Abstract
• Background and Aims Previous research has made clear the intrinsic taxonomic difficulties in identifying species in the genus Orobanche. The aim of this study, therefore, was to investigate the systematic utility of seed characteristics.
• Methods Light and scanning electron microscopy was used to examine the seeds of 33 taxa of Orobanche from the Iberian Peninsula and the Balearic Islands.
• Key Results Characters such as size, shape and ornamentation of the seeds were not found to be very useful in differentiation of taxa; however, other characters of the epidermal seed coat cells proved to be very helpful in this respect. Ornamentation of the periclinal walls could be used to discriminate four morphological types. Other features related to the anticlinal walls of the cells, such as thickness, presence/absence of a narrow trough, or relative depth, all contributed to the characterization of a large number of species.
• Conclusions The usefulness of micromorphological studies on seeds of Orobanche in relation to differentiating taxa is demonstrated, and a key is provided to distinguish species or groups of species.
Key words: Orobanche, seed, seed‐coat, morphology, systematic significance, SEM
INTRODUCTION
Orobanche is a large genus mainly distributed throughout subtropical and temperate regions of the northern hemisphere, with the Mediterranean region one of the most important centres of diversity. Orobanche is a genus with approx. 100 species, 30–35 % of which are present in the Iberian Peninsula and the Balearic Islands. Thirty‐two taxa were recognized by Foley (2001), although the floristic contributions of Pujadas and Lora González (1995, 1996, 1997), Pujadas et al. (1997), Pujadas (1997, 2000), Pujadas and Gómez (2000) have indicated that there are 34 taxa (Pujadas, 2002).
The genus Orobanche is divided into two well‐delimited sections, Trionychon Wallr. and Orobanche L., traditionally recognized by authors such as Beck von Mannagetta (1930) and Chater and Webb (1972), and more recently by Foley (2001) and Pujadas (2002). The most important differential characteristics of the section Trionychon are branched stems, bracteolate flowers, an entire and campanulated calyx, a blue or purple corolla, white anthers, and the stigma usually white. This contrasts with the single stems, non‐bracteolate flowers, calyx divided into two lateral segments, corolla white, yellow, brown, amethyst or red, anthers yellow, brown‐grey or greyish, and stigma yellow, orange, red or purple of the section Orobanche (Pujadas, 2002). From a molecular point of view, according to Schneeweiss (2001), the separation of both sections is pronounced, and supports the treatment of the section Trionychon as a separate genus Phelipanche, as previously stated by Teryokhin (1991, 1997).
The Orobanchaceae is a family of holoparasitic plants, which has been traditionally considered to be closely related to the Scrophulariaceae. Some authors, like Stace (1985), have pointed out the absence of a clear separation between the families, and have highlighted the relationship between hemiparasitic and holoparasitic taxa of both families due to the continuity of their morphological features (Kuijt, 1969; Weber, 1980). Previously, Boeshore (1920) had carried out a study concerning morphological continuity between the Scrophulariaceae and Orobanchaceae based on different characters such as roots, leaves or inflorescences in order to delimit these families. Other authors such as Hutchinson (1969), Takhtajan (1980) and Cronquist (1981) consider Orobanchaceae as a different and more advanced family, probably originating from the subfamily Rhinantoideae of the Scrophulariaceae. Later work by Teryokhin (1997) considered it as a subfamily Orobanchoideae within the Scrophulariaceae. Recent molecular phylogenetic research on the Scrophulariaceae and Orobanchaceae has provided new data about their delimitation. Young et al. (1999) suggest that the traditional Orobanchaceae, the hemiparasitic Scrophulariaceae, and the Lindenbergia genus should make up the family Orobanchaceae. Finally, Olmstead et al. (2001) suggest that this complex should be subdivided into at least five different families and that the Scrophulariaceae parasites should be included within the Orobanchaceae.
The difficulty in identifying species in the genus Orobanche has been shown by the research of various authors. Among these, we can highlight the palinology carried out by Minkin (1987), Minkin and Eshbaugh (1989) and Abu Sbaih et al. (1994), and the biomolecular studies by Schneeweiss (2001), Benharrat et al. (2001), Román et al. (2003). The intrinsic taxonomic difficulties of the genus Orobanche are further increased by the fact that important differential characters are observed only with difficulty, or not at all, on dried specimens. With regard to seeds, there are several studies that analyse different aspects such as their chemical composition (Velasco et al., 2000), observations through fluorescence microscopy techniques (Joel, 1987a), or research with scanning electron microscopy (SEM; Abu Sbaih and Jury, 1994a, b; Musselman and Mann, 1976; Joel, 1987b, 1988a, b; Deif et al., 2000).
The use of SEM allows for the observation of structures which would be difficult by other means, and some authors have highlighted the importance of this technique for the study of seed‐coats, especially for those families in which identification is complicated, such as the Portulacaceae (Danin et al., 1979) and, particularly, the Scrophulariaceae (Canne, 1979; Sutton, 1988; Juanet al., 1994, 1997, 1999, 2000), where features of seed morphology have been widely used to distinguish the different taxa or to find affinities between them.
The aim of the present study was to illustrate the role of seed features in the identification of species of Orobanche in the Iberian Peninsula and the Balearic Islands as an aid to agriculture by identifying soil‐born pathogens, and to relate such characters to the systematics of the group.
MATERIALS AND METHODS
Seed morphology was studied in 33 taxa of the Orobanche L. genus from the Iberian Peninsula and the Balearic Islands. For every taxon, specimens from the populations within the study area were sampled, except for O. teucrii and O. reticulata. In these cases, it being impossible to obtain samples from Iberian populations, specimens from France (courtesy of the Herbarium at the Pirenaic Institute of Ecology, CSIC, Jaca) and from Germany, respectively, were used. For O. artemisiae‐campestris, which is present in the Iberian Peninsula, it proved impossible to find seeds for analysis.
The study was based on dry herbarium specimens from the Department of Sciences and Agronomy and Forestry Resources of the University of Córdoba (COA), although some of the samples used came from specimens provided by the herbaria of the Aranzadi Science Society in San Sebastián (ARAN) (O. caryophyllaceae), by the Department of Science and Natural Resources at the University of Alicante (ABH) (O. olbiensis) and by the Pirenaic Institute of Ecology (JACA) (O. teucrii). Collectors and localities are shown in the Appendix.
Data on the width and length of seeds is based on the measurement of 30–150 seeds per taxon, by means of direct observation under optical microscopy.
For scanning electron microscopy, dry seeds were mounted directly on stubs using double‐sided adhesive tape and coated with gold/palladium in a sputter coater. Morphological observations were made using a Philips XL‐30 microscope of the Electronic Microscopy Service of the University of Seville.
The terminology of seed‐coat surface sculpturing basically follows Stearn (1992) and Font Quer (1993). The abbreviations of the authorities of plant names follow the standard of Brummit and Powell (1992), and acronyms of the herbariums follow Holmgren et al. (1990).
RESULTS
Seeds of Orobanche are extremely small, being less than 1 mm in size, with a wide variety of shapes (ellipsoid, oblongoid, ovoid, globose, trigonous or tetragonous) and a terminal funicular attachment. The seed coat is reticulated with polygonal cells, which range from more or less isodiametric to tangentially elongated, being sometimes irregular. Orobanche seeds usually have a smooth membranous outer periclinal wall, although they can sometimes have a fibrillar aspect due to epicuticular waxes. Both kinds collapse on the inner periclinal wall, thus showing the structure of the latter, which is of very changeable aspect (perforate, granulate, rugulose, etc.).
Observations made using scanning electron microscopy indicated that there are four morphological seed types, recognizable as listed in Table 1.
Table 1.
Four morphological seed types of Orobanche as indicated by observations made using scanning electron microscopy
| 1. | Epidermal cells with fibrillar outer periclinal wall | Type IV |
| 1′. | Epidermal cells with smooth outer periclinal wall | 2 |
| 2. | Outer periclinal walls impede vision of the inner one | Type I |
| 2′. | Outer periclinal walls allow vision of the inner one | 3 |
| 3. | Inner periclinal wall perforate | Type II |
| 3′. | Inner periclinal wall granulate or rugulose | Type III |
Type I
Seeds 0·28–0·47 × 0·17–0·30 mm, ellipsoid to ovoid shape; very dark brown. Seed‐coat surface of isodiametric cells, 0·06–0·11 × 0·04–0·06 mm; anticlinal walls with remarkably deep thickening at the vertex, and with a narrow trough marking the wall junctures between cells; outer periclinal walls smooth, impeding vision of the inner one (Fig. 1A–C).
