Abstract
Autophagy is an evolutionarily conserved process that cells use to degrade and recycle cellular proteins and remove damaged organelles. During the past decade, there has been a growing interest in defining the basic cellular mechanism of autophagy and its roles in health and disease. However, the functional role of autophagy in kidney fibrosis remains poorly understood. Here, using GFP-LC3 transgenic mice, we show that autophagy is induced in renal tubular epithelial cells (RTECs) of obstructed kidneys after unilateral ureteral obstruction (UUO). Deletion of LC3B (LC3−/− mice) resulted in increased collagen deposition and increased mature profibrotic factor TGF-β levels in obstructed kidneys. Beclin 1 heterozygous (beclin 1+/−) mice also displayed increased collagen deposition in the obstructed kidneys after UUO. We also show that TGF-β1 induces autophagy in primary mouse RTECs and human renal proximal tubular epithelial (HK-2) cells. LC3 deficiency resulted in increased levels of mature TGF-β in primary RTECs. Under conditions of TGF-β1 stimulation and autoinduction, inhibition of autolysosomal protein degradation by bafilomycin A1 increased mature TGF-β protein levels without alterations in TGF-β1 mRNA. These data suggest a novel intracellular mechanism by which mature TGF-β1 protein levels may be regulated in RTECs through autophagic degradation, which suppresses kidney fibrosis induced by UUO. The dual functions of TGF-β1, as an inducer of TGF-β1 autoinduction and an inducer of autophagy and TGF-β degradation, underscore the multifunctionality of TGF-β1.
In the kidney, fibrosis is responsible for chronic progressive kidney failure, and the prevalence of CKD is increasing worldwide.1,2 Extracellular matrix (ECM) protein production and progressive accumulation are hallmarks of renal tubulointerstitial fibrosis in progressive kidney disease. Collagens are the main components of the ECM in the kidney, and type I collagen (Col-I) is the major type associated with disease states.3,4 The cellular mechanisms that facilitate tubulointerstitial fibrosis after injury remain incompletely defined. Recent lineage tracing or genetic fate mapping studies have strongly challenged the theory that renal tubular epithelial cells (RTECs) traverse the tubular basement membrane to become myofibroblasts in a process of epithelial-to-mesenchymal transition (EMT), but rather, that interstitial pericytes/perivascular fibroblasts are the myofibroblast progenitor cells.5–7 It also has been proposed that profibrotic factors, such as TGF-β1, are upregulated in the tubular interstitial area on injury, leading to kidney fibrosis.8 TGF-β1 induces production of ECM proteins, including fibronectin and collagens, and inhibits degradation of ECM proteins mainly by matrix metalloproteinases.9–11 Given the recent evidence that casts doubts about the role of EMT in vivo, how RTECs contribute to the development of renal tubulointerstitial fibrosis is not entirely clear.
TGF-β is synthesized as a single polypeptide precursor that includes a preregion signal peptide, which is removed by proteolytic cleavage, and pro–TGF-β, containing a proregion called the latency-associated peptide and a mature TGF-β, and it converts to homodimeric pro–TGF-β through disulfide bonds.12 After cleavage by proprotein convertases, such as furin, latency-associated peptide remains noncovalently associated with the dimeric form of mature TGF-β as the small latent complex (SLC).13 SLC formation occurs in the Golgi apparatus, and mature TGF-β is secreted as part of SLC and associated with latent TGF-β–binding protein to form TGF-β large latent complex, which interacts with ECM. On stimulus, the dimeric form of mature TGF-β is dissociated from large latent complex and becomes the bioactive mature TGF-β ligand, which can then bind TGF-β receptors to trigger downstream Smad-dependent or -independent signaling pathways.12,13 Thus, the availability of mature TGF-β is the limiting factor of TGF-β activity and not TGF-β synthesis per se, because the body generates more pro–TGF-β than necessary. Whereas TGF-β/TGF-β receptor downstream signaling pathways have been extensively investigated, the regulation of TGF-β maturation and bioavailability has not been well studied but may serve as an important target for fibrotic diseases that alter TGF-β signaling.
Macroautophagy, hereafter referred to as autophagy, is a fundamental cellular homeostatic process that cells use to degrade and recycle cellular proteins and remove damaged organelles. The process of autophagy involves the formation of double membrane–bound vesicles called autophagosomes that envelop and sequester cytoplasmic components, including macromolecular aggregates and cellular organelles, for bulk degradation by a lysosomal degradative pathway.14 Autophagy can be induced in response to either intracellular or extracellular factors, such as amino acid or growth factor deprivation, hypoxia, low cellular energy state, endoplasmic reticulum stress or oxidative stress, organelle damage, and pathogen infection.15–22 To date, over 30 genes involved in autophagy have been identified in yeast, and they have been termed autophagy-related genes (Atgs). The mammalian ortholog of Atg8 is comprised of a family of proteins known as microtubule-associated protein 1 light chain 3 (LC3) that functions as a structural component in the formation of autophagosomes.23 LC3B (herein referred to as LC3) is the best characterized form and the most widely used as an autophagic marker. The conversion of the cytosolic form of LC3 (LC3-I) to lipidated form (LC3-II) indicates autophagosome formation. In contrast to LC3, Beclin 1, encoded by the beclin 1 gene, is the mammalian ortholog of yeast Atg6 that is required for the initiation of autophagy through its interaction with Vps34. Homozygous deletion of beclin 1 (beclin 1−/−) exhibits early embryonic lethality, whereas heterozygous deletion (beclin 1+/−) results in increased incidence of spontaneous tumorigenesis, abnormal proliferation of mammary epithelial cells and germinal center B lymphocytes, and increased susceptibility to neurodegeneration.24–27
We previously reported that autophagy promotes intracellular degradation of Col-I induced by TGF-β1 in glomerular mesangial cells.28 In the present study, we explored the functional role of autophagy in an in vivo model of progressive kidney fibrosis induced by unilateral ureteral obstruction (UUO) in autophagy-deficient LC3 null (LC3−/−) and heterozygous (beclin 1+/−) mice and green fluorescent protein (GFP)-LC3 transgenic mice. We also performed functional studies in primary cultured mouse RTECs and human renal proximal tubular epithelial (HK-2) cells. We hypothesized that induction of autophagy in RTECs promotes TGF-β degradation and thereby reduces TGF-β secretion and suppresses development of kidney fibrosis.
Results
Autophagy Is Induced Primarily in RTECs of Obstructed Kidneys after UUO
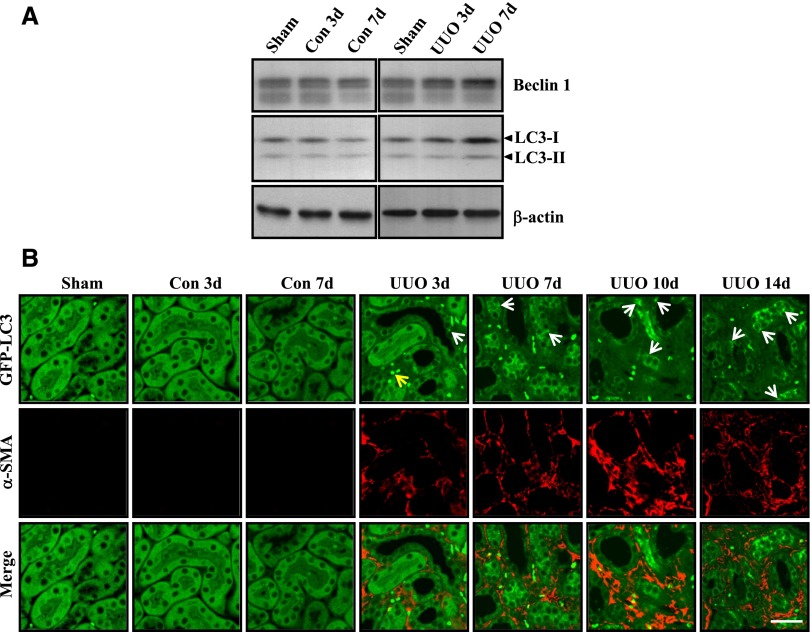
We had previously reported that, in glomerular mesangial cells, autophagy promotes intracellular degradation of collagen induced by TGF-β1.28 To investigate the functional role of autophagy in kidney fibrosis, we examined the induction of autophagy in the kidney after injury induced by UUO in mice. We first determined the expression of two autophagy-related proteins, Beclin 1 and LC3, in both obstructed and contralateral kidneys at 3 and 7 days after UUO and compared it with sham-operated kidneys. As shown in Figure 1A, the levels of Beclin 1, LC3-I, and LC3-II proteins were increased in the obstructed kidneys at 3 and 7 days after UUO but not the corresponding contralateral kidneys compared with sham-operated kidneys.
