Abstract
Circulating Tumor cells (CTCs) represent tumor cells in the blood stream dislodged from the primary tumor. The presence of CTCs in the bloodstream provides a unique opportunity to sample cancer tissue by means of a relatively less-invasive “liquid biopsy.” Over the past decade, there has been a tremendous increase in the amount of research examining the potential clinical utility of CTCs in the management of cancer. A number of techniques to refine the sensitivity and range of CTC assays are also in development. In this article, we review the recent developments in the current and potential clinical applications of CTCs in breast cancer. CTC enumeration already has an established role as a prognostic biomarker in metastatic breast cancer, while molecular characterization of CTCs can serve as a potential predictive biomarker for therapy selection, pharmacodynamic evaluation, and identification of novel actionable targets for novel therapies. The role of CTCs in breast cancer screening and detection of recurrence is currently limited. Further development in techniques will be pivotal in enhancing the broad applicability of CTCs and advancing the field of personalized breast cancer therapy.
Keywords: circulating tumor cells, molecular characterization, breast cancer, personalized medicine
Introduction
Circulating tumor cells (CTCs), represent tumor cells in the blood stream dislodged from the primary tumor. CTCs comprise of a heterogeneous population of cells, including apoptotic tumor cells, i.e. cells passively shed from tumors due to vascular compromise, as well as viable tumor cells capable of initiating and establishing cancer metastasis by seeding peripheral organs. CTCs were first described as early as 1869 as carcinoma associated circulating epithelial cells, by Dr. T.R. Ashworth [1]. Subsequent studies demonstrated their neoplastic origin and common clonality with the primary tumor [2].
The presence of CTCs in the bloodstream provides a unique opportunity to study cancer cells using a simple peripheral blood test rather than an invasive tissue biopsy. Unfortunately CTCs are rare, even in patients with metastatic disease, circulate for unknown lengths of time, and are outnumbered by surrounding blood cells at a ratio of about a million to one making their detection technologically challenging. In the last few years, tremendous strides have been made in isolating and characterizing CTCs in a wide multitude of ways. In this review, we will summarize the recent developments in the technology, and current and potential clinical applications of CTCs in breast cancer.
Current methods of isolating CTCs
Over the past decade several innovative techniques have been developed to detect CTCs in the bloodstream. Most of the techniques combine an enrichment step prior to detection and analysis. The key methods of CTC isolation are summarized below:
Epithelial Based: Most current commercial systems, including the FDA-approved CellSearch™ System, utilize EpCAM (Epithelial Cell Adhesion molecule) and CK (Cytokeratin) based immunomagnetic systems to distinguish tumor cells from normal blood cells. In this technique, first enrichment of CTCs is accomplished by labeling the cells with anti-EpCAM antibodies (attached to ferrofluid nanoparticles) and separating them by applying a magnetic field. Subsequently, the cells are stained for cytokeratins to separate the CTCs from contaminating cell types [3]. While this technology is helpful for CTC enumeration, the cell fixation techniques restricts detailed RNA-based molecular assays and preclude functional analyses. Refinements of this technology, such as utilizing dielectrophoretic and microfluidics sorting, are being developed to overcome these limitations[4].
Size based: Several methods utilize the larger size of CTCs, in comparison to other blood cells to enrich them from blood samples. The ISET (Isolation by size of epithelial tumor cells) system is a prime example of using this principle [5]. There is however significant variation in the size of CTCs and this could lead to variability in the analysis.
qRT-PCR based: These extremely sensitive assays identify CTCs by detection of multiple RNA transcripts that are characteristic of cancer cells rather than the contaminating leukocytes [6]. This method can often be technically challenging and can have a high number of false positive and false negative cells [7].
Fiber optic array laser scanning technology (FAST) based: This cytometric analysis has the ability to simultaneously evaluate at the fluorescent emission of a large number of cells at the same time, without a need for prior enrichment [8]. As a result, it is very useful to detect rare CTCs in blood samples in a comparatively faster way. This technique might not be fully suitable for further downstream molecular characterization.
Microfluidics and herringbone based: The microfluidics based cell sorting technique utilizes custom built micro-chips to identify and separate CTCs from blood samples with increased yield and purity, compared to currently available technologies. The “Herringbone” design, in which specially constructed grooves in the ceiling of the microfluidic chamber creates microvortices of flow directing cells toward the walls of the device for increased capture. The tumor cells are unfixed and captured under conditions that allow sophisticated molecular analyses, including genomic sequencing [9, 10].
Role of CTCs in management of Metastatic Breast Cancer
A) Prognostic marker
The use of CTC enumeration as a prognostic factor has been well established in several tumor types including breast cancer. In the landmark breast cancer CTC study, Cristofanilli et al. prospectively evaluated CTC counts from 177 patients with measurable metastatic breast cancer prior to and after the initiation of a new therapy utilizing the Veridex CellSearch® assay. Of these 177 patients, 61% were found to have detectable CTCs in their blood. The authors demonstrated that that the number of CTCs was an independent predictor of progression-free survival and overall survival. For instance, women with ≥5 CTCs per 7.5 ml prior to initiation of therapy had a significantly shorter median overall survival than did those with <5 CTCs/7.5 ml blood (10.2 months versus >18 months). Furthermore, patients with persistent CTCs (>/=5 CTCs per 7.5 ml) despite initiation of therapy also had significantly shorter median overall survival (8.2 months versus > 18 months). Several subsequent studies have demonstrated the role of CTC enumeration in determining prognosis of patients with metastatic breast cancer [3, 11, 12]. The key studies and trials evaluating CTCs in breast cancer are summarized in Table 1.
