Abstract
Familial adenomatous polyposis (FAP) is an autosomal dominant inherited syndrome characterized by multiple adenomatous polyps (predisposing to colorectal cancer development) and numerous extracolonic manifestations. The underlying genetic burden generates variable clinical features that may influence operative management. As a precancerous hereditary condition, the rationale of performing a prophylactic surgery is a mainstay of FAP management. The purpose of the present paper is to bring up many controversial aspects regarding surgical treatment for FAP, and to discuss the results and perspectives of the operative choices and approaches. Preferably, the decision-making process should not be limited to the conventional confrontation of pros and cons of ileorectal anastomosis or restorative proctocolectomy. A wide discussion with the patient may evaluate issues such as age, genotype, family history, sphincter function, the presence or risk of desmoid disease, potential complications of each procedure and chances of postoperative surveillance. Therefore, the definition of the best moment and the choice of appropriate procedure constitute an individual decision that must take into consideration patient’s preferences and full information about the complex nature of the disease. All these facts reinforce the idea that FAP patients should be managed by experienced surgeons working in specialized centers to achieve the best immediate and long-term results.
Keywords: Familial adenomatous polyposis, Surgical treatment, Restorative proctocolectomy, Ileal pouch-anal anastomosis, Ileorectal anastomosis, Adenocarcinomas
Core tip: This paper is an extensive review of the literature focusing the options and criteria for surgical treatment of patients with Familial Adenomatous Polyposis. The author put together a great number of dilemas and the current recommendations in order to help readers to understand how complex the disease is and to summarize the current knowledge.
INTRODUCTION
Familial adenomatous polyposis (FAP) is a genetic complex syndrome that may affect 1-8 per 10000 persons. Clinically, it is characterized by early development of a wide range of colorectal adenomatous polyps after the second decade of life and many extracolonic manifestations. If not treated by prophylactic colectomy, patients will have an almost 100% risk of developing a colorectal cancer (CRC).
Classic FAP and the attenuated form (AFAP) result from germline APC (adenomatous polyposis coli) mutations. FAP is an inherited autosomal dominant disease in which the majority of patients will be affected in a context of familial history, and almost 30% may have a “de novo” mutation. The less aggressive variant AFAP exhibits fewer colorectal adenomatous polyps (usually 10-100), later age of adenoma appearance and a lower cancer risk[1]. A subset of patients will exhibit a mutation in the base-excision-repair MUTYH gene, whose carriers may present a less severe and later polyposis when compared to FAP. Discovered in 2002, this recessive syndrome is called MUTYH associated polyposis (MAP), and CRC are frequent and discovered at the same moment as the polyposis[2].
FAP patients must be operated due to the almost inevitable adenoma-carcinoma sequence, and surgery has a positive impact on life expectancy[3]. In the past, most patients died from CRC, but this risk has been gradually reduced since the founding of the first Polyposis Registry by Lockhart-Mummery in St Mark’s Hospital in 1925 (Figure 1). Subsequently, the establishment of National Polyposis Registries in many countries changed morbidity patterns through early diagnosis and prophylactic colectomy, with a resulting improved prognosis[4]. Besides this, extracolonic manifestations such as desmoid disease, duodenal and pouch neoplasia may still pose important challenges to patients and surgeons.
Figure 1.

British surgeon John Percy Lockhart-Mummery (1875-1957) who established the famous Polyposis Registry in St. Marks Hospital (London).
Genetic testing and familial counseling should follow clinical and endoscopic diagnosis. Certain genotypes have been linked to specific extracolonic manifestations and may influence polyposis severity; moreover, it has been suggested that these associations should be considered in surveillance and therapeutic decisions (Table 1).
Table 1.
Phenotype-Genotype correlations in familial adenomatous polyposis patients
| Phenotype | Mutations | Authors |
| AFAP | APC extreme ends (exons 3,4,5) and exon 9 | Spirio et al[5] 1993 |
| Profuse polyposis (approximately 5000 polyps) | Between codons 1250-1464 | Nagase et al[6] 1992 |
| Severe polyposis and early CRC onset | Deletion in codon 1309 | Caspari et al[7] 1995 |
| Desmoid tumor | Between codons 1444-1580 | Caspari et al[7] 1995 |
| CHRPE | Between codons 463-1387 | Olschwang et al[8] 1993 |
| Thyroid cancer | 5' to codon 1220 | Cetta et al[9] 2000 |
| Duodenal adenomas | Between codons 976-1067 | Bertario et al[10] 2003 |
| Rectal cancer | Codons 1250 to 1464 | Bertario et al[11] 2000 |
AFAP: Attenuated familial adenomatous polyposis; APC: Adenomatous polyposis coli; CRC: Colorectal cancer; CHRPE: Congenital hypertrophy of the retinal pigment epithelium.
After diagnosis, surgery represents the mainstay of treatment for FAP patients (Figure 2). Surgical options include total colectomy with ileorectal anastomosis (IRA), total proctocolectomy with ileostomy (TPI), and restorative proctocolectomy (RPC) with or without mucosectomy and ileal-pouch anal anastomosis.
Figure 2.
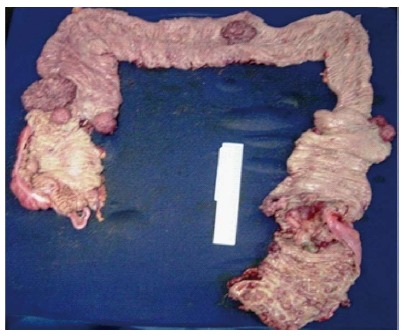
PAF specimen showing numerous polyps along the colon and at least seven simultaneous malignant lesions.
SURGICAL DECISION MAKING
As a rare and complex genetic disease, it is well recognized that FAP is better managed by a collaborative group of specialists. Several clinical and genetic features may influence surgical decisions regarding timing and extension of resection.
The present manuscript aims to raise and discuss several controversial issues concerning the surgical treatment of patients diagnosed with FAP.
Timing of surgery
There are no guidelines regarding the timing of surgery and most classical FAP patients undergo surgery between 15 and 25 years of age. Preferably prophylactic, an elective resection may be planned considering individual and family features, as well as patient’s preferences[12]. The main factors involved in timing of surgery are listed in Table 2.
Table 2.
Factors to be considered in the timing decision for surgery
| Reasons to indicate or postpone surgery | Timing for surgery |
| Presence of symptoms (> risk of CRC) | As soon as possible |
| Asymptomatic patient with mild disease | Discuss opportunity (before 20 years?) |
| Sized lesions or with high-grade dysplasia, not amenable to endoscopic resection | Immediately |
| Severe disease at colonoscopy or by family history/genotype | As soon as practicable |
| Attenuated polyposis at colonoscopy or by family history/genotype | Personal decision (16-20 years if mild or 21-25 years if attenuated polyposis ) |
| Preoperative diagnosis, positive family history or genetically susceptible for desmoids | Delay surgery (after evaluating CRC risk) |
CRC: Colorectal cancer.
