Abstract
There have been numerous reports on the relationship between eosinophilia and toxocariasis. The present study investigated seropositive rates of toxocariasis among healthy people with or without eosinophilia in urban and rural areas, and assessed risk factors for positive antibody test. A total of 610 healthy people, who visited health check-up (Medicheck®, Korea Association of Health Promotion), 310 from Seoul and 300 from Gyeongsangnam-do, were subjected for this study. Their serum samples were tested by ELISA with the crude antigen of Toxocara canis larvae. Cross-reactions with other tissue invading helminth antigens were also investigated. Total antibody positive rate of toxocariasis was 8.7% of the 610 subjects. When the subjects were grouped into 3 by their eosinophil counts, the antibody positive rates significantly differed by the groups; 5.9% (18/306) in the group<350/µL, 10.0% (11/110) in the group 350-500/µL, and 12.4% (24/194) in the group>500/µL (P=0.028). A total of 22 serum samples cross-reacted with other tissue-invading helminth antigens. A questionnaire analysis recognized drinking alcohol and smoking as significant risk factors of toxocariasis. In conclusion, toxocariasis antibody positive rate is correlated with eosinophil counts. It is recommended that healthy subjects with eosinophilia by routine health examination and risk factors undergo Toxocara serology by multiantigen ELISA to investigate etiology.
Graphical Abstract
Keywords: Toxocara canis, Toxocariasis, Eosinophilia, ELISA, Larval Crude Antigen
INTRODUCTION
Migrating larvae of Toxocara canis or T. cati not only reside in the tissue of dogs or cats, but also infect other animals, including human bodies. The infection route of humans is well-known; accidental swallowing of embryonated eggs in soil or in contaminated food, or ingestion of encapsulated larvae of Toxocara canis in the tissue of a paratenic host (1). When humans are infected with larvae of Toxocara canis or Toxocara canis, it is commonly associated with eosinophilia.
Since both eosinophilia and toxocariasis themselves are usually asymptomatic, the diagnosis is inevitably delayed. Most of patients with toxocariasis with light infection live undiagnosed and those with heavy worm burden have been diagnosed after clinical manifestations appear due to damage of infected organs. The migrating Toxocara larvae may infiltrate into the liver, lungs, brain, or eyes, and result in hepatic abscess, pulmonary or pleural inflammation with effusion, meningitis, retinal inflammation, or other organ damage (2, 3, 4, 5).
In Korea, Toxocara canis is commonly found in the intestine of canids (6, 7, 8). There have been continuous interests on the antibody positivity in human serum, which greatly varied with the demography and health status of subjects. For example, the antibody positive rate of toxocariasis among rural adults was approximately 5% in Gangwon-do (9), 68% among patients with eosinophilia (more than 500/µL or more than or equal to 10% WBC) who visited the hospital in Gwacheon (10), 45.5% among patients with eosinophilia in Chungcheongnam-do (11), and 64.9% among healthy residents with eosinophilia in Seoul (12). In the United States, Toxocara serum antibody positive rate was reported up to 13.9% (13) while the infection rates of Toxocara canis and Toxocara canis were 1.62% and 4.03% respectively in pet dogs and cats (14). In addition, the laboratory findings with eosinophilia, hypergammaglobulinemia, and increased isohemagglutinin A and B titers were correlated with acute toxocariasis (15). Toxocariasis is a common infection of public health importance but neglected by physicians and public health workers in the United States (15).
In this context, there have been no data comparing the seropositive rate of toxocariasis among healthy populations between those with and without eosinophilia in Korea. The present study assessed the serum antibody positive rate of two groups in two different locations, Seoul and Gyeongsangnam-do and evaluated related risk factors.
MATERIALS AND METHODS
Subjects
From May 9 to October 22 2012, 610 subjects were randomly selected from the healthy people who received regular healthcare check-ups (MediCheck®) at the Korea Association of Health Promotion (KAHP); 310 from metropolitan Seoul and 300 from rural area in Gyeongsangnam-do, the southern part of Korea. These subjects were screened of eosinophil counts in the peripheral blood. Half of them were recruited from those with eosinophil count over 7%. The subjects who were taking any medications with a diagnosis within 6 months were excluded.
Antibody test for toxocariasis
A total of 610 serum samples were used in this study. The inhouse ELISA developed in the Institute of Endemic Disease, Seoul National University was used for serologic diagnosis of human toxocariasis, which detects human IgG antibodies to Toxocara canis larva antigen (TCLA) (16). The preparation process of TCLA was described by Jin et al. (16). The positive reactions by ELISA were examined again by western blotting.
Western blotting
The crude antigen of Toxocara canis larvae was separated by 10% SDS-PAGE (sodium dodecyl sulphate-polyacrylamide gel electrophoresis) and transferred onto polyvinylidene fluoride (PVDF) membrane. The membrane was cut into strips and blocked with 5% skim milk. The strips were incubated with serum samples (diluted 1:100 in 5% skim milk solution) at 4 overnight, and was followed by 1.3 mg/mL polyclonal rabbit anti-human IgG antiserum conjugated with horseradish peroxidase (HRP; Dako, Glostrup, Denmark) which was used at a dilution of 1:2,000 with 5% skim milk. The reaction was visualized using ECL™ Western blotting detection reagents (Amersham, Piscataway, NJ, USA).