Table 1.
Summary of the key studies and trials evaluating CTCs in metastatic breast cancer
| Literature | Objective | Study Design | Method of detection | Outcomes |
|---|---|---|---|---|
| Cristofanalli et al, 2004 [3] | To evaluate whether CTC counts are associated with survival | CTC counts done before the start of a new line of therapy and at the 1st follow-up visit | CellSearch system | Patients with a higher baseline CTC count had shorter median PFS (2.7 months vs. 7.0 months) and shorter OS (10.1 months vs. >18 months). The difference was also seen at the 1st follow-up CTC count. |
| Liu et al, 2009 [17] | To evaluate whether CTC counts are associated with radiographic disease progression | Serial CTC counts done over time in patients starting a new therapy with progressive metastatic breast cancer Radiographic studies were performed in 9- to 12-week intervals |
CellSearch system | There was a strong correlation between CTC counts and radiographic disease progression in patients receiving chemotherapy or endocrine therapy for metastatic breast cancer |
| Bardia et al, 2010 [20] | To evaluate the individual and joint contribution of CTCs, with the commercial tumor marker CA 27–29 in the prospective trial (TBCRC 005) | CTC counts done before the start of a new line of therapy and at the 1st follow-up visit | CellSearch system | Change in CA2729 change from baseline to week 3–4 post-therapy was independently and more strongly associated with progression of disease as compared to change in CTC count |
| Pierga et al, 2012 [10] | To examine whether changes in CTC counts between inclusion and before the 2nd cycle are associated with PFS and OS | CTC counts were done at baseline, before cycle 2, before cycle 3/4 (timed with scans) and at the time of progression | CellSearch System | Elevated CTCs before the 2nd cycle are an early sign of poor PFS and OS |
| Min and Bardia et al, 2012 [12] | To investigate the changes in epithelial and mesenchymal composition of CTCs metastatic breast cancer | CTC evaluation done before the start of a new line of therapy and various serial intervals after start of therapy | RNA-ISH and sequencing | CTCs display dynamic shifts in their molecular expression profiles between an epithelial state and a mesenchymal state (EMT) in response to therapies, and the shifts correlate with a response to therapy (epithelial predominant) and progression of disease (more mesenchymal expression) |
CTC = Circulating Tumor Cell
PFS = Progression Free Survival
OS = Overall Survival
TBCRC = Translational Breast Cancer Research Symposium
ISH = In-situ Hybridization
EMT = Epithelial and Mesenchymal Transition
Are elevated CTC counts associated with worse prognosis in all breast cancer subtypes? This was recently evaluated in a retrospective analysis of 517 patients, which indicated that higher numbers of CTCs were strongly associated with a poor prognosis in all subtypes except in HER2-positive patients [13]. Similarly another prospective multicenter trial with 468 patients confirmed ability of CTCs to predict an inferior PFS in all groups except patients with HER2-positive cancer. However, a strong improvement in OS based on low CTC numbers was demonstrated regardless of subtype [14]. Further investigation is warranted to clarify whether CTCs would be useful in HER2-positive patients or if their utility would be restricted only to other subtypes.
B) Monitoring therapeutic response
Like any biomarker, CTC enumeration can potentially be utilized to evaluate response to therapy [3, 15]. This can be particularly helpful for patients with bone metastasis where radiological changes in bone scans can be difficult to detect and can take months to manifest. Studies have confirmed that patients with extensive bone metastases have higher mean CTC counts [16]. In a small prospective study by Liu et al. (2009), patients with metastatic breast cancer starting a new line of therapy (N=68) for a measurable progression underwent CTC counts at baseline and every 3–4 weeks after initiation of new therapy. These counts were subsequently correlated with imaging conducted every 9–12 weeks. This study noted a strong correlation between CTC counts and traditional radiology based progression [17]. This association was not restricted to only CTC counts at the time of imaging, but rather was seen as far back as 7–9 weeks before imaging. Thus, the CTC counts could provide vital information about therapeutic response prior to radiological imaging.
Prior to incorporation into clinical practice, a new biomarker needs to be compared to existing biomarkers, and relative predictive contribution in addition to existing predictive factors needs to be assessed [18, 19]. So, how does CTC enumeration compare to changes in traditional tumor markers, CA 27–29 and CEA? To evaluate the individual and joint contribution of CTCs, the investigators compared the CTCs with the commercial tumor marker CA 27–29 in a prospective trial (Transitional Breast Cancer Research Consortium 005). The authors reported that while CTCs was an informative prognostic marker as per prior reports, CA 27–29 change from baseline to week 3–4 post-therapy was independently and more strongly associated with progression of disease (hazard ratio: 23.7, p value =0.0006), as compared to change in CTC count [20]. Thus, the additional utility of CTC enumeration, as compared to traditional tumor markers is unclear.