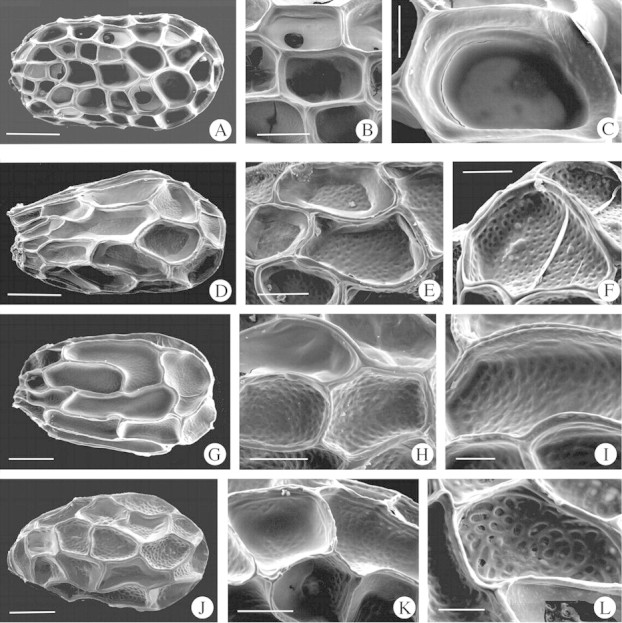
Fig. 1. Scanning electron micrographs of Orobanche seeds. (A–C) Type I, O. rapum genistea. (D–L) Type II. (D–G) Subtype IIA: (D) O. alba, (E) O. reticulata subsp. reticulata, (F) O. minor, (G) O. calendulae. (H, I) Subtype IIB: (H) O. elatior subsp. icterica, (I) O. amethystea. (J–L) Subtype IIC: (J) O. laserpitii‐sileris, (K, L) O. clausonis. Scale bars = 100 µm (A, D, G, J); 50 µm (B, E, H, K); 20 µm (C, F, I, L).
Among the taxa examined, only seeds of O. rapum‐genistae have this seed type.
Type II
Seeds 0·27–0·57 × 0·14–0·41 mm, ovoid, oblongoid, ellipsoid, trigonous, tetragonous or subglobose; dark brown to greyish. Epidermal seed‐coat of polygonal ± isodiametric cells or tangentially elongated 0·06–0·2 × 0·04–0·10 mm, with anticlinal walls from slightly to remarkably deep, evenly or unevenly thickened, and with a narrow trough marking the wall junctures between cells, sometimes just evident at the vertex; inner periclinal wall perforate (Fig. 1D–L; Table 2).
Table 2.
Relation of characters studied for species included in Type II (IIA, IIB, IIC)
| Seed subtype | Taxon | Seed shape | Seed size (mm) | Cell shape | Depth of anticlinal walls | Thickness of anticlinal walls | Narrow trough |
| IIA | O. alba | Oblongoid to ellipsoid | 0.29–0.46 × 0.17–0.28 | Isodiametric to tangentially elongated | Medium (Fig. 1D) | Even | Evident all around the edge |
| O. ballotae | Ovoid | 0.29–0.43 × 0.19–0.29 | Isodiametric | Medium | Even | More evident at the vertices | |
| O. calendulae | Ovoid | 0.30–0.41 × 0.18–0.27 | Isodiametric | Medium (Fig. 1G) | Even | More evident at the vertices | |
| O. crenata | Ellipsoid | 0.28–0.48 × 0.19–0.30 | Isodiametric | Medium | Even | Evident all around the edge | |
| O. minor | Oblongoid to ellipsoid | 0.27–0.36 × 0.15–0.24 | Isodiametric to tangentially elongated | Medium (Fig. 1F) | Even | Evident all around the edge | |
| O. reticulata subsp. reticulata | Ellipsoid to ovoid | 0.32–0.48 × 0.18–0.31 | Isodiametric | Medium (Fig. 1E) | Even | More evident at the vertices | |
| IIB | O. amethystea | Ovoid to ellipsoid | 0.27–0.43 × 0.17–0.28 | Isodiametric | Medium (Fig. 1I) | Even | More evident at the vertices |
| O. caryophyllaceae | Ovoid to ellipsoid | 0.25–0.47 × 0.16–0.30 | Isodiametric | Medium | Even | More evident at the vertices | |
| O. cernua | Oblongoid to ovoid | 0.30–0.50 × 0.17–0.24 | Isodiametric to irregular | Medium | Even | Evident all around the edge | |
| O. crinita | Ovoid | 0.24–0.40 × 0.14–0.23 | Tangentially elongated | Medium | Even | More evident at the vertices | |
| O. elatior subsp. icterica | Ellipsoid to ovoid | 0.30–0.45 × 0.20–0.27 | Isodiametric | Medium (Fig. 1H) | Even | More evident at the vertices | |
| O. foetida | Ellipsoid to ovoid | 0.27–0.39 × 0.14–0.23 | Isodiametric to tangentially elongated, irregular | Medium | Even | Evident all around the edge | |
| O. gracilis | Subglobose to ovoid | 0.31–0.49 × 0.17–0.31 | Isodiametric | Remarkable | Even | More evident at the vertices | |
| O. hederae | Ovoid – oblongoid to ellipsoid | 0.33–0.48 × 0.20–0.31 | Isodiametric to tangentially elongated | Medium to remarkable | Even | Evident all around the edge | |
| O. montserratii | Ovoid to subglobose | 0.31–0.52 × 0.20–0.40 | Isodiametric | Remarkable | Even | Evident all around the edge | |
| O. teucrii | Ellipsoid to subglobose | 0.33–0.47 × 0.22–0.30 | Isodiametric | Medium | Even | Evident all around the edge | |
| IIC | O. clausonis | Ellipsoid to ovoid | 0.30–0.50 × 0.16–0.28 | Isodiametric | Remarkable (Figs. 1K, L) | Uneven | More evident at the vertices |
| O. haenseleri | Trigonous or tetragonous | 0.29–0.57 × 0.18–0.41 | Isodiametric | Medium to slight | Even | More evident at the vertices | |
| O. laserpitii‐sileris | Ovoid to oblongoid | 0.25–0.46 × 0.16–0.29 | Isodiametric | Medium (Fig 1J) | Even | More evident at the vertices |
The differences observed in the diameter of the perforations of the inner periclinal wall suggest separation into three subtypes, as shown in Table 3.
Table 3.
Three subtypes of Type II seeds as indicated by differences observed in the diameter of the perforations of the inner periclinal wall
| 1. | Inner periclinal walls of epidermal seed coat cells with perforations smaller in diameter than the thickness of the walls that separate them; a small central mammilla sometimes present on cells | Subtype IIA |
| 1′. | Inner periclinal walls of epidermal seed coat cells with perforations similar in diameter or superior to the thickness of the walls that separate them; small central mammilla absent on cells | 2 |
| 2. | Inner periclinal walls of epidermal seed coat cells with perforations similar in diameter to the thickness of the walls that separate them | Subtype IIB |
| 2′. | Inner periclinal walls of epidermal seed coat cells with perforations larger in diameter than the thickness of the walls that separate them | Subtype IIC |
Subtype IIA.
Epidermal seed coat cells more or less isodiametric to tangentially elongated, anticlinal walls of medium depth and evenly thickened, with a narrow trough that is noticeable all around the edge of the cells or just at the vertices. Inner periclinal wall of epidermal seed coat cells with perforations of smaller diameter than the thickness of the walls that separate them, sometimes with a small central mammilla. This subtype includes O. alba, O. ballotae, O. calendulae, O. crenata, O. minor and O. reticulata subsp. reticulata.
Subtype IIB.
