Abstract
Minute granules of sporopollenin, called orbicules, can be observed on the innermost tangential and/or radial walls of secretory tapetum cells. Orbicules were investigated in 53 species of 34 Gentianaceae genera using light microscopy, scanning electron microscopy and transmission electron microscopy. This selection covered all different tribes and subtribes recognized in Gentianaceae (87 genera, ±1650 species). Orbicules were found in 38 species (23 genera) distributed among the six tribes recognized in Gentianaceae. The orbicule typology is based on those described previously in Rubiaceae. Of the six orbicule types described previously, Type II orbicules are lacking. Type III orbicules are most common (17 species). Hockinia Gardner is the only representative with Type I orbicules. The number of representatives with orbicules belonging to the other orbicule types are equally distributed among the species studied: seven species possess Type IV orbicules, six species Type V and six species Type VI. The systematic usefulness of this typology is discussed in comparison with the latest systematic insights within the family, and palynological trends in Gentianaceae. Orbicule data have proven to be useful for evaluating tribal delimitation within Rubiaceae and Loganiaceae s.l.; however, they seem not to be useful for tribal delimitation in Gentianaceae. In the tribes Potalieae and Gentianeae orbicule data may be useful at subtribal level.
Key words: Orbicules, Ubisch bodies, pollen, Gentianaceae, SEM, TEM, morphology, ultrastructure, typology, tapetum, systematics
INTRODUCTION
In gymnosperms, seed ferns, and anthers of flowering plants, tiny (<4 µm) granules may occur on the radial and innermost tangential walls of secretory tapetum cells. These granules are sometimes in close contact with the pollen grains, and are called orbicules (Erdtman et al., 1961) or Ubisch bodies (Kosmath, 1927). Orbicules were observed for the first time by Rosanoff (1865). Despite their early discovery, little attention was paid to these structures and in many palynological studies they have been overlooked.
Orbicules develop simultaneously with the growing pollen exine and are composed of sporopollenin, similar to the pollen exine. Orbicules may thus serve as a model for the study of sporopollenin biosynthesis and polymerization in comparison with the exine, since orbicules are acellular structures independent of cytoplasmic control (Clément and Audran, 1993a, b, c). Pro‐orbicules, which are produced by the endoplasmic reticulum of secretory tapetal cells, are the progenitors of orbicules (Echlin and Godwin, 1968). The pro‐orbicules are extruded through the radial and innermost tangential walls of the secretory tapetum cells, at the beginning of the tetrad stage (Christensen et al., 1972; El‐Ghazaly and Jensen, 1986; Clément and Audran, 1993a, b, c; Vijayaraghavan and Chaudry, 1993). Pro‐orbicules are spherical structures, which are composed of a complex organic mixture. They are surrounded by a glycocalyx. The deposition of sporopollenin precursors, produced by the protoplast of the secretory tapetum cells on the glycoprotein filaments of the glycocalyx and on the growing pollen exine, starts at the late tetrad stage. The mature orbicule wall is composed of structural and filling elements (Clément and Audran, 1993a, b, c). Proteins, pectins, polysaccharides and glycolipids, organized in a three‐dimensional polygonal frame (Rowley, 1990) are part of the structural elements. The filling elements consist of unsaturated lipids and polyphenols (Clément and Audran, 1993a, b, c). At the ultrastructural level, Clément and Audran (1993a, b, c) could distinguish four different zones of electron density in mature orbicules of Lilium L. ‘Enchantment’: (1) an electron‐translucent orbicule cavity; (2) a thin and very electron‐dense orbicule cavity–wall interface; (3) a homogeneous electron‐dense orbicule wall; and (4) a thin, very electron‐dense peripheral layer.
Orbicules are considered to be a general feature of species characterized by a secretory tapetum. Huysmans et al. (1998) reported, however, that orbicules are lacking in several taxa with such a tapetum. The predominance of the secretory tapetum among land plants was demonstrated by Furness and Rudall (2001). This observation supports the hypothesis of Pacini et al. (1985) that the secretory type is plesiomorphic in angiosperms. Species with an amoeboid tapetum, which is believed to be apomorphic, characteristically do not produce orbicules (Sporne, 1973; Pacini et al., 1985), although there are exceptions such as Gentiana acaulis L. (Lombardo and Carraro, 1976; Vinckier and Smets, 2002a, c). Tiwari and Gunning (1986) reported the presence of sporopollenin granules on the tapetal remnants in species with amoeboid tapeta, generally smaller in size than the orbicules produced by secretory tapeta. Orbicules have been recorded in some Cretaceous (Mesozoic) (Taylor and Alvin, 1984; Osborn et al., 1991; Serbet and Stockey, 1991; Archangelsky and Taylor, 1993) and Mioceneous (Cenozoic) (Zetter et al., 2002) seed plants which may indicate, along with the proposed plesiomorphic nature of the secretory tapetum (Pacini et al., 1985), that the presence of orbicules is a plesiomorphic feature of seed plants.
During the past 20 years different hypotheses have been suggested to attribute a function to orbicules. However, none of the proposed functions are satisfactorily proven. For a summary of some of the proposed functions see Huysmans et al. (1998).
In angiosperms, the ornamentation of the pollen exine and that of the orbicule wall often show striking analogies (Nilsson and Robyns, 1974; El‐Ghazaly and Jensen, 1986; Hesse, 1986). These parallels are explained by Hesse (1985) as rooted in the homology of tapetum and sporogeneous tissue. Since ornamentation of the pollen exine offers useful characters for systematics, orbicules might also have taxonomic value. In the subfamily Cinchonoideae of Rubiaceae (Huysmans et al., 1997) an orbicule typology has been worked out based on the morphological and ultrastructural orbicule diversity. It was concluded that orbicule characteristics can be useful for systematics at the generic and tribal level. Apart from the study on Cinchonoideae taxa (Huysmans et al., 1997), the taxonomic usefulness of orbicule characters has so far only been studied in four angiosperm taxa: the genera Dioscorea L. (Schols et al., 2001) and Euphorbia L. (El‐Ghazaly, 1989; El‐Ghazaly and Chaudhary, 1993), the Chloanthaceae, now included in Lamiaceae (Raj and El‐Ghazaly, 1987), and the order Gentianales (Vinckier et al., 2000; Vinckier and Smets, 2002a, b, c; Vinckier, 2003).
Recently a phylogenetic, genus‐level classification of the Gentianaceae, based on cladistic analyses of the trnL intron, matK and internal transcribed spacer sequence (ITS) data, was presented by Struwe et al. (2002). In this new classification, the family Gentianaceae consists of approx. 87 genera with approx. 1615–1688 species distributed among six tribes: Saccifolieae, Exaceae, Chironieae, Helieae, Potalieae and Gentianae (Struwe et al., 2002). It is the third largest family in Gentianales, with Rubiaceae and Apocynaceae s.l. being distinctly larger. They have a cosmopolitan distribution from temperate to tropical regions. The representatives of this family are trees, shrubs, vines, or perennial and annual herbs. Flowers are usually bisexual (unisexual in Veratrilla Baill. ex Franch.), hypogynous, 4‐ or 5‐merous, or rarely 3‐ or 6–16‐merous. The calyx is actinomorphic, rarely zygomorphic (Exacum L.). Inflorescences are terminal or axillary, and are usually cymose, but less often racemose, capitate, clustered or spicate; the flowers are seldom solitary. Members of the family do not produce latex and stipules are absent (Struwe et al., 2002). Interesting to the scope of the present paper is the highly variable tapetum; both secretory and amoeboid tapetum types occur, as well as very variable pollen exine characteristics (Nilsson, 1967; Sankara Rao and Chinnappa, 1983). Pollen grains may be released as monads, tetrads or polyads (Nilsson, 2002).
The present study has five major aims: (1) to investigate the presence of orbicules in Gentianaceae; (2) to provide detailed descriptions of the morphology and ultrastructure of orbicules present; (3) to construct an orbicule‐typology; (4) to investigate pollen exine characters by examining the possible parallels between pollen exine and orbicule wall ornamentation; and (5) to discuss the systematic usefulness of the orbicule typology in comparison with the latest systematic insights within the family Gentianaceae.
MATERIALS AND METHODS
This study is based on herbarium material: 53 species of 34 Gentianaceae genera were selected for examination (Table 1; Appendix). This selection covered all different tribes and subtribes recognized in Gentianaceae (Struwe et al., 2002).
Table 1.
