Abstract
Background
Congenital Long QT syndrome (LQTS) is an arrhythmogenic disorder that causes syncope and sudden death. While its genetic basis has become well-understood, the mechanisms whereby mutations translate to arrhythmia susceptibility in the in situ human heart have not been fully defined. We used noninvasive ECG imaging (ECGI) to map the cardiac electrophysiologic substrate and examine whether LQTS patients display regional heterogeneities in repolarization, a substrate which promotes arrhythmogenesis.
Methods and Results
25 subjects (9 LQT1, 9 LQT2, 5 LQT3 and 2 LQT5) with genotype and phenotype positive LQTS underwent ECGI. Seven normal subjects provided control. Epicardial maps of activation, recovery times (RT), Activation-recovery intervals (ARI) and repolarization dispersion were constructed. Activation was normal in all patients. However, RT and ARI were prolonged relative to control, indicating delayed repolarization and abnormally long APD (312 ± 30 ms vs. 235 ± 21 ms in control). ARI prolongation was spatially heterogeneous, with repolarization gradients much steeper than control (119 ± 19 ms/cm vs. 2.0 ± 2.0 ms/cm). There was variability in steepness and distribution of repolarization gradients between and within LQTS types. Repolarization gradients were steeper in symptomatic patients (130 ± 27 ms/cm in 12 symptomatic patients vs. 98 ± 19 ms/cm in 13 asymptomatic patients; P < 0.05).
Conclusions
LQTS patients display regions with steep repolarization dispersion caused by localized APD prolongation. This defines a substrate for reentrant arrhythmias, not detectable by surface ECG. Steeper dispersion in symptomatic patients suggests a possible role for ECGI in risk stratification.
Keywords: congenital long-QT syndrome, arrhythmia (mechanisms), electrophysiology mapping, noninvasive imaging, ECGI
Introduction
Congenital Long QT syndrome (LQTS) is an inherited cardiac channelopathy which affects 1 in 20001 patients worldwide, predisposing healthy young adults with structurally normal hearts to syncope and sudden cardiac death due to polymorphic ventricular tachycardia (torsades de pointes).
Molecular and genetic studies in humans have uncovered many of the genes2 associated with LQTS, thus providing molecular insight into the pathogenesis of the disease. LQT1 is caused by loss of function mutations in KCNQ13, which encodes the α subunit of the slowly activating potassium channel IKs, leading to reduced IKs current. Similarly, LQT2 is associated with loss of function mutations in KCNH2 (hERG)3 and a reduced rapidly activating potassium current IKr. LQT3 is caused by gain of function mutations in SCN5A4 and increased INa current during the plateau and late phase of the action potential (AP). The rare LQT5 is associated with mutations in the KCNE1 (mink)4, which encodes the auxiliary β subunit of the IKs channel, causing loss of current. The loss of repolarizing current (LQT1, LQT2 and LQT5) or the increase in depolarizing current (LQT3) leads to prolongation of the ventricular AP duration (APD) and delayed ventricular repolarization, which is reflected in a long QT interval on the ECG (the LQTS phenotype).
The diagnostic characteristic for a positive phenotype is prolongation of the QT interval (corrected for heart rate using Bazett's equation; QTc) on the 12-lead ECG, with values over 450 ms in male and 460 ms in female5. QT interval longer than 500 ms indicates increased arrhythmia risk6. Diagnosis can also be based on the Schwartz score2 which includes patient's age, medical and family history, symptoms and QTc. Genotype-specific changes in T- wave morphology7,8 that are suggestive but not diagnostic (broad-based in LQT1, notched in LQT2, late-appearing in LQT3) and occasional alternans are present on the body-surface ECG. Clinical arrhythmia triggers are distinct for each subtype9. Importantly, while genetic testing is increasingly used in the diagnosis, it is not useful for risk stratification among mutation carriers since the disease is characterized by incomplete penetrance and variable expressivity. Experiments10,11,12 and modeling studies7,13-16 have shed valuable light on the cellular and tissue-level mechanisms of arrhythmia in LQTS. Prolongation of the AP leading to the development of early after depolarizations (EADs)17 has been proposed as a trigger, and amplification of spatial dispersion of repolarization has been proposed as the substrate for development of reentry. Body surface potential mapping in LQTS patients has demonstrated multiple distributions and high nondipolar content, suggestive of regional electrical disparities in the heart18. However, the arrhythmogenic substrate in the intact human heart of patients with congenital LQTS has not been characterized due to lack of noninvasive, high resolution, panoramic mapping techniques. In the absence of such methods, attempts have been made to infer dispersion of repolarization in LQTS patients from body-surface ECG characteristics19 such as QTc dispersion (difference between the maximum and minimum QTc on the 12-lead ECG). Because the signal in each ECG lead is generated by the integrated activity over the entire heart, geometrical relationships of repolarization in the heart are not preserved in the body surface ECG. Hence, the surface ECG cannot distinguish between regional and global repolarization abnormalities, and global ECG-based measures such as QTc dispersion are not well defined and indeed have not proven useful. In this study, we used noninvasive Electrocardiographic Imaging (ECGI; also called Electrocardiographic Mapping, ECM) to map the electrophysiologic (EP) substrate in patients with genotype positive, phenotype positive LQTS. It has been established, through canine experiments, that ECGI can noninvasively reconstruct repolarization properties accurately and localize areas of increased dispersion of repolarization in the heart20. In the intact human heart, ECGI has been used to map normal repolarization21 and conditions associated with altered repolarization, including WPW syndrome22 and ventricular pacing23.
We hypothesized that the EP substrate in patients with hereditary LQTS comprises of regions with delayed repolarization and steep spatial dispersion of repolarization on the ventricular epicardium, providing the substrate for reentrant arrhythmias and torsades de pointes.