Epidermal seed coat cells more or less isodiametric to tangentially elongated, anticlinal walls very deep or of medium depth and evenly thickened with a narrow trough that is evident all around the edge of the cells or just at the vertices. Inner periclinal wall of epidermal seed coat cells with perforations of similar diameter to the thickness of the walls that separate them. This subtype includes O. amethystea, O. caryohyllaceae, O. cernua, O. crinita, O. elatior subsp. icterica, O. foetida, O. gracilis, O. hederae, O. montserratii and O. teucrii.
Subtype IIC.
Epidermal seed coat cells more or less isodiametric, slightly to very deep and anticlinal walls evenly or unevenly thickened, with a narrow trough that is noticeable just at the vertices of the cells. Inner periclinal wall of seed coat cells with perforations of larger diameter than the thickness of the walls that separate them. This subtype includes O. clausonis, O. haenseleri and O. laserpitii‐sileris.
Type III
Seeds 0·25–0·50 × 0·16–0·30 mm, ovoid, oblongoid or ellipsoid, dark brown. Epidermal seed coat made up of polygonal, more or less isodiametric, tangentially elongated, or irregular cells 0·07–0·20 × 0·03–0·07 mm; anticlinal walls medium to very deep and evenly thickened, with a narrow trough marking the wall junctures between cells, noticeable all around the edge of the cells, inner periclinal walls granulate or rugulose (Fig. 2A–C, Table 4). This type includes O. cumana, O. densiflora and O. santolinae.
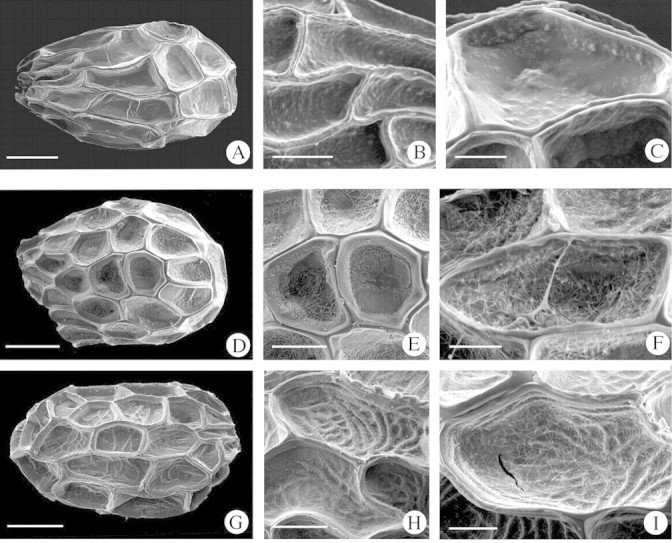
Fig. 2. Scanning electron micrographs of Orobanche seeds. (A–C) Type III: (A) O. densiflora, (B) O. cumana, (C) O. santolinae. (D–I) Type IV. (D–F) Subtype IVA: (D, E) O. latisquama, (F) O. ramosa. (G–I) Subtype IVB: (G) O. purpurea, (H) O. schultzii, (I) O. lavandulacea. Scale bars = 100 µm (A, D, G); 50 µm (B, E, H); 20 µm (C, F, I).
Table 4.
Relation of characters studied for species included in Types III and IV (IVA, IVB)
| Seed type or subtype | Taxon | Seed shape | Seed size (mm) | Cell shape | Depth of anticlinal walls | Thickness of anticlinal walls | Narrow trough |
| III | O. cumana | Oblongoid | 0·36–0·50 × 0·16–0·25 | Tangentially elongated or irregular | Medium (Fig. 2B) | Even | Evident all around the edge |
| O. densiflora | Ovoid to ellipsoid | 0·27–0·43 × 0·17–0·27 | Isodiametric to tangentially elongated | Medium (Fig. 2A) | Even | Evident all around the edge | |
| O. santolinae | Ellipsoid to ovoid | 0·26–0·48 × 0·16–0·29 | Isodiametric to irregular | Remarkable (Fig. 2C) | Even | Evident all around the edge | |
| IVA | O. arenaria | Ovoid to ellipsoid | 0·35–0·58 × 0·23–0·38 | Isodiametric | Medium | Even | More evident at the vertices |
| O. latisquama | Ellipsoid to ovoid | 0·30–0·45 × 0·18–0·28 | Isodiametric | Slight (Figs. 2D, E) | Even | Evident all around the edge | |
| O. nana | Ovoid to ellipsoid | 0·29–0·51 × 0·17–0·30 | Isodiametric to irregular | Medium | Even | More evident at the vertices | |
| O. ramosa | Ellipsoid to ovoid | 0·33–0·52 × 0·19–0·33 | Isodiametric | Medium (Fig. 2F) | Uneven | More evident at the vertices | |
| IVB | O. lavandulacea | Ovoid to oblongoid | 0·32–0·52 × 0·18–0·38 | Isodiametric | Remarkable (Fig. 2I) | Uneven | More evident at the vertices |
| O. mutelii | Ovoid | 0·35–0·53 × 0·21–0·35 | Isodiametric to irregular | Remarkable | Even | More evident at the vertices | |
| O. olbiensis | Ellipsoid to ovoid | 0·28–0·61 × 0·17–0·41 | Isodiametric | Slight | Uneven | More evident at the vertices | |
| O. purpurea | Ellipsoid to ovoid | 0·36–0·48 × 0·19–0·31 | Isodiametric | Medium (Fig. 2G) | Even | More evident at the vertices | |
| O. schultzii | Ellipsoid to ovoid | 0·39–0·57 × 0·26–0·38 | Isodiametric to tangentially elongated | Remarkable (Fig. 2H) | Uneven | More evident at the vertices | |
| O. tunetana | Ellipsoid to globose | 0·28–0·37 × 0·22–0·30 | Isodiametric | Medium | Even | More evident at the vertices |
Type IV
Seeds 0·28–0·61 × 0·17–0·41 mm, ovoid, oblongoid, globose to ellipsoid, light or dark grey‐brown. Epidermal seed coat made up of cells that are polygonal, more or less isodiametric to tangentially elongated, or irregular cells 0·07–0·15 × 0·03–0·08 mm; anticlinal walls slightly to very deep, evenly or unevenly thickened, with a narrow trough marking the wall junctures between cells more evident at the vertices; outer periclinal walls fibrillar, through which the reticulate inner ones can sometimes be seen (Fig. 2D–I, Table 4).
The appearance of periclinal walls suggests separation into two subtypes:
Subtype IVA: inner periclinal walls hidden by the outer.
Subtype IVB: reticulate inner periclinal wall visible through the outer.
Subtype IVA.
Seed shape from ellipsoid to ovoid. Epidermal seed coat cells with inner periclinal wall hidden by fibrillar outer periclinal wall; anticlinal walls of slight or medium depth. This subtype includes O. arenaria, O. latisquama, O. nana and O. ramosa.
Subtype IVB.
Seeds oblongoid, ellipsoid, ovoid to globose. Epidermal seed coat with reticulate inner periclinal wall visible through the fibrillar outer periclinal wall; anticlinal walls slightly to very deep. This subtype includes O. lavandulaceae, O. mutelii, O. olbiensis, O. purpurea, O. schultzii and O. tunetana.
DISCUSSION
The results obtained confirm the usefulness of seed characters for the identification of most of the species studied, and highlight the division of the genus into two sections (Trionychon and Orobanche), as has been traditionally recognized (Beck von Mannagetta, 1930; Chater and Webb, 1972; Foley, 2001; Pujadas, 2002).
Within each taxon, the size of the seeds is very variable. This is the reason why this character is not very useful for distinguishing groups of species below the category section, such as the ‘grex’ established by Beck von Mannagetta (1930). However, at species level, seed size helps to identify some taxa such as O. reticulata, O minor, O. nana and O. arenaria. Seed shape is also a variable character, ranging from oblongoid, ellipsoid to ovoid, although they are sometimes globose or subglobose. This character is related to its relative position in the capsule, as in the case of the Scrophulariaceae (Juan et al., 2000). The most extreme case is O. haenseleri, whose seeds allow the separation of this species from the rest of the taxa under study.