Orbicule morphology and ultrastructure of all species studied (classification is according to Struwe et al., 2002)
| Orbicule ultra‐structure | ||||||||||||||
| Tribe | Subtribe | Indent‐ | Aggre‐ | |||||||||||
| (genera/species) | (genera/species) | Species | Size (µm) | Shape | ations | gates | Density | ETC | EDC | EDW | EDP | Type | Pollen exine | Figures |
| Saccifolieae (5/16–20) | Curtia tenuifolia | – | – | – | – | – | – | mret‐perf | 6G and H | |||||
| Hockinia montana | 0·652 ± 0·179 | sp | + | + | a | I | msp‐mret‐perf | 1A and B | ||||||
| Exaceae (6/144–184) | Exacum gracilipes | 0·975 ± 0·206 | ir | + | + | a | V | stri‐perf | ||||||
| Exacum macranthum | 1·240 ± 0·244 | ir‐sgr | + | + | a | V | rug | 5E–F | ||||||
| Exacum oldenlandioides | 1·080 ± 0·268 | ir‐sgr | + | + | a | + | + | + | + | V | stri‐perf | 5D | ||
| Sebaea albidiflora | 0·451 ± 0·119 | aif | + | + | va | + | – | + | + | IV | stri‐mret | 4C–D | ||
| Sebaea ovata | 0·958 ± 0·282 | aif | + | + | a | IV | stri‐mret | 4A and B | ||||||
| Chironieae (23/159) | Chironiinae (12/106) | Blackstonia perfoliata | 0·307 ± 0·163 | ir‐ro | – | + | a | – | – | + | + | IIIa | stri‐perf | 2F–H |
| Centaurium serpentinicola | 1·151 ± 0·272 | (em) ro | – | – | a | VI | stri‐perf | |||||||
| Centaurium spicatum | 1·300 ± 0·377 | (em) ro | – | – | a | VI | stri‐perf | 5G | ||||||
| Centaurium tenuiflorum | 0·467 ± 0·076 | (em) ro | – | – | a | VI | stri‐perf | |||||||
| Chironia pegleriae | 0·201 ± 0·064 | (em) ro | – | – | a | IIIa | stri‐perf | |||||||
| Chironia purpurascens | 0·210 ± 0·051 | (em) ro | – | – | a | – | – | + | + | IIIa | stri‐perf | 2A–D | ||
| Eustoma grandiflorum | 0·280 ± 0·081 | ro | – | – | a | IIIa | stri‐perf‐mret | 2E | ||||||
| Ixanthus viscosus | 0·516 ± 0·126 | ro | – | + | va | IIIa | stri‐perf‐mret | 1G and H | ||||||
| Sabatia bartramii | – | – | – | – | – | – | stri‐perf | |||||||
| Sabatia brachiata | – | – | – | – | – | – | stri‐perf | |||||||
| Sabatia quadrangula | – | – | – | – | – | – | stri‐perf | |||||||
| Canscorinae (6/24) | Canscora decussata | 0·189 ± 0·068 | (em) ro | – | – | la | IIIa | stri‐perf | ||||||
| Canscora heteroclita | 0·208 ± 0·045 | (em) ro | – | – | la | IIIa | stri‐perf | |||||||
| Hoppea fastigiata | 0·309 ± 0·083 | (em) ro | – | + | va | VI | perf‐mret | 5H | ||||||
| Coutoubeinae (5/29) | Coutoubea spicata | – | – | – | – | – | – | tetr: ret‐hbr | 7A and B | |||||
| Schultesia guianensis | – | – | – | – | – | – | tetr: ret | 6D | ||||||
| Schultesia pachyphylla | – | – | – | – | – | – | tetr: stri‐ret | |||||||
| Helieae (22/184) | Calolisianthus pedunculatus | 1·229 ± 0·227 | ir | + | + | a | IV | tetr: ret isl. hbr, loop‐like | 4G and H | |||||
| Irlbachia purpurascens | – | – | – | – | – | – | pol: perf‐mret‐hbr, polar loops | 6E and F | ||||||
| Macrocarpaea rubra | – (c TEM) | – | – | – | – | – | ret‐mret | |||||||
| Prepusa montana | 2·985 ± 0·772 | ro | – | – | la | IIIa | tetr: gr | 3C and D | ||||||
| Tachia guianensis | 0·384 ± 0·100 | (em) ro | – | + | a | IIIa | psil‐gr | 3A and B | ||||||
| Potalieae (13/154) | Faroinae (9/31) | Enicostema verticillatum | – | – | – | – | – | – | perf‐mret | |||||
| Faroa affinis | – | – | – | – | – | – | stri‐perf‐mret | |||||||
| Lisianthiinae (1/30) | Lisianthius areopolus | 0·227 ± 0·048 | (em) ir | – | + | a | VI | ret | ||||||
| Lisianthius nigrescens | 0·439 ± 0·097 | (em) ir | – | + | a | VI | ret | 6A and B | ||||||
| Potaliinae (3/93) | Anthocleista amplexicaulis | 0·573 ± 0·144 | ir | + | + | a | V | psil‐perf | 5B | |||||
| Anthocleista scandens | 0·873 ± 0·288 | ir | + | + | a | V | perf | 5A | ||||||
| Fagraea racemosa | 1·512 ± 0·366 | ir | + | + | a | + | – | + | + | V | stri‐ret‐hbr | 5C | ||
| Potalia amara | 0·719 ± 0·323 | ir | – | + | a | VI | psil‐perf | |||||||
| Gentianeae (17/939–968) | Gentianinae (3/400–403) | Gentiana acaulis | 0·376 ± 0·127 | aif | – | + | va | IV | stri‐mret | |||||
| Gentiana algida | 0·384 ± 0·140 | aif | – | + | va | IV | stri‐mret | |||||||
| Gentiana alpina | 0·487 ± 0·166 | aif | – | + | va | IV | stri‐mret | |||||||
| Tripterospermum fasciculatum | 0·285 ± 0·086 | aif | – | + | a | – | – | + | + | IV | stri | 4E–F | ||
| Swertiinae (14/539–565) | Frasera albicaulis | 0·229 ± 0·056 | ir‐ro | – | – | la | IIIa | stri‐perf | ||||||
| Frasera parryi | 0·247 ± 0·069 | ir‐ro | – | – | la | – | – | + | + | IIIa | stri‐perf | 3E–G | ||
| Gentianella amarella | 0·373 ± 0·180 | ro | – | – | la | IIIa | perf‐mret | |||||||
| Gentianella bellidifolia | 0·409 ± 0·200 | ro | – | – | la | IIIa | stri‐mret | 1F | ||||||
| Gentianopsis barbata | 0·168 ± 0·053 | ro | – | + | a | IIIa | mret | |||||||
| Gentianopsis procera | 0·249 ± 0·066 | ro | – | + | a | – | – | + | + | IIIa | mret | 1C–E | ||
| Halenia corniculata | – (c TEM) | – | – | – | – | – | psil‐perf | 7C | ||||||
| Lomatogonium rotatum | – | – | – | – | – | – | stri‐perf | |||||||
| Lomatogonium carinthiacum | – (c TEM) | – | – | – | – | – | stri‐perf | |||||||
| Obolaria virginica | – (c TEM) | – | – | – | – | – | perf‐mret | 6C | ||||||
| Swertia bimaculata | 0·199 ± 0·082 | ro | – | – | la | IIIa | stri‐perf | 3H | ||||||
| Swertia perennis | 0·145 ± 0·032 | ro | – | – | la | IIIa | stri‐perf‐mret | |||||||
| Genus incertae sedis | Voyria caerulea | psil | 7D | |||||||||||
Species in bold possess orbicules.
Tribe and subtribe, sensu Struwe et al. (2002); Size, mean ± standard deviation; Type, orbicule type sensu Huysmans et al. (1997) and Vinckier et al. (2000).
a, abundant, aif, angular irregular folded; c TEM, confirmed by TEM observation; EDC, electron‐dense cavity wall interface; EDP, very electron‐dense peripheral layer; EDW, homogeneous electron‐dense orbicule wall; em, embedded in tapetal membrane; ETC, electron translucent cavity; gr, granulate exine; hbr, heterobrochate; ir, irregular; isl, islands; la, only few orbicules are present on locule‐surface; mret, microreticulate; msp, microspinulose; perf, perforate; pol, polyad; psil, psilate; r, regular; ret, reticulate; ro, rounded oblate; rug, rugulate; sp, spiny; stri, striate; sgr, sporopollenin granulae present on the orbicule surface; tetr, tetrad; va, very abundant.
For scanning electron microscopy (SEM) of orbicules and pollen, dried flowers or buds were rehydrated in the wetting agent Agepon® (Agfa Gevaert, Leverkusen, Germany) for 1–2 h. The anthers were picked out from the flowers and the tips of the anthers were removed with a razor blade to facilitate rehydration of the locules. After dissection, the anthers remained for another hour in the wetting agent. Following dehydration in a graded acetone series, the material was critical point dried (CPD 030, Balzers) and mounted on stubs with double‐sided adhesive tape before further preparation. The pollen grains were slightly removed from the opened locules with a fine cactus spine to facilitate observation of the locule surface. The removed pollen grains were collected on the same stubs for further observations. The stubs were sputter coated with gold (SPI‐MODULE™ Sputter Coater; SPI Supplies, West Chester, PA, USA). We used a Jeol JSM‐6400 microscope, at 10–20 kV, for morphological observations. Comparative size measurements of orbicules were ascertained from SEM micrographs using Carnoy 2·0 (Schols et al., 2002). From each anther investigated, 100 orbicules were measured, and mean values and standard deviations were calculated (Table 1).