Methods
Subject enrollment
25 patients participated in the study (9 LQT1, 9 LQT2, 5 LQT3 and 2 LQT5). Twenty four of them underwent ECGI at Washington University in St. Louis (six were referred by Vanderbilt University School of Medicine and drove to St. Louis). One patient underwent ECGI at Bordeaux University Hospital and raw data was forwarded electronically to Washington University for processing, epicardial reconstruction and analysis. All patients were genotype positive (class I mutation in KCNQ1, KCNH2, SCN5A and KCNE1) and phenotype positive for LQTS (QTc ≥ 450 ms male, ≥ 460 ms female). Subjects with (symptomatic) or without (asymptomatic) history of syncope or sudden cardiac arrest and appropriate defibrillation were included. Table 1 summarizes the clinical characteristics of the LQTS group. The control group21 consisted of seven healthy volunteers with normal hearts (4 male, 3 female; mean QTc: 384 ± 12 ms) and no history of cardiac events. All subjects signed a written informed consent and all protocols were reviewed and approved by the Human Research Protection Office at Washington University in Saint Louis and Bordeaux University Hospital.
Table 1.
Clinical Characteristics of the LQTS Subjects.
| Gene | Mutation | LQTS Type | ID | Age (YRS.) | Sex | QTc (ms) | Cardiac Events (Symptoms) | Therapy |
|---|---|---|---|---|---|---|---|---|
| KCNQ1 | 340delF | LQT1 | 14 | 40 | F | 485 | none (found during family screen) | Toprolol |
| KCNQ1 | 340delF | LQT1 | 15 | 11 | F | 541 | none (found during family screen) | Nadolol |
| KCNQ1 | G26D | LQT1 | 19 | 23 | F | 458 | VF and appropriate ICD shock | ICD* |
| KCNQ1 | T312I | LQT1 | 20 | 59 | F | 520 | none (found during family screen) | None |
| KCNQ1 | T312I | LQT1 | 21 | 37 | F | 488 | several episodes of syncope (index case for family) | Propranolol |
| KCNQ1 | T312I | LQT1 | 23 | 7 | M | 563 | none (found during family screen) | Atenolol |
| KCNQ1 | Q359X | LQT1 | 18 | 39 | F | 516 | none (found during family screen) | Metoprolol + ICD* |
| KCNQ1 | Unknown | LQT1 | 13 | 16 | F | 495 | none (found during family screen) | Nadolol |
| KCNQ1 | Unknown | LQT1 | 24 | 22 | F | 481 | none (found during family screen) | Nadolol |
| KCNH2 | H70R | LQT2 | 3 | 28 | M | 469 | none (found during family screen) | Nadolol + ICD* |
| KCNH2 | H70R | LQT2 | 6 | 51 | F | 503 | palpitations at rest | Atenolol + ICD* |
| KCNH2 | Q376sp | LQT2 | 1 | 11 | M | 558 | presycope (index case for family) | Nadolol |
| KCNH2 | Q376sp | LQT2 | 4 | 39 | F | 463 | presyncope | Nadolol |
| KCNH2 | Q376sp | LQT2 | 5 | 10 | M | 481 | none (found during family screen) | Nadolol |
| KCNH2 | S783P | LQT2 | 25 | 27 | F | 493 | several episodes of syncope | Nadolol |
| KCNH2 | V796G | LQT2 | 11 | 33 | F | 488 | several episodes of VF (index case for family) | Metoprolol + ICD* |
| KCNH2 | Y652X | LQT2 | 16 | 46 | M | 480 | syncope and seizure | Lopressor |
| KCNH2 | Y652X | LQT2 | 17 | 16 | M | 500 | none (found during family screen) | Nadolol |
| SCN5A | E1784K | LQT3 | 2 | 20 | M | 500 | presyncope | ICD* |
| SCN5A | E1784K | LQT3 | 7 | 34 | M | 478 | VF and appropriate shock | ICD* |
| SCN5A | E1784K | LQT3 | 12 | 59 | M | 533 | none (found during family screen) | ICD* |
| SCN5A | R1644H | LQT3 | 8 | 36 | F | 496 | palpitations and syncope (index case for family) | Metoprolol + ICD* |
| SCN5A | R1644H | LQT3 | 22 | 8 | M | 543 | none (found during family screen) | Nadolol |
| KCNE1 | D76N | LQT5 | 9 | 35 | F | 486 | none (found during family screen) | ICD* |
| KCNE1 | D76N | LQT5 | 10 | 58 | F | 500 | syncope | ICD* |
The patients with ICD were not paced. The average duration of ICD implant was 5 ± 3 years at the time of the ECGI study
Electrocardiographic Imaging
The ECGI methodology has been described previously21,22,23. Briefly, 256 body-surface ECGs were recorded in sinus rhythm. Following this, the patients underwent a thoracic CT scan gated at 70% of the R-R interval while wearing the electrodes. The patient-specific heart-torso geometry, digitized from the CT images, was combined with the body surface potentials using custom-developed mathematical algorithms to noninvasively reconstruct electric potentials and 502 ventricular unipolar electrograms (EGMs) on the epicardial surface of the heart.
Analysis
For each subject, the epicardial EGMs were analyzed as described below. Local activation time (AT) was determined from the time of steepest negative deflection (-dV/dt) during the QRS complex. Local recovery time (RT) was determined from the time of maximum dV/dt during the upstroke of the T wave. As shown, this determination is independent of T wave polarity and morphology24. The local activation-recovery interval (ARI), shown to be a surrogate for APD24,25,26, was computed as the difference between local AT and local RT for each epicardial site. The ARI values were corrected for heart rate using Bazett's formula (ARIc = ARI/(RR0.5); RR = R-R interval). From the RT map and ARI map, epicardial dispersion of repolarization was computed as the maximum differences ΔRT and ΔARIc between two adjacent EGM sites on the epicardium. Their gradients (ΔRT/Δx, ΔARIc/Δx) were computed through division by the distance Δx between the two adjacent sites.