Questionnaire
The interviewer gave a structured questionnaire to the examinees in both Seoul and Gyeongsangnam-do to collect data regarding subjects' characteristics. We obtained an informed consent from each examinee. The questionnaire includes housing, economic status, and social status. Also a history and a frequency of eating raw liver or meat of cows or other animals were questioned. The questionnaire also contained a question whether subjects had a recent history of keeping dogs or cats. Social history such as smoking, drinking alcohol, and travel history was also in the questionnaire.
Statistical analysis
All data analyses were performed with a commercial software program (IBM SPSS Statistics, version 21.0; SPSS, Chicago, IL, USA). A P value of <0.05 was considered as a statistical significance.
Ethics statement
The study protocol was reviewed and permitted by the institutional review board of the KAHP (12-C-04). All of the subjects submitted informed consent for this study at the enrollment.
RESULTS
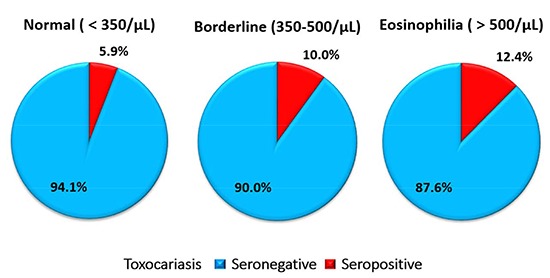
A total of 610 subjects came from Seoul (n=310) and Gyeongsangnam-do (n=300). Demographic data of the subjects showed no significant difference by the locality (Table 1). All of the subjects were grouped into three, based on the absolute eosinophil count in the peripheral blood; less than 350/µL as Group 1 (normal), between 350/µL and 500/µL as Group 2 (borderline), and over 500/µL Group 3 (eosinophilia). The count of 350/µL was derived by tentative criteria from the KAHP healthcare system, which defines eosinophilia as over 7% of peripheral white blood cells. Assuming that the number of circulating leukocytes is 5,000 per µL, the absolute eosinophil count based on the criteria could be deduced as 350 per µL.
Table 1.
Basic demography and the distribution of the subjects
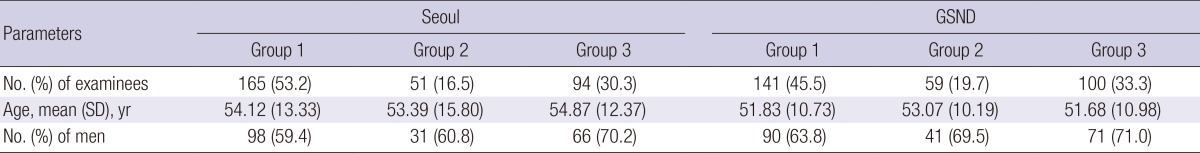
GSND, Gyeongsangnam-do; SD, standard deviation; Group 1, peripheral eosinophil count less than 350 per µL; Group 2, peripheral eosinophil count between 350 and 500 per µL; Group 3, peripheral eosinophil count over 500 per µL.
A total of 306 subjects (165 from Seoul and 141 from Gyeongsangnam-do) were in Group 1, 110 (51 from Seoul and 59 from Gyeongsangnam-do) in Group 2, and 194 subjects (94 from Seoul and 100 from Gyeongsangnam-do) in Group 3. Mean age of the subjects was similar in Groups 1, 2, and 3. The percentage of male was 61.4%, 65.5%, and 70.6% in the Group 1, 2, and 3, respectively. The detailed demographic profile of each group in Seoul and Gyeongsangnam-do is shown in Table 1. Overall, little variation by age or sex was shown in the demographic profile between Seoul and Gyeongsangnam-do and between the groups by eosinophil counts.
The cut-off value of absorbance for TCLA ELISA was defined to be an absorbance of 0.250 (16). Based on the cut-off value, the serum antibody positive rate was 6.1% in Seoul and 11.3% in Gyeongsangnam-do (P=0.033). In terms of the eosinophil group, the antibody positive rate increased as with the increase of the eosinophil count; 5.9% in Group 1, 10.0% in Group 2, and 12.4% in Group 3 (P=0.037). The rates of each group in Gyeognsangnam-do were higher than those in Seoul (Table 2).
Table 2.
Antibody positivity to antigens of tissue invading helminthes by multi-antigen ELISA in Seoul and Gyeongsangnam-do, Korea
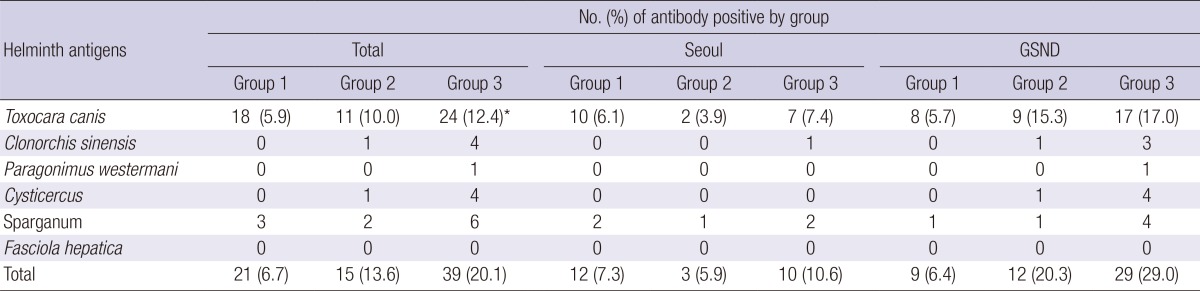
GSND, Gyeongsangnam-do; Group 1, peripheral eosinophil counts<350/µL; Group 2, 350-500/µL; Group 3, >500/µL. *P value=0.037.