For observations using transmission electron microscopy (TEM) anthers of selected flowers were fixed with 2 % glutaraldehyde at pH 7·4, buffered with 0·05 m sodium cacodylate. Selected anthers are embedded in LR‐White Resin (Polysciences Inc., Warrington, PA, USA). Prior to embedding in LR‐White Resin the material was dehydrated in a graded ethanol series. Semi‐thin (±1 µm) sections were cut with a Reichert Jung Ultracut E microtome and stained with 0·1 % thionin/0·1 % methylene blue. The semi‐thin sections were observed with a Leica DM LB light microscope. The ultra‐thin (±70 nm) sections, on copper grids, were stained with uranyl acetate and lead citrate in a LKB 2168 Ultrostainer and were observed in a Zeiss EM 900 transmission electron microscope at 50–80 kV.
RESULTS
Orbicule characters
Orbicules were found in 38 species (23 genera) distributed among the six tribes recognized in Gentianaceae (Struwe et al., 2002).
The size and shape of the orbicules, presence of indentations in the orbicule wall, occurrence of aggregated orbicules, their distribution density, and ultrastructure are summarized in Table 1.
Size
In Gentianaceae orbicules are often very small, the smallest (0·145 µm) being observed in Swertia perennis L. The largest orbicules (2·985 µm) occur in Prepusa montana Mart. (Table 1).
Shape
There are three distinct shapes: (1) more or less rounded oblate (Figs 1C and G, 2C, E and F and 3B, F and H); (2) irregular‐shaped granular orbicules (Fig. 5B–F); and (3) irregular angular and ‘folded’ orbicules (Fig. 4B, C, E and H; Table 1). Apart from these clearly definable shapes, the orbicular elements were generally amorphous.
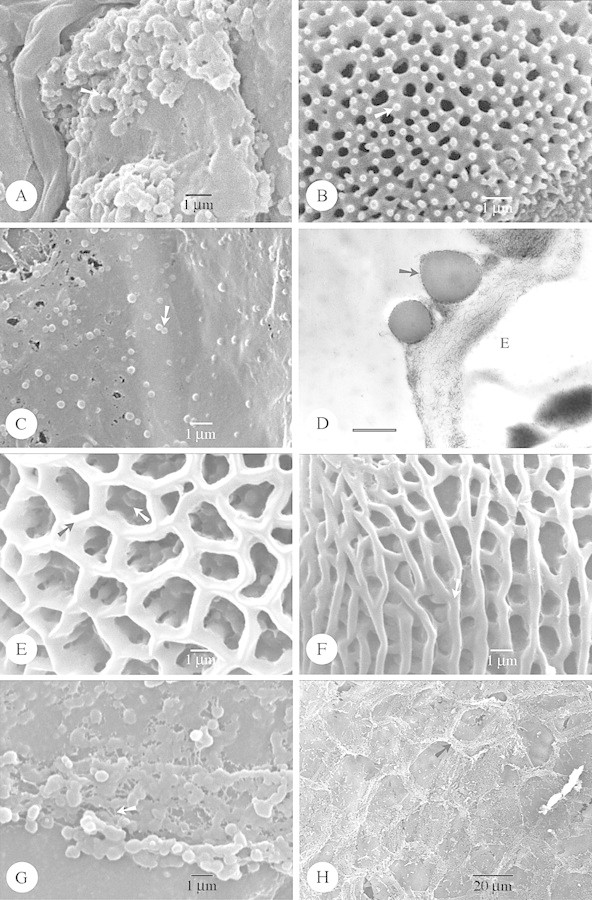
Fig. 1. SEM of species with Type I orbicules (A and B) and SEM and TEM of Type IIIa (C–H) orbicules. Abbreviations used in figures: E, endothecium; ED, electron dense; ET, electron translucent; PE, pollen exine; SEM, scanning electron microscopy; TEM, transmission electron microscopy. A and B, Hockinia montana. A, SEM observation of the spiny orbicules (arrow), covering the locule wall surface. B, Detail of the exine ornamentation. Microspines (arrow) are present upon the tectum. C–E, Gentianopsis procera. C, SEM observation of the small spherical orbicules (arrow). D, TEM observation of the orbicules. An electron‐transparent orbicule cavity is absent. The orbicule wall is delimited by a very electron‐dense and granular peripheral layer (arrow). Bar = 0·6 µm. E, Detail of the reticulate exine. The lumina are beset with granules (white arrow). The muri are sharply crested (black arrow). F, Gentianella bellidifolia. SEM observation of the striato‐reticulate exine. The muri are oriented parallel and criss‐crossed with smaller muri. The muri are rounded (arrow). G and H, Ixanthus viscosus. G, SEM observation of single or compound orbicules. Sporopollinous threads occur between the orbicules (arrow). H, General view of the locule wall surface, imprints of pollen grains are striking. Orbicules (black arrow) are present on the edges between the imprints.
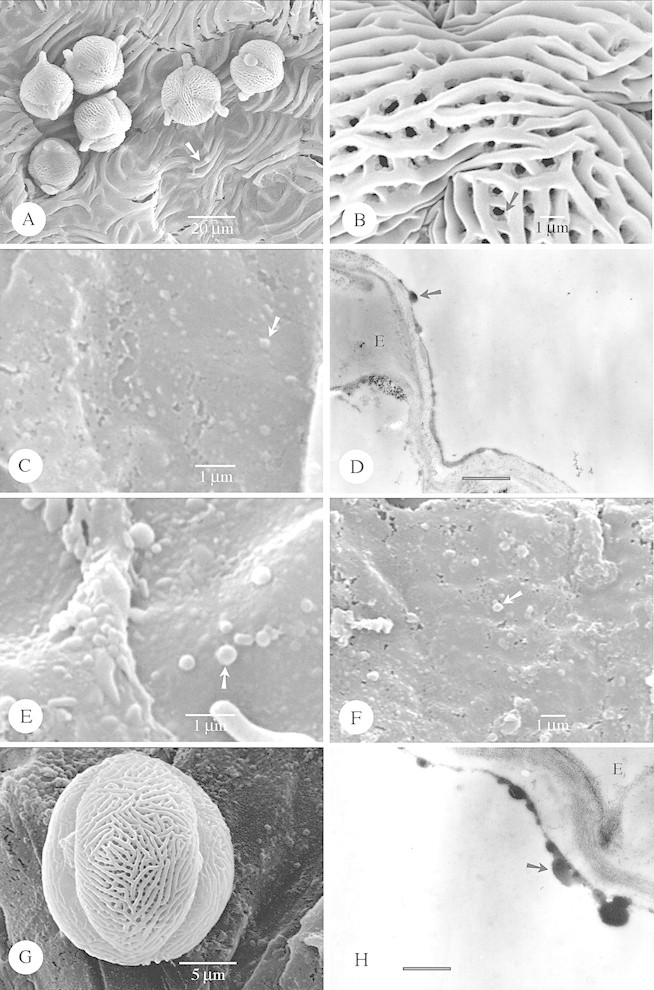
Fig. 2. SEM and TEM of species with Type IIIa orbicules. A–D, Chironia purpurascens. A, General observation of six pollen grains lying on the locule wall surface, which consists of wavy ridges (arrow). B, Detail of the striate exine ornamentation in the polar region. The muri possess granules (arrow) on their side walls. C, SEM observation of the small spherical orbicules (arrow) covering the locule wall surface. D, TEM observation of the small orbicules (arrow). An electron‐transparent orbicule cavity is lacking. Bar = 0·6 µm. E, Eustoma grandiflorum. SEM observation of the small spherical orbicules (arrow). F–H, Blackstonia perfoliata. F, SEM observation of small orbicules (arrow) present on the locule wall surface. G, Equatorial view of a striate pollen grain lying on the locule wall surface. The muri are oriented in various directions. H, TEM observation of the orbicules, lacking an electron‐transparent orbicule cavity. Bar = 0·6 µm.

