Abstract
• Background and Aims All plants synthesize pantothenate but its synthesis and regulation are not well understood. The aim of this work is to study the effect of exogenous supply of precursor compounds on pantothenate levels in leaves.
• Methods Precursor compounds were supplied in solution to excised leaves and the pantothenate content was measured using a microbial method.
• Key Results Pantothenate levels in excised leaves of Limonium latifolium, tomato (Lycopersicon esculentum), bean (Phaseolus vulgaris) and grapefruit (Citrus × paradisi) were examined following an exogenous supply of the precursor compounds pantoyl lactone or β-alanine. Significantly higher levels of extractable pantothenate were found when pantoyl lactone was supplied, but not when β-alanine was supplied despite a measurable uptake of β-alanine into the leaf.
• Conclusions The results suggested that the pantoate supply may be rate-limiting or regulating pantothenate synthesis in leaves.
Keywords: β-Alanine, β-alanine betaine, Limonium latifolium, Lycopersicon esculentum, osmoprotection, pantoate, pantothenate
INTRODUCTION
Pantothenate (vitamin B5) is the metabolic precursor of coenzyme A and acyl carrier protein. Bacteria, fungi and plants synthesize pantothenate, but animals obtain it from their diet (Smith and Song, 1996). Pantothenate synthesis has been thoroughly characterized in bacteria (Brown and Williamson, 1987; Merkel and Nichols, 1996). Few studies have focused on the synthesis, regulation and role of pantothenate in plants (Ottenhof et al., 2004), although plants are one of the major dietary sources of pantothenate for animals and humans (Raman and Rathinasabapathi, 2004), and pantothenate is vital for the growth of plant cells (Savage et al., 1979).
Radiotracer experiments by Jones et al. (1994) showed that pea leaves converted radiolabelled valine to pantothenate. Characterization of intermediates suggested that the plant pathway to pantothenate was comparable to the bacterial pathway (Jones et al., 1994; Fig. 1). The final step of pantothenate synthesis, the ATP-dependent condensation of pantoate and β-alanine, was catalysed by pantothenate synthetase, a cytosolic enzyme expressed in both leaves and roots (Genschel et al., 1999).
Fig. 1.
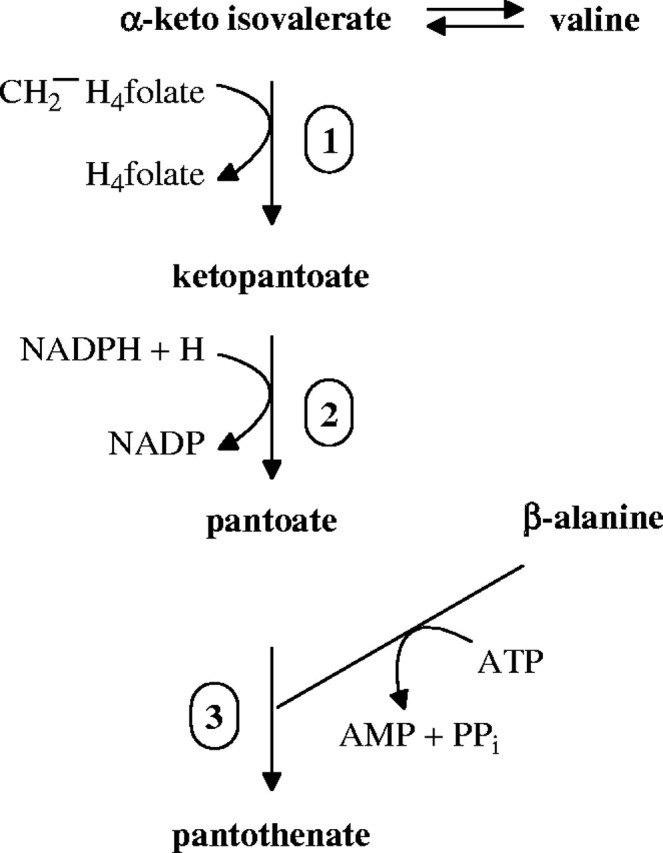
Biosynthesis of pantothenate in plants (after Jones et al., 1994). Steps numbered are catalysed by the following enzymes: 1, ketopantoate hydroxymethyltransferase; 2, ketopantoate reductase; and 3, pantothenate synthetase.
