Abstract
Taurine (2-aminoethanesulfonic acid) is widely distributed in animal tissues and has diverse pharmacological effects. However, the role of taurine in modulating smooth muscle contractility is still controversial. We propose that taurine (5-80 mM) can exert bidirectional modulation on the contractility of isolated rat jejunal segments. Different low and high contractile states were induced in isolated jejunal segments of rats to observe the effects of taurine and the associated mechanisms. Taurine induced stimulatory effects on the contractility of isolated rat jejunal segments at 3 different low contractile states, and inhibitory effects at 3 different high contractile states. Bidirectional modulation was not observed in the presence of verapamil or tetrodotoxin, suggesting that taurine-induced bidirectional modulation is Ca2+ dependent and requires the presence of the enteric nervous system. The stimulatory effects of taurine on the contractility of isolated jejunal segments was blocked by atropine but not by diphenhydramine or by cimetidine, suggesting that muscarinic-linked activation was involved in the stimulatory effects when isolated jejunal segments were in a low contractile state. The inhibitory effects of taurine on the contractility of isolated jejunal segments were blocked by propranolol and L-NG-nitroarginine but not by phentolamine, suggesting that adrenergic β receptors and a nitric oxide relaxing mechanism were involved when isolated jejunal segments were in high contractile states. No bidirectional effects of taurine on myosin phosphorylation were observed. The contractile states of jejunal segments determine taurine-induced stimulatory or inhibitory effects, which are associated with muscarinic receptors and adrenergic β receptors, and a nitric oxide associated relaxing mechanism.
Keywords: Taurine, Bidirectional modulation, Enteric nervous system, Calcium dependent, Homeostasis, Intestinal motility
Introduction
Taurine (2-aminoethanesulfonic acid) is widely distributed in animal tissues, and plays an important role in diverse physiological processes including membrane stabilization (1), osmoregulation (2), regulation of cell apoptosis (3), antioxidation (4,5), modulation of neuronal excitability (6,7), and maintenance of Ca2+ homeostasis (8).
The function of taurine in the cardiovascular system has been well studied (9-11). Oral supplementation of taurine is shown to reduce blood pressure and improve cardiac function in hypertensive subjects (12-14). Ex vivo studies have shown that contractile responses to norepinephrine (NE) and KCl in aortic rings are attenuated both in taurine-treated normal rats and diabetic rats compared with controls (15,16); NE-, KCl-, and adenosine-receptor-agonist-induced hypercontractility of the aorta are enhanced in taurine-depleted rats compared with the effects in control animals (17,18). Although most studies show that taurine induces inhibitory effects in precontracted vessels, some reports indicate that taurine at concentrations of 20-60 mM inhibits phenylephrine-induced contraction in normal arteries of rats without affecting the basal tone of the arteries (19). However, taurine further enhances the NE- or KCl-induced contraction of arteries in the insulin-resistant rat (20). The studies cited above indicate that the effects of taurine on vascular smooth muscle are still controversial and the associated mechanisms remain unclear.
The divergent effects of taurine on vascular smooth muscle attracted our interest. We proposed a bidirectional modulation of taurine on smooth muscle and noted that the effects of taurine on intestinal smooth muscle contraction are rarely reported. It is known that the contraction of intestinal smooth muscle is modulated by the enteric nervous system (ENS), which can control functions of the intestine even when it is completely separated from the central nervous system (CNS) (21). To characterize the effects of taurine on intestinal contractility and reveal the possible mechanism, isolated jejunal segments and 3 different pairs of low and high contractile states of the segments were established by modification of ionic concentrations or by inhibitory and stimulatory neurotransmitters.
Material and Methods
Material
Ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid (EGTA) was obtained from Wako (Japan). Calmodulin (CaM) was generously provided by Professor K. Kohama, Gumma University School of Medicine, Japan. Tetrodotoxin (TTX) was obtained from Aladdin Chemistry Co., Ltd. (China). Taurine and other reagents were purchased from Sigma (USA).
Determination of jejunal contractility
All rats were treated according to the Guidelines for the Care and Use of Laboratory Animals of Dalian Medical University, and all experimental procedures were carried out in accordance with the Declaration of Helsinki. Healthy male Sprague-Dawley rats (certificate No. SCXK 2008-0002), weighing 180-220 g, were used for measurement of jejunal contractility, Rats were fasted for 24 h but allowed water before experiments.
