Abstract
Feingold syndrome (FGLDS1) is an autosomal dominant disorder caused by mutations in the MYCN oncogene on the short arm of chromosome 2 (2p24.1). It is characterised by microcephaly, digital abnormalities, oesophageal and duodenal atresias, and often learning disability or mental retardation. In 2011, individuals sharing the skeletal abnormalities of FGLDS1 but lacking mutations in MYCN, were found to harbour hemizygous deletions of the MIR17HG gene on chromosome 13q31.3. These individuals share many of the characteristics of FGLDS1 except for gastrointestinal atresia. The condition was termed Feingold syndrome type 2 (FGLDS2). We describe the presentation and management of a fourth known case of FGLDS2 in an 18-year-old girl with microcephaly, short stature, mildly dysmorphic features, digital malformations and significant cognitive and psychiatric symptoms. Comparative genomic hybridisation array testing confirmed a 7.4 Mb microdeletion in chromosome region 13q31.1q.31.3 corresponding to the MIR17HG gene.
Background
Feingold syndrome type 1 (FGLDS1) is an autosomal dominant syndrome characterised by microcephaly, short stature, brachymesophalangy, brachysyndactyly of the toes, oesophageal and duodenal atresias and intellectual compromise.1 2 It is caused by mutations in or deletions of MYCN (2p24.1). MYCN encodes N-myc, a transcription factor highly expressed in the fetal brain and crucial for normal brain development.3
Feingold syndrome type 2 (FGLDS2) was described by de Pontual et al.4 In a study of 10 participants with skeletal abnormalities consistent with FGLDS1 but lacking mutations in MYCN, two individuals were found to have germline hemizygous microdeletions in chromosome region 13q31.3. Animal models suggested that MIR17HG was the gene responsible for the Feingold syndrome phenotype.4
A search of the Database of Chromosomal Imbalance and Phenotype in Humans Using Ensembl Resources (DECIPHER)5 identified a third participant (participant ID: 248412) with a 13q31.3 deletion encompassing the entire MIR17HG gene. This individual did not carry the diagnosis of Feingold syndrome but had phenotypic characteristics consistent with the diagnosis such as microcephaly, intellectual deficits, brachymesophalangy and toe syndactyly. Here, we describe a fourth case of FGLDS2 with a deletion encompassing MIR17HG including the clinical presentation and management of psychiatric symptoms. To our knowledge, this is the first detailed description of an individual with FGLDS2 in the literature.
Case presentation
The proband is an 18-year-old girl with microcephaly, short stature, mild cognitive delay and numerous psychological and behavioural difficulties. She was born at 36 weeks gestational age, weighing seven pounds, three ounces. Labour was precipitous, but delivery was uneventful. She was described as dysmorphic, but chromosomal analysis did not reveal any abnormalities. She was discharged from hospital at 5 days of age.
She began walking at 1 year, but had delays in talking and toilet-training. She always struggled academically, with increasing difficulties as she progressed through school. She was functioning at an IQ of 70–75. At age 12, she began to develop anxiety and an urge to pick at her skin. Her neurological examination was normal. The patient's immediate family appears to be healthy.