In Limonium latifolium, a halophytic species of the family Plumbaginaceae, β-alanine was also methylated to β-alanine betaine, an osmoprotectant (Rathinasabapathi et al., 2000). There is interest in engineering the synthesis of β-alanine betaine in crops that do not naturally accumulate it (Rathinasabapathi et al., 2002). With this in mind, an investigation was carried out to see if β-alanine levels limited the synthesis of pantothenate in plants. It was expected that β-alanine might limit pantothenate synthesis in L. latifolium because β-alanine was shared between β-alanine betaine and pantothenate synthesis pathways in this species (Rathinasabapathi et al., 2000; Raman and Rathinasabapathi, 2003). The objective of the current study was to determine whether pantoate or β-alanine would be rate-limiting for, or regulating, synthesis of pantothenate in the leaves of L. latifolium and other plant species.
MATERIALS AND METHODS
Chemicals
Pantothenic acid and pantoyl lactone were obtained from Sigma Chemical Co (St Louis, MO, USA) and d-(–)-pantoic acid was synthesized by treating d-(–)-pantoyl lactone with 1 n potassium hydroxide (Shimizu et al., 1988). Solvents were from Fisher Scientific (Pittsburg, PA, USA). [1-14C]β-Alanine (specific activity 52 mCi mmol−1), purchased from American Radiolabeled Chemical Inc. (St Louis, MO, USA) was used without further purification.
Plants, bacteria and growth conditions
Seeds of Lycopersicon esculentum Mill. ‘Celebrity’, Phaseolus vulgaris L. ‘Kentucky Wonder’ and Limonium latifolium (Sm.) O. Kuntze were obtained from Park Seeds (Greenwood, SC, USA). Plants were grown in a greenhouse as described previously (Rathinasabapathi et al., 2000). Arabidopsis thaliana (L.) Heynh. ecotype Columbia (Lehle Seeds, Round Rock, TX, USA) was cultivated in a commercial potting medium in a temperature-controlled room (24 °C day/23 °C night) in an 8-h day length of 200 μmol m−2 s−1 PPFD light provided by cool white fluorescent lamps. Citrus × paradisi ‘Red Ruby’ leaves were obtained from a tree at the orchard of the Horticultural Sciences Department, University of Florida, Gainesville. Lactobacillus plantarum ATCC 8014 from the American Type Culture Collection, was cultivated at 30 °C in pantothenate assay medium (Sigma, St Louis, MO, USA; catalogue no. P6851) supplemented with 100 µg L−1 pantothenate.
Treatment of excised leaves
Young leaf tissue free of diseases and pests was sampled from different species and rinsed in distilled water. The cut ends of excised leaves were placed in 10 mL (for Arabidopsis thaliana) or 50 mL (for all other species) of Hoagland nutrient medium (Hoagland and Arnon, 1950) and incubated at 25 °C at 150 μmol m−2 s−1 PPFD (cool white fluorescent light) for 24 h. For treatments, pantoyl lactone, pantoate, β-alanine or valine were added to the nutrient medium at 1 mm final concentration.
