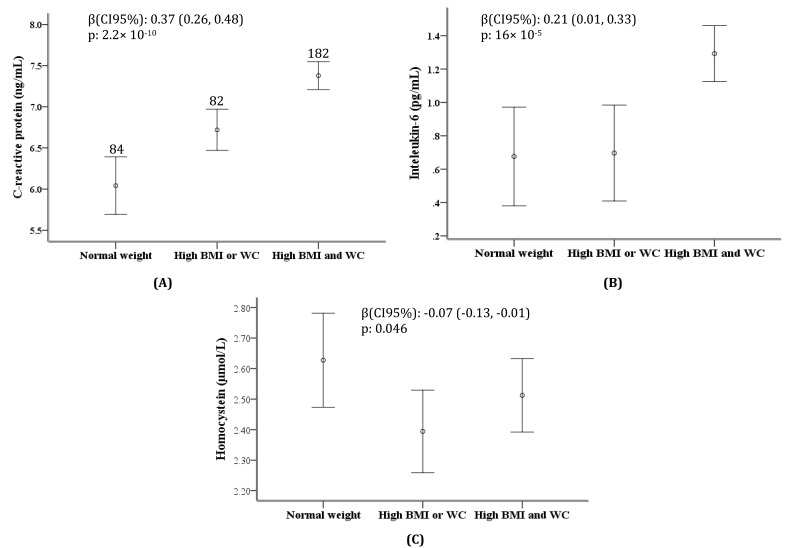
Assessing the levels of hs-CRP, IL-6 and Hcy between the obesity categorizes showed a gradual and significant increase in the levels of hs-CRP and IL-6. A strong linear augmentation in the levels of hs-CRP and IL-6 was observed from the first group (subjects with normal BMI and waist circumference) to third group (individuals, who had both general and abdominal obesity), with the median hs-CRP levels of 6.21, 6.65, 7.75 ng/ml (Ptrend: 2.33×10-10) and median levels of IL-6 of 0.55, 0.84, 1.35 pg/ml (Ptrend: 18×10-5). Additionally, an increase of 0.37 ng/ml (CI95%: 0.24-0.48; P: 2.2×10-10) in the hs-CRP levels and 0.21 pg/ml (CI95%: 0.10-0.33; P: 16×10-5) in IL-6 levels were observed from normal weight group to subjects with both general and abdominal obesity after adjustment for sex and age (Figure 1).
Figure 1.
Trends of (A) C-reactive protein (hs-CRP), (B) interleukin-6 (IL-6), (C) homocystein (Hcy) levels in association with increasing the grade of obesity
Discussion
In the present study, an attempt was made to assess possible associations between IMs, such as hs-CRP, IL-6, Hcy and obesity-related factors. Participants with abdominal obesity had higher levels of hs-CRP and IL-6. Furthermore, in our study, the levels of hs-CRP and IL-6 increased considerably in association with obesity, a finding consistent with previous reports, which showed that elevated levels of these markers are associated with obesity(18); according to the results of this study, there was strong association between hs-CRP, IL-6, Hcy and WC, WHtR and wrist among the subjects with abdominal obesity.
