Abstract
Background and Purpose
Orexins have been demonstrated to play important roles in many physiological processes. However, it is not known how orexin A affects the activity of the hypoglossal motoneuron (HMN) and genioglossus (GG) muscle.
Experimental Approach
GG muscle electromyograms (GG-EMG) were recorded in anaesthetized adult rats after orexin A or orexin receptor antagonists were applied to the hypoglossal nucleus, and in adult rats in which orexin neurons were lesioned with the neurotoxin orexin-saporin (orexin-SAP). HMN membrane potential and firing were recorded from neonatal rat brain slices using whole-cell patch clamp after an infusion of orexin A or orexin receptor antagonists.
Key Results
Unilateral micro-injection of orexin A (50, 100 or 200 μM) into the hypoglossal nucleus significantly enhanced ipsilateral GG activity in adult rats. Orexin A (4, 20, 100 or 500 nM) depolarized the resting membrane potential and increased the firing rate of HMNs in a dose-dependent manner in the medullary slices of neonatal rats. Both SB 334867, a specific OX1 receptor antagonist and TCS OX2 29, a specific OX2 receptor antagonist not only blocked the depolarized membrane potential and the increased firing rate of HMNs by orexin A in the neonatal model but also attenuated GG-EMG in the adult model. A significant decrease in GG-EMG was observed in adult orexin neuron-lesioned rats compared with sham animals.
Conclusion and Implications
Orexin A activates OX1 and OX2 receptors within the hypoglossal motor pool and promotes GG activity, indicating that orexin A is involved in controlling respiratory motor activity.
Table of Links
| TARGETS | LIGANDS |
|---|---|
| OX1 receptor | Orexin A |
| OX2 receptor | TCS OX2 29 |
This Table lists protein targets and ligands which are hyperlinked to corresponding entries in http://www.guidetopharmacology.org, the common portal for data from the IUPHAR/BPS Guide to PHARMACOLOGY (Pawson et al., 2014) and the Concise Guide to PHARMACOLOGY 2013/14 (Alexander et al., 2013).
Introduction
As peptide transmitters, orexins, including orexin A and B, have been demonstrated to play an important role in many physiological processes, such as feeding behaviour (Willie et al., 2001), energy homeostasis (Kukkonen et al., 2002), sleep/wake cycles (Saper et al., 2005), motivation (Harris and Aston-Jones, 2006), stress responses (Zhang et al., 2006), regulation of the cardiovascular system (Shirasaka et al., 2003) and breathing (Young et al., 2005). Orexin neurons are restricted to the lateral hypothalamus (LH). They project widely throughout the brain, with the exception of the cerebellum. Orexins act on two specific GPCRs, OX1 and OX2 receptors, in target cells (Elias et al., 1998; Peyron et al., 1998; Sakurai et al., 1998; Nambu et al., 1999).
The genioglossus (GG) muscle, which is innervated by the medial branch of the hypoglossal nerve, is the largest upper airway dilator muscle. Contraction of the GG muscle promotes anterior tongue movement and oropharyngeal airway widening (Jordan and White, 2008), which prevents obstructive sleep apnoea (OSA) in humans (Remmers et al., 1978). There are two entirely opposite results reporting that plasma orexin A was lower (Nishijima et al., 2003) or higher (Igarashi et al., 2003) in patients with OSA, suggesting that dysfunction of orexin systems might be involved in the pathophysiology of OSA. Importantly, how orexin regulates GG muscle activity is still not clear. Some reports about the effect of orexins on upper airway patency in vivo have been published. Specifically, orexin B micro-injected into the rat pontine Kolliker-Fuse nucleus was reported to enhance the pre-inspiratory activity of the hypoglossal nerve (Dutschmann et al., 2007). Also orexin A micro-injected into the hypoglossal nucleus was reported to activate the GG muscle in decerebrate cats (Peever et al., 2003). However, the effect of orexin on the hypoglossal motoneuron (HMN), the key role of endogenous orexin in controlling GG activity and the receptor mechanism involved in this process, warrants further investigation.
Thus, in the present study, a GG muscle electromyogram (GG-EMG) was recorded in anaesthetized adult rats before and after the application of orexin A or orexin receptor antagonists to the hypoglossal nucleus, and in adult rats whose orexin neurons were lesioned by the neurotoxin orexin-saporin (orexin-SAP; micro-injected into the bilateral LH 14 days before the recording). To elucidate the receptors involved, a whole-cell patch clamp was used to record the membrane potential and firing of the HMN in medullary slices of neonatal rats. Our findings indicate that endogenous orexin targeting the HMN enhanced GG activity via OX1 and OX2 receptors.
Methods
Animals
Male Sprague-Dawley (SD) rats (250–350 g) and neonatal pups (P6-P10) were obtained from the Sino-British SIPPR/BK Laboratory Animal Limited Company (Shanghai, China). The animals were maintained at an ambient temperature of 22 ± 0.5°C, with a relative humidity of 60 ± 2%. A 12 h light/ dark cycle (lights on at 07:00 h, illumination intensity ≈ 100 lux) was automatically controlled. Food and water were given ad libitum. Experimental protocols were approved by the Medical Experimental Animal Administrative Committee of Shanghai Medical College of Fudan University, in accordance with the guidelines implemented by the NIH in the US regarding the care and use of animals for experimental procedures. In the in vivo experiments, the animal number in every group was 5 or 6. In the in vitro experiments, each concentration of orexin A was applied to at least 6 slices. Every effort was made to minimize the number of animals used and any pain or discomfort experienced by the animals. All studies involving animals are reported in accordance with the ARRIVE guidelines for reporting experiments involving animals (Kilkenny et al., 2010; McGrath et al., 2010).
Surgical procedures and EMG recording
Animals were anaesthetized with urethane (1.0 g·kg−1, i.p.) and then tracheotomized to maintain an unobstructed airway. Anaesthetic depth was assessed by observing the reflex responses to paw pinches. The femoral artery and external jugular vein were cannulated for blood pressure measurements and i.v. infusions respectively. The rats received continuous i.v. saline (1.0 mL·h−1) during the experiment to stabilize body fluids. They were laid on a servo-controlled electric heating pad (Temperature Controller, Quanshui, Nanjing, China) to keep the body temperature consistent, which was monitored with a rectal probe to ensure the core body temperature was at 37.0 ± 0.5°C. Vagal nerves were cut bilaterally to enhance GG muscle activity (Fenik et al., 1997; Stettner and Kubin, 2013). The GG muscle was exposed. Then two insulated, multi-stranded stainless steel wires (Cooner Wire Company, Chatsworth, CA, USA) were implanted into the muscle, as demonstrated previously (Jelev et al., 2001). To further ensure adequate electrode placement during surgery, GG signals were monitored on a chart recorder (model SMUP-E, Bio-electric Signals Processing System, Shanghai, China; Polygraph System Amplifier, Tokyo, Japan) to document tonic and respiratory-related activity. GG muscle activity was significantly weakened and almost non-detectable when the medial branches of the hypoglossal nerve was cut at the end of the experiment. This indicated that the recordings came mainly from the GG muscle with these electrode placements.
Micro-injection studies
Sixteen urethane-anaesthetized rats were positioned in a stereotaxic frame (Kopf model 962, Tujunga, CA, USA) with blunt ear bars. A stainless steel tube (0.20 mm o.d.) was unilaterally introduced into the hypoglossal nucleus [anteroposterior (AP) –13.0 to –14.5 mm, lateral (L) 0.2–0.3 mm, dorsoventral (DV) −9.0 to −9.2 mm] as per the rat atlas of Paxinos and Watson (2007). The signals from the GG muscle were continuously recorded for at least 1 h after micro-injection of saline into the hypoglossal nucleus (0.1 μL) over a period of 1 min through the cannula, followed by orexin A (50, 100 or 200 μM) dissolved in saline. Right after finishing the recording, pontamine sky blue dye (2%, 0.1 μL) was micro-injected into the same site to mark the injection site left by the cannula. In a separate group of 11 urethane-anaesthetized rats, GG muscle signals were recorded after micro-injection of a specific OX1 antagonist, SB 334867 (10 mM), or a specific OX2 antagonist, TCS OX2 29 (1 mM), into the hypoglossal nucleus (receptor nomenclature follows Alexander et al., 2013). The concentration of SB 334867 selected was based on previous reports (Azhdari-Zarmehri et al., 2013).
Lesion study
After being anaesthetized with chloral hydrate (420 mg·kg−1, i.p.), another 13 rats were placed in the stereotaxic apparatus. A fine glass pipette containing the neurotoxin orexin-SAP (0.43 mg·mL−1, 400 nl·per side) was lowered into the LH (AP –2.6 to –3.6 mm, L 1.6–1.8 mm, DV –8.6 to –8.8 mm) as per the rat atlas of Paxinos and Watson (2007). Orexin-SAP was injected with nitrogen gas pulses of 20–40 psi using the compressed air delivery system described previously (Scammell et al., 1998) over the course of 15 min in 7 rats, and saline was administered to the other 6 rats that served as controls. The incision was then closed by suture. Fourteen days later, GG muscle activity was recorded.