Time course experiments were carried out with tomato leaflets from 3-month-old plants, grown in 4-gallon pots in a commercial potting medium in a greenhouse. The leaflet samples weighed about 150–200 mg each. Following the supply of pantoyl lactone (at 0·1 or 1·0 mm) or β-alanine at 1·0 mm under light (150 μmol m−2 s−1 PPFD, cool white fluorescent light), the leaflet samples were collected at different time intervals, frozen in liquid nitrogen and stored at −80 °C. For monitoring β-alanine uptake, leaflets were supplied with 1 mm β-alanine ([14C]β-alanine, 50 µCi mol−1) and the samples, harvested at various incubation times, were ground in 2 mL of methanol : chloroform : water (12 : 5 : 1 v/v/v) mixture. Radioactivity in an aliquot of this extract was measured using a liquid scintillation counter (Beckman Instruments Inc., Fullerton, CA, USA) in 50 % (v/v) ready gel with 70 % counting efficiency.
Extraction of tissue
The extraction method was adapted from Gonthier et al. (1998). The tissue was blotted dry, weighed and ground to a fine powder after freezing in liquid nitrogen. A buffer of 20 mm MOPS–KOH, pH 7·0, was added at the rate of 5 mL per every 1 g tissue and mixed in a mortar and pestle. The brei was filtered through four layers of cheese-cloth and the extract was centrifuged at 20 000 g for 20 min at 4 °C. The supernatant was collected and stored frozen at −80 °C and used for pantothenate determinations.
Determination of pantothenic acid in tissue extracts
Pantothenate in tissue extracts was determined using a microbial assay as described by Wyse et al. (1985). A 2-mL aliquot of extract dilution containing 10–100 ng of pantothenic acid was added to 3 mL of growth medium. After mixing for a few seconds, the tubes were heated in an autoclave for 5 min at 115 °C, cooled, aseptically inoculated by one drop of a Lactobacillus plantarum suspension and incubated for 24 h at 30 °C water bath. Growth was measured by the optical density of growth medium at 620 nm in a UV-visible spectrophotometer (Beckman Instruments Inc., Fullerton, CA, USA). Quantitative values on pantothenate levels for the time course experiment were analysed using the analysis of variance and a t-test at P ≤ 0·05 was used for mean separation.
RESULTS AND DISCUSSION
Most studies on pantothenate levels were on a large number of food products including some fruits and vegetables (Mozafar, 1994). Pantothenate levels in leaves, however, had not been investigated in most of these studies. Therefore, in this study extractable free pantothenate levels were determined in Limonium latifolium leaves and compared with six other species. A ten-fold variation was noticed among these species (Table 1). Limonium latifolium leaves had approx. 3 nmol g−1 f. wt. Other reports show that lentil, avocado and rice had about 1·2, 0·25 and 0·2 nmol pantothenate g−1 f. wt (Gonthier et al., 1998).
Table 1.
Extractable pantothenate levels in mature leaves of different plant species
| Species |
Pantothenate (nmol g−1 f. wt) |
|---|---|
| Limonium latifolium | 2·85 ± 0·1 |
| Arabidopsis thaliana | 1·30 ± 0·2 |
| Basella alba | 1·22 ± 0·02 |
| Citrus × paradisi | 6·96 ± 1·1 |
| Nicotiana tabacum | 4·28 ± 0·5 |
| Rosa sinensis | 0·70 ± 0·11 |
| Lycopersicon esculentum | 1·31 ± 0·03 |
Fresh leaves were processed immediately after harvest.
Values are means and standard error from three independent determinations.
Excised leaves of L. latifolium, Phaseolus vulgaris, Lycopersicon esculentum and Citrus × paradisi were incubated in nutrient medium or nutrient medium supplemented with either 1 mm β-alanine or 1 mm pantoyl lactone. Among the four species, only L. latifolium was known to accumulate β-alanine betaine (Rathinasabapathi et al., 2000). Pantoyl lactone, inside the plant cell, could be expected to yield pantoate via hydrolysis. Activities of ketopantoyl lactone reductase and a pantoyl lactone-dependent pantothenate synthetase were known in spinach (Julliard, 1994) but no direct evidence is available for a biosynthetic pathway leading to pantothenate via the lactone forms of ketopantoate and pantoate in any organism (Ottenhoff et al., 2004).