Previous studies have shown a positive association between IL-6 and obesity; for instance, Salzburg et al reported that IL-6 is positively associated with the degree of overweight and obesity; leptin as the best known adipokine, and WHR are the best predictors for IL-6 plasma levels in juveniles and adults respectively (19). In the present study we observed a significantly positive trend for IL-6 with the obesity categorizes (20), and the best predictor for IL-6 was WHtR. Compaired to BMI, WHtR is a more sensitive predictor of health risk, and could predict morbidity and mortality, better than BMI (21) as it is more closely associated with abdominal obesity. In our study, WHtR could predict the levels of hs-CRP and IL-6. Waist circumference, WHR and WHtR as direct anthropometric measures of abdominal obesity, are predictive for insulin resistance, metabolic syndrome (MetS) and coronary artery disease (22). Association between abdominal obesity and increased levels of IMs, such as hs-CRP have been noted in previous studies (23, 24); it is reported that abdominal adipose tissues are a major source of cytokines, including TNF-α and IL-6, which in turn increase hepatic hs-CRP production (25, 26). Weight loss associated with concomitant decrease in hs-CRP levels (27, 28). The results of a study examining the relationship between hs-CRP and MetS components, showed that of the various components of MetS, waist circumference is the best predictor of hs-CRP elevation; this study provides evidence that hs-CRP level is related to obesity, specially abdominal obesity (18), and in agreement with their finding, our results also showed that abdominally obese individuals had higher levels of hs-CRP, and in the same group, WC was the best predictor for elevated hs-CRP. In addition, another study of 326 healthy Japanese men, aged 30-59 years, showed that participants with abdominal obesity had higher hs-CRP and IL-6 and lower adiponectin levels, serum levels of hs-CRP and IL-6 increased in association with clustering of MetS components in subjects with abdominal obesity, however, adiponectin levels showed a little change in this group (29).
Another study of an adult population in Spain showed the association between obesity and hyper homocysteinemia, and indicated that increased levels of Hcy are mostly related to abdominal obesity; WC and FBS are the independent predictor for this inflammatory marker (30); in contrast to results mentioned, in our population, the increasing trend in Hcy levels were not significant; it could be explained by changes in the ratio of adipose/ muscle tissue in obese subjects and changes in oxidation level. In our study, the best predictors for Hcy among the subjects with and without abdominal obesity were Wrist and Hip respectively.
Findings of the present study, which to the best of our knowledge is the first to be conducted on a Tehran population, added to the growing evidence that obesity is considered as a low-grade inflammatory disease. Nevertheless, this study has some limitations that need to be addressed; first, absence of longitudinal follow-up toward clinical endpoints such as myocardial infarction and stroke. Second, our small sample size prohibits us from generalization and studies with larger sample size are recommended. Third, no detailed inquiries were made regarding dietary habits that might potentially affect hs-CRP, IL-6 and Hcy production and association between them and obesity-related factors.
Conclusion
This study provides evidence that abdominally obese individuals had higher levels of hs-CRP, IL-6 and Hcy, and in the abdominally obese group, wrist, waist and WHtR were the best predictors for Hcy, hs-CRP and IL-6 respectively.
Acknowledgment