Medullary slice preparation
SD neonatal rats were anaesthetized by hypothermia (placed on ice for 10–15 min) and decapitated, as described previously (Miles et al., 2004; Gonzalez-Forero et al., 2012). Shortly after this procedure, rat brainstems were quickly removed and placed in ice-cold cutting artificial cerebrospinal fluid (ACSF) bubbled continuously with 95% O2 and 5% CO2. Transverse medullary slices (300 μm thick) containing HMNs were formed with a vibratome (VT 1200, Leica, Nussloch, Germany). These slices were then incubated at 30°C for 30 min in the normally-oxygenated ACSF and superfused at 3–4 mL·min−1. Subsequently, the slices were treated at room temperature in ACSF for whole-cell patch-clamp recordings.
Whole-cell patch-clamp recordings
The patch electrodes were pulled from borosilicate glass capillaries (1.5 mm o.d., 0.86 mm i.d.; Harvard Apparatus, Holliston, MA, USA) on a Brown-Flaming micropipette puller (model P-97, Sutter Instrument, Novato, CA, USA), which carried a resistance of 3–5 MΩ when filled with internal solution. A single coronal slice was transferred into a recording chamber (0.4 mL), where it was held down with a platinum ring. The neurons were visually identified as HMNs, based on their location within the hypoglossal nucleus and characteristic size, using infrared-differential interference contrast optics (Olympus, Tokyo, Japan) (Nunez-Abades and Cameron, 1995). The input resistance value of HMN should be between 50 and 150 MΩ as an electrophysiological criterion (Nunez-Abades and Cameron, 1995; Berger et al., 1996). The current recordings were obtained when low-pass Bessel-filtered at 10 kHz with a MultiClamp 700B amplifier (Axon Instruments, Union City, CA, USA); the data were digitized with a digidata 1440A analogue-to-digital converter and acquired using pClamp 10.3 software (Molecular Devices, Union City, CA, USA). A high resistance seal (>1 GΩ) with the HMN membrane was obtained before the whole-cell patch-clamp mode was established. The acceptable HMN recordings presented a resting membrane potential of −55 mV or more negative, with leak currents and liquid junction potentials not corrected.
HMNs were current clamped to record repetitive firing and/or membrane potentials, and the former was evoked by rectangular depolarizing current steps (100–800 pA) of 1000 ms in duration once a minute. Steady-state repetitive firing was defined as the mean firing frequency of all action potentials (APs) after the third AP in response to an individual current step, and acceptable APs were >70 mV in amplitude. The steady firing rate min−1 before drug administration was defined as the control. At the end of the experiment, orexin A at 20 nM was administered alone to the cell. Cells that failed to respond were excluded.
Patch-clamp procedure
The firing rate of all cells was left steady for at least 3 min until application of orexin A at 3–4 mL·min−1 for 6 min per cell. Each cell was recorded for 26 min, during which it received a depolarizing current of 1000 ms in duration at the beginning of every minute. When the control rate was recorded, orexin A was applied to the chamber at the fifth minute, which lasted for 6 min. Afterwards, each cell was washed for 15 min. The firing rate before the application of orexin A was considered to be the control, and the highest firing rate after the treatment was defined as the peak rate.
Orexin receptor antagonists, SB4867 or TCS OX2 29, were applied continuously to the bath solution 5 min before, during and after orexin A administration at 100 nM. Concentrations of antagonists (10 μM) were selected based on previous reports (Sakurai et al., 1998; Smart et al., 2001).The incubation time with orexin A for all cells was 6 min.
Histology
At the end of each in vivo experiment, the rats were given urethane (1 g·kg−1, i.p.) and intracardially perfused with 150 mL of saline followed by 250 mL of 4% paraformaldehyde. The medullary or hypothalamic regions were blocked, transferred to 20 and 30% sucrose, respectively, until they sank, before being cut into coronal sections (30 μm thick) with a cryostat (CM 1850, Leica). In the in vivo micro-injection experiments, the medullary sections were stained with neutral red solution to localize the micro-injection sites. In the lesion study, the LH sections were stained with Nissl solution or anti-orexin A antibody to determine whether the related neurons were lesioned.
Quantification of EMG
The GG-EMG was recorded in vivo and analysed using MFLab 200 software (Fudan University, Shanghai, China). GG-EMG includes two parts, tonic and respiratory-related GG-EMG. To estimate the activity of GG during each tonic and respiratory-related phase, the average intensity of GG-EMG (y) was obtained from the following formula respectively:
where |x(i)| is the absolute value of GG-EMG and N is the length of original GG-EMG data. Tonic GG-EMG is the equivalent of the average intensity of GG-EMG during the expiratory phase, and respiratory-related GG-EMG is the difference between inspiratory and expiratory GG activity. It should be noted that tonic GG-EMG is not basal noise from the instruments but active electrical activity from GG muscle. However, in the experiment, EMG recorded during the expiratory phase includes not only tonic GG-EMG but also basal noise; it is difficult to distinguish between them. We strictly controlled the insert position and depth of the electrode (parameters of instrument), so the basal noise is assumed to be similar between different groups. Hence, we used EMG recorded during the expiratory phase to represent tonic GG-EMG and absolute values are reported instead of normalized EMG data.
Statistical analysis
The results are presented as arithmetic means ± SEM. All analyses were performed using GraphPad Prism software 5.0 package. Statistics were performed using one-way anova followed by the Newman–Keuls test or the Student's paired or unpaired t-test. We set P < 0.05 as the minimal significant level and also reported P < 0.01 wherever it was detected.
Chemicals and solutions
Orexin A and anti-orexin A antibody were purchased from Sigma Chemical Company (St. Louis, MO, USA); SB 334867 [N-(2-methyl-6-benzoxazolyl)-N′-1,5-naphthyridin-4-yl urea] and TCS OX2 29 {(2S)-1-[3,4-dihydro-6,7-dimethoxy-2(1H)-isoquinolinyl]-3,3-dimethyl-2-[(4-pyridinylmethyl)amino]-1-butanone hydrochloride} from Tocris Bioscience Company (Bristol, UK); orexin-SAP [(orexin B peptide)-saporin] from Advanced Targeting Systems (San Diego, CA, USA); and TTX (tetrodotoxin) from Ziyi Reagent Factory (Shanghai, China). Orexin A and TCS OX2 29 were dissolved in saline, and SB 334867 in DMSO.
Cutting ACSF contained (in mM): NaHCO3 26, KCl 3, NaH2PO4 1.25, MgCl2 2, sucrose 218 and glucose 10. Normal ACSF was composed of (in mM): NaHCO3 26, KCl 3, NaH2PO4 1.25, MgCl2 2, NaCl 130, CaCl2 2 and glucose 10. The solution was bubbled with a gas mixture of 95% O2 and 5% CO2 to maintain the pH of the solution at 7.4. The internal solution in the patch electrode contained (in mM): potassium gluconate 122.5, KCl 17.5, NaCl 9, MgCl2 1, HEPES 10, EGTA 0.2, Mg-ATP 3, Na3-GTP 0.3, with pH buffered to 7.2 using KOH.
Results
Orexin A micro-injected in the hypoglossal nucleus of the adult rats enhanced ipsilateral respiratory-related GG-EMG in vivo
To investigate the effects of exogenous orexin A on GG-EMG in vivo, orexin A at 50, 100 or 200 μM (0.1 μL) was micro-injected unilaterally into the hypoglossal nucleus in the adult rats. As shown in Figure 1B, orexin A at 50, 100 or 200 μM increased GG activity. The injection site made by a micro-injection cannula in all rats was confirmed by the neutral red staining (Figure 1A).
Figure 1.
