Abstract
Background:
Sonographic features of lymph nodes on endobronchial ultrasound (EBUS) have been shown to be useful in prediction of malignancy in mediastinum and hilum. The aim of this study was to assess the utility of morphologic features of mediastinal and/or hilar lymph nodes obtained by EBUS in patients with sarcoidosis.
Materials and Methods:
We retrospectively reviewed the records of 224 patients with mediastinal/hilar lymph node enlargements who underwent EBUS for diagnostic purpose. The lymph nodes were characterized based on the EBUS images as follows: (1) Size; based on short-axis dimension, <1 cm or ≥1 cm, (2) shape; oval or round, (3) margin; distinct or indistinct, (4) echogenicity; homogeneous or heterogeneous, (5) presence or absence of central hilar structure, and (6) presence or absence of granular (sandpaper) appearance.
Results:
One hundred (24.4%) nodes exhibited indistinct margins while 309 (75.6%) had distinct margins. One hundred and ninety nine (48.7%) nodes were characterized as homogeneous, and 210 (51.3%) nodes as heterogeneous. Granular appearance was observed in 130 (31.8%) lymph nodes. The presence of granules in lymph nodes on EBUS had the highest specificity (99.3%) for the diagnosis of sarcoidosis. Logistic regression analysis revealed the finding of distinct margin alone as an independent predictive factor for the diagnosis of sarcoidosis.
Conclusions:
The presence of granular appearance in lymph nodes by EBUS had the highest specificity (99.3%) for the diagnosis of sarcoidosis. Lymph nodes having distinct margins tend to suggest sarcoidosis.
Keywords: Diagnosis, echoic features, endobronchial ultrasonography, sarcoidosis
INTRODUCTION
Sarcoidosis is a multisystem disorder of unknown etiology characterized by noncaseating epitheloid cell granulomas. The incidence of sarcoidosis has been increasing, possibly explained by greater awareness and recognition of the condition.[1] The lung is the most common organ involved, and thoracic adenopathy is present in up to 85% of cases.[2] The diagnosis of sarcoidosis can be greatly substantiated by excluding other disease possibilities, using appropriate clinicoradiological, cytological or histological tissue examinations, especially when treatment with systemic steroids is contemplated. Transbronchial lung biopsy (TBLB) is the recommended procedure in most cases. The diagnostic yield, however, depends largely upon the experience of the operator and number of biopsy specimens.[3] Conventional (i.e., nonendobronchial ultrasound [EBUS]-guided) transbronchial needle aspiration (TBNA) has also been used as a diagnostic tool in pulmonary sarcoidosis with yields similar to those of TBLB.[4] Mediastinoscopy has been the method of choice when TBLB is unsuccessful.[5] However, it is invasive, costly and requires in-patient care with a morbidity rate of 2-3%.[6]
Endobronchial ultrasonography has been used to identify mediastinal and hilar lymph nodes.[7] EBUS-TBNA has emerged as an accurate, minimally invasive, safe technique for assessing undiagnosed mediastinal adenopathies.[8] EBUS-TBNA can image the paratracheal, subcarinal, and hilar lymph nodes are allowing to obtain needle biopsies real-time under direct visualization, resulting in a high diagnostic yield. This is performed with convex probe-EBUS (CP-EBUS), which has a 7.5-MHz ultrasound probe on the tip of a bronchovideoscope. CP-EBUS is capable of detecting lymph nodes as small as 2 mm, which are sometimes not visible on computed tomography (CT) imaging.
It has been reported that sonographic features are useful imaging tools in the evaluation of cervical lymph node metastasis in head and neck cancers, breast cancers, and thoracic malignancies.[9,10] Furthermore, sonographic features during endoscopic ultrasound (EUS) have been shown to be useful for the prediction of malignant lymph nodes in the mediastinum and the hilum.[11,12] Until date, there are no reports on the classification of sonographic features during EBUS-TBNA or on the usefulness of these features in the diagnosis of sarcoidosis in mediastinal and hilar lymph nodes.
The aim of this study was to assess the utility of the morphologic features of mediastinal and/or hilar lymph nodes obtained by EBUS in patients with sarcoidosis.