C) Predictive marker for therapy selection
While elevated CTC counts are associated with inferior prognosis, can CTC counts be utilized as a predictive biomarker in clinical decision making? The basic idea is to try to quickly identify effective chemotherapy based on a decline in CTC counts to move on to the subsequent lines of therapy with the hope that rapid identification of sensitivity or resistance will spare patients the toxicity of ineffective chemotherapy and potentially lead them to earlier initiation of alternate, more effective therapies. The SWOG S0500 clinical trial has evaluated the clinical utility of CTC enumeration in clinical decision making. In the large prospective randomized clinical trial, women with newly diagnosed metastatic breast cancer (N = 595) had CTC counts evaluated at baseline and week 3–4 after therapy [23]. Patients who had elevated CTCs after one cycle of chemotherapy were randomized to either change therapy to a different regimen, or to continue the same chemotherapy. Early results from SWOG S0500 reported at the 2013 San Antonio Breast Cancer Symposium, unfortunately demonstrated no significant differences in patient outcome by early change in chemotherapy versus change at the time of clinical/radiological progression [21], indicating that the finding of elevated CTCs after one cycle of chemotherapy does not have clinical utility for therapy selection.
As opposed to CTC enumeration, molecular characterization of the CTCs can potentially be helpful as a predictive biomarker for therapy selection for several reasons. First, there is evidence that metastatic tumor may not share the same molecular characteristics as the primary tumor [22, 23]. Real time CTC characterization might be a more accurate snapshot of the current biological state of the cancer, in comparison to biopsies taken at the time of diagnosis [24–28]. For example, various groups have reported detection of HER2-positive CTCs among patients with HER2-negative primary tumors [29–32]. If such tumors now respond to anti-HER2 directed therapies, the detection of HER2-positive CTCs would have therapeutic implications. In the DETECT III trial HER2-negative patients with metastatic breast cancer who have HER2-positive CTCs by immunofluorescence will be randomized to either standard therapy or standard therapy with the addition of lapatinib (ClinicalTrials.gov # NCT01619111) [33]. Another ongoing clinical trial is investigating combination of vinorelbine with trastuzumab (ClinicalTrials.gov # NCT01185509) for patients with previously HER2-negative breast cancer who are found to have HER2-positive CTCs. The key ongoing trials evaluating CTCs are summarized in Table 2.
Table 2.
Summary of the ongoing clinical trials evaluating CTCs in breast cancer
| Trial | Clinical trials.gov # | Patient Population | Study Design | Objective | Primary outcome measures |
|---|---|---|---|---|---|
| SWOGS0500 | NCT00382018 | 595 patients starting 1st line chemotherapy for MBC | Phase 3 trial - standard chemotherapy or change in chemotherapy with increased CTC count | To evaluate the strategy of changing therapy vs. maintaining therapy in patients with increased CTC counts at 1st follow-up | OS, PFS |
| DETECT III (Germany) | NCT01619111 | 228 HER2 negative MBC patients with HER2 positive CTCs | Phase III study with 2 groups - standard therapy and +/− lapatinib | To evaluate the efficacy of HER2 targeted therapy in these patients as well as the significance of CTC as an early predictive marker for treatment response | PFS |
| TREAT-CTC (Europe) | NCT01548677 | 2175 HER2 negative primary breast cancer who have detectable HER2 positive CTCs after surgery | Phase II study with 2 groups- observation or Trastuzumab for 18 weeks (6 cycles) | To evaluate the efficacy of Herceptin in treating HER2 negative primary BC with HER2 positive CTCs | Compare CTC detection rate at week 18 |
| COMETI Phase II | NCT01701050 | 200 ER+, HER2 negative, progressive MBC patients who are starting a new endocrine therapy | Biomarker trial (no therapy) | Determine a CTC- Endocrine Therapy index in ER+, HER2- MBC patients for the identification of patients that will progress rapidly | Rapid Disease Progression (Within 3 months of new line of endocrine therapy) |
CTC = Circulating Tumor Cell; PFS = Progression Free Survival; OS = Overall Survival; MBC = Metastatic Breast Cancer; ER = Estrogen Receptor; HER2 = Human Epidermal Growth Factor Receptor.
Second, molecular interrogation of CTCs could help in identification of actionable mutations and subsequent selection of therapy [34–36]. As an illustrative example, recently Schneck et al explored the possibility of identifying specific hotspot mutations from CTCs [37]. They utilized PCR based SNaPshot technology to successfully identify 12 different mutations from the enrolled patients (N=44). Similarly, other clinical trials are ongoing to rapidly characterize CTCs to detect actionable changes and personalize therapy for patients with metastatic breast cancer [38].