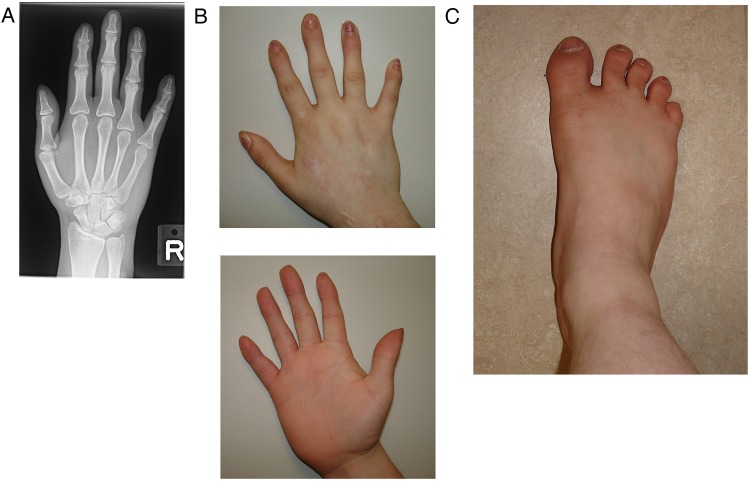
At age 18 years, her height was 145.5 cm (<3rd centile, 50th centile for 11 years) and weight was 55.1 kg (25–50th centile). Head circumference was 51.5 cm (<3rd centile, 50th centile for 7 years). Inner canthal distance was 2.8 cm (25th centile), and outer canthal distance was 7.5 cm (50th centile for 3 years), with a palpebral fissure length of 2.4 cm (50th centile for 2 years). Hand length was 14.5 cm (50th centile for 8–1/2 years) and middle finger length was 6.0 cm (50th centile for 7–1/2 years). Ear length was 6.0 cm (50th centile). Foot length was 20 cm (50th centile for 8 years). She had minimal dysmorphic features. Palpebral fissures were short and up slanting. Eyes appeared deep set. Nose was long and straight with narrow nares. Ears were normal in position, but had significant swelling secondary to self-injury. Philtrum and lips were normal. Palate was intact and teeth were normally positioned. The left lower lateral incisor was congenitally absent. Hands were small with short middle phalanges. There was clinodactyly of the second and fifth digits bilaterally. Fingers were spindle shaped with tapered distal phalanges. The fifth metacarpals were short bilaterally with proximally positioned fifth digits (figure 1A, B). The distal interphalangeal creases on the second digits were decreased. Feet were short and broad with bilateral hallux valgus and short, broad halluces. There was bilateral 2–3 syndactyly of the toes. Halluces were also short and broad (figure 1C). Deep tendon reflexes were 3+ at the biceps, brachioradialis and knees, and 2+ at the triceps and ankle. A brain MRI did not reveal any abnormalities. Genetic testing revealed a microdeletion mapping to chromosome 13q31.1.
Figure 1.
Radiograph (A) and photograph (B) of right hand of proband showing skeletal deformities in digits. Photograph (C) right foot of proband showing deformities in halluces.
Investigations
DNA was extracted from specimens of whole blood using QIAcube (Qiagen, Hilden, Germany). Array comparative genomic hybridisation (CGH) was performed using the 180k ISCA Cytochip (Bluegnome, Cambridge, UK) and high-resolution microarray scanner (Agilent Technologies, Santa Clara, USA). Copy number variants were analysed using BlueFuse (Bluegnome) and further filtered and categorised using Cartagenia Software (Leuven, Belgium). A 7.44 Mb deletion was detected with breakpoints estimated to be between chr13:85 175 188–92 617 860 (GRCh37/hg19). The deletion was confirmed to be de novo using BAC BlueFISH clone RP11–275J18 (Bluegnome, Cambridge), which maps to 13q31.1 nucleotide position chr13:89 679 232–89 844 740 (GRCh37/hg19 assembly). The deletion encompasses the OMIM morbid gene MIR17HG (OMIM #614326), in addition to SLITRK6, SLITRK5, MIR17, MIR18A, MIR19A, MIR20A, MIR19B1, MIR92A1 and GPC5.
The diagnosis of FGLDS2 was made based on chromosomal analysis and clinical features. Table 1 summarises the patient's clinical features that are consistent with FGLDS2 as described by de Pontual et al,4 and aspects of her presentation that are not (yet) known to be a part of the syndrome.
Table 1.
Clinical presentation of proband
| Signs and symptoms consistent with Feingold syndrome type 2 | Signs and symptoms not described in Feingold syndrome type 2 |
|---|---|
| Microcephaly | Obsessions |
| Short stature | Anxiety |
| Intellectual disability | Mood dysregulation |
| Skeletal abnormalities | Disruptive behaviour |
Treatment
As the patient entered adolescence, she began to struggle with severe anxiety and obsessive thoughts. She was tried on several SSRIs, including escitalopram, fluoxetine, fluvoxamine and sertraline, that appeared to make her more agitated. Antipsychotic medication, including risperidone, paliperidone, quetiapine and chlorpromazine, would either make her more agitated, or oversedate her. The violent outbursts and mood dysregulation would rebound when the effect of the neuroleptic wore off. Benzodiazepines including clonazepam and lorazepam were useful for sleep but did not improve mood or behaviour.