Fig. 3. SEM and TEM of species with Type IIIa orbicules. A and B, Tachia guianensis. A, Equatorial view of a granulate pollen grain lying on the locule wall surface. The granulate exine consist of numerous granules (arrow) on the smooth sexine. B, Detail of the spherical orbicules (arrow), resembling the granules present on the exine. C and D, Prepusa montana. C, SEM observation of a granulate tetrad lying on the locule wall surface. The granulate exine consist of numerous macroprocesses (black arrow) intermingled with microprocesses (white arrow). D, Detailed observation of the spherical orbicules (arrow), resembling the macroprocesses present on the exine. E–G, Frasera parryi. E, Equatorial view of a striate pollen grain lying on the locule wall surface. F, Observation of the small orbicules (arrow) present on the locule wall surface at a very low distribution density. G, TEM observation of a part of a pollen grain, close to the tapetal remnants on which orbicules (arrow) are present. Bar = 0·6 µm. H, Swertia bimaculata. SEM observation of the small orbicules (arrow), distributed upon the locule wall surface at a very low distribution density.
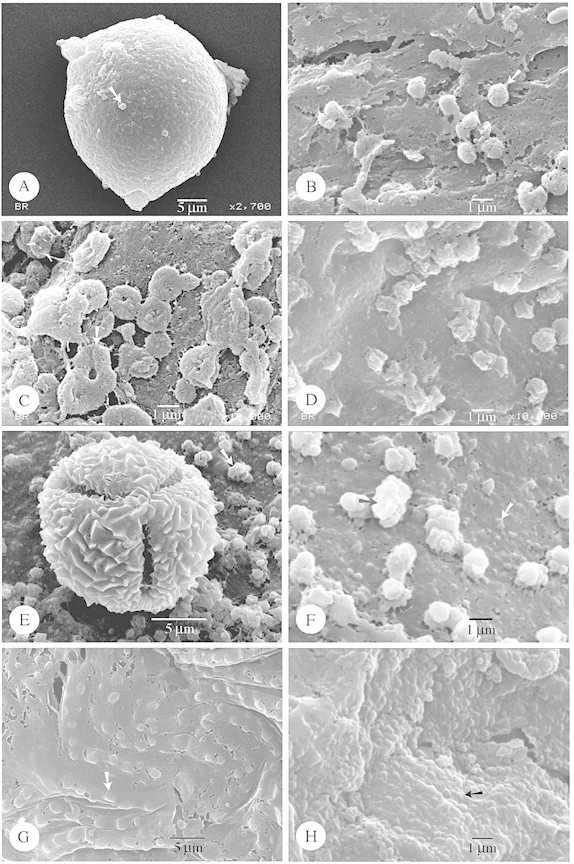
Fig. 5. SEM of species with irregular Type V (A–F) and strongly embedded Type VI (G–H) orbicules. A, Anthocleista scandens. Polar view of a tri‐porate perforate pollen grain. Orbicules (arrow) are attached to the exine. B, Anthocleista amplexicaulis. Detail of the irregular orbicules (arrow) covering the locule wall surface. C, Fagraea racemosa. SEM observation of the irregular and compound orbicules (arrow). D, Exacum oldenlandioides. SEM observation of the irregular orbicules covering the locule wall surface. On the orbicule wall small sporopollinous granulae are present (arrow). E and F, Exacum macranthum. E, SEM observation of a rugulate pollen grain lying on the locule wall surface which is covered by many orbicules (arrow). F, Detailed SEM observation of the orbicules (black arrow) covering the locule wall surface. Intermingled with the orbicules small granules of sporopollenin (white arrow) may cover the locule wall surface. G, Centaurium spicatum. SEM observation of the distinctly embedded orbicules (arrow). H, Hoppea fastigiata. SEM observation of the distinctly embedded orbicules (arrow).
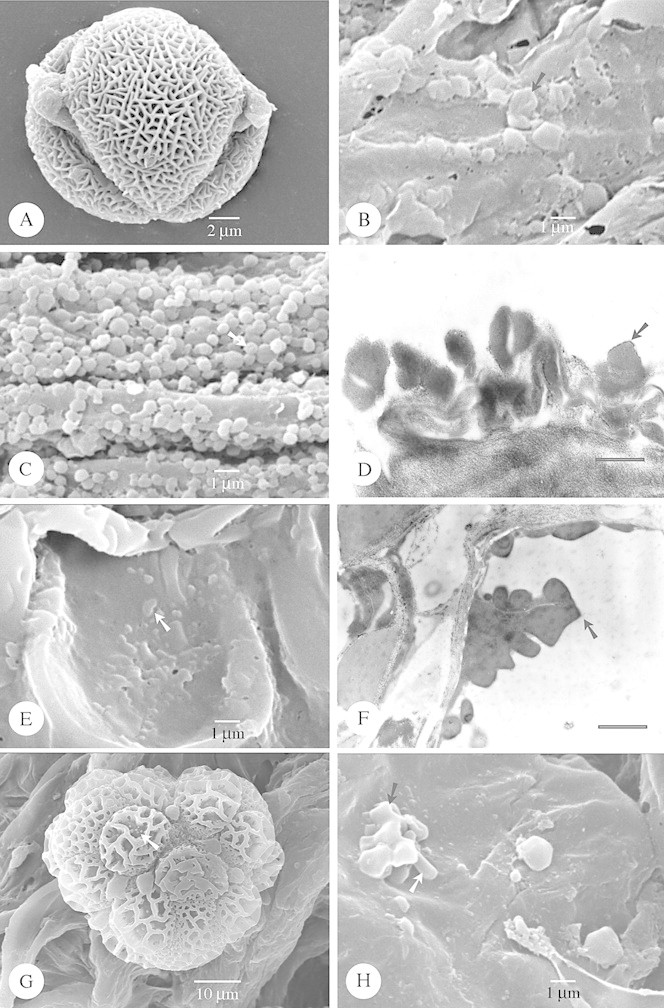
Fig. 4. SEM and TEM of species with irregular and folded Type IV orbicules. A and B, Sebaea ovata. A, Equatorial view of a striato‐micro‐reticulate pollen grain. B, Detail of the irregular and folded orbicules (arrow), resembling parts of the muri building up the exine. C and D, Sebaea albidiflora. C, SEM observation of the irregular and folded orbicules (arrow). D, TEM observation of the irregular orbicules positioned on the remnants of the tapetum cells. The orbicules posses an elongated electron‐transparent cavity and the homogeneous ED orbicule wall is delimited by a electron‐dense peripheral layer (arrow). Bar = 0·6 µm. E and F, Triptospermum fasciculatum. E, SEM observation of the irregular and folded orbicules (arrow) covering the locule wall surface. F, TEM observation of the irregular orbicules, lacking an electron‐transparent cavity. The homogeneous ED orbicule wall is delimited by an ED peripheral layer (arrow). Bar = 1 µm. G and H, Calolisianthus pedunculatus. G, SEM observation of a tetrad lying on the locule wall surface. The muri of the reticulate pattern are organized in heterobrochate reticulate islands. The lumina are beset with granules (arrow). H, SEM observation of the orbicules on the locule wall surface. The orbicules consist of columellae‐like particles (white arrow) which support the muri‐like part (black arrow) of the orbicules.
Indentations and other surface characters
In the majority of species the orbicules have a smooth surface. However, in some species the orbicule wall is indented (Figs 4B and C and 5B and C) or covered by sporopolleninous granules (Fig. 5D and F) or micro‐spinulae (Fig. 1A). Thin threads may occur, connecting the orbicules with each other, or between the tapetal membrane and the orbicules (Fig. 1G).
Aggregations
The apparently large orbicules are often aggregations of smaller orbicules. Irregular layers of small to large aggregations of orbicules embedded in residual tapetal material occur in some species (Figs 1G and 5C).
Distribution density and location of orbicules
When present, orbicules are dispersed over the entire locule surface. Orbicules may be attached to the exine of the pollen grains (Fig. 5A). The position of the orbicules relative to the tapetal membrane varies. They may be completely embedded (Fig. 5G and H), or lightly attached to the surface of the tapetal membrane (Figs 1C, 2E and 5F). A continuum between both extremes may occur within one species (Fig. 3B).
Orbicules are always irregularly arranged. The number of orbicules present on the locule surface is well in excess to the number of pollen grains within the locule. Three terms are used (Table 1) to describe the distribution density: ‘very abundant’ denotes that the locule surface is obscured by the orbicule density (Fig. 4C), while ‘abundant’ denotes that the orbicules are not in direct contact, so that the locule surface is visible between the orbicules (Fig. 1C). In Gentianaceae, it was observed (for the first time in Gentianales) that small orbicules may be dispersed over the locule surface in very low distribution densities (Fig. 3F and H).
Ultrastructure
Thirteen species (13 genera) were investigated at the ultrastructural level (TEM, Table 1). These species covered all the tribes (excluding Saccifolieae) defined recently in Gentianaceae (Struwe et al., 2002). The selection was based on the observation of variations in morphological characters of the orbicules. In the descriptions of the ultrastructure (Table 1) much attention is paid to the occurrence of the different zones of electron density, as defined by Clément and Audran (1993a, b, c) (see Introduction).
