Abstract
To assess the risk of acute kidney injury (AKI) attributable to aminoglycosides (AGs) in patients with severe sepsis or septic shock, we performed a retrospective cohort study in one medical intensive care unit (ICU) in France. Patients admitted for severe sepsis/septic shock between November 2008 and January 2010 were eligible. A propensity score for AG administration was built using day 1 demographic and clinical characteristics. Patients still on the ICU on day 3 were included. Patients with renal failure before day 3 or endocarditis were excluded. The time window for assessment of renal risk was day 3 to day 15, defined according to the RIFLE (risk, injury, failure, loss, and end-stage renal disease) classification. The AKI risk was assessed by means of a propensity-adjusted Cox proportional hazards regression analysis. Of 317 consecutive patients, 198 received AGs. The SAPS II (simplified acute physiology score II) score and nosocomial origin of infection favored the use of AGs, whereas a preexisting renal insufficiency and the neurological site of infection decreased the propensity for AG treatment. One hundred three patients with renal failure before day 3 were excluded. AGs were given once daily over 2.6 ± 1.1 days. AKI occurred in 16.3% of patients in a median time of 6 (interquartile range, 5 to 10) days. After adjustment to the clinical course and exposure to other nephrotoxic agents between day 1 and day 3, a propensity-adjusted Cox proportional hazards regression analysis showed no increased risk of AKI in patients receiving AGs (adjusted relative risk = 0.75 [0.32 to 1.76]). In conclusion, in critically septic patients presenting without early renal failure, aminoglycoside therapy for less than 3 days was not associated with an increased risk of AKI.
INTRODUCTION
Aminoglycosides (AGs) have bactericidal activity, which has been proven to be synergic with that of β-lactams. Although adding an AG to a standard antibiotic treatment did not translate into a reduction of mortality in Gram-negative-bacterial sepsis in subgroups of patients with globally moderate degrees of severity (1), a decrease in mortality was recently reported in a meta-analysis comparing a bitherapy to therapy with a β-lactam alone for patients with septic shock (2). In addition, AGs widen the spectrum of the antibiotic treatment, which should be advantageous in populations with an increased risk of resistant bacteria, such as intensive care unit (ICU) patients (3, 4). Empirical antibiotic treatments including AGs could be more appropriate in up to 15 to 20% of cases than a β-lactam alone (5, 6). In the ICU setting, modifications of the empirical antibiotic treatment or the addition of a new antibiotic occurs less frequently after bitherapy including an AG than after monotherapy (7).
Unfortunately, nephrotoxicity is an important potential limitation of AGs. There is a consensus that AG-related nephrotoxicity has decreased over the years due to better consideration of both reduced duration of treatment and once-daily administration (8). However, for ICU patients, higher doses have been recommended (9) because an increased volume of distribution (V) has been described (10). These aggressive doses could be responsible for a higher nephrotoxicity incidence. To date, a decrease in renal function has been observed in 5 to 14% of patients receiving AGs according to a wide definition (a >33% decrease in creatinine clearance [CLCR] with or without an increase of creatinine in plasma of ≥0.3 mg/dl) or a more restrictive definition (a >50% decrease in CLCR with or without a plasma creatinine increase of ≥0.5 mg/dl) (11–13). Both definitions may overestimate AG-associated renal toxicity, and the addition of criteria for tubular damage to the definitions based on plasma creatinine levels lowers the incidence of AG-related nephrotoxicity by 2- or 3-fold (13, 14). Conversely, a recent score designed to assess acute kidney injury (AKI) in severely septic patients (the risk, injury, failure, loss, and end-stage renal disease [RIFLE] score [15]) found 24% AKI in patients receiving AGs (16).
These controversial data suggest that AG-associated AKI might not be attributable solely to AGs because of the frequent confounding factors associated with AKI (17–19), such as septic shock per se, other nephrotoxic drugs (20), direct effect of bacterial toxins, and comorbidities, such as diabetes or altered baseline renal function. The aim of this study was to assess the AG-attributed AKI (the real AG nephrotoxicity) in patients with septic shock or severe sepsis.
MATERIALS AND METHODS
Design and ethical aspects.
This was a retrospective, observational study performed in a single 28-bed medical ICU from November 2008 to January 2010. The study protocol was approved by the Ethical Committee of the Société de Réanimation de Langue Française. Written consent was waived because of the observational nature of the study.
Inclusion and exclusion criteria and recruitment period.
The first part of the study consisted in including all patients admitted for or who developed septic shock or severe sepsis. Factors associated with the administration of AGs in septic shock or severe sepsis on day 1 were studied in this first set of patients.
Adult patients still in the ICU on day 3 were eligible for inclusion in the second part of the study, aiming at the determination of the nephrotoxicity risk associated with AG treatment (primary objective). Patients who had another indication for AGs, such as endocarditis, even complicated by severe sepsis, were excluded. Patients with renal failure before day 3 (either those under chronic renal replacement therapy or with acute tubular necrosis needing renal replacement therapy) were excluded since the distinction between aspecific renal failure and the specific AG-associated nephrotoxicity would be impossible with these patients. Patients with day 1 renal clearance of less than 56.25 ml/mn/1.73 m2 (I category of the RIFLE score [21]) followed by a severe decrease until day 3 without the need for renal replacement therapy (day 1/day 3 renal clearance > 1 plus day 3 renal clearance < 37.5 ml/mn/1.73 m2 before day 3) were also excluded, since this decrease also cannot be attributed to AGs. Studied parameters were collected from day 1 to the end of study, i.e., day 15, day of discharge, or day of death when occurring before day 15.