Obviously, adenoma-associated symptoms such as diarrhea, bleeding, malnutrition or growth retard may encourage surgical indication on the next available opportunity. Also, specific endoscopic and histological features (presence of numerous adenomas, sized lesions or high-grade dysplasia) represent good reasons to indicate a prophylactic colectomy as soon as possible. Even asymptomatic patients with severe polyposis should not have their surgical procedure postponed.
It is important to emphasize that those patients with severe polyposis or CRC are more likely to be symptomatic. Among our own patients, CRC incidence was much lower in asymptomatic patients (1.1% vs 65.8%). Moreover, patients without CRC presented a shorter length of symptoms (15.2 mo vs 26.4 mo) and less frequent weight loss (11.4% vs 33.9%)[13]. Similarly, Bülow et al[14] reported a 60% incidence of CRC in patients with symptomatic FAP.
Besides symptoms, age is also an important factor related with CRC risk. In untreated patients, the mean age of CRC diagnosis and subsequent death have been reported to be 39 and 42 years, respectively[15]. In a review of 1073 patients from European countries, the risk of having a carcinoma at an age less than 20 years was estimated to be approximately 1%[16]. Within our own series, average age of patients without CRC was lower at treatment (29.5 years vs 40.0 years, P = 0.001)[13]. Age distribution revealed a cumulative incidence of 1.9% and 32.1% in patients with less than 20 and 30 years of age, respectively. This data is similar to the 15% incidence in patients before the age of 25 years at the St Mark’s Hospital[17].
These data explain why surgery may be deferred till the late teens or beginning of the third decade in most patients[18], after physical, emotional and social maturation are established[19]. Once FAP is diagnosed, there is a recommendation for immediate surgery for severe cases (> 1000 colonic and/or > 20 rectal polyps) as soon as practicable[20]. A similar approach should be employed in patients with family history of severe disease[12].
In another situations, FAP diagnosis does not require the need to perform surgery immediately. For example, an elective procedure may be delayed if an asymptomatic (at-risk or mutation carrier) young patient is compliant with surveillance and polyposis is not severe, since he understands there is a risk for CRC. In this setting, a personal decision based on psychological well-being and patient/family preferences related to school education or professional issues may also affect the planning of surgery. Considering that most patients will be treated around their twenties, doctors should discuss with young patients the possibility of sexual dysfunction following rectal resection, a technique-associated complication due to pelvic plexus lesion occurring in less than 4% of the patients[1].
Also, patients presenting an AFAP (by phenotype or genotype) or with a suggestive family history may be operated later, as CRC will generally occur in the early to mid-50s[21]. Another reason to delay surgery would be a preoperative diagnosis or the treatment of high-risk patients (positive family history or genetically susceptible) for desmoid tumor, if there were no imminent risk for CRC[22].
Selecting the best procedure: Ileal-anal or IRA?
The decision-making process to surgical alternatives must be tailored to the disease severity as well as to patient’s age, clinical conditions and personal preferences. Certainly, surgeon’s experience and technical skills may influence the final choice[21]. The factors that may affect surgical decision are listed in Table 3.
Table 3.
Patient’s and disease factors affecting operative choices
| Patient | Disease |
| Age, sex, obesity, prior surgery | Number and location of polyps |
| Genetics - family history | Colorectal cancer or metastatic disease |
| Female fecundity | Presence or risk of desmoid disease |
| Compliance with follow-up | AFAP |
| Acceptance of a temporary stoma | MAP |
MAP: MYH associated polyposis; AFAP: Attenuated familial adenomatous polyposis.
Proctocolectomy with ileostomy is the less common operation performed and must be reserved for patients with low rectal cancer, sphincter dysfunction, when a mesenteric desmoid prevents pouch construction or when it is impossible to pull the pouch down to the pelvis. This operative choice may lead to profound body image and emotional alterations related to the stoma and sexual dysfunction after pelvic dissection. Differently, surgical decision for most patients will confront IRA and RPC, and until now the published literature reached no consensus about this issue, making the choice between RPC and IRA a continuous matter of debate[23]. Discussion of the pros and cons of these techniques must not be oversimplified, cause several operative, oncological and functional variables must come into debate (see Table 4).
Table 4.
Recommendations for surgical treatment based on clinical and genetic features
| Operation | IRA | RPC |
| Indications | Mild FAP or MAP (< 20 rectal or < 1000 colonic polyps)AFAP by family history, endoscopy or genetic testingNo colorectal carcinomaYoung women without definitive offspringMetastatic CRC | Many (> 20) rectal adenomas or > 3 cm or high-grade dysplasiaSevere colonic phenotype(> 1000) or family historyColorectal carcinomaMutations in codon 1309Mesenteric desmoid or family history or APC mutation (codons 1403-1578) |
| Pros | Technically simple, good function, low morbidity, no pelvic dissection | |
| Cons | Metachronous rectal cancer | Technically demandingHigh morbidity |
AFAP: Attenuated familial adenomatous polyposis; MAP: MYH associated polyposis; IRA: Ileorectal anastomosis; CRC: Colorectal cancer; RPC: Restorative proctocolectomy; APC: Adenomatous polyposis coli.
IRA is generally recommended for patients with few rectal polyps, with AFAP, a family history of mild phenotype and for those young women with desire to be pregnant. IRA should not be performed in patients with a diseased rectum (adenomas > 3 cm, with severe dysplasia, cancer or sphincter dysfunction) or colon cancer. Patients with other features should undergo RPC[24-26].
Besides being a rectal sparing prophylactic colectomy, IRA provides good surgical and functional outcomes, but requires long-term follow-up of the rectum. During the pre-pouch era, metachronous rectal cancer rates varied from 15% to 40%, decreasing to less than 10% after pouch surgery came into practice[21,25]. After IRA, development of rectal cancer risk depends on several risk factors such as length of follow-up, chronologic age and local of APC mutation[11,26].
Certainly, bad selection criteria in the era before RPC accounts for the high rates of rectal recurrence after IRA. Today it is possible to select better candidates for IRA, once the risk of proctectomy is much lower in those presenting less than 20 rectal or 1000 colonic polyps[27]. In a series of 776 IRA from a multicenter Scandinavian study, proctectomy was performed in 229 of 576 patients (40%) during the prepouch period vs 26 of 200 (13%) during the pouch period[25].