A shape variation in the cross‐sections of orbicules was observed: spherical (Figs 1D, 2D and H and 3G), irregular (Fig. 4D; Figs 46 and 48 of Vinckier and Smets, 2002a), and many‐sided appearances occur (Fig. 4F). In many species the orbicules lack an electron‐translucent orbicule cavity (Figs 1D, 2D and H, 3G and 4F). When an electron‐translucent orbicule cavity is present it might have an elongated (Fig. 4D) or a rounded oblate appearance (Fig. 48 of Vinckier and Smets, 2002a). In some species, the electron‐dense orbicule wall possesses a very intense, homogeneous staining (Figs 2D and H and 3G). The only species in which a thin electron‐dense orbicule cavity–wall interface is observed, between the orbicule cavity and the homogeneous electron‐dense orbicule wall, was Exacum oldenlandioides (S. Moore) Klack (Fig. 48 of Vinckier and Smets, 2002a). The presence of a very electron‐dense peripheral layer, delimiting the homogeneous electron‐dense orbicule wall, is a general feature in the species studied. However, orbicules with very intense, homogeneous staining (e.g. Fig. 2D and H) do not show this electron‐dense peripheral layer. This electron‐dense peripheral layer sometimes has a granular appearance (Fig. 1D).
Orbicule types
The typology (Table 1) follows the treatment of orbicule types described for the rubiaceous subfamilies, Cinchonoideae (Huysmans et al., 1997) and Ixoroideae (Vinckier et al., 2000). In the Cinchonoideae, Huysmans et al. (1997) defined four orbicule types: spiny orbicules (Type I), microrugulate orbicules (Type II), smooth and rounded oblate orbicules (Type III), and irregular and folded orbicules (Type IV). Two additional orbicule types were defined by Vinckier et al. (2000) in the Ixoroideae: large flattened and irregular orbicules (Type V), and distinctly embedded orbicules (Type VI). In Ixoroideae (Vinckier et al., 2000) Type III orbicules were split up into two subtypes: rounded oblate orbicules (Subtype IIIa), and doughnut‐shaped orbicules (Subtype IIIb) which are characterized by a central indentation in the orbicule wall. For a more extensive description of the different orbicule types, refer to Huysmans et al. (1997), Vinckier et al. (2000), and Vinckier and Smets (2002a).
Orbicules in Gentianaceae fit well into the types described previously in Rubiaceae, and hence these are used in the present paper (Table 1). Orbicule aggregation does not affect their placement into types, due to the fact that single orbicules or aggregations may occur within one species (Fig. 1G). Of the six orbicule types, Type II orbicules are lacking in the study group. In the majority of species (17 species) Type III orbicules are recorded (Figs 1C–H, 2 and 3; Table 1). Hockinia Gardner is the only representative with Type I orbicules (Figs 1A and B). The number of representatives with orbicules belonging to the other orbicule types are equally distributed among the species studied: seven species possess Type IV orbicules (Fig. 4A–H), six species Type V (Fig. 5A–F) and seven species Type VI (Figs 5G and H and 6A and B; Table 1).
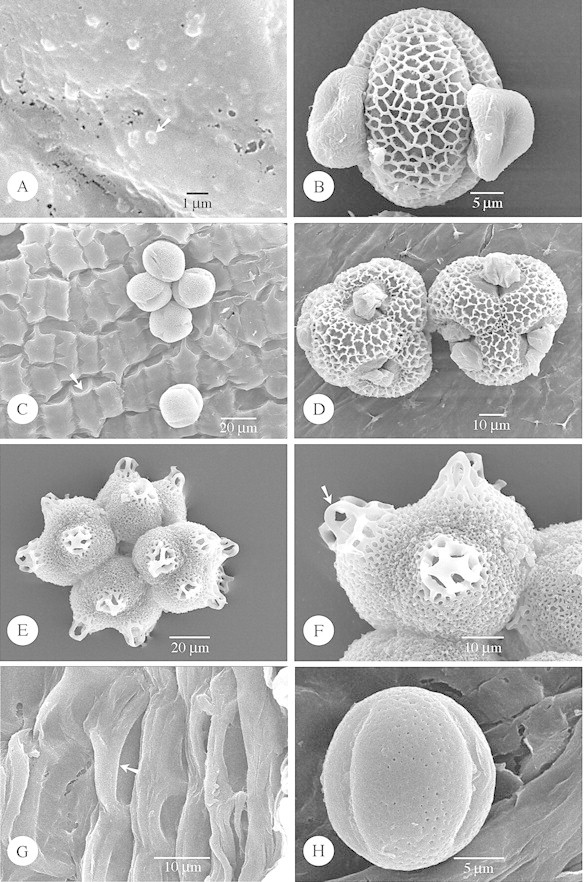
Fig. 6. SEM of species with strongly embedded Type VI (A and B) orbicules, and species lacking orbicules (C–H). A and B, Lisianthius nigrescens. A, General observation of the strongly embedded orbicules (arrow). B, Equatorial view of a reticulate pollen grain. C, Lomatogonium carinthiacum. General view of five pollen grains lying on the locule wall surface, which lacks orbicules. Deep grooves in the locule surface reveal the position of the underlying endothecial cells, the U‐shaped endothecial thickenings (arrow) can be distinguished. D, Schultesia guianensis. SEM observation of two reticulate tetrads lying on the smooth locule wall surface. E and F, Irlbachia purpurascens. E, General view of a micro‐reticulate polyad. F, Detailed observation of the muri of the reticulate pattern, at the distal regions, which are loop‐like. The loops of reticulum (arrow) are raised from the general surface of the pollen grain. G and H, Curtia tenuifolia. G, SEM observation of the locule surface consisting of wavy ridges (arrow). H, Equatorial view of a perforate pollen grain lying on the locule wall surface.
Pollen characters
The pollen grains are released as monads (Figs 2A and G, 3A and E, 4A, 5A and E, 6B, C and H and 7D), tetrads (Figs 3C, 4G, 6D and 7A) or polyads (Fig. 6E and F). Pollen grains are mostly 3‐colporate (Figs 2A and G, 3A and E, 4A, 5E and 6B, C and H) to 3‐porate (Fig 5A), but 2‐porate pollen (Fig. 7D) also occurs in the Gentianaceae. The colpi or pores are bordered with a distinct margo or annulus. The characteristics of the pollen sexine are summarized in Table 1, according to the terminology of Punt et al. (1994). In the palynologically diverse Gentianaceae a considerable variation in pollen ornamentation types is observed among the species studied. Most species possess a striate (Fig. 3E), striato‐reticulate (Figs 1F, 2B and G and 4A) to reticulate exine pattern (Figs 1E, 4G, 6B and D and 7A). Apart from these ornamentation types microreticulate (Fig. 6F), microspinulose (Fig. 1B), perforate (Figs 5A and 6H), psilate (Fig. 7D), rugulate (Fig. 5E) and granulate (Figs 3A and C) exine types are present. Of the Gentianaceae species studied by Sankara Rao and Chinnappa (1983), Hoppea fastigata (Griseb.) Clarke and Frasera albicaulis Douglas ex Griseb were also investigated in the present study. For both species the exine data substantiate the results of Sankara Rao and Chinnappa (1983). The granulate sexines may consist of numerous macroprocesses intermingled with microprocesses (Fig. 3C), or only of granules present on a psilate exine (Fig. 3A). The reticulate patterns can be heterobrochate (Figs 4G, 6F and 7A) or, as in most cases, homobrochate. The columellae of the reticulate patterns may be of variable length and the sexine organized in heterobrochate reticulate islands (Fig. 4G). In Irlbachia purpurascens (Aubl.) Maas (Fig. 6E and F) polar loops of reticulum that is raised from the general surface of the pollen grain are present. Lumina can be beset with granules (Figs 1E and 4G). Muri are either sharply crested (Fig. 1E) or rounded (Fig. 1F). The muri are oriented parallel (Fig. 3E) or in various directions (Fig. 2G), or criss‐crossed and interlaced (Figs 1F, 2B and 4A), in a few cases with granules present on the sidewalls of the muri (Fig. 2B).