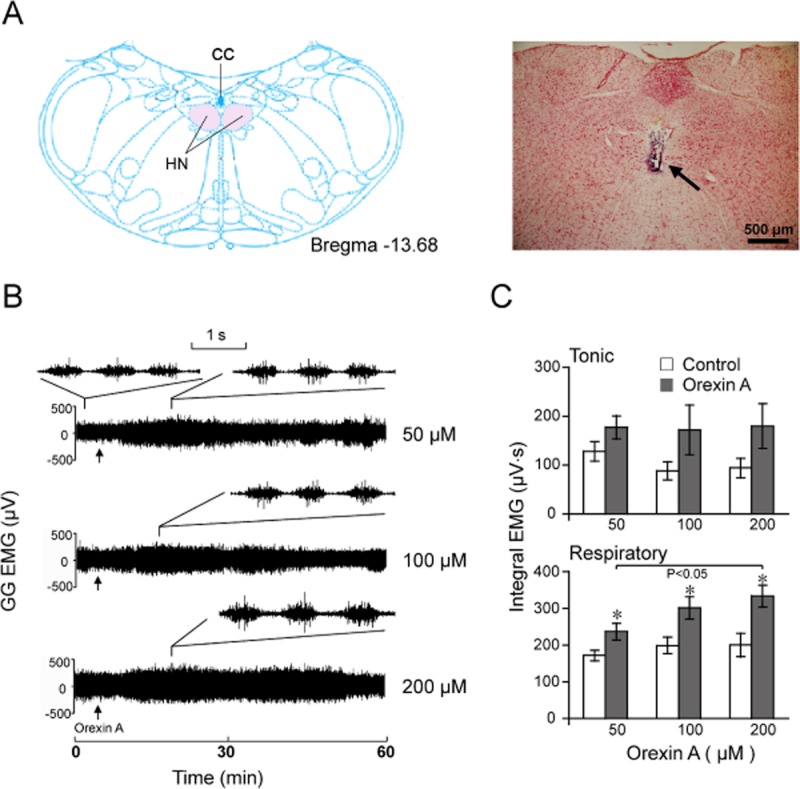
Effects of unilateral micro-injection of orexin A (0.1 μl) into the hypoglossal nucleus on the GG-EMG in adult rats. (A) Location of the hypoglossal nucleus at bregma −13.68 mm in the atlas (left) and micro-injection trace in the hypoglossal nucleus (right) indicated by an arrow. (B) A typical example illustrating GG-EMG increased by orexin A micro-injection at 50 (n = 6), 100 (n = 5) and 200 μM (n = 5). Arrow indicates the time of orexin A injection. The traces of tonic and respiratory-related GG activities induced by different doses of orexin A are shown with smaller time window upon the compressed graphics. (C) Effect of orexin A on the tonic and respiratory-related GG-EMG. Values are means ± SEM. *P < 0.05, vs control. HN, hypoglossal nucleus; CC, central canal.
As shown in Figure 1B,C, orexin A at 50, 100 or 200 μM enhanced respiratory-related GG-EMG whereas it did not increase tonic GG-EMG. Meanwhile, the breathing rate, heart rate and mean arterial pressure of each animal were not affected by orexin A administration when compared with those vital sign readings obtained before the micro-injection (P > 0.05, n = 6 or 5, data not shown). Orexin A at 200 μM enhanced respiratory-related GG-EMG more strongly than at 50 μM (P < 0.05, n = 6; Figure 1B,C). These results clearly indicate that exogenous orexin A increased respiratory-related GG-EMG activity in a dose-dependent manner, but did not alter tonic activity, breathing rate, heart rate and mean arterial pressure in urethane-anaesthetized rats.
Orexin A increased the firing rate and decreased the membrane potential of the HMNs of the neonatal rats in vitro
The mechanism by which orexin A increased GG-EMG was further investigated by whole-cell patch-clamp experiments. All HMNs selected for recording met the locational, morphological and electrophysiological criteria, with a stable resting membrane potential in the normal ACSF of −55 mV or more hyperpolarized prior to the injection of any holding current. Figure 2 depicts a typical example illustrating the location (Figure 2A, panel a) of the hypoglossal nucleus and the morphology of one HMN (Figure 2A, panel b). The effect of orexin A (4–500 nM) on HMNs was tested on 83 neurons from 83 different medullary slices of neonatal rats. Orexin A activated 76 HMNs, whereas the remaining 7 cells did not response to it even though bath concentration of orexin A is up to 500 nM. All responsive cells were included (n = 76), and all unresponsive cells were excluded (n = 7). All HMNs fired regularly at a rate greater than 10 APs·s−1 when the rectangular depolarizing current steps were produced at 100–800 pA of 1000 ms in duration. The mean resting membrane potential was −63.67 ± 0.45 mV (n = 76). The mean evoked current step was 374 ± 20 pA, and the mean AP amplitude generated by depolarizing current steps was 75.0 ± 3.0 mV.
Figure 2.
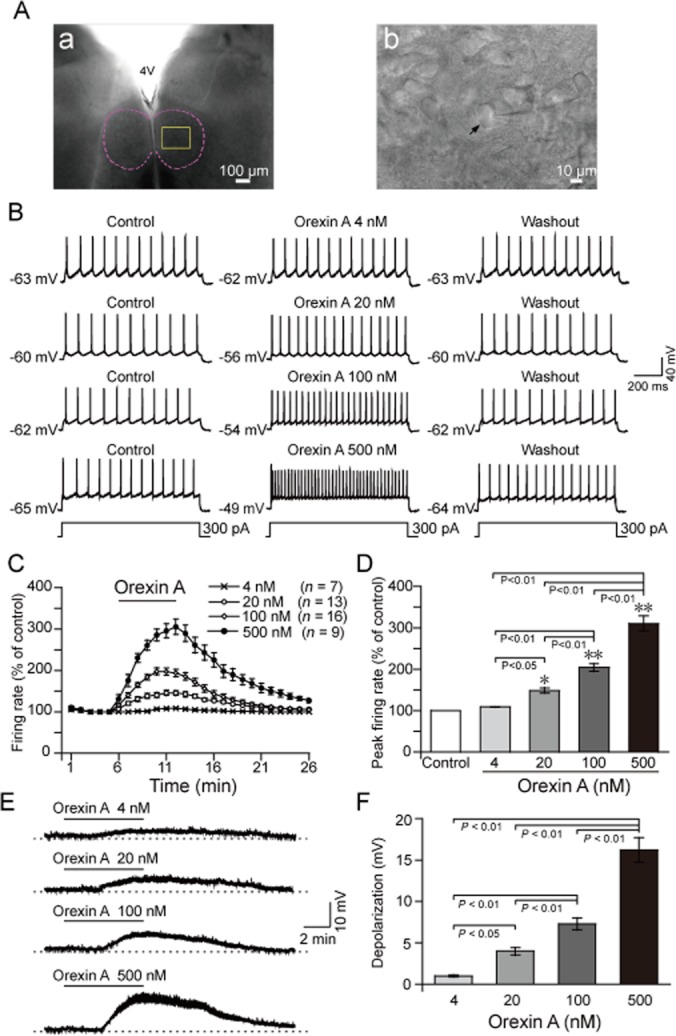
Orexin A increased the firing rate and decreased the membrane potential of HMNs in a concentration-dependent and reversible manner in neonatal rats. (A) Typical example illustrating the location of the hypoglossal nucleus (a) indicated by a dashed line. (b) Higher magnification of the square area in (a), arrow indicating a hypoglossal motoneuron. The recording electrode is shown entering from the right side of the field. (B) Typical examples illustrating repetitive firing of HMNs before (left), during (middle) and 15 min after (right) perfusion with orexin A at 4 (n = 7), 20 (n = 13), 100 (n = 16) and 500 nM (n = 9), respectively. The evoked depolarizing current steps of these 4 neurons were the same as 300 pA of 1000 ms in duration. (C) Time course of the change in the firing rate induced by orexin A at 4, 20, 100 and 500 nM. (D) Mean peak firing rate (% of control) induced by orexin A. (E) Typical examples illustrating orexin A decreased the membrane potential of HMNs at 4, 20, 100 and 500 nM. (F) Effect of orexin A on the membrane potential of HMNs. Values are means ± SEM. *P < 0.05; **P < 0.01, vs control. 4 V, the fourth ventricle.
The HMNs recorded exhibited dose-dependent and reversible excitatory responses to orexin A at 4, 20, 100 or 500 nM, with an increase in the peak firing rate compared with those in the control (Figure 2B,D). As indicated by the firing rate time course, the firing rate of HMNs increased quickly after the addition of orexin A at the fifth minute and decreased after orexin A was washed off. The firing rate returned to basal level in about 15 min (Figure 2C).
In addition to increasing the firing rate, orexin A decreased HMN membrane potential. As shown in Figure 2E,F, orexin A induced depolarization in a dose-dependent and reversible manner, with a decreased membrane potential at concentrations of 4, 20, 100 or 500 nM. When administered at 100 nM, orexin A immediately depolarized the neuron in normal ACSF, and the membrane potential soon returned to the resting level after removal of orexin A (Figure 3A, left). In the presence of TTX (Figure 3A, right), the same effect was observed, suggesting that orexin A-induced depolarization in HMNs was a direct postsynaptic effect. Similar results were observed in other HMNs from five medullary slices. These results indicate that exogenous orexin A increased the firing rate and decreased the membrane potential of the HMNs in a dose-dependent and reversible manner in medullary slices of neonatal rats.
Figure 3.
