Abstract
While significant advances in radiotherapy have increased its effectiveness in many cancer settings, general strategies to widen the therapeutic window between normal tissue toxicity and malignant tumor destruction would still offer great value. CD47 blockade has been found to confer radioprotection to normal tissues while enhancing tumor radiosensitivity. Here we report that CD47 blockade directly enhances tumor immunosurveillance by CD8+ T cells. Combining CD47 blockade with irradiation did not affect fibrosarcoma growth in T cell-deficient mice, whereas adoptive transfer of tumor-specific CD8+ T cells restored combinatorial efficacy. Further, ablation of CD8+ T cells abolished radiotherapeutic response in immunocompetent syngeneic hosts. CD47 blockade in either target cells or effector cells was sufficient to enhance antigen-dependent CD8+ CTL-mediated tumor cell killing in vitro. In CD47-deficient syngeneic hosts, engrafted B16 melanomas were 50% more sensitive to irradiation, establishing that CD47 expression in the microenvironment was sufficient to limit tumor radiosensitivity. Mechanistic investigations revealed increased tumor infiltration by cytotoxic CD8+ T cells in a CD47-deficient microenvironment, with an associated increase in T cell-dependent intratumoral expression of granzyme B. Correspondingly, an inverse correlation between CD8+ T cell infiltration and CD47 expression was observed in human melanomas. Our findings establish that blocking CD47 in the context of radiotherapy enhances antitumor immunity by directly stimulating CD8+ cytotoxic T cells, with the potential to increase curative responses.
Introduction
CD47 is a widely expressed counter-receptor for the inhibitory phagocyte receptor SIRPα. Blocking this interaction enhances macrophage-mediated clearance of tumor cells (1–3). Correspondingly, elevated CD47 expression on cancer cells is proposed to suppress anti-tumor innate immunity (4, 5). However, CD47 also functions as a signaling receptor that determines cell fate through the regulation of several death/survival pathways, mainly through its interactions with the matricellular protein thrombospondin-1 (TSP1). Binding of the C-terminal signature domain of TSP1 to CD47 causes a profound inhibition of the nitric oxide/cGMP signaling in vascular cells and T cells (6–8). In the immune system binding of TSP1 to CD47 inhibits T cell activation (9–11), in part by inhibiting the autocrine activating function of hydrogen sulfide signaling in T cells (12). TSP1 is the relevant CD47 ligand in T cells because these cells do not express detectable levels of SIRPα (13, 14). Signaling through CD47 also regulates T cell differentiation and adhesion as well as NK and dendritic cell functions that regulate adaptive immunity (15–22). Thus, we propose that treatment of tumor-bearing animals with CD47 blocking antibodies, which are known to inhibit both SIRPα and TSP1 binding to CD47, could directly modulate adaptive as well as innate anti-tumor immunity. Indeed, cytotoxic T cells were recently implicated in the anti-tumor effects of a CD47-blocking antibody, but this outcome was attributed to an indirect effect of inhibiting SIRPα engagement on macrophages (23).
We previously demonstrated that blockade of CD47 enhances the radiation-induced delay in tumor growth in two syngeneic mouse models (24). The reduction of tumor burden when CD47 blockade was combined with ionizing radiation (IR) was associated with radioprotection of the cells in the tumor microenvironment, increased oxygenation of the tumor by increasing blood flow, and enhanced migration of cytotoxic lymphocytes. More recently we have demonstrated that blocking CD47 signaling provides radioprotection in T cells and endothelial cells through an up-regulation of pro-survival autophagy (25). Thus, the increased survival of these cells in the irradiated tumor stroma could enhance anti-tumor immunity. IR activates the immune system, and its role in the abscopal effect of radiation therapy is primarily attributed to activation of T-cell anti-tumor immunity (26–28). These results suggested that CD47 expression by stromal cells may play a significant role in modulating T cell anti-tumor immunity activated as a consequence of damage to tumor cells caused by IR.
To date, the ablation of tumor growth by CD47 blockade has been attributed to restoration of macrophage-mediated immune surveillance by reducing the ability of CD47 on tumor cells to engage SIRPα on tumor-associated macrophages. In contrast, here we show that the reduction in tumor growth by CD47 blockade is dependent on an intact adaptive immune system, specifically CD8+ cytotoxic T cells. Moreover, blockade or loss of CD47 signaling in effector T cells is sufficient to directly increase CD8+ T cell killing of irradiated cancer cells and to reduce tumor burden in vivo.
Materials and Methods
Model of T-Cell Adoptive Transfer
Athymic nu/nu mice in a BALB/c background (NCI-Frederick) were injected in the hind limbs with 1×106 15-12RM fibrosarcoma cells expressing HIV gp160 (29). Treatment was initiated once tumors reached an average 100 mm3 volume. Tumor irradiation was accomplished by securing each animal in a Lucite jig fitted with lead shielding that protected the body from radiation while allowing exposure of the tumor-bearing leg in a single field of uniform size. A Therapax DXT300 X-ray irradiator microprocessor-controlled orthovoltage system (Pantak, Inc., East Haven, CT) using 2.0 mm A1 filtration (300 KV/10 mA) at a dose rate of 2.53 Gy/min was used as the X-ray source. Irradiated tumors received one 10 Gy dose. For the adoptive transfer of RT-1 T cells, we immunized RT-1 TCR transgenic mice with a recombinant vaccinia virus expressing gp160, vPE16 (one million pfu). More than 8 weeks after the immunization, CD8+ T cells were purified from spleens using a CD8 T cell isolation kit from Miltenyi. Cell suspensions were injected intravenously, and a group of mice were concurrently treated with saline or 750 µL of 10 µM CD47 translation-blocking antisense morpholino (24). Mice were randomized, and some groups of animals were locally irradiated with 10 Gy as previously described (24). Tumor size was measured every other day for 30 days after treatment or until humane endpoints were reached. All experimental procedures were carried out under approved protocols following the guidelines of the National Cancer Institute Animal Care and Use Committee.
15-12 RM Syngeneic Fibrosarcoma Model
WT BALB/c mice were injected with 1×106 15-12RM cells as shown previously (29). Once tumors reached an average of 100 mm3 a group of mice was treated with saline or CD47 morpholino. Forty eight hours later tumors in some groups of mice were locally irradiated at 10 Gy. CD8 depletion was performed as shown previously (30). Briefly, mice were treated with 0.2 mg of anti-CD8 antibody on the same day as morpholino treatment, the day after, and the day of IR treatment. Subsequent depletions were done at days 14, 21 and 28. To confirm depletion, trunk blood was collected at time of sacrifice (Day 30), and circulating CD8+ cells were measured by flow cytometry as shown before (31). Tumor volume was measured every other day using calipers.