Objectives.
The primary objective of this study was the assessment of the AKI risk from day 3 to day 15 for patients having received AGs, in comparison with that for patients who did not receive AGs.
The secondary objectives were the identification of factors associated with the administration of AGs in severe sepsis or septic shock and the description of AG treatment in a population of ICU patients with severe sepsis or septic shock.
AKI was defined by an increase in the RIFLE score (21) from 1 (no risk plus creatinine clearance of >56.25 ml/min/1.73 m2 on day 3) to risk, injury, or failure from day 3 to day 15 or from a score of 2 (risk category [CLCR of >37.5 ml/min/1.73 m2 and <56.25 ml/min/1.73 m2]) to injury or failure).
Creatinine clearance was calculated on a daily basis using daily serum creatinine and the simple modified modification in diet in renal disease (MDRD), which is supposed to provide an estimation of the glomerular filtration rate (GFR) (15). For each patient included in the cohort, the charts were reviewed and the following data were recorded: demographic variables, SAPS II (simplified acute physiology score II) at the end of day 1, sequential organ failure assessment (SOFA) at the end of day 1 and day 3, type, cause, and severity of infection (severe sepsis and septic shock were defined according to the international guidelines [22]), nephrotoxic drugs and intravenous (i.v.) iodate contrast used at admission and all along the stay, other risk factors for nephrotoxicity (i.e., diabetes mellitus, presence of a single functional kidney, cirrhosis, kidney graft, preexisting renal failure, and rhabdomyolysis [defined by a creatine phosphokinase [CPK] level of >500 IU/liter]).
The duration of therapy, the dose, and serum concentrations of AGs were also recorded.
AG treatment.
In the ICU, AGs were always combined with β-lactams. Gentamicin (GEN) was recommended for treatment of infections due to Gram-positive or community-acquired Gram-negative bacteria, whereas amikacin (AMK) was recommended for treatment of nosocomially acquired Gram-negative bacteria infections. Tobramycin was not recommended. Doses and adjustments were checked daily by senior physicians with expertise in infectious diseases (A. Boyer and D. Gruson). The AG loading dose was calculated according to the total body weight (TBW) (20 mg/kg of body weight for AMK and 7 mg/kg for GEN) (10, 23). This regimen was defined according to an expected mean V of 0.3 to 0.4 liters/kg and a target peak concentration of 25 to 30 mg/liter for GEN and 40 to 50 mg/liter for AMK. If the observed peak was <20 mg/liter for GEN or <35 mg/liter for AMK, the daily dose was increased by 1.25- to 1.3-fold. Inversely if the peak was >35 mg/liter for GEN or >55 mg/liter for AMK, the daily dose was reduced in the same proportions. If the trough was >2.5 mg/liter for GEN and 5 mg/liter for AMK, the AG injection was delayed until the target trough was obtained. Peak was assessed 30 min after the end of AG infusion, and trough was assessed just before the following one, except for the last administration. A maximum of 5 days of AG therapy was recommended. The area under the concentration-time curve (AUC) was calculated from the peak and trough of the same interval by using a one-compartment pharmacokinetics (PK) model.
Statistical analysis.
The categorical variables were compared using the chi-squared test, and continuous variables were compared using the Student t test. In order to adjust for confounding factors of AKI that could have participated in the decision to administrate AGs, a propensity score of AG treatment was built with day 1 variables (the day of sepsis and antibiotic initiation). All the variables supposed to interact with the decision were considered. On day 3, once patients meeting prespecified criteria had been excluded, the time to AKI was examined by Kaplan-Meier and propensity-adjusted Cox proportional hazards regression analyses. The dependent binary variable was the AKI occurrence, and AG treatment was the primary independent variable. Most of the other independent variables having been already considered in the propensity score, only new nephrotoxic treatments (after day 1 and until either AKI or the end of the study, i.e., day 15 or before in the case of death or discharge) and the evolution of sepsis by delta SOFA (day 3 to day 1) were added to this model. Adjusted relative risks were computed for variables in the final model. A P value of 0.05 was considered significant. All calculations were computed using the software program SAS, version 9.3 (SAS corporation, Cary, NC).
RESULTS
Day 1 results and propensity score.
Between November 2008 and January 2010, 317 adult patients were consecutively included in the study. AGs had been administered to 198 patients. Septic shock was present in 227 (71.6%), whereas 90 (28.4%) had severe sepsis (Table 1). In Table 2, the variables constituting the propensity score for AG treatment are described. The most significant factors associated with AG administration were the SAPS II score and the nosocomial origin of infection, which both favored the decision to use AG treatment. In turn, a preexisting renal insufficiency and the neurological site of infection both decreased the propensity for AG treatment. The distribution of the propensity score for receiving AGs according to the true AG administration shows that a number of patients had an intermediate score (data not shown). Once adjusted for the propensity score, no difference between patients receiving or not receiving AGs was observed (data not shown).
TABLE 1.