MATERIALS AND METHODS
Patients
The records of 224 patients with mediastinal and/or hilar lymphadenopathy were retrospectively investigated. The patients’ chart accessed for the procedure records, radiologic studies, pathology reports, laboratory results, and clinic notes. A chest radiograph and CT scan of the chest (plain and contrast-enhanced) were performed in all patients. Conventional flexible bronchoscopy (model BF-240 bronchoscope; Olympus; Tokyo, Japan) was first performed in a standard fashion to examine the tracheobronchial tree, followed by CP-EBUS (model XBF-UC260F-OL8; Olympus with ultrasound processor model EU-C2000; Olympus, Tokyo, Japan). The same bronchoscopist performed both bronchoscopy procedures under local anesthesia and conscious sedation (midazolam).
PROCEDURE
Bronchoscopy procedures were performed orally. Following conventional flexible bronchoscopic examination of the tracheobronchial tree, CP-EBUS was performed to first identify the lymph nodes. Lymph nodes were identified according to the International Staging System.[13] Blood vessels were further confirmed using the Doppler mode. A 22-gauge, 4-cm cytology needle (Olympus) was used to obtain specimens. A dedicated TBNA needle was inserted through the working channel of the bronchoscope, and the designated lymph node was punctured under direct EBUS guidance. The aspirated material was smeared onto glass slides. Smears were air-dried as well as fixed in 95% alcohol. For each site, the median number of passes was two (range: 1-5). Histologic specimens obtained in some cases were fixed in formalin before being sent to the Pathology Department. Rapid on-site cytologic evaluation (ROSE) was not available.
The final diagnosis of pulmonary sarcoidosis was based on lymph node aspirates revealing epitheliod, noncaseating granulomas without necrosis in the absence of identifiable malignancy, lymphoma, or infection (i.e., tuberculosis or fungal disease) and appropriate clinical picture. In cases in which cytology was nondiagnostic, histology samples (obtained by EBUS-TBNA, TBLB, endobronchial biopsy) revealing characteristic granulomas or clinical/radiologic findings consistent with sarcoidosis was considered to be adequate for diagnosis.
ENDOBRONCHIAL ULTRASOUND IMAGE CHARACTERISTICS OF LYMPH NODES
The lymph nodes were characterized based on EBUS images as follows:
Size; short-axis dimension <1 cm or ≥1 cm,
Shape; oval or round,
Margin; distinct or indistinct,
Echogenicity; homogeneous or heterogeneous,
Presence or absence of central hilar structure (CHS), and
Presence or absence of granular (sandpaper) appearance. [Figures 1–5].
Figure 1.
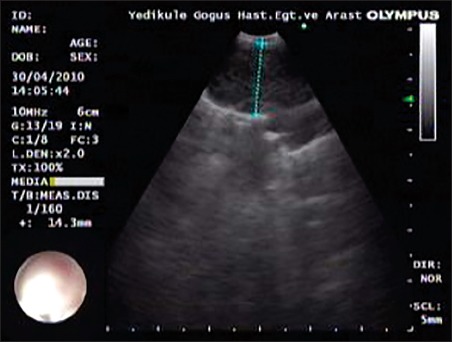
A lymph node of a case with distinct margin in the endobronchial ultrasound image
Figure 5.
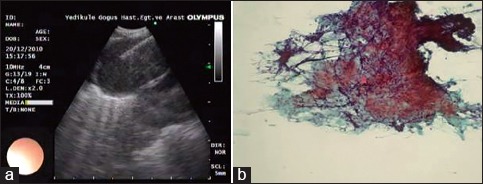
A representative case of a lymph node with granules in the endobronchial ultrasound image (a), which is associated with a noncaseating granuloma in histopathologic image (b) (sarcoidosis)
Figure 2.
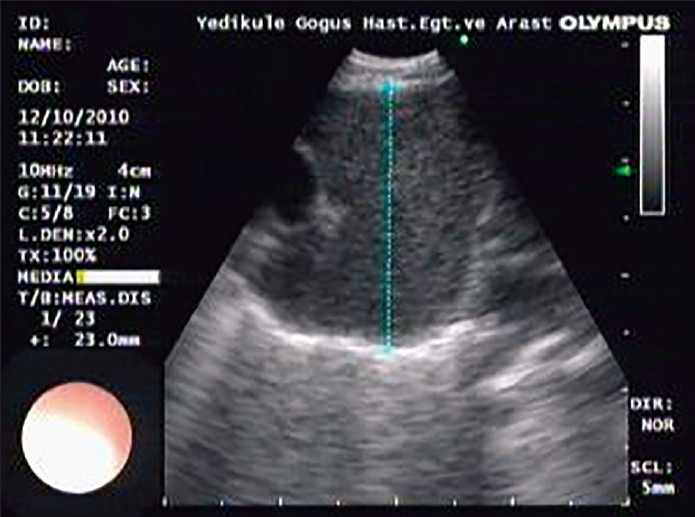
A lymph node of a case with round shape
Figure 3.