In vivo B16 melanoma model
C57BL/6 WT or CD47-null mice on the same background (Jackson Labs) were injected with B16F10 melanoma cells (ATCC) (1 ×106 cells) into the hind limb to induce tumor growth. When tumors reached an average of 100 mm3 a group of WT and CD47-null tumor bearing mice was treated with local irradiation (10 Gy). Tumor size was measured every third day using calipers, and wet weight was determined at the end of the study. CD8+ T cell depletion studies in B16 melanoma were carried out as explained above. The first depletion was performed the day of IR and weekly after that. CD8+ depletion was confirmed as stated above.
In vitro cytotoxicity assays
CTLs from mice that express a TCR specific for gp160 were isolated from P18I10-Dd specific CTL clone, RT-1 (32). P18I10 is a CTL epitope from HIV gp160. For the in vitro CTL assay, spleens of naïve RT-1 transgenic mice (33) were stimulated with P18I10-pulsed wild-type BALB/c spleen cells for seven to ten days before use. CTLs were left untreated or treated with CD47 morpholino. Groups of cells were used untreated or exposed to ionizing radiation. The cells were co-cultured with fibrosarcoma 15-12 RM cells expressing HIV gp160 or the corresponding Neo control transfectant (29), and target cell viability was measured using the RTCS-ACEA system or by lactate dehydrogenase (LDH) release.
Immunohistochemistry
Tumor tissue sections were paraffin embedded and treated as previously reported (25). Sections were stained with antibodies specific for CD8+, granzyme B, and CD47. Anonymized human melanoma patient sections were handled appropriately in accordance with institutional review board-approved protocol. Immunoreactivity was examined through light microscopy.
Results
Blockade of CD47 enhances radiation growth delay in a model of T-cell adoptive transfer
We previously reported that blockade of CD47 enhances the radiation-induced tumor growth delay in a syngeneic model of lung squamous cell carcinoma (24). When this experiment was repeated using athymic mice, we observed no additive effect of CD47 blockade on growth of irradiated tumors (Supplemental Fig. 1). This indicated that the increase in radiation-induced growth delay observed with CD47 blockade depends on T cell mediated anti-tumor immunity. To determine the role of CD47 blockade on cytotoxicity of CD8+ T cells in vivo, we injected 15-12RM fibrosarcoma cells into the hindlimbs of athymic mice. Once tumors reached an average of 100 mm3, some groups of mice received primed CD8+ T cells isolated from spleens of transgenic RT-1 mice that recognize an epitope derived from the gp160 expressed on 15-12RM tumors (29). At 48 h after treatment, tumors in some groups of mice were exposed to local irradiation. Radiation induced the expected tumor growth delay, but blockade of CD47 did not significantly decrease tumor volume when used alone and did not increase the delay when used in combination with IR (Fig. 1A). Therefore, innate antitumor immunity was not significantly activated by CD47 blockade in the absence of T cells in this syngeneic fibrosarcoma model. These data also suggested that T cells are required for the ability of CD47 to enhance tumor radiosensitivity in this model. Adoptive transfer of tumor-specific CD8+ T cells caused significant reductions in tumor growth when compared to saline and radiation alone (Fig. 1A), and blockade of CD47 did not interfere with the cytotoxic effect of CD8+ T cells in the absence of IR (Fig. 1A and 1B). On the other hand, a profound reduction of tumor growth was observed in mice that were administered CD47 morpholino in combination with CD8+ T cell transfer and exposure to IR, when compared to a group of mice irradiated in combination with CD8+ T cell transfer (Fig. 1A and 1B).
Fig. 1. CD47 blockade combined with irradiation increases the efficacy of adoptive T cell transfer.
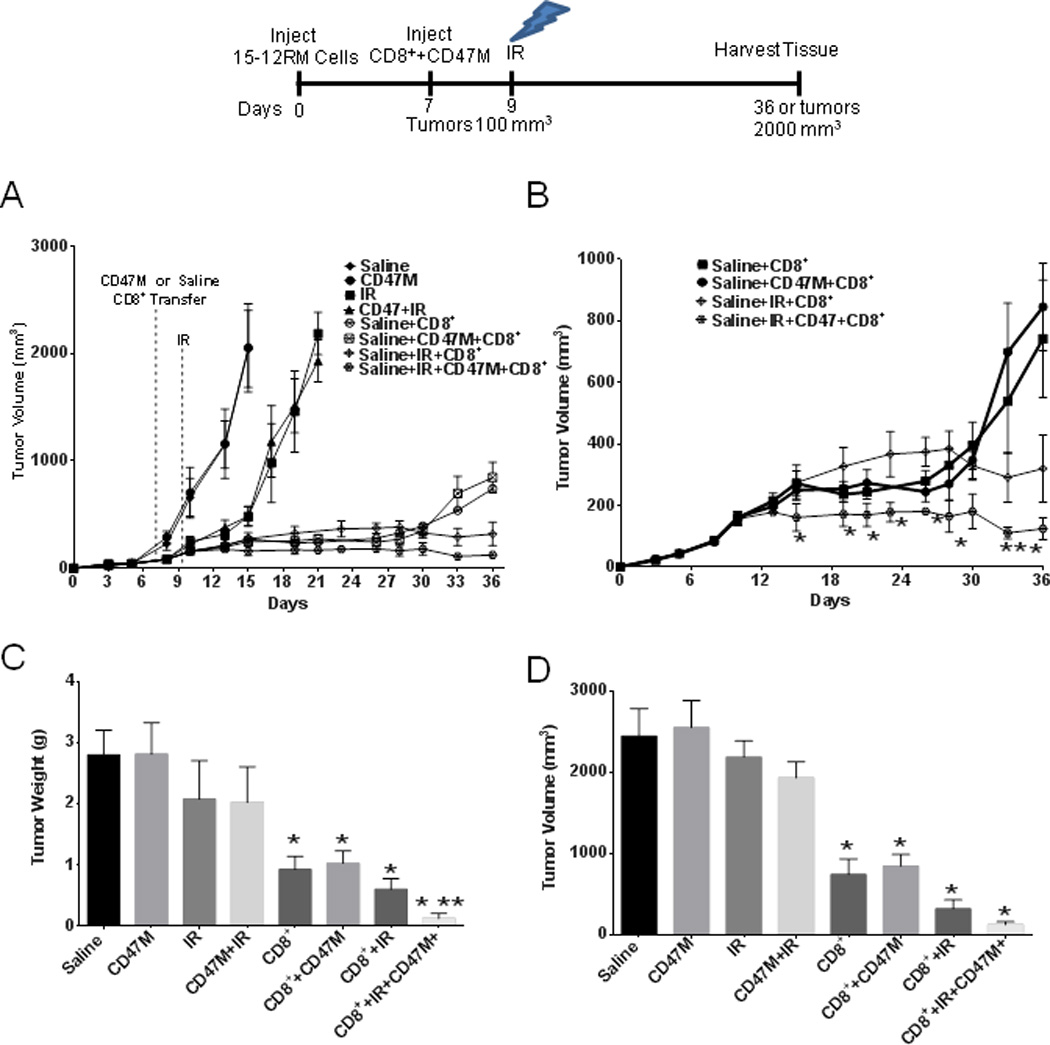
Athymic BALB/c nu/nu mice were injected in one hind limb with 15-12 RM fibrosarcoma cells expressing HIV gp160. Mice were injected IP with saline or 10 µM CD47 morpholino in saline or received these treatments in combination with IR at 10 Gy. (A), Tumor-bearing mice received adoptive transfer (I.V.) of RT-1 CD8+ T cells in the presence or absence of CD47 morpholino (CD47M) treatment (A and B) or received these treatments in combination with IR treatment (A and C) *P<0.05. At the end of the study (day 36) mice were sacrificed, and tumor volume (D) and wet weights (E) were measured. N = 6–8, * statistically significant versus vehicle control, P < 0.05, ** statistically significant CD47+CD8++IR versus CD8++IR.