As far as ornamentation is concerned, our results agree with other authors (Ungurean, 1986; Joel, 1987a, b; Abu Sbaih and Jury, 1994a, b), showing that the genus Orobanche has reticulate seeds, although Teryokhin (1997) described them as alveolate due to invagination of the thin outer wall of the epidermal seed coat cells. The periclinal walls of the epidermal seed coat cells are of particular taxonomic interest, especially the inner wall, which remains intact since it is lignified. However, Abu Sbaih and Jury (1994a) pointed out that this wall is perforate‐reticulate in all the species that they studied. Therefore, they considered the outer wall to be of greater interest as it allowed them to distinguish four groups of species.
In the present study, the epidermal seed coat cells permitted the discrimination of four morphological types, based on observations of both inner and outer periclinal walls. Among the four established types, Type IV corresponds to the section Trionychon, except for O. latisquama, whereas Types I, II and III are included in section Orobanche. The differences found in the periclinal walls of the epidermal cells support the morphological differentiation of the taxa of both sections, thus corroborating the taxonomic separation that Beck von Mannagetta (1890, 1930) established and which has been noted by other authors, including Polo (1988), Valdés and Díez (1990), and Abu Sbaih et al. (1994) based on pollen data; Andary (1994) in a chemotaxonomical study; and Velasco et al. (2000) in their study concerning fatty acid and tocochromanol compositions of seeds. However, it has not been possible to find any correlation between the types or subtypes established in the present study and the sections or groups (grex) based on the vegetative characters of the different taxa established by Beck von Mannagetta (1930), or on the results of the recent molecular analyses by Schneeweiss (2001), Benharrat et al. (2001) and Román et al. (2001, 2003).
Type I, which includes O. rapum‐genistae (section Orobanche), is clearly separate from the other three established types because the epidermal cells present a smooth outer periclinal wall, which impedes vision of its inner one. Previously, Andary (1994) had already found morphological differences in the seeds of this taxon when comparing them with those of other species in the section Orobanche. In a phylogenetic study carried out on 20 Andalusian Orobanche taxa belonging to both sections, Román et al. (2003) showed this species (O. rapum‐genistae) as the most different (with a distance of 0·864). Its genetic and morphological separation in relation to the rest of the species of the section Orobanche is therefore obvious.
Type II (section Orobanche), which shows the epidermal seed coat cells with perforate inner periclinal wall that are visible through the smooth outer periclinal wall, includes a greater number of species. Abu Sbaih and Jury (1994a) observed that in four species of the section Orobanche (O. alba, O. cernua, O. crenata and O. minor), which were also analysed in our study, the outer wall is perforate. Moreover, it has been observed that within this group the diameter of such perforations on the inner wall vary amongst species, thus allowing for the distinction of three separate subgroups. This character has previously been used in species of the section Trionychon by Ungurean (1986), who distinguished two groups: one with numerous and small perforations, and the other with scarce, large ones. We agree with this author that the thickening of the inner wall gives seeds their reticulate aspect.
In Type III only three species of the section Orobanche are included, in which the outer periclinal wall shows a granulated‐rugulose inner wall. This pattern has not previously been observed by other authors (Ungurean, 1986; Joel, 1987a, b; Abu Sbaih and Jury, 1994a, b). Even through they show the same seed type, these species (O. cumana, O. densiflora and O. santolinae) are different from a genetic point of view, as shown in the dendrogram reported by Román et al. (2003) in which these species are not clustered together, despite two of them belonging to same grex (grex Minores Beck).
Type IV, as has been pointed out above, includes all species belonging to the section Trionychon except for O. latisquama, which is found in the section Orobanche grex Galeatae Beck. The separation of O. latisquama from the section Orobanche has been supported by several authors, some of whom have even placed it under a different genus. Thus, Schultz (1848) includes it in Boulardia under the name of B. latisquama, whereas others such as Cosson (1848) and Lange (1864) consider it to be within the genus Ceratocalyx as C. macrolepis and C. fibriata, respectively. More recently, Schneeweiss (2001), despite the fact that he includes it once again within the genus Orobanche as O. macrolepis, regards it as a complex taxon that remains in a middle position between the rest of the taxa in the genus Orobanche (section Trionychon) and the genus Cistanche, thus constituting a separate clade.
This type shows a fibrillar periclinal outer wall, as ‘group A’ recognized by Abu Sbaih and Jury (1994a), who also include within it species of the section Trionychon. Previously, some authors such as Teryokhin (1991) have included certain species of the Orobanche genus (section Trionychon) in the genus Phelipanche, the presence of fibrillar outer periclinal walls of epidermal seed coat cells being one of distinctive characters. Results subsequently obtained in the molecular study carried out by Schneeweiss (2001) support this separation. However, in the present work it has been shown that O. latisquama (section Orobanche) features a fibrillar outer periclinal wall, a character more compatible with the section Trionychon or genus Phelipanches. Besides, a recent phylogenetic study points out that O. clausonis, another species of the section Orobanche, is closer to the taxa of the section Trionychon than to those of the section Orobanche (Román et al., 2003). In conclusion, it is clear that there are still several issues to solve.
Regarding the two subgroups, IVA and IVB, established on the basis of the visibility of the inner wall of the epidermal cells, these differ from subgroups obtained by other means of analysis (Velasco et al., 2000; Schneeweiss, 2001; Román et al., 2003) and from the groups proposed by Novopokrovsky and Tzvelev (2000): Holocladas Novopokr. and Pleioclada Novopokr. (depending on whether they present a simple or a ramificated stem, respectively).
In spite of the homogeneity of the seeds in this genus, pointed out by Abu Sbaih and Jury (1994a, b), our results have enabled us to produced a key (Table 5) in a similar manner to the one designed by Joel (1988a) for six species from Israel. In our key, most taxa have been separated except for six cases where two or three species have been grouped together (O. foetida and O. hederae; O. amethystea and O. caryophyllaceae; O. tunetana and O. purpurea; O. lavandulaceae and O. schultzii; O. ballotae, O. calendulae and O. reticulata; O. cernua, O. elatior and O. teucrii), even though they are taxa that are not close phylogenetically.
Table 5.