Figure 1.
Autophagy is induced in epithelial cells of obstructed kidneys after UUO. (A) Expression of Beclin 1 and LC3 proteins examined by Western blot analysis of tissue lysates from obstructed and contralateral kidneys at 3 days (UUO 3d and Con 3d, respectively) and 7 days (UUO 7d and Con 7d, respectively) after UUO or sham operation in C57BL/6 mice. Immunoblotting for β-actin was used as sample loading controls. Similar results were obtained from three independent experiments, and representative blots are shown. (B) Immunofluorescence staining for α-SMA (red) in the renal cortex of obstructed kidneys of GFP-LC3 (green) transgenic mice 3, 7, 10, and 14 days after UUO (UUO 3d, UUO 7d, UUO 10d, and UUO 14d, respectively), corresponding contralateral kidneys (Con 3d and 7d), and sham-operated kidneys. Similar results were observed in three independent experiments, and representative images are shown. White arrows indicate GFP-LC3 puncta (autophagosome) formation, and the yellow arrow points to a red blood cell. Scale bar, 30 μm.
Puncta formation of GFP-LC3 fusion protein is a widely used marker to visualize autophagosomes, and it represents the accumulation of a membrane-bound form of LC3 in formation of autophagic vesicles.29 Using GFP-LC3 transgenic mice, we examined overall cellular distribution of the autophagosome marker LC3 in the kidneys of mice subjected to UUO for 3, 7, 10, and 14 days. Punctate distribution of GFP-LC3 was visualized by confocal fluorescence microscopy, and representative images are shown in Figure 1B. We observed a high level of constitutive autophagy in the glomeruli under basal conditions, consistent with a previous report in podocytes compared with other intrinsic renal cells30 that did not change after UUO (data not shown). In contrast, there was little constitutive autophagy within the renal tubules. However, after UUO, the abundance of GFP-LC3 puncta (Figure 1B, green dots) in RTECs of obstructed kidneys increased, but it did not increase in contralateral kidneys compared with sham-operated kidneys. We also used α-smooth muscle actin (α-SMA) as an interstitial myofibroblast marker to examine whether autophagosomes can be observed in myofibroblasts after UUO. As shown in Figure 1B, an increased number of α-SMA–expressing cells (Figure 1B, red) was detected in the interstitium of the obstructed kidneys, compared with the contralateral and sham-operated kidneys. In the obstructed kidneys, increased abundance of GFP-LC3 dots was observed predominantly in RTECs and rarely in α-SMA–positive interstitial cells.
Deficiency of Autophagic Proteins LC3 and Beclin 1 Results in Increased Collagen Deposition in Obstructed Kidneys
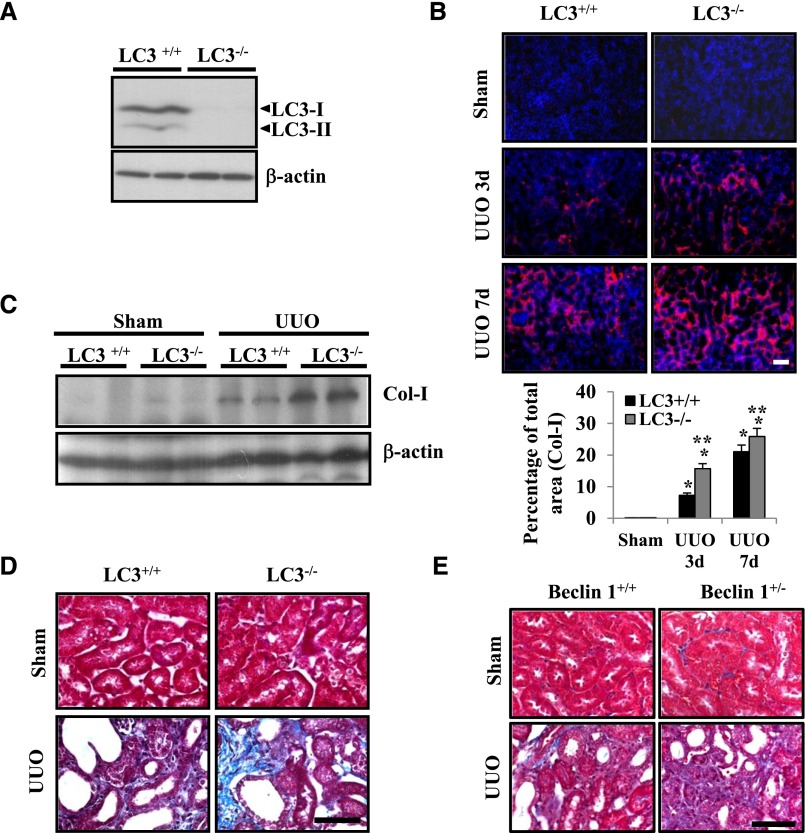
To assess the function of the autophagy-related protein LC3 in kidney fibrosis, we examined the kidneys of LC3 null (LC3−/−) mice compared with wild-type littermate (LC3+/+) mice 3 and 7 days after UUO. We first confirmed the deletion of LC3 expression in the kidneys from LC3−/− mice by Western blot analysis (Figure 2A). Next, immunofluorescence staining for Col-I revealed that collagen accumulation was significantly enhanced in the tubulointerstitial areas of kidneys of LC3−/− mice at 7 days after UUO compared with sham-operated LC3−/− mice and LC3+/+ control mice (Figure 2B). Western blot analysis further confirmed the findings that disruption of LC3-enhanced Col-I protein expression in the 7-day UUO kidney compared with LC3+/+ mice and sham-operated LC3−/− mice (Figure 2C). Consistently, Masson Trichrome staining revealed that LC3−/− mice developed increased collagen deposition in the kidneys 7 days after UUO compared with sham-operated LC3−/− mice and LC3+/+ control mice (Figure 2D). Similarly, mice deficient in autophagic protein Beclin 1 through heterozygous deletion (beclin 1+/−) also exhibited increased collagen deposition, confirming our previous report that reduced Beclin 1 expression leads to increased collagen levels and profibrotic phenotype.28 Here, we also show that the beclin 1+/− mice developed increased collagen deposition in the kidneys 7 days after UUO compared with sham-operated beclin 1+/− mice and beclin 1+/+ control mice (Figure 2E). Taken together, these data showed that deficiency of LC3 or Beclin 1 leads to increased collagen protein levels, suggesting that LC3 and Beclin 1 may function to inhibit kidney fibrosis in the obstructed kidneys.
Figure 2.
Deletion of LC3 results in enhanced collagen deposition in the kidneys after UUO. (A) LC3 protein expression levels in the kidneys of LC3+/+ and LC3−/− mice were examined by Western blot analysis. Similar results were obtained from at least three different mice of each genotype, and representative blots are shown. (B) Representative confocal photomicrographs of immunofluorescence labeling for Col-I (red) on kidney sections of LC3+/+ and LC3−/− mice 3 and 7 days after UUO or sham operation (upper panel). (Lower panel) Relative quantification of immunofluorescence staining in kidney sections that were labeled with antibody to Col-I. *P<0.001 versus sham mice; **P<0.05 versus LC3+/+ mice with UUO at each time point. (C) Levels of Col-I expression in the kidneys of LC3+/+ and LC3−/− mice 7 days after UUO or sham operation were assessed by Western blot analysis. Similar results were obtained from three independent experiments, and representative data from two different mice in each group are shown. Immunoblotting for β-actin was used as the sample loading control. (D) Representative photomicrographs of Masson Trichrome–stained kidneys of LC3+/+ and LC3−/− mice 7 days after UUO or sham operation. (E) Representative photomicrographs of Masson Trichrome–stained kidneys of beclin 1+/+ and beclin 1+/− mice at 7 days after UUO or sham operation. Scale bars, 50 μm.