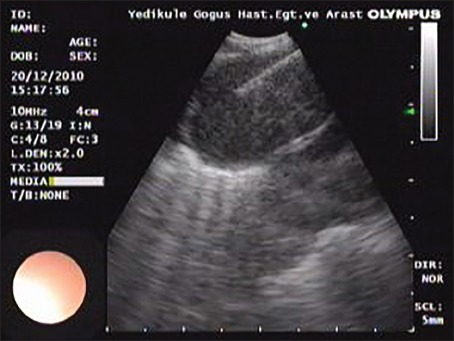
A lymph node of a case with granules
Figure 4.
A lymph node of a case with homogeneous echogenicity
We measured both the long and the short axis of all lymph nodes. When the ratio of the short to a long axis of lymph nodes was, <1.5, we defined the lymph nodes as round. On the other hand, if the ratio was >1.5, we defined it as oval, as described by Fujiwara et al.[14] EBUS images were interpreted by at least two operators having over 5 years experience with experience of over 800 EBUS procedures performed annually in a tertiary pulmonary medicine hospital.
ANALYSIS
All six sonographic characteristics of the lymph nodes were compared with the final pathologic result of the lymph nodes. The sensitivity, specificity, positive predictive value, and negative predictive value were calculated by standard definitions. Logistic regression analysis was used to predict the independent factors (sonographic features of lymph nodes) for the diagnosis of sarcoidosis.
RESULTS
The characteristics of the 224 patients who were enrolled and evaluated in this study are summarized in Table 1. The histopathologic diagnoses of lesions were sarcoidosis (n = 75), nonsmall cell carcinoma (n = 94), small cell carcinoma (n = 11), and tuberculosis (n = 44).
Table 1.
Patient characteristics
A total of 409 lymph nodes was analyzed. The proportion of each station is summarized in Table 2. None of the patients had any complications related to EBUS-TBNA.
Table 2.
Lymph node stations included in the study

The size of evaluated lymph nodes ranged from 5 to 70 mm. The number of nodes with a short axis <1 cm was 30 (7.3%), with a short axis >1 cm was 379 (92.7%). For the shape, 123 (30.1%) lymph nodes were characterized as oval and 286 (69.9%) as a round. For margin, 100 (24.4%) nodes exhibited indistinct margins and 309 (75.6%) had distinct margins. For echogenicity, 199 (48.7%) nodes were characterized as homogeneous, and 210 (51.3%) nodes as heterogeneous. A CHS was observed in 20 (4.9%) nodes, and the presence of granules in 130 (31.8%) nodes.
The EBUS feature concerning size >10 mm had the highest sensitivity in the diagnosis of sarcoidosis (100%), but the lowest specificity (9.3%). By contrast, an EBUS feature relating to the presence of granules had the highest specificity (99.3%) for the diagnosis of sarcoidosis. Positive and negative predictive value relating to the presence of granules was also high (98.4% and 91.9% respectively). The diagnostic yield of each EBUS image for sarcoidosis is shown on Table 3.
Table 3.
Diagnostic yield of each endobronchial ultrasound image for sarcoidosis
Logistic regression analysis revealed that only the presence of a distinct margin was independent predictive factor for the diagnosis of sarcoidosis, with a hazard ratio of 9.01 (P = 0.045, 95% confidence interval 1.04-77.7).