Baseline characteristics of patients for propensity score elaboration
| Variablea | Value for patient group (nb)c |
P value | ||
|---|---|---|---|---|
| Total (317) | Aminoglycosides |
|||
| No (103) | Yes (214) | |||
| No. (%) male | 191 (60.2) | 61 (59.2) | 130 (60.7) | 0.80 |
| Mean age, yrs | 59 (17) | 59 (16) | 59 (18) | 0.67 |
| Mean SAPS II score | 59.2 (20.7) | 60.7 (20.2) | 56.0 (21.3) | 0.06 |
| No. (%) positive for nosocomial sepsis | 175 (55.2) | 44 (42.7) | 131 (61.2) | <10–1 |
| No. (%) with sepsis origin | 0.02 | |||
| Blood or catheter | 21 (6.6) | 6 (5.8) | 15 (7.0) | |
| Abdominal, urinary or gynecological tract | 34 (10.7) | 8 (7.8) | 26 (12.1) | |
| Lung, skin soft tissue, osteitis or arthritis | 232 (73.2) | 71 (68.9) | 161 (75.2) | |
| Neurological | 13 (4.1) | 8 (7.8) | 5 (2.3) | |
| No identified origin | 17 (5.4) | 10 (9.7) | 7 (3.3) | |
| No. (%) with sepsis severity | ||||
| Septic shock | 227 (71.6) | 71 (68.9) | 156 (72.9) | 0.47 |
| Severe sepsis | 90 (28.4) | 32 (31.1) | 58 (27.1) | |
| No. (%) with organ failure status | ||||
| None | 35 (11.0) | 10 (9.7) | 25 (11.7) | 0.60 |
| At least one | 282 (89.0) | 93 (90.3) | 189 (88.3) | |
| No. (%) with: | ||||
| Rhabdomyolysisd | 40 (12.62) | 17 (16.50) | 23 (10.75) | 0.16 |
| Renal graft or single functional kidney | 18 (5.7) | 6 (5.8) | 12 (5.6) | 0.94 |
| Diabetes mellitus | 55 (17.3) | 14 (13.6) | 41 (19.2) | 0.21 |
| Cirrhosis | 41 (12.93) | 11 (10.7) | 30 (14.0) | 0.40 |
| Preexisting renal failure | 30 (9.5) | 13 (12.6) | 17 (7.9) | 0.19 |
| No. (%) given specific nephrotoxic treatment(s) | ||||
| ACE inhibitors/ARB | 85 (27.0) | 26 (25.2) | 59 (27.8) | 0.63 |
| Diuretics | 97 (30.8) | 29 (28.2) | 68 (32.1) | 0.48 |
| High osmolar radiocontrast agent | 27 (8.6) | 10 (9.7) | 17 (8.0) | 0.62 |
| Hydroxyethyl starches | 15 (4.8) | 5 (4.8) | 10 (4.7) | 0.96 |
| NSAIDs | 18 (5.7) | 4 (3.9) | 14 (6.6) | 0.31 |
| Antimicrobial agente | 32 (10.2) | 10 (9.7) | 22 (10.4) | 0.85 |
| Immunosuppressive treatmentf | 19 (6.0) | 7 (6.8) | 12 (5.7) | 0.69 |
| No. (%) with nephrotoxic treatment status | ||||
| No | 128 (40.6) | 48 (46.6) | 80 (37.7) | 0.13 |
| At least one | 187 (59.4) | 55 (53.4) | 132 (62.3) | |
SAPS, simplified acute physiology score; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blockers; NSAIDs, nonsteroidal anti-inflammatory drugs.
n, no. of patients in each group.
Qualitative variables are represented with number (%), quantitative variables with mean (SD).
Rhabdomyolysis is defined by a CPK value of >500 IU/liter.
Vancomycin, amphotericin B, acyclovir, foscavir.
Calcineurin inhibitors.
TABLE 2.
Multivariate analysis of factors associated with aminoglycoside treatment
| Variablea | Value for all patientsb | OR (95% CI) |
|---|---|---|
| No. (%) in sex group | ||
| Male | 191 (60.2) | 1.00 |
| Female | 126 (39.8) | 0.92 (0.52–0.61) |
| Age (yrs) | 59 (17) | 0.99 (0.97–1.01) |
| SAPS II | 59.2 (20.7) | 1.02 (1.00–1.04) |
| No. (%) with: | ||
| Nosocomial sepsis | ||
| No | 142 (44.8) | 1.00 |
| Yes | 175 (55.2) | 2.10 (1.22–3.63) |
| Sepsis origin | ||
| Blood or catheter | 21 (6.6) | 1.00 |
| Abdominal, urinary or gynecological tract | 34 (10.7) | 1.25 (0.32–4.91) |
| Lung, skin soft tissue, osteitis or arthritis | 232 (73.2) | 0.93 (0.31–2.80) |
| Neurological | 13 (4.1) | 0.14 (0.03–0.76) |
| No identified origin | 17 (5.4) | 0.23 (0.05–1.01) |
| Sepsis severity | ||
| Septic shock | 227 (71.6) | 1.00 |
| Severe sepsis | 90 (28.4) | 0.87 (0.43–1.74) |