Statistics
Continuous variables (AT, RT, ARIc, ΔRT, ΔARIc, ΔRT/Δx, ΔARIc/Δx) were plotted as mean ± standard deviation. A one-way ANOVA was used to identify group differences. All pairwise comparisons between LQT groups and control were conducted (4 tests) with Bonferroni correction to control the type I error. An unpaired two-tailed Satterthwaite's modified t-test was used to quantify the differences in mean ECGI-derived steepness of repolarization gradients and differences in mean body-surface QTc between symptomatic and asymptomatic LQTS patients. All tests were considered statistically significant if P < 0.05.
Results
The results section focuses on the more common types of LQTS (LQT1, 2 and 3); results from two patients with the rare LQT5 are also presented. The figures below provide representative examples; Supplemental Figures 2-5 provide repolarization (ARI) maps for all individual patients in each LQTS group. Supplemental Tables 1, 2 and 3 contain results for all LQT1, LQT2 and LQT3 patients, respectively, and Supplemental Table 4 (Section 3) relates ECGI derived parameters to cardiac events history for family members with the same mutation. Online Supplement Section 4 provides results from statistical analysis. Graphs from multiple comparison tests between control and the four LQTS groups are provided for each variable being compared. A sensitivity analysis was conducted using the natural logarithm for all continuous variables due to concern of variance heterogeneity between groups. The results of all pairwise comparisons, after Bonferroni correction, were consistent with the unadjusted (non-logarithmic) analyses. These results are also included in Section 4 of the Online Supplement.
Epicardial Activation Patterns
Figure 1 shows representative ECGI epicardial activation isochrone maps for control, LQT1 (patient 21), LQT2 (patient 3) and LQT3 (patient 7). The epicardial activation pattern in LQTS during sinus rhythm was characterized by normal right ventricular (RV) breakthrough (marked by *) followed by rapid activation of the ventricles, with the excitation spreading uniformly without lines of block, regions of delayed activation, or regions of slow conduction (isochrone crowding). The latest region to activate was the left ventricular (LV) base. This sequence of activation was the same as that of the control group. The total ventricular activation time, measured as the difference between latest and earliest AT, was around 50 ms - comparable to control (Figure 1). The mean ventricular activation times (Supplemental Tables 1, 2 and 3) were 54 ± 5 ms, 49 ± 9 ms and 55 ± 11 ms for LQT1, LQT2 and LQT3 groups, respectively; control was 47 ± 9 ms. Similarly, the mean ventricular activation time for LQT5 was 53 ± 8 ms. These results indicate that ventricular activation in all LQTS types was normal.
Figure 1.

Epicardial Activation Times (AT) Isochrone Maps. Examples of activation in (left to right) control, LQT1 (patient 21), LQT2 (patient 3), and LQT3 (patient 7). In all LQTS types as in normal control, epicardial activation starts from breakthrough at anterior RV (shown by white asterisk) near the RVOT region, 20-30 ms after the onset of QRS. It proceeds in a uniform fashion to activate the ventricles synchronously. The latest region to activate is the LV basal region (dark blue). The total ventricular activation time (TVAT) in all LQTS types was around 50 ms, comparable to normal control. RA = right atrium, LA = left atrium, RV = right ventricle, LV = left ventricle, AO = aorta, ms = milliseconds.
Epicardial Recovery Pattern
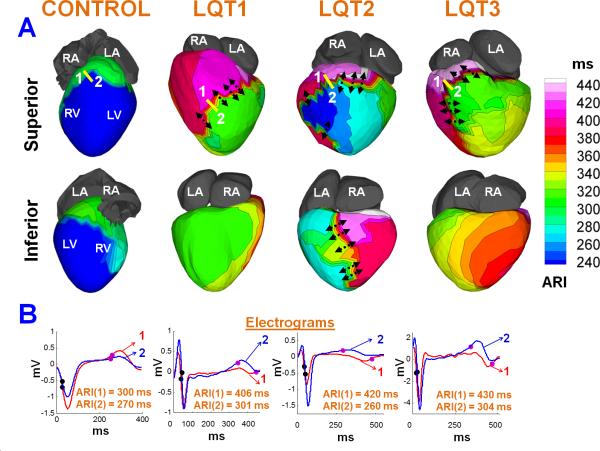
Figure 2 shows representative epicardial RT maps for control, LQT1 (patient 15), LQT2 (patient 16) and LQT3 (patient 8) in superior and inferior views. There were marked local changes in recovery pattern in all LQTS types compared to control; the LQTS subjects showed regions of delayed ventricular recovery on the epicardium. For instance, maximum RT in the LQT1 subject was 443 ms compared to 328 ms in control. This led to recovery time differences (ΔRT) that were abnormally large compared to control. For instance, maximum ΔRT in LQT1 was 100 ms (Figure 2) compared to only 28 ms in control. This resulted in a steep gradient of recovery in LQTS (shown by black arrows in figure 2) which was much greater than that of control (Table 2). The mean RT values in the three LQT groups were 372 ± 30 ms in LQT1, 369 ± 42 ms in LQT2 and 404 ± 35 ms in LQT3 compared to control value of 257 ± 15 ms (P < 0.05 for each LQTS type versus control). The mean ΔRT values were 95 ± 19 ms in LQT1, 134 ± 37 ms in LQT2 and 112 ± 9 ms in LQT3 compared to 16 ± 12 ms in control (P < 0.05). The mean ΔRT/Δx values were 89 ± 18 ms/cm in LQT1, 115 ± 25 ms/cm in LQT2 and 133 ± 6 ms/cm in LQT3 compared to only 5.0 ± 3.0 ms/cm in control (P < 0.05). The mean RT, ΔRT and ΔRT/Δx for the two LQT5 patients were 343 ± 24 ms, 136 ± 23 ms and 134 ± 13 ms/cm respectively, as shown in Table 2. Online Supplement Section 4 provides graphs of pairwise comparison of mean RT, ΔRT and ΔRT/Δx between control and the four LQTS groups. Note that the morphologies of EGMs from locations across the region of steep dispersion differ markedly (figure 2, panel B). In particular, EGMs in region 1 of delayed repolarization exhibit a predominantly negative T wave compared to EGMs from region 2. This reflects a change in the direction of the voltage gradient across the region of steep dispersion. As shown24, the maximum dV/dt determines recovery time independent of T-wave polarity.