When 1 mm β-alanine was supplied to excised leaves, pantothenate level did not increase significantly in any of the four species examined (Table 2). An exogenous supply of 1 mm pantoyl lactone to excised leaves significantly (P < 0·05) increased pantothenate levels in the leaves from all the four species (Table 2). A supply of 1 mm valine to L. latifolium leaves had a similar effect (Table 2). Valine and pantoyl lactone themselves did not support the growth of L. plantarum in the assay medium. Hence, precursor compounds pantoyl lactone and valine had presumably been transformed to pantothenate in the tissue during the incubation.
Table 2.
Effect of 1 mm exogenous supply of β-alanine, pantoyl lactone and valine for 24 h on the levels of pantothenate in excised leaves of L. latifolium, tomato, bean and grapefruit
| Pantothenate (nmol g−1 f. wt) |
|||||||
|---|---|---|---|---|---|---|---|
| Species |
Control |
β-Alanine |
Pantoyl lactone |
Valine |
|||
| Limonium latifolium | 3·27 ± 0·5 | 3·7 ± 0·5 | 10·3 ± 0·8 | 5·1 ± 0·5 | |||
| Lycopersicon esculentum | 2·93 ± 0·1 | 2·7 ± 0·6 | 51·1 ± 0·5 | ND | |||
| Phaseolus vulgaris | 3·54 ± 0·8 | 3·73 ± 1·1 | 37·1 ± 2·2 | ND | |||
| Citrus × paradisi | 21·36 ± 0·4 | 23·3 ± 1·1 | 42·9 ± 6·5 | ND | |||
Values are means and standard error from three independent leaf samples.
ND, not determined.
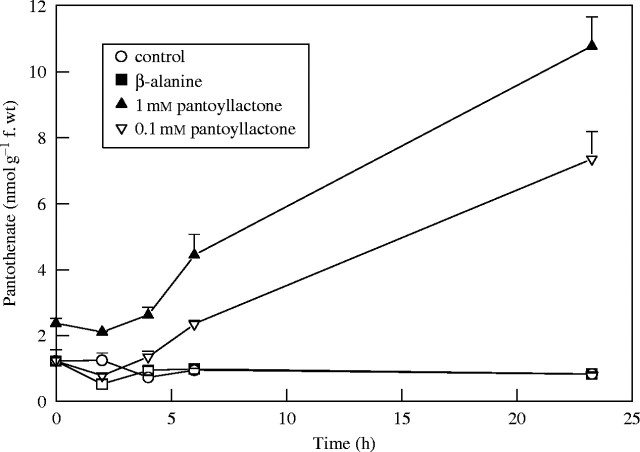
Results from a time-course experiment, wherein tomato leaflets were supplied with pantoyl lactone and β-alanine individually, reinforced these observations. Pantothenate in leaflets increased linearly following pantoyl lactone supply over time (Fig. 2). At 6-h and 24-h sampling times, significantly more pantothenate was found in the leaflets supplied with 1 mm pantoyl lactone compared with leaflets supplied with 0·1 mm pantoyl lactone or control (Fig. 2). In a similar experiment, tomato leaflets, supplied with 1 mm d-pantoate for 24 h, had significantly increased pantothenate levels (90 ± 10 nmol g−1 f. wt; n = 3). Such increases were not observed for the negative control or β-alanine-supplied leaflets (Fig. 2).
Fig. 2.
Pantothenate levels in tomato leaflets supplied with nutrient medium alone (control) or nutrient medium supplemented with 0·1 or 1·0 mm of pantoyllactone or 1 mm β-alanine for periods of time up to 24 h. Each point represents mean and standard error of three replicates.