RTEC Apoptosis Is Enhanced in Obstructed Kidneys of beclin 1+/− Mice
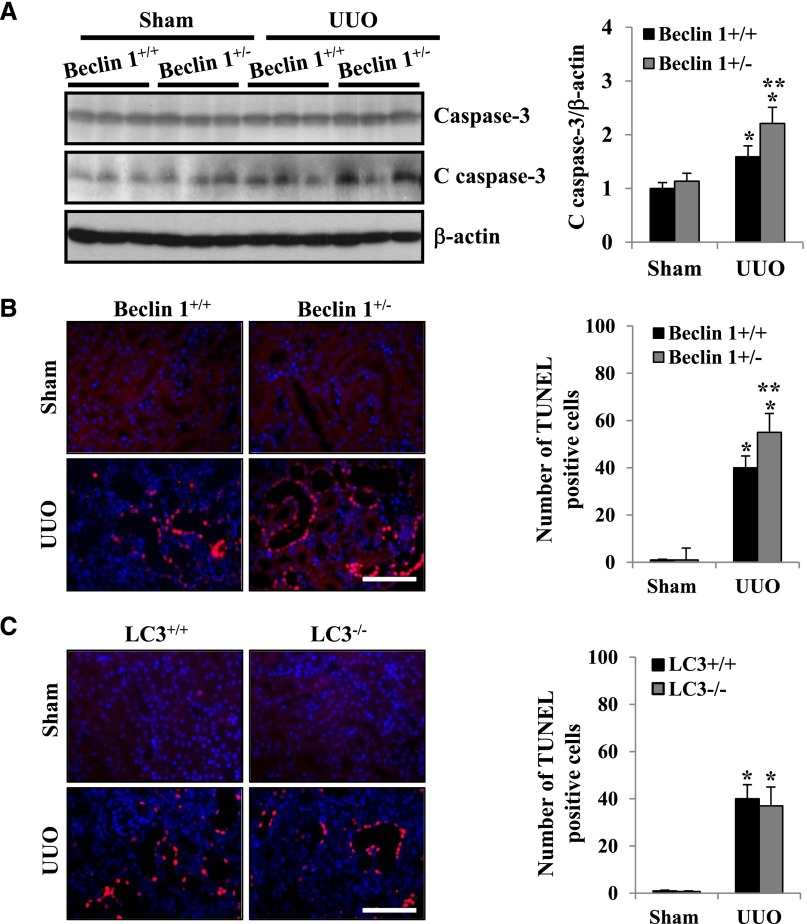
There is emerging evidence indicating that Beclin 1 regulates crosstalk between apoptosis and autophagy and has an antiapoptotic role in several settings, including chemotherapy, irradiation, immunotherapy, angiogenesis inhibitors, nutrient deprivation, and hypoxia.31 It has been previously shown that apoptosis is increased in obstructed renal tubules.32 However, the functional role of Beclin 1 and LC3 in UUO-induced kidney injury and apoptosis has not been shown. Therefore, we examined whether deficiency of Beclin 1 or LC3 influences RTEC apoptosis in the UUO kidney. Although the beclin 1−/− mice are embryonic lethal, the beclin 1+/− mice are viable and display approximately 50% reduction in autophagic activity compared with beclin 1+/+ littermates.25,33 As shown in Figure 3A, increased apoptosis in the obstructed kidneys of beclin 1+/− mice was revealed by higher levels of cleaved caspase-3 expression, a marker of apoptosis, compared with beclin 1+/+ mice. The number of terminal deoxynucleotidyl transferase–mediated digoxigenin-deoxyuridine nick-end labeling (TUNEL)-positive cells was also significantly greater in the obstructed kidneys of beclin 1+/− mice, indicating that tubular epithelial cell apoptosis was significantly increased at 7 days after UUO injury compared with beclin 1+/+ mice (Figure 3B). In contrast, there were no significant differences in the number of TUNEL-positive cells in the obstructed kidneys of LC3−/− mice compared with LC3+/+ mice at 7 days after UUO (Figure 3C). Thus, these data suggest that Beclin 1 functions to protect RTECs from apoptosis after UUO injury.
Figure 3.
Reduced expression of Beclin 1 by heterozygous deletion of beclin 1 is associated with increased tubular epithelial cell apoptosis in kidneys after UUO. (A) Western blot for expression of cleaved caspase-3 (C caspase-3) in the kidney cortex of beclin 1+/+ and beclin 1+/− mice 7 days after UUO or sham operation (left panel). The levels of C caspase-3 were quantitated as the ratio to β-actin by densitometry, and data are presented as the mean±SEM of three independent experiments (right panel). *P<0.001 versus sham mice; **P<0.05 versus beclin 1+/+ mice with UUO. (B) Representative images (left panel) and quantification of TUNEL-positive apoptosis (right panel) in the renal cortex of beclin 1+/+ mice and beclin 1+/− mice 7 days after UUO or sham operation. Data are presented as the mean±SEM of three independent experiments. *P<0.001 versus sham mice; **P<0.05 versus beclin 1+/+ mice with UUO. (C) Representative images (left panel) and quantification of TUNEL-positive apoptosis (right panel) in the renal cortex of LC3+/+ mice and LC3−/− mice 7 days after UUO or sham operation. Data are presented as the mean±SEM of three independent experiments. *P<0.001 versus sham mice. Scale bars, 50 μm.
TGF-β1 Induces Autophagy in Cultured RTECs
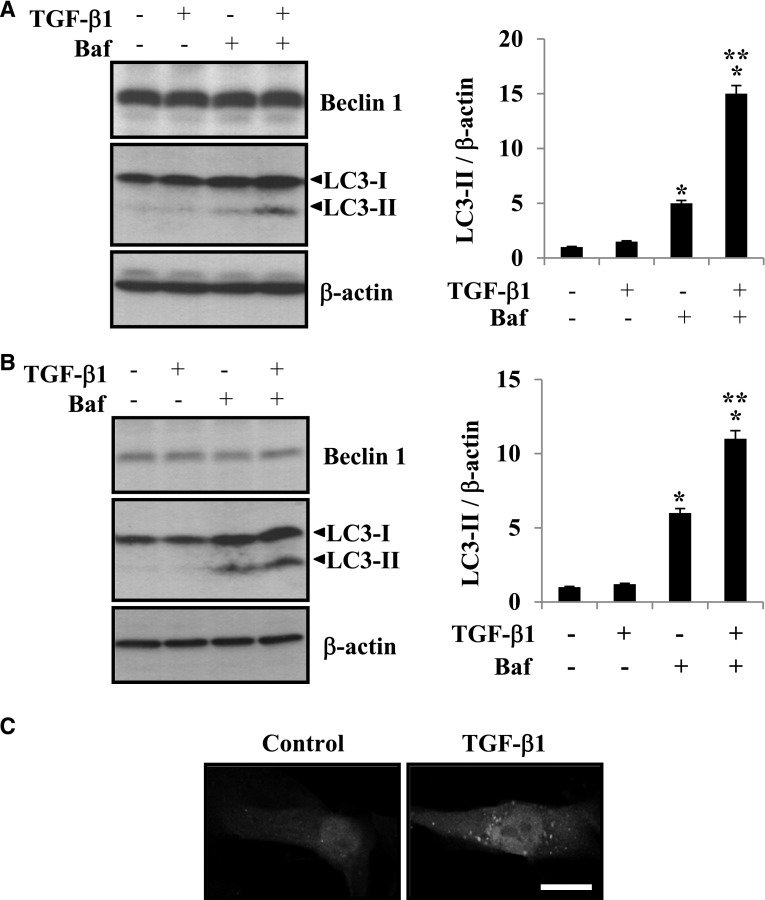
TGF-β1 has been reported to be upregulated in response to injurious stimuli, including UUO, and mediates synthesis of ECM proteins and kidney fibrosis.8 Our findings shown above indicate that autophagy is primarily induced in tubular epithelial cells of obstructed kidneys after UUO injury and that LC3 deficiency promotes kidney fibrosis in vivo. We next assessed whether TGF-β1 induces autophagy in primary cultured RTECs and HK-2 cells in vitro. Because autophagy is a dynamic process involving LC3-II degradation, bafilomycin A1, an inhibitor of autolysosomal degradation pathway, was used to assess autophagic activity or flux. As shown in Figure 4A, treatment with bafilomycin A1 and TGF-β1 resulted in greater accumulation of LC3-II in primary cultured RTECs (Figure 4A) and HK-2 cells (Figure 4B) compared with treatment with TGF-β1 or bafilomycin A1 alone, indicating that autophagic flux is increased with TGF-β1 treatment. Furthermore, we used primary cultured RTECs obtained from GFP-LC3 transgenic mice and showed that treatment with TGF-β1 resulted in increased abundance of GFP-LC3 puncta, indicating increased autophagosome formation compared with the nonstimulated control cells (Figure 4C). These data show that TGF-β1 induces autophagy in RTECs.
Figure 4.