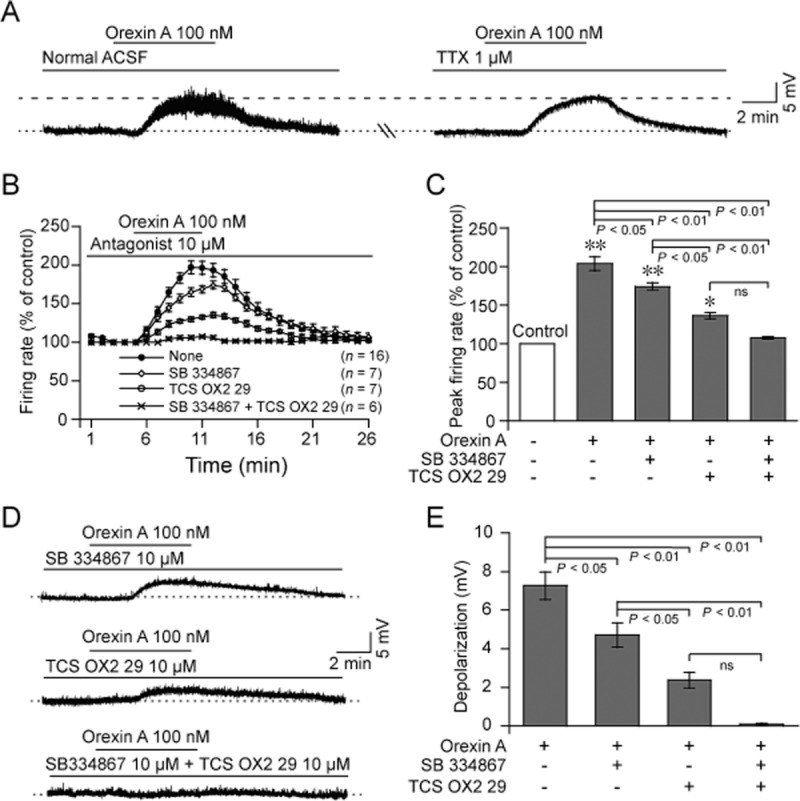
Effects of OX receptor antagonists on orexin A-induced firing rate and depolarization of HMNs in neonatal rats. (A) Typical examples indicating that orexin A (100 nM) induced depolarization of HMN in normal ACSF (left), and this effect persisted in TTX-containing ACSF (right) in the same cell. (B) Time course of orexin A (100 nM)-induced firing rate in ACSF containing OX1 receptor antagonist, SB 334867 (10 μM, n = 7) and OX2 receptor antagonist, TCS OX2 29 (10 μM, n = 7) alone or together (n = 6). (C) Average peak firing rate (% of control) induced by orexin A in ACSF containing SB 334867 and TCS OX2 29 alone or together. (D) Typical examples illustrating that orexin A (100 nM)-induced depolarization was blocked by SB 334867 (up, n = 7), TCS OX2 29 (middle, n = 7), or together (down, n = 6). (E) Change of orexin A-induced depolarization after addition of SB 334867 (10 μM) and TCS OX2 29 (10 μM) alone or together. Values are means ± SEM. *P < 0.05; **P < 0.01, vs control.
Orexin receptor antagonists blocked orexin A-induced increases in firing rate and depolarization of HMNs of the neonatal rats in vitro
To investigate the contribution of OX1 and OX2 receptors to the excitatory effects of orexin A on HMNs, an OX1-selective antagonist SB 334867 (10 μM) and OX2-selective antagonist TCS OX2 29 (10 μM) were used.
In the presence of SB 334867, orexin A (100 nM) increased the firing rate of HMNs, but the peak firing rate was lower than that induced by the neuropeptide alone (Figure 3B,C), while still higher than that of the control (P < 0.01, n = 7; Figure 3B,C). With TCS OX2 29, the firing rate induced by orexin A was significantly suppressed (P < 0.01, n = 7; Figure 3B,C) but still higher than that of the control (P < 0.05, n = 7; Figure 3B,C). In the presence of both SB 334867 and TCS OX2 29, orexin A (100 nM) failed to increase the firing rate of HMNs (P > 0.05, n = 6; Figure 3B,C).
Orexin A depolarized the membrane potential of HMNs. Both SB 334867 (P < 0.05, n = 7) and TCS OX2 29 (P < 0.01, n = 7) blocked orexin A-induced depolarization (Figure 3D,E). Amplitudes of orexin A-induced depolarization in the presence of TCS OX2 29 were significantly smaller than those in the presence of SB 334867 (Figure 3D,E). Furthermore, such depolarization was completely abolished in the ACSF containing SB 334867 and TCS OX2 29 (P < 0.01, n = 6; Figure 3D,E). These results indicate that orexin A increased the firing rate and depolarized the membrane potential of the HMNs in medullary slices of neonatal rats by stimulation of both OX1 and OX2 receptors.
Orexin receptor antagonists decreased the GG-EMG, especially respiratory-related GG activity of the adult rats in vivo
To investigate the contribution of endogenous orexin and its two receptors distributed throughout HMNs to the GG-EMG in vivo, blockade of receptors within the HMNs was employed.
Micro-injections of OX1-selective antagonist SB 334867 (10 mM, 0.1 μL) into the hypoglossal nucleus attenuated respiratory-related GG-EMG (P < 0.01, n = 6; Figure 4A,B), whereas it tended to suppress tonic GG-EMG, although there was no significant difference between vehicle and antagonist treatment (P > 0.05, n = 6; Figure 4A,B). In contrast, OX2-selective antagonist TCS OX2 29 (1 mM, 0.1 μL) decreased tonic GG activity (P < 0.05, n = 5; Figure 4C,D) and respiratory-related GG activity (P < 0.01, n = 5; Figure 4C,D). These results indicate that both OX1 and OX2 receptors are involved in the physiological control mediated by orexin A of GG activity in adult rats.
Figure 4.
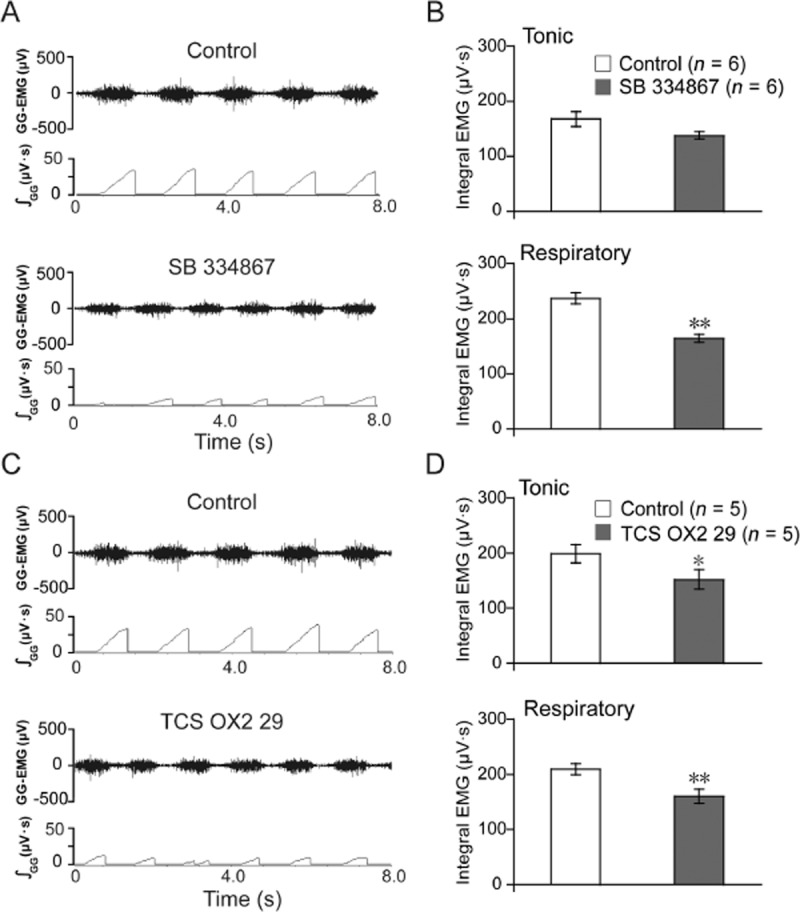
Micro-injection of OX1 receptor antagonist, SB 334867 (10 mM, 0.1 μl, n = 6) or OX2 receptor antagonist, TCS OX2 29 (1 mM, 0.1 μl, n = 5) into the hypoglossal nucleus attenuated GG-EMG in adult rats. (A) Typical example illustrating that SB 334867 mainly decreased the respiratory-related GG-EMG compared with control (saline, 0.1 μl). (B) Changes in the tonic and respiratory-related GG-EMG following a micro-injection of SB 334867. (C) Typical example illustrating that TCS OX2 29 decreased both the tonic and the respiratory-related GG-EMG compared with the control (saline, 0.1 μl). (D) Changes in the tonic and respiratory-related GG-EMG following microinjection of TCS OX2 29. Values are means ± SEM. *P < 0.05, **P < 0.01, vs control.