DISCUSSION
The results of this study indicate that sonographic features on EBUS such as the presence of a distinct margin and/or granular (sandpaper) appearance of the lymph node may be suggestive of sarcoidosis. The diagnosis of sarcoidosis can be difficult and relies on considerably upon other examinations for the exclusion of other diseases. The most common mode of presentation of sarcoidosis is hilar and mediastinal lymphadenopathy, and up to 90% of patients show evidence of hilar node enlargement on chest radiography.[15] Tissue diagnosis using these features is, therefore, reasonable, as they are a likely target for confirming the diagnosis, especially since TBLB yielded 30% of patients with undiagnosed suspected sarcoidosis.[16] Conventional TBNA was shown to have a diagnostic yield of 42-76% for sarcoidosis, with a higher yield in stage I of the disease.[17,18,19] However, this is performed via blind needle aspiration. TBNA guided by CT fluoroscopy has been reported to have a high diagnostic yield.[20] However, CT scan guidance can be costly, and patients as well as the examiner are exposed to radiation. Mediastinoscopy has a high diagnostic rate and, therefore, has been the procedure of choice when TBLB is nondiagnostic, but perihilar lymph nodes cannot be accessed by this method.[5]
The recent advances in the endoscopic sampling of mediastinal lymph nodes using EBUS-TBNA and EUS-guided fine needle aspiration (EUS-FNA) have been further developed for the diagnosis of mediastinal lymphadenopathy. Recent trials of EUS-FNA in sarcoidosis showed a diagnostic value of 82%,[16] a sensitivity of 89-100% and a specificity of 94-96%.[21,22] The results of another study demonstrated that direct real-time CP-EBUS-guided TBNA is a safe and accurate method of evaluating both mediastinal and hilar lymph nodes. It had a sensitivity of 95.7% (45 of 47 patients), the specificity of 100%, and accuracy of 97.1% (68 of 70 patients) in distinguishing benign from malignant mediastinal and/or hilar lymph nodes. CP-EBUS is capable of detecting lymph nodes as small as 3 mm in short axis not visible on CT imaging, and these lymph nodes can be punctured under real-time ultrasound guidance.[23] Recently, Fujiwara et al.[14] reported EBUS sonographic features that are suggestive of benign lymph nodes. They classified the different sonographic features of mediastinal lymph nodes and applied this EBUS image classification to the prediction of lymph node metastasis. Their results show that sonographic EBUS features of lymph nodes during EBUS-TBNA are helpful for the prediction of benign lymph nodes in lung cancer patients. They showed that a short axis of more than 1 cm, round shape, distinct margin, heterogeneous echogenicity, absence of CHS, or the presence of coagulation necrosis sign tended to suggest metastatic lymph nodes. In another study sonography was reported as a useful imaging tool in the evaluation of cervical lymph nodes in patients with malignancies of the head, neck, and thorax.[9]
Choi et al.[24] retrospectively reviewed the clinical and sonographic features of eight patients with 10 tall-cell variants of papillary thyroid carcinoma. They reported that the tall-cell variants often appeared as microlobulated markedly hypoechoic nodules with microcalcifications and extrathyroidal extensions on sonography which were always associated with lymph node metastasis. Sonographic images in were also found helpful for suggesting Kikuchi's disease.[25] Cervical lymphadenopathies in patients with Kikuchi's disease have a smaller size, are less round, a less micronodular reticular echotexture, and show more signs of matting and cortical widening than those with lymphoma examined under ultrasound. Another study showed that metastatic cervical lymph nodes were ultrasonographically characterized by a smaller long axis to short axis ratio (L/S ratio), the absence of a hilus and a hypoechoic center.[26]
The echogenic feature of EBUS is closely related to cell arrangement and the amount of fibrous stroma. Lesions of the gastrointestinal system visualized with EUS, have showed that a heterogeneous echogenicity was more likely to indicate a malignancy than homogeneous echogenicity.[27,28,29,30] In the present study homogeneous echogenicity was associated with the diagnosis of sarcoidosis. Furthermore, presence of granules in a lymph node visualized by EBUS standard for the diagnosis of sarcoidosis with high positive predictive value.
When a mediastinal/hilar lymph node in a patient with a suspected diagnosis of sarcoidosis by radiological/clinical data shows only benign lymphocytes on cytology, ultrsonographical features of lymph nodes such as granular (sandpaper) appearance and distinct margin would reassure the diagnosis of sarcoidosis and helps to limit multiple passes in a lymph node and sampling of multiple stations where ROSE is available.