Tumors excised from animals receiving the CD8+ transfer alone showed a 68% reduction in tumor weight when compared to saline alone, and this was a greater reduction than from a single dose of IR (Fig. 1C). On the other hand, tumors of animals receiving CD47 morpholino and T cell transfer combined with IR showed a 95% reduction when compared to saline and a remarkable reduction in tumor growth when compared to the rest of the treatment groups (Fig. 1C and 1D). Moreover, 30% of these mice had no detectable residual tumor. CD47 morpholino treatment yielded a better response to IR than those receiving only CD8+ cells and IR (tumor volume 320±219 mm3 CD8+IR vs. 124 ±72 mm3 CD47M+IR+CD8, p = 0.018) and lower tumor wet weight (0.60± 0.34 g CD8+IR vs. 0.15 ± 0.20g CD47M+IR+CD8, p=0.03) at the end of the study (Fig. 1 C and D). CD47 knockdown in the tumors was confirmed by western blot hybridization (Supplemental Fig. 3A). These observations indicate that the enhanced radiation growth delay observed in two syngeneic models with blockade of CD47 depends on a competent adaptive immune system and is at least in part mediated by a cytotoxic T cell response.
The radiation growth delay observed with CD47 treatment is dependent on CD8+ T cells in a mouse syngeneic model of fibrosarcoma
To confirm that CD47 blockade can enhance a radiation-induced growth delay in 15-12RM fibrosarcoma growth in an immunocompetent syngeneic host, we injected 15-12RM fibrosarcoma cells into immune-competent BALB/c mice. Once tumors reached a palpable size, we treated groups of animals with saline or 10 µM CD47 morpholino IP. At 48 h after treatment when the tumors reached an average of 100 mm3, a subset of mice from each treatment group was exposed to local IR at a dose of 10 Gy. Tumors of the saline-injected mice continued to grow. In contrast to 15-12RM tumor growth in athymic mice (Fig. 1), we observed a significant (p=0.04) reduction in tumor volume in mice that were administered CD47 morpholino alone (Fig. 2A). This could be explained by a T cell immune response that was directly enhanced by CD47 blockade or by CD47-dependent macrophage priming of T-cell immunity as reported by others (23). We observed the expected radiation growth delay with local IR, and IR treatment slowed tumor growth more than blockade of CD47 alone. On the other hand, administration of the CD47 morpholino in combination with IR caused a significant reduction in tumor growth when compared to the other treatment groups. As shown on Fig. 2B, tumors of mice treated with the combination treatment of IR and CD47 morpholino showed a 96% reduction in tumor growth at the end of the study when compared to saline alone, 90% when compared to CD47 morpholino alone and 79% when compared to IR treatment alone (0.22±0.13 g in the combination treatment vs. 4.5±1.9 g in saline alone, p = 0.001, vs. 2.1±1.8 g CD47 morpholino alone, p = 0.03). No gross pathologies were observed during the study in mice receiving the combination treatment, consistent with the previously reported lack of toxicity resulting from systemic CD47 blockade (24, 34).
Fig. 2. CD8+ T cells are necessary for blockade of CD47 to enhance radiation growth delay in an immunocompetent mouse syngeneic fibrosarcoma model.
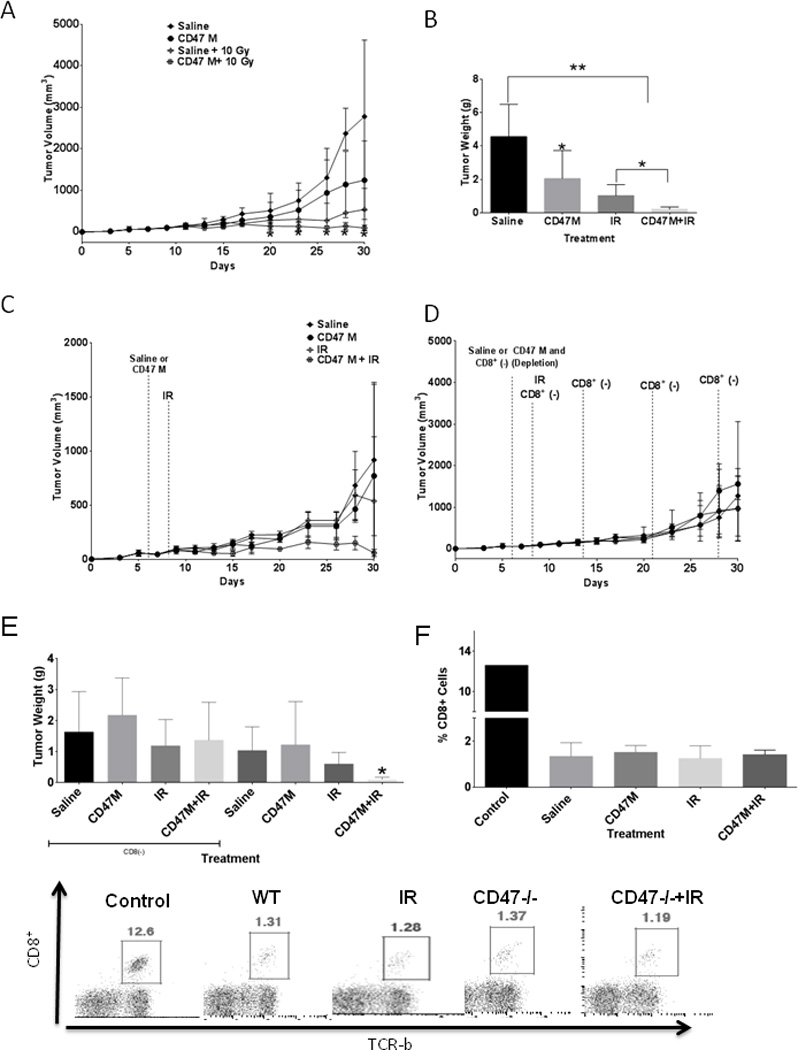
15-12RM cells were injected in immunocompetent BALB/c mice. Groups of mice were treated with saline or 10 µM CD47 morpholino (CD47M), and a subset from each treatment was exposed to IR. (A). At the end of the study (day 30) tumors were excised and wet weights were measured (B, N=6 *p<0.05). In a different set of experiments mice were treated in the same manner in combination with anti-CD8 to deplete CD8+ T cells (C). A concurrent control study was carried out (D, N=6). At the end of the study mice were sacrificed, and wet weight was measured (E). At the end of the study trunk blood was collected from each animal, and circulating CD8+ T cells were measured by flow cytometry to determine the effectiveness of depletion.