Finally, molecular characterization of CTCs could potentially help in evaluation of pharmacodynamic effect and clinical decision making. For example, the COMETI P2 trial is investigating the utility of a CTC-based Endocrine Therapy Index (ETI) in hormone receptor-positive breast cancer. Up to 30% of patients with metastatic hormone receptor-positive breast cancer have disease progression within 2–3 months of subsequent endocrine therapy [39]. However, currently there is no predictive biomarker that can accurately ascertain whether an individual tumor will respond to subsequent endocrine therapy or not. The purpose of the COMETI P2 trial is to evaluate an Endocrine Therapy Index, based on expression of the estrogen receptor (ER), HER2, Ki67, and Bcl-2 in the CTCs, to identify patients at risk for early progression based on specific CTC-based markers [40]. If validated, the study will establish the clinical utility of molecular interrogation of CTCs to guide clinical decision making in metastatic breast cancer.
D) Biomarker to understand mechanism of therapeutic resistance
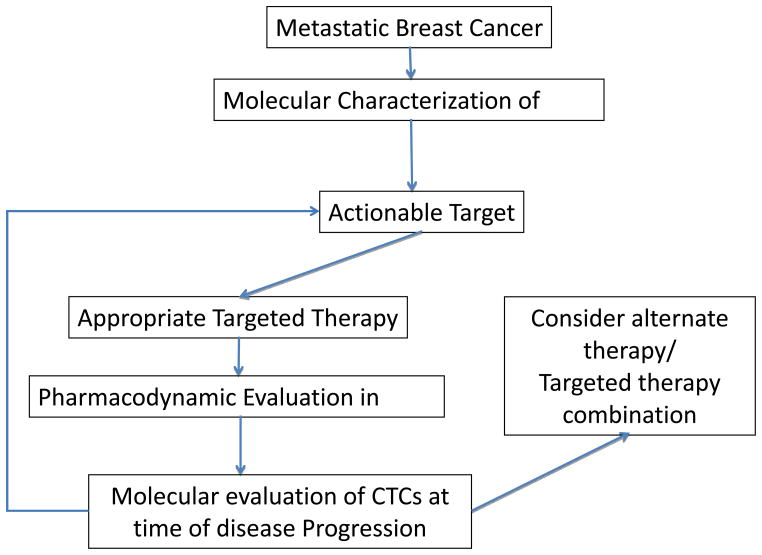
Besides therapy selection, molecular characterization of CTCs can play an important role in understanding mechanism of therapeutic resistance and in identification of novel actionable targets in breast cancer. The actionable targets identified can be evaluated and validated in clinical trials. For example, it has been demonstrated that CTCs display dynamic shifts in their molecular expression profiles between an epithelial state and a mesenchymal state in response to therapies, and the shifts correlate with a response to therapy (epithelial predominant) and progression of disease (more mesenchymal expression) [10]. Furthermore, based on RNA sequencing of CTCs, authors have identified that aberrant expression of FOXC1, besides TGF-b activation, contributes to the epithelial-mesenchymal- transition (EMT) switch and therapeutic resistance in breast cancer. The clinical significance of FOXC1 and TGF-b expression as a potential biomarker of therapeutic resistance and actionable target in breast cancer is under investigation in ongoing clinical trials. Thus molecular characterization of CTCs can potentially be utilized for selection of therapy, to monitor pharmacodynamic response, and identification of novel actionable targets in metastatic breast cancer. This is conceptually outlined in figure 1.
Figure 1.
Schema outlining how molecular characterization of CTCs can potentially be utilized for selection of therapy, to monitor pharmacodynamic response, and identification of novel actionable targets in metastatic breast cancer.
Role of CTCs in Localized Breast Cancer
A) Cancer screening
Between 20–30% of patients with no clinically detected metastasis have been observed to be positive for CTCs in several studies [24, 41, 42]. However, the low sensitivity of current CTC identification tests in early breast cancer is a significant impediment to their use as screening tools to diagnose breast cancer. Even in the metastatic setting, current CTC platforms such as the CellSearch™ system can detect CTC only in about 70% of patients with metastatic breast cancer, and in the early breast cancer setting the sensitivity is much lower. Mathematical modeling of CTC loads at various tumor stages indicate that to reduce the rate of distant metastases to 1% in operable early breast cancer would require a sensitivity improvement of approximately 15-fold from current CTC identification methods [43]. Interestingly the combination of leukapheresis and the CellSearch™ system has recently been reported to identify CTCs in 90% of patients in a small non-metastatic breast cancer cohort [44]. Thus, improvements in sensitivity using enrichment techniques broaden applicability of CTC detection as a method for early breast cancer diagnosis or screening. An ongoing clinical trial is examining the potential role of CTCs as a screening tool assessing CTC counts from patients who are undergoing a diagnostic or therapeutic breast procedure and who have no prior history of invasive carcinoma or clinically apparent metastatic disease (ClinicalTrials.gov # NCT01322750) [45].
B) Early diagnosis of Disease Recurrence
Theoretically, CTC detection, if sufficiently sensitive, could detect micrometastatic or occult metastatic disease among patients thought to have early breast cancer. This would include both patients who have early presence of CTCs without established metastases and others who have established metastases which are below the detection threshold of imaging. Studies have shown both these subtypes of patients are definitely at increased risk of progression in comparison to those who are negative for CTCs [42]. It has also been shown that CTC detection can predict the risk of axillary lymph node metastasis and could have potential utility as an adjunct to sentinel lymph node sampling [46].