Rats were sacrificed by cervical dislocation and the jejunum was carefully removed and kept in Krebs buffer (118 mM NaCl, 4.7 mM KCl, 1.2 mM KH2PO4, 1.2 mM MgSO4, 4.2 mM NaHCO3, 2.5 mM CaCl2, 10 mM glucose, pH 7.4). The isolated jejunum was rinsed, clipped into segments (about 12 mm long), and the mesenteric border was removed. The segments were suspended in longitudinal orientation in 20-mL organ baths of warm (37°C), aerated Krebs buffer. The distal end of each segment was tied to an organ holder, and the proximal end was secured with a silk thread to an isometric force transducer. A 1-g/cm resting tension was applied to each segment, which was equilibrated for about 1 h. Rhythmic spontaneous contractions developed in all preparations. The contractile responses of the isolated jejunal segment were recorded with a BL-420 physiological recording system (Chengdu Taimeng Software Co., Ltd., China).
The contractility of isolated jejunal segments in Krebs buffer was selected as the normal contractile state (NCS). Modified low-Ca2+ (1.25 mM), low-K+ (2.5 mM), or adrenaline (5 μM) Krebs buffers were selected as low contractile states (LCS). Modified high-Ca2+ (5.0 mM), high-K+ (10.0 mM), or carbachol (CBC, 1 μM) Krebs buffers were selected as high contractile states (HCS). To study the mechanisms of taurine-induced bidirectional modulation, the HCS induced by high Ca2+ (5.0 mM) Krebs buffer and LCS induced by low Ca2+ (1.25 mM) Krebs buffer were selected as the representative HCS and representative LCS, respectively. A sodium channel blocker (TTX, 0.3 μM for 3 min); a voltage-dependent L-type calcium channel blocker (verapamil, 1 μM for 3 min); an adrenergic α-receptor antagonist (phentolamine, 1 μM for 3 min); an adrenergic β-receptor antagonist (propranolol, 1 μM for 3 min); a cholinergic M receptor antagonist (atropine, 1 μM for 3 min); a histamine H1-receptor antagonist (diphenhydramine, 10 μM for 3 min); a histamine H2-receptor antagonist (cimetidine, 10 μM for 3 min), and a nitric oxide (NO) synthase inhibitor [L-NG-nitroarginine (L-NNA), 300 μM for 3 min] were used in the assay. The contractile amplitude of isolated jejunal segments is reported as a percentage of the contractile amplitude in the NCS. The contractile amplitude at NCS was set to a relative value of 100%.
Measurement of myosin phosphorylation
Myosin and myosin light chain kinase (MLCK) used in the assay were purified from chicken gizzard smooth muscle as described previously (22). Myosin phosphorylation was carried out in a 20 mM Tris-HCl, pH 7.4, buffer containing 1 mM dithiothreitol (DTT), 5 mM MgCl2, 60 mM KCl, 0.1 mM CaCl2, 0.6 μM calmodulin, 4 μM myosin, and 2 mM ATP with or without taurine at 25°C for 20 min. MLCK (2.0 and 0.02 μM) was used to phosphorylate MLC20, exerting a high-extent and low-extent of phosphorylation, respectively. The extent of 20-kDa myosin light chain (MLC20) phosphorylation was measured by 10% glycerol polyacrylamide gel electrophoresis (PAGE) (23,24). A Gel-Pro Analyzer (Media Cybernetics, USA) was used to scan the density and size of phosphorylated MLC20. The extent of phosphorylated MLC20 is reported as the percentage of phosphorylated MLC20 in total MLC20. The extent of full mono-phosphorylation was set at 100% (control).
Statistical analysis
All data are reported as means±SE. Statistical significance was tested by one-way analysis of variance, followed by the Dunnett multiple comparisons test, using the SPSS software 13.0 (SPSS Inc., USA). Statistical significance was defined as P<0.05.