The T cell requirement for CD47 blockade to enhance the tumor response to IR could be mediated by regulatory or effector T cell functions. To determine if the enhancement by CD47 blockade requires a CD8+ effector T cell response, mice were treated with anti-CD8 the same day as morpholino treatment (day 7), and the day after. To avoid any CD8+ T cell responses elicited by radiation treatment, the mice were also treated with anti-CD8 the day of radiation treatment and weekly after that until the end of the study. CD8+ T cell depletion increased the growth rate of tumors in the initial weeks of the study when compared to the concurrent control study (Fig. 2C–E). The effectiveness of 10 Gy IR decreased following CD8 depletion (1.4±1.2 vs, 1.2±.9 saline vs. IR in CD8 (−) animals and 1.0±.8 vs. 0.6±0.4 saline and IR animals in immunocompetent mice). However, CD8+ depletion abolished the enhancement from combining ionizing radiation and CD47 blockade (1.2±.9 vs 1.4±1.2 IR vs CD47 IR tumor weight in CD8+-depleted animals. Approximately 90% depletion of circulating CD8+ T cells was achieved under all treatment conditions as assessed by flow cytometry (Fig. 2F).
Blockade of CD47 enhances recruitment of CD8+ cytotoxic T cells in the syngeneic model and T cell transfer mouse models of fibrosarcoma
We stained tumor sections of mice bearing fibrosarcoma tumors with an antibody specific for CD8+ T cells (Fig. 3). As expected sections of tumors grown in athymic mice showed minimal CD8 immunoreactivity (Fig. 3A top panels). On the other hand we observed similar densities of CD8+ T cells in all groups that received adoptive transfer (Fig. 3A bottom panels and 3C),. CD8+ T cell transfer alone caused a 9-fold increase in CD8+ density in tumors, whereas addition of CD47 morpholino caused and over 8-fold increase over saline alone. Transfer of CD8+ in combination with IR and CD47 knockdown caused an over 8-fold increase in CD8+ density when compared to saline alone. We performed the same staining in fibrosarcoma tumors grown in immunocompetent mice and observed a significant 59% (p=0.02) increase in CD8+ density in tumors of mice that were treated with IR and CD47 morpholino when compared to saline and mice receiving IR alone (P=0.006, Fig. 3B and 3D).
Fig. 3. Blockade of CD47 enhances CD8+ T cell recruitment into irradiated tumors.
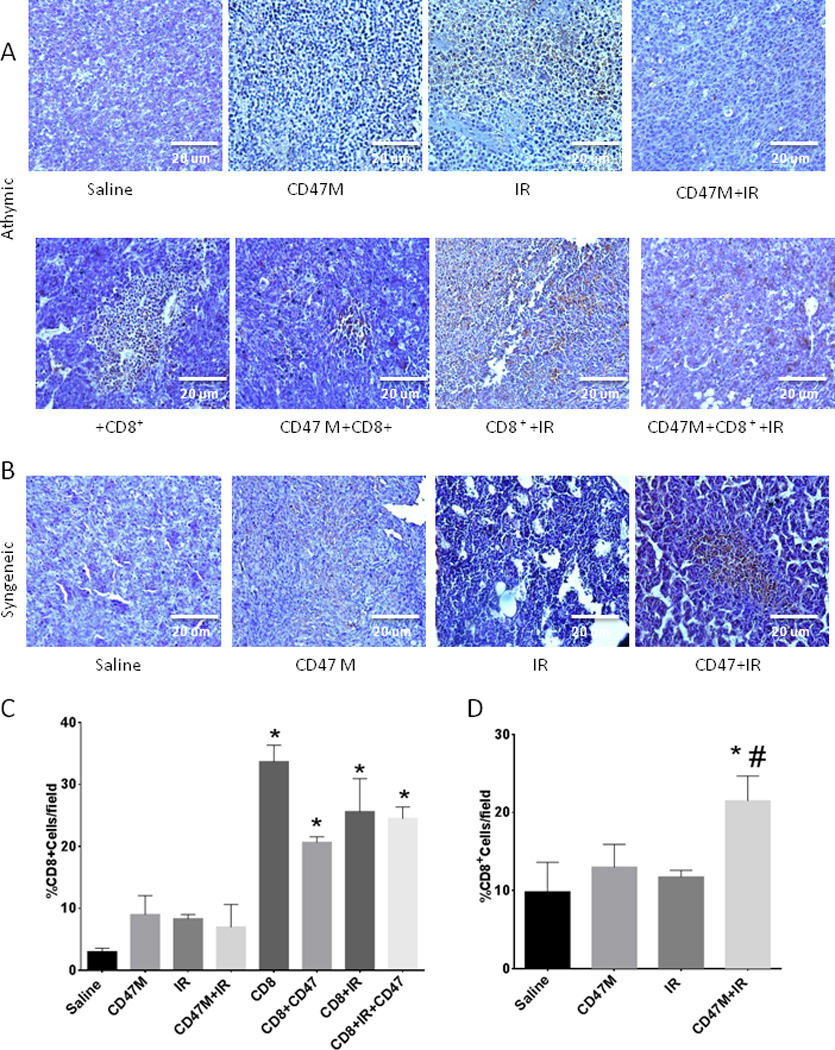
Sections from fibrosarcoma tumors grown in BALB/c athymic mice (A) or BALB/c immunocompetent mice (C) that received the indicated treatments were stained with an antibody to CD8+ cells. Sections were photographed under light microscopy (20×) and quantified using imageJ software (B and D). N=3,4 (4 sections/animal)*p<0.05 from saline #p<0.05 from IR.