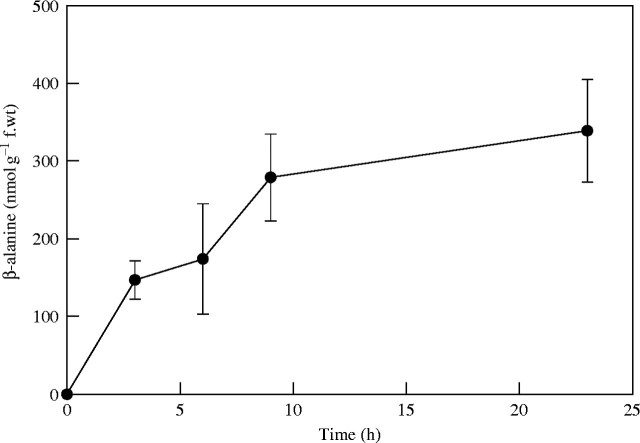
In a similar experiment, excised tomato leaflets were also incubated in 1 mm β-alanine in the nutrient medium mixed with [1-14C]β-alanine. Uptake of β-alanine was monitored by measuring the radioactivity in the extracts of the samples. About 300 nmol of β-alanine g−1 f. wt leaflet were taken up by the tissues during the 24-h incubation period (Fig. 3). This suggested that lack of increase in the level of pantothenate following β-alanine supply was not due to poor uptake of the exogenous supply.
Fig. 3.
Uptake of exogenously supplied β-alanine by excised tomato leaflets over a period of time up to 24 h. Each point represents the mean and standard error of four replicates. Uptake was monitored by using [14C]β-alanine as described in Materials and methods.
Data presented here are consistent with previous observations that pantothenate synthesis was active in the leaf tissue (Jones et al., 1994; Genschel et al., 1999). Additionally, the present data showed that exogenous pantoyl lactone increased the pantothenate levels, suggesting that pantoate could be rate-limiting for pantothenate synthesis in the leaf tissue in the species examined. This phenomenon, contrasting with observations in bacteria (Jackowski and Rock, 1981), appeared to be common to both accumulators and non-accumulators of β-alanine betaine.
An alternative explanation for increased pantothenate levels following the supply of pantoyl lactone can be that pantoyl lactone or its derivatives reduced coenzyme A synthesis by inhibiting pantothenate kinase, thus elevating the free pantothenate levels. These explanations are not mutually exclusive, but regulation of plant pantothenate kinase by pantoyl lactone or its derivatives has not been tested (Falk and Guerra, 1993).
Currently, there is interest in engineering important crops for the synthesis of the osmoprotectant β-alanine betaine to test its possible role in stress tolerance (Rathinasabapathi et al., 2002). Such engineering might deplete the β-alanine pool to negatively affect pantothenate synthesis. Results presented here, however, imply that some of the β-alanine pool in the leaf will be available for the synthesis of β-alanine betaine without adversely reducing the pantothenate pool.
Acknowledgments
This work was supported by a grant to B.R. from the United States Department of Agriculture (NRICGP-2001-35318-10947). We thank Dr Maria Gallo-Meagher for comments on the manuscript and Ms Celia Sigua and Ms Keri Roberts for excellent technical assistance. This is Florida Agricultural Experiment Station Journal Series Number R-09227.
Footnotes
Present address: Department of Medicine, University of Florida, Gainesville, FL 32608, USA.