Thus, IRA may provide good results in AFAP, MAP and mild FAP patients that agree to undergo follow-up, and RPC should be reserved for those with profuse polyposis[21,22]. Consequently, a policy of blanket RPC is not a good idea for patients with mild or attenuated disease, mainly for those asymptomatic and at a young age.
During the last decades, RPC progressively turned out to be the most common procedure despite its surgical morbidity. For this reason, candidates to RPC must be aware of its technical complexity and the reported high complication rates[28]. Comparative studies with IRA have reported higher complications after RPC[29,30], although this has not been a unanimous finding[31]. In our own series[26], we also observed more complications after RPC (48.1%) compared to proctocolectomy with ileostomy (26.6%) and IRA (19.0%) (P = 0.03).
Furthermore, pelvic dissection may lead to urinary and sexual dysfunction such as decreased fecundity in women (but it doesn’t risk pregnancy) and male impotence[32,33]. Indeed, it has been reported a 50% reduction in female fecundity after RPC[32]. As IRA doesn’t involve pelvic dissection, young females of childbearing age must be informed of this fact in order to refine the decision-making process.
Although not yet proven, RPC has been associated with a greater risk of desmoid disease when compared to IRA[34]. If this information turns out to be true, this chance must be critically evaluated as desmoid disease is an important cause of mortality among FAP patients.
Long-term functional results have been generally better after IRA[35,36]. In a meta-analysis of 12 selected studies (1002 FAP) comparing functional outcome and quality of life between RPC and IRA[37], bowel frequency, night defecation and use of incontinence pads were significantly less in the IRA group, although fecal urgency was more frequent with IRA compared with ileal pouch-anal anastomosis (IPAA). Reoperation within 30 d was more common after IPAA. There was no significant difference between the procedures in terms of sexual dysfunction, dietary restriction or postoperative complications (bowel obstruction, hemorrhage, intra-abdominal sepsis, and anastomotic leak). Rectal cancer was only observed in the IRA group (5%). In addition, abdominal reoperation on the rectum was more frequent after IRA (28%) vs IPAA (3%). The study demonstrated the individual merits and weaknesses of IRA and IPAA. Generally, better functional results are attributed to IRA, although there are no significant differences regarding quality of life[35,38].
Another capital issue regarding RPC is oncological. Although it was initially thought that RPC would abolish the risk of neoplasia, adenomas may develop within the ileal pouch many years after the surgical treatment[39]. Furthermore, there is a risk of malignant transformation attested by reports of cancer at the ileal pouch or at the ATZ[40,41]. These data clearly reflect that RPC is not a cancer-free procedure.
Then, surgical experience with RPC assumes a great importance. FAP patients should be advised to have the operation performed in medical centers that are familiar with FAP and by surgeons with proper training to perform this procedure.
CONTROVERSIAL ISSUES
Should genetic guide surgery?
As already stated, the final decision depends on many factors that include a fully informed patient. However, no consensus has been reached regarding the use of genotype to guide surgical option, mainly in patients with a polyp-free rectum. This idea is based on the relations between APC genotype and colonic polyposis. There are mutations associated with severe (between codons 1250 and 1464, especially at codon 1309), mild (extreme ends of the gene and in the alternatively spliced part of exon 9) and intermediate (in the remaining parts of the gene) forms of the disease[42].
In an important study containing data from four National Polyposis Registries, there were analyzed cumulative risks of proctectomy and cancer twenty years after primary colectomy in four hundred and seventy-five polyposis patients[43]. The authors registered cumulative risks of secondary proctectomy of 10%, 39%, and 61% in the attenuated, intermediate, and severe genotype groups, respectively (P < 0.05). Risks of cancer were not different. This data clearly shows that mutation analysis may be used to predict the risk of secondary proctectomy, and thus may help select the best option in patients with a few rectal adenomas.
Surgical traumas, hormonal influences in women, family history and genotype (APC mutation between codons 1445 and 1578) have been listed into a desmoid risk scoring system for FAP patients[44]. Recognition of this genotype-phenotype association has influenced surgical decision-making and has also served as an alert to delay or avoid surgery in order to prevent desmoid disease. This recommendation is especially important in those with 3′ APC mutations, which are associated with a 65% risk of developing mesenteric desmoids[45].
Handsewn or mechanical anastomosis?
IPAA may be accomplished either by handsewn or stapled anastomosis, and the relative benefits of each technique are a source of intense debate, as these two options may impact the outcome after RPC.
At the beginning, IPAA was performed manually after anorectal mucosectomy above the dentate line. Subsequently, this supposed advantage of removing all at-risk mucosa in ulcerative colitis and FAP patients proved to be insufficient after reports of islands of rectal mucosa left behind during dissection. A natural technical advance in FAP surgery was achieved with the introduction of stapled anastomosis. Besides been technically easier and faster, it promotes better function when compared to hand-sewn anastomosis due to the less sphincter manipulation and the preservation of a small rectal cuff above the anastomosis.
Against the premise that RPC would eliminate cancer risk, there is accumulating evidence that adenomas may develop within the pouch in 8%-74% and also at the anal transition zone (ATZ). Still controversial, potential factors for the development of pouch polyposis have been investigated. Ileal pouch adenomas are more common in patients older than 50 years of age or those presenting more than 1000 colonic adenomas. Age of the pouch is also important, with risk varying from 7%-16% after 5 years to 35%-42% after 10 years and 75% after 15 years[40,46].
As shown in Table 5, the incidence of polyps at the ATZ is usually lower after handsewn when compared to double-stapled anastomosis[36,47,48,49]. The presumable thinking that the remnant mucosal left behind after stapled anastomosis carries the same risk of malignization as the original disease, one could think that mucosectomy is probably the ideal choice. The risk of malignization within the pouch is probably low, as there are only thirty cases of cancer reported both at the anastomotic site and in the ileal pouch so far[39]. But since invasive cancer has been reported to occur either from the preserved ATZ or from retained mucosal remnants, a reduction of the cancer risk should not guide solely the operative choice. Consequently, as the natural course and significance of pouch polyps are better understood, the facts discussed here raise the need for long-term endoscopic pouch surveillance regardless of the anastomosis performed.
Table 5.
Incidence of adenomatous polyps at the anal transition zone or anastomotic site after handsewn or double-stapled anastomosis
| Study | Handsewn anastomosis | Double-stapled anastomosis | Follow-up (yr) |
| Remzi et al[47] | 21% in the pouch14.3% in the ATZ | 11% in the pouch28% in the ATZ | 5.8 |
| Von Roon et al[48] | 27% | 54% | 10 |
| Friederich et al[49] | 29% | 64% | 7 |
| Van Duijvendijk et al[36] | 10% | 31% | 7 |
ATZ: Anal transition zone.