Obsessive thoughts and behavioural difficulties worsened over time, necessitating multiple visits to the emergency department and admissions. The outbursts became more aggressive and self-injurious behaviours became more prominent (ie, biting, scratching and punching herself on the side of the head). She would often run away and caused a great deal of property damage at home.
At age 17, there was a suicide attempt by drug overdose. At the time of admission, she presented as a petite female who was significantly dishevelled and malodorous. Her mood was low. She was extremely labile and had frequent behavioural outbursts. Chlorpromazine had been used for sleep and agitation on admission, but was felt to cause more agitation, therefore was discontinued, while benzodiazepines and zopiclone were introduced for sleep. Gabapentin was started at a dose of 100 mg three times daily and later increased to 200 mg three times daily. There was a significant calming effect with improvement in volatility and agitation and decrease in self-injurious behaviour, but her mood remained low.
Eventually, aripiprazole was started at 5 mg every morning. Within a few days, significant changes were observed. Her anxiety, mood, and ability to tolerate frustration improved, such that she was observed smiling, joking and laughing with staff and copatients. Her self-injurious behaviours stopped. Adding to the evidence that aripiprazole was effective in this patient, there was a significant decline in her mood and increase in her lability during a 3-day period when she refused to take the medication, and a more subtle worsening of her psychiatric symptoms during a week when the dose was decreased. On both occasions, reintroducing the medication caused a rapid improvement within days. A gradual increase to 15 mg per day was well-tolerated and further improved her mood and behaviours.
Outcome and follow-up
At the time of submission of this manuscript, the patient remains admitted to the inpatient psychiatric unit awaiting placement. Her mood has consistently remained stable and her behaviours are significantly improved.
Discussion
Using array CGH, a fourth case of Feingold syndrome type 2 (FGLDS2) has been identified in a patient with 7.4 Mb deletion of chromosome region 13q31.1q.31.3. Features in our patient include microcephaly, short stature, digital abnormalities with short digits and clinodactyly of the second and fifth digits bilaterally and cognitive delay. We have described the psychiatric symptomatology in detail. Unlike cases of Feingold syndrome type 1, there is no history of gastrointestinal atresia. In this patient, although the deletion encompasses numerous genes, the loss of MIR17HG and GPC5 is consistent with other cases of FGLDS2. Neither parent harboured this deletion indicating that that was a de novo germline mutation.
Unlike the previously described individuals with MIR17HG deletions, the patient described here does not have hypoplastic thumbs. Other than mental retardation (ranging from mild to severe), the mental and behavioural features of the three previously described individuals with FGLDS2 is not known. All of the three other cases have had a deletion of MIR17HG and at least the first exon of the GPC5 gene. There is evidence from animal models to support the suggestion that the deletion of MIR17HG results in the skeletal manifestations of FGLDS2.4 However, it is not known whether the deletion of MIR17HG or any of the other genes encompassed in the deletion is contributing to our patient's psychiatric manifestations.
It is also difficult to determine how the patient's unique response to medication related to her genetic condition. We describe how the patient's psychiatric symptoms worsened with serotonergic medication (SSRIs) and most antipsychotic medication. Aripiprazole, an antipsychotic with a unique mechanism of action was well-tolerated and effective. Unlike most antipsychotics which are antagonists of dopamine receptors, aripiprazole acts either as a partial agonist at D2 and D3 receptors or through functional selectivity.6 Indeed, aripiprazole has been shown to be an effective medication in patients with developmental brain disorders and other organic brain conditions. A recent systematic review of the use aripiprazole for problematic behaviour in autistic spectrum disorders, intellectual disability and/or developmental delay demonstrated efficacy of the medication.7 It has also been shown to be effective in treating psychosis and aggression in dementia8–10 as well as behavioural problems and irritability in Fragile X syndrome.11 The other medications that the patient tolerated well were the benzodiazepines, zopiclone, and gabapentin, all of which are GABA-ergic medications. Gabapentin is a GABA analogue antiepileptic medication also used for neuropathic pain and many off label conditions such as anxiety and aggression.12
In summary, we describe the fourth known case of MIR17HG mutation associated with FGLDS2. Given the paucity of clinical information associated with this condition in the literature, we describe in detail the physical features and psychiatric symptoms of this patient, as well as the medical management of these symptoms. The possibility of an underlying genomic imbalance needs to be considered and evaluated through microarray CGH.