Results
Effects of taurine on the contractility of isolated jejunal segments
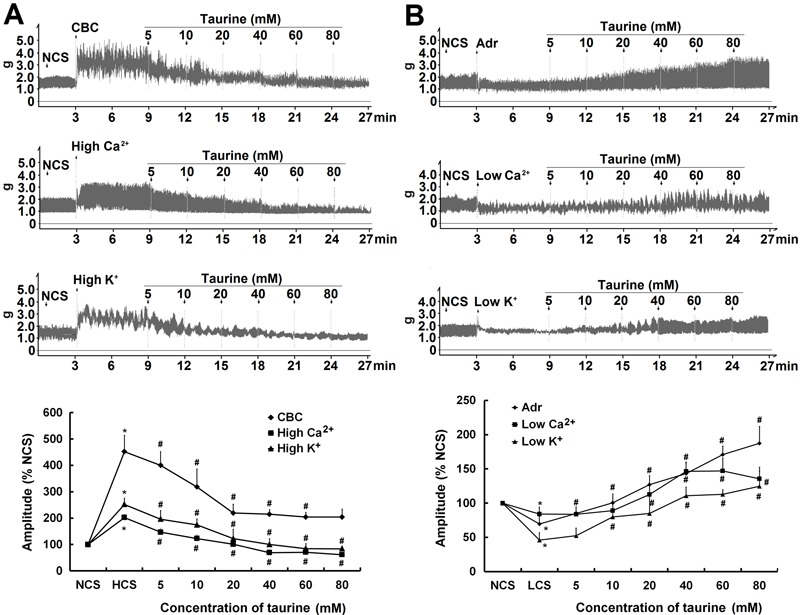
Taurine (5-80 mM) enhanced the contractile amplitude of isolated jejunal segments in a concentration-dependent manner (Figure 1, Figure S1). The contractile amplitude in both HCS and LCS was statistically different from that of NCS (Figure 2, Figure S2). Taurine (5-80 mM) significantly enhanced the contractile amplitude of jejunal segments in all 3 LCS (P<0.05), and significantly decreased the amplitude in all 3 HCS (P<0.05, Figure 2, Figure S2). These data indicated that taurine induced bidirectional modulation of the spontaneous contractility of isolated jejunal segments.
Figure 1. Effects of taurine on the contractile amplitude of isolated jejunal segments. Representative traces and statistical analysis (n=6) of taurine-induced effects on the contractile amplitude of isolated jejunal segments in the normal contractile state (NCS, control). The contractile amplitude in NCS is set to 100%; the effects of taurine on the contractile amplitude are the relative values compared with NCS. *P<0.05 vs the contractile amplitude in NCS before taurine administration (one-way ANOVA).
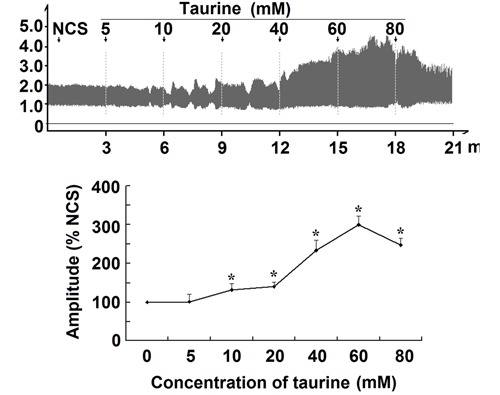
Figure 2. Taurine-induced bidirectional modulations on the contractile amplitude of isolated jejunal segments. A, Representative traces and statistical analysis (n=6) of the inhibitory effects of taurine on the contractile amplitude of isolated jejunal segments in 3 high contractile states (HCS). B, Representative traces and statistical analysis (n=6) of the stimulatory effects of taurine on the contractile amplitude of isolated jejunal segments in 3 low contractile states (LCS). The contractile amplitude in the normal contractile state (NCS, control) is set to 100%; the contractile amplitude in HCS and LCS are the relative values compared with NCS. CBC: carbachol; Adr: adrenaline. *P<0.05 vs the contractile amplitude in NCS; #P<0.05 vs the contractile amplitude in LCS or HCS before taurine administration (one-way ANOVA).
Underlying mechanism of taurine-induced bidirectional modulation
In the presence of TTX, neither an inhibitory effect of taurine (10-60 mM) on the contractile amplitude of isolated jejunal segments in the HCS induced by high Ca2+ (5.0 mM) Krebs buffer nor a stimulatory effect on the contractile amplitude in LCS induced by low Ca2+ (1.25 mM) Krebs buffer were observed (Figure 3). These data showed that TTX abolished bidirectional modulation of taurine on jejunal contractility.
Figure 3. Effects of taurine on the contractile amplitude of isolated jejunal segments pretreated with tetrodotoxin (TTX). Representative traces and statistical analysis (n=6) of taurine-induced effects on the contractile amplitude of isolated jejunal segments in the normal contractile state (NCS, control), high contractile state (HCS) induced by high Ca2+ (5.0 mM) Krebs buffer and low contractile state (LCS) induced by low Ca2+ (1.25 mM) Krebs buffer pretreated with TTX (0.3 μM). The contractile amplitude in NCS is set to 100%; other data are the relative values compared with NCS. CS: contractile state.