Another important question regarding handsewn and stapled anastomosis is the confrontation of functional results and morbidity. As already reported, even though mucosectomy may reduce the risk of adenoma and cancer formation, it has been related to worse functional outcomes[50].
Is it safe to perform an ileal-pouch anastomosis without a diverting ileostomy?
Despite its intrinsic technical complexity, RPC is safe (mortality: 0.5%-1%) and carries an acceptable risk of non-life-threatening complications (10%-25%), achieving good long-term functional outcome with excellent patient satisfaction (over 95%).
A temporary protective ileostomy proximal to pouch has been classically performed in order to mitigate the effects of anastomosis leakage and to prevent pelvic sepsis (reported in 6% and 37%), fistulization and thus compromise pouch function. Consequently, it should also prevent the need for re-laparotomy and, most importantly, pouch failure[51]. Although most patients exhibit a very good acceptance of this temporary stoma, it may be a source of several complications.
Ileostomy omission has been advocated in selected cases, with the rationale that it is associated with similar rates of septic complications and may also provide economic advantages[52]. Selection criteria should exclude clinical (high doses of steroids, malnutrition, toxicity or anemia) and technical factors (difficult procedures with intraoperative complications). Furthermore, surgeons must be sure that the ileoanal anastomosis is tension-free, that it is supplied with adequate blood flow, that the tissue rings are intact and that air leaks are absent[53,54].
Several studies identified the underlying disease (ulcerative colitis) as a risk factor for pouch-related sepsis[50,51]. Within this context, its omission is attributed to the general fewer RPC complications rates in FAP than in ulcerative colitis. At diagnosis, FAP patients usually exhibit few symptoms and good general conditions, a different picture from those with ulcerative colitis. And when comparing septic complications with and without ileostomy, most cases have been attributed to steroid use.
In a paper from Saint Antoine Hospital[55], the authors reported their experience with 71 patients (38 females) who underwent laparoscopic RPC between November 2004 and February 2010. Indications were FAP (34), ulcerative colitis (35), indeterminate colitis (1) and Lynch syndrome (1). Laparoscopic RPC was performed as a one-stage procedure in 49 patients, and after a sub-total colectomy in 22. Seven patients in each group underwent the formation of a diverting stoma. Sixteen patients experienced at least one postoperative complication. The postoperative morbidity was 29% (n = 4/14) and 21% (n = 12/21) in patients with and without a stoma (P = 0.8), and the rate of fistula was 21% and 5%, respectively (P = 0.08). Seven percent of patients with a stoma and 16% without stoma had an intra-abdominal collection (P = 0.7). Nine patients required reoperation, which was not influenced by the presence or absence of a diverting stoma. The results of this study are similar to other laparoscopic RPC series.
Omission of ileostomy may have a great impact on young patients at school age. Once large-bowel techniques are evolving rapidly, the selection criteria for not performing an ileostomy after laparoscopic RPC, especially in FAP, still needs to be clarified. López-Rosales et al[56] reported good results in eight out of ten patients who underwent IPAA without protection. Ky et al[57] registered eleven postoperative complications and three reoperations among 32 patients. In our own series, one patient who had undergone one-stage procedure developed a postoperative fistula successfully treated with intestinal deviation. So far, we have preferred to perform laparoscopic RPC with ileostomy, and this choice is also based on the potential risk of desmoid tumors in FAP, which has been associated with surgical trauma among other predictive factors[58].
The review of the pertinent literature leads to the recognition that selective omission of a protective ileostomy may be safe and associated with similar septic complications and failure rates when compared with stoma patients. However, this finding forces us to critically evaluate FAP patient selection criteria, in which an experienced surgical team, a patient with a good clinical status and a procedure without adverse intraoperative outcomes should necessarily be included[59].
Laparoscopic or open approach?
During the last decade, surgical technique has evolved significantly, mainly with the crescent incorporation of laparoscopic techniques to accomplish complex procedures such as total abdominal (procto) colectomy. Acceptance of laparoscopic extensive resections is still controversial due to obstacles such as technical intraoperative difficulties, greater length of surgical procedure and the need for specialized training and instruments. Furthermore, most of the current literature includes case-matched studies and case series that deal with patients suffering from different diseases.
Regardless of these limitations, many publications have demonstrated the feasibility, safety, and good functional outcome in patients with ulcerative colitis and FAP. These patients are ideal candidates for a minimally invasive approach as these diseases usually affect young, motivated and body image conscious patients. Thus, the laparoscopic approach may be a useful mean to minimize morbidity in this population[60].
A review of the literature concerning laparoscopy usually reveals greater operative time, no difference in mortality, complication, reoperation and readmission rates; higher cosmesis scores and less blood loss have also been widely reported[61-63]. In our own experience with 49 patients, we registered a very low conversion rate (2%) and no patient required blood transfusion. Moreover, we observed 24.5% complications, 2% mortality and only 14.3% reoperations. Median length of hospital stay was only 6.2 d[58].
Besides lessening the body image impact, another potential advantages may be associated with laparoscopy. There exists an amount of evidence suggesting a reduction in abdominal and pelvic adhesions, which could result in less small bowel obstruction (SBO)[64] and improved fertility[65]. But the idea regarding a lower incidence of SBO has been critically debated[66,67]. RPC is known to be associated with postoperative infertility in open surgery, which may be caused by pelvic adhesions affecting the fallopian tubes. However, fertility after laparoscopic IPAA has been rarely assessed. The group from Paris analyzed 63 patients aged 45 or less by questionnaire[65]. The results were compared with those of controls undergoing laparoscopic appendectomy. Most patients (73%) suffered from ulcerative colitis. The authors found that 73% of the patients who attempted pregnancy after IPAA were able to conceive, and suggest that the infertility rate appears to be lower after laparoscopic IPAA than after open surgery.
Another important conclusion came from another institution[68] where 50 patients were evaluated by questionnaire after attempting to conceive. This study grouped patients with FAP (12), ulcerative colitis (37) and colonic ischemia (1). Comparison of open (23; 46%) and laparoscopic (27; 54%) RPC revealed a higher pregnancy rate after laparoscopic ileal anal-pouch anastomosis (log-rank, P = 0.023), suggesting laparoscopy to be the best approach in young women.
Taking into account all the controversial issues, one could argue whether those complex procedures should be performed only in specialized centers and by skilled and experienced surgical teams. The analysis of a greater number of patients allocated in randomized controlled trials may adequately elucidate the real dimension of the supposed advantages of laparoscopy for extended colorectal resections.
Is there a role for chemoprevention?