| No. of organ failures | ||
| No failure | 35 (11.0) | 1.00 |
| At least one | 282 (89.0) | 0.46 (0.16–1.30) |
| Rhabdomyolysisc | ||
| No | 277 (87.4) | 1.00 |
| Yes | 40 (12.6) | 0.61 (0.28–1.30) |
| Renal graft or single functional kidney | ||
| No | 299 (94.3) | 1.00 |
| Yes | 18 (5.7) | 1.53 (0.43–5.41) |
| Diabetes mellitus | ||
| No | 262 (82.6) | 1.00 |
| Yes | 55 (17.3) | 1.69 (0.78–3.64) |
| Cirrhosis | ||
| No | 276 (87.1) | 1.00 |
| Yes | 41 (12.9) | 1.53 (0.66–3.58) |
| Preexisting renal failure | ||
| No | 287 (90.5) | 1.00 |
| Yes | 30 (9.5) | 0.37 (0.14–1.00) |
| Nephrotoxic treatments | ||
| ACE inhibitors/ARB | ||
| No | 230 (73.0) | 1.00 |
| Yes | 85 (27.0) | 0.81 (0.38–1.76) |
| Diuretics | ||
| No | 218 (69.2) | 1.00 |
| Yes | 97 (30.8) | 0.70 (0.33–1.53) |
| High osmolar radiocontrast agent | ||
| No | 288 (91.4) | 1.00 |
| Yes | 27 (8.6) | 0.63 (0.23–1.73) |
| Hydroxyethyl starches | ||
| No | 300 (95.2) | 1.00 |
| Yes | 15 (4.8) | 0.77 (0.21–2.81) |
| NSAIDs | ||
| No | 297 (94.3) | 1.00 |
| Yes | 18 (5.7) | 1.91 (0.47–7.70) |
| Antimicrobial agentd | ||
| No | 283 (89.8) | 1.00 |
| Yes | 32 (10.2) | 0.97 (0.37–2.56) |
| Immunosuppressive treatmente | ||
| No | 296 (94.0) | 1.00 |
| Yes | 19 (6.0) | 0.31 (0.09–1.03) |
| Use of nephrotoxic treatments | ||
| No | 128 (40.6) | 1.00 |
| At least one | 187 (59.4) | 1.99 (0.80–4.95) |
SAPS, simplified acute physiology score; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blockers; NSAIDs, nonsteroidal anti-inflammatory drugs.
Qualitative variables are represented with no. (%), quantitative variables with mean (SD).
Rhabdomyolysis is defined by a CPK level of >500 IU/liter.
Vancomycin, amphotericin B, acyclovir, foscavir.
Calcineurin inhibitors.
Day 3 results.
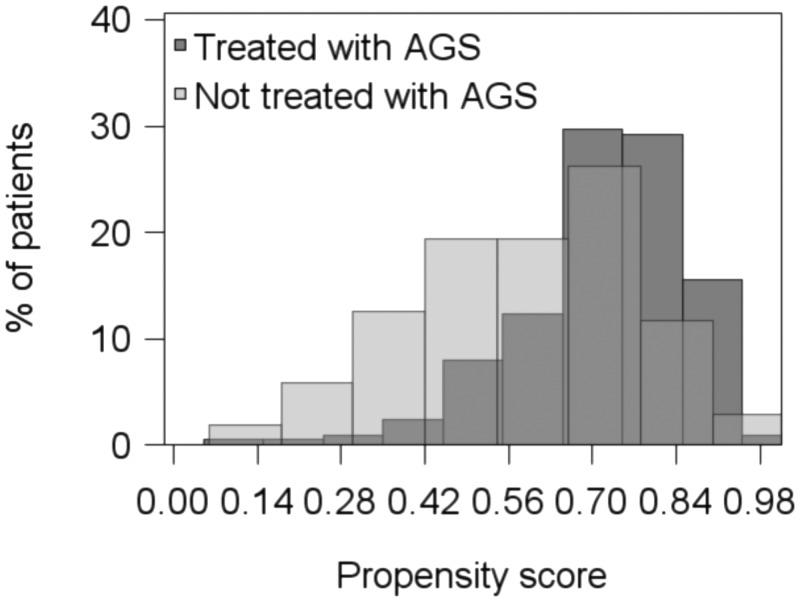
Between day 1 and day 3, 103 patients among 317 were excluded, among whom 72 were excluded for either initial severe AKI (n = 58) or renal replacement therapy (n = 14) (AGs were administered in two-thirds of these patients [n = 48]). The remaining population of 214 patients had mean lengths of ICU and hospital stay of 16 ± 16 and 38 ± 60 days, respectively. Twenty-one (10%) died before day 15. Among these 214 patients, 150 received AGs. The mean duration of AG therapy was 2.6 ± 1.1 days. Amikacin was prescribed for 74% and gentamicin for 26%. Results of drug monitoring, including trough and peak determinations, are shown in Table 3. The areas under the curve (AUCs) were 200 ± 114 mg · h/liter for gentamicin and 537 ± 590 mg · h/liter for amikacin. Figure 1 represents the distribution of the propensity score, according to whether AGs were given or not, in the 214 patients. AKI occurred in 35 patients (16.3%), with a median delay of 6 (interquartile range, 5 to 10) days. The results of the multivariate analysis of risk factors for acute renal failure are shown in Table 3. The SOFA score improved from day 1 to day 3 for 154 patients (72%), whereas it decreased for 60 (28%). At least one additional nephrotoxic agent had been administered for 133 patients (62%) in the period from day 1 to the index date. Variables associated with the occurrence of AKI risk in the multivariate Cox model are shown in Table 4. After adjustment to delta SOFA, exposure to additional nephrotoxic agents (day 1 to the index date), and propensity score for AG treatment, the administration of AGs was not associated with a significant risk of AKI (Table 4).
TABLE 3.