Key to Iberian Peninsula and the Balearic Islands species of Orobanche based on seed features
| 1. | Epidermal seed coat cells with smooth outer periclinal walls | 2 |
| 1′. | Epidermal seed coat cells with fibrillar outer periclinal walls | 20 |
| 2. | Outer periclinal walls impede vision of inner ones; anticlinal walls thickening at the vertices | O. rapum‐genisteae |
| 2′. | Outer periclinal walls allow vision of inner ones; anticlinal walls of even or uneven thickness | 3 |
| 3. | Epidermal seed coat cells with perforate inner periclinal wall | 4 |
| 3′. | Epidermal seed coat cells with rugulose or granulate inner periclinal wall | 18 |
| 4. | Cells of inner periclinal wall with a small central mammilla | 5 |
| 4′. | Cells of inner periclinal wall without a central mammilla | 6 |
| 5. | Anticlinal walls with a narrow trough that is more evident at the vertices; seeds 0·32–0·48 mm | O. reticulata subsp. reticulata |
| 5′. | Anticlinal walls with a narrow trough that is visible all around the edge; seeds 0·27–0·36 mm | O. minor |
| 6. | Inner periclinal wall of epidermal seed coat cells with perforations of smaller diameter than the thickness of the walls that separate them | 7 |
| 6′. | Inner periclinal wall of epidermal seed coat cells with perforations of diameter similar to or greater than the thickness of the walls that separate them | 9 |
| 7. | Epidermal seed coat cells in the mid and insertion areas tangentially elongated and isodiametric at the opposite end | O. alba |
| 7′. | Epidermal seed coat cells isodiametric on the entire surface | 8 |
| 8. | Anticlinal wall junctures with a narrow trough that is visible all around the edge of the cells | O. crenata |
| 8′. | Anticlinal wall junctures with a narrow trough that is evident at the vertices of the cells | O. ballotae, O. calendulae,O. reticulata subsp. reticulata |
| 9. | Perforations with a similar diameter to the thickness of the wall that separates them | 10 |
| 9′. | Perforations with a diameter which is larger than the thickness of the separating wall | 16 |
| 10. | Epidermal seed coat cells with anticlinal walls of remarkable depth | 11 |
| 10′. | Epidermal seed coat cells with anticlinal walls of medium depth | 13 |
| 11. | Anticlinal wall junctures with a narrow trough that is evident at the vertices of the cells | O. gracilis |
| 11′. | Anticlinal wall junctures with a narrow trough that is visible all around the edge of the cells | 12 |
| 12. | Tangentially elongated epidermal seed coat cells, whose size decreases at the area of insertion; seeds ovoid to oblong | O. hederae |
| 12′. | Isodiametric epidermal seed coat cells on the entire surface; seeds ovoid to subglobose | O. montserratii |
| 13. | Anticlinal wall junctures with a narrow trough that is visible all around the edge of the cells | 14 |
| 13′. | Anticlinal wall junctures with a narrow trough that is evident at the vertices of the cells | 15 |
| 14. | Epidermal seed coat cells tangentially elongated which and narrow and decrease at the area of insertion | O. foetida, O. hederae |
| 14′. | Epidermal seed coat cells more or less isodiametric or irregular | O. cernua, O. elatior subsp. icterica, O. teucrii |
| 15. | Epidermal seed coat cells tangentially elongated | O. crinita |
| 15′. | Epidermal seed coat cells more or less isodiametric | O. amethystea, O. caryophyllaceae |
| 16. | Epidermal seed coat cells with anticlinal walls of remarkable depth and unevenly thickened | O. clausonis |
| 16′. | Epidermal seed coat cells with anticlinal walls of slight or medium depth and evenly thickened | 17 |
| 17. | Seeds trigonous to tetragonal | O. haenseleris |
| 17′. | Seeds ovoid to oblongoid | O. laserpitii‐sileris |
| 18. | Epidermal seed coat cells with anticlinal walls of remarkable depth | O. santolinae |
| 18′. | Epidermal seed coat cells with anticlinal walls of medium depth | 19 |
| 19. | Epidermal seed coat cells tangentially elongated or irregular; seeds oblongoid | O. cumana |
| 19′. | Epidermal seed coat cells more or less isodiametric at the vertices, tangentially elongated over the rest of the surface; seeds ovoid to ellipsoid | O. densiflora |
| 20. | Inner periclinal wall hidden by the outer one | 21 |
| 20′. | Reticulate inner periclinal wall visible through the outer one | 24 |
| 21. | Epidermal seed coat cells with anticlinal walls of slight depth, and a narrow trough that is visible all around the edge of the cells | O. latisquama |
| 21′. | Epidermal seed coat cells with anticlinal walls of medium depth and a narrow trough that is evident just at the vertices | 22 |
| 22. | Epidermal seed coat cells with anticlinal walls unevenly thickened | O. ramosa |
| 22′. | Epidermal seed coat cells with anticlinal walls evenly thickened | 23 |
| 23. | Seeds <0·40 mm; epidermal seed coat cells isodiametric to irregular | O. nana |
| 23′. | Seeds ≥0·40 mm; epidermal seed coat cells isodiametric | O. arenaria |
| 24. | Epidermal seed coat cells with anticlinal walls of slight depth; seeds light grey‐brown | O. olbiensis |
| 24′. | Epidermal seed coat cells with anticlinal walls of medium or remarkable depth; seeds dark grey‐brown | 25 |
| 25. | Epidermal seed coat cells with anticlinal walls of medium depth | O. tunetana, O. purpurea |
| 25′. | Epidermal seed coat cells with anticlinal walls of remarkable depth | 26 |
| 26. | Anticlinal walls evenly thickened | O. mutelii |
| 26′. | Anticlinal walls unevenly thickened | O. lavandulaceae, O. schultzii |
Some of the features of the Orobanche seeds, such as the terminal insertion and the reticulate ornamentation, show the similarity that exists between this genus and the hemiparasite species of Scrophulariaceae (Chuang and Heckard, 1972; Musselman and Mann, 1976; Canne, 1980; Juan et al., 1998), with which the Orobanchaceae family has some morphological characters in common (see above).
In addition, the results of the present study highlight, to some extent, the importance of the morphology of seeds in relation to dispersal or to the establishment of new plants. Thus, due to their small size, Orobanche seeds can be regarded as ‘dust‐seeds’ that are mainly dispersed by wind, and which are frequent in other families with parasitic or hemiparasitic representatives, such as Orchidaceae (van der Pijl, 1982). According to Musselman and Mann (1976), the size of the seeds could be the result of an adaptation of these plants in an attempt to be dispersed through vegetation so as to be as close as possible to the host plant. Similarly, according to Stace (1985), the high production of seeds per capsule is an adaptation of this type of plant aimed at increasing the probability of finding an appropriate host. The membranous outer periclinal wall of the epidermic cells would favour water absorption, thus enhancing the adherence and germination capacity, as in some examples of the family Lamiaceae (Ryding, 1992).
ACKNOWLEDGEMENTS
We thank the Herbaria that provided samples for the research: the Aranzadi Science Society in San Sebastián (ARAN), the Department of Science and Natural Resources at the University of Alicante (ABH) and the Pirenean Institute of Ecology (JACA). We would also like to acknowledge the help of the Electron Microscopy Unit at the University of Seville, particularly Dña. A. Fernández and Dña. A. Tejada from the Department of Sciences and Agronomy and Forestry Resources, and D. Alberto Herrero of the Royal Botanic Garden of Madrid.
APPENDIX
Localities and dates of collections studied.
Orobanche alba Stephan ex Willd. SPAIN. Almería, Peña Lobera, Sierra Lúcar, 15 Jun. 1994, Pujadas and Pallarés (COA 17323). Granada, Sierra Nevada, Nigüelas, Pista barranco de los Alisos, 22 Jul.1997, Prados, Parra and Jiménez (COA 17635). Jaén, Sierra Mágina, El Almadén, 13 Jun. 1997, Pujadas (COA 23546). Jaén, Sierra Mágina, Torres, base de Monteagudo, 4 Jul. 1998, Pujadas and Plaza (COA 29897).
Orobanche amethystea Thuill. SPAIN. Alicante, Cap de les Hortes, 17 Apr.1997, Pujadas and Aguilella (COA 23735). Almería, Cóbdar, Cerro Pinar, 25 May 1994, Pujadas (COA 16475). Burgos, Villabáscones, 26 Jul.1991, Pujadas and García Salmones (COA 17608). Cádiz, Jerez de la Frontera, 11 May 1988, Pujadas and Oliveira (COA 13539). Girona, El Port de la Selva, naer Tamariua beach, 15 May 1998, Pujadas (COA 30473).
Orobanche arenaria Borkh. SPAIN. Almería, Sierra de las Estancias, Puerto Santa María de Nieva, 17 Jun. 2000, Pujadas (COA 29507). Granada, Puebla from Don Fadrique to Santiago de la Espada, Km 6, 19 Jul. 1999, Pujadas (COA 28906). Granada, Km 4 N from Trevélez, near Río Trevélez, 19 Jul. 2000, Pujadas and López (COA 30477). Murcia, Sierra Espuña, Pico Espuña, 17 Jun. 2000, Pujadas (COA 29508).
Orobanche ballotae A. Pujadas SPAIN. Almería, Agua Amarga, 6 May 1994, Pujadas (COA 17427). Almería, Níjar, N del Barranco Sabinar, 19 Apr. 1994, Pujadas and Jiménez (COA 17447). Almería, Níjar, Cerro de Los Frailes, 2 May 1994, Pujadas (COA 17446). Jaén, Los Villares, Los Cañones, 12 Jun. 1991, Pujadas and Lora (COA 17618).
Orobanche calendulae Pomel SPAIN. Ciudad Real, Piedrabuena, 10 May 1992, Carrasco, Garrido and Blanco (COA 22062). Toledo, Borrox, 24 May 1995, Pujadas (COA 30527).
Orobanche caryophyllaceae Sm. SPAIN. Álava, Peñalta, Arán, 10 Jun. 1983. Huesca, La Muria, Bisaurri, Ribagorza, 1 Jul. 2001, Pujadas (COA 31075).