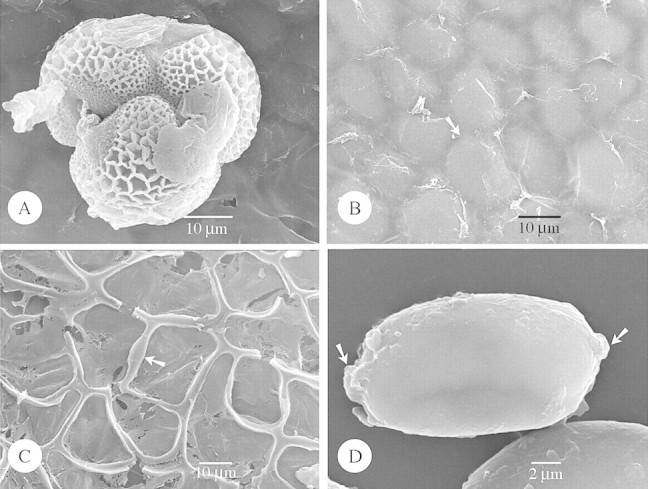
Fig. 7. SEM of species lacking orbicules. A and B, Coutoubea spicata. A, General observation of a heterobrochate reticulate tetrad. B, General observation of the locule wall surface lacking orbicules. Smooth grooves (arrow) in the locule surface reveal the position of the underlying endothecial cells. C, Halenia corniculata. The locule surface possesses ridges (arrow) forming more or less a reticulum. D, Voyria caerulea. General view of a psilate 2‐porate (arrows) pollen grain.
Locule surface characters
The appearance of the locule surface is often affected by dehydration. Consequently any definition of the surface morphology must be approached with caution. A ribbed surface, with the ribs parallel to the long axis of the anther, is a common feature in the group studied (Figs 4C and 5G). In some species the locule surface consists of wavy ridges (Figs 2A and 6G), in other species the ridges on the locule surface form a distinct reticulum (Fig. 7C). Sometimes smooth to deep grooves in the locule surface reveal the position of the underlying endothecial cells (Figs 6C and 7B) and the endothecial thickenings (Fig. 6C). Intermingled with the orbicules, small granules of sporopollenin may cover the locule wall surface (Fig. 5F). Imprints of pollen grains in the locule wall surface are also observed (Fig. 1H).
DISCUSSION
The 53 species studied out of approx. 1650 in Gentianaceae represent only 3·2 % of the species in this family. Despite the limited sample size, a good representation of the family is presented due to the fact that the selected species cover all the different tribes and subtribes recognized recently in the family (Struwe et al., 2002).
Correlations between pollen exine and orbicule surface ornamentation within the same species
As mentioned in the Introduction, a notable correlation in ornamentation between the pollen exine and the orbicule wall can be present. In the Gentianaceae species studied several examples of such a correlation were observed.
Species characterized by a granulate exine possess orbicules that resemble those granules. This correlation was observed in Tachia guianensis Aubl. and Prepusa montana Mart. (Fig. 3A–D). The orbicules present in Hockinia montana Gardner possess similar microspinulae on their orbicule wall to those that occur on the micro‐spinulose pollen tectum (Fig. 1A and B). The characteristics of the rugulate exine pattern in Exacum macranthum Arn. are reflected in the morphology of the orbicules present, which have also a rugulate appearance (Fig. 5E and F).
The irregularly folded orbicules (Type IV), present in species with striate, striato‐microreticulate or reticulate pollen grains, resemble the morphology of the elements comprising the muri of the pollen exine (Fig. 4A–H). In Calolisianthus pedunculatus (Cham. & Schltdl.) Gilg an extreme form of this correlation is observed: the orbicules consist of columellae‐like particles which support muri‐like parts (Fig. 4G and H). The surface of the orbicules is usually smooth. The species where this correlation occurs are Calolisianthus pedunculatus, Gentiana acaulis, G. algida Pall., G. alpina Adam, Sebaea albidiflora F Muell., S. ovata (Labill.) R. Br. and Tripterospermum fasciculatum (Wall.) A.O. Chater. In many Gentianaceae species with a striate or striato‐microreticulate exine pattern no correlation between the orbicule surface ornamentation and the pollen exine surface has been observed. The orbicules in these species can be regular (Figs 1C–H, 2 and 3E–H) or irregular in shape, and may possess sporopolleninous granulae on their orbicule wall surface (Fig. 5C and D). Spiny orbicules correlated with a striate exine, as reported in Dioscorea (Schols et al., 2001), do not occur in Gentianaceae.
Correlation of orbicule typology with trends in Gentianaceae palynology and systematic usefulness of orbicules
All specimens investigated within one species are characterized by the same orbicule type. No intraspecific variation has been observed.
No Gentianaceae tribe is characterized by a distinctive orbicule type: absence and different orbicule types occur in each tribe (Table 1), which reflects the observed palynological diversity in Gentianaceae. Tribe Saccifolieae is regarded as the most basal clade, followed by the clade Exaceae which is sister to the rest of Gentianaceae (Struwe et al., 2002). Both tribes are characterized by the presence of orbicules [excluding Curtia tenuifolia (Aubl.) Knobl., Fig. 6G and H; Table 1]. In Exaceae Type V (Fig. 5E and F) and Type IV (Fig. 4A–D) orbicules are recorded, in Saccifolieae Type I orbicules are present (Fig. 1A; Table 1). The tribe Chironieae is a more apomorphic clade, followed by the crown clade comprising the monophyletic tribes Helieae, Potalieae and Gentianeae. In this clade no particular sister‐group relationships are well supported (Struwe et al., 2002).
In tribe Chironieae, subtribe Chironiinae species possess orbicules of Type III, Type VI, or lack orbicules. In Canscorinae Type III and VI orbicules are present, and all Coutoubeinae species lack orbicules (Table 1).
In the majority of Gentianaceae genera monads are present, but tetrads have evolved twice, in Chironieae–Coutoubeinae (Coutoubea Aubl. and Schultesia Mart.) and in the tribe Helieae (Calolisianthus Gilg) (Struwe et al., 2002). The Chironieae–Coutoubeinae genera with tetrads are characterized by the absence of orbicules (see above) (Figs 6D, 7A and B). However, Calolisianthus (Helieae), which has tetrads, possesses orbicules (Fig. 4G and H). In Helieae polyads occur in Irlbachia Mart. (Fig. 6E and F). These polyads are regarded as being derived from tetrads (Struwe et al., 2002). Irlbachia is characterized by the absence of orbicules. Thus, comparable with the palynological trend observed in Apocynaceae s.l. (Vinckier and Smets, 2002b), a similar trend is observed in Helieae palynology from the genera with monads and tetrads possessing orbicules (Types III and IV), to the genus Irlbachia with polyads which lacks orbicules (Table 1). However, no comparable trend in orbicule typology, as recorded in Apocynaceae s.l. (Type III → embedded Type VI orbicules → orbicules absent) (Vinckier and Smets, 2002b), was present in the family Gentianaceae.
As a result of increasing molecular evidence (Struwe et al., 1994, 2002) the three genera Anthocleista Afzel. ex R. Br., Fagraea Thunb. and Potalia Aubl. have been included in Gentianaceae. However, this grouping seems unnatural from a palynological point of view (Nilsson, 2002), as Anthocleista (Fig. 5A) and Potalia possess 3(‐4)‐porate pollen grains, which is a pollen type not found in the rest of the Gentianaceae, although this type has arisen several times in Gentianales (Geniostoma J.R. Forst. & G. Forst., Rauvolfia L. (Fig. 1E of Vinckier and Smets, 2002b), Baissea A. DC.). On the other hand, Fagraea is characterized by 3‐colporate pollen, resembling gentianaceous pollen. In both genera Anthocleista and Fagraea the same orbicule type (Type V, Fig. 5A–D) is present, but in Potalia Type VI orbicules are observed (Fig. 52 of Vinckier and Smets, 2002a). The subtribes recognized in the tribe Potalieae (Struwe et al., 2002) are consistent with the orbicule typology: subtribe Faroinae lacks orbicules, Lisianthiinae possess Type VI orbicules (Fig. 6A and B), and Potaliinae are characterized by Type V orbicules (excluding Potalia) (Table 1).
In Gentianeae, the subdivision into subtribes Gentianinae and Swertiinae is strongly supported by molecular data (trnL intron, matK, ITS) (Struwe et al., 2002). Both subtribes are also characterized by the presence of distinct orbicule types: all Gentianinae species studied possess Type IV orbicules (Fig. 4E and F), and species of Swertiinae studied possess Type IIIa orbicules (Figs 1C–F and 3E–H), or lack orbicules (Figs 6C and 7C; Table 1).
Recognizing the systematic significance of the individual orbicule characters, the following points can be revealed. In Exaceae, the orbicules of each species have indentations and are aggregated. In the majority of Chironieae species indentations or aggregated orbicules are lacking and the orbicules possess a rounded oblate shape, similar to the orbicules of subtribe Swertiinae. In subtribe Gentianinae, aggregated angular irregular folded orbicules are present which lack indentations.