Drug monitoring parameters of aminoglycoside administrationa
| Drug | No. (%) of patients given drug | Dose (mg/kg) | Duration (days) | Cpeak (mg/liter)b | % of target attainment (value achieved [mg/liter]) for: |
|
|---|---|---|---|---|---|---|
| Cpeak | Ctrough | |||||
| Gentamicin | 39 (26) | 6.0 ± 1.6 | 2.9 ± 1.1 | 24.9 ± 11.4 | 47 (>25) | 85.4 (<2.5) |
| Amikacin | 111 (74) | 18.4 ± 5.5 | 2.5 ± 1.0 | 44.3 ± 16.0 | 66 (>40) | 73.3 (<5) |
n = 150 patients.
For gentamicin, n = 101; for amikacin, n = 259.
FIG 1.
Propensity scores for aminoglycoside treatment at baseline for the studied patients.
TABLE 4.
Multivariate risk for acute kidney injury (Cox proportional hazards ratio)
| Factor | HRa (95% CI) | P value |
|---|---|---|
| Aminoglycoside (yes vs no) | 0.75 (0.32–1.76) | 0.51 |
| Decrease in SOFA score between day 1 and day 3 (yes vs no) | 1.02 (0.48–2.15) | 0.96 |
| Nephrotoxic treatment after day 1 (yes vs no) | 0.75 (0.37–1.51) | 0.43 |
The relative risk (hazards ratio [HR]) has been adjusted based on the propensity score.
Ninety-one patients (28.7%) died before day 28 among the entire day 1 population, and the variables independently associated with the occurrence of death in the multivariate Cox model were age and gender, whereas AG treatment was not significantly associated with day 28 mortality (Table 5).
TABLE 5.
Risk for day 28 mortality
| Factor | RRa (95% CI) | P value |
|---|---|---|
| Aminoglycoside (yes vs no) | 0.78 (0.47–1.30) | 0.34 |
| Age | 1.02 (1.01–1.04) | <0.01 |
| Sex (female vs male) | 0.52 (0.33–0.81) | <0.01 |
| Day 1 SOFA | 1.05 (0.98–1.13) | 0.16 |
| Severe sepsis vs septic shock | 1.05 (0.52–2.09) | 0.90 |
The relative risk (RR) has been adjusted based on the propensity score and the SAPS II score.
DISCUSSION
The main finding of our study is that aminoglycosides are not associated with an increase in AKI in patients admitted to the ICU for severe sepsis or septic shock without early renal failure after adjustment for the clinical evolution of patients in the first three ICU days (delta SOFA day 3 to day 1 and exposure to nephrotoxic agents) and, most importantly, for the propensity score for receiving AGs.
The propensity score was based on the risk factors for AG nephrotoxicity listed in two meta-analyses (24, 25), to which we added specific characteristics of sepsis. The patient severity level (SAPS II score, odds ratio [OR] = 1.02 [1.00 to 1.04]) was a major determinant in the decision to use AGs, reflecting the prescribers' confidence in the bactericidal and synergistic action of AGs (26). Another factor positively influencing AG treatment was the nosocomial origin of infection (OR = 2.10 [1.22 to 3.63]). This could be expected, since hospital infections are commonly caused by multiresistant bacteria (27). Conversely, the neurological origin of sepsis discouraged the initiation of AGs (OR = 0.14 [0.03 to 0.76]), which is in accordance with their pharmacokinetic characteristics, no more than 10% of the total intravenous dose penetrating cerebrospinal fluid (28). Patients with preexisting renal failure (OR = 0.37 [0.14 to 1.00]) were less prone to be treated by AGs, probably due to a benefit-risk approach in this subset of patients.
The overall incidence of AKI in our study was 16% (35/214), which should be considered low compared to the data of a recent Italian study reporting a 40% incidence of acute renal failure in 279 septic ICU patients (29). This might be explained by our decision to exclude patients with AKI occurring before day 3. Adding these excluded patients (n = 72) would have led to a comparable overall risk of AKI (37%; 107/286). Many studies have attempted to determine the specific renal risk of AGs, and the risk prevalence of AKI is highly variable. The vast majority of published studies suffer from the absence of a combination of validated acute renal failure criteria and sufficient severity of cases to allow the extrapolation of results to critically ill patients. The Acute Dialysis Quality Initiative group developed the RIFLE classification, accounting for a broad range of acute impairment of kidney function through the consensus of experts (15). It has been validated with ICU patients and can detect AKI with high sensitivity and specificity (15, 21). A recent retrospective study using these criteria has shown a 24% AG nephrotoxicity, but only 12% of patients experiencing AKI had severe sepsis (16). A strength of our study was to use a validated AKI classification with an exclusively ICU population. We also carefully avoided other methodological flaws. Patients with kidney injury, failure, or loss of function assessed by RIFLE scoring before day 3 were excluded in order to increase the specificity of AG-associated acute renal failure. Indeed, AG-associated nephrotoxicity is known to occur at the end of the AG treatment or later, i.e., from day 3 to day 15. This time window was chosen based on a previously reported mean delay between AG treatment and nephrotoxicity of 8.8 ± 3.4 days (30).
Moreover, AKI in a severe sepsis or septic shock population is a complex issue. While septic shock remains the leading cause of AKI in this population (31), the pressure on renal function is increased by many other factors, such as nephrotoxic drugs (20), a direct effect of bacterial toxins, vascular nephritis, etc. (32). We performed a quasirandomized study in which all variables supposed to interact with AKI occurrence were considered in a propensity-adjusted Cox proportional hazards regression analysis. It allowed a better assessment of the causal link between the administration of AGs and AKI occurrence. It would therefore make sense to abandon the assessment of AG nephrotoxicity by the rate of acute renal failure occurring after the administration of AGs and to prefer a better methodological assessment of AG-associated AKI (19).