Learning points.
Consider the diagnosis of Feingold syndrome type 2 in patients with microcephaly, short stature, short digits, clinodactyly, development delay and psychiatric symptoms including depression, anxiety and behavioural difficulties.
Consider the use of aripiprazole in the treatment of mood disorders and behavioural difficulties in patients with Feingold syndrome type 2 or other developmental delay as there is a growing body of evidence supporting its use in patients with organic brain conditions.
Gabapentin may be a useful medication in treating agitation and behavioural difficulties in individuals with developmental delay.
Acknowledgments
The authors would like to thank the family for their involvement and willingness to participate in this case report. No funding source was involved in the collection of data or preparation of this manuscript.
Footnotes
Contributors: HG conceived the project and contributed to drafting and revising the manuscript. VMS and MS performed the genomic analysis, interpreted the data, contributed to drafting and revising the manuscript. PAM contributed to drafting and revising the manuscript.
Competing interests: None.
Patient consent: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Celli J, van Bokhoven H, Brunner HG. Feingold syndrome: clinical review and genetic mapping. Am J Med Genet A 2003;122A:294–300. [DOI] [PubMed] [Google Scholar]
- 2.Feingold M, Hall BD, Lacassie Y et al. Syndrome of microcephaly, facial and hand abnormalities, tracheoesophageal fistula, duodenal atresia, and developmental delay. Am J Med Genet 1997;69:245–9. [PubMed] [Google Scholar]
- 3.Knoepfler PS, Cheng PF, Eisenman RN. N-myc is essential during neurogenesis for the rapid expansion of progenitor cell populations and the inhibition of neuronal differentiation. Genes Dev 2002;16:2699–712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.de Pontual L, Yao E, Callier P et al. Germline deletion of the miR-17 approximately 92 cluster causes skeletal and growth defects in humans. Nat Genet 2011;43:1026–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Firth HV, Richards SM, Bevan AP et al. DECIPHER: database of chromosomal imbalance and phenotype in humans using ensembl resources. Am J Hum Genet 2009;84:524–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shapiro DA, Renock S, Arrington E et al. Aripiprazole, a novel atypical antipsychotic drug with a unique and robust pharmacology. Neuropsychopharmacology 2003;28:1400–11. [DOI] [PubMed] [Google Scholar]
- 7.Deb S, Farmah BK, Arshad E et al. The effectiveness of aripiprazole in the management of problem behaviour in people with intellectual disabilities, developmental disabilities and/or autistic spectrum disorder—a systematic review. Res Dev Disabil 2014;35:711–25. [DOI] [PubMed] [Google Scholar]
- 8.Streim JE, Porsteinsson AP, Breder CD et al. A randomized, double-blind, placebo-controlled study of aripiprazole for the treatment of psychosis in nursing home patients with Alzheimer disease. Am J Geriatr Psychiatry 2008;16:537–50. [DOI] [PubMed] [Google Scholar]
- 9.De Deyn P, Jeste DV, Swanink R et al. Aripiprazole for the treatment of psychosis in patients with Alzheimer's disease: a randomized, placebo-controlled study. J Clin Psychopharmacol 2005;25:463–7. [DOI] [PubMed] [Google Scholar]
- 10.Mintzer JE, Tune LE, Breder CD et al. Aripiprazole for the treatment of psychoses in institutionalized patients with Alzheimer dementia: a multicenter, randomized, double-blind, placebo-controlled assessment of three fixed doses. Am J Geriatr Psychiatry 2007;15:918–31. [DOI] [PubMed] [Google Scholar]
- 11.Erickson CA, Stigler KA, Posey DJ et al. Aripiprazole in autism spectrum disorders and fragile X syndrome. Neurotherapeutics 2010;7:258–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Letterman L, Markowitz JS. Gabapentin: a review of published experience in the treatment of bipolar disorder and other psychiatric conditions. Pharmacotherapy 1999;19:565–72. [DOI] [PubMed] [Google Scholar]