In the present study, we have found that, although lymph nodes of patients with sarcoidosis are often round, may have a granular appearance, homogeneous echogenicity, and distinct margin, logistic regression analysis revealed that the only independent predictive factor for the diagnosis of sarcoidosis was the presence of a distinct margin. Further prospective studies are recommended to confirm the utility of sonographic features during EBUS-TBNA.
Footnotes
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- 1.Perng RP, Chen JH, Tsai TT, et al. Sarcoidosis among Chinese in Taiwan. J Formos Med Assoc. 1997;96:697–9. [PubMed] [Google Scholar]
- 2.Lynch JP, 3rd, Kazerooni EA, Gay SE. Pulmonary sarcoidosis. Clin Chest Med. 1997;18:755–85. doi: 10.1016/s0272-5231(05)70417-2. [DOI] [PubMed] [Google Scholar]
- 3.Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med. 1999;160:736–55. doi: 10.1164/ajrccm.160.2.ats4-99. [DOI] [PubMed] [Google Scholar]
- 4.Trisolini R, Lazzari Agli L, Cancellieri A, et al. Transbronchial needle aspiration improves the diagnostic yield of bronchoscopy in sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2004;21:147–51. [PubMed] [Google Scholar]
- 5.Gossot D, Toledo L, Fritsch S, et al. Mediastinoscopy vs thoracoscopy for mediastinal biopsy. Results of a prospective nonrandomized study. Chest. 1996;110:1328–31. doi: 10.1378/chest.110.5.1328. [DOI] [PubMed] [Google Scholar]
- 6.Hammoud ZT, Anderson RC, Meyers BF, et al. The current role of mediastinoscopy in the evaluation of thoracic disease. J Thorac Cardiovasc Surg. 1999;118:894–9. doi: 10.1016/s0022-5223(99)70059-0. [DOI] [PubMed] [Google Scholar]
- 7.Okamoto H, Watanabe K, Nagatomo A, et al. Endobronchial ultrasonography for mediastinal and hilar lymph node metastases of lung cancer. Chest. 2002;121:1498–506. doi: 10.1378/chest.121.5.1498. [DOI] [PubMed] [Google Scholar]
- 8.Herth FJ, Eberhardt R, Vilmann P, et al. Real-time endobronchial ultrasound guided transbronchial needle aspiration for sampling mediastinal lymph nodes. Thorax. 2006;61:795–8. doi: 10.1136/thx.2005.047829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ahuja AT, Ying M. Sonographic evaluation of cervical lymph nodes. AJR Am J Roentgenol. 2005;184:1691–9. doi: 10.2214/ajr.184.5.01841691. [DOI] [PubMed] [Google Scholar]
- 10.Kim TH, Kang DK, Kim SY, et al. Sonographic differentiation of benign and malignant papillary lesions of the breast. J Ultrasound Med. 2008;27:75–82. doi: 10.7863/jum.2008.27.1.75. [DOI] [PubMed] [Google Scholar]
- 11.Lee N, Inoue K, Yamamoto R, et al. Patterns of internal echoes in lymph nodes in the diagnosis of lung cancer metastasis. World J Surg. 1992;16:986–93. doi: 10.1007/BF02067013. [DOI] [PubMed] [Google Scholar]
- 12.Bhutani MS, Hawes RH, Hoffman BJ. A comparison of the accuracy of echo features during endoscopic ultrasound (EUS) and EUS-guided fine-needle aspiration for diagnosis of malignant lymph node invasion. Gastrointest Endosc. 1997;45:474–9. doi: 10.1016/s0016-5107(97)70176-7. [DOI] [PubMed] [Google Scholar]
- 13.Mountain CF, Dresler CM. Regional lymph node classification for lung cancer staging. Chest. 1997;111:1718–23. doi: 10.1378/chest.111.6.1718. [DOI] [PubMed] [Google Scholar]
- 14.Fujiwara T, Yasufuku K, Nakajima T, et al. The utility of sonographic features during endobronchial ultrasound-guided transbronchial needle aspiration for lymph node staging in patients with lung cancer: A standard endobronchial ultrasound image classification system. Chest. 2010;138:641–7. doi: 10.1378/chest.09-2006. [DOI] [PubMed] [Google Scholar]