No AG-associated AKI risk was found in this study (adjusted OR, 0.75; 95% confidence interval [CI],0.32 to 1.76), and there might even be a slight trend toward a reduced renal risk in AG patients, considering that the study population did not include patients with early acute renal failure. This might be related to both pharmacological and physiological mechanisms. Recent pharmacological concepts of AGs administration, such as the once-daily dose schedule for AG administration, which result in a decrease on AG-associated AKI (9) have been respected in the present study, as shown by the use of once-daily doses of gentamicin and amikacin during a short course achieving a correct rate of target obtention (Table 3). A physiological mechanism has also been suggested by several authors (33–35). Lipcsey et al. compared four groups of pigs (endotoxinemia plus tobramycin, endotoxinemia plus saline, saline plus tobramycin, and saline alone) and suggested that sepsis-induced hypoperfusion was predominant over specific AG toxicity for AKI occurrence (34). More recently, Langenberg et al. compared 3 groups of sheep (Escherichia coli infusion, E. coli infusion followed by gentamicin IV, and a control group) and showed a lower rate of NO synthase and hypoxia-inducible factor in the gentamicin group than in the septic group. This provides evidence that AGs might stop endothelial and cellular signals at the origin of sepsis-related AKI (35).
There are some limitations to this study. This was a retrospective cohort study conducted at a single ICU, possibly limiting its extrapolation. In particular, even if the AG administration was consistent with the standards of care at the time of the study (100% once-daily dose, 61% of peak concentration of drug in serum (Cpeak) target attainment, and short duration) (36), recent recommendations have emphasized the use of higher doses of amikacin (25 to 30 mg/kg) potentially changing the AG-associated AKI risk (37). The general characteristics of the population are also representative of medical ICU septic patients (SAPS II score of 59 ± 20, 72% septic shock, and mean duration of ICU stay of 14 ± 16 days). In critically ill patients, assessing the glomerular filtration rate by means of serum creatinine levels can be questioned. Urinary CLCR, though more specific, bears the same limitations (38). We used the MDRD formula for basal estimation of the glomerular filtration rate, following the recommendation for RIFLE classification. However, recently, the KDIGO (Kidney Disease: Improving Global Outcomes) initiative proposed a modified RIFLE classification that should be used in the future (39). However, to date, RIFLE remains the most validated classification, with more than half a million studied patients. We excluded from the analysis patients who had impaired renal function that worsened between day 1 and day 3, since we expect aminoglycoside nephrotoxicity to be manifest some days after starting. However, we performed a sensitivity analysis in reincorporating patients excluded for initial renal failure (defined by creatinine clearance of <37.5) (n = 58) and patients needing immediate renal replacement therapy (n = 14). Among this population (n = 286), 107 (37.4%) patients developed AKI. The AKI adjusted relative risk of aminoglycoside treatment (yes versus no) given by the Cox model was 0.95 (95% CI, 0.59 to 1.47; P = 0.76). Finally, the finding of no increased risk might be attributed to a lack of power despite the relatively large number of patients included. However, the probability that the trend in the reduction of risk obtained in this study could be reversed to an increase is low, especially with the statistical methods used. With traditional multivariable regression techniques, models become unstable when the sample size is small and the number of covariates included in the model is large relative to the number of outcome events. Adjusting on propensity scores is a means of preventing overloading subsequent regression models, especially when studying a rare outcome (40). In this case, though the choice of treatment was not randomized and it might be surmised that these drugs would be used in patients at lower risk of renal failure, the use of a propensity score built with all known risk factors for renal failure should protect against such confounding. Of course, the lack of association we found does not mean that AGs are no longer nephrotoxic, just that the choice of patients and the way the drugs were given was judicious. Further studies in similar patients in other settings would be useful to confirm these results, ideally under randomized controlled circumstances, which unfortunately are hardly possible in this context.
In conclusion, with modern modalities of administration, including a high dose given once daily and a short duration of treatment, AGs did not appear to increase the AKI risk in ICU patients treated for severe sepsis or septic shock without early acute renal failure.
ACKNOWLEDGMENTS
We thank the patients and the clinical and clerical teams involved in this study.
We have no relevant conflict of interest in the field of aminoglycoside treatment.