Figure 2.
Epicardial Recovery Time (RT) Maps. A. Maps are shown in superior (top row) and inferior (bottom row) views for control, LQT1 (patient 15), LQT2 (patient 16), and LQT3 (patient 8). All three LQTS subjects had regions with abnormally long RT as shown by predominant magenta and white colors in the maps. The maximum RT value in LQTS was 470 ms. The maximum RT value in the normal heart (left most column) was 360 ms (predominant blue and green colors in the map). The heterogeneity in ventricular recovery resulted in large RT differences in all three LQTS types. The solid yellow line (top panels) connects two closest neighboring EGMs (from site 1 and site 2) with maximum ΔRT. In all three LQTS patients, ΔRT (RT(1)-RT(2)) exceeded 100 ms (compared to normal value of only 28 ms in the left most column). As a result, there was a steep gradient of repolarization ΔRT/Δx across this region (shown by black arrows); it was much steeper than control (Normal: 6 ms/cm, LQT1: 102 ms/cm, LQT2: 159 ms/cm, LQT3: 139 ms/cm). B. ECGI-reconstructed unipolar EGMs from the three LQT patients exhibited drastic changes in T-wave morphology across the yellow line. The T waves obtained from site 1 (red) were inverted or predominantly negative compared to those from site 2 (blue; upright or predominantly positive). Such T-wave changes over a short distance (<10 mm) were absent in the control group. RT (time of dV/dt max during upstroke of T wave) is indicated by the pink dot on the corresponding EGMs (site 1 red; site 2 blue). Corresponding 12-lead ECG tracings are provided in Supplemental Figure 1. mV = millivolts.
Table 2.
Summary of repolarization parameters for all groups.
| Parameter (Mean ± SD) | NORMAL (n=7) | LQT1 (n=9) | LQT2 (n=9) | LQT3 (n=5) | LQT5 (n=2) |
|---|---|---|---|---|---|
| RT (ms) | 257± 15 | 372 ± 30 | 369 ± 42 | 404 ± 35 | 343 ± 24 |
| ΔRT (ms) | 16 ± 12 | 95 ± 19 | 134 ± 37 | 112 ± 9 | 136 ± 23 |
| ΔRT/Δx (ms/cm) | 5.0 ± 3.0 | 89 ± 18 | 115 ± 25 | 133 ± 6 | 134 ± 13 |
| ARIc (ms) | 235 ± 21 | 316 ± 28 | 307 ± 36 | 335 ± 18 | 288 ± 36 |
| ΔARIc (ms) | 19 ± 13 | 99 ± 20 | 136 ± 36 | 110 ± 14 | 139 ± 28 |
| ΔARIc/Δx (ms/cm) | 2.0 ± 2.0 | 92 ± 18 | 117 ± 29 | 129 ± 14 | 137 ± 17 |
All results were statistically significant (P < 0.05 for each LQTS type versus control). However, LQT5 group had only two patients. The difference in mean ARIc between control and LQT5 group was not statistically significant. Online Supplement Section 4 provides graphs showing pairwise comparisons between control and each LQTS group.
Activation-Recovery Intervals
Figure 3 shows the ARI maps for the patients of figure 2 in two views. ARI was computed at each epicardial site by subtracting local AT from local RT and was corrected for heart rate. ARI reflects local repolarization and was shown to be a surrogate for local APD24,25,26. Note that the RT maps (Figure 2) and the ARI maps (Figure 3) are very similar. RT is determined by both the activation sequence and local repolarization, while ARI is determined by local repolarization only (independent of the activation sequence). Therefore, the close similarity of the maps indicates that local repolarization is the major determinant of the repolarization sequence in LQTS. A similar property was observed in the normal human heart21. However, as seen from figure 3, all LQTS subtypes had regions with significant prolongation of ARI compared to control. For instance, the maximum ARIc in LQT3 (Figure 3) was 430 ms compared to control value of 320 ms. The maximum ARI dispersion in each LQTS group (shown by black arrows) was much greater than that of control (Table 2). The location of maximum ARI gradient varied within and among the LQTS types as shown in Supplemental Figures 2, 3, 4 and 5. The mean ARIc values in the three LQT groups were 316 ± 28 ms in LQT1, 307 ± 36 ms in LQT2 and 335 ± 18 ms in LQT3 compared to 235 ± 21 ms in the control group (P < 0.05 for each LQTS type versus control). The mean ΔARIc values were 99 ± 20 ms in LQT1, 136 ± 36 ms in LQT2, 110 ± 14 ms in LQT3 compared to 19 ± 13 ms in control (P < 0.05). The mean ΔARIc/Δx values were 92 ± 18 ms/cm in LQT1, 117 ± 29 ms/cm in LQT2, 129 ± 14 ms/cm in LQT3 compared to only 2.0 ± 2.0 ms/cm in control (P < 0.05). The mean ARI, ΔARI and ΔARI/Δx for the two LQT5 patients were 288 ± 36 ms, 139 ± 28 ms and 137 ± 17 ms/cm, respectively (Table 2). Online Supplement Section 4 provides graphs of pairwise comparison of mean ARI, ΔARI and ΔARI/Δx between control and the four LQTS groups.