Swertia perennis possesses the smallest orbicules in the study group, and this species is known to have an amoeboid tapetum (Sankara Rao and Chinnappa, 1983). Also Canscora decussata (Roxb.) Schult. & Schult. f., characterized by an amoeboid tapetum (Maheswari Devi, 1962), produces very small orbicules (0·189 µm). It was observed that, in both species, the very small orbicules were distributed in a very low density on the locule wall surface. Thus it is suggested that presence of very small orbicules (mean 0·2 µm), along with a very low distribution density (mean 2 orbicules 25 µm–2), may give an indication of the presence of an amoeboid tapetum in the species, since sporopollenin granules are reported in taxa with an amoeboid tapetum (Tiwari and Gunning, 1986). Ontogenetical research may elucidate whether the small orbicules present in Canscora decussata and Swertia perennis are homologous with the sporopollenin granules produced by the amoeboid tapetum in Tradescantia (Tiwari and Gunning, 1986) or can be considered as orbicules. Additional information on the tapetum type of species lacking orbicules is required to answer the question as to whether orbicules are lacking in species with amoeboid tapeta.
CONCLUSIONS
Orbicule typology has proved to be useful for evaluating tribal delimitation within Rubiaceae (Huysmans et al., 1997; Vinckier et al., 2000) and Loganiaceae s.l. (Vinckier and Smets, 2002a); however, they seem not to be useful for tribal delimitation in Gentianaceae. In the tribes Potalieae and Gentianeae, orbicule typology may be useful at the subtribal level.
In the crown clade comprising Helieae, Potalieae and Gentianeae, no particular sister‐group relationships is well supported in the three‐gene analysis of Struwe et al. (2002). Further data are thus required to resolve these relationships. Despite the lack of congruence between orbicule and molecular data, individual orbicule characters along with pollen and other morphological characters can be used in a future combined analysis. Orbicule types should not be used in a cladistic analysis because they are aggregations of characters.
ACKNOWLEDGEMENTS
We thank the following people for excellent technical assistance: Dr Eric Schoeters (TEM, Leuven), Koen Collart (TEM, Leuven), and Ir. Marcel Verhaegen (SEM, National Botanic Garden of Belgium). Thanks are due to the curator of The National Botanic Garden of Belgium (BR) for the generous supply of anther samples. We are grateful for the valuable suggestions on the manuscript by Dr Carol Furness and Dr Favio Gonzàlez. This research was financially supported by projects No. 2·0038.91 (SEM) and G.0104.01 (general research project) from the Fund for Scientific Research‐Flanders (FWO) and a grant from the Research Council of the K.U.Leuven (OT/01/25)
APPENDIX
Alphabetical list of Gentianaceae specimens giving species, country, collector, specimen number and herbarium.
Anthocleista amplexicaulis Baker, Madagascar, A.J.M. Leeuwenberg & G.R. Rafamantanantsoa 14437 (BR)
Anthocleista scandens Hook. f., Madagascar, Bioko Fernando‐Po (s.n.) (BR)
Blackstonia perfoliata (L.) Huds., Algeria, D. Podlech 37064 (BR)
Calolisianthus pedunculatus (Cham. & Schltdl.) Gilg, Brazil, G. Hatschbach 47336 (BR)
Canscora decussata (Roxb.) Schult. & Schult. f., India, H.B. Naithouri 3954 (BR)
Canscora heteroclita (L.) Gilg, Sri Lanka, A.G. Robyns 7206 (BR)
Centaurium serpentinicola Carlström, Turkey, M. Nydegger 44139 (BR)
Centaurium spicatum (L.) Fernald, France, Berger 2310 (BR)
Centaurium tenuiflorum (Hoffmgg. & Link) Fritsch, France, C. Van den Berghen (s.n.) (BR); France, P. Bamps 7463 (BR); Portugal, J. Bouharmont 27871 (BR)
Chironia pegleriae Prain, South Africa, J. Lambinon & M. Reekmans 82/301 (BR)
Chironia purpurascens (E. Meyer) Benth. & Hook. f., South‐Africa, E.S. Kemp 818 (BR)
Coutoubea spicata Aubl., French Guyana, F. Billiet & B. Jadin 1145 (BR)
Curtia tenuifolia (Aubl.) Knobl., Brazil, G. Hatschbach 48278 (BR)
Enicostema verticillatum (L.) Engl. ex Gilg, Sri Lanka, N. Balakrishnan 53619 (BR)
Eustoma grandiflorum (Raf.) Shinners, USA, Geerinck‐Coutrez 4373 (BR)
Exacum gracilipes Balf. f., Oman, S.A. Ghazanfar 2337 (BR)
Exacum macranthum Arn., Sri Lanka, P.L. Comanor 419 (BR)
Exacum oldenlandioides (S. Moore) Klack, Guinea‐Bissau, Centro de botanico Lisboa 3761 (BR)
Fagraea racemosa Jack, Borneo, Veldkamp 8516 (BR)
Faroa affinis De Wild., Democratic Republic of Congo, Dikumbwa, Kisimba & Muzinga 3591 (BR)
Frasera albicaulis Douglas ex Griseb., USA, E.B. Babcock 1790 (BR)
Frasera parryi Torr., USA, V. Durand 3488 (BR)
Gentiana acaulis L., Switzerland, C. Vanden Berghen 30 (BR); France, G.F. De Witte 16457 (BR)
Gentiana algida Pall., Russia, Kharkevich (s.n.) (BR)
Gentiana alpina Adam, Spain, J. Auoyo, I. Fernandez & J. Pastor 11518 (BR)
Gentianella amarella (L.) Borner, Poland, S. Lisowski 57556 (BR)
Gentianella bellidifolia (Hook.f.) Holub, New Zealand, M.T. Mangaweka, P.D. Buckley & K.W. Julley 15/2/77 (BR)
Gentianopsis barbata (Froel.) Ma, Russia, Kharkevich & Buch (s.n.) (BR)
Gentianopsis procera (Th. Holm) Ma, Canada, F. Marie‐Victorin & F. Rolland‐Germain 49464 (BR)
Halenia corniculata (L.) Cornaz, Russia, Primorskiy, Kharkevich & Buch (s.n.) (BR)
Hockinia montana Gardner, Brazil, unknown 17150 (BR)
Hoppea fastigiata (Griseb.) Clarke, Sri Lanka, A.G. Robyns 7206bis (BR)
Irlbachia purpurascens (Aubl.) Maas, French Guyana, F. Billiet & B. Jadin 1550 (BR)
Ixanthus viscosus Griseb., Canary Islands, J. Bouharmont 25535 (BR)
Lisianthius areopolus B.L. Rob., Mexico, R.L. Wilbur 35600 (BR)
Lisianthius nigrescens Schltdl. & Cham., Mexico, Arnold arboretum, Universidad Nacional Autonoma de Mexico 446 (BR)
Lomatogonium carinthiacum (Wulfen) Rchb., Kazach stan, V. Zuev 6649 (BR)
Lomatogonium rotatum (L.) Fries ex Fern., Russia, S. Kharkevich & T. Buch (s.n.) (BR)
Macrocarpaea rubra Malme, Brazil, R. Kummrow 2385 (BR)
Obolaria virginica L., USA, H.E. Ahles & J. Haesloop 53200 (BR)
Potalia amara Aubl., French Guyana, collector of the Orstom centre 1188 (BR); French Guyana, F. Billiet & B. Jadin 1821 (BR); F. Billiet & B. Jadin 1900 (BR)
Prepusa montana Mart., Brazil, G. Hatschbach 47930 (BR)
Sabatia bartramii Wilbur, USA, S. Mc Daniel 6455 (BR)
Sabatia brachiata Elliott, USA, A.E. Radford 45410 (BR)
Sabatia quadrangula Wilbur, USA, J.F. Matthews, L. Kesler, P. Campbell & W. Weston (s.n.) (BR)
Schultesia guianensis (Aubl.) Malme, French Guyana, F. Billiet & B. Jadin 1157 (BR)
Schultesia pachyphylla Grisrb., Brazil, G. Hatschbach 47936 (BR)
Sebaea albidiflora F Muell., Australia, A. Mubold 7276 (BR)
Sebaea ovata (Labill.) R. Br., D.J.E. Whibley 8862 (BR)
Swertia bimaculata (Siebold & Zucc.) Hook. f. & Thomson ex C.B. Clarke, China, Sino‐American Guizhou Botanical Expedition 402 (BR)
Swertia perennis L., France, J. Bouharmont 7718 (BR)
Tachia guianensis Aubl., Brazil, P. Bamps 5416 (BR); Peru, F. Woytkowski 5348 (BR); Peru, S. Mc Daniel & M. Rimachi 16483 (BR)
Tripterospermum fasciculatum (Wall.) A.O. Chater, India, G. Panigrahi 19807 (BR)
Voyria caerulea Aubl., French Guyana, F. Billiet & B. Jadin 4381 (BR)
Supplementary Material
Received: 14 April 2003;; Returned for revision: 14 May 2003. Accepted: 28 July 2003; Published electronically: 19 September 2003
References
- ArchangelskyS, Taylor TN.1993. The ultrastructure of in situ Clavatipollenites pollen from the early Cretaceous of Patagonia. American Journal of Botany 80: 879–885. [Google Scholar]