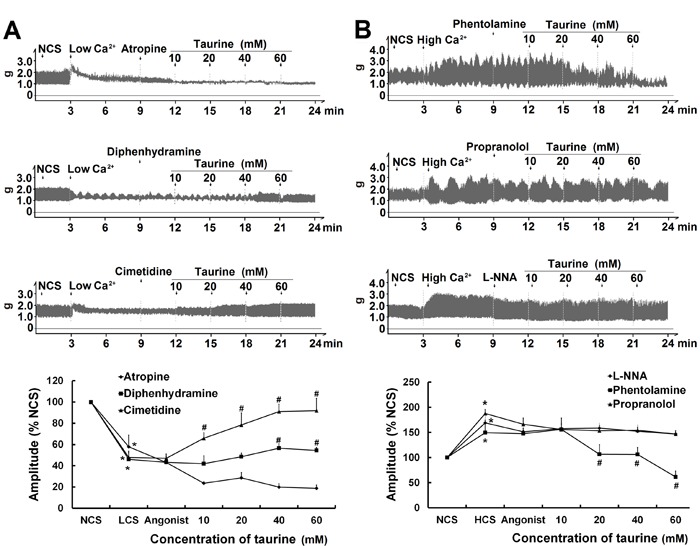
The nonselective muscarinic receptor antagonist atropine blocked the stimulatory effect of taurine (10-60 mM) on the contractile amplitude of isolated jejunal segments in LCS induced by low Ca2+ (1.25 mM) Krebs buffer. Neither the histamine H1-receptor antagonist diphenhydramine nor the H2-receptor antagonist cimetidine blocked taurine (10-60 mM)-induced stimulatory effects on the contractile amplitude in the LCS induced by low Ca2+ (1.25 mM) Krebs buffer (Figure 4). In the HCS induced by high Ca2+ (5.0 mM) Krebs buffer, β-adrenergic receptor antagonist propranolol, and NO synthase inhibitor L-NNA blocked the taurine (10-60 mM)-induced inhibitory effect on the contractile amplitude of isolated jejunal segments. However, the α-adrenergic receptor antagonist phentolamine did not block the taurine-induced (10-60 mM) inhibitory effects on the contractile amplitude in the HCS induced by high Ca2+ (5.0 mM) Krebs buffer (Figure 4).
Figure 4. Effects of taurine on the contractile amplitude of isolated jejunal segments pretreated with receptor antagonists. A, Representative traces and statistical analysis (n=6) of taurine-induced effects on the contractile amplitude of isolated jejunal segments pretreated with 1 μM atropine, 10 μM diphenhydramine, and 10 μM cimetidine in low contractile states (LCS) induced by low Ca2+ (1.25 mM) Krebs buffer. B, Representative traces and statistical analysis (n=6) of taurine-induced effects on the contractile amplitude of isolated jejunal segments pretreated with 1 μM phentolamine, 1 μM propranolol, and 300 μM L-NNA in high contractile states (HCS) induced by high Ca2+ (5.0 mM) Krebs buffer. The contractile amplitude in the normal contractile state (NCS, control) is set to 100%; other data are the relative values compared with NCS. *P<0.05 vs the contractile amplitude in NCS; #P<0.05 vs the contractile amplitude in LCS or HCS before taurine administration (one-way ANOVA).
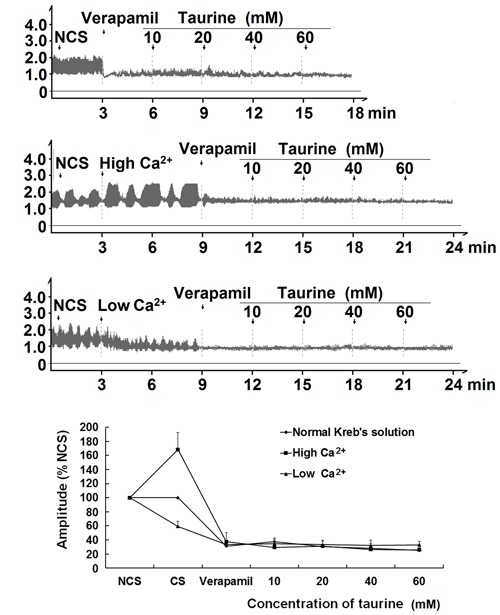
Pre-incubation of isolated jejunal segments with the Ca2+ channel blocker verapamil at NCS, HCS induced by high Ca2+ (5.0 mM) Krebs buffer, and LCS induced by low Ca2+ (1.25 mM) Krebs buffer abolished the effects of taurine (10-60 mM) on the contractile amplitude of isolated jejunal segments (Figure 5). This suggests that verapamil blocked the effects of taurine-induced contractility of isolated jejunal segments.