Figure 3.
Activation-Recovery Interval (ARI) Maps. A. Maps are shown in superior (top row) and inferior (bottom row) views for the patients of figure 2. ARI (surrogate for local APD) values were abnormally long (magenta and white regions) in all three LQTS patients compared to control. The maximum ARI value in LQTS was 450 ms compared to only 340 ms (green in the left most column) in control. The localized prolongation of APD resulted in large ARI differences in all three LQTS types. The solid yellow line (top panels) connects two closest neighboring EGMs (from site 1 and site 2) with maximum ΔARIc. In all three LQTS patients, ΔARIc (ARIc(1)-ARIc(2)) exceeded 100 ms (compared to normal ΔARIc of only 30 ms in the left most column). As a result, there was a steep gradient of repolarization ΔARIc/Δx across this region (indicated by black arrows) which was two orders of magnitude greater than control (Normal: 7 ms/cm, LQT1: 104 ms/cm, LQT2: 146 ms/cm, LQT3: 140 ms/cm). B. The ECGI-reconstructed EGMs depict the time instances of AT (black dots) and RT (pink dots). The corresponding ARI values (RT – AT) are indicated below.
Discussion
This is the first study of its kind, characterizing the electrophysiologic substrate in patients with hereditary LQTS. Noninvasive ECGI made it possible to map with high resolution the entire ventricular epicardium of the intact heart in unanaesthetized patients. The panoramic mapping was essential for the characterization of the substrate.
The results indicate that there is significant prolongation of the action potential on the ventricular epicardium of congenital LQTS patients compared to normal control. The prolongation is consistent with the clinical phenotype of long QT interval on the body-surface ECG. While the epicardial activation was normal in all types of LQTS studied (LQT1, 2, 3 and 5), there was a marked increase in heterogeneity of ventricular recovery on the epicardium which caused significant delay in repolarization in certain regions. These regions were located in close proximity (< 10 mm) to regions with earlier recovery, resulting in abnormally large differences (> 100 ms) in recovery time and ARIs on the epicardium. This is in marked contrast with the normal heart21, where the mean LV apex-to-base ARI dispersion was only 42 ms and average LV ARI exceeded RV ARI by only 32 ms. With ARI being the surrogate for local APD, these findings reflect spatially heterogeneous prolongation of the action potential, causing the formation of regions with steep dispersion of repolarization.
The epicardial regions with delayed recovery and long ARIs exhibited marked changes in the T-wave morphology (negative or bi-phasic) of the epicardial EGMs compared to the neighboring regions with normal recovery and ARIs (Figures 2 and 3). Such changes in the T-wave over a short distance (< 10 mm) reflected the large spatial dispersion of repolarization and differences in APD.
The location and magnitude of the steep gradients varied from patient to patient. Interestingly, patients with the same genetic mutation had different epicardial repolarization patterns with different regions of prolonged APD. For instance, in Supplemental Figure 3 (LQT2), patient 1 and patient 5 are family members who have the same mutation (KCNH2-Q376sp) but markedly different ARI maps. Patient 1 has long ARIs throughout the RV and a steep ARI gradient across the entire septum. Patient 5 shows long ARIs and a steep repolarization gradient in the basal and free walls of the RV. In the same figure, patient 3 and patient 6 are family members with the same mutation (KCNH2-H70R). Patient 3 has long ARIs and steep gradient in the basal and free walls of the RV. Patient 6 has long ARIs throughout the anterior RV and a steep ARI gradient across the anterior aspect of the septum. In the same figure, patient 16 and patient 17 are family members with the same mutation (KCNH2-Y652X) with markedly different ARI maps. In Supplemental Figure 2 (LQT1), patient 14 and patient 15 are family members with the same mutation (KCNQ1-340delF). Patient 14 has long ARIs in the anterior aspect of the septum and inferior RV base, but patient 15 has long ARIs only in the anterior RV. Similar observations were made for LQT1 patients 20, 21 and 23 (KCNQ1-T312I) in Supplemental Figure 2, LQT3 patients 8 and 22 (SCN5A-R1644H) and patients 7 and 12 (SCN5A-E1784K) in Supplemental Figure 4, and LQT5 patients 9 and 10 (KCNE1-D76N) in Supplemental Figure 5. This finding is consistent with earlier studies27,28 which demonstrated considerable variability in the ECG-based clinical phenotype of LQTS (measured with body-surface QTc intervals and T-wave characteristics) in family members with the same gene mutation. We suggest that such variation among patients can be attributed to the following factors: differences in the spatial distribution of ion channels expression levels (the LQTS mutant channel and/or other ion channels), differences in gap junction distribution causing spatial heterogeneity of electric loading on repolarizing cells29, spatial variations in neural inputs and hormonal effects, coronary blood flow, and effects of modifier genes that result in variable expressivity of LQTS. A seminal multistage genome-wide association study showed that common variations of NOS1AP, a regulator of neuronal nitric oxide synthase, is significantly associated with QT interval variations in a general population derived from three cohorts30, thereby establishing the important role of modifier genes. It should be clarified that heterogeneous distribution of the LQTS mutant ion channel itself is not a necessary condition for the resultant dispersion of repolarization. Action potential repolarization and APD are determined by a delicate balance among multiple ionic currents which are expressed heterogeneously in the heart. On this background of heterogeneous substrate, even uniform distribution of the mutant channel can shift the delicate balance of currents and cause large and heterogeneous changes in APD.