The questionnaire answers, possible risk factors or possible conditions that could lead to toxocariasis, have been analyzed based on the eosinophil group in Table 3. Notwithstanding some limitations, which will be discussed later, gender, location, smoking status, drinking habit, and the history of raw meat ingestion are significantly related with the peripheral eosinophil count (Table 3).
Table 3.
Risk factors of toxocariasis in daily lives based on the questionnaires
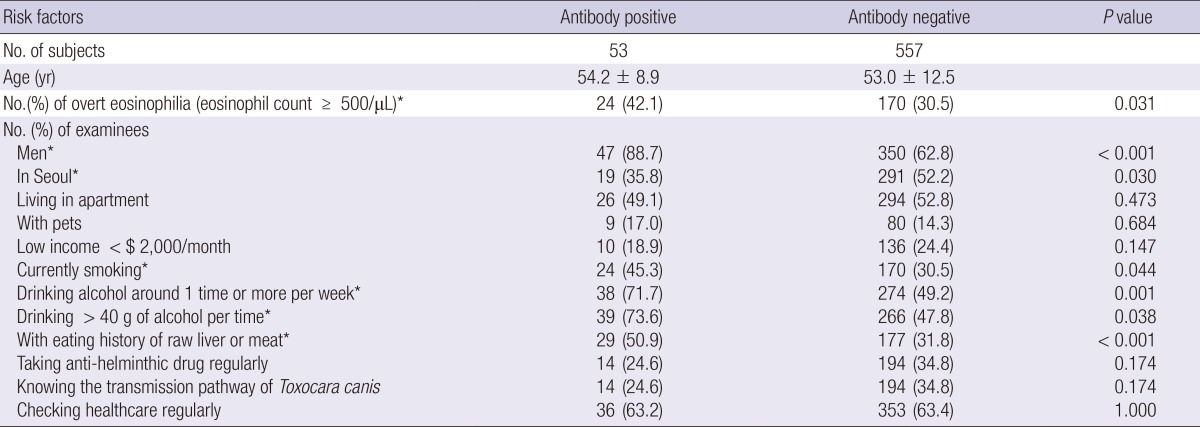
Seropositive, ELISA absorbance ≥0.250; *statistically significant.
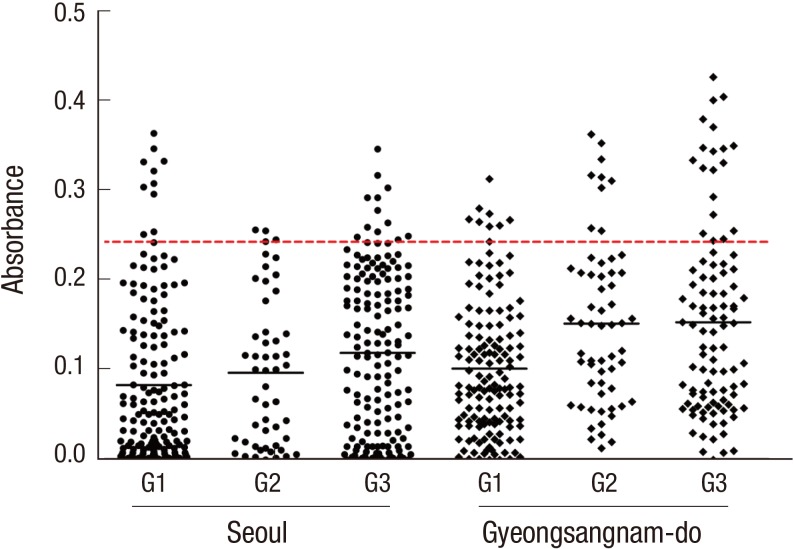
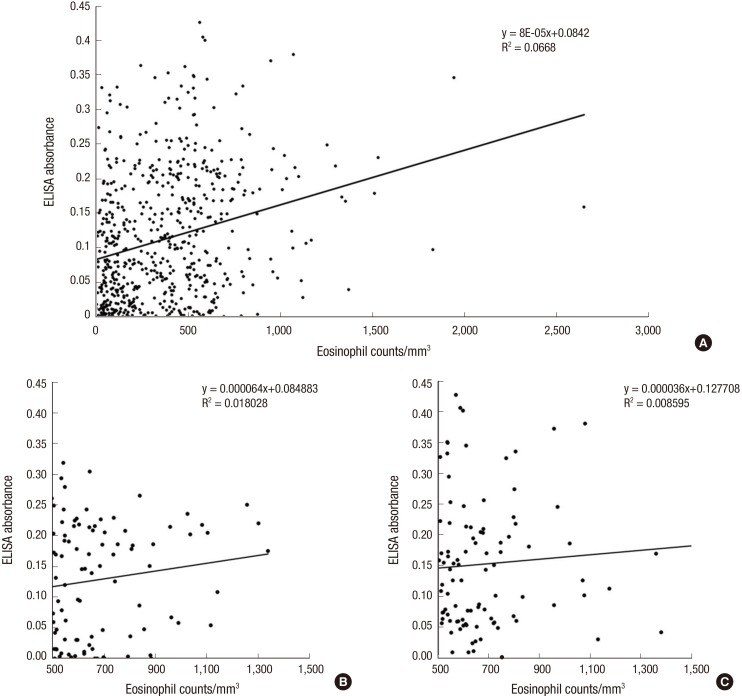
The distribution of ELISA absorbance for serum antibody is plotted by groups in Fig. 1. The absorbance demonstrated no significant correlation with the eosinophil counts (r=0.258, Fig. 2).