The next logical question is whether or not early detection of such high risk patients, and potential escalation of treatment for them, impacts survival outcomes. While more research is required in this area, small studies have shown encouraging results about utility of molecular characterization of CTCs for subsequent therapy selection. In one small Phase II trial among patients with persistent HER2-positive CTCs detected after completion of standard adjuvant therapy the administration of trastuzumab reduced the risk of recurrence and increased disease free survival [47]. The investigators in this study specifically evaluated patients with primary HER2-negative breast cancer with detectable HER2-positive CTCs (N=75). After therapy with trastuzumab, 75% of the treated women became free of detectable CTCs while only 17.9% did so in the control observation group. Based on these results, the ongoing TREAT-CTC randomized trial will investigate the potential clinical utility of adjuvant trastuzumab among patients with HER2 negative non-metastatic breast cancer who have detectable HER2-positive CTCs [48].
Finally, recent work has also identified molecular signatures of CTCs that can preferentially form metastases in selected organs [49]. Investigators identified a specific protein signature from cultured CTC cell lines and showed the ability of these selected cells to form extensive metastases in an animal model. Further work in this area could be very important in predicting expected sites of metastases in patients based on molecular characterization of their isolated CTCs.
Limitations of current methods
Current CTC detection methods are heavily dependent on identification based on epithelial markers. Though the sensitivity of epithelial identifiers helps in efficiently picking up cancerous cells in the circulation, there are a number of situations where this aspect becomes a significant limitation. Subgroups of CTCs often undergo epithelial mesenchymal transition (EMT) which has been shown to increase the ability of these cells to survive in the circulation and establish micrometastases in peripheral tissues. EMT has been implicated in preventing cell death, protecting from the immune system and lack of response to therapy [50]. Tumor cells which have already undergone EMT reduce their expression of classical epithelial markers and express a mesenchymal profile. This consequently, leads to an inability of several detection methods to detect this highly significant and dangerous subpopulation of CTCs [51]. The choice of markers used can also make them incapable of being used for non-epithelial cancers. Newer techniques have now been developed which can detect mesenchymal CTCs as well [10].
Furthermore detection of circulating tumor DNA could potentially offer a more sensitive alternative to CTC enumeration. Recently, Dawson et al (2013) reported results of a prospective comparison of CTC counts with circulating tumor DNA in patients undergoing therapy for metastatic breast cancer (N=30) [52]. Circulating tumor DNA detects tumor specific DNA sequences from the peripheral blood, and in this study was shown to have a superior sensitivity to a CTC enumeration with the CellSearch system (sensitivity of 90% vs. 67%). However, circulating tumor DNA can only be performed in patients who have a detectable mutation, in contrast to the generalized applicability of CTCs. Circulating tumor DNA could potentially be extremely useful in the detection of specific actionable cancer mutations from a blood sample. This has been demonstrated with the identification of specific PIK3CA mutations from these DNA fragments in the blood [53].
Future Directions
CTC detection and molecular characterization is rapidly evolving towards further clinical applications, including but not limited to improving diagnosis, predicting prognosis, determining therapeutic response, and assisting therapy selection. Currently there are no standardized methods for molecular characterization and it is essential to identify methods which are sensitive, accurate, and reproducible. Further improvement in molecular characterization of low numbers of CTCs and determining tumor heterogeneity by genetic sequencing of isolated single cells are vital.
Combining two or more techniques of CTC detection may also help in improving sensitivity of detection. Further refinements in sensitivity are necessary before CTCs become useful tools for screening and diagnosis of early breast cancer. Additionally, it will be important to develop and validate characterization methods which can maintain cell viability. Captured, viable CTCs would enable real-time assessment of their molecular signatures and responses to drugs, potentially enabling clinicians to stay ‘one step ahead’ of evolving drug tolerance/resistance. It is likely that modern CTC technologies will facilitate a new age of real-time cancer monitoring and significantly advance the field of personalized breast cancer therapy.
Footnotes
Conflict of Interest
Sasmit Sarangi, Kailash Mosulpuria, Michaela J. Higgins, Aditya Bardia have nothing to disclose.