Figure 5. Effects of taurine on the contractile amplitude of isolated jejunal segments pretreated with verapamil. Representative traces and statistical analysis (n=6) of taurine-induced effects on the contractile amplitude of isolated jejunal segments pretreated with 1 μM verapamil in the normal contractile state (NCS, control), CS induced by high Ca2+ (5.0 mM) Krebs buffer and CS induced by low Ca2+ (1.25 mM) Krebs buffer. The mean contractile amplitude without verapamil treatment in NCS is set to 100%; other data are the relative values compared with NCS. CS: contractile state.
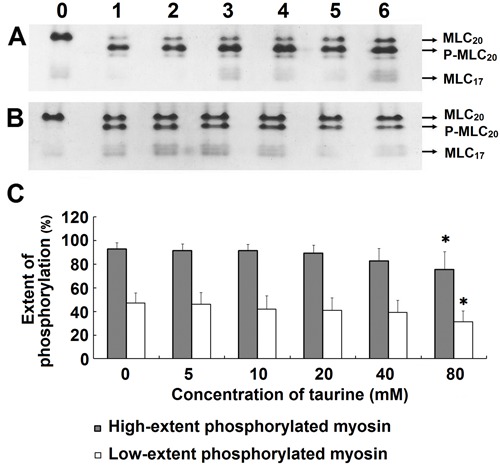
No significant effects of taurine on myosin phosphorylation were observed at concentrations of 5-40 mM. Although the effects of 80 mM taurine were significant, both high-extent and low-extent of myosin phosphorylation were affected (Figure 6). These data indicate that taurine did not modulate myosin phosphorylation bidirectionally.
Figure 6. Effects of taurine on phosphorylated myosin. A, Effects of taurine on high-extent phosphorylated myosin, 0.02 μM myosin light chain kinase (MLCK), and 4.0 μM myosin purified from chicken gizzard smooth muscle used in the assay. B, Effects of taurine on low-extent phosphorylated myosin, 2.0 μM MLCK and 4.0 μM myosin purified from chicken gizzard smooth muscle used in the assay. Lanes 0-6 represent unphosphorylated myosin (without MLCK and taurine), high- or low-extent phosphorylated control (without taurine), high- or low-extent phosphorylated myosin with 5, 10, 20, 40, and 80 mM taurine, respectively. C, Extent of myosin phosphorylation, which was analyzed using the Gelpro software. The extent of phosphorylated myosin was calculated as percentage of phosphorylated regulatory myosin light chain of 20 kDa (MLC20) in total MLC20. Mono-phosphorylation was calculated as 100% phosphorylation. *P<0.05 vs high- or low-extent phosphorylated control (without taurine) (one-way ANOVA) (lane 1). MLC20: unphosphorylated MLC20 (20 kDa regulatory myosin light chain); p-MLC20: mono-phosphorylated MLC20; MLC17: 17 kDa myosin essential light chains.
Discussion
Taurine exerted stimulatory and inhibitory effects on the contractility of isolated jejunal segments in both low and high contractile states. Considering that the HCS and LCS were established under different assay conditions and by using agents with different mechanisms, the results suggest that taurine-induced bidirectional modulation on isolated rat jejunal segments depends on its contractile state. The evidence that taurine induced a bidirectional modulation on jejunal contractility depending on its contractile state indicates that taurine plays an important homeostatic role in intestinal function.
Ca2+/CaM-dependent phosphorylation of myosin light chains by MLCK is generally considered to be the primary mechanism for regulating the contraction of smooth muscle. Nevertheless, the effects of taurine on phosphorylation of myosin from chicken gizzard were not consistent with its bidirectional effect on isolated segments of rat jejunum. We only observed a slight inhibition effect of taurine on both high- and low-extent phosphorylation of myosin from chicken gizzard. Considering that such a high intracellular concentration of taurine can hardly be achieved in the muscle, we believe that the inhibition of 80 mM taurine on myosin phosphorylation was due to the toxic effects induced by such high concentration. These results indicated that taurine might exert dual modulation not on myogenic contractions of intestinal smooth muscle directly, but by some other mechanisms, e.g., regulation of neurotransmitter or hormone release. However, due to species differences, we cannot completely exclude the possibility that taurine has a direct effect on phosphorylation of myosin light chains in rats.