When facing an inherited cancer syndrome, management may be accomplished by genetic counseling, screening for at-risk lesions, chemoprevention, prophylactic surgery and lifetime surveillance. Current guidelines recommend that patients at risk for FAP should initiate endoscopic examination at 10-12 years of age, with continuing regular endoscopic surveillance until colectomy is advisable due to polyp burden, size or degree of dysplasia[16]. During this period, all significant sized adenomas should be removed if surgery has not been advised yet.
Ideally, FAP treatment would be pharmacological, as the NSAIDS, sulindac, celecoxib (selective cyclo-oxygenase-2 inhibitor) and aspirin may cause regression of established adenomatous polyps in individuals with FAP and may also reduce the number and size of colorectal adenomas. Thus, these drugs may act as an adjunct to postpone surgery in patients with mild polyposis or after ileal-rectal anastomosis. Besides these potential effects, chemoprevention alone is not suitable since CRC may develop even in patients with polyp suppression with NSAIDs[16].
Otherwise, chemoprevention with celecoxib may be an acceptable therapeutic option in cautiously selected FAP patients who present a high risk of rectal or duodenal cancer but a low risk of cardiovascular and thromboembolic events[69]. Within a context of primary therapy, it may be indicated for those who refuse surgery or have a high surgical risk, for patients with extensive desmoid disease or for ileal pouch polyposis whose treatment means an end-ileostomy[70].
Due to these properties, celecoxib was approved by the Food and Drug Administration (FDA) in 1999 and by the European Medicines Agency (EMEA) in 2003 to reduce the number of adenomatous colorectal polyps in individuals with FAP in conjunction with usual care (e.g., endoscopic surveillance and surgery)[69]. As an alternative to chemoprevention with celecoxib, it has been recognized that sulindac may present a stronger effect on the number of colorectal adenomas (although it doesn’t prevent them), and that its gastrointestinal-related toxicity may be managed with proton pump inhibitors[16].
CONCLUSION
All the data presented here clearly show how complex the decisions regarding FAP surgical treatment are. In this context, many disease and patient factors must be considered when taking the final choice. As a genetic disease associated with a great risk of CRC, the rationale of performing a prophylactic colectomy is a mainstay of FAP management. Patients should undergo an appropriate clinical evaluation and receive psychological support, since a great part of this population is young and recognize they suffer from a hereditary condition that usually affects other family members and deserves surveillance for life. In this way, the challenge of the working team (surgeon, gastroenterologist, genetic counselors and others) is to take individual decisions throughout the disease evolution based on the best available evidences and recommendations.
Footnotes
P- Reviewer: Ganschow P, Nagpal AP, O'Riordan JM S- Editor: Qi Y L- Editor: A E- Editor: Zhang DN
References
- 1.Half E, Bercovich D, Rozen P. Familial adenomatous polyposis. Orphanet J Rare Dis. 2009;4:22. doi: 10.1186/1750-1172-4-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bolocan A, Ion D, Stoian RV, Serban MB. Map syndrome (MYH Associated Polyposis) colorectal cancer, etiopathological connections. J Med Life. 2011;4:109–111. [PMC free article] [PubMed] [Google Scholar]
- 3.Phillips R, Clark SK. Polyposis Syndromes. In: Wolf BC, Fleshman JW, Beck DE, Pemberton JH, Wexner SD, et al., editors. The ASCRS Textbook of Colon and Rectal Surgery. New York: Springer; 2007. pp. 373–384. [Google Scholar]
- 4.Barrow P, Khan M, Lalloo F, Evans DG, Hill J. Systematic review of the impact of registration and screening on colorectal cancer incidence and mortality in familial adenomatous polyposis and Lynch syndrome. Br J Surg. 2013;100:1719–1731. doi: 10.1002/bjs.9316. [DOI] [PubMed] [Google Scholar]
- 5.Spirio L, Olschwang S, Groden J, Robertson M, Samowitz W, Joslyn G, Gelbert L, Thliveris A, Carlson M, Otterud B. Alleles of the APC gene: an attenuated form of familial polyposis. Cell. 1993;75:951–957. doi: 10.1016/0092-8674(93)90538-2. [DOI] [PubMed] [Google Scholar]
- 6.Nagase H, Miyoshi Y, Horii A, Aoki T, Ogawa M, Utsunomiya J, Baba S, Sasazuki T, Nakamura Y. Correlation between the location of germ-line mutations in the APC gene and the number of colorectal polyps in familial adenomatous polyposis patients. Cancer Res. 1992;52:4055–4057. [PubMed] [Google Scholar]
- 7.Caspari R, Olschwang S, Friedl W, Mandl M, Boisson C, Böker T, Augustin A, Kadmon M, Möslein G, Thomas G. Familial adenomatous polyposis: desmoid tumours and lack of ophthalmic lesions (CHRPE) associated with APC mutations beyond codon 1444. Hum Mol Genet. 1995;4:337–340. doi: 10.1093/hmg/4.3.337. [DOI] [PubMed] [Google Scholar]
- 8.Olschwang S, Tiret A, Laurent-Puig P, Muleris M, Parc R, Thomas G. Restriction of ocular fundus lesions to a specific subgroup of APC mutations in adenomatous polyposis coli patients. Cell. 1993;75:959–968. doi: 10.1016/0092-8674(93)90539-3. [DOI] [PubMed] [Google Scholar]
- 9.Cetta F, Montalto G, Gori M, Curia MC, Cama A, Olschwang S. Germline mutations of the APC gene in patients with familial adenomatous polyposis-associated thyroid carcinoma: results from a European cooperative study. J Clin Endocrinol Metab. 2000;85:286–292. doi: 10.1210/jcem.85.1.6254. [DOI] [PubMed] [Google Scholar]