Footnotes
Published ahead of print 6 October 2014
REFERENCES
- 1.Paul M, Benuri-Silbiger I, Soares-Weiser K, Leibovici L. 2004. Beta lactam monotherapy versus beta lactam-aminoglycoside combination therapy for sepsis in immunocompetent patients: systematic review and meta-analysis of randomised trials. BMJ 328:668. 10.1136/bmj.38028.520995.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kumar A, Safdar N, Kethireddy S, Chateau D. 2010. A survival benefit of combination antibiotic therapy for serious infections associated with sepsis and septic shock is contingent only on the risk of death: a meta-analytic/meta-regression study. Crit. Care Med. 38:1651–1664. 10.1097/CCM.0b013e3181e96b91. [DOI] [PubMed] [Google Scholar]
- 3.Durante-Mangoni E, Grammatikos A, Utili R, Falagas ME. 2009. Do we still need the aminoglycosides? Int. J. Antimicrob. Agents 33:201–205. 10.1016/j.ijantimicag.2008.09.001. [DOI] [PubMed] [Google Scholar]
- 4.Leibovici L, Vidal L, Paul M. 2009. Aminoglycoside drugs in clinical practice: an evidence-based approach. J. Antimicrob. Chemother. 63:246–251. 10.1093/jac/dkn469. [DOI] [PubMed] [Google Scholar]
- 5.Beardsley JR, Williamson JC, Johnson JW, Ohl CA, Karchmer TB, Bowton DL. 2006. Using local microbiologic data to develop institution-specific guidelines for the treatment of hospital-acquired pneumonia. Chest 130:787–793. 10.1378/chest.130.3.787. [DOI] [PubMed] [Google Scholar]
- 6.Bhat S, Fujitani S, Potoski BA, Capitano B, Linden PK, Shutt K, Paterson DL. 2007. Pseudomonas aeruginosa infections in the intensive care unit: can the adequacy of empirical beta-lactam antibiotic therapy be improved? Int. J. Antimicrob. Agents 30:458–462. 10.1016/j.ijantimicag.2007.05.022. [DOI] [PubMed] [Google Scholar]
- 7.Marcus R, Paul M, Elphick H, Leibovici L. 2011. Clinical implications of beta-lactam-aminoglycoside synergism: systematic review of randomised trials. Int. J. Antimicrob. Agents 37:491–503. 10.1016/j.ijantimicag.2010.11.029. [DOI] [PubMed] [Google Scholar]
- 8.Slaughter RL, Cappelletty DM. 1998. Economic impact of aminoglycoside toxicity and its prevention through therapeutic drug monitoring. PharmacoEconomics 14:385–394. 10.2165/00019053-199814040-00005. [DOI] [PubMed] [Google Scholar]
- 9.Drusano GL, Ambrose PG, Bhavnani SM, Bertino JS, Nafziger AN, Louie A. 2007. Back to the future: using aminoglycosides again and how to dose them optimally. Clin. Infect. Dis. 45:753–760. 10.1086/520991. [DOI] [PubMed] [Google Scholar]
- 10.Rea RS, Capitano B, Bies R, Bigos KL, Smith R, Lee H. 2008. Suboptimal aminoglycoside dosing in critically ill patients. Ther. Drug Monit. 30:674–681. 10.1097/FTD.0b013e31818b6b2f. [DOI] [PubMed] [Google Scholar]
- 11.Buijk SE, Mouton JW, Gyssens IC, Verbrugh HA, Bruining HA. 2002. Experience with a once-daily dosing program of aminoglycosides in critically ill patients. Intensive Care Med. 28:936–942. 10.1007/s00134-002-1313-7. [DOI] [PubMed] [Google Scholar]
- 12.Fayed DF, Dahmash NS, al-Zeer AH, Shibl AM, Huraib SO, Abu-Aisha H. 1996. Efficacy and safety of once-daily amikacin in combination with ceftazidime in critically ill adults with severe gram-negative infections. J. Chemother. 8:457–464. 10.1179/joc.1996.8.6.457. [DOI] [PubMed] [Google Scholar]
- 13.Schentag JJ, Plaut ME, Cerra FB. 1981. Comparative nephrotoxicity of gentamicin and tobramycin: pharmacokinetic and clinical studies in 201 patients. Antimicrob. Agents Chemother. 19:859–866. 10.1128/AAC.19.5.859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.French MA, Cerra FB, Plaut ME, Schentag JJ. 1981. Amikacin and gentamicin accumulation pharmacokinetics and nephrotoxicity in critically ill patients. Antimicrob. Agents Chemother. 19:147–152. 10.1128/AAC.19.1.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P. 2004. Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit. Care 8:R204–R212. 10.1186/cc2872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Selby NM, Shaw S, Woodier N, Fluck RJ, Kolhe NV. 2009. Gentamicin-associated acute kidney injury. QJM 102:873–880. 10.1093/qjmed/hcp143. [DOI] [PubMed] [Google Scholar]
- 17.Li SC, Ioannides-Demos LL, Spicer WJ, Berbatis C, Spelman DW, Tong N, McLean AJ. 1989. Prospective audit of aminoglycoside usage in a general hospital with assessments of clinical processes and adverse clinical outcomes. Med. J. Aust. 151:224–232. [DOI] [PubMed] [Google Scholar]
- 18.Shrimpton SB, Milmoe M, Wilson AP, Felmingham D, Drayan S, Barrass C, Gruneberg RN, Ridgway GL. 1993. Audit of prescription and assay of aminoglycosides in a UK teaching hospital. J. Antimicrob. Chemother. 31:599–606. 10.1093/jac/31.4.599. [DOI] [PubMed] [Google Scholar]
- 19.Boyer A, Gruson D, Bouchet S, Clouzeau B, Hoang-Nam B, Vargas F, Gilles H, Molimard M, Rogues AM, Moore N. 2013. Aminoglycosides in septic shock: an overview, with specific consideration given to their nephrotoxic risk. Drug Saf. 36:217–230. 10.1007/s40264-013-0031-0. [DOI] [PubMed] [Google Scholar]
- 20.Taber SS, Mueller BA. 2006. Drug-associated renal dysfunction. Crit. Care Clin. 22:357–374, viii. 10.1016/j.ccc.2006.02.003. [DOI] [PubMed] [Google Scholar]