TGF-β1 stimulation induces autophagy in RTECs. (A and B) Western blot analysis of LC3 in (A) HK-2 cells and (B) RTECs incubated in the absence or presence of TGF-β1 (2 ng/ml) and bafilomycin A1 (Baf; 10 nM) as indicated for 24 hours. Immunoblotting for β-actin was used as the protein loading control. The levels of LC3-II were quantitated as the ratio to β-actin by densitometry. Data are presented as the mean±SEM of three independent experiments. *P<0.05 versus no treatment; **P<0.05 versus Baf treatment only. (C) Representative image of the punctate formation of GFP-LC3 visualized by confocal fluorescence microscopy. RTECs obtained from GFP-LC3 transgenic mice were incubated in the absence (control) or presence of TGF-β1 (2 ng/ml) for 24 hours. Scale bar, 10 μm.
LC3 Deficiency Results in Increased Mature TGF-β in Obstructed Kidneys
TGF-β activity is controlled by the availability of mature TGF-β. The Golgi apparatus is an important site for the TGF-β maturation process that is necessary for generation and secretion of the bioactive mature TGF-β ligand that can bind TGF-β receptors for signal transduction.13,34 The Golgi is also a potential membrane source for autophagy, and LC3 is known to form a stable association with the membrane component in the formation of autophagosomes.35,36 Thus, we sought to determine whether autophagy regulated the levels of mature TGF-β, thereby controlling the availability and secretion of mature TGF-β to regulate TGF-β activity induced by UUO injury. We examined the levels of pro–TGF-β and mature TGF-β proteins in the kidneys of LC3+/+ and LC3−/− mice after UUO. We found that mature TGF-β but not pro–TGF-β levels were significantly increased in the obstructed kidneys of LC3−/− mice compared with wild-type LC3+/+ mice (Figure 5) after UUO, indicating that LC3 functions to suppress mature TGF-β levels induced by UUO.
Figure 5.
Autophagy suppresses mature TGF-β levels in the kidneys after UUO. Expression of pro–TGF-β and mature TGF-β proteins in the kidney lysates from LC3+/+ and LC3−/− mice 7 days after UUO or sham operation. Representative Western blot data from two different mice in each group are shown. β-Actin was used as a loading control. The relative abundance of mature TGF-β was quantitated as the ratio of mature TGF-β to β-actin by densitometry, and data are presented as the mean±SEM of three independent experiments. *P<0.01 versus sham mice; **P<0.05 versus LC3+/+ mice with UUO.
Mature TGF-β Is Degraded by Autophagic Pathway under TGF-β1 Stimulation and Autoinduction
Autoinduction of TGF-β1 mRNA by TGF-β1 has been shown in RTECs and other cell types, such as mesangial cells.37,38 We next determined whether LC3 deficiency leads to increased mature TGF-β levels under TGF-β stimulation and autoinduction in RTECs in vitro. Primary cultured RTECs obtained from the kidneys of LC3+/+ and LC3−/− mice were analyzed for levels of pro–TGF-β and mature TGF-β proteins. As shown in Figure 6A, TGF-β1 stimulation increased mature TGF-β levels in RTECs from LC3+/+ mice and LC3-deficient RTECs from LC3−/− mice. TGF-β1–stimulated increases in mature TGF-β protein levels were further enhanced. These findings indicate that disruption of LC3 leads to increased mature TGF-β protein levels under TGF-β stimulation and autoinduction in RTECs. We further examined whether autophagy function regulates the level of mature TGF-β levels in HK-2 cells by the inhibition of autolysosomal degradation using bafilomycin A1.39,40 As shown in Figure 6B, mature TGF-β protein levels were significantly increased in HK-2 cells by TGF-β1 stimulation (Figure 6B, middle panel, lane 2) and further increased on cotreatment with bafilomycin A1 (Figure 6B, middle panel, lane 4). Thus, these data suggest that the autolysosomal protein degradation pathway may be responsible for the intracellular degradation of mature TGF-β protein.
Figure 6.

Autophagy promotes degradation of mature TGF-β in RTECs. (A) Levels of pro–TGF-β and mature TGF-β in primary cultured RTECs from LC3+/+ and LC3−/− mice incubated in the absence (control) or presence of TGF-β1 (1 ng/ml) as indicated for 24 hours. Immunoblotting for β-actin was used as the protein loading control. The relative abundance of mature TGF-β was quantitated as the ratio to β-actin by densitometry, and data are presented as the mean±SEM of three independent experiments. *P<0.05 versus LC3+/+ RTECs treated with TGF-β1. (B) Levels of pro–TGF-β and mature TGF-β in HK-2 cells incubated in the absence or presence of TGF-β1 (2 ng/ml) and Baf (10 nM) as indicated for 24 hours. Immunoblotting for β-actin was used as the protein loading control. The relative abundance of mature TGF-β was quantitated as the ratio to β-actin by densitometry, and data are presented as the mean±SEM of three independent experiments. *P<0.05 versus no treatment; **P<0.05 versus TGF-β1 treatment only. (C) Semiquantitative RT-PCR for TGF-β1 mRNA expression in HK-2 cells incubated in the absence or presence of TGF-β1 (2 ng/ml) and Baf (10 nM) as indicated for 24 hours. Amplified cDNA level of TGF-β1 was standardized with that of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Densitometric analysis data are presented as the mean±SEM of three independent experiments. *P<0.05 versus no treatment. (D) Representative confocal photomicrographs of immunofluorescence labeling for LC3 (red) and TGF-β1 (green) in HK-2 cells incubated in the absence (control) or presence of TGF-β1 (2 ng/ml) and Baf (10 nM) as indicated for 24 hours. (E) Effects of conditioned media (CM) from untreated (control) or TGF-β1–treated LC3+/+ and LC3−/− RTECs on Col-I expression in NIH3T3 fibroblasts. The relative abundance of Col-I was quantitated as the ratio to β-actin by densitometry, and data are presented as the mean±SEM of three independent experiments. *P<0.05 versus control CM; **P<0.05 versus TGF-β1–treated CM from LC3+/+ RTECs.
To assure that the increases in mature TGF-β protein levels observed with bafilomycin A1 treatment were not because of corresponding increases in TGF-β1 mRNA expression and confirm the TGF-β autoinduction in RTECs, we performed semiquantitative RT-PCR, and representative results are shown in Figure 6C. No significant increases in the levels of TGF-β1 mRNA were detected in HK-2 cells on treatment with bafilomycin A1 compared with untreated cells (Figure 6C, lane 3 versus lane 1). Stimulation with TGF-β1 significantly increased TGF-β1 mRNA, confirming TGF-β autoinduction in RTECs as expected, but cotreatment with bafilomycin A1 did not further increase TGF-β1 mRNA (Figure 6C, lane 2 versus lane 4). Thus, inhibition of autophagy increased mature TGF-β1 protein levels without similarly inducing TGF-β1 mRNA expression, indicating that the increases in mature TGF-β protein levels are a result of decreased degradation rather than increased synthesis.
We further determined whether intracellular TGF-β1 is colocalized with LC3 puncta, which represent autophagosome formation.36 We assessed for punctate distribution of LC3 (Figure 6D, red) and subcellular localization of TGF-β1 (Figure 6D, green) visualized by confocal fluorescence microscopy, and representative images are shown in Figure 6D. We observed that treatment with bafilomycin A1 alone or TGF-β1 increased the abundance of autophagosomes (Figure 6D, red dots) compared with the nonstimulated control cells. These results are consistent with the Western blot analysis (Figure 4A) detecting increases in endogenous LC3-II. We also observed that cotreatment with bafilomycin A1 and TGF-β1 increased the TGF-β1 staining (Figure 6D, green) compared with bafilomycin A1 or TGF-β1 treatment alone or nonstimulated control cells. Moreover, cotreatment with bafilomycin A1 and TGF-β1 resulted in increased colocalization of LC3 and TGF-β1 (seen as orange-colored dots in the merged images in Figure 6D) compared with bafilomycin A1 or TGF-β1 treatment alone or nonstimulated control cells.