- ChristensenJE, Horner HT Jr, Lersten NR.1972. Pollen wall and tapetal orbicular wall development in Sorghum bicolor (Gramineae). American Journal of Botany 59: 43–58. [Google Scholar]
- ClémentC, Audran J‐C.1993a. Cytochemical and ultrastructural evolution of orbicules in Lilium Plant Systematics and Evolution 7: 63–74. [Google Scholar]
- ClémentC, Audran J‐C.1993b. Orbicule wall surface characteristics in Lilium (Liliaceae). Grana 32: 348–353. [Google Scholar]
- ClémentC, Audran, J‐C.1993c. Electron microscope evidence for a membrane around the core of the Ubisch body in Lilium (Liliaceae). Grana 32: 311–314. [Google Scholar]
- EchlinP, Godwin H.1968.The ultrastructure and ontogeny of pollen in Helleborus foetidus L. I. The development of the tapetum and Ubisch bodies. Journal of Cell Science 3: 161–174. [DOI] [PubMed] [Google Scholar]
- El‐GhazalyG.1989. Pollen and orbicule morphology of some Euphorbia species. Grana 28: 243–259. [Google Scholar]
- El‐GhazalyG, Chaudhary R.1993. Morphology and taxonomic application of orbicules (Ubisch bodies) in the genus Euphorbia Grana Suppl. 2: 26–32. [Google Scholar]
- El‐GhazalyG, Jensen WA.1986. Studies of the development of wheat (Triticum aestivum) pollen. Grana 25: 1–29. [Google Scholar]
- ErdtmanG, Berglund B, Praglowski J.1961.An introduction to a Scandinavian pollen flora. Stockholm: Almqvist and Wiksell. [Google Scholar]
- FurnessC, Rudall P.2001. The tapetum in basal angiosperms: early diversity. International Journal of Plant Sciences 162: 375–392. [Google Scholar]
- HesseM.1985. Hemispheric surface processes of exine and orbicules in Calluna (Ericaceae). Grana 24: 93–98. [Google Scholar]
- HesseM.1986. Orbicules and the ektexine are homologous sporopollenin concretions in Spermatophyta. Plant Systematics and Evolution 153: 37–48. [Google Scholar]
- HuysmansS, El‐Ghazaly G, Nilsson S, Smets E.1997. Systematic value of tapetal orbicules: a preliminary survey of the Cinchonoideae (Rubiaceae). Canadian Journal of Botany 75: 815–826. [Google Scholar]
- HuysmansS, El‐Ghazaly G, Smets E.1998. Orbicules in angiosperms. Morphology, function, distribution, and relation with tapetum types. Botanical Review 64: 240–272. [Google Scholar]
- KosmathL.1927. Studie über das Antherentapetum. Österrechische Botanische Zeitschrift 76: 235–241. [Google Scholar]
- LombardoG, Carraro L.1976. Tapetal ultrastructural changes during pollen development. III. Studies on Gentiana acaulis Caryologia 29: 345–349. [Google Scholar]
- MasheswariDeviH.1962. Embryological studies in Gentianaceae (Gentianoideae and Menyanthoideae). Proceedings of the Indian Academy of Sciences, Sect. B, 60: 52–65. [Google Scholar]
- NilssonS.1967. Pollen morphological studies in Gentianaceae‐Gentianinae. Grana Palynologica 7: 46–145. [Google Scholar]
- NilssonS.2002. Gentianaceae: a review of palynology. In: Struwe L, Albert VA, eds. Gentianaceae. Systematics and natural history Cambridge: Cambridge University Press, 377–497. [Google Scholar]
- NilssonS, Robyns A.1974. Pollen morphology and taxanomy of the genus Quararibea s.l (Bombacaceae). Bulletin du Jardin Botanique National de Belgique – Bulletin van de Nationale Plantentuin van België 44: 77–99. [Google Scholar]
- OsbornJM, Taylor TN, Crane PR.1991. The ultrastructure of Sahnia pollen (Pentoxylales). American Journal of Botany 78: 1560–1569. [Google Scholar]
- PaciniE.1997. Tapetum character states: analytical keys for tapetum types and activities. Canadian Journal of Botany 75: 1448–1459. [Google Scholar]
- PaciniE, Franchi GG, Hesse M.1985. The tapetum: its form, function and possible phylogeny in embryophyta. Plant Systematics and Evolution 149: 155–185. [Google Scholar]
- PuntW, Blackmore S, Nilsson S, Le Thomas A.1994.Glossary of pollen and spore terminology. Utrecht: LPP Foundation. [Google Scholar]
- RajB, El‐Ghazaly G.1987. Morphology and taxonomic application of orbicules (Ubisch bodies) in Chloanthaceae. Pollen and Spores 29: 151–166. [Google Scholar]
- RosanoffS.1865. Zur kenntniss des baues und der entwickel ungsgeschichte des pollens der Mimosaceae. Jahrbücher für Wisschen schaft Botaniker 4: 441–450. [Google Scholar]
- RowleyJR.1990. The fundamental structure of the pollen exine. Plant Systematics and Evolution Suppl. 5: 13–29. [Google Scholar]
- SankaraRaoK, Chinnappa CC.1983. Studies in Gentianaceae. Microsporangium and pollen. Canadian Journal of Botany 61: 324–336. [Google Scholar]
- ScholsP, Dessein S, D’Hondt C, Huysmans S, Smets E.2002. Carnoy: a new digital measurement tool for palynology. Grana 41: 124–126. [Google Scholar]
- ScholsP, Furness CA, Wilkin P, Huysmans S, Smets E.2001. Morphology of pollen and orbicules in some Dioscorea species and its systematic implications. Botanical Journal of the Linnean Society 136: 295–311. [Google Scholar]
- SerbetR, Stockey RA.1991. Taxodiaceous pollen cones from the Upper Cretaceous (Horseshoe Canyon Formation) of Drumheller, Alberta, Canada. Review of Palaeobotany and Palynology 70: 67–76. [Google Scholar]
- SporneKR.1973. A note on the evolutionary status of tapetal types in dicotyledons. New Phytologist 72: 1173–1174. [Google Scholar]
- StruweL, Albert VA, Bremer B.1994.Cladistics and family level classification of the Gentianales. Cladistics 10: 175–206. [Google Scholar]
- StruweL, Kadereit JW, Klackenberg J, Nilsson S, Thiv M, Von Hagen KB, Albert VA.2002. Systematics, character evolution, and biogeography of Gentianaceae, including a new tribal and subtribal classification. In: Struwe L, Albert VA, eds. Gentianaceae. Systematics and natural history Cambridge: Cambridge University Press, 21–309. [Google Scholar]
- TaylorTN, Alvin KL.1984. Ultrastructure and development of Mesozoic pollen, Classopollis American Journal of Botany 71: 575–587. [Google Scholar]
- TiwariSC,Gunning BES.1986. An ultrastructural, cytochemical and immuno‐fluorescence study of postmeiotic development of plasmodial tapetum in Tradescantia virginiana L. and its relevance to the pathway of sporopollenin secretion. Protoplasma 133: 100–114. [Google Scholar]
- VijayaraghavanMR, Chaudhry B.1993. Structure and development of orbicules in the tapetum of Prosopsis juliflora (Leguminosae, Mimosoideae). Phytomorphology 43: 41–48. [Google Scholar]
- VinckierS.2003.Orbicules: morphology, ultrastructure, and develop ment in Gentianales and their possible role as vector of allergens. PhD Thesis, K. U. Leuven. [Google Scholar]
- VinckierS, Smets E.2002a. Morphology, ultrastructure and typology of orbicules in family Loganiaceae s.l and related genera, in relation to systematics. Review of Palaeobotany and Palynology 119(3–4): 161–189. [Google Scholar]
- VinckierS, Smets E.2002b. Morphological and ultrastructural diversity of orbicules in relation to evolutionary tendencies in Apocynaceae s.l. Annals of Botany 90: 647–662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VinckierS, Smets E.2002c. Systematic importance of orbicule diversity in Gentianales. Grana 41: 158–182. [Google Scholar]
- VinckierS, Huysmans S, Smets E.2000. Morphology and ultrastructure of orbicules in the subfamily Ixoroideae (Rubiaceae). Review of Palaeobotany and Palynology 108: 151–174. [DOI] [PubMed] [Google Scholar]
- ZetterR, Weber M, Hesse M, Pingen M.2002. Pollen, Pollenkitt, and Orbicules in Craigia bronnii flower buds (Tilioideae, Malvaceae) from the Miocene of Hambach, Germany. International Journal of Plant Sciences 163(6): 1067–1071. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