Compliance with Ethics Guidelines
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
References
• - Of Major importance
- 1.Ghossein RA, Bhattacharya S, Rosai J. Molecular detection of micrometastases and circulating tumor cells in solid tumors. Clin Cancer Res. 1999;5(8):1950–60. [PubMed] [Google Scholar]
- 2.Racila E, Euhus D, Weiss AJ, et al. Detection and characterization of carcinoma cells in the blood. Proc Natl Acad Sci U S A. 1998;95(8):4589–94. doi: 10.1073/pnas.95.8.4589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cristofanilli M, Budd GT, Ellis MJ, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004;351(8):781–91. doi: 10.1056/NEJMoa040766. [DOI] [PubMed] [Google Scholar]
- 4.Peeters DJ, De Laere B, Van den Eynden GG, et al. Semiautomated isolation and molecular characterisation of single or highly purified tumour cells from CellSearch enriched blood samples using dielectrophoretic cell sorting. Br J Cancer. 2013;108(6):1358–67. doi: 10.1038/bjc.2013.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vona G, Sabile A, Louha M, et al. Isolation by size of epithelial tumor cells : a new method for the immunomorphological and molecular characterization of circulatingtumor cells. Am J Pathol. 2000;156(1):57–63. doi: 10.1016/S0002-9440(10)64706-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aerts J, Wynendaele W, Paridaens R, et al. A real-time quantitative reverse transcriptase polymerase chain reaction (RT-PCR) to detect breast carcinoma cells in peripheral blood. Ann Oncol. 2001;12(1):39–46. doi: 10.1023/a:1008317512253. [DOI] [PubMed] [Google Scholar]
- 7.Ko Y, Klinz M, Totzke G, Gouni-Berthold I, Sachinidis A, Vetter H. Limitations of the reverse transcription-polymerase chain reaction method for the detection of carcinoembryonic antigen-positive tumor cells in peripheral blood. Clin Cancer Res. 1998;4(9):2141–6. [PubMed] [Google Scholar]
- 8.Krivacic RT, Ladanyi A, Curry DN, et al. A rare-cell detector for cancer. Proc Natl Acad Sci U S A. 2004;101(29):10501–4. doi: 10.1073/pnas.0404036101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nagrath S, Sequist LV, Maheswaran S, et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature. 2007;450(7173):1235–9. doi: 10.1038/nature06385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- •10.Yu M, Bardia A, Wittner BS, et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science. 2013;339(6119):580–4. doi: 10.1126/science.1228522. This study reports a novel method of identify mesenchymal CTCs in breast cancer and correlates them with disease progression. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hayes DF, Cristofanilli M, Budd GT, et al. Circulating tumor cells at each follow-up time point during therapy of metastatic breast cancer patients predict progression-free and overall survival. Clin Cancer Res. 2006;12(14 Pt 1):4218–24. doi: 10.1158/1078-0432.CCR-05-2821. [DOI] [PubMed] [Google Scholar]
- •12.Pierga JY, Hajage D, Bachelot T, et al. High independent prognostic and predictive value of circulating tumor cells compared with serum tumor markers in a large prospective trial in first-line chemotherapy for metastatic breast cancer patients. Ann Oncol. 2012;23(3):618–24. doi: 10.1093/annonc/mdr263. This prospective series of patients confirms the independent prognostic impact of CTCs and compares them to established tumor markers in metastatic breast cancer. [DOI] [PubMed] [Google Scholar]
- 13.Giordano A, Giuliano M, De Laurentiis M, et al. Circulating tumor cells in immunohistochemical subtypes of metastatic breast cancer: lack of prediction in HER2-positive disease treated with targeted therapy. Ann Oncol. 2012;23(5):1144–50. doi: 10.1093/annonc/mdr434. [DOI] [PubMed] [Google Scholar]
- 14.Wallwiener M, Hartkopf AD, Baccelli I, et al. The prognostic impact of circulating tumor cells in subtypes of metastatic breast cancer. Breast Cancer Res Treat. 2013;137(2):503–10. doi: 10.1007/s10549-012-2382-0. [DOI] [PubMed] [Google Scholar]
- 15.Budd GT, Cristofanilli M, Ellis MJ, et al. Circulating tumor cells versus imaging--predicting overall survival in metastatic breast cancer. Clin Cancer Res. 2006;12(21):6403–9. doi: 10.1158/1078-0432.CCR-05-1769. [DOI] [PubMed] [Google Scholar]
- 16.De Giorgi U, Valero V, Rohren E, et al. Circulating tumor cells and bone metastases as detected by FDG-PET/CT in patients with metastatic breast cancer. Ann Oncol. 2010;21(1):33–9. doi: 10.1093/annonc/mdp262. [DOI] [PubMed] [Google Scholar]
- 17.Liu MC, Shields PG, Warren RD, et al. Circulating tumor cells: a useful predictor of treatment efficacy in metastatic breast cancer. J Clin Oncol. 2009;27(31):5153–9. doi: 10.1200/JCO.2008.20.6664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hayes DF, Bast RC, Desch CE, et al. Tumor marker utility grading system: a framework to evaluate clinical utility of tumor markers. J Natl Cancer Inst. 1996;88(20):1456–66. doi: 10.1093/jnci/88.20.1456. [DOI] [PubMed] [Google Scholar]
- 19.Harris L, Fritsche H, Mennel R, et al. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol. 2007;25(33):5287–312. doi: 10.1200/JCO.2007.14.2364. [DOI] [PubMed] [Google Scholar]
- •20.Bardia A, Huang P, Zhang Z, Sokoll L, Ingle JN, Carey LA, et al. Circulating Tumor Cells (CTCs) and CA27.29 as Predictors of Outcome in Metastatic Breast Cancer (MBC) Patients in the Prospective TBCRC 005 Biomarker Study. J Clin Oncol. 2010;28(15s):suppl; abstr 1001. Clinical trial demonstrated that change in CA2729 change from baseline to week 3–4 post-therapy was independently and more strongly associated with progression of disease as compared to change in CTC count. [Google Scholar]