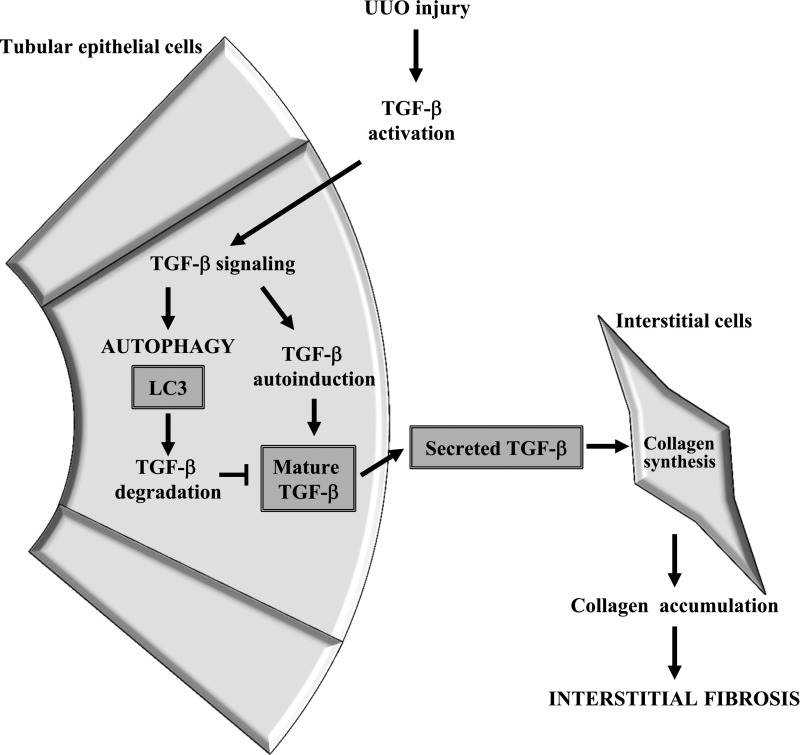
Increases in mature TGF-β lead to increased TGF-β secretion, and we examined whether conditioned media from TGF-β1–treated LC3-deficient RTECs induce Col-I expression in fibroblasts. As shown in Figure 6E, we observed that the conditioned media from TGF-β1–treated LC3−/− RTECs enhanced Col-I production in NIH3T3 fibroblasts in vitro. These data suggest that LC3 may negatively regulate the level of mature TGF-β in RTECs and that LC3 deficiency enhances the level of secreted active TGF-β that leads to increase in collagen production by adjacent interstitial fibroblasts (Figure 7).
Figure 7.
Schematic representation of the proposed model for the effects of autophagy induction in RTECs on tubulointerstitial fibrosis. Kidney injury induced by UUO results in activation of TGF-β and leads to the induction of autophagy. Activation of autophagy promotes degradation of mature TGF-β, thereby decreasing TGF-β secretion, and suppresses tubulointersitial fibrosis induced by UUO.
Discussion
In the present study, we investigated the functional role of autophagy in kidney injury and fibrosis. We show that kidney injury induced by UUO, a well established in vivo model of progressive kidney fibrosis, results in induction of autophagy and that deficiency of autophagic protein LC3 leads to increased collagen deposition and mature TGF-β levels in obstructed kidneys. Our findings suggest that autophagy regulates TGF-β expression and suppresses kidney fibrosis.
An emerging body of evidence suggests that the induction of autophagy constitutes a cytoprotective mechanism as a stress-adaptive response to tissue injury and that dysregulation of autophagy leads to the pathogenesis of human diseases.41,42 In the kidney, it has been shown that podocytes, which are terminally differentiated epithelial cells lining the outer aspect of the glomerular basement membrane, exhibit a high basal level of autophagy,30 which is thought to represent a self-repair/survival mechanism that is especially important in postmitotic cells, such as podocytes, that, like neurons, have a very limited capacity for cell division and replacement. In contrast, our studies using GFP-LC3 transgenic mice show that autophagic activity in RTECs is not high constitutively compared with the podocytes but potently induced with injury after UUO. In the obstructed kidneys, our studies show increased abundance of GFP-LC3 puncta formation in tubular epithelial cells and increased expression of Beclin 1, LC3-I, and LC3-II proteins similar to previous report showing upregulated Beclin 1 and LC3 and increased autophagic vacuoles in obstructed renal tubules using transmission electron microscopy.6 Thus, these findings indicate that kidney injury after UUO potently induces autophagy in the RTECs.
Recent studies implicate dysregulated autophagy in disorders characterized by fibrosis in various tissues, including cardiac fibrosis, liver fibrosis, and idiopathic pulmonary fibrosis.43 We have recently reported a critical role of autophagy in negatively regulating matrix production in glomerular mesangial cells by promoting degradation of intracellular Col-I induced by TGF-β1.28 Moreover, inhibition of autophagy by a selective chemical inhibitor, 3-methyladenine, has been shown to enhance interstitial fibrosis in the obstructed kidneys after UUO in rats.44 Here, we use a genetic approach to inhibit autophagy and show that deletion of the LC3 gene in mice results in increased collagen expression and deposition in the tubulointerstitial areas of kidneys after injury induced by UUO, suggesting that LC3 may function to inhibit kidney fibrosis.
Given that beclin 1 homozygous knockout mice display embryonic lethality and die at about embryonic day 7.5,24 whereas other autophagy-related gene knockout mice, such as LC3−/− mice, can survive to birth, Beclin 1 likely has additional functions other than its role in the initiation of autophagy. Indeed, Beclin 1 is known to mediate crosstalk between autophagy and apoptosis. Beclin 1, which contains a Bcl-2 homology-3 domain, was initially identified as a Bcl-2–interacting protein and has an antiapoptotic role in certain conditions, such as nutrient deprivation and hypoxia, possibly as a stress-adaptive mechanism.31 Caspase-mediated cleavage of Beclin 1 generates N- and C-terminal fragments, resulting in loss of its proautophagic activity and, sensitizes cells to apoptotic signals.45 The depletion of Beclin 1 has been shown to trigger caspase-dependent programmed cell death in Caenorhabditis elegans.31 In the UUO model, both induction of autophagy and apoptosis have been shown to occur in a time-dependent manner. Indeed, increased cell death because of apoptosis leading to tubular epithelial loss is a prominent feature in UUO, and our findings show that inhibition of Beclin 1 through heterozygous deletion of beclin 1 gene enhanced RTEC apoptosis in the obstructed kidneys. These data suggest that Beclin 1 functions to protect RTECs from apoptosis after UUO injury.
A hallmark of CKDs is tubulointerstitial fibrosis with excessive matrix deposition produced by myofibroblasts,46 and tubular epithelial cells have been thought to have an active role in this process by EMT and directly contribute to the myofibroblast pool.47–50 However, recent studies of epithelial lineage in models of kidney injury indicate that EMT is unlikely to occur in vivo,5,6 therefore raising the significance of paracrine actions by which tubular epithelial contribution to interstitial fibrosis is through paracrine signaling rather than the EMT. Interestingly, our data from the GFP-LC3 transgenic mice revealed that the RTECs and not the interstitial cells most prominently display autophagic activity, which was evidenced by increased abundance of GFP-LC3 puncta after kidney injury induced by UUO. Furthermore, we found that treatment with exogenous TGF-β1 induces autophagy in primary cultured mouse RTECs and HK-2 cells. Actions of TGF-β1 are well known as potent inducers of ECM production and fibrogenesis, and they are induced during kidney injury, including the actions induced by UUO. Moreover, TGF-β1 can potentiate its actions through autoinduction. Therefore, in the present study, we focused on investigating the mechanism by which autophagy in tubular epithelial cells regulates ECM proteins produced by interstitial fibroblasts in response to TGF-β1 and kidney injury and fibrosis induced by UUO. We found that the deletion of LC3 resulted in increased mature TGF-β proteins in the obstructed kidneys after UUO. Similarly, blockade of autophagy in RTECs by the use of LC3-deficient RTECs isolated from LC3−/− mice or inhibition of autolysosomal protein degradation by treatment with bafilomycin A1 also resulted in additional increases in mature TGF-β protein levels without corresponding alterations in TGF-β1 mRNA when its expression was induced by exogenous TGF-β1. These data suggest that LC3 functions to decrease mature TGF-β levels in RTECs through autophagic degradation and consequently, reduces TGF-β secretion and suppresses development of interstitial fibrosis induced by UUO.
Evidence for paracrine effects by tubular cell-derived TGF-β1 is corroborated in a transgenic mouse model of conditional overexpression of TGF-β1 in renal tubules, which displayed widespread peritubular deposition of collagen and fibrosis and focal degeneration of nephrons with empty collapsed remnants of tubular basement membrane embedded into a dense collagenous fibrous tissue but no evidence for transition of tubular cells into myofibroblasts.7 Moreover, in a rat model of ischemia-reperfusion injury, tubular production of TGF-β1 was increased; through paracrine stimulation, in turn, it activated interstitial fibroblasts and led to tubulointerstitial fibrosis, whereas treatment with a pharmacological inhibitor of TGF-β type I receptor blocked TGF-β signaling and ameliorated interstitial fibrosis.51 Our studies show that the conditioned media from TGF-β1–treated LC3-deficient (LC3−/−) RTECs enhanced Col-I production in NIH3T3 fibroblasts and suggest that LC3 may play a role in regulating secreted mature TGF-β by RTECs and acting in a paracrine fashion to regulate collagen production by adjacent interstitial fibroblasts. Thus, paracrine signaling by tubular epithelial cells through secreted bioactive TGF-β may be an important mechanism that causes interstitial fibrosis.