Orobanche cernua L. SPAIN. Almería, Cerro Santa Fe, near Cerro Los Carneros, SE de la Mina Santa Bárbara, 23 Mar. 1994, Pujadas (COA 17355). Jaén, Bélmez de la Moraleda, Sierra de la Cruz, 3 May 1998, Pujadas (COA 25297). Granada, Cullar‐Baza, El Margen, Arroyo El Margen, 08 Jun. 2000, Pujadas (COA 29902).
Orobanche clausonis Pomel SPAIN. Córdoba, Km 5 from Rute to Carcabuey, 15 Jun. 1995, Pérez (COA 17591). Granada, Sierra Parapanda, 28 Jun. 2000, Pujadas, Plaza and Román (COA 29903). Murcia, Sierra Espuña, Río Espuña, 15 Apr. 1997, Peláez (COA 23732).
Orobanche crenata Forssk. SPAIN. Cádiz, near Grazalema, 20 Apr. 1995, Pujadas (COA 17507). Córdoba, Km 24 Puente Genil – Écija, 17 Jun.1987, Hernández et al. (COA 13682). Granada, Trevélez, near Río Trevélez, 30 Jun. 1990, Pujadas and Poyato (COA 13583). Málaga, near Guaro, 24 Jun.1992, Lora, Prados and Pujadas (COA 17519).
Orobanche crinita Viv. SPAIN. Baleares, Menorca, Sant Lluis, Biniancolla, 13 Agu. 2000, Fraga, Poyato and Pujadas (COA 29572). Menorca, ES Castell, 13 Apr. 2001, Fraga and Pujadas (COA 31046).
Orobanche cumana Wallr. SPAIN. Córdoba, Huerta San Luis, 2 Jul. 1996, Lora and Pujadas (COA 22077). Córdoba, Cortijo del Partido, 2 Jul. 1997, Pujadas (COA 23745). Málaga, near Laguna de la Fuente de Piedra, 8 Jun.1995, Lora and Pujadas (COA 17628). Sevilla, Km 3 from Ecija to Estepa, 1 Jul. 1996, Pujadas, Lora and Jiménez (COA 22079). Sevilla, El Coronil, 16 Jun. 1986, García (COA 13636).
Orobanche densiflora Reut. SPAIN. Cádiz, Rota, Punta Candor, 26 Apr. 1998, Pujadas (COA 25299). Cádiz, Chiclana, Sancti‐Petri, 14 May 2000, Pujadas (COA 29912). Cádiz, Barbate beach, 15 Jun. 2000, Sánchez (COA 29586). Cádiz, Tarifa, Bolonia, 15 Jun. 2000, Plaza. (COA 29881).
Orobanche elatior Sutton subsp. icterica (Pau) A. Pujadas SPAIN. Alicante, Palomaret‐Agrost, 17 May 1996, Pujadas (COA 29595). Alicante, Castalla, Rambla de Cañoles, 8 Jun. 1996, Pujadas (COA 30537).
Orobanche foetida Poir. SPAIN. Granada, Río Trevélez, near Trevélez, 16 Jul. 1990, Pujadas and Poyato (COA 17375), Granada, Km 2 N Trevélez, near Río Trevélez, 19 Jul. 2000, Pujadas and López (COA 30538). Huelva, El Conquero, 10 Jun. 1998, E. Sánchez‐Gullón (COA 28430).
Orobanche gracilis Sm. SPAIN. Cádiz, Sierra del Aljibe, Garganta del Pozo Oscuro, 26 Apr. 1997, Villamandos (COA 23746). Málaga, Campillos, Sierra de Peñarrubia, La Herriza, 26 Mar. 1998, Pujadas, Plaza and Rubio (COA 25553). Huelva, Bailones, cumbres de San Bartolomé, 22 Apr. 1998, López (COA 25302).
Orobanche haenseleri Reut. SPAIN. Jaén, Sierra Mágina, Almadén, Torres, 16 Jun. 2000, Román and Martínez (COA 29921). Granada, Sierra Nevada, Barranco de los Alisos near Río Dúrcal, 22 Jul.1997, Prados, Parra and Jiménez (COA 23555). Granada, Trevélez, Km 5 N from Río Trevélez, 19 Jul. 2000, Pujadas and López (COA 30481). Málaga, Sierra de las Nieves, El Corona‐Tolox, 27 Agu. 1997, Plaza (COA 25719).
Orobanche hederae Duby SPAIN. Burgos, Munilla, 31 Jul. 1990, Pujadas and Poyato (COA 17479). Burgos, Villanés, 30 Jul. 1990, Pujadas and Poyato (COA 17483). Cádiz, P.N. Los Alcornocales, Canuto de la Alcaria, between Sierra La Palma and Sierra Luna, 20 Jun. 1995, Lora, Plaza and Escuín (COA 17469). Córdoba, Cabra, La Nava, 6 Jun. 1995, Lora (COA 17665).
Orobanche laserpitii‐sileris Jordan FRANCIA, Lescum, Pyr. Atlantiques Le Piquet de Lhurs, 25 Agu. 1999, Villar and Benito (COA 30541). SPAIN. Lleida, Tredós, Val d’Aran, 29 Jun. 2001, Pujadas (COA 31053).
Orobanche latisquama (F.W. Schultz) Batt. in Batt. & Trabut SPAIN. Ciudad Real, Fuencaliente, 20 May 1997, Carrillo (COA 28909). Granada, Km 6 from Puebla de Don Fadrique to Santiago de la Espada, 19 Jul.1999, Pujadas (COA 28909). Murcia, Totana, ermita Santa Eulalia, 30XG2784, 19 Jun. 1997, Pujadas (COA 23752).
Orobanche lavandulacea Reichenb SPAIN. Córdoba, Iznájar, cerro del Azucarón, 2 May 1998 (COA 25304). Girona, Km 2 from Port de la Selva to Cadaqués, 12 May 1998, Pujadas (COA 25542). Jaén, Los Cañones, Secos, 1 Jun. 2000, Román and Fiestas (COA 29501).
Orobanche minor Sm SPAIN. Girona, Cadaqués, Faro de Cap de Creus, 15 May 1998, Pujadas (COA 28172). Girona, El Port de la Selva, near Tamariua beach, 15 May 1998, Pujadas (COA 28200). Huelva, Km 3 from Linares de la Sierra a Aracena, 07 May 1997, Lora and Pujadas (COA 30528).
Orobanche montserratii A. Pujadas & D. Gómez SPAIN. Huesca, Fanlo, Cañón de Añisclo, near Ermita San Urbez, 21 Jul. 1997, Pujadas, Montserrat and Gómez (COA 27731). Huesca, Fanlo, San Urbez, Cañón de Añisclo, 2 Agu. 1975, Montserrat (COA 24394). Huesca, Fanlo, Cañón de Añisclo, 21 Jul. 1997, Pujadas, Montserrat and Gómez (COA 27730).
Orobanche mutelii F.W. Schultz in Mutel SPAIN. Almería, Santa María de Nieva, Puertecico‐Cruce a Los Cabreras de Abajo, 11 May 1994, Pujadas (COA 17451). Granada, Cúllar de Baza, Arroyo El Margen, 8 Jun. 2000, Pujadas (COA 29503). Huesca, Jaca, near Barbastro, 19 May 1995, Benito (COA 13590). Toledo, 2 Km N from Ciruelos, 24 May 1995, Pujadas (COA 17452).
Orobanche nana (Reut.) Beck SPAIN. Albacete, Sierra Alcaraz, Puerto Crucetilla, 9 Jun. 1992, Pujadas, Lora and Prados (COA 17461). Córdoba, Alcolea, near Pantano de San Rafael, 16 May 2000, Pujadas, Plaza and Román (COA 29505). Granada, Sierra Parapanda, 28 Jun. 2000, Pujadas, Plaza and Román (COA 29509).
Orobanche olbiensis (Coss.) Nyman SPAIN. Alicante. Pinet beach, Santa Pola, 22 Jun.2000, Crespo (COA 29597).
Orobanche purpurea Jacq. SPAIN. Jaén, Sierra Mágina, Torres, El Almadén, 5 Sep.1997, Pujadas and Mazariegos (COA 25416).