Fig. 1.
Distribution of ELISA absorbance to TCLA by eosinophil groups from Seoul and Gyeongsangnam-do. G1, peripheral eosinophil counts < 350/µL; G2, 350-500/µL; G3, > 500/µL.
Fig. 2.
Correlation between the TCLA ELISA absorbances and total eosinophil counts in the peripheral blood. (A) Total subjects. (B) Eosinophilia group (> 500/µL) in Seoul. (C) Eosinophilia group (> 500/µL) in Gyeongsangnam-do.
Among 610 subjects, there were 22 subjects who were presented as positive to other helminth antigens; 5 to Clonorchis sinensis, 1 to Paragonimus westermani, 5 to cysticercus, and 11 to sparganum (Table 2). Three of those with toxocariasis recognized positive reactions with other antigens.
In the questionnaire, 2 subjects in Group 2 and 5 in Group 3 answered to have allergy-related symptoms or signs (Table 4). However, the eosinophil group found no significant correlation with any of the conditions (P=0.498).
Table 4.
Underlying medical conditions among the subjects
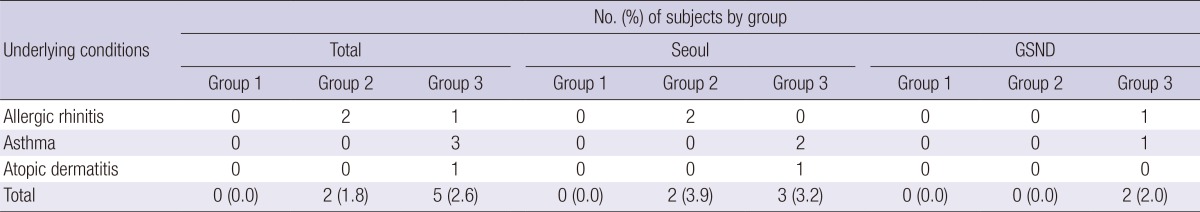
GSND, Gyeongsangnam-do; Group 1, peripheral eosinophil counts <350/µL; Group 2, 350-500/µL; Group 3, >500/µL.
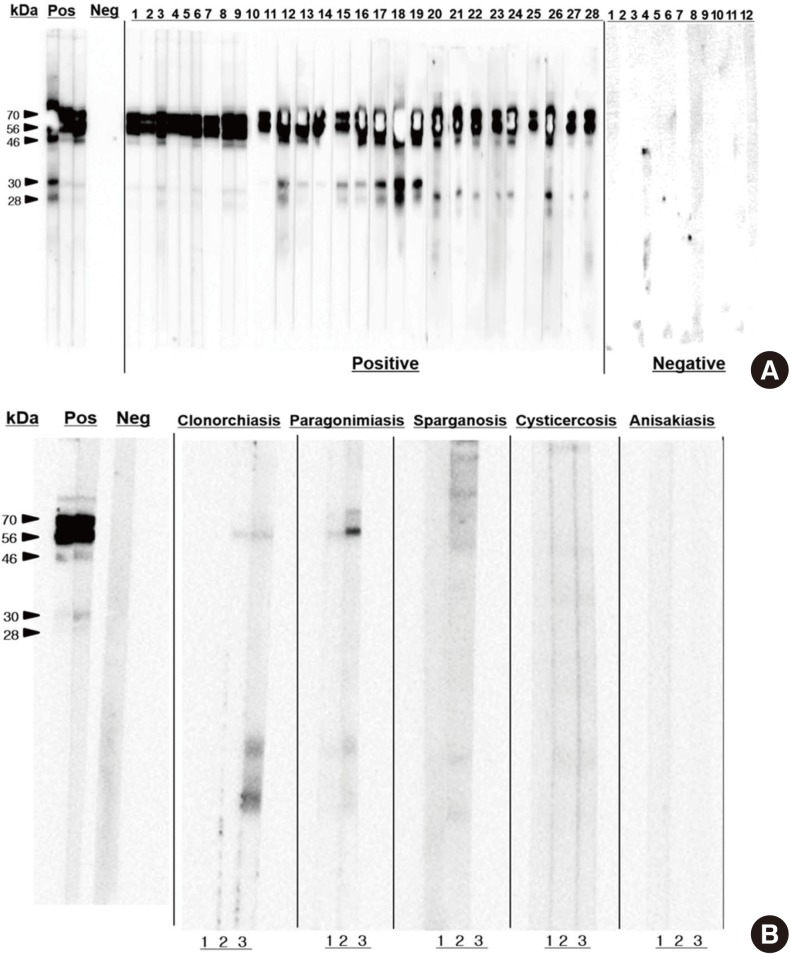
Western blotting for IgG antibodies to TCLA observed several reacting bands in the range of 28-70 kDa (Fig. 3). The reacting bands were grouped into high (56, 70 kDa) and low (28, 30 kDa). Western blotting with the serum samples that were positive with antigens of other tissue-invading helminthes observed no reacting bands with those of sparganum, cysticercus, and Anisakis. However, some cross-reactions were recognized in the high bands in positive serum samples to Clonorchis sinensis or Paragonimus westermani antigen.