Regulation of cytokine signaling through autophagic degradation, thereby limiting the availability of the mature cytokine for subsequent processing and secretion, is a novel intracellular mechanism for regulating TGF-β. Interestingly, autophagy recently has been shown to play a role in regulating IL-1 family cytokines. IL-1β is a proinflammatory cytokine that is first produced as a proform, similar to TGF-β, and autophagy controls IL-1β secretion by targeting intracellular pro–IL-1β for degradation.52 Induction of autophagy occurs in cells stimulated with Toll-like receptor ligands and leads autophagosomes to sequester and degrade pro–IL-1β, damaged mitochondria, and inflammasome components, suggesting that autophagy may regulate inflammation through the degradation.52–57 The induction of autophagy in mice using rapamycinreduced LPS-induced elevation of serum IL-1β, and inhibition of autophagy prevented IL-1β degradation and increased mature IL-1β.52 Moreover, inhibition of autophagy increased inflammasome activation, whereas induction of autophagy repressed it.53 These studies suggest that the induction of autophagy by inflammatory stimuli may serve as a self-regulatory mechanism by which autophagy controls inflammatory cytokine secretion and reduce active inflammasomes, thereby limiting potentially harmful inflammatory responses. Similarly, our findings suggest a novel role for autophagy as a cytoprotective mechanism to negatively regulate the production of mature TGF-β proteins in RTECs, consequently limiting TGF-β secretion and suppressing development of interstitial fibrosis in kidney injury.
ECM synthesis is important for the initial stage of repair of tubular injury, and transient peritubular myofibroblast differentiation of interstitial fibroblasts may provide beneficial role on tubular recovery as described by Fujigaki et al.58 Similarly, TGF-β promotes wound repair and regeneration, thereby exerting paradoxical cytoprotective effects to mitigate tissue injury. However, extended dysregulation of expression and activation of TGF-β results in relentless ECM synthesis and accumulation, development of kidney fibrosis, and ultimately, end stage kidney failure. Therefore, the beneficial effects of TGF-β activation and ECM synthesis are transient. Suzuki et al.59 also reported that tubular injury led to the development of surrounding matrix protein deposition and contributed to the pathogenesis of interstitial fibrosis. Indeed, Fujigaki et al.58 used a single intravenous injection of uranyl acetate (5 mg/kg) into Sprague–Dawley rats; tubular injury reached its peak 7 days after the injection, and then, tubules underwent a repair and regeneration process. However, our UUO model induces tubular injury consistently without recovery process. Thus, effects of TGF-β activation, ECM synthesis, and activation of interstitial fibroblasts on renal injury may be protective/reparative or deleterious/progressive depending on, for instance, the duration and extent of injury.
In summary, we investigated the functional role of autophagy proteins LC3 and Beclin 1 in kidney injury and fibrosis. The process of autophagy provides a system for protein degradation that is essential for tissue homeostasis and cell survival. We show that the induction of autophagy in RTECs protects against apoptosis and promotes TGF-β degradation, thereby reducing TGF-β secretion and development of renal interstitial fibrosis. Our findings suggest a novel intracellular mechanism for regulating TGF-β by autophagy. Kidney injury, including that induced by UUO, leads to activation of TGF-β, which is a potent inducer of ECM production and fibrogenesis, and can also potentiate its actions through autoinduction. Autophagy may serve as an adaptive mechanism that allows tight control of TGF-β actions.
Concise Methods
Reagents
Recombinant human TGF-β1 was purchased from R&D Systems (Minneapolis, MN). Bafilomycin A1, anti-LC3 antibody, and Cy3-conjugated anti–α-SMA antibody were from Sigma-Aldrich (St. Louis, MO). Anti–Col-I and anti–TGF-β1 antibodies were from Calbiochem (San Diego, CA) and Abcam (Cambridge, MA), respectively. Antibodies against cleaved caspase-3, caspase-3, and TGF-β were purchased from Cell Signaling Technology (Danvers, MA). Antibodies against Beclin 1 and β-actin were from Santa Cruz Biotechnology (Santa Cruz, CA). Cy3-conjugated anti-rabbit IgG, Cy3-conjugated anti-mouse IgG, and FITC-conjugated anti-rabbit IgG were from Jackson ImmunoResearch Laboratories (West Grove, PA).
Mice and UUO
Male C57BL/6 mice were from The Jackson Laboratory (Bar Harbor, ME). GFP-LC3 transgenic mice were provided by Riken Laboratories (Hirosawa, Japan). LC3+/+ and LC3−/− mice were provided by Marlene Rabinovitch (Standford, CA).60 Beclin 1+/+ and beclin 1+/− mice were provided by Beth Levine (Dallas, TX).61 For UUO, mice (8–12 weeks of age) were anesthetized with pentobarbital, and the left ureter was exposed by a left dorsal incision. The ureter was obstructed by two point ligations with silk sutures. Sham-operated mice underwent the same procedure, except for the obstruction of the left ureter. The incision was closed, and mice were allowed to recover. Mice were euthanized 3, 7, 10, or 14 days after surgery. Kidneys were fixed with 4% paraformaldehyde before embedding in paraffin. Paraffin sections (5-µm thick) were used for Masson Trichrome staining or immunostaining. All experimental protocols were approved by the Institutional Animal Care and Use Committee at Harvard Medical School.
Cell Culture
HK-2 cells were obtained from American Type Culture Collection (Manassas, VA) and cultured in DMEM/F-12 medium containing insulin (5 µg/ml), transferrin (5 µg/ml), selenium (5 ng/ml), hydrocortisone (36 ng/ml), triiodothyronine (4 pg/ml), and 10% FCS. Primary cultured RTECs were obtained from C57BL/6, LC3+/+ and LC3−/−, and GFP-LC3 transgenic mice using procedures as previously described.62 Briefly, to isolate RTECs, the renal cortex was divided into small pieces (<1 mm) and incubated in HBSS containing 0.2% collagenase II for 1 hour at 37°C in an oxygen-saturated atmosphere. The samples were then passed through sieves of descending pore sizes (250, 150, 68, and 40 µm). The resultant tubular epithelial cell isolates were resuspended and cultured in modified K1 medium containing 5% FCS (Gibco, Eggenstein, Germany), 10 µg/ml EGF, 10 mM Hepes, 0.5 mg/ml prostaglandin E2, 180 µg/ml hydrocortisone, 100 units/ml penicillin, and 100 µg/ml streptomycin. Cells were used for experiments at passages 3–5 and rendered quiescent in media containing 0.5% FCS for 24 hours before treatment with TGF-β1. Mouse fibroblast (NIH3T3) cells were obtained from American Type Culture Collection and cultured in DMEM with 10% FCS. Supernatants of primary cultured RTECs from LC3+/+ and LC3−/− mice with or without TGF-β1 treatment for 24 hours were collected and used as a source of conditioned medium to stimulate NIH3T3 fibroblasts. The NIH3T3 cells were serum-starved for 24 hours, washed two times with PBS, and then incubated with the conditioned medium for 24 hours. Because it has been previously shown that the half-life of exogenous TGF-β in the culture is about 50 minutes and after 12 hours of incubation, the exogenously added TGF-β (labeled with 125I) is no longer detectable in the culture media,63 the effects could not simply be because of the presence of exogenous TGF-β in the conditioned media.
Immunofluorescence Microscopy
Paraffin-embedded kidney sections (5 μm) were analyzed by immunofluorescence staining using anti–Col-I and anti–α-SMA or anti–TGF-β1 and anti-LC3 as primary antibodies Cy3-conjugated anti-rabbit IgG, Cy3-conjugated anti-mouse IgG, and FITC-conjugated anti-rabbit IgG were used to detect the corresponding primary antibodies. Images were analyzed using a Nikon D-eclipse C1 confocal fluorescence microscope. Exposure settings were unchanged throughout acquisition. Relative area labeled with antibodies to Col-I was assessed in predetermined high-power fields (×200) of the cortex (15 fields), and the percentage of the total tissue area that stained positively for Col-I was determined using ImageJ software (National Institutes of Health, Bethesda, MD).