Effect of lesions of orexin neurons on GG-EMG in adult rats in vivo
Next, we lesioned the bodies of orexin neurons in the bilateral LH, and observed the EMG activity of GG. As shown in Figure 5A,B, both Nissl and anti-orexin A antibody immunohistochemical staining in the LH depict a significant reduction in orexin neurons after orexin-SAP injection into the nucleus 14 days before the observation. After the lesions, the body weight of the rats obviously decreased (data not shown). Lesioning of orexin neurons in bilateral LH was observed to significantly decrease respiratory-related GG-EMG (P < 0.01, n = 6) but did not significantly suppress tonic GG-EMG (n = 7, P > 0.05; Figure 5C,D). Together, with the results from the receptor antagonist experiments, these findings indicate that endogenous orexin A stimulates GG activity in adult rats.
Figure 5.
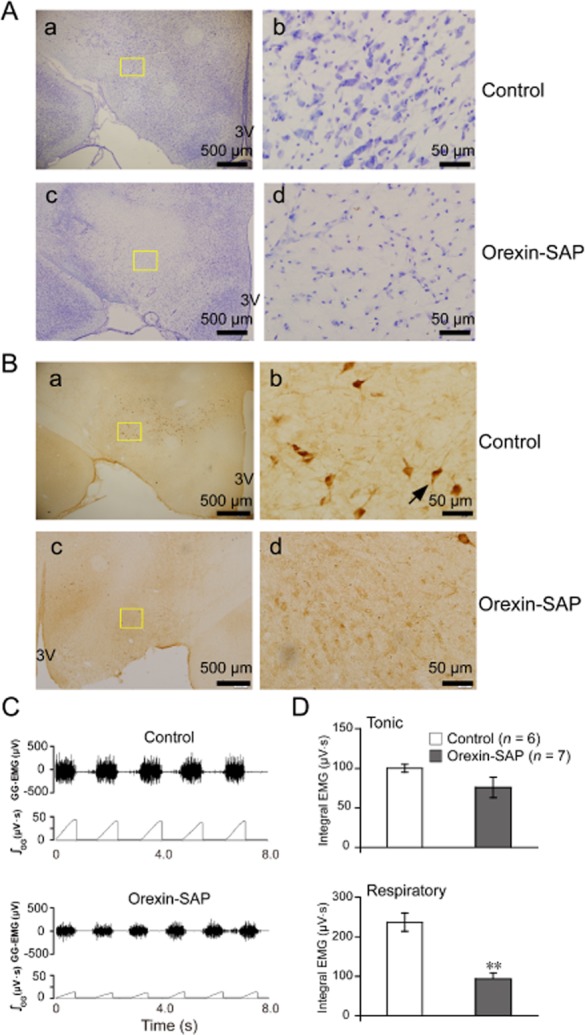
Lesions of orexin neurons in the bilateral LH after orexin-SAP (400 nl per side, 0.43 mg ml−1) micro-injection decreased the respiratory-related GG-EMG in adult rats. (A) Loss of Nissl bodies in the LH after orexin-SAP treatment. (a) Nissl staining showing cells in the saline-treated rats and (c) the cells in the orexin-SAP lesioned rats. Higher magnification of the square area in a (b) and in c (d). (B) Loss of orexin A-immunoreactive neurons in the LH after orexin-SAP treatment. (a) Anti-orexin A antibody immunohistochemistry staining showing cells indicated by an arrow in the saline-treated rats and (c) the cells in the orexin-SAP lesioned rats. Higher magnification of the square area in a (b) and in c (d). (C) Typical example illustrating orexin neurons lesions decreased the respiratory-related GG-EMG. (D) Changes in the tonic and respiratory-related GG-EMG after orexin-SAP treatment (n = 7) compared with the control (n = 6). Values are means ± SEM. **P < 0.01, vs control. 3 V, the third ventricle; orexin-SAP, orexin-saporin.
Discussion
We report that orexin A modulated GG activity, as indicated by the major findings depicted here. Exogenous orexin A administered to the hypoglossal motor pool significantly increased baseline respiratory-related GG activity without affecting breathing rate, heart rate and arterial pressure of adult rats in vivo, and exogenous orexin A excited in vitro HMN by depolarizing the membrane potential and increasing the firing rate of these neurons in neonatal medullary slices. Moreover, GG-EMG was significantly attenuated after the orexin neurons were lesioned, suggesting an endogenously excitatory orexinergic drive to hypoglossal motor outputs to control GG activity under physiological conditions. In addition, in vitro and in vivo experiments verified that the effects of orexin A on GG activity and HMNs were mediated by two types of orexin receptors: OX1 and OX2.
Our finding that orexin A micro-injected into the hypoglossal nucleus increased ipsilateral GG-EMG is consistent with the results that orexin A micro-injected into the trigeminal motor nucleus or hypoglossal nucleus in decerebrate cats markedly increased ipsilateral masseter or GG muscle tone (Peever et al., 2003).
In the present study, we also observed that orexin A had neuroexcitatory activity to HMNs. That is, the neuropeptide depolarized the membrane potential and increased the firing rate of HMNs in medullary slices. In 1998, orexin was reported to significantly increase the frequency of the postsynaptic potential of cultured hypothalamic neurons, suggesting that orexins have neuroexcitatory activity (de Lecea et al., 1998). Also, Yamuy et al. (2004) showed that orexin-activated spinal motoneurons in the cat. This can be explained by the resting membrane potential of HMNs which was depolarized by orexin A and reached the threshold potential to fire the AP. Therefore, orexin A enhanced the firing rate of HMN.
Based upon our data, it took about 3–5 min for the orexin injections to have an effect. This delayed effect may be caused by the diffusion of orexin A to the HMNs and signal transduction process of orexin system. In experiments with neonatal medullary slices, the effect time of orexin is also not immediate. It takes time for orexin signal transduction. On the one hand, orexins directly activate G protein-coupled calcium channels and induce opening of channels and calcium ions influx. On the other hand, orexins activate G protein-coupled phospholipase C, which hydrolyses PIP2 to IP3. IP3 then induces rising of intracellular calcium by activating IP3 receptor. Finally, a series of reactions in target cells happen.
Although orexin A was observed to be involved in regulating GG activity and the firing of HMNs in the present study, the receptor subtypes mediating this effect have not been addressed. It has been reported that orexin A activates OX1 and OX2 receptors, two subtypes of the GPCR family (de Lecea et al., 1998; Sakurai et al., 1998), and that orexin A has similar affinity for each (Sakurai et al., 1998). It is well recognized that HMNs not only receive the projection of orexin neurons (Nixon and Smale, 2007) but also express OX1 (Fung et al., 2001) and OX2 receptors (Volgin et al., 2002). Furthermore, lumbar motoneurons activated by orexin partially depended upon OX1 receptors (Yamuy et al., 2004). Therefore, we investigated the effect of different orexin receptor subtypes on membrane potential and firing rate of HMNs in vitro and GG activity in vivo via specific receptor antagonism in the hypoglossal nucleus. Orexin A-induced depolarization was not blocked by TTX in in vitro experiments, indicating that the depolarization of orexin A on the HMNs was a direct postsynaptic effect. Therefore, highly selective antagonists for postsynaptic OX1 and OX2 receptors were applied to clarify the underlying receptor mechanisms. SB 334867 and TCS OX2 29 separately blocked the orexin A-induced increase in firing rate and depolarization in part at a concentration 100 times higher than that of orexin A. This concentration should be sufficient for blocking orexin A activation of OX1 and OX2 receptors (Sakurai et al., 1998; Smart et al., 2001).
In our work, we observed that GG activity significantly decreased when the hypoglossal nucleus were treated with specific OX1 or OX2 antagonists, indicating that these receptors contribute to the process of respiratory outflow to control GG activity in which orexin A, released from the orexinergic nerve terminal, bound to OX1 and OX2 receptors in HMNs to excite HMNs and increase GG muscle tone.
Data from in vitro experiments were slightly different from in vivo experiments. For example, the amplitudes of orexin A-induced depolarization and firing rates with TCS OX2 29 were significantly less than those with SB 334867, indicating that OX2 receptor might play a more important role in orexin A-induced depolarization and firing rate increasing of HMNs than OX1 receptor. Moreover, orexin A-induced depolarization and firing rate increasing were completely abolished after an addition of both SB 334867 and TCS OX2 29. These two antagonists might not completely block GG activity in vivo. Reports suggest that GG activity is regulated by diverse neurotransmitters, such as serotonin (Jelev et al., 2001; Sood et al., 2005), noradrenaline (Fenik et al., 2005; Chan et al., 2006), histamine (Bastedo et al., 2009), acetylcholine (Liu et al., 2005), GABA (Morrison et al., 2003) and glycine (Morrison et al., 2002), among others. These neurotransmitters regulate the sleep/wake cycle, and orexins play an important role in sleep stability. Orexin neurons are wake-active neurons. They exhibit slow (<10 Hz) tonic discharges during wakefulness and cease firing shortly after the onset of slow wave sleep, but display transient discharges during rapid eye movement (REM) sleep and increase firing before the end of REM sleep. Their activity plays an important wake-promoting role and antagonizes cortical deactivation (EEG synchronization) and loss of muscle tone (Lee et al., 2005; Takahashi et al., 2008). Therefore, people with narcolepsy who lack orexin neurons will suppress postural muscle tone during cataplexy. Orexin may facilitate airway opening when people/animals are actively awake. Serotonin and histamine are also wake-active monoamines. However, both of them endogenously played minimal roles in the normal modulation of GG activity across natural sleep-wake states (Sood et al., 2005; Bastedo et al., 2009), despite the fact that they delivered to the hypoglossal nucleus induced robust GG activation in conscious rats in vivo (Jelev et al., 2001; Bastedo et al., 2009). However, an endogenous noradrenergic drive contributes to GG activation across sleep/wake states (Fenik et al., 2005; Chan et al., 2006). Although the data in our study were recorded in urethane-anaesthetized rats, we do not deny that orexin-driven hypoglossal-GG activity may be meaningful and possibly more apparent during wakefulness. Next we should study more about the orexinergic drive activating hypoglossal motor outflow to GG muscle across sleep/wake states.