The neuronal regulation of intestinal contraction involves the ENS, as well as extrinsic nerves (25); TTX is a blocker of neuronal conduction (26); taurine-induced modulation on the contractility of isolated jejunal segments was blocked by TTX, suggesting that neural regulation of ENS is involved in taurine-induced effects.
Activation of muscarinic or histamine receptor increases intestinal contractility, and stimulation of α and β adrenoceptors inhibits intestinal contractility. Inhibition of intestinal contractility is also mediated by NO, a nonadrenergic, noncholinergic neurotransmitter that produces its effect by directly acting on smooth muscle and by indirectly inhibiting acetylcholine and substance P release (27,28). In LCS induced by low Ca2+ (1.25 mM) Krebs buffer, atropine blocked the stimulatory effects of taurine on the contractility of isolated jejunal segments, but diphenhydramine and cimetidine did not, implicating that the stimulatory effects of taurine on the jejunal contractility are correlated with muscarinic receptor-linked stimulation. The results are in line with reports that found the effect of taurine (10−6-10−1 mM) on acid secretion in the rat stomach was completely or partially inhibited by atropine (29). Although the doses of taurine used in our study were different from those in the above report, the difference might be due to the different target tissues of taurine. In HCS induced by high Ca2+ (5.0 mM) Krebs buffer, propranolol and L-NNA abolished the inhibitory effects of taurine on the contractility of isolated jejunal segments, however, phentolamine did not modify the inhibitory effects of taurine, suggesting that the inhibitory effects of taurine are correlated with the activation of adrenergic β receptors, as well as NO synthase-linked relaxation mechanisms, rather than adrenergic α receptors. Consistent with our results, taurine has been reported to increase serum levels of NO, NO synthase, and reactive nitrogen oxide species in the rat model of hypertension (30) and in guinea pig spleen tissue (4). However, the possible mechanisms of taurine on NO synthase in our study and the above studies may differ, and remains uncertain in this study.
Smooth muscle contraction is initiated by an increase in cytosolic free Ca2+, brought about either by release of Ca2+ from intracellular stores or by influx of Ca2+ through voltage-dependent Ca2+ channels (31). Our results indicated that verapamil, an L-type Ca2+ channel antagonist, blocked the effect of taurine on the contractility of isolated jejunal segments in NCS and HCS induced by high Ca2+ (5.0 mM) Krebs buffer and LCS induced by low Ca2+ (1.25 mM) Krebs buffer, suggesting that modulation of intestinal contractility by taurine is Ca2+ dependent and involves L-type Ca2+ channels. It has been demonstrated that taurine may exert a normalizing action on the [Ca2+]i by directly or indirectly modulating the activity of the voltage-dependent Ca2+ channels or other transmembrane ion channels and transporters in the cell organelles (16,32-34). Such dual effects on [Ca2+]i may explain the bidirectional modulation of taurine on smooth muscle contraction.
Although the present research characterized a homeostatic modulation of taurine on contractility of intestinal smooth muscle, it should still be considered as preliminary. Further studies are needed to reveal the detailed mechanism, including the possible neurotransmitters involved and the details of cell signal transduction.
Supplementary Material
Acknowledgments
Research supported by the National Natural Science Foundation of China (No. 30772601).
Footnotes
First published online.
References
- 1.Junyent F, de Lemos L, Utrera J, Paco S, Aguado F, Camins A, et al. Content and traffic of taurine in hippocampal reactive astrocytes. Hippocampus. 2011;21:185–197. doi: 10.1002/hipo.20739. [DOI] [PubMed] [Google Scholar]