Western Blot Analysis
Immunoblotting was carried out as previously described.64 Briefly, proteins were extracted with buffer containing 0.05 M Tris-HCl (pH 8.0), 0.15 M NaCl, 5.0 mM EDTA, 1% NP-40, and protease inhibitors [2.0 mM N-ethylmaleimide, 2.0 mM 4-(2-aminoethyl)-benzenesulfonyl fluoride, 1 µg/ml leupeptin, and pepstatin] at 4°C. After centrifugation (14,000 rpm for 20 minutes), supernatant (detergent-soluble), and pellet (detergent-insoluble) fractions were collected and subjected to Western blotting, and target proteins were detected by using LumiGLO (Cell Signaling Technology, Beverly, MA) and exposed to x-ray films.
RT-PCR
RT-PCR was carried out as previously described.64 The PCR primer sets for human TGF-β1 were (forward) 5′-ACATTGACTTCCGCAAGGAC-3′, (reverse) 5′-GTCCAGGCTCCAAATGTAGG-3′. The PCR primer sets for human glyceraldehyde 3-phosphate dehydrogenase were (forward) 5′-CAATGACCCCTTCATTGACC-3′ and (reverse) 5′-TTGATTTTGGAGGGATCTCG-3′.
TUNEL Assay
TUNEL assay was performed to detect apoptotic cell death using the ApopTag Plus Peroxidase In Situ Apoptosis Detection Kit (Chemicon, Temecula, CA) as described in the manufacturer’s instructions. In each kidney, the numbers of tubular and interstitial apoptotic cells were counted separately in 20 nonoverlapping fields observed at ×400 magnification.
Statistical Analyses
Statistical significance of the experimental data from three independent experiments was derived by the t test or ANOVA, and P values<0.05 were considered significant. All of the experiments were performed at least three times. Densitometric analyses for the quantitation of Western blot and RT-PCR data were carried out by using ImageJ software.
Disclosures
None.
Acknowledgments
We thank Drs. C. Brooks and T. Ichimura (Brigham and Women’s Hospital, Harvard Medical School) for technical assistance.
This work was supported by the National Institutes of Health grants R01-HL079904, P01-HL114501, and R01-DK57661 to M.E.C.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
References
- 1.Coresh J, Astor BC, Greene T, Eknoyan G, Levey AS: Prevalence of chronic kidney disease and decreased kidney function in the adult US population: Third National Health and Nutrition Examination Survey. Am J Kidney Dis 41: 1–12, 2003 [DOI] [PubMed] [Google Scholar]
- 2.Lameire N, Jager K, Van Biesen W, de Bacquer D, Vanholder R: Chronic kidney disease: A European perspective. Kidney Int Suppl 68: S30–S38, 2005 [DOI] [PubMed] [Google Scholar]
- 3.Yoshioka K, Tohda M, Takemura T, Akano N, Matsubara K, Ooshima A, Maki S: Distribution of type I collagen in human kidney diseases in comparison with type III collagen. J Pathol 162: 141–148, 1990 [DOI] [PubMed] [Google Scholar]
- 4.Chin BY, Mohsenin A, Li SX, Choi AM, Choi ME: Stimulation of pro-alpha(1)(I) collagen by TGF-beta(1) in mesangial cells: Role of the p38 MAPK pathway. Am J Physiol Renal Physiol 280: F495–F504, 2001 [DOI] [PubMed] [Google Scholar]
- 5.Grgic I, Duffield JS, Humphreys BD: The origin of interstitial myofibroblasts in chronic kidney disease. Pediatr Nephrol 27: 183–193, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li L, Zepeda-Orozco D, Black R, Lin F: Autophagy is a component of epithelial cell fate in obstructive uropathy. Am J Pathol 176: 1767–1778, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Koesters R, Kaissling B, Lehir M, Picard N, Theilig F, Gebhardt R, Glick AB, Hähnel B, Hosser H, Gröne HJ, Kriz W: Tubular overexpression of transforming growth factor-beta1 induces autophagy and fibrosis but not mesenchymal transition of renal epithelial cells. Am J Pathol 177: 632–643, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sato M, Muragaki Y, Saika S, Roberts AB, Ooshima A: Targeted disruption of TGF-beta1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J Clin Invest 112: 1486–1494, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vu TH, Werb Z: Matrix metalloproteinases: Effectors of development and normal physiology. Genes Dev 14: 2123–2133, 2000 [DOI] [PubMed] [Google Scholar]
- 10.Nagase H, Woessner JF, Jr.: Matrix metalloproteinases. J Biol Chem 274: 21491–21494, 1999 [DOI] [PubMed] [Google Scholar]
- 11.Ignotz RA, Massagué J: Transforming growth factor-beta stimulates the expression of fibronectin and collagen and their incorporation into the extracellular matrix. J Biol Chem 261: 4337–4345, 1986 [PubMed] [Google Scholar]
- 12.Annes JP, Munger JS, Rifkin DB: Making sense of latent TGFbeta activation. J Cell Sci 116: 217–224, 2003 [DOI] [PubMed] [Google Scholar]
- 13.Dubois CM, Laprise MH, Blanchette F, Gentry LE, Leduc R: Processing of transforming growth factor beta 1 precursor by human furin convertase. J Biol Chem 270: 10618–10624, 1995 [DOI] [PubMed] [Google Scholar]
- 14.Xie Z, Klionsky DJ: Autophagosome formation: Core machinery and adaptations. Nat Cell Biol 9: 1102–1109, 2007 [DOI] [PubMed] [Google Scholar]
- 15.Ecker N, Mor A, Journo D, Abeliovich H: Induction of autophagic flux by amino acid deprivation is distinct from nitrogen starvation-induced macroautophagy. Autophagy 6: 879–890, 2010 [DOI] [PubMed] [Google Scholar]
- 16.Liang J, Shao SH, Xu ZX, Hennessy B, Ding Z, Larrea M, Kondo S, Dumont DJ, Gutterman JU, Walker CL, Slingerland JM, Mills GB: The energy sensing LKB1-AMPK pathway regulates p27(kip1) phosphorylation mediating the decision to enter autophagy or apoptosis. Nat Cell Biol 9: 218–224, 2007 [DOI] [PubMed] [Google Scholar]
- 17.Meijer AJ, Codogno P: Autophagy: Regulation by energy sensing. Curr Biol 21: R227–R229, 2011 [DOI] [PubMed] [Google Scholar]
- 18.Nijholt DA, de Graaf TR, van Haastert ES, Oliveira AO, Berkers CR, Zwart R, Ovaa H, Baas F, Hoozemans JJ, Scheper W: Endoplasmic reticulum stress activates autophagy but not the proteasome in neuronal cells: Implications for Alzheimer’s disease. Cell Death Differ 18: 1071–1081, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Majmundar AJ, Wong WJ, Simon MC: Hypoxia-inducible factors and the response to hypoxic stress. Mol Cell 40: 294–309, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Scherz-Shouval R, Shvets E, Fass E, Shorer H, Gil L, Elazar Z: Reactive oxygen species are essential for autophagy and specifically regulate the activity of Atg4. EMBO J 26: 1749–1760, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Levine B, Kroemer G: Autophagy in the pathogenesis of disease. Cell 132: 27–42, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mizushima N: Physiological functions of autophagy. Curr Top Microbiol Immunol 335: 71–84, 2009 [DOI] [PubMed] [Google Scholar]
- 23.Suzuki K, Kirisako T, Kamada Y, Mizushima N, Noda T, Ohsumi Y: The pre-autophagosomal structure organized by concerted functions of APG genes is essential for autophagosome formation. EMBO J 20: 5971–5981, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yue Z, Jin S, Yang C, Levine AJ, Heintz N: Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci U S A 100: 15077–15082, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Qu X, Yu J, Bhagat G, Furuya N, Hibshoosh H, Troxel A, Rosen J, Eskelinen EL, Mizushima N, Ohsumi Y, Cattoretti G, Levine B: Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J Clin Invest 112: 1809–1820, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Arsov I, Li X, Matthews G, Coradin J, Hartmann B, Simon AK, Sealfon SC, Yue Z: BAC-mediated transgenic expression of fluorescent autophagic protein Beclin 1 reveals a role for Beclin 1 in lymphocyte development. Cell Death Differ 15: 1385–1395, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pickford F, Masliah E, Britschgi M, Lucin K, Narasimhan R, Jaeger PA, Small S, Spencer B, Rockenstein E, Levine B, Wyss-Coray T: The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid beta accumulation in mice. J Clin Invest 118: 2190–2199, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kim SI, Na HJ, Ding Y, Wang Z, Lee SJ, Choi ME: Autophagy promotes intracellular degradation of type I collagen induced by transforming growth factor (TGF)-β1. J Biol Chem 287: 11677–11688, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T: LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19: 5720–5728, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hartleben B, Gödel M, Meyer-Schwesinger C, Liu S, Ulrich T, Köbler S, Wiech T, Grahammer F, Arnold SJ, Lindenmeyer MT, Cohen CD, Pavenstädt H, Kerjaschki D, Mizushima N, Shaw AS, Walz G, Huber TB: Autophagy influences glomerular disease susceptibility and maintains podocyte homeostasis in aging mice. J Clin Invest 120: 1084–1096, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kang R, Zeh HJ, Lotze MT, Tang D: The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18: 571–580, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chevalier RL: Pathogenesis of renal injury in obstructive uropathy. Curr Opin Pediatr 18: 153–160, 2006 [DOI] [PubMed] [Google Scholar]