Interestingly, in many of the pairs above, only one of the family members had a history of cardiac events (patient 1, patient 6, patient 16, patient 7, patient 8 and patient 10). These patients had steeper repolarization (ARI) gradients (patient 1 - 143 ms/cm, patient 6 - 159 ms/cm, patient 16 - 146 ms/cm, patient 7 - 134 ms/cm, patient 8 - 141 ms/cm, patient 10 - 149 ms/cm) compared to their respective family members with no events (patient 5 - 91 ms/cm, patient 3 - 106 ms/cm, patient 17 - 88 ms/cm, patient 12 - 110 ms/cm, patient 22 - 121 ms/cm, and patient 9 - 125 ms/cm). For the entire population of 25 LQT patients, 12 were symptomatic and 13 asymptomatic (Table 1). There was a statistically significant difference in the mean ARI between the two groups; it was 130 ± 27 ms/cm in symptomatic patients compared to 98 ± 19 ms/cm in asymptomatic patients (P = 0.002). In contrast, the difference in mean body-surface QTc was non-significant; it was 492 ± 25 ms in symptomatic patients compared to 508 ± 29 ms in asymptomatic patients (P = 0.1). The ECGI-derived steepness of repolarization gradients (ΔRT/Δx, ΔARIc/Δx) did not correlate (R = 0.2) with the patient's QTc measured from the body-surface ECG. This is not surprising because QTc, being a global marker, cannot adequately reflect the underlying cardiac EP substrate and its regional spatial properties. Thus the steepness of the repolarization gradients (EP substrate) better correlated with the patient's history of cardiac events (syncope, ventricular fibrillation, ICD shock) than the body-surface QTc. While this observation is preliminary, it could potentially be of clinical relevance. If found to be consistent in a larger study, it could be the basis for noninvasive arrhythmia risk stratification, with ECGI adding to the already established risk factors2. Also, whether the patterns of repolarization dispersion persist and remain unchanged over time in a given patient remains to be explored in future studies.
Some clinical studies have suggested that there is a genotype specific T-wave morphology8 which can help to identify the LQTS subtypes based on the body-surface ECG. However, this possibility is limited because often family members with the same mutation have very different T waves27. This limitation is consistent with the ECGI finding that considerable overlap exists in the locations of maximum ventricular repolarization gradients among the LQTS subtypes. Interestingly though, in many patients the region of maximum gradient involved the RV and septum (Supplemental Figure 2, 3 and 4). This finding is consistent with clinical EP studies in LQTS patients with severe syncope and arrhythmia, which recorded in-vivo human intracardiac monophasic action potentials from RV endocardium31. These studies showed marked spatial differences in ventricular recovery, with abnormally long APD recorded in regions of the RV31.
Also of interest is a study32 that found that mERG (mouse ERG) protein expression in the developing embryonic mouse heart was not homogeneous but was greater in the RV and in the RV outflow tract, and that certain mutations in hERG caused developmental abnormalities that mainly affected the same regions. Taken together, these findings support the possible role of RV in creating spatial heterogeneities of repolarization in LQTS.
Prolonged APD and steep spatial dispersion of repolarization have long been known to form the substrate for EADs, unidirectional block and reentrant arrhythmias. The role of reentry in LQTS arrhythmias (torsades de pointes) has been established3. However, the only noninvasive marker of dispersion of ventricular repolarization is the QT dispersion measured on the body-surface ECG. This marker lacks sensitivity and specificity and, more importantly, is not based on a solid principle. In fact, each body-surface ECG electrode records electrical signal generated by the integrated electrical activity over the entire heart. Therefore, spatial relationships in the heart are lost in the body-surface ECG and “spatial dispersion” cannot be defined in a meaningful way. ECGI overcomes this limitation by reconstructing the actual spatial properties and dispersion on the heart itself.
Although all 25 patients exhibited significantly longer ARI (APD) compared to the 7 controls, ARI dispersion was not clearly seen on the epicardium for some patients. Patient 4 (Supplemental Figure 3), patients 18, 20 and 21 (Supplemental Figure 2), patient 12 (Supplemental Figure 4) and patient 9 (Supplemental Figure 5) showed large ARI dispersion only along the basal region of the ventricles. This does not rule out the possibility that steep dispersion exists in the depth of the myocardium, as ECGI mapping is limited to the epicardium. Being the first ECGI study of LQTS in human subjects, only small cohorts from the most prevalent LQT types were included. As such, genotype-specific patterns of epicardial repolarization could not be identified. Relationships between genotype and patterns of repolarization need to be examined in a study with larger numbers of subjects from the diverse LQTS types. In addition, genotype positive / phenotype negative patients should be included in future studies.
Supplementary Material
Acknowledgments
We thank Mr. Tim Street and Mr. Michael Harrod for expert assistance with the CT scans, and Mr. Eric Novak for assistance with statistical analysis. We also appreciate greatly the helpful discussions with Junjie Zhang and Christopher Andrews during many meetings of the ECGI research team in the Rudy lab.
Funding Sources: This study was supported by NIH–NHLBI grants R01-HL-033343 and R01-HL-049054 (to Y. Rudy) and by the Washington University Institute of Clinical and Translational Sciences grant UL1 TR000448 from the National Center for Advancing Translational Sciences (NCATS) of the NIH. Dr. Yoram Rudy is the Fred Saigh Distinguished Professor at Washington University.