- 2.Morales I, Dopico JG, Sabate M, Gonzalez-Hernandez T, Rodriguez M. Substantia nigra osmoregulation: taurine and ATP involvement. Am J Physiol Cell Physiol. 2007;292:C1934–C1941. doi: 10.1152/ajpcell.00593.2006. [DOI] [PubMed] [Google Scholar]
- 3.Zhang LY, Zhou YY, Chen F, Wang B, Li J, Deng YW, et al. Taurine inhibits serum deprivation-induced osteoblast apoptosis via the taurine transporter/ERK signaling pathway. Braz J Med Biol Res. 2011;44:618–623. doi: 10.1590/S0100-879X2011007500078. [DOI] [PubMed] [Google Scholar]
- 4.Bircan FS, Balabanli B, Turkozkan N, Ozan G. Effects of taurine on nitric oxide and 3-nitrotyrosine levels in spleen during endotoxemia. Neurochem Res. 2011;36:1978–1983. doi: 10.1007/s11064-011-0521-3. [DOI] [PubMed] [Google Scholar]
- 5.Silva LA, Silveira PC, Ronsani MM, Souza PS, Scheffer D, Vieira LC, et al. Taurine supplementation decreases oxidative stress in skeletal muscle after eccentric exercise. Cell Biochem Funct. 2011;29:43–49. doi: 10.1002/cbf.1716. [DOI] [PubMed] [Google Scholar]
- 6.Gao X, Yang X, Zhang B. Neuroprotection of taurine against bilirubin-induced elevation of apoptosis and intracellular free calcium ion in vivo . Toxicol Mech Methods. 2011;21:383–387. doi: 10.3109/15376516.2010.546815. [DOI] [PubMed] [Google Scholar]
- 7.Haojun Z, Yaoling W, Ke Z, Jin L, Junling W. Effects of NaF on the expression of intracellular Ca2+ fluxes and apoptosis and the antagonism of taurine in murine neuron. Toxicol Mech Methods. 2012;22:305–308. doi: 10.3109/15376516.2012.657259. [DOI] [PubMed] [Google Scholar]
- 8.Huxtable RJ. Physiological actions of taurine. Physiol Rev. 1992;72:101–163. doi: 10.1152/physrev.1992.72.1.101. [DOI] [PubMed] [Google Scholar]
- 9.Wojcik OP, Koenig KL, Zeleniuch-Jacquotte A, Costa M, Chen Y. The potential protective effects of taurine on coronary heart disease. Atherosclerosis. 2010;208:19–25. doi: 10.1016/j.atherosclerosis.2009.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Abebe W, Mozaffari MS. Role of taurine in the vasculature: an overview of experimental and human studies. Am J Cardiovasc Dis. 2011;1:293–311. [PMC free article] [PubMed] [Google Scholar]
- 11.Souza MM, Boyle RT. A moderate decrease in temperature inhibits the calcium signaling mechanism(s) of the regulatory volume decrease in chick embryo cardiomyocytes. Braz J Med Biol Res. 2001;34:137–141. doi: 10.1590/S0100-879X2001000100018. [DOI] [PubMed] [Google Scholar]
- 12.Ideishi M, Miura S, Sakai T, Sasaguri M, Misumi Y, Arakawa K. Taurine amplifies renal kallikrein and prevents salt-induced hypertension in Dahl rats. J Hypertens. 1994;12:653–661. doi: 10.1097/00004872-199406000-00005. [DOI] [PubMed] [Google Scholar]
- 13.Anuradha CV, Balakrishnan SD. Taurine attenuates hypertension and improves insulin sensitivity in the fructose-fed rat, an animal model of insulin resistance. Can J Physiol Pharmacol. 1999;77:749–754. doi: 10.1139/y99-060. [DOI] [PubMed] [Google Scholar]
- 14.Militante JD, Lombardini JB. Treatment of hypertension with oral taurine: experimental and clinical studies. Amino Acids. 2002;23:381–393. doi: 10.1007/s00726-002-0212-0. [DOI] [PubMed] [Google Scholar]
- 15.Abebe W, Mozaffari MS. Effects of chronic taurine treatment on reactivity of the rat aorta. Amino Acids. 2000;19:615–623. doi: 10.1007/s007260070011. [DOI] [PubMed] [Google Scholar]
- 16.Abebe W. Effects of taurine on the reactivity of aortas from diabetic rats. Life Sci. 2008;82:279–289. doi: 10.1016/j.lfs.2007.11.012. [DOI] [PubMed] [Google Scholar]
- 17.Abebe W, Mozaffari MS. Effect of taurine deficiency on adenosine receptor-mediated relaxation of the rat aorta. Vascul Pharmacol. 2003;40:219–228. doi: 10.1016/j.vph.2003.08.001. [DOI] [PubMed] [Google Scholar]