Fig. 3.
Western blotting of serum samples. (A) Toxocariasis and control. (B) Other helminthiases.
DISCUSSION
The present study recognized overall serum antibody positive rate of human toxocariasis as 8.7% of the healthy subjects in Korea using TCLA ELISA. By area, the rate was 6.1% in Seoul (urban) and 11.3% Gyeongsangnam-do (rural). The rate in Gyeongsangnam-do was significantly higher than that in Seoul. Although excretory-secretory antigen of Toxocara canis larvae (TES) is the standard for serodiagnosis (17, 18, 19, 20, 21, 22) and most of previous studies in Korea used it, it was confirmed that the TCLA produced as good diagnostic values of 92.2% sensitivity and 86.6% specificity as high as TES (16). Furthermore western blotting of the TCLA recognized same band patterns with that of TES (Fig. 3). This fact enables us to compare the seropositivity records together (Table 5).
Table 5.
Comparison of seroprevalence of toxocariasis in Korea
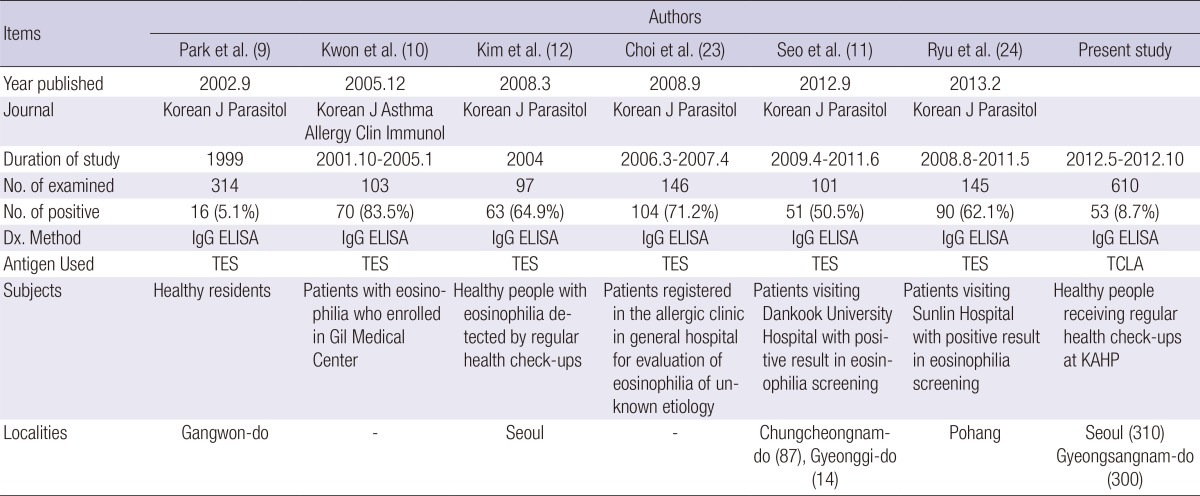
TES, excretory-scretory antigen of T. canis larvae; TCLA, crude antigen of T. canis larvae; KAHP, the Korea Association of Health Promotion.
One study described serum antibody positive rate of 5.1% in general population in a rural area (9) but others recorded 50% to 80% positive rates among people with eosinophilia (10, 11, 12, 23, 24). The reported serum antibody positive rate has varied quite widely by locality or subjects. The 300 subjects from Gyeongsangnam-do of the present study were similar with those reported by Park et al. (9) in Gangwon-do, both of whom lived in rural or suburb areas. All of the subjects of two studies were healthy adults; the present seropositive rate, however, was 11.3%, as twice high as 5.1% of the previous one. The previous one investigated at Gangwon-do in 1999 but the present study was done at Gyeongsangnam-do in 2012. It is hard to explain what made the difference of positive rate. Further studies subjecting more population at various localities may provide clues of the difference. The present results clearly demonstrated that the positive rate in metropolitan Seoul is significantly lower than that in rural areas.
The subjects of present study were classified into 3 by eosinophil counts; normal (<350/µL), borderline (350-500/µL), and eosinophilia (>500/µL). The serum antibody positive rate in the 3 groups was 5.9%, 10.0%, and 12.4% in order (P=0.037). The positive rate of 12.4% of eosinophilia group was very low compared to those over 50% of previous reports (10, 11, 12, 23, 24). Those positive rates of previous studies were rather high because they were suspected with some symptoms or signs of toxocariasis at hospitals but the subjects of the present study were healthy without any related clinical symptoms. All of them were recruited from healthcare check-ups in the present study. This shows that, even among the eosinophilia groups, the positive rate may vary significantly by individual subjects.