Orobanche ramosa L. SPAIN. Granada, El Jau, 21 Sep. 2000, Pujadas (COA 30534). Granada, Cullar de Vega, 5 Oct. 1989, Pujadas and Oliveira (COA 13605). Granada, La Zubia, 27 Agu. 1988, Oliveira (COA 13816).
Orobanche rapum‐genistae Thuill. SPAIN. Albacete, Sierra Alcaraz, near from Río Mundo, 10 Jun. 1992, Pujadas, Prados and Lora (COA 17594). Granada, Puebla de Don Fadrique, near from Puerta del Pinar, 03 Jun. 2000, Pujadas and López (COA 29576). Huelva, Parque Natural Doñana, La Rocina, Almonte, 15 Apr. 2000, Sánchez Gullón and Cobos (COA 29585). Madrid, El Escorial, 23 Jun. 1972, Hernández and Benítez (COA 13608).
Orobanche reticulata Wallr. subsp. reticulata GERMANY. Bayern, Traithen Kar, Östhang, swischen Oberem Sudelfeld und Kleinem Traithen, 3 Agu.1996, Angerer and Pusch (COA 29393).
Orobanche santolinae Loscos SPAIN. Almería, Tabernas, Venta de los Yesos, 18 May 1996, Pujadas (COA 23566). Córdoba, Rute, Sierra Alta, 29 Jun. 1996, Pujadas and Lora (COA 29460). Granada, Sierra Parapanda, Illora, 07 Jun.2000, Pujadas (COA 29929).
Orobanche schultzii Mutel SPAIN. Almería, Puerto de Santa María de Nieva, 30 May 1994, Pujadas (COA 17465). Huelva, Casa de la Madrina, Sanlúcar de Guadiana, 15 Jun. 2001, Sánchez Gullón (COA 31015).
Orobanche tunetana Beck SPAIN. Alicante, Cabo de Huertas, 17 May 1997, Pujadas and Aguilella (COA 22550).
Orobanche teucrii Holandre FRANCIA. Laruns, Valle de Ossau, Hourat, Cirque de Pan, 07 Jul. 1999, Villar et al. (JACA 97599).
Received: 24 December 2003; Returned for revision: 3 March 2004; Accepted: 18 March 2004, Published electronically: 24 May 2004
References
- Abu SbaihHA, Jury SL.1994a. Seed micromorphology and taxonomy in Orobanche (Orobanchaceae). Flora Mediterranea 4: 41–48. [Google Scholar]
- Abu SbaihHA, Jury SL.1994b. The seed micromorphology of the genus Orobanche (Orobanchaceae). In: Pieterse AH, Verkleij JAC, ter Borg SJ, eds. Proceedings of the 3rd International Workshop on Orobanche and related Striga research Amsterdam, The Netherlands: Royal Tropical Institute, 112–120. [Google Scholar]
- Abu SbaihHA, Keith‐Lucas DM, Jury SL, Harborne JB, Tubaileh AS.1994. Pollen morphology of the genus Orobanche (Orobanchaceae). In: Pieterse AH, Verkleij JAC, ter Borg SJ, eds. Proceedings of the 3rd International Workshop on Orobanche and related Striga research Amsterdam, The Netherlands: Royal Tropical Institute, 99–111. [Google Scholar]
- AndaryC.1994. Chemotaxonomical study of the genus Orobanche In: Pieterse AH, Verkleij JAC, ter Borg SJ, eds. Proceedings of the 3rd International Workshop on Orobanche and related Striga research Amsterdam, The Netherlands: Royal Tropical Institute, 121–126. [Google Scholar]
- Beck von MannagettaG.1890. Monographie der Gattung Orobanche Bibliotheca Botanica 19: 1–275. [Google Scholar]
- Beck von MannagettaG.1930. Orobanchaceae. In: Engler A, ed. Das Pflanzenreich, vol IV. Leipzig: Verlag von Wilhelm Engelmann, 1–348. [Google Scholar]
- BenharratH, Théodet C, Veronesi V, Thalouarn P.2001. Broomrape identification using inter simple sequence repeat (ISSR). Application to Tryonychon Wallr and Osproleon section species. In: Fer A, Thalouarn P, Joel DM, Musselman LJ, Parker C, Verkleij JAC, eds. Proceeding of the 7th International Parasitic Weed Symposium. Nantes, France, 79. [Google Scholar]
- BoeshoreI.1920. The morphological continuity of Scrophulariaceae and Orobanchaceae. Contributions from the Botanical Laboratory of the Morris Arboretum of the University of Pennsylvania 5: 139–177. [Google Scholar]
- BurmmitRK, Powell CE.1992.Authors of plant names. Richmond: Royal Botanic Gardens, Kew. [Google Scholar]
- CanneJM.1979. A light and scanning electron microscope study of seed morphology in Agalinis (Scrophulariaceae) and its taxonomic significance. Systematic Botany 4: 281–296. [Google Scholar]
- CanneJM.1980. Seed surface features in Aureolaria, Brachystima, Tomanthera, and certain South American Agalinis (Scrophulariaceae). Systematic Botany 5: 241–252. [Google Scholar]
- ChaterAO, Webb DA.1972. Orobanchaceae. In: Tutin TG, Heywood VH, Burges NA, Moore DM, Valentine DH, Walters SM, Webb DA, eds. Flora Europaea, 3 Cambridge: Cambridge University Press, 285–294. [Google Scholar]
- ChuangTI, Heckard LR.1972. Seed coat morphology in Cordylanthus (Scrophulariaceae) and its taxonomic significance. American Journal of Botany 59: 258–265. [Google Scholar]
- CossonESC.1848. Sur un genre nouveau de la famille des Orobanchées. Annales des Sciences Naturelles Botanique Paris, series 3 9: 145–147. [Google Scholar]
- CronquistA.1981.An integrated system of classification of flowering plants. New York: Columbia University Press. [Google Scholar]
- DaninA, Baker HG, Baker I.1979. Cytogeography and taxonomy of the Portulaca oleracea L. polyploid complex. Israel Journal of Botany 27: 177–211. [Google Scholar]
- DeifHAR, Al‐Menoufi OA, Ahmed MF.2000. A taxonomic study on the populations of three common species of the genus Orobanche L. (Orobanchaceae) in Egypt. 2. Macro‐ and micromorphology of the seeds and their taxonomic implications. In: Nordernstam B, El Ghazaly G, Kassas M, eds. Plant systematics for the 21st Century London: Portland Press, 251–266. [Google Scholar]
- FoleyMJY.2001.Orobanche In: Paiva J, Sales F, Hedge IC, Aedo C, Aldasoro JJ, Castroviejo S, Herrero A, Velayos M, eds. Flora Iberica, 14. Madrid: CSIC, 32–72. [Google Scholar]
- Font QuerP.1993.Diccionario de Botánica. Barcelona: Labor. [Google Scholar]
- HolmgrenPK, Holmgren NH, Barnett LC.1990.Index Herbariorum. Part 1. The herbaria of the world, ed. 8. Regnum Vegetabile 120. New York: New York Botanical Garden. [Google Scholar]
- HutchinsonJ.1969.Evolution and phylogeny of flowering plants. New York: Academic Press. [Google Scholar]
- JoelDM.1987a. Detection and identification of Orobanche seeds using fluorescence microscopy. Seed Science and Technology 15: 119–124. [Google Scholar]
- JoelDM.1987b. Identification of Orobanche seeds. In: Weber HC, Forstreuter W, eds. Parasitic flowering plants (Proceedings of the 4th ISPFP) Marburg: Philipps‐Universität, 437–444. [Google Scholar]
- JoelDM.1988a. A key for the identification or Orobanche seeds. Phytoparasitica 16: 376–377. [Google Scholar]
- JoelDM.1988b.Orobanche cumana, a new adventive weed in Israel. Phytoparasitica 16: 375. [Google Scholar]
- JuanR, Fernández I, Pastor J.1997. Systematic consideration of microcharacters of fruits and seeds in the genus Verbascum (Scrophulariaceae). Annals of Botany 80: 591–598. [Google Scholar]
- JuanR, Pastor J, Fernández I.1994. Seed morphology in Veronica L. (Scrophulariaceae) from south‐west Spain. Botanical Journal of the Linnean Society 115: 133–143. [Google Scholar]