- 10.Bertario L, Russo A, Sala P, Varesco L, Giarola M, Mondini P, Pierotti M, Spinelli P, Radice P. Multiple approach to the exploration of genotype-phenotype correlations in familial adenomatous polyposis. J Clin Oncol. 2003;21:1698–1707. doi: 10.1200/JCO.2003.09.118. [DOI] [PubMed] [Google Scholar]
- 11.Bertario L, Russo A, Radice P, Varesco L, Eboli M, Spinelli P, Reyna A, Sala P. Genotype and phenotype factors as determinants for rectal stump cancer in patients with familial adenomatous polyposis. Hereditary Colorectal Tumors Registry. Ann Surg. 2000;231:538–543. doi: 10.1097/00000658-200004000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ellis CN. Colonic adenomatous polyposis syndromes: clinical management. Clin Colon Rectal Surg. 2008;21:256–262. doi: 10.1055/s-0028-1089940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Campos FG, Nicácio De Freitas I, Imperiale AR, Seid VE, Perez RO, Nahas SC, Cecconello I. [Colorectal cancer in familial adenomatous polyposis: Are there clinical predictive factors?] Cir Esp. 2010;88:390–397. doi: 10.1016/j.ciresp.2010.05.013. [DOI] [PubMed] [Google Scholar]
- 14.Bülow S. Results of national registration of familial adenomatous polyposis. Gut. 2003;52:742–746. doi: 10.1136/gut.52.5.742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bussey HJ. Familial polyposis coli. Pathol Annu. 1979;14 Pt 1:61–81. [PubMed] [Google Scholar]
- 16.Vasen HF, Möslein G, Alonso A, Aretz S, Bernstein I, Bertario L, Blanco I, Bülow S, Burn J, Capella G, et al. Guidelines for the clinical management of familial adenomatous polyposis (FAP) Gut. 2008;57:704–713. doi: 10.1136/gut.2007.136127. [DOI] [PubMed] [Google Scholar]
- 17.Nugent KP, Phillips RK. Rectal cancer risk in older patients with familial adenomatous polyposis and an ileorectal anastomosis: a cause for concern. Br J Surg. 1992;79:1204–1206. doi: 10.1002/bjs.1800791136. [DOI] [PubMed] [Google Scholar]
- 18.McGrath DR, Spigelman AD. In the beginning there was colectomy: current surgical options in familial adenomatous polyposis. Hered Cancer Clin Pract. 2004;2:153–160. doi: 10.1186/1897-4287-2-4-153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Warrier SK, Kalady MF. Familial adenomatous polyposis: challenges and pitfalls of surgical treatment. Clin Colon Rectal Surg. 2012;25:83–89. doi: 10.1055/s-0032-1313778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Church JM, McGannon E, Burke C, Clark B. Teenagers with familial adenomatous polyposis: what is their risk for colorectal cancer? Dis Colon Rectum. 2002;45:887–889. doi: 10.1007/s10350-004-6322-x. [DOI] [PubMed] [Google Scholar]
- 21.Smith KD, Rodriguez-Bigas MA. Role of surgery in familial adenomatous polyposis and hereditary nonpolyposis colorectal cancer (Lynch syndrome) Surg Oncol Clin N Am. 2009;18:705–715. doi: 10.1016/j.soc.2009.07.006. [DOI] [PubMed] [Google Scholar]
- 22.Sturt NJ, Clark SK. Current ideas in desmoid tumours. Fam Cancer. 2006;5:275–85; discussion 287-8. doi: 10.1007/s10689-005-5675-1. [DOI] [PubMed] [Google Scholar]
- 23.Wolthuis AM, Leonard D, Kartheuser A, Bruyninx L, Van De Stadt J, Van Cutsem E, D’Hoore A. Different surgical strategies in the treatment of familial adenomatous polyposis: what’s the role of the ileorectal anastomosis? Acta Gastroenterol Belg. 2011;74:435–437. [PubMed] [Google Scholar]
- 24.Church J, Burke C, McGannon E, Pastean O, Clark B. Risk of rectal cancer in patients after colectomy and ileorectal anastomosis for familial adenomatous polyposis: a function of available surgical options. Dis Colon Rectum. 2003;46:1175–1181. doi: 10.1007/s10350-004-6710-2. [DOI] [PubMed] [Google Scholar]
- 25.Bülow S, Bülow C, Vasen H, Järvinen H, Björk J, Christensen IJ. Colectomy and ileorectal anastomosis is still an option for selected patients with familial adenomatous polyposis. Dis Colon Rectum. 2008;51:1318–1323. doi: 10.1007/s10350-008-9307-3. [DOI] [PubMed] [Google Scholar]
- 26.Campos FG, Perez RO, Imperiale AR, Seid VE, Nahas SC, Cecconello I. Surgical treatment of familial adenomatous polyposis: ileorectal anastomosis or restorative proctolectomy? Arq Gastroenterol. 2009;46:294–299. doi: 10.1590/s0004-28032009000400009. [DOI] [PubMed] [Google Scholar]
- 27.Church J, Burke C, McGannon E, Pastean O, Clark B. Predicting polyposis severity by proctoscopy: how reliable is it? Dis Colon Rectum. 2001;44:1249–1254. doi: 10.1007/BF02234779. [DOI] [PubMed] [Google Scholar]
- 28.Fazio VW, Kiran RP, Remzi FH, Coffey JC, Heneghan HM, Kirat HT, Manilich E, Shen B, Martin ST. Ileal pouch anal anastomosis: analysis of outcome and quality of life in 3707 patients. Ann Surg. 2013;257:679–685. doi: 10.1097/SLA.0b013e31827d99a2. [DOI] [PubMed] [Google Scholar]
- 29.Björk J, Akerbrant H, Iselius L, Svenberg T, Oresland T, Påhlman L, Hultcrantz R. Outcome of primary and secondary ileal pouch-anal anastomosis and ileorectal anastomosis in patients with familial adenomatous polyposis. Dis Colon Rectum. 2001;44:984–992. doi: 10.1007/BF02235487. [DOI] [PubMed] [Google Scholar]
- 30.Tonelli F, Valanzano R, Monaci I, Mazzoni P, Anastasi A, Ficari F. Restorative proctocolectomy or rectum-preserving surgery in patients with familial adenomatous polyposis: results of a prospective study. World J Surg. 1997;21:653–68; discussion 659. doi: 10.1007/s002689900289. [DOI] [PubMed] [Google Scholar]
- 31.Ambroze WL, Dozois RR, Pemberton JH, Beart RW, Ilstrup DM. Familial adenomatous polyposis: results following ileal pouch-anal anastomosis and ileorectostomy. Dis Colon Rectum. 1992;35:12–15. doi: 10.1007/BF02053332. [DOI] [PubMed] [Google Scholar]