When the serum antibody positive rate was analyzed in detail by area, the correlation between eosinophil counts and antibody positivity was not significant in Seoul (P=0.698) but significant in Gyeongsangnam-do (P=0.014). The correlation has been consistently confirmed by several studies (10, 11, 12, 23, 24, 25, 26, 27). The insignificant correlation in Seoul might have been caused by small number of antibody positive subjects in each group. Nonetheless, the subjects with higher eosinophil count showed significantly higher seropositive rate, and we need to keep in mind that symptomless eosinophilia is the most frequent sign of adult-onset toxocariasis. Furthermore, the borderline group of eosinophil counts should also be suspected to confirm its diagnosis. Adults with eosinophilia or borderline counts, especially those who live in rural areas, could be considered as high risk group of toxocariasis and should receive serum antibody test for correct evaluation of etiology.
In 2013, the Korea Association of Health Promotion (KAHP) healthcare report released the healthcare data that 4.9% of examined subjects observed their eosinophil counts over 7% in Seoul and Gyeongsangnam-do. The present results indicated 11.5% (35/304) of them were antibody positive for toxocariasis. Consequently, if we extrapolate the findings based on this calculation, the serum antibody positive of toxocariasis would be on average 563.5 per 100,000 healthcare examinees in 2013. Surely, the serum antibody positive rates vary by methods, target subjects, localities, or sampling period. All of them are not clinical patients but some of them may be clinical patients in the future because of year-long survival of the larvae in the tissue. Furthermore, 5.9% of subjects with normal eosinophil counts (95.1% of screened subjects) were serum antibody positive. These results suggest that there are many hidden cases of covert toxocariasis in Korea. One editorial mentioned that about 4% of whole patients in a tertiary hospital in Seoul showed eosinophilia and most of them had history of eating raw liver of cows in Korea (28). The article focused on the fact that covert toxocariasis is neglected or abandoned by most physicians and even by patients in Korea (28).
Toxocariasis of the liver usually induces few symptoms unless heavy burden infections. In other words, if there is any symptom, it indicates that the parasite burden is heavy or does already have migrated into other vital organs such as the lungs, eyes, or brain. A few studies continuously claimed Toxocara canis as a cause of idiopathic complications of those vital organs (25, 29, 30). When the symptoms came to be called 'idiopathic,' it usually means that the progress has already been made and the tissue damage has occurred. Especially ocular toxocariasis, which induces visual disturbance, is critical for covert toxocariasis cases (31). Therefore, early detection of the infection is important, and this study illustrates the rationale and methods of serum antibody screening of symptomless toxocariasis. Of course it is recommended to medicate antibody positive cases when they have related symptoms or signs because the larvae may spread to other vital organs although it is not fatal.
In addition to the peripheral eosinophil count, gender, geographic location, current smoking status, drinking habits, and history of raw meat ingestion were found as risk factors of toxocariasis antibody positivity. This is plausible in that a man who lives in a rural area with drinking habit could hardly maintain his health in good status. Moreover, in other countries such as Brazil or Poland, toxocariasis in children is more concerned than that in adults. Main route of infection is by hand-to-mouth delivery of eggs of Toxocara spp. in the sand or grass in public squares (26, 27, 32). In Korea, however, infection in adults is more prevalent and problematic because common route of infection is ingestion of raw liver or meat of mammals or birds as side-dishes while drinking alcohol (33). The present study added drinking alcohol and smoking as significant risk factors of toxocariasis. These risk factors are common behaviors of aged men in rural areas in Korea.
Polyparasitism and consequent cross-reaction with Toxocara antigen should be considered in the present study. It is known that TCLA cross-reacts with serum samples of other helminthiases (16), so the multi-antigen ELISA is recommended. Especially nematode infections are cross-reacting more frequently (16, 18, 19, 27, 32), but it is fortunate that intestinal nematodes are rare in Korea (34). It is also plausible that the multiantigen positive subjects were genuine cases of polyparasitism. The multiantigen positive subjects were commonly found with antigens of sparganum, cysticercus, and Clonorchis sinensis in the eosinophilia group (Table 2). The findings suggest that some of them may be real positive to the antigens rather than cross-reaction because the reacting bands by western blotting were different from those of toxocariasis (Fig. 3). Moreover, underlying medical conditions of subjects that can increase the eosinophil count in the peripheral blood were investigated. Eosinophils increase as a result of immunologic response or diseases such as allergic rhinitis, asthma, and atopic dermatitis. The results were arranged in Table 4, but none of these diseases seemed noteworthy because of the small number of subjects.
The present study has some limitations. The subjects were recruited only from 2 localities, Seoul and Gyeongsangnam-do, which limited its generalization. Also the number of positive subjects was not large enough to confirm various significant parameters as risk factors. Lastly, all of the subjects were selected from healthcare examinees of the KAHP, which might have contributed a selection bias.