- 15.Newman LS, Rose CS, Maier LA. Sarcoidosis. N Engl J Med. 1997;336:1224–34. doi: 10.1056/NEJM199704243361706. [DOI] [PubMed] [Google Scholar]
- 16.Annema JT, Veseliç M, Rabe KF. Endoscopic ultrasound-guided fine-needle aspiration for the diagnosis of sarcoidosis. Eur Respir J. 2005;25:405–9. doi: 10.1183/09031936.05.00098404. [DOI] [PubMed] [Google Scholar]
- 17.Bilaçeroğlu S, Perim K, Günel O, et al. Combining transbronchial aspiration with endobronchial and transbronchial biopsy in sarcoidosis. Monaldi Arch Chest Dis. 1999;54:217–23. [PubMed] [Google Scholar]
- 18.Trisolini R, Lazzari Agli L, Cancellieri A, et al. The value of flexible transbronchial needle aspiration in the diagnosis of stage I sarcoidosis. Chest. 2003;124:2126–30. doi: 10.1378/chest.124.6.2126. [DOI] [PubMed] [Google Scholar]
- 19.Cetinkaya E, Yildiz P, Altin S, et al. Diagnostic value of transbronchial needle aspiration by Wang 22-gauge cytology needle in intrathoracic lymphadenopathy. Chest. 2004;125:527–31. doi: 10.1378/chest.125.2.527. [DOI] [PubMed] [Google Scholar]
- 20.Garpestad E, Goldberg S, Herth F, et al. CT fluoroscopy guidance for transbronchial needle aspiration: An experience in 35 patients. Chest. 2001;119:329–32. doi: 10.1378/chest.119.2.329. [DOI] [PubMed] [Google Scholar]
- 21.Wildi SM, Judson MA, Fraig M, et al. Is endosonography guided fine needle aspiration (EUS-FNA) for sarcoidosis as good as we think? Thorax. 2004;59:794–9. doi: 10.1136/thx.2003.009472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fritscher-Ravens A, Sriram PV, Topalidis T, et al. Diagnosing sarcoidosis using endosonography-guided fine-needle aspiration. Chest. 2000;118:928–35. doi: 10.1378/chest.118.4.928. [DOI] [PubMed] [Google Scholar]
- 23.Yasufuku K, Chiyo M, Sekine Y, et al. Real-time endobronchial ultrasound-guided transbronchial needle aspiration of mediastinal and hilar lymph nodes. Chest. 2004;126:122–8. doi: 10.1378/chest.126.1.122. [DOI] [PubMed] [Google Scholar]
- 24.Choi YJ, Shin JH, Kim JH, et al. Tall cell variant of papillary thyroid carcinoma: Sonographic and clinical findings. J Ultrasound Med. 2011;30:853–8. doi: 10.7863/jum.2011.30.6.853. [DOI] [PubMed] [Google Scholar]
- 25.Lo WC, Chang WC, Lin YC, et al. Ultrasonographic differentiation between Kikuchi's disease and lymphoma in patients with cervical lymphadenopathy. Eur J Radiol. 2012;81:1817–20. doi: 10.1016/j.ejrad.2011.04.030. [DOI] [PubMed] [Google Scholar]
- 26.Khanna R, Sharma AD, Khanna S, et al. Usefulness of ultrasonography for the evaluation of cervical lymphadenopathy. World J Surg Oncol. 2011;9:29. doi: 10.1186/1477-7819-9-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee SJ, Paik YH, Lee DK, et al. The diagnostic value of endoprobe for small esophageal leiomyomas derived from the muscularis mucosae. Yonsei Med J. 2005;46:61–5. doi: 10.3349/ymj.2005.46.1.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen TK, Wu CH, Lee CL, et al. Endoscopic ultrasonography in the differential diagnosis of giant gastric folds. J Formos Med Assoc. 1999;98:261–4. [PubMed] [Google Scholar]
- 29.Melzer E, Fidder H. The natural course of upper gastrointestinal submucosal tumors: An endoscopic ultrasound survey. Isr Med Assoc J. 2000;2:430–2. [PubMed] [Google Scholar]
- 30.Akatsu T, Aiura K, Shimazu M, et al. Endoscopic ultrasonography of nonfunctioning pancreatic islet cell tumors with histologic correlation. Hepatogastroenterology. 2004;51:1590–4. [PubMed] [Google Scholar]