CD47 blockade enhances antigen-dependent T cell killing of irradiated 15-12 RM fibrosarcoma cells
To determine whether CD47 blockade can enhance T cell-mediated anti-tumor immunity in the absence of macrophages, we performed dynamic monitoring of CD8+ T cell-mediated tumor cell cytolysis. CTLs from RT-1 TCR transgenic mice recognizing a gp160 peptide were isolated and incubated with 15-12RM fibrosarcoma cells in the RT-CES system, which measures killing of adherent target tumor cells by a decrease in surface impedance, and is presented as a normalized cell index. Cell index is derived from the measured electrical impedance and is a quantitative measure of the number of viable adherent target cells in each well. Therefore, a decreased cell index indicates loss of viable target tumor cells. This is an established quantitative real-time method for assessing antigen-dependent CTL activity (35–37) Cells were plated at a 1:5 T/E ratio, which yielded optimal cytotoxic activity (supplemental Fig. 2A). As observed in Fig. 4A, 15-12RM fibrosarcoma cells continued to grow over time at a similar rate as the antigen-independent Neo control cell line. Treatment of these two cell lines with IR attenuated growth of the target cells, but the cells maintained stable growth over time. Blockade of CD47 on fibrosarcoma cells did not affect proliferation of this cell line (Fig. 4A, supplemental Fig. 2B), however incubation of CTLs with 15-12RM fibrosarcoma cells caused an antigen-dependent reduction in target cell viability, indicating specific tumoricidal activity of T cells against the 15-12RM fibrosarcoma cells. Blockade of CD47 in the fibrosarcoma cells caused a steady enhancement in T-cell mediated cytotoxicity when compared to untreated CTL incubated with fibrosarcoma cells. Combination with radiation and CD47 blockade caused a 5-fold increase in cell death over untreated 15-12RM fibrosarcoma cells and 3-fold increase in cell death when compared to CTLs from irradiated and untreated pairs at 24 h. Blocking CD47 in the fibrosarcoma cells and irradiating the cells caused 1.5-fold increase in tumoricidal activity, showing further enhancement of cancer cell killing when combining CD47 blockade and irradiation.
Fig. 4. CD47 blockade enhances antigen-dependent T cell killing of 15-12RM fibrosarcoma when combined with irradiation.
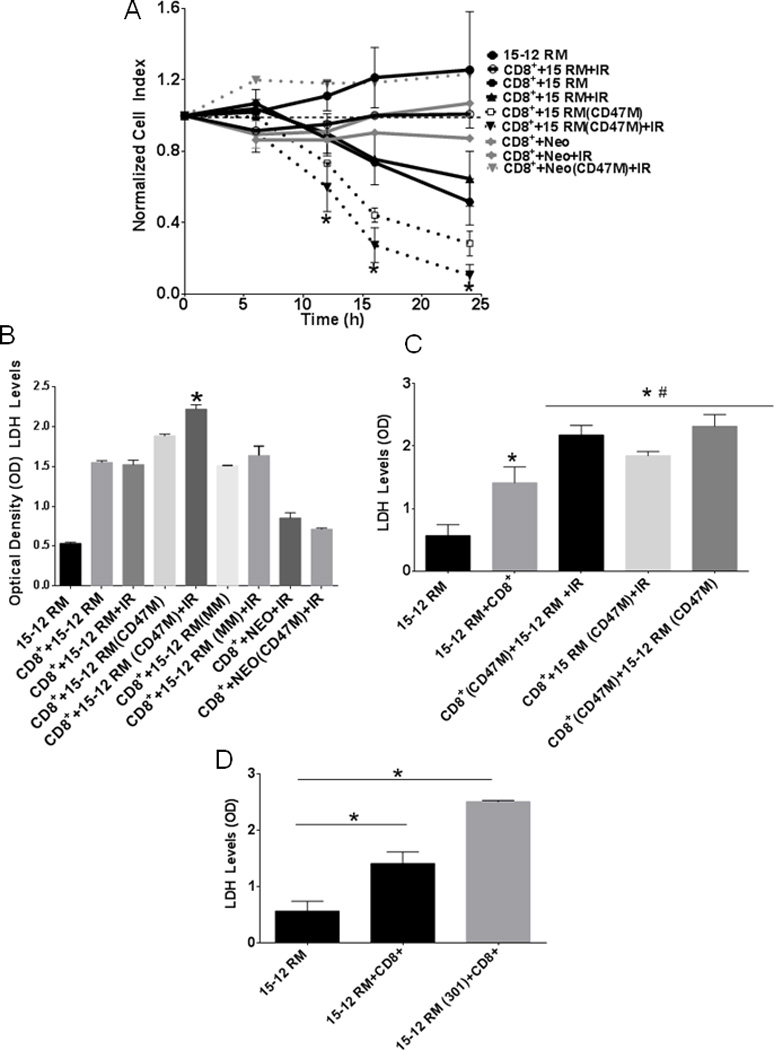
(A) 15-12RM target cells were seeded into 16-well plates. RT-1-derived effector T cells were co-cultured with target cells at a ratio of 1:5, and target cell growth and viability was dynamically monitored using the RT-CES system. Target cell viability monitored by surface impedance is presented as a normalized cell index (N=3 in triplicate, *p<0.05). Cells were plated into 96-well plates and effector and or targets cells were treated with 10 µM CD47 Morpholino (CD47 M; A, C and D). Mismatched morpholino (MM A and B) or mouse antibody to CD47 clone 301 (D), and LDH release was quantified after 24 h as a measure of cell cytotoxicity, N=3 *p<0.05.
We measured LDH release to confirm that isolated CTLs demonstrated tumoricidal activity against the fibrosarcoma cells. Blockade of CD47 alone increased LDH release over untreated effector plus target co-cultures, and this effect was further enhanced with IR (Fig 4B). Administration of a control mismatched morpholino (MM) did not affect cytolysis (Fig. 4B). In both assays we did not observe increased killing with CD47 blockade when Neo cells lacking gp160 expression were used as the target, indicating that the enhanced T cell-mediated killing upon blocking CD47 is antigen-dependent.
In similar experiments we tested whether blocking CD47 in the effector T-cells would enhance the tumoricidal activity of CD8+ T cells by measuring LDH release. Isolated CTLs were treated with 10 µM CD47 morpholino for 48 h and then were co-cultured with untreated 15-12RM cells (Fig. 4C). Blocking CD47 in the CTL with morpholino caused a significant increase in tumoricidal activity at 24 h of T cells over untreated CTL (Fig. 4C, p = 0.0007). CD47 knockdown on the CD8+ T cells was confirmed by western blot hybridization (Supplemental Fig. 3B). Blocking CD47 in the target cells as well as in both effector and target enhanced cytotoxic killing as well over untreated effector plus target cells (p=0.046, p=0074 respectively). Consistent with our previous report of radioprotection in Jurkat T cells (25), blocking CD47 in CD8+ T cells caused radioprotection of these cells (Supplemental Fig. 2C). These results demonstrate that blockade of CD47 directly increases the viability and tumoricidal activity of effector CD8+ cytotoxic T cells.
To independently confirm that CD47 directly regulates CTL-mediated tumor cell killing, we tested the well-characterized murine CD47-blocking antibody clone 301 (Fig. 4D). Treatment with the CD47 antibody increased T cell mediated cytotoxicity when incubated with effector and target cells. This further indicates that CD47 signaling in the T cells plays a major role in regulating target tumor cell killing.