The authors wish to thank Ms N Shiva for critical editing of English grammar and syntax of the manuscript; the staff and participants in the TLGS for their cooperation. This work was supported by the Iran National Science Foundation under grant No: 83075.
References
- 1.Klein J, Perwitz N, Kraus D, Fasshauer M. Adipose tissue as source and target for novel therapies. Trends Endocrinol Metab. 2006;17:26–32. doi: 10.1016/j.tem.2005.11.008. [DOI] [PubMed] [Google Scholar]
- 2.Organization WH. Obesity: Preventing and managing the global epidemic. WHO Consultation. 2004. ISBN: 9241208945. [PubMed]
- 3.Ronti T, Lupattelli G, Mannarino E. The endocrine function of adipose tissue: an update. Clin Endocrinol (Oxf) 2006;64:355–365. doi: 10.1111/j.1365-2265.2006.02474.x. [DOI] [PubMed] [Google Scholar]
- 4.Fantuzzi G. Adipose tissue, adipokines, and inflammation. J Allergy Clin Immunol. 2005;115:911–919. doi: 10.1016/j.jaci.2005.02.023. [DOI] [PubMed] [Google Scholar]
- 5.Greenberg AS, Obin MS. Obesity and the role of adipose tissue in inflammation and metabolism. Am J Clin Nutr. 2006;83:461S–465S. doi: 10.1093/ajcn/83.2.461S. [DOI] [PubMed] [Google Scholar]
- 6.Trayhurn P, Wood IS. Adipokines: inflammation and the pleiotropic role of white adipose tissue. Br J Nutr. 2004;92:347–355. doi: 10.1079/bjn20041213. [DOI] [PubMed] [Google Scholar]
- 7.Bermudez EA, Rifai N, Buring J, Manson JE, Ridker PM. Interrelationships among circulating interleukin-6, C-reactive protein, and traditional cardiovascular risk factors in women. Arterioscler Thromb Vasc Biol. 2002;22:1668–1673. doi: 10.1161/01.atv.0000029781.31325.66. [DOI] [PubMed] [Google Scholar]
- 8.Anty R, Bekri S, Luciani N, Saint-Paul MC, Dahman M, Iannelli A, et al. The inflammatory C-reactive protein is increased in both liver and adipose tissue in severely obese patients independently from metabolic syndrome, Type 2 diabetes, and NASH. Am J Gastroenterol. 2006;101:1824–1833. doi: 10.1111/j.1572-0241.2006.00724.x. [DOI] [PubMed] [Google Scholar]
- 9.Bisoendial RJ, Birjmohun RS, Akdim F, van 't Veer C, Spek CA, Hartman D, et al. C-reactive protein elicits white blood cell activation in humans. Am J Med. 2009;122:582, e1–589. doi: 10.1016/j.amjmed.2008.11.032. [DOI] [PubMed] [Google Scholar]
- 10.Ridker PM. C-reactive protein and the prediction of cardiovascular events among those at intermediate risk: moving an inflammatory hypothesis toward consensus. J Am Coll Cardiol. 2007;49:2129–2138. doi: 10.1016/j.jacc.2007.02.052. [DOI] [PubMed] [Google Scholar]
- 11.Dudeja V, Misra A, Pandey RM, Devina G, Kumar G, Vikram NK. BMI does not accurately predict overweight in Asian Indians in Northern India. Br J Nutr. 2001;86:105–112. doi: 10.1079/bjn2001382. [DOI] [PubMed] [Google Scholar]
- 12.Scott JM. Homocysteine and cardiovascular risk. Am J Clin Nutr. 2000;72:333–334. doi: 10.1093/ajcn/72.2.333. [DOI] [PubMed] [Google Scholar]
- 13.Azizi F, Rahmani M, Emami H, Mirmiran P, Hajipour R. Tehran Lipid and Glucose Study. IJEM. 2000;2:77–86. [Google Scholar]
- 14.Azizi F, Rahmani M, Emami H, Mirmiran P, Hajipour R, Madjid M, et al. Cardiovascular risk factors in an Iranian urban population: Tehran lipid and glucose study (phase 1) Soz Praventivmed. 2002;47:408–426. doi: 10.1007/s000380200008. [DOI] [PubMed] [Google Scholar]
- 15.Azizi F, Ghanbarian A, Momenan AA, Hadaegh F, Mirmiran P, Hedayati M, et al. Prevention of non-communicable disease in a population in nutrition transition: Tehran Lipid and Glucose Study phase II. Trials. 2009;10:5. doi: 10.1186/1745-6215-10-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Delavari A, Forouzanfar MH, Alikhani S, Sharifian A, Kelishadi R. First nationwide study of the prevalence of the metabolic syndrome and optimal cutoff points of waist circumference in the Middle East: the national survey of risk factors for noncommunicable diseases of Iran. Diabetes Care. 2009;32:1092–1097. doi: 10.2337/dc08-1800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502. [PubMed] [Google Scholar]