To ascertain whether there was an endogenous orexinergic excitatory drive to the GG muscle as was observed in exogenous activity, we blocked orexin receptors in the hypoglossal nucleus or lesioned orexin neurons in the bilateral LH. Because the distribution of the neurons is restricted to the bilateral LH (Peyron et al., 1998; Nambu et al., 1999), specific lesions at this site will remove the endogenous resource of orexins. In the present study, therefore, a specific neurotoxin orexin-SAP was used to lesion the neurons in the bilateral LH; more than 90% of orexin neurons are reported to undergo apoptosis 14 days after neurotoxin administration (Gerashchenko et al., 2001). Gerashchenko et al. also reported that melanin-concentrating hormone (MCH) neurons in the LH were found to be partly decreased at the same time after orexin-SAP injection. So we did not stain the immunoreactivity of MCH neurons in these rats in the present study. During the last decades, growing experimental evidence indicates that MCH may play a crucial role in the homeostatic regulation of REM sleep (Peyron et al., 2009). Saito et al. (2001) reported that hypoglossal nucleus express MCH receptors. However, there is no convincing evidence demonstrating MCH neurons control motor outflow of HMNs and activity of GG muscle. Therefore, our results that GG-EMG was markedly reduced in lesioned rats suggest that endogenous orexins mainly excited GG activity. Stettner and Kubin (2013) also investigated the effect of orexin neurons on hypoglossal nerve activity (XIIa), respiratory rate in urethane-anaesthetized, vagotomized rats. They reported that XIIa and respiratory rate were reduced when OX1 and OX2 receptors were antagonized in the perifornical (PF) region of the posterior hypothalamus, but not in the hypoglossal nucleus. These results suggested that hypoglossal motoneuronal activation is mediated by local orexin-dependent mechanisms within the posterior hypothalamus. Orexin receptor antagonism in the PF in Stettner's experiment is the equation of orexin neurons lesion. In our study, we lesioned the orexin neurons by using neurotoxin orexin-SAP and found the weakening of GG-EMG in lesioned animals, consistent with Stettner's result. Additionally, GG-EMG decreased after antagonizing activation of OX1 or OX2 receptors in the hypoglossal nucleus using their specific antagonists in vivo. These data suggest that endogenous orexins released from the LH enhanced GG tone by acting on OX1 and OX2 receptors in the medullary HMNs.
We think anaesthesia is not completely equal to sleep. The mechanisms of anaesthesia and sleep are not the same. During sleep, orexin system is not active. Orexin system induces wakefulness. However, orexin is not the sole factor to prompt wakefulness. Orexin neurons in bilateral LH of the rats were lesioned 14 days before GG-EMG was recorded in our studies. Although most of orexin neurons were verified to be lesioned, the lesioned rats did not remain under sleep condition all day. Patients with narcolepsy are also wakeful for some time. We have not found direct evidence to verify that orexin system could be activated under anesthetized conditions. We believe that the effect of orexin exists under anaesthetized conditions because the GG-EMG significantly decreased when the hypoglossal nucleus was micro-injected with orexin receptor antagonists in our study. In addition, some reported studies indirectly supported this idea. For example, Kayaba et al. (2003) reported that basal blood pressure in orexin knockout mice was lower in both anaesthetized and conscious conditions. In another paper, Gompf et al. reported coordinated inhibition of hypothalamic orexinergic and locus coeruleus noradrenergic neurons is not required for anesthetic induction. In this study, the authors observed that normal emergence from halothane-induced hypnosis in orexin-deficient mice, which suggests that additional wake-promoting systems likely remain active during general anesthesia produced by halothane (Gompf et al., 2009). Finally, Kushikata et al. (2010) also reported that plasma concentration of orexin A did not change during propofol–fentanyl anaesthesia (1 and 2 h after the induction of anaesthesia) in patients undergoing eye surgery. All these results suggest that the effect of orexin system does not completely disappear under anaesthetized state.
The current findings are important to research regarding upper airway physiology, especially with respect to the pathophysiology of OSA and narcolepsy. Either a lack of orexin neurons or a defect in OX2 receptors in humans or animals causes severe narcolepsy, a condition in which the awake patient can lose muscle tone suddenly and fall asleep, depicting the disorder of sleep stability (Chemelli et al., 1999; Lin et al., 1999; Siegel, 1999; Peyron et al., 2000; Thannickal et al., 2000; Hara et al., 2001; Mochizuki et al., 2004). Also, dysfunction of orexin systems might be involved in the pathophysiology of OSA because plasma orexin A was lower in patients with OSA (Nishijima et al., 2003). Previous studies have shown that orexin plays an important role in the promotion and maintenance of arousal. OSA patients suffer frequent arousal during sleep. However, so far, the correlation between OSA and orexin remained controversial. About 25% of patients with narcolepsy occur with OSA at the same time (Sansa et al., 2010), and human narcolepsy-cataplexy is associated with central orexin deficiency (Nishino et al., 2000). All these reports suggested hypothalamic dysfunction in narcolepsy. Sansa et al. (2010) also reported that there may be a connection between narcolepsy and OSA. Narcolepsy with cataplexy occurring during prepubertal age is frequently accompanied by overweight or obesity (Poli et al., 2013). There is evidence showing that OSA occurs more frequently and may be more severe in children and adolescents who are overweight or obese (Narang and Mathew, 2012). Both conditions can potentiate the adverse impact of each. The obesity may cause airway obstruction during sleep (OSA). An important and possible pathophysiological mechanism contributing to this association is adenotonsillar hypertrophy due to increased somatic growth (Arens and Muzumdar, 2010). In addition, functional factors that operate to promote upper airway obstruction OSA in obese individuals during sleep include altered neuromuscular tone resulting in greater upper airway collapsibility during sleep (Narang and Mathew, 2012). We believe that it is necessary to know whether orexin is the connection between narcolepsy and OSA.
In conclusion, endogenous orexin A released from the hypothalamic orexin neurons increased GG activity via activating OX1 and OX2 receptors in medullary HMNs, indicating that hypothalamic orexin neurons can participate in controlling upper airway patency. Furthermore, inhibition of OX1 and OX2 receptors can cause airway collapse, which may play an important role in the pathogenesis of OSA.
Acknowledgments
This study was supported in part by Grants-in-Aid for Scientific Research from the Science and Technology Commission of Shanghai Municipality (09JC1402100, 14JC1400900, 13dz2260700), the National Natural Science Foundation of China (81070001, J1210041, 31200867 and 31121061), the National Basic Research Program of China (2011CB711000), Shanghai Leading Academic Discipline Project (B119), a key laboratory program of the Education Commission of Shanghai Municipality (ZDSYS14005) and the Shanghai Medical Image Computing and Computer Assisted Intervention Key Laboratory (13DZ2272200). We are grateful to Dr. P. Feng in the Division of Pulmonary, Critical Care and Sleep Medicine, Case Western Reserve University for providing technical supports.
Glossary
- AP
action potential
- GG
genioglossus
- GG-EMG
GG muscle electromyogram
- HMN
hypoglossal motoneuron
- LH
lateral hypothalamus
- OSA
obstructive sleep apnoea
- orexin-SAP
orexin-saporin
- PF
perifornical
- REM
rapid eye movement
- XIIa
hypoglossal nerve activity
Author contributions
G. H. Z., Z. L. L, Z. L. H., and L. L. S. conceived and designed the experiments. G. H. Z., Z. L. L., B. J. Z., W. Y. G., N. N. S. and W. Z. performed the experiments. G. H. Z., Z. L. L., Y. X. C. and S. Q. L. analysed the data. L. L. S. contributed reagents/materials/analysis tools. G. H. Z., Z. L. L., Z. L. H. and L. L. S. wrote the article.
Conflicts of interest
All authors declare no conflict of interest.