- 33.Haspel J, Shaik RS, Ifedigbo E, Nakahira K, Dolinay T, Englert JA, Choi AM: Characterization of macroautophagic flux in vivo using a leupeptin-based assay. Autophagy 7: 629–642, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Miyazono K, Olofsson A, Colosetti P, Heldin CH: A role of the latent TGF-beta 1-binding protein in the assembly and secretion of TGF-beta 1. EMBO J 10: 1091–1101, 1991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Geng J, Klionsky DJ: The Golgi as a potential membrane source for autophagy. Autophagy 6: 950–951, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mizushima N, Yoshimori T, Levine B: Methods in mammalian autophagy research. Cell 140: 313–326, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dockrell ME, Phanish MK, Hendry BM: Tgf-beta auto-induction and connective tissue growth factor expression in human renal tubule epithelial cells requires N-ras. Nephron, Exp Nephrol 112: e71–e79, 2009 [DOI] [PubMed] [Google Scholar]
- 38.Choi ME, Kim EG, Huang Q, Ballermann BJ: Rat mesangial cell hypertrophy in response to transforming growth factor-beta 1. Kidney Int 44: 948–958, 1993 [DOI] [PubMed] [Google Scholar]
- 39.Yamamoto A, Tagawa Y, Yoshimori T, Moriyama Y, Masaki R, Tashiro Y: Bafilomycin A1 prevents maturation of autophagic vacuoles by inhibiting fusion between autophagosomes and lysosomes in rat hepatoma cell line, H-4-II-E cells. Cell Struct Funct 23: 33–42, 1998 [DOI] [PubMed] [Google Scholar]
- 40.Klionsky DJ, Elazar Z, Seglen PO, Rubinsztein DC: Does bafilomycin A1 block the fusion of autophagosomes with lysosomes? Autophagy 4: 849–850, 2008 [DOI] [PubMed] [Google Scholar]
- 41.Mizushima N, Levine B, Cuervo AM, Klionsky DJ: Autophagy fights disease through cellular self-digestion. Nature 451: 1069–1075, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Choi AM, Ryter SW, Levine B: Autophagy in human health and disease. N Engl J Med 368: 1845–1846, 2013 [DOI] [PubMed] [Google Scholar]
- 43.Del Principe D, Lista P, Malorni W, Giammarioli AM: Fibroblast autophagy in fibrotic disorders. J Pathol 229: 208–220, 2013 [DOI] [PubMed] [Google Scholar]
- 44.Kim WY, Nam SA, Song HC, Ko JS, Park SH, Kim HL, Choi EJ, Kim YS, Kim J, Kim YK: The role of autophagy in unilateral ureteral obstruction rat model. Nephrology (Carlton) 17: 148–159, 2012 [DOI] [PubMed] [Google Scholar]
- 45.Djavaheri-Mergny M, Maiuri MC, Kroemer G: Cross talk between apoptosis and autophagy by caspase-mediated cleavage of Beclin 1. Oncogene 29: 1717–1719, 2010 [DOI] [PubMed] [Google Scholar]
- 46.Wynn TA: Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J Clin Invest 117: 524–529, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Iwano M, Plieth D, Danoff TM, Xue C, Okada H, Neilson EG: Evidence that fibroblasts derive from epithelium during tissue fibrosis. J Clin Invest 110: 341–350, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kalluri R, Neilson EG: Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest 112: 1776–1784, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Burns WC, Kantharidis P, Thomas MC: The role of tubular epithelial-mesenchymal transition in progressive kidney disease. Cells Tissues Organs 185: 222–231, 2007 [DOI] [PubMed] [Google Scholar]
- 50.Zeisberg M, Neilson EG: Mechanisms of tubulointerstitial fibrosis. J Am Soc Nephrol 21: 1819–1834, 2010 [DOI] [PubMed] [Google Scholar]
- 51.Geng H, Lan R, Singha PK, Gilchrist A, Weinreb PH, Violette SM, Weinberg JM, Saikumar P, Venkatachalam MA: Lysophosphatidic acid increases proximal tubule cell secretion of profibrotic cytokines PDGF-B and CTGF through LPA2- and Gαq-mediated Rho and αvβ6 integrin-dependent activation of TGF-β. Am J Pathol 181: 1236–1249, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Harris J, Hartman M, Roche C, Zeng SG, O’Shea A, Sharp FA, Lambe EM, Creagh EM, Golenbock DT, Tschopp J, Kornfeld H, Fitzgerald KA, Lavelle EC: Autophagy controls IL-1beta secretion by targeting pro-IL-1beta for degradation. J Biol Chem 286: 9587–9597, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Shi CS, Shenderov K, Huang NN, Kabat J, Abu-Asab M, Fitzgerald KA, Sher A, Kehrl JH: Activation of autophagy by inflammatory signals limits IL-1β production by targeting ubiquitinated inflammasomes for destruction. Nat Immunol 13: 255–263, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Saitoh T, Fujita N, Jang MH, Uematsu S, Yang BG, Satoh T, Omori H, Noda T, Yamamoto N, Komatsu M, Tanaka K, Kawai T, Tsujimura T, Takeuchi O, Yoshimori T, Akira S: Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1beta production. Nature 456: 264–268, 2008 [DOI] [PubMed] [Google Scholar]
- 55.Crişan TO, Plantinga TS, van de Veerdonk FL, Farcaş MF, Stoffels M, Kullberg BJ, van der Meer JW, Joosten LA, Netea MG: Inflammasome-independent modulation of cytokine response by autophagy in human cells. PLoS ONE 6: e18666, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schroder K, Tschopp J: The inflammasomes. Cell 140: 821–832, 2010 [DOI] [PubMed] [Google Scholar]
- 57.Nakahira K, Haspel JA, Rathinam VA, Lee SJ, Dolinay T, Lam HC, Englert JA, Rabinovitch M, Cernadas M, Kim HP, Fitzgerald KA, Ryter SW, Choi AM: Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol 12: 222–230, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Fujigaki Y, Muranaka Y, Sun D, Goto T, Zhou H, Sakakima M, Fukasawa H, Yonemura K, Yamamoto T, Hishida A: Transient myofibroblast differentiation of interstitial fibroblastic cells relevant to tubular dilatation in uranyl acetate-induced acute renal failure in rats. Virchows Arch 446: 164–176, 2005 [DOI] [PubMed] [Google Scholar]
- 59.Suzuki T, Kimura M, Asano M, Fujigaki Y, Hishida A: Role of atrophic tubules in development of interstitial fibrosis in microembolism-induced renal failure in rat. Am J Pathol 158: 75–85, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Cann GM, Guignabert C, Ying L, Deshpande N, Bekker JM, Wang L, Zhou B, Rabinovitch M: Developmental expression of LC3alpha and beta: Absence of fibronectin or autophagy phenotype in LC3beta knockout mice. Dev Dyn 237: 187–195, 2008 [DOI] [PubMed] [Google Scholar]
- 61.Liang XH, Jackson S, Seaman M, Brown K, Kempkes B, Hibshoosh H, Levine B: Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature 402: 672–676, 1999 [DOI] [PubMed] [Google Scholar]
- 62.Wang L, Lee JY, Kwak JH, He Y, Kim SI, Choi ME: Protective effects of low-dose carbon monoxide against renal fibrosis induced by unilateral ureteral obstruction. Am J Physiol Renal Physiol 294: 508–517, 2008 [DOI] [PubMed] [Google Scholar]
- 63.Rollins BJ, O’Connell TM, Bennett G, Burton LE, Stiles CD, Rheinwald JG: Environment-dependent growth inhibition of human epidermal keratinocytes by recombinant human transforming growth factor-beta. J Cell Physiol 139: 455–462, 1989 [DOI] [PubMed] [Google Scholar]
- 64.Ding Y, Kim JK, Kim SI, Na HJ, Jun SY, Lee SJ, Choi ME: TGF-beta1 protects against mesangial cell apoptosis via induction of autophagy. J Biol Chem 285: 37909–37919, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]