Footnotes
Conflict of Interest Disclosures: Dr. Yoram Rudy co-chairs the scientific advisory board of CardioInsight Technologies (CIT). He holds equity in CIT and receives royalties. CIT does not support any research conducted in Dr. Rudy's laboratory. Dr. Haïssaguerre is a stockholder of CardioInsight Technologies. Dr. Strom is a paid employee and stockholder of CardioInsight Technologies. Dr. Silva received consultant fees and speaker honoraria from Medtronic, AliveCor.
References
- 1.Schwartz PJ, Stramba-Badiale M, Crotti L, Pedrazzini M, Besana A, Bosi G, Gabbarini F, Goulene K, Insolia R, Mannarino S, Mosca F, Nespoli L, Rimini A, Rosatti E, Salice P, Spazzolini C. Prevalence of the congenital long-QT syndrome. Circulation. 2009;120:1761–1767. doi: 10.1161/CIRCULATIONAHA.109.863209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schwartz PJ, Crotti L, Insolia R. Long-QT Syndrome: from genetics to management. Circ Arrhythm Electrophysiol. 2012;5:868–877. doi: 10.1161/CIRCEP.111.962019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Roden DM, Balser JR, George AL, Anderson ME. Cardiac ion channels. Annu Rev Physiol. 2002;64:431–475. doi: 10.1146/annurev.physiol.64.083101.145105. [DOI] [PubMed] [Google Scholar]
- 4.Schwartz PJ, Ackerman MJ, George AL, Wilde AM. Impact of genetics on the clinical management of channelopathies. J Am Coll Cardiol. 2013;62:169–180. doi: 10.1016/j.jacc.2013.04.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rautaharju PM, Surawicz B, Gettes LS, Bailey JJ, Childers R, Deal BJ, Gorgels A, Hancock EW, Josephson M, Kligfield P, Kors JA, Macfarlane P, Mason JW, Mirvis DM, Okin P, Pahlm O, van Herpen G, Wagner GS, Wellens H. American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; American College of Cardiology Foundation; Heart Rhythm Society. AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part IV: the ST segment, T and U waves, and the QT interval: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society: endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol. 2009;53:982–991. doi: 10.1016/j.jacc.2008.12.014. [DOI] [PubMed] [Google Scholar]
- 6.Priori SG, Schwartz PJ, Napolitano C, Bloise R, Ronchetti E, Grillo M, Vicentini A, Spazzolini C, Nastoli J, Bottelli G, Folli R, Cappelletti D. Risk stratification in the long-QT syndrome. N Engl J Med. 2003;348:1866–1874. doi: 10.1056/NEJMoa022147. [DOI] [PubMed] [Google Scholar]
- 7.Gima K, Rudy Y. Ionic current basis of electrocardiographic waveforms: a model study. Circ Res. 2002;90:889–896. doi: 10.1161/01.res.0000016960.61087.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moss AJ, Zareba W, Benhorin J, Locati EH, Hall WJ, Robinson JL, Schwartz PJ, Towbin JA, Vincent GM, Lehmann MH. ECG T-wave patterns in genetically distinct forms of the hereditary long QT syndrome. Circulation. 1995;92:2929–2934. doi: 10.1161/01.cir.92.10.2929. [DOI] [PubMed] [Google Scholar]
- 9.Schwartz PJ, Priori SG, Spazzolini C, Moss AJ, Vincent MG, Napolitano C, Denjoy I, Guicheney P, Breithardt G, Keating MT, Towbin JA, Beggs AH, Brink P, Wilde AM, Toivonen L, Zareba W, Robinson JL, Timothy KW, Corfield V, Wattanasirichaigoon D, Corbett C, Haverkamp W, Schulze-Bahr E, Lehmann MH, Schwartz K, Coumel P, Bloise R. Genotype phenotype correlation in the long-QT syndrome: gene specific triggers for life-threatening arrhythmias. Circulation. 2001;103:89–95. doi: 10.1161/01.cir.103.1.89. [DOI] [PubMed] [Google Scholar]
- 10.Yan GX, Antzelevitch C. Cellular basis for the normal T wave and the electrocardiographic manifestations of the long-QT syndrome. Circulation. 1998;98:1928–1936. doi: 10.1161/01.cir.98.18.1928. [DOI] [PubMed] [Google Scholar]
- 11.El-Sherif N, Caref EB, Yin H, Restivo M. The electrophysiological mechanism of ventricular arrhythmias in the long QT syndrome: tridimensional mapping of activation and recovery patterns. Circ Res. 1996;79:474–492. doi: 10.1161/01.res.79.3.474. [DOI] [PubMed] [Google Scholar]
- 12.Shimizu W, Antzelevitch C. Cellular basis for long QT, transmural dispersion of repolarization, and torsade de pointes in the long QT syndrome. J Electrocardiol. 1999;32(Suppl):177–184. doi: 10.1016/s0022-0736(99)90077-8. [DOI] [PubMed] [Google Scholar]
- 13.Clancy CE, Rudy Y. Linking a genetic defect to its cellular phenotype in a cardiac arrhythmia. Nature. 1999;400:566–569. doi: 10.1038/23034. [DOI] [PubMed] [Google Scholar]
- 14.Clancy CE, Rudy Y. Cellular consequences of HERG mutations in the long QT syndrome: precursors to sudden cardiac death. Cardiovasc Res. 2001;50:301–313. doi: 10.1016/s0008-6363(00)00293-5. [DOI] [PubMed] [Google Scholar]
- 15.Viswanathan PC, Rudy Y. Cellular arrhythmogenic effects of congenital and acquired long-QT syndrome in the heterogeneous myocardium. Circulation. 2000;101:1192–1198. doi: 10.1161/01.cir.101.10.1192. [DOI] [PubMed] [Google Scholar]