References
- Alexander SPH, Benson HE, Faccenda E, Pawson AJ, Sharman JL, Spedding M, Peters JA, Harmar AJ, CGTP Collaborators The Concise Guide to PHARMACOLOGY 2013/14: G protein-coupled receptors. Br J Pharmacol. 2013;170:1459–1562. doi: 10.1111/bph.12445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arens R, Muzumdar H. Childhood obesity and obstructive sleep apnea syndrome. J Appl Physiol. 2010;108:436–444. doi: 10.1152/japplphysiol.00689.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azhdari-Zarmehri H, Esmaeili MH, Sofiabadi M, Haghdoost-Yazdi H. Orexin receptor type-1 antagonist SB-334867 decreases morphine-induced antinociceptive effect in formalin test. Pharmacol Biochem Behav. 2013;112:64–70. doi: 10.1016/j.pbb.2013.09.018. [DOI] [PubMed] [Google Scholar]
- Bastedo T, Chan E, Park E, Liu H, Horner RL. Modulation of genioglossus muscle activity across sleep-wake states by histamine at the hypoglossal motor pool. Sleep. 2009;32:1313–1324. doi: 10.1093/sleep/32.10.1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger AJ, Bayliss DA, Viana F. Development of hypoglossal motoneurons. J Appl Physiol. 1996;81:1039–1048. doi: 10.1152/jappl.1996.81.3.1039. [DOI] [PubMed] [Google Scholar]
- Chan E, Steenland HW, Liu H, Horner RL. Endogenous excitatory drive modulating respiratory muscle activity across sleep-wake states. Am J Respir Crit Care Med. 2006;174:1264–1273. doi: 10.1164/rccm.200605-597OC. [DOI] [PubMed] [Google Scholar]
- Chemelli RM, Willie JT, Sinton CM, Elmquist JK, Scammell T, Lee C, et al. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell. 1999;98:437–451. doi: 10.1016/s0092-8674(00)81973-x. [DOI] [PubMed] [Google Scholar]
- Dutschmann M, Kron M, Morschel M, Gestreau C. Activation of Orexin B receptors in the pontine Kolliker-Fuse nucleus modulates pre-inspiratory hypoglossal motor activity in rat. Respir Physiol Neurobiol. 2007;159:232–235. doi: 10.1016/j.resp.2007.06.004. [DOI] [PubMed] [Google Scholar]
- Elias CF, Saper CB, Maratos-Flier E, Tritos NA, Lee C, Kelly J, et al. Chemically defined projections linking the mediobasal hypothalamus and the lateral hypothalamic area. J Comp Neurol. 1998;402:442–459. [PubMed] [Google Scholar]
- Fenik V, Kubin L, Okabe S, Pack AI, Davies RO. Differential sensitivity of laryngeal and pharyngeal motoneurons to iontophoretic application of serotonin. Neuroscience. 1997;81:873–885. doi: 10.1016/s0306-4522(97)00215-7. [DOI] [PubMed] [Google Scholar]
- Fenik VB, Davies RO, Kubin L. REM sleep-like atonia of hypoglossal (XII) motoneurons is caused by loss of noradrenergic and serotonergic inputs. Am J Respir Crit Care Med. 2005;172:1322–1330. doi: 10.1164/rccm.200412-1750OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fung SJ, Yamuy J, Sampogna S, Morales FR, Chase MH. Hypocretin (orexin) input to trigeminal and hypoglossal motoneurons in the cat: a double-labeling immunohistochemical study. Brain Res. 2001;903:257–262. doi: 10.1016/s0006-8993(01)02318-6. [DOI] [PubMed] [Google Scholar]
- Gerashchenko D, Kohls MD, Greco M, Waleh NS, Salin-Pascual R, Kilduff TS, et al. Hypocretin-2-saporin lesions of the lateral hypothalamus produce narcoleptic-like sleep behavior in the rat. J Neurosci. 2001;21:7273–7283. doi: 10.1523/JNEUROSCI.21-18-07273.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gompf H, Chen J, Sun Y, Yanagisawa M, Aston-Jones G, Kelz MB. Halothane-induced hypnosis is not accompanied by inactivation of orexinergic output in rodents. Anesthesiology. 2009;111:1001–1009. doi: 10.1097/ALN.0b013e3181b764b3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Forero D, Montero F, Garcia-Morales V, Dominguez G, Gomez-Perez L, Garcia-Verdugo JM, et al. Endogenous Rho-kinase signaling maintains synaptic strength by stabilizing the size of the readily releasable pool of synaptic vesicles. J Neurosci. 2012;32:68–84. doi: 10.1523/JNEUROSCI.3215-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara J, Beuckmann CT, Nambu T, Willie JT, Chemelli RM, Sinton CM, et al. Genetic ablation of orexin neurons in mice results in narcolepsy, hypophagia, and obesity. Neuron. 2001;30:345–354. doi: 10.1016/s0896-6273(01)00293-8. [DOI] [PubMed] [Google Scholar]
- Harris GC, Aston-Jones G. Arousal and reward: a dichotomy in orexin function. Trends Neurosci. 2006;29:571–577. doi: 10.1016/j.tins.2006.08.002. [DOI] [PubMed] [Google Scholar]
- Igarashi N, Tatsumi K, Nakamura A, Sakao S, Takiguchi Y, Nishikawa T, et al. Plasma orexin-A levels in obstructive sleep apnea-hypopnea syndrome. Chest. 2003;124:1381–1385. doi: 10.1378/chest.124.4.1381. [DOI] [PubMed] [Google Scholar]
- Jelev A, Sood S, Liu H, Nolan P, Horner RL. Microdialysis perfusion of 5-HT into hypoglossal motor nucleus differentially modulates genioglossus activity across natural sleep-wake states in rats. J Physiol. 2001;532((Pt 2)):467–481. doi: 10.1111/j.1469-7793.2001.0467f.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan AS, White DP. Pharyngeal motor control and the pathogenesis of obstructive sleep apnea. Respir Physiol Neurobiol. 2008;160:1–7. doi: 10.1016/j.resp.2007.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kayaba Y, Nakamura A, Kasuya Y, Ohuchi T, Yanagisawa M, Komuro I, et al. Attenuated defense response and low basal blood pressure in orexin knockout mice. Am J Physiol Regul Integr Comp Physiol. 2003;285:R581–R593. doi: 10.1152/ajpregu.00671.2002. [DOI] [PubMed] [Google Scholar]
- Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG. Animal research: reporting in vivo experiments: the ARRIVE guidelines. Br J Pharmacol. 2010;160:1577–1579. doi: 10.1111/j.1476-5381.2010.00872.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kukkonen JP, Holmqvist T, Ammoun S, Akerman KE. Functions of the orexinergic/hypocretinergic system. Am J Physiol Cell Physiol. 2002;283:C1567–C1591. doi: 10.1152/ajpcell.00055.2002. [DOI] [PubMed] [Google Scholar]
- Kushikata T, Yoshida H, Kudo M, Kudo T, Hirota K. Changes in plasma orexin A during propofol-fentanyl anaesthesia in patients undergoing eye surgery. Br J Anaesth. 2010;104:723–727. doi: 10.1093/bja/aeq098. [DOI] [PubMed] [Google Scholar]
- de Lecea L, Kilduff TS, Peyron C, Gao X, Foye PE, Danielson PE, et al. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc Natl Acad Sci U S A. 1998;95:322–327. doi: 10.1073/pnas.95.1.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee MG, Hassani OK, Jones BE. Discharge of identified orexin/hypocretin neurons across the sleep-waking cycle. J Neurosci. 2005;25:6716–6720. doi: 10.1523/JNEUROSCI.1887-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin L, Faraco J, Li R, Kadotani H, Rogers W, Lin X, et al. The sleep disorder canine narcolepsy is caused by a mutation in the hypocretin (orexin) receptor 2 gene. Cell. 1999;98:365–376. doi: 10.1016/s0092-8674(00)81965-0. [DOI] [PubMed] [Google Scholar]
- Liu X, Sood S, Liu H, Horner RL. Opposing muscarinic and nicotinic modulation of hypoglossal motor output to genioglossus muscle in rats in vivo. J Physiol. 2005;565((Pt 3)):965–980. doi: 10.1113/jphysiol.2005.084657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGrath JC, Drummond GB, McLachlan EM, Kilkenny C, Wainwright CL. Guidelines for reporting experiments involving animals: the ARRIVE guidelines. Br J Pharmacol. 2010;160:1573–1576. doi: 10.1111/j.1476-5381.2010.00873.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miles GB, Lipski J, Lorier AR, Laslo P, Funk GD. Differential expression of voltage-activated calcium channels in III and XII motoneurones during development in the rat. Eur J Neurosci. 2004;20:903–913. doi: 10.1111/j.1460-9568.2004.03550.x. [DOI] [PubMed] [Google Scholar]