- •21.Smerage JB, Barlow WE, Hayes DF, et al. SWOG S0500 – A randomized phase III trial to test the strategy of changing therapy versus maintaining therapy for metastatic breast cancer patients who have elevated circulating tumor cell (CTC) levels at first follow-up assessment. Presented at San Antonio Breast Cancer Symposium; December, 2013; Clinical trial demonstrated that changing therapy based on earlier detection of elevated CTC count after therapy does not result in superior outcomes. [Google Scholar]
- 22.Lindstrom LS, Karlsson E, Wilking UM, et al. Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J Clin Oncol. 2012;30(21):2601–8. doi: 10.1200/JCO.2011.37.2482. [DOI] [PubMed] [Google Scholar]
- 23.Hoefnagel LD, van de Vijver MJ, van Slooten HJ, et al. Receptor conversion in distant breast cancer metastases. Breast Cancer Res. 2010;12(5):R75. doi: 10.1186/bcr2645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Riethdorf S, Muller V, Zhang L, et al. Detection and HER2 expression of circulating tumor cells: prospective monitoring in breast cancer patients treated in the neoadjuvant GeparQuattro trial. Clin Cancer Res. 2010;16(9):2634–45. doi: 10.1158/1078-0432.CCR-09-2042. [DOI] [PubMed] [Google Scholar]
- 25.Sieuwerts AM, Mostert B, Bolt-de Vries J, et al. mRNA and microRNA expression profiles in circulating tumor cells and primary tumors of metastatic breast cancer patients. Clin Cancer Res. 2011;17(11):3600–18. doi: 10.1158/1078-0432.CCR-11-0255. [DOI] [PubMed] [Google Scholar]
- 26.Somlo G, Lau SK, Frankel P, et al. Multiple biomarker expression on circulating tumor cells in comparison to tumor tissues from primary and metastatic sites in patients with locally advanced/inflammatory, and stage IV breast cancer, using a novel detection technology. Breast Cancer Res Treat. 2011;128(1):155–63. doi: 10.1007/s10549-011-1508-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aktas B, Muller V, Tewes M, et al. Comparison of estrogen and progesterone receptor status of circulating tumor cells and the primary tumor in metastatic breast cancer patients. Gynecol Oncol. 2011;122(2):356–60. doi: 10.1016/j.ygyno.2011.04.039. [DOI] [PubMed] [Google Scholar]
- 28.Fehm T, Hoffmann O, Aktas B, et al. Detection and characterization of circulating tumor cells in blood of primary breast cancer patients by RT-PCR and comparison to status of bone marrow disseminated cells. Breast Cancer Res. 2009;11(4):R59. doi: 10.1186/bcr2349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hayes DF, Walker TM, Singh B, et al. Monitoring expression of HER-2 on circulating epithelial cells in patients with advanced breast cancer. Int J Oncol. 2002;21(5):1111–7. doi: 10.3892/ijo.21.5.1111. [DOI] [PubMed] [Google Scholar]
- 30.Wulfing P, Borchard J, Buerger H, et al. HER2-positive circulating tumor cells indicate poor clinical outcome in stage I to III breast cancer patients. Clin Cancer Res. 2006;12(6):1715–20. doi: 10.1158/1078-0432.CCR-05-2087. [DOI] [PubMed] [Google Scholar]
- 31.Flores LM, Kindelberger DW, Ligon AH, et al. Improving the yield of circulating tumour cells facilitates molecular characterisation and recognition of discordant HER2 amplification in breast cancer. Br J Cancer. 2010;102(10):1495–502. doi: 10.1038/sj.bjc.6605676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tewes M, Aktas B, Welt A, et al. Molecular profiling and predictive value of circulating tumor cells in patients with metastatic breast cancer: an option for monitoring response to breast cancer related therapies. Breast Cancer Res Treat. 2009;115(3):581–90. doi: 10.1007/s10549-008-0143-x. [DOI] [PubMed] [Google Scholar]
- 33. [Accessed: 12/30/2013];DETECT III - A Multicenter, Phase III Study to Compare Standard Therapy +/− Lapatinib in HER2-ve MBC-Patients With HER2+ve CTCs [database on the Internet] Available from: http://clinicaltrials.gov/show/NCT01619111.
- 34.Gallant JN, Matthew EM, Cheng H, Harouaka R, Lamparella NE, Kunkel M, et al. Predicting therapy response in live tumor cells isolated with the flexible micro spring array device. Cell Cycle. 2013;12(13):2132–43. doi: 10.4161/cc.25165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hou HW, Warkiani ME, Khoo BL, et al. Isolation and retrieval of circulating tumor cells using centrifugal forces. Sci Rep. 2013;3:1259. doi: 10.1038/srep01259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ozkumur E, Shah AM, Ciciliano JC, et al. Inertial focusing for tumor antigen-dependent and -independent sorting of rare circulating tumor cells. Sci Transl Med. 2013;5(179):179ra47. doi: 10.1126/scitranslmed.3005616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- •37.Schneck H, Blassl C, Meier-Stiegen F, et al. Analysing the mutational status of PIK3CA in circulating tumor cells from metastatic breast cancer patients. Mol Oncol. 2013;7(5):976–86. doi: 10.1016/j.molonc.2013.07.007. This study reports a new method to molecularly characterize and identify actionable mutations in isolated CTCs. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. [Accessed: 12/30/2013];Characterization of Circulating Tumor Cells (CTCs) to Direct Pre-Operative and Systemic Therapy in Patients With Locally Advanced or Metastatic Stage IV Breast Cancer [database on the Internet] Available from: http://clinicaltrials.gov/show/NCT01048918.