- 18.Belfki H, Ben Ali S, Bougatef S, Ben Ahmed D, Haddad N, Jmal A, et al. Relationship of C-reactive protein with components of the metabolic syndrome in a Tunisian population. Eur J Intern Med. 2012;23:e5–9. doi: 10.1016/j.ejim.2011.10.011. [DOI] [PubMed] [Google Scholar]
- 19.Stelzer I, Zelzer S, Raggam RB, Prüller F, Truschnig-Wilders M, Meinitzer A, et al. Link between leptin and interleukin-6 levels in the initial phase of obesity related inflammation. Translat Res. 2012;159:118–124. doi: 10.1016/j.trsl.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 20.Hsieh SD, Yoshinaga H, Muto T, Sakurai Y, Kosaka K. Health risks among Japanese men with moderate body mass index. Int J Obes Relat Metab Disord. 2000;24:358–362. doi: 10.1038/sj.ijo.0801157. [DOI] [PubMed] [Google Scholar]
- 21.Cox BD, Whichelow M. Ratio of waist circumference to height is better predictor of death than body mass index. BMJ. 1996;313:1487. doi: 10.1136/bmj.313.7070.1487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nakamura H, Ito H, Egami Y, Kaji Y, Maruyama T, Koike G, et al. Waist circumference is the main determinant of elevated C-reactive protein in metabolic syndrome. Diabetes Res Clin Pract. 2008;79:330–336. doi: 10.1016/j.diabres.2007.09.004. [DOI] [PubMed] [Google Scholar]
- 23.Saijo Y, Kiyota N, Kawasaki Y, Miyazaki Y, Kashimura J, Fukuda M, et al. Relationship between C-reactive protein and visceral adipose tissue in healthy Japanese subjects. Diabetes Obes Metab. 2004;6:249–258. doi: 10.1111/j.1462-8902.2003.0342.x. [DOI] [PubMed] [Google Scholar]
- 24.You T, Ryan AS, Nicklas BJ. The metabolic syndrome in obese postmenopausal women: relationship to body composition, visceral fat, and inflammation. J Clin Endocrinol Metab. 2004;89:5517–5522. doi: 10.1210/jc.2004-0480. [DOI] [PubMed] [Google Scholar]
- 25.Yudkin JS, Stehouwer CD, Emeis JJ, Coppack SW. C-reactive protein in healthy subjects: associations with obesity, insulin resistance, and endothelial dysfunction: a potential role for cytokines originating from adipose tissue? . Arterioscler Thromb Vasc Biol. 1999;19:972–978. doi: 10.1161/01.atv.19.4.972. [DOI] [PubMed] [Google Scholar]
- 26.Mohamed-Ali V, Goodrick S, Rawesh A, Katz DR, Miles JM, Yudkin JS, et al. Subcutaneous adipose tissue releases interleukin-6, but not tumor necrosis factor-alpha, in vivo. J Clin Endocrinol Metab. 1997;82:4196–4200. doi: 10.1210/jcem.82.12.4450. [DOI] [PubMed] [Google Scholar]
- 27.O'Brien KD, Brehm BJ, Seeley RJ, Bean J, Wener MH, Daniels S, et al. Diet-induced weight loss is associated with decreases in plasma serum amyloid a and C-reactive protein independent of dietary macronutrient composition in obese subjects. J Clin Endocrinol Metab. 2005;90:2244–2249. doi: 10.1210/jc.2004-1011. [DOI] [PubMed] [Google Scholar]
- 28.Ryan AS, Nicklas BJ. Reductions in plasma cytokine levels with weight loss improve insulin sensitivity in overweight and obese postmenopausal women. Diabetes Care. 2004;27:1699–1705. doi: 10.2337/diacare.27.7.1699. [DOI] [PubMed] [Google Scholar]
- 29.Nishida M, Moriyama T, Sugita Y, Yamauchi-Takihara K. Abdominal obesity exhibits distinct effect on inflammatory and anti-inflammatory proteins in apparently healthy Japanese men. Cardiovasc Diabetol. 2007;6:27. doi: 10.1186/1475-2840-6-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vaya A, Rivera L, Hernandez-Mijares A, de la Fuente M, Sola E, Romagnoli M, et al. Homocysteine levels in morbidly obese patients: its association with waist circumference and insulin resistance. Clin Hemorheol Microcirc. 2012;52:49–56. doi: 10.3233/CH-2012-1544. [DOI] [PubMed] [Google Scholar]