CD47 in the tumor microenvironment regulates anti-tumor T-cell responses
Several studies have shown that reducing CD47 expression in tumor cells results in enhanced innate immunosurveillance (38, 39). However, the preceding results indicate that the role of selective CD47 blockade in the tumor microenvironment must also be considered. We examined tumor sections of human melanoma patients to correlate expression of CD47 and CD8+ cells. (Fig. 5A and 5B) Our data demonstrated a significant inverse correlation between CD47 expression and infiltration of CD8+ T cells (p=0.021). This suggests that expression of CD47 by tumor cells or in the tumor microenvironment can limit the infiltration of cytotoxic T cells into human tumors.
Fig. 5. Lack of CD47 in the tumor microenvironment increases the IR-induced growth delay in a syngeneic mouse melanoma model.
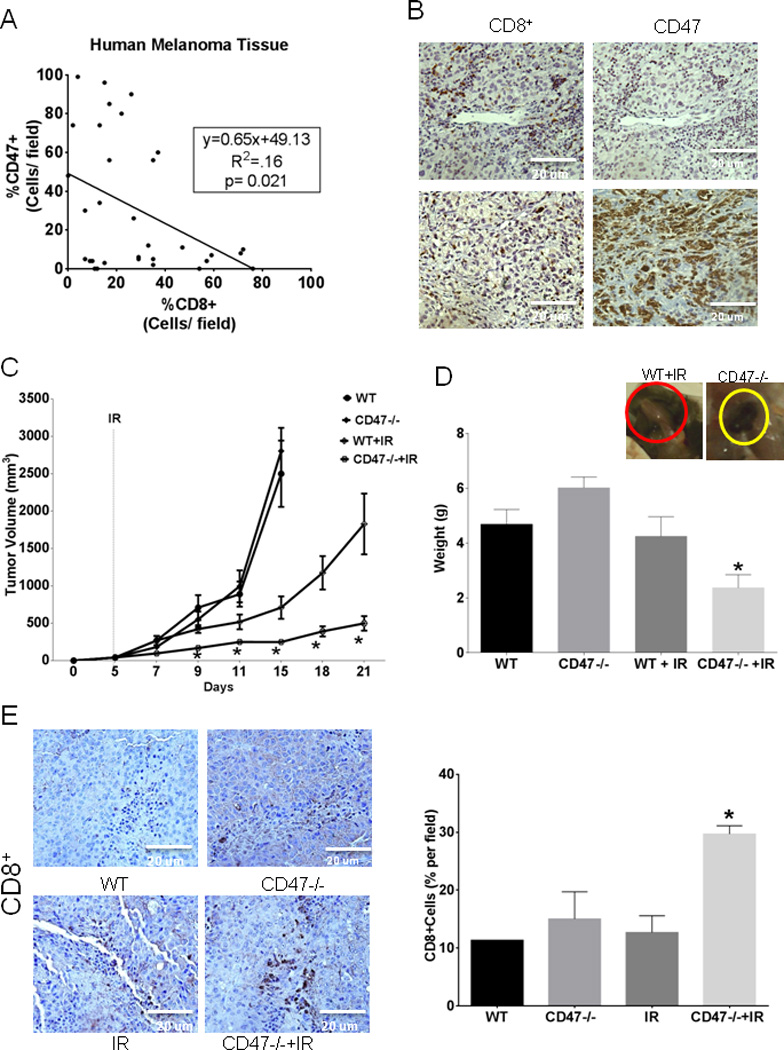
(A) Thirty-eight paired serial sections from 13 human melanoma tumors were stained with antibodies to human CD47 and CD8. Numbers of CD47+ and CD8+ cells were inversely correlated (p=0.021 by Pearson correlation). (B) Representative stained tumor sections (20×) from melanoma patients with low (upper panels) and high CD47 expression (lower panels). (C) B16F10 melanoma cells were injected into WT and CD47−/− mice hind limbs and were irradiated at 10 Gy on day 5. Tumor dimensions were measured ever other day using calipers, and tumor volume was calculated as W2 × L/2, where W = shortest diameter and L = longest diameter. (D) Tumors were excised, and weights were recorded at the end of the study. N=5, *p<0.05. (E) Immunohistochemical analysis to detect CD8+ infiltrating cells (Brown stain) in melanoma tumor sections counterstained Hematoxilin, sections were visualized under light microscopy (20×) and quantified using ImageJ software N=3 (4 sections/animal) *p<0.05.
To specifically examine the role of CD47 in the tumor microenvironment we injected B16 melanoma cells into syngeneic WT or CD47-null C57BL/6 mice and followed tumor growth. As shown in Fig. 5C, tumors grew at similar rates in WT and CD47-null microenvironments, with no significant differences in tumor volume. Treatment of tumor-bearing WT mice with IR caused the expected radiation growth delay. However, treatment of tumor-bearing CD47-null mice with IR caused a significant further reduction in melanoma tumor growth compared to irradiated WT throughout the study, with 73% smaller tumor volume at day 21 (1830±410 WT+IR vs. 500±100 CD47−/−+IR, p<0.02). At the end of the study we also observed a 45% reduction in tumor wet weight in irradiated tumors grown in a CD47-null microenvironment versus irradiated alone (Fig. 5D). To determine if T cell-mediated antitumor immunity played a role in this effect we assessed CD8+ T cell infiltration by immunohistochemistry. Sections of irradiated melanoma tumors grown in a CD47-null microenvironment exhibited a significant 60% increase in CD8+ T cell infiltration compared to tumor sections of saline and irradiated tumors grown in WT mice (Fig. 5E). In a similar set of experiments WT and CD47-null mice bearing B16F10 melanoma tumors were depleted of CD8+ T cells by weekly treatment with anti-CD8 and where indicated treated with IR (Supplemental Fig. 4A and B). CD8 depletion was confirmed at the time of sacrifice (Supplemental Fig. 4C). CD8 depletion did not affect the growth of melanoma tumors in a WT or CD47-null microenvironment. However depletion of CD8+ cells attenuated the effect of ionizing radiation when compared to immunocompetent animals (Supplemental Fig. 4A and B). The enhanced IR-induced tumor growth delay observed in a CD47-null microenvironment was also attenuated when compared to the enhancement observed in immunocompetent mice (compare Fig. 5C).
Tumor CD47 expression regulates granzyme B and Foxp3 expression
Since we observed similar T cell numbers in our fibrosarcoma model but enhanced T cell-mediated killing associated with CD47 blockade, we proposed that CD47 signaling limits functional CTL activation. As observed in Fig. 6A irradiated 15-12RM tumors grown in nu/nu BALB/c mice and treated with CD47 morpholino and receiving T cell adoptive transfer showed a 10-fold increase in granzyme B mRNA expression over untreated mice (WT). CD8+ T cell transfer or IR treatment alone did not increase granzyme B mRNA expression. Combining CD8+ T cell transfer or treatment with CD47 morpholino showed elevated levels of granzyme B over WT but did not achieve significance. Moreover, we observed a 20-fold increase in granzyme B gene expression in tumors of mice developed in Balb/c immunocompetent mice that were treated with CD47 morpholino and irradiation (Fig. 6B). We also observed a similar fold increase in granzyme B mRNA expression in B16 melanoma tumors developed in a CD47-null microenvironment that were irradiated when compared to untreated B16 tumors (Fig. 6C). To determine if the increased granzyme B expression simply reflects CD8+ T cell infiltration we determined CD8α mRNA expression in tumors and calculated the ratio of granzyme B to CD8α expression (supplemental Fig. 5). The ratio was highest with combined IR and CD47 morpholino or in a CD47 null microenvironment, indicating that CD47 blockade increased expression. Therefore, combining CD47 blockade and IR treatment of 15 RM fibrosarcoma tumors and B16 melanoma tumors has similar effects to increase granzyme B expression independent of increased CD8+ T cell infiltration.