- 21.Ostermann M., Chang RW. 2007. Acute kidney injury in the intensive care unit according to RIFLE. Crit. Care Med. 35:1837–1843, 1852. 10.1097/01.CCM.0000277041.13090.0A. [DOI] [PubMed] [Google Scholar]
- 22.Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, Sprung CL, Douglas IS, Jaeschke R, Osborn TM, Nunnally ME, Townsend SR, Reinhart K, Kleinpell RM, Angus DC, Deutschman CS, Machado FR, Rubenfeld GD, Webb SA, Beale RJ, Vincent JL, Moreno R. 2013. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit. Care Med. 41:580–637. 10.1097/CCM.0b013e31827e83af. [DOI] [PubMed] [Google Scholar]
- 23.American Thoracic Society. 2005. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am. J. Respir. Crit. Care Med. 171:388–416. 10.1164/rccm.200405-644ST. [DOI] [PubMed] [Google Scholar]
- 24.Pannu N, Nadim MK. 2008. An overview of drug-induced acute kidney injury. Critical care medicine. 36:S216–223. 10.1164/rccm.200405-644ST. [DOI] [PubMed] [Google Scholar]
- 25.Moore RD, Smith CR, Lipsky JJ, Mellits ED, Lietman PS. 1984. Risk factors for nephrotoxicity in patients treated with aminoglycosides. Ann. Intern. Med. 100:352–357. 10.7326/0003-4819-100-3-352. [DOI] [PubMed] [Google Scholar]
- 26.Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, Suppes R, Feinstein D, Zanotti S, Taiberg L, Gurka D, Kumar A, Cheang M. 2006. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit. Care Med. 34:1589–1596. 10.1097/01.CCM.0000217961.75225.E9. [DOI] [PubMed] [Google Scholar]
- 27.da Silva Winter J, Dos Santos RP, de Azambuja AZ, Cechinel AB, Goldani LZ. 2013. Microbiologic isolates and risk factors associated with antimicrobial resistance in patients admitted to the intensive care unit in a tertiary care hospital. Am. J. Infect. Control 41:846–848. 10.1016/j.ajic.2012.11.010. [DOI] [PubMed] [Google Scholar]
- 28.Fulnecky EJ, Wright D, Scheld WM, Kanawati L, Shoham S. 2005. Amikacin and colistin for treatment of Acinetobacter baumannii meningitis. J. Infect. 51:e249–e251. 10.1016/j.jinf.2005.04.003. [DOI] [PubMed] [Google Scholar]
- 29.Rocco M, Montini L, Alessandri E, Venditti M, Laderchi A, De Gennaro P, Raponi G, Vitale M, Pietropaoli P, Antonelli M. 2013. Risk factors for acute kidney injury in critically ill patients receiving high intravenous doses of colistin methanesulfonate and/or other nephrotoxic antibiotics: a retrospective cohort study. Crit. Care 17:R174. 10.1186/cc12853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rybak MJ, Abate BJ, Kang SL, Ruffing MJ, Lerner SA, Drusano GL. 1999. Prospective evaluation of the effect of an aminoglycoside dosing regimen on rates of observed nephrotoxicity and ototoxicity. Antimicrob. Agents Chemother. 43:1549–1555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ricci Z, Polito A, Polito A, Ronco C. 2011. The implications and management of septic acute kidney injury. Nat. Rev. Nephrol. 7:218–225. 10.1038/nrneph.2011.15. [DOI] [PubMed] [Google Scholar]
- 32.Philipponnet C, Guerin C, Canet E, Robert R, Mariat C, Dijoud F, Azoulay E, Souweine B, Heng AE. 2013. Kidney biopsy in the critically ill patient, results of a multicentre retrospective case series. Minerva Anestesiol. 79:53–61. [PubMed] [Google Scholar]
- 33.Zager RA. 1992. Endotoxemia, renal hypoperfusion, and fever: interactive risk factors for aminoglycoside and sepsis-associated acute renal failure. Am. J. Kidney Dis. 20:223–230. 10.1016/S0272-6386(12)80694-9. [DOI] [PubMed] [Google Scholar]
- 34.Lipcsey M, Carlsson M, Larsson A, Algotsson L, Eriksson M, Lukinius A, Sjolin J. 2009. Effect of a single dose of tobramycin on systemic inflammatory response-induced acute kidney injury in a 6-hour porcine model. Crit. Care Med. 37:2782–2790. 10.1097/CCM.0b013e3181a988f8. [DOI] [PubMed] [Google Scholar]
- 35.Langenberg C, Gobe G, Hood S, May CN, Bellomo R. 2014. Renal histopathology during experimental septic acute kidney injury and recovery. Crit. Care Med. 42:e58–e67. 10.1097/CCM.0b013e3182a639da. [DOI] [PubMed] [Google Scholar]
- 36.Drusano GL, Louie A. 2011. Optimization of aminoglycoside therapy. Antimicrob. Agents Chemother. 55:2528–2531. 10.1128/AAC.01314-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Matthaiou DK, De Waele J, Dimopoulos G. 2014. What is new in the use of aminoglycosides in critically ill patients? Intensive Care Med. 40:1553–1555. 10.1007/s00134-014-3376-7. [DOI] [PubMed] [Google Scholar]
- 38.Bragadottir G, Redfors B, Ricksten SE. 2013. Assessing glomerular filtration rate (GFR) in critically ill patients with acute kidney injury—true GFR versus urinary creatinine clearance and estimating equations. Crit. Care 17:R108. 10.1186/cc12777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kellum JA, Lameire N. 2013. Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (part 1). Crit. Care 17:204. 10.1186/cc11454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Braitman LE, Rosenbaum PR. 2002. Rare outcomes, common treatments: analytic strategies using propensity scores. Ann. Intern. Med. 137:693–695. 10.7326/0003-4819-137-8-200210150-00015. [DOI] [PubMed] [Google Scholar]