In conclusion, the results of this study clearly demonstrate that toxocariasis is rather common and a major cause of eosinophilia in adult Koreans. To detect toxocariasis in routine healthcare examination, it is recommended to screen serum samples of those with eosinophilia or borderline counts and also with risk factors by multi-antigen ELISA using TCLA.
ACKNOWLEDGEMENTS
All of the authors appreciate Ms. Hye-Seong Kim, Institute of Endemic Diseases, Seoul National University, for her technical supports of ELISA.
Footnotes
This study was supported by a grant from the Korea Association of Health Promotion, 2012.
The present authors have no potential conflicts to disclose.
References
- 1.Rubinsky-Elefant G, Hirata CE, Yamamoto JH, Ferreira MU. Human toxocariasis: diagnosis, worldwide seroprevalences and clinical expression of the systemic and ocular forms. Ann Trop Med Parasitol. 2010;104:3–23. doi: 10.1179/136485910X12607012373957. [DOI] [PubMed] [Google Scholar]
- 2.Ashwath ML, Robinson DR, Katner HP. A presumptive case of toxocariasis associated with eosinophilic pleural effusion: case report and literature review. Am J Trop Med Hyg. 2004;71:764. [PubMed] [Google Scholar]
- 3.Jung JK, Jung JT, Lee CH, Kim EY, Kwon JG, Kim BS. A case of hepatic abscess caused by Toxocara. Korean J Hepatol. 2007;13:409–413. doi: 10.3350/kjhep.2007.13.3.409. [DOI] [PubMed] [Google Scholar]
- 4.Park SP, Park I, Park HY, Lee SU, Huh S, Magnaval JF. Five cases of ocular toxocariasis confirmed by serology. Korean J Parasitol. 2000;38:267–273. doi: 10.3347/kjp.2000.38.4.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee JY, Kim BJ, Lee SP, Jeung YJ, Oh MJ, Park MS, Paeng JW, Lee BJ, Choi DC. Toxocariasis might be an important cause of atopic myelitis in Korea. J Korean Med Sci. 2009;24:1024–1030. doi: 10.3346/jkms.2009.24.6.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cho SY, Kang SY, Ryang YS. Helminthes infections in the small intestine of stray dogs in Ejungbu City, Kyunggi Do, Korea. Korean J Parasitol. 1981;19:55–59. doi: 10.3347/kjp.1981.19.1.55. [DOI] [PubMed] [Google Scholar]
- 7.Huh S, Sohn WM, Chai JY. Intestinal parasites of cats purchased in Seoul. Korean J Parasitol. 1993;31:371–373. doi: 10.3347/kjp.1993.31.4.371. [DOI] [PubMed] [Google Scholar]
- 8.Kim YH, Huh S. Prevalence of Toxocara canis, Toxascaris leonina and dirofilaria immitis in dogs in Chuncheon, Korea (2004) Korean J Parasitol. 2005;43:65–67. doi: 10.3347/kjp.2005.43.2.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Park HY, Lee SU, Huh S, Kong Y, Magnaval JF. A seroepidemiological survey for toxocariasis in apparently healthy residents in Gangwon-do, Korea. Korean J Parasitol. 2002;40:113–117. doi: 10.3347/kjp.2002.40.3.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kwon NH, Oh MJ, Lee SP, Lee BJ, Choi DC. The clinical impact of toxocariasis in patients with unknown eosinophilia. Korean J Asthma Allergy Clin Immunol. 2005;25:299–304. [Google Scholar]
- 11.Seo M, Yoon SC. A seroepidemiological survey of toxocariasis among eosinophilia patients in Chungcheongnam-do. Korean J Parasitol. 2012;50:249–251. doi: 10.3347/kjp.2012.50.3.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kim YH, Huh S, Chung YB. Seroprevalence of toxocariasis among healthy people with eosinophilia. Korean J Parasitol. 2008;46:29–32. doi: 10.3347/kjp.2008.46.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Won KY, Kruszon-Moran D, Schantz PM, Jones JL. National seroprevalence and risk factors for zoonotic Toxocara spp. infection. Am J Trop Med Hyg. 2008;79:552–557. [PubMed] [Google Scholar]
- 14.Companion Animal Parasite Council. Parasite prevalence maps. [accessed on 26 September 2014]. Available at http://www.capcvet.org/parasite-prevalence-maps/
- 15.Woodhall DM, Eberhard ML, Parise ME. Neglected parasitic infections in the United States: toxocariasis. Am J Trop Med Hyg. 2014;90:810–813. doi: 10.4269/ajtmh.13-0725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jin Y, Shen C, Huh S, Sohn WM, Choi MH, Hong ST. Serodiagnosis of toxocariasis by ELISA using crude antigen of Toxocara canis larvae. Korean J Parasitol. 2013;51:433–439. doi: 10.3347/kjp.2013.51.4.433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jacquier P, Gottstein B, Stingelin Y, Eckert J. Immunodiagnosis of toxocarosis in humans: evaluation of a new enzyme-linked immunosorbent assay kit. J Clin Microbiol. 1991;29:1831–1835. doi: 10.1128/jcm.29.9.1831-1835.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mohamad S, Azmi NC, Noordin R. Development and evaluation of a sensitive and specific assay for diagnosis of human toxocariasis by use of three recombinant antigens (TES-26, TES-30USM, and TES-120) J Clin Microbiol. 2009;47:1712–1717. doi: 10.1128/JCM.00001-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Magnaval JF, Fabre R, Maurières P, Charlet JP, de Larrard B. Application of the western blotting procedure for the immunodiagnosis of human toxocariasis. Parasitol Res. 1991;77:697–702. doi: 10.1007/BF00928685. [DOI] [PubMed] [Google Scholar]