Fig. 6. Deficiency of CD47 increases granzyme B and regulates Foxp3 gene expression in tumors.
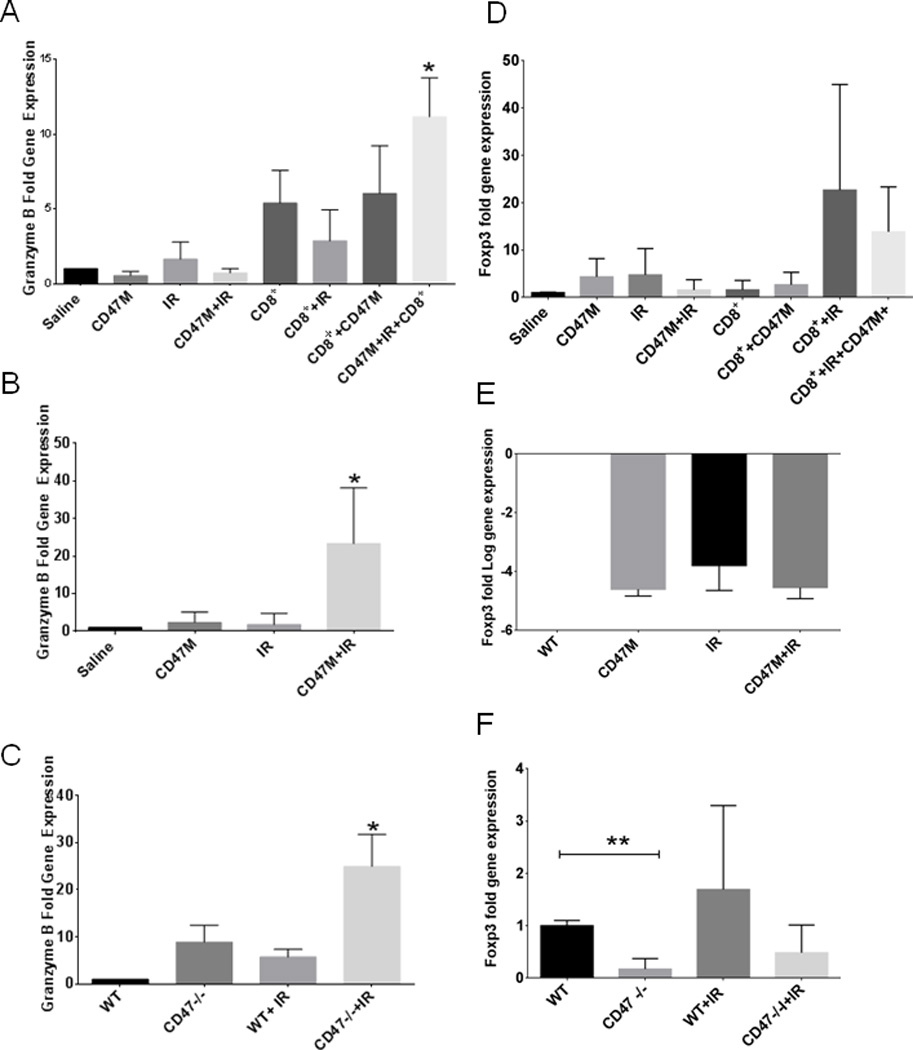
Expression of granzyme B and Foxp3 was determined by RT-PCR in 15-12RM tumors grown in athymic BALB/c mice (A,D), immunocompetent BALB/c mice (B,E), or B16 melanoma tumors grown in WT or CD47-null C57BL/6 mice (C,F) (*p<0.05.).
We also examined tumor sections for expression of Foxp3, a marker of regulatory T cells that can result in an immunosuppressive microenvironment. We did not observe a significant regulation of Foxp3 mRNA in our fibrosarcoma model (Fig. 6D–E), but we observed a 70% (*p<0.05) reduction of Foxp3 mRNA in B16 tumors grown in a CD47-null microenvironment (Fig. 6F). This is consistent with a previous study of CD47-null mice (40), and suggests that expression of CD47 in certain tumor microenvironments may alter T-reg differentiation or infiltration of T-regs and thereby limit adaptive tumor immunity.
CD47 regulates granzyme B expression in isolated CD8+ T cells and in spleen tissue
To determine whether blockade of CD47 caused autonomous up-regulation of granzyme B in T cells, we treated isolated CD8+ T cells with 10 µM CD47 morpholino for 48 h. Blockade of CD47 increased granzyme B protein expression in lysates from the CD8+ T cells used for our cytotoxicity assays (Fig. 7A). A corresponding 3-fold increase in granzyme B mRNA expression was observed in extracts from cells treated with the CD47 morpholino (Fig. 7B). We stained spleen sections from WT and CD47-null mice and observed an increase in granzyme B protein expression in sections of spleen from CD47-null mice represented by the strong staining shown in the lower panel of Fig. 7C. This indicates that blockade or deficiency of CD47 causes an autonomous up-regulation of granzyme B in effector T cells that is consistent with their increased cytotoxic killing of irradiated tumor targets and decreased tumor burden when CD47 is blocked.
Fig. 7. Deficiency of CD47 increases granzyme B in CD8+ cells and in spleen tissue.