- Mochizuki T, Crocker A, McCormack S, Yanagisawa M, Sakurai T, Scammell TE. Behavioral state instability in orexin knock-out mice. J Neurosci. 2004;24:6291–6300. doi: 10.1523/JNEUROSCI.0586-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrison JL, Sood S, Liu X, Liu H, Park E, Nolan P, et al. Glycine at hypoglossal motor nucleus: genioglossus activity, CO(2) responses, and the additive effects of GABA. J Appl Physiol. 2002;93:1786–1796. doi: 10.1152/japplphysiol.00464.2002. [DOI] [PubMed] [Google Scholar]
- Morrison JL, Sood S, Liu H, Park E, Nolan P, Horner RL. GABAA receptor antagonism at the hypoglossal motor nucleus increases genioglossus muscle activity in NREM but not REM sleep. J Physiol. 2003;548((Pt 2)):569–583. doi: 10.1113/jphysiol.2002.033696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nambu T, Sakurai T, Mizukami K, Hosoya Y, Yanagisawa M, Goto K. Distribution of orexin neurons in the adult rat brain. Brain Res. 1999;827:243–260. doi: 10.1016/s0006-8993(99)01336-0. [DOI] [PubMed] [Google Scholar]
- Narang I, Mathew JL. Childhood obesity and obstructive sleep apnea. Journal of nutrition and metabolism. 2012;2012:134202. doi: 10.1155/2012/134202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishijima T, Sakurai S, Arihara Z, Takahashi K. Plasma orexin-A-like immunoreactivity in patients with sleep apnea hypopnea syndrome. Peptides. 2003;24:407–411. doi: 10.1016/s0196-9781(03)00055-x. [DOI] [PubMed] [Google Scholar]
- Nishino S, Ripley B, Overeem S, Lammers GJ, Mignot E. Hypocretin (orexin) deficiency in human narcolepsy. Lancet. 2000;355:39–40. doi: 10.1016/S0140-6736(99)05582-8. [DOI] [PubMed] [Google Scholar]
- Nixon JP, Smale L. A comparative analysis of the distribution of immunoreactive orexin A and B in the brains of nocturnal and diurnal rodents. Behav Brain Funct. 2007;3:28. doi: 10.1186/1744-9081-3-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nunez-Abades PA, Cameron WE. Morphology of developing rat genioglossal motoneurons studied in vitro: relative changes in diameter and surface area of somata and dendrites. J Comp Neurol. 1995;353:129–142. doi: 10.1002/cne.903530112. [DOI] [PubMed] [Google Scholar]
- Pawson AJ, Sharman JL, Benson HE, Faccenda E, Alexander SP, Buneman OP, Davenport AP, McGrath JC, Peters JA, Southan C, Spedding M, Yu W, Harmar AJ, NC-IUPHAR The IUPHAR/BPS Guide to PHARMACOLOGY: an expert-driven knowledgebase of drug targets and their ligands. Nucl. Acids Res. 2014;42:D1098–D1106. doi: 10.1093/nar/gkt1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. 6th edn. San Diego, CA: Academic Press; 2007. [Google Scholar]
- Peever JH, Lai YY, Siegel JM. Excitatory effects of hypocretin-1 (orexin-A) in the trigeminal motor nucleus are reversed by NMDA antagonism. J Neurophysiol. 2003;89:2591–2600. doi: 10.1152/jn.00968.2002. [DOI] [PubMed] [Google Scholar]
- Peyron C, Tighe DK, van den Pol AN, de Lecea L, Heller HC, Sutcliffe JG, et al. Neurons containing hypocretin (orexin) project to multiple neuronal systems. J Neurosci. 1998;18:9996–10015. doi: 10.1523/JNEUROSCI.18-23-09996.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peyron C, Faraco J, Rogers W, Ripley B, Overeem S, Charnay Y, et al. A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat Med. 2000;6:991–997. doi: 10.1038/79690. [DOI] [PubMed] [Google Scholar]
- Peyron C, Sapin E, Leger L, Luppi PH, Fort P. Role of the melanin-concentrating hormone neuropeptide in sleep regulation. Peptides. 2009;30:2052–2059. doi: 10.1016/j.peptides.2009.07.022. [DOI] [PubMed] [Google Scholar]
- Poli F, Pizza F, Mignot E, Ferri R, Pagotto U, Taheri S, et al. High prevalence of precocious puberty and obesity in childhood narcolepsy with cataplexy. Sleep. 2013;36:175–181. doi: 10.5665/sleep.2366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remmers JE, De Groot WJ, Sauerland EK, Anch AM. Pathogenesis of upper airway occlusion during sleep. J Appl Physiol. 1978;44:931–938. doi: 10.1152/jappl.1978.44.6.931. [DOI] [PubMed] [Google Scholar]
- Saito Y, Cheng M, Leslie FM, Civelli O. Expression of the melanin-concentrating hormone (MCH) receptor mRNA in the rat brain. J Comp Neurol. 2001;435:26–40. doi: 10.1002/cne.1191. [DOI] [PubMed] [Google Scholar]
- Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell. 1998;92:573–585. doi: 10.1016/s0092-8674(00)80949-6. [DOI] [PubMed] [Google Scholar]
- Sansa G, Iranzo A, Santamaria J. Obstructive sleep apnea in narcolepsy. Sleep Med. 2010;11:93–95. doi: 10.1016/j.sleep.2009.02.009. [DOI] [PubMed] [Google Scholar]
- Saper CB, Scammell TE, Lu J. Hypothalamic regulation of sleep and circadian rhythms. Nature. 2005;437:1257–1263. doi: 10.1038/nature04284. [DOI] [PubMed] [Google Scholar]
- Scammell TE, Griffin JD, Elmquist JK, Saper CB. Microinjection of a cyclooxygenase inhibitor into the anteroventral preoptic region attenuates LPS fever. Am J Physiol. 1998;274((3 Pt 2)):R783–R789. doi: 10.1152/ajpregu.1998.274.3.R783. [DOI] [PubMed] [Google Scholar]
- Shirasaka T, Takasaki M, Kannan H. Cardiovascular effects of leptin and orexins. Am J Physiol Regul Integr Comp Physiol. 2003;284:R639–R651. doi: 10.1152/ajpregu.00359.2002. [DOI] [PubMed] [Google Scholar]
- Siegel JM. Narcolepsy: a key role for hypocretins (orexins) Cell. 1999;98:409–412. doi: 10.1016/s0092-8674(00)81969-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smart D, Sabido-David C, Brough SJ, Jewitt F, Johns A, Porter RA, et al. SB-334867-A: the first selective orexin-1 receptor antagonist. Br J Pharmacol. 2001;132:1179–1182. doi: 10.1038/sj.bjp.0703953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sood S, Morrison JL, Liu H, Horner RL. Role of endogenous serotonin in modulating genioglossus muscle activity in awake and sleeping rats. Am J Respir Crit Care Med. 2005;172:1338–1347. doi: 10.1164/rccm.200502-258OC. [DOI] [PubMed] [Google Scholar]
- Stettner GM, Kubin L. Antagonism of orexin receptors in the posterior hypothalamus reduces hypoglossal and cardiorespiratory excitation from the perifornical hypothalamus. J Appl Physiol. 2013;114:119–130. doi: 10.1152/japplphysiol.00965.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K, Lin JS, Sakai K. Neuronal activity of orexin and non-orexin waking-active neurons during wake-sleep states in the mouse. Neuroscience. 2008;153:860–870. doi: 10.1016/j.neuroscience.2008.02.058. [DOI] [PubMed] [Google Scholar]
- Thannickal TC, Moore RY, Nienhuis R, Ramanathan L, Gulyani S, Aldrich M, et al. Reduced number of hypocretin neurons in human narcolepsy. Neuron. 2000;27:469–474. doi: 10.1016/s0896-6273(00)00058-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volgin DV, Saghir M, Kubin L. Developmental changes in the orexin 2 receptor mRNA in hypoglossal motoneurons. Neuroreport. 2002;13:433–436. doi: 10.1097/00001756-200203250-00014. [DOI] [PubMed] [Google Scholar]
- Willie JT, Chemelli RM, Sinton CM, Yanagisawa M. To eat or to sleep? Orexin in the regulation of feeding and wakefulness. Annu Rev Neurosci. 2001;24:429–458. doi: 10.1146/annurev.neuro.24.1.429. [DOI] [PubMed] [Google Scholar]
- Yamuy J, Fung SJ, Xi M, Chase MH. Hypocretinergic control of spinal cord motoneurons. J Neurosci. 2004;24:5336–5345. doi: 10.1523/JNEUROSCI.4812-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young JK, Wu M, Manaye KF, Kc P, Allard JS, Mack SO, et al. Orexin stimulates breathing via medullary and spinal pathways. J Appl Physiol. 2005;98:1387–1395. doi: 10.1152/japplphysiol.00914.2004. [DOI] [PubMed] [Google Scholar]
- Zhang W, Shimoyama M, Fukuda Y, Kuwaki T. Multiple components of the defense response depend on orexin: evidence from orexin knockout mice and orexin neuron-ablated mice. Auton Neurosci. 2006;126-127:139–145. doi: 10.1016/j.autneu.2006.02.021. [DOI] [PubMed] [Google Scholar]