- 39.Osborne CK, Pippen J, Jones SE, et al. Double-blind, randomized trial comparing the efficacy and tolerability of fulvestrant versus anastrozole in postmenopausal women with advanced breast cancer progressing on prior endocrine therapy: results of a North American trial. J Clin Oncol. 2002;20(16):3386–95. doi: 10.1200/JCO.2002.10.058. [DOI] [PubMed] [Google Scholar]
- 40. [Accessed: 01/16/2014];Characterization of Circulating Tumor Cells (CTC) From Patients With Metastatic Breast Cancer Using the CTC-Endocrine Therapy Index (COMETI P2) [database on the Internet] Available from: http://clinicaltrials.gov/show/NCT01701050.
- 41.Krishnamurthy S, Cristofanilli M, Singh B, et al. Detection of minimal residual disease in blood and bone marrow in early stage breast cancer. Cancer. 2010;116(14):3330–7. doi: 10.1002/cncr.25145. [DOI] [PubMed] [Google Scholar]
- 42.Lucci A, Hall CS, Lodhi AK, et al. Circulating tumour cells in non-metastatic breast cancer: a prospective study. Lancet Oncol. 2012;13(7):688–95. doi: 10.1016/S1470-2045(12)70209-7. [DOI] [PubMed] [Google Scholar]
- 43.Coumans FA, Siesling S, Terstappen LW. Detection of cancer before distant metastasis. BMC Cancer. 2013;13(1):283. doi: 10.1186/1471-2407-13-283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fischer JC, Niederacher D, Topp SA, et al. Diagnostic leukapheresis enables reliable detection of circulating tumor cells of nonmetastatic cancer patients. Proc Natl Acad Sci U S A. 2013;110(41):16580–5. doi: 10.1073/pnas.1313594110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. [Accessed: 12/30/2013];Circulating Tumor Cells (CTCs): A Potential Screening Test for Clinically Undetectable Breast Carcinoma [database on the Internet] Available from: http://clinicaltrials.gov/show/NCT01322750.
- 46.Nakagawa T, Martinez SR, Goto Y, et al. Detection of circulating tumor cells in early-stage breast cancer metastasis to axillary lymph nodes. Clin Cancer Res. 2007;13(14):4105–10. doi: 10.1158/1078-0432.CCR-07-0419. [DOI] [PubMed] [Google Scholar]
- •47.Georgoulias V, Bozionelou V, Agelaki S, et al. Trastuzumab decreases the incidence of clinical relapses in patients with early breast cancer presenting chemotherapy-resistant CK-19mRNA-positive circulating tumor cells: results of a randomized phase II study. Ann Oncol. 2012;23(7):1744–50. doi: 10.1093/annonc/mds020. This Phase-II study demostrates the ability of trastuzumab to effectively eliminate HER2+ CTCs, in patients with HER2- primary tumors. Trastuzumab treated patients had a longer DFS and reduced number of relapses. [DOI] [PubMed] [Google Scholar]
- 48. [Accessed: 12/30/2013];Efficacy Study of Herceptin to Treat HER2-negative CTC Breast Cancer (TREAT-CTC) [database on the Internet] Available from: http://clinicaltrials.gov/show/NCT01548677.
- 49.Zhang L, Ridgway LD, Wetzel MD, et al. The identification and characterization of breast cancer CTCs competent for brain metastasis. Sci Transl Med. 2013;5(180):180ra48. doi: 10.1126/scitranslmed.3005109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tiwari N, Gheldof A, Tatari M, Christofori G. EMT as the ultimate survival mechanism of cancer cells. Semin Cancer Biol. 2012;22(3):194–207. doi: 10.1016/j.semcancer.2012.02.013. [DOI] [PubMed] [Google Scholar]
- 51.Kasimir-Bauer S, Hoffmann O, Wallwiener D, Kimmig R, Fehm T. Expression of stem cell and epithelial-mesenchymal transition markers in primary breast cancer patients with circulating tumor cells. Breast Cancer Res. 2012;14(1) doi: 10.1186/bcr3099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- •52.Dawson SJ, Tsui DW, Murtaza M, et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N Engl J Med. 2013;368(13):1199–209. doi: 10.1056/NEJMoa1213261. This prospective study shows a higher sensitivity of detection of circulating tumor DNA in comparison to CTCs, in selected patients with specific mutations. [DOI] [PubMed] [Google Scholar]
- •53.Higgins MJ, Jelovac D, Barnathan E, et al. Detection of tumor PIK3CA status in metastatic breast cancer using peripheral blood. Clin Cancer Res. 2012;18(12):3462–9. doi: 10.1158/1078-0432.CCR-11-2696. This study reports a novel technique to identify specfic cancer mutations from a blood sample, by detecting cirtculating tumor DNA. [DOI] [PMC free article] [PubMed] [Google Scholar]