- 32.Olsen KØ, Juul S, Bülow S, Järvinen HJ, Bakka A, Björk J, Oresland T, Laurberg S. Female fecundity before and after operation for familial adenomatous polyposis. Br J Surg. 2003;90:227–231. doi: 10.1002/bjs.4082. [DOI] [PubMed] [Google Scholar]
- 33.Church J. Surgery for Familial Adenomatous Polyposis. In: Rodriguez-Bigas MA, Cutai R, Lynch PM, Tomlinson I, Vasen HFA, et al., editors. Hereditary Colorectal Cancer. New York: Springer; 2010. pp. 203–218. [Google Scholar]
- 34.Vogel J, Church JM, LaGuardia L. Minimally invasive pouch surgery predisposes to desmoid tumor formation in patients with familial adenomatous polyposis. Dis Colon Rectum. 2005;48:662. [Google Scholar]
- 35.Soravia C, Klein L, Berk T, O’Connor BI, Cohen Z, McLeod RS. Comparison of ileal pouch-anal anastomosis and ileorectal anastomosis in patients with familial adenomatous polyposis. Dis Colon Rectum. 1999;42:1028–1033; discussion 1028-1033. doi: 10.1007/BF02236696. [DOI] [PubMed] [Google Scholar]
- 36.van Duijvendijk P, Slors JF, Taat CW, Oosterveld P, Vasen HF. Functional outcome after colectomy and ileorectal anastomosis compared with proctocolectomy and ileal pouch-anal anastomosis in familial adenomatous polyposis. Ann Surg. 1999;230:648–654. doi: 10.1097/00000658-199911000-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Aziz O, Athanasiou T, Fazio VW, Nicholls RJ, Darzi AW, Church J, Phillips RK, Tekkis PP. Meta-analysis of observational studies of ileorectal versus ileal pouch-anal anastomosis for familial adenomatous polyposis. Br J Surg. 2006;93:407–417. doi: 10.1002/bjs.5276. [DOI] [PubMed] [Google Scholar]
- 38.Van Duijvendijk P, Slors JF, Taat CW, Oosterveld P, Sprangers MA, Obertop H, Vasen HF. Quality of life after total colectomy with ileorectal anastomosis or proctocolectomy and ileal pouch-anal anastomosis for familial adenomatous polyposis. Br J Surg. 2000;87:590–596. doi: 10.1046/j.1365-2168.2000.01442.x. [DOI] [PubMed] [Google Scholar]
- 39.Wasmuth HH, Tranø G, Myrvold HE, Aabakken L, Bakka A. Adenoma formation and malignancy after restorative proctocolectomy with or without mucosectomy in patients with familial adenomatous polyposis. Dis Colon Rectum. 2013;56:288–294. doi: 10.1097/DCR.0b013e31827c970f. [DOI] [PubMed] [Google Scholar]
- 40.Tonelli F, Ficari F, Bargellini T, Valanzano R. Ileal pouch adenomas and carcinomas after restorative proctocolectomy for familial adenomatous polyposis. Dis Colon Rectum. 2012;55:322–329. doi: 10.1097/DCR.0b013e318241e6f2. [DOI] [PubMed] [Google Scholar]
- 41.Campos FG, Imperiale AR, Seid VE, Perez RO, da Silva e Sousa AH, Kiss DR, Habr-Gama A, Cecconello I. Rectal and pouch recurrences after surgical treatment for familial adenomatous polyposis. J Gastrointest Surg. 2009;13:129–136. doi: 10.1007/s11605-008-0606-8. [DOI] [PubMed] [Google Scholar]
- 42.Nieuwenhuis MH, Mathus-Vliegen LM, Slors FJ, Griffioen G, Nagengast FM, Schouten WR, Kleibeuker JH, Vasen HF. Genotype-phenotype correlations as a guide in the management of familial adenomatous polyposis. Clin Gastroenterol Hepatol. 2007;5:374–378. doi: 10.1016/j.cgh.2006.12.014. [DOI] [PubMed] [Google Scholar]
- 43.Nieuwenhuis MH, Douma KF, Bleiker EM, Bemelman WA, Aaronson NK, Vasen HF. Female fertility after colorectal surgery for familial adenomatous polyposis: a nationwide cross-sectional study. Ann Surg. 2010;252:341–344. doi: 10.1097/SLA.0b013e3181e9829f. [DOI] [PubMed] [Google Scholar]
- 44.Sinha A, Tekkis PP, Gibbons DC, Phillips RK, Clark SK. Risk factors predicting desmoid occurrence in patients with familial adenomatous polyposis: a meta-analysis. Colorectal Dis. 2011;13:1222–1229. doi: 10.1111/j.1463-1318.2010.02345.x. [DOI] [PubMed] [Google Scholar]
- 45.Speake D, Evans DG, Lalloo F, Scott NA, Hill J. Desmoid tumours in patients with familial adenomatous polyposis and desmoid region adenomatous polyposis coli mutations. Br J Surg. 2007;94:1009–1013. doi: 10.1002/bjs.5633. [DOI] [PubMed] [Google Scholar]
- 46.Tajika M, Niwa Y, Bhatia V, Tanaka T, Ishihara M, Yamao K. Risk of ileal pouch neoplasms in patients with familial adenomatous polyposis. World J Gastroenterol. 2013;19:6774–6783. doi: 10.3748/wjg.v19.i40.6774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Remzi FH, Church JM, Bast J, Lavery IC, Strong SA, Hull TL, Harris GJ, Delaney CP, O’Riordain MG, McGannon EA, et al. Mucosectomy vs. stapled ileal pouch-anal anastomosis in patients with familial adenomatous polyposis: functional outcome and neoplasia control. Dis Colon Rectum. 2001;44:1590–1596. doi: 10.1007/BF02234377. [DOI] [PubMed] [Google Scholar]
- 48.von Roon AC, Will OC, Man RF, Neale KF, Phillips RK, Nicholls RJ, Clark SK, Tekkis PP. Mucosectomy with handsewn anastomosis reduces the risk of adenoma formation in the anorectal segment after restorative proctocolectomy for familial adenomatous polyposis. Ann Surg. 2011;253:314–317. doi: 10.1097/SLA.0b013e318f3f498. [DOI] [PubMed] [Google Scholar]
- 49.Friederich P, de Jong AE, Mathus-Vliegen LM, Dekker E, Krieken HH, Dees J, Nagengast FM, Vasen HF. Risk of developing adenomas and carcinomas in the ileal pouch in patients with familial adenomatous polyposis. Clin Gastroenterol Hepatol. 2008;6:1237–1242. doi: 10.1016/j.cgh.2008.06.011. [DOI] [PubMed] [Google Scholar]