- 20.Yamasaki H, Araki K, Lim PK, Zasmy N, Mak JW, Taib R, Aoki T. Development of a highly specific recombinant Toxocara canis second-stage larva excretory-secretory antigen for immunodiagnosis of human toxocariasis. J Clin Microbiol. 2000;38:1409–1413. doi: 10.1128/jcm.38.4.1409-1413.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Romasanta A, Romero JL, Arias M, Sánchez-Andrade R, López C, Suárez JL, Diaz P, Díez-Baños P, Morrondo P, Paz-Silva A. Diagnosis of parasitic zoonoses by immunoenzymatic assays--analysis of cross-reactivity among the excretory/secretory antigens of Fasciola hepatica, Toxocara canis, and Ascaris suum. Immunol Invest. 2003;32:131–142. doi: 10.1081/imm-120022974. [DOI] [PubMed] [Google Scholar]
- 22.Ishida MM, Rubinsky-Elefant G, Ferreira AW, Hoshino-Shimizu S, Vaz AJ. Helminth antigens (Taenia solium, Taenia crassiceps, Toxocara canis, Schistosoma mansoni and Echinococcus granulosus) and cross-reactivities in human infections and immunized animals. Acta Trop. 2003;89:73–84. doi: 10.1016/j.actatropica.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 23.Choi D, Lim JH, Choi DC, Paik SW, Kim SH, Huh S. Toxocariasis and ingestion of raw cow liver in patients with eosinophilia. Korean J Parasitol. 2008;46:139–143. doi: 10.3347/kjp.2008.46.3.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ryu BH, Park JS, Jung YJ, Kang SK, Lee SH, Choi SJ. Clinical and serological findings in patients with toxocariasis in the Pohang region: the features of toxocariasis in Pohang. Korean J Med. 2013;84:203–210. [Google Scholar]
- 25.Kang YR, Kim SA, Jeon K, Koh WJ, Suh GY, Chung MP, Kim H, Kwon OJ, Kang ES, Um SW. Toxocariasis as a cause of new pulmonary infiltrates. Int J Tuberc Lung Dis. 2013;17:412–417. doi: 10.5588/ijtld.12.0273. [DOI] [PubMed] [Google Scholar]
- 26.Zukiewicz M, Kaczmarski M, Topczewska M, Sidor K, Tomaszewska BM. Epidemiological and clinical picture of parasitic infections in the group of children and adolescents from north-east region of Poland. Wiad Parazytol. 2011;57:179–187. [PubMed] [Google Scholar]
- 27.Schoenardie ER, Scaini CJ, Brod CS, Pepe MS, Villela MM, McBride AJ, Borsuk S, Berne ME. Seroprevalence of Toxocara infection in children from southern Brazil. J Parasitol. 2013;99:537–539. doi: 10.1645/GE-3182. [DOI] [PubMed] [Google Scholar]
- 28.Lim JH. Foodborne eosinophilia due to visceral larva migrans: a disease abandoned. J Korean Med Sci. 2012;27:1–2. doi: 10.3346/jkms.2012.27.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kim YJ, Moon CH, Chang JH. Toxocariasis of the optic disc. J Neuroophthalmol. 2013;33:151–152. doi: 10.1097/WNO.0b013e3182893ff9. [DOI] [PubMed] [Google Scholar]
- 30.Yoon YS, Lee CH, Kang YA, Kwon SY, Yoon HI, Lee JH, Lee CT. Impact of toxocariasis in patients with unexplained patchy pulmonary infiltrate in Korea. J Korean Med Sci. 2009;24:40–45. doi: 10.3346/jkms.2009.24.1.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ahn SJ, Woo SJ, Jin Y, Chang YS, Kim TW, Ahn J, Heo JW, Yu HG, Chung H, Park KH, et al. Clinical features and course of ocular toxocariasis in adults. PLoS Negl Trop Dis. 2014;8:e2938. doi: 10.1371/journal.pntd.0002938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Manini MP, Marchioro AA, Colli CM, Nishi L, Falavigna-Guilherme AL. Association between contamination of public squares and seropositivity for Toxocara spp. in children. Vet Parasitol. 2012;188:48–52. doi: 10.1016/j.vetpar.2012.03.011. [DOI] [PubMed] [Google Scholar]
- 33.Noh Y, Hong ST, Yun JY, Park HK, Oh JH, Kim YE, Jeon BS. Meningitis by Toxocara canis after ingestion of raw ostrich liver. J Korean Med Sci. 2012;27:1105–1108. doi: 10.3346/jkms.2012.27.9.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Korea Center for Disease Control and Prevention. The 8th National Survey for Intestinal Parasites in Korea. Osong: Korea Center for Disease Control and Prevention; 2013. [Google Scholar]