- 18.Abebe W, Mozaffari MS. Taurine depletion alters vascular reactivity in rats. Can J Physiol Pharmacol. 2003;81:903–909. doi: 10.1139/y03-088. [DOI] [PubMed] [Google Scholar]
- 19.Niu LG, Zhang MS, Liu Y, Xue WX, Liu DB, Zhang J, et al. Vasorelaxant effect of taurine is diminished by tetraethylammonium in rat isolated arteries. Eur J Pharmacol. 2008;580:169–174. doi: 10.1016/j.ejphar.2007.10.039. [DOI] [PubMed] [Google Scholar]
- 20.Xue W, Zhang M, Li J, Wu D, Niu L, Liang Y. Effects of taurine on aortic rings isolated from fructose-fed insulin resistance Sprague-Dawley rat are changed. Cardiovasc Drugs Ther. 2008;22:461–468. doi: 10.1007/s10557-008-6124-9. [DOI] [PubMed] [Google Scholar]
- 21.Schemann M. Control of gastrointestinal motility by the “gut brain” - the enteric nervous system. J Pediatr Gastroenterol Nutr. 2005;41((Suppl 1)):S4–S6. doi: 10.1097/01.scs.0000180285.51365.55. [DOI] [PubMed] [Google Scholar]
- 22.Lin Y, Ishikawa R, Okagaki T, Ye LH, Kohama K. Stimulation of the ATP-dependent interaction between actin and myosin by a myosin-binding fragment of smooth muscle caldesmon. Cell Motil Cytoskeleton. 1994;29:250–258. doi: 10.1002/cm.970290308. [DOI] [PubMed] [Google Scholar]
- 23.Lin Y, Sun H, Dai S, Tang Z, He X, Chen H. The bi-directional regulation of filamin on the ATPase activity of smooth muscle myosin. Chin Med Sci J. 2000;15:162–164. [PubMed] [Google Scholar]
- 24.Yang J, Wang X, Tang Z, Chen H, Dai S, Lin Y. The characterization of myosin light chain phosphorylation by the constitutively active fragment of MLCK. Chinese Med Sci J. 2003;18:206–212. http://www.cqvip.com/Main/Detail.aspx?id=9051177 (Chung-kuo i hsueh k'o hsueh tsa chih) [Google Scholar]
- 25.Hansen MB. Neurohumoral control of gastrointestinal motility. Physiol Res. 2003;52:1–30. [PubMed] [Google Scholar]
- 26.Llewellyn LE. Sodium channel inhibiting marine toxins. Prog Mol Subcell Biol. 2009;46:67–97. doi: 10.1007/978-3-540-87895-7_3. [DOI] [PubMed] [Google Scholar]
- 27.Li M, Johnson CP, Adams MB, Sarna SK. Cholinergic and nitrergic regulation of in vivo giant migrating contractions in rat colon. Am J Physiol Gastrointest Liver Physiol. 2002;283:G544–G552. doi: 10.1152/ajpgi.00114.2001. [DOI] [PubMed] [Google Scholar]
- 28.Mang CF, Truempler S, Erbelding D, Kilbinger H. Modulation by NO of acetylcholine release in the ileum of wild-type and NOS gene knockout mice. Am J Physiol Gastrointest Liver Physiol. 2002;283:G1132–G1138. doi: 10.1152/ajpgi.00192.2002. [DOI] [PubMed] [Google Scholar]
- 29.Huang KH, Chang CC, Ho JD, Lu RH, Tsai LH. Role of taurine on acid secretion in the rat stomach. J Biomed Sci. 2011;18:11. doi: 10.1186/1423-0127-18-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hu J, Xu X, Yang J, Wu G, Sun C, Lv Q. Antihypertensive effect of taurine in rat. Adv Exp Med Biol. 2009;643:75–84. doi: 10.1007/978-0-387-75681-3_8. [DOI] [PubMed] [Google Scholar]
- 31.Aviello G, Scalisi C, Fileccia R, Capasso R, Romano B, Izzo AA, et al. Inhibitory effect of caffeic acid phenethyl ester, a plant-derived polyphenolic compound, on rat intestinal contractility. Eur J Pharmacol. 2010;640:163–167. doi: 10.1016/j.ejphar.2010.04.040. [DOI] [PubMed] [Google Scholar]
- 32.Sawamura A, Sada H, Azuma J, Kishimoto S, Sperelakis N. Taurine modulates ion influx through cardiac Ca2+ channels. Cell Calcium. 1990;11:251–259. doi: 10.1016/0143-4160(90)90001-B. [DOI] [PubMed] [Google Scholar]
- 33.Satoh H, Sperelakis N. Review of some actions of taurine on ion channels of cardiac muscle cells and others. Gen Pharmacol. 1998;30:451–463. doi: 10.1016/S0306-3623(97)00309-1. [DOI] [PubMed] [Google Scholar]
- 34.Li F, Obrosova IG, Abatan O, Tian D, Larkin D, Stuenkel EL, et al. Taurine replacement attenuates hyperalgesia and abnormal calcium signaling in sensory neurons of STZ-D rats. Am J Physiol Endocrinol Metab. 2005;288:E29–E36. doi: 10.1152/ajpendo.00168.2004. [DOI] [PubMed] [Google Scholar]