- 50.Lovegrove RE, Constantinides VA, Heriot AG, Athanasiou T, Darzi A, Remzi FH, Nicholls RJ, Fazio VW, Tekkis PP. A comparison of hand-sewn versus stapled ileal pouch anal anastomosis (IPAA) following proctocolectomy: a meta-analysis of 4183 patients. Ann Surg. 2006;244:18–26. doi: 10.1097/01.sla.0000225031.15405.a3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Heuschen UA, Hinz U, Allemeyer EH, Autschbach F, Stern J, Lucas M, Herfarth C, Heuschen G. Risk factors for ileoanal J pouch-related septic complications in ulcerative colitis and familial adenomatous polyposis. Ann Surg. 2002;235:207–216. doi: 10.1097/00000658-200202000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Joyce MR, Kiran RP, Remzi FH, Church J, Fazio VW. In a select group of patients meeting strict clinical criteria and undergoing ileal pouch-anal anastomosis, the omission of a diverting ileostomy offers cost savings to the hospital. Dis Colon Rectum. 2010;53:905–910. doi: 10.1007/DCR.0b013e3181d5e0fd. [DOI] [PubMed] [Google Scholar]
- 53.Remzi FH, Fazio VW, Gorgun E, Ooi BS, Hammel J, Preen M, Church JM, Madbouly K, Lavery IC. The outcome after restorative proctocolectomy with or without defunctioning ileostomy. Dis Colon Rectum. 2006;49:470–477. doi: 10.1007/s10350-006-0509-2. [DOI] [PubMed] [Google Scholar]
- 54.Rockall TA, Watkinson JC, Clark SE, Douek EE. Scintigraphic evaluation of glomus tumours. J Laryngol Otol. 1990;104:33–36. doi: 10.1017/s0022215100111727. [DOI] [PubMed] [Google Scholar]
- 55.Hor T, Zalinski S, Lefevre JH, Shields C, Attal E, Tiret E, Parc Y. Feasibility of laparoscopic restorative proctocolectomy without diverting stoma. Dig Liver Dis. 2012;44:118–122. doi: 10.1016/j.dld.2011.09.007. [DOI] [PubMed] [Google Scholar]
- 56.López-Rosales F, González-Contreras Q, Muro LJ, Berber MM, de León HT, Fernández OV, Veana RR. Laparoscopic total proctocolectomy with ileal pouch anal anastomosis for ulcerative colitis and familial adenomatous polyposis: initial experience in Mexico. Surg Endosc. 2007;21:2304–2307. doi: 10.1007/s00464-007-9523-2. [DOI] [PubMed] [Google Scholar]
- 57.Ky AJ, Sonoda T, Milsom JW. One-stage laparoscopic restorative proctocolectomy: an alternative to the conventional approach? Dis Colon Rectum. 2002;45:207–10; discussion 210-1. doi: 10.1007/s10350-004-6149-5. [DOI] [PubMed] [Google Scholar]
- 58.Campos FG, Araújo SE, Melani AG, Pandini LC, Nahas SC, Cecconello I. Surgical outcomes of laparoscopic colorectal resections for familial adenomatous polyposis. Surg Laparosc Endosc Percutan Tech. 2011;21:327–333. doi: 10.1097/SLE.0b013e31822b0f65. [DOI] [PubMed] [Google Scholar]
- 59.Campos FG. Current trends regarding protective ileostomy after restorative proctocolectomy. J Coloproctol. 2013;33:1–15. [Google Scholar]
- 60.da Luz Moreira A, Church JM, Burke CA. The evolution of prophylactic colorectal surgery for familial adenomatous polyposis. Dis Colon Rectum. 2009;52:1481–1486. doi: 10.1007/DCR.0b013e3181ab58fb. [DOI] [PubMed] [Google Scholar]
- 61.Ahmed Ali U, Keus F, Heikens JT, Bemelman WA, Berdah SV, Gooszen HG, van Laarhoven CJ. Open versus laparoscopic (assisted) ileo pouch anal anastomosis for ulcerative colitis and familial adenomatous polyposis. Cochrane Database Syst Rev. 2009;(1):CD006267. doi: 10.1002/14651858.CD006267.pub2. [DOI] [PubMed] [Google Scholar]
- 62.Tilney HS, Lovegrove RE, Heriot AG, Purkayastha S, Constantinides V, Nicholls RJ, Tekkis PP. Comparison of short-term outcomes of laparoscopic vs open approaches to ileal pouch surgery. Int J Colorectal Dis. 2007;22:531–542. doi: 10.1007/s00384-006-0177-7. [DOI] [PubMed] [Google Scholar]
- 63.Antolovic D, Kienle P, Knaebel HP, Schmidt J, Gutt CN, Weitz J, Koch M, Büchler MW, Seiler CM. Totally laparoscopic versus conventional ileoanal pouch procedure--design of a single-centre, expertise based randomised controlled trial to compare the laparoscopic and conventional surgical approach in patients undergoing primary elective restorative proctocolectomy--LapConPouch-Trial. BMC Surg. 2006;6:13. doi: 10.1186/1471-2482-6-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Dolejs S, Kennedy G, Heise CP. Small bowel obstruction following restorative proctocolectomy: affected by a laparoscopic approach? J Surg Res. 2011;170:202–208. doi: 10.1016/j.jss.2011.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Beyer-Berjot L, Maggiori L, Birnbaum D, Lefevre JH, Berdah S, Panis Y. A total laparoscopic approach reduces the infertility rate after ileal pouch-anal anastomosis: a 2-center study. Ann Surg. 2013;258:275–282. doi: 10.1097/SLA.0b013e3182813741. [DOI] [PubMed] [Google Scholar]
- 66.El-Gazzaz GS, Kiran RP, Remzi FH, Hull TL, Geisler DP. Outcomes for case-matched laparoscopically assisted versus open restorative proctocolectomy. Br J Surg. 2009;96:522–526. doi: 10.1002/bjs.6578. [DOI] [PubMed] [Google Scholar]
- 67.Fraser JD, Garey CL, Laituri CA, Sharp RJ, Ostlie DJ, St Peter SD. Outcomes of laparoscopic and open total colectomy in the pediatric population. J Laparoendosc Adv Surg Tech A. 2010;20:659–660. doi: 10.1089/lap.2010.0086. [DOI] [PubMed] [Google Scholar]
- 68.Bartels SA, DʼHoore A, Cuesta MA, Bensdorp AJ, Lucas C, Bemelman WA. Significantly increased pregnancy rates after laparoscopic restorative proctocolectomy: a cross-sectional study. Ann Surg. 2012;256:1045–1048. doi: 10.1097/SLA.0b013e318250caa9. [DOI] [PubMed] [Google Scholar]
- 69.Huang K, Gutierrez LP, Bülow S, Gallinger S, Castells A, Eagle CJ, Church JM. Clinical characteristics and outcomes in familial adenomatous polyposis patients with a long-term treatment of celecoxib: a matched cohort study. Fam Cancer. 2011;10:303–308. doi: 10.1007/s10689-011-9423-4. [DOI] [PubMed] [Google Scholar]
- 70.Lynch HT, Thorson AG, Smyrk T. Rectal cancer after prolonged sulindac chemoprevention. A case report. Cancer. 1995;75:936–938. doi: 10.1002/1097-0142(19950215)75:4<936::aid-cncr2820750407>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]