- JuanR, Pastor J, Fernández I.1998. Estudio de frutos y semillas en Parentucellia Viv. (Scrophulariaceae) y sus implicaciones taxonómicas. Acta Botánica Malacitana 23: 51–57. [Google Scholar]
- JuanR, Pastor J, Fernández I.1999. Morphological and anatomical studies of Linaria species from south‐west Spain: seeds. Annals of Botany 84: 11–19. [Google Scholar]
- JuanR, Pastor J, Fernández I.2000. SEM and light microscope observations on fruit and seeds in Scrophulariaceae from southwest Spain and their systematics significance. Annals of Botany 86: 323–338. [Google Scholar]
- KuijtJ.1969.The biology of parasitic flowering plant. Berkeley and Los Angeles, California: University of California Press. [Google Scholar]
- LangeJMCh.1864.Pugillus plantarum imprimis hispanicarum, quas in itinere 1851–52 legit Jon. Lange. III. Videnskabelige Meddelelser Dansk Naturhistorisk Forening Kjøbenhavn 1861. [Google Scholar]
- MinkinPJ.1987. Pollen morphology of the Orobanchaceae and rhinanthoid Scrophulariaceae with reference to their taxonomy. In: Weber HC, Forstreuter W, eds. Parasitic flowering plants (Proceedings of the 4th ISPFP) Marburg: Philipps‐Universität, 575–589. [Google Scholar]
- MinkinPJ, Eshbaugh WH.1989. Pollen morphology of the Orobanchaceae and rhinanthoid Scrophulariaceae. Grana 28: 1–18. [Google Scholar]
- MusselmanLJ, Mann WF.1976. A survey of surface characteristics of seeds of Scrophulariaceae and Orobanchaceae using scanning electron microscopy. Phytomorphology 26: 370–378. [Google Scholar]
- NovopokrovskyNN, Tzelev P.2000. Orobanchaceae. In: Schischkin BK, ed. Flora of the USSR Vol. XXIII (English edition). Enfield, NH: Science Publishers, 21–145. [Google Scholar]
- OlmsteadRG, de Pamphilis CW, Wolfe AD, Young ND, Elisens WJ, Reeves PJ.2001. Disintegration of Scrophulariaceae. American Journal of Botany 88: 348–361. [PubMed] [Google Scholar]
- van der PijlL.1982.Principles of dispersal in higher plants. Berlin: Springer‐Verlag. [Google Scholar]
- PoloJM.1988. Aportación a la morfología polínica del género Orobanche L. Significados taxonómicos. In: Civit J, Valle MF, eds. Actas VI Simposio de Palinologia APLE Salamanca: 137–140. [Google Scholar]
- PujadasA.1997.Orobanche ballotae A. Pujadas (Orobanchaceae), especie nueva. Acta Botánica Malacitana 22: 29–34. [Google Scholar]
- PujadasA.2000.Orobanche L. (Orobanchaceae), Sect. Trionychon Wallr. en Andalucía. Acta Botánica Malacitana 25: 93–106. [Google Scholar]
- PujadasA.2002.Orobanche L. In: López JA, Catalán P, Sáez LI, eds. Plantas parásitas de la Península Ibérica y Baleares. Madrid: Mundi‐Prensa. [Google Scholar]
- PujadasA, Gómez D.2000.Orobanche montserratii A. Pujadas & D. Gómez (Orobanchaceae), especie nueva del pirineo oscense. Anales del Jardín Botánico de Madrid 57: 267–275. [Google Scholar]
- PujadasA, Lora González A.1995.Orobanche almeriensis A. Pujadas (Orobanchaceae), nueva especie del sureste de la Península Ibérica. Anales del Jardín Botánico de Madrid 53: 47–53. [Google Scholar]
- PujadasA, Lora González A.1996. El género Orobanche L. (Orobancheceae) en la provincia de Almería SE de España. Acta Botánica Malacitana 21: 199–220. [Google Scholar]
- PujadasA, Lora González A.1997. Distribución de Orobanche clausonis Pomel (Orobanchaceae) en la Península Ibérica y Baleares. Anales del Jardín Botánico de Madrid 55: 477–479. [Google Scholar]
- PujadasA, Lora González A, Crespo MB.1997.Orobanche tunetana G. Beck (Orobanchaceae), especie nueva para el continente europeo. Anales del Jardín Botánico de Madrid 55: 277–283. [Google Scholar]
- RománB, Alfaro C, Torres A, Satovic Z, Pujadas A, Rubiales D.2003. Genetic relationship among Orobanche species as revealed by RAPD analysis. Annals of Botany 91: 637–642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RománB, Alfaro C, Torres A, Satovic Z, Kharrat M, Pujadas A, Rubiales D.2001. An analysis of genetic variation in natural populations of Orobanche foetida from Spain and Tunisia. In: Fer A, Thalouarn P, Joel DM, Musselman LJ, Parker C, Verkleij JAC, eds. Proceeding of the 7th International Parasitic Weed Symposium. Nantes, France, 57–60. [Google Scholar]
- RydingO.1992. Pericarp structure and phylogeny within Lamiaceae subfamily Nepetoideae tribe Ocimeae. Nordic Journal of Botany 12: 273–298. [Google Scholar]
- SchneeweissGM.2001. Relationships within Orobanche sect. Tryonychon: insights from its sequences markers. In: Fer A, Thalouarn P, Joel DM, Musselman LJ, Parker C, Verkleij JAC, eds. Proceeding of the 7th International Parasitic Weed Symposium. Nantes, France, 49–52. [Google Scholar]
- SchultzFW.1848.Archives de la flore de France et d’Allemagne Bitche: Haguenau & Deux‐Ponts, 99–154. [Google Scholar]
- StaceCA.1985. Orobanchaceae. In: Heywood VH, ed. Las plantas con flores Barcelona: Reverté, 244–245. [Google Scholar]
- StearnWT.1992.Botanical Latin. London: David & Charles. [Google Scholar]
- SuttonDA.1988.A revision of the tribe Antirrhineae. London: British Museum (Natural History)/Oxford: Oxford University Press. [Google Scholar]
- TakhtajanA.1980. Outline of the classification of flowering plants (Magnoliophyta). Botanical Review 46: 225–339. [Google Scholar]
- TeryokhinES.1991. Orobanche research in the USSR. In: Wegman K, Musselman LJ, eds. Progress in Orobanche research. Proceedings of the International Workshop on Orobanche Research Tübingen, Germany: Eberhard‐Karls University, 30–34. [Google Scholar]
- TeryokhinES.1997.Weed broomrapes, systematics, ontogenesis, biology, evolution. Landshut: Aufstieg‐Verlag. [Google Scholar]
- UngureanL.1986. Recherches sur le tegument seminal dans le cadre des espèces de la section Trionychon Wallr. du genre Orobanche L. Revue Roumaine de Biologie, Série de Biologie Végétale 31: 121–125. [Google Scholar]
- ValdésB, Díez M J.1990. Aportación de los datos polínicos en taxonomía vegetal. In: Blanca G, Díaz de la Guardia C, Fernández MC, Garrido M, Rodríguez‐García MI, Romero AT, eds. Polen, esporas y sus aplicaciones. Granada: Facultad de Ciencias, 153–165. [Google Scholar]
- VelascoL, Goffman FG, Pujadas A.2000. Fatty acids and tocochromanols in seeds of Orobanche Phytochemistry 54: 295–300. [DOI] [PubMed] [Google Scholar]
- WeberHC.1980. Zur Evolution des parasitismus bei den Scrophulariaceae und Orobanchaceae. Plant Systematics and Evolution 136: 217–232. [Google Scholar]
- YoungND, Steiner KE, de Pamphilis CW.1999. The evolution of parasitism in Scrophulariaceae/Orobanchaceae: plastid Gene sequences refute an evolutionary transition series. Annals of the Missouri Botanical Garden 86: 876–893. [Google Scholar]