LITERATURE CITED
- Brown GM, Williamson JM. 1987. Biosynthesis of folic acid, riboflavin, thiamine, and pantothenic acid. In: Neidhardt FC, ed. Escherichia coli and Salmonella typhimurium cellular and molecular biology, Vol. 1. Washington DC: American Society for Microbiology, 521–538. [Google Scholar]
- Falk KL, Guerra DJ. 1993. Coenzyme A biosynthesis in plants: partial purification and characterization of pantothenate kinase from spinach. Archives of Biochemistry and Biophysics 301: 424–430. [DOI] [PubMed] [Google Scholar]
- Genschel U, Powell CA, Abell C, Smith AG. 1999. The final step of pantothenate biosynthesis in higher plants: cloning and characterization of pantothenate synthetase from Lotus japonicus and Oryza sativa (rice). Biochemistry Journal 341: 669–678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonthier A, Fayol V, Viollet J, Hartmann DJ. 1998. Determination of pantothenic acid in foods: influence of the extraction method. Food Chemistry 63: 287–294. [Google Scholar]
- Hoagland DR, Arnon DI. 1950. The water-culture method for growing plants without soil. California Agricultural Experiment Station Bulletin No. 347. [Google Scholar]
- Jackowski S, Rock CO. 1981. Regulation of coenzyme A biosynthesis. Journal of Bacteriology 148: 926–932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones CE, Dancer JE, Smith AG, Abell C. 1994. Evidence for the pathway to pantothenate in plants. Canadian Journal of Chemistry 72: 261–263. [Google Scholar]
- Julliard JH. 1994. Purification and characterization of oxopantoyl lactone reductase from higher plants: role in pantothenate synthesis. Botanica Acta 107: 191–200. [Google Scholar]
- Merkel WK, Nichols BP. 1996. Characterization and sequence of the Escherichia coli panBCD gene cluster. FEMS Microbiology 143: 247–252. [DOI] [PubMed] [Google Scholar]
- Mozafar A. 1994.Plant vitamins: agronomic, physiological and nutritional aspects. Boca Raton, FL: CRC Press. [Google Scholar]
- Ottenhoff HH, Ashurst JL, Whitney HM, Saldanha SA, Schmitzberger F, Gweon HS, Blundell TL, Abell C, Smith AG. 2004. Organisation of the pantothenate (vitamin B5) biosynthesis pathway in higher plants. Plant Journal 37: 61–72. [DOI] [PubMed] [Google Scholar]
- Raman SB, Rathinasabapathi B. 2003. β-Alanine N-methyltransferase of Limonium latifolium cDNA cloning and functional expression of a novel N-methyltransferase implicated in the synthesis of the osmoprotectant β-alanine betaine. Plant Physiology 132: 1642–1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raman SB, Rathinasabapathi B. 2004. Pantothenate synthesis in plants. Plant Science 167: 961–968. [Google Scholar]
- Rathinasabapathi B, Raman SB, Sigua C, Aly MAM. 2002. β-Alanine betaine: a nitrogenous osmoprotectant implicated in plant tolerance to salinity and hypoxic stress. In: Wood AJ, ed. Biochemical and molecular responses of plants to the environment. Trivandrum, India: Research Signpost, 69–77. [Google Scholar]
- Rathinasabapathi B, Sigua C, Ho J, Gage DA. 2000. Osmoprotectant β-alanine betaine synthesis in the Plumbaginaceae: S-adenosyl-l-methionine dependent N-methylation of β-alanine to its betaine via N-methyl and N,N-dimethyl β-alanines. Physiologia Plantarum 109: 225–231. [Google Scholar]
- Savage AD, King J, Gamborg OL. 1979. Recovery of a pantothenate auxotroph from a cell suspension culture of Datura innoxia Mill. Plant Science Letters 16: 367–376. [Google Scholar]
- Shimizu S, Kataoka M, Chung MC, Yamada H. 1988. Ketopantoic acid reductase of Pseudomonas maltophila 845: purification, characterization, and role in pantothenate biosynthesis. Journal of Biological Chemistry 263: 12077–12084. [PubMed] [Google Scholar]
- Smith CM, Song WO. 1996. Comparative nutrition of pantothenic acid. Journal of Nutrition and Biochemistry 7: 312–321. [Google Scholar]
- Wyse BW, Song WO, Walsh JH, Hansen RG. 1985. Pantothenic acid. In: Augustin J, Klein BP, Becker D, Venugopal PB (eds). Methods in vitamin assay. New York: Wiley-Interscience Publications, 399–416, ISBN 0-471-86957-0. [Google Scholar]