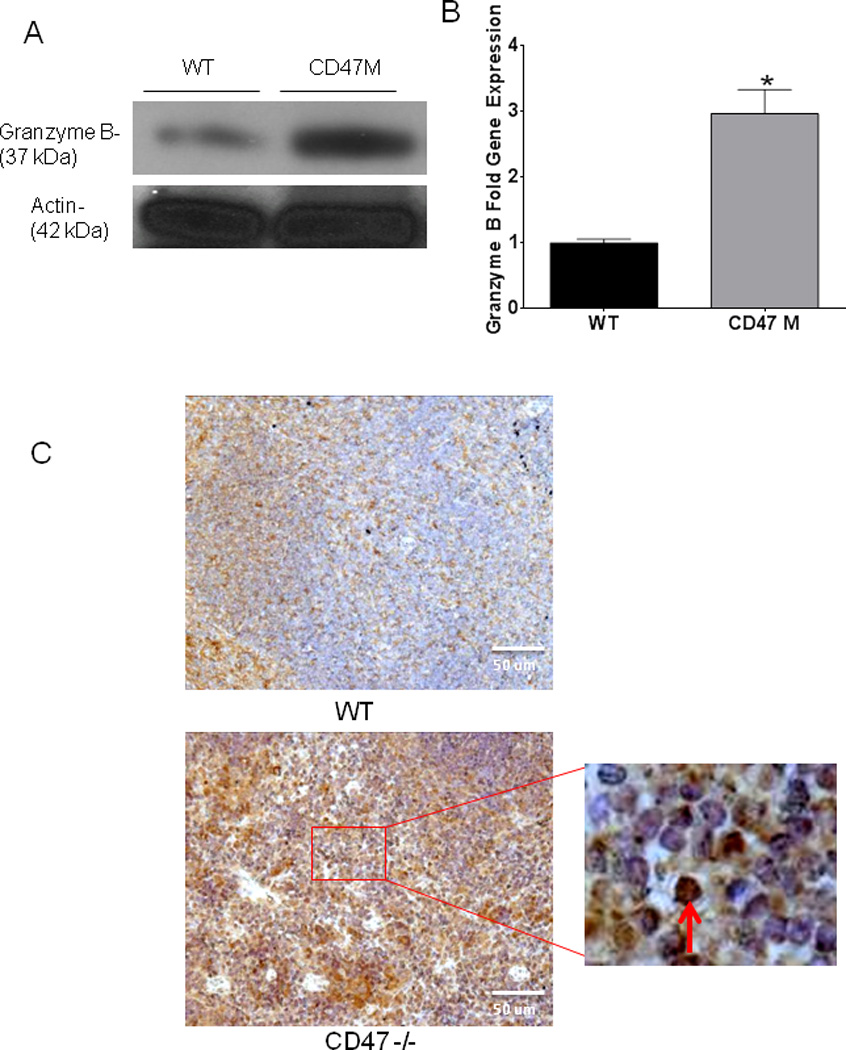
Protein expression (A) and mRNA expression (B) was determined by Western Blot Hybridization and qRT-PCR respectively in CD8+ T cells used for cytotoxicity assays (*p<0.007). Tissue sections of spleens from WT and CD47-null mice bearing B16 melanoma tumors were stained with an antibody specific to granzyme B arrow denotes granular pattern of stain (C).
Discussion
This study provides new evidence that blockade of CD47 directly facilitates the cytolytic activity of CTL to improve anti-tumor immunity and further expands our previous studies showing that blockade of CD47 potentiates the pro-death effects of ionizing radiation in tumors. Blocking CD47 in tumor cells as well as in cytotoxic T cells enhances anti-tumor responses of cytotoxic T cells in vitro and in the tumor microenvironment in vivo. Blockade of CD47 in tumor cells enhances T cell-mediated fibrosarcoma cell killing. On the other hand blocking CD47 in effector cells is also sufficient to cause a significant enhancement of T cell-mediated tumor cell cytotoxicity. Suppression of CD47 in effector T cells causes an autonomous increase in granzyme B expression that can mediate target tumor cell death. Enhanced T cell activation following CD47 blockade is consistent with the well-established inhibitory CD47 signaling pathways activated by TSP1 binding to CD47 on T cells (9, 12, 16, 18, 20, 41).
We previously reported that suppression of CD47 by systemic delivery of a CD47 morpholino enhances the radiation-induced tumor regrowth delay in two syngeneic tumor models (24). We observed increased leukocyte migration into the treated tumors and predicted that the radioprotective effects of CD47 blockade preserve anti-tumor immunity, which would then target tumor cells expressing radiation-induced damage-associated molecular patterns (DAMPs) (27). These DAMPs can stimulate the killing by tumor-associated macrophages, but performing our squamous cell carcinoma and fibrosarcoma models in mice lacking T cell-mediated immunity (42) or in immunocompetent mice depleted of CD8+ T cells demonstrated that effector T cells are necessary for blockade of CD47 to synergize with IR to inhibit tumor growth. These results also indicate that the reported function of CD47 expressed on tumor cells to inhibit killing by tumor-associated macrophages does not play a significant role in the enhancement of radiation responses following blockade of CD47 in our models. Although blockade of CD47 alone had no effect on tumor growth in the 15-12RM model in athymic mice, a profound tumor ablation was observed when T cell transfer is combined with IR. Moreover, when the same fibrosarcoma tumors were injected into immunocompetent mice we observed a similar additive effect between radiation and CD47 blockade to delay tumor growth that was lost following CD8+ T cell depletion. Others have reported that tumor responses to ionizing radiation are impaired in the absence of T-cell immunity (28). Our results confirm this observation but also indicate that expression of CD47 on CTLs and tumor cells limits CTL-mediated anti-tumor immunity.
In addition to the direct roles of CD47 in T cell immunity shown here, anti-CD47 antibody-mediated phagocytosis by macrophages was reported to initiate an anti-tumor T-cell response (23). OVA-specific CD8+ T cells harvested from lymph nodes and co-cultured with macrophages treated with a CD47-blocking antibody, B6H12, showed increased antigen-dependent proliferation only after CD47-mediated phagocytosis. In vivo, the cytotoxic effect of CD8+ cells was also dependent on priming by macrophages. Although macrophage antigen presentation can initiate T cell-mediated anti-tumor responses, our studies indicate that blocking CD47 on T cells is sufficient to directly activate T cell cytotoxicity towards tumors. Our in vitro killing assays clearly demonstrate that CD47 blockade increases CTL activity independent of macrophage priming since we primed CTLs prior to transfer. Moreover, our in vitro data and in vivo B16 melanoma models confirm that blocking CD47 on the effector cells or the absence of CD47 in the tumor microenvironment is sufficient to enhance anti-tumor T cell immunity. Conversely, elevated CD47 expression in human melanoma sections correlated with decreased CD8+ T cell infiltration, suggesting immunosuppressive effects of intratumoral CD47 caused by inhibition of effector T cell recruitment as well as activation.
Most studies to date concerning the role of CD47 in cancer have focused on the immunosuppressive role of CD47 expression by tumor cells and how reducing this expression can enhance anti-tumor innate immunity mediated by macrophages or NK cells. However, CD47 expression on T cells is well known to regulate their activation and functional differentiation in vitro and in vivo (21, 43). Our results show for the first time that blocking CD47 in effector cytotoxic T cells can directly enhance their anti-tumor response. This is evidenced by reduced tumor burden when B16 melanoma tumors are treated with ionizing radiation in a CD47-null host. In this model an increased CD8+ infiltrate was observed in the tumors (Fig. 5), and increased gene expression of granzyme B was observed in tumors grown in CD47-null hosts. Furthermore, the effect of IR in the 15-12RM CTL transfer model was significantly enhanced only when CD8 CTL and CD47 morpholino treatments were combined but not by either one alone, which strongly supports this conclusion. Conversely, depletion of CD8+ T cells decreased the benefit of CD47 blockade, indicating that effector CTL rather than regulatory