- 16.Silva JR, Pan H, Wu D, Nekouzadeh A, Decker KF, Cui J, Baker NA, Sept D, Rudy Y. A multiscale model linking ion-channel molecular dynamics and electrostatics to the cardiac action potential. Proc Natl Acad Sci U S A. 2009;106:11102–11106. doi: 10.1073/pnas.0904505106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Keating MT, Sanguinetti MC. Molecular and cellular mechanisms of cardiac arrhythmias. Cell. 2001;104:569–580. doi: 10.1016/s0092-8674(01)00243-4. [DOI] [PubMed] [Google Scholar]
- 18.Ambroggi LD, Bertoni T, Locati E, Stramba-Badiale M, Schwartz PJ. Mapping of body surface potentials in patients with the idiopathic long QT syndrome. Circulation. 1986;74:1334–1345. doi: 10.1161/01.cir.74.6.1334. [DOI] [PubMed] [Google Scholar]
- 19.Day CP, McComb JM, Campbell RW. QT dispersion: an indication of arrhythmia risk in patients with long QT intervals. Br Heart J. 1990;63:342–344. doi: 10.1136/hrt.63.6.342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ghanem RN, Burnes JE, Waldo AL, Rudy Y. Imaging dispersion of myocardial repolarization, II: noninvasive reconstruction of epicardial measures. Circulation. 2001;104:1306–1312. doi: 10.1161/hc3601.094277. [DOI] [PubMed] [Google Scholar]
- 21.Ramanathan C, Jia P, Ghanem R, Ryu K, Rudy Y. Activation and repolarization of the normal human heart under complete physiological conditions. Proc Natl Acad Sci U S A. 2006;103:6309–6314. doi: 10.1073/pnas.0601533103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ghosh S, Rhee EK, Avari JN, Woodard PK, Rudy Y. Cardiac memory in patients with Wolff-Parkinson-White syndrome: noninvasive imaging of activation and repolarization before and after catheter ablation. Circulation. 2008;118:907–915. doi: 10.1161/CIRCULATIONAHA.108.781658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Marrus SB, Andrews CM, Cooper DH, Faddis MN, Rudy Y. Repolarization changes underlying long-term cardiac memory due to right ventricular pacing: noninvasive mapping with electrocardiographic imaging. Circ Arrhythm Electrophysiol. 2012;5:773–781. doi: 10.1161/CIRCEP.112.970491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Coronel R, de Bakker JM, Wilms-Schopman FJ, Opthof T, Linnenbank AC, Belterman CN, Janse MJ. Monophasic action potentials and activation recovery intervals as measures of ventricular action potential duration: experimental evidence to resolve some controversies. Heart Rhythm. 2006;3:1043–1050. doi: 10.1016/j.hrthm.2006.05.027. [DOI] [PubMed] [Google Scholar]
- 25.Millar CK, Kralios FA, Lux RL. Correlation between refractory periods and activation-recovery intervals from electrograms: effects of rate and adrenergic interventions. Circulation. 1985;72:1372–1379. doi: 10.1161/01.cir.72.6.1372. [DOI] [PubMed] [Google Scholar]
- 26.Haws CW, Lux RL. Correlation between in vivo transmembrane action potential durations and activation-recovery intervals from electrograms: effects of interventions that alter repolarization time. Circulation. 1990;81:281–288. doi: 10.1161/01.cir.81.1.281. [DOI] [PubMed] [Google Scholar]
- 27.Schwartz PJ, Priori SG, Napolitano C. The long-QT syndrome. In: Zipes DP, Jalife J, editors. Cardiac Electrophysiology: From Cell to Bedside. 3rd ed. WB Saunders Co.; Philadelphia: 2000. pp. 597–615. [Google Scholar]
- 28.Benhorin J, Moss AJ, Bak M, Zareba W, Kaufman ES, Kerem B, Towbin JA, Priori S, Kass RS, Attali B, Brown AM, Ficker E. Variable expression of Long QT syndrome among gene carriers from families with five different HERG mutations. Ann Noninvasive Electrocardiol. 2002;7:40–46. doi: 10.1111/j.1542-474X.2001.tb00137.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Viswanathan PC, Shaw RM, Rudy Y. Effects of IKr and IKs heterogeneity on action potential duration and its rate dependence: a simulation study. Circulation. 1999;99:2466–2474. doi: 10.1161/01.cir.99.18.2466. [DOI] [PubMed] [Google Scholar]
- 30.Arking DE, Pfeufer A, Post W, Kao WH, Newton-Cheh C, Ikeda M, West K, Kashuk C, Akyol M, Perz S, Jalilzadeh S, Illig T, Gieger C, Guo CY, Larson MG, Wichmann HE, Marban E, O'Donnell CJ, Hirschhorn JN, Kaab S, Spooner PM, Meitinger T, Chakravarti A. A common genetic variant in the NOS1 regulator NOS1AP modulates cardiac repolarization. Nat Genet. 2006;38:644–651. doi: 10.1038/ng1790. [DOI] [PubMed] [Google Scholar]
- 31.Bonatti V, Rolli A, Botti G. Recording of monophasic action potentials of the right ventricle in long QT syndromes complicated by severe ventricular arrhythmias. Eur Heart J. 1983;4:168–179. doi: 10.1093/oxfordjournals.eurheartj.a061436. [DOI] [PubMed] [Google Scholar]
- 32.Teng GQ, Zhao X, Lees-Miller JP, Quinn FR, Li P, Rancourt DE, London B, Cross JC, Duff HJ. Homozygous missense N629D hERG (KCNH2) potassium channel mutation causes developmental defects in the right ventricle and its outflow tract and embryonic lethality. Circ Res. 2008;103:1483–1491. doi: 10.1161/CIRCRESAHA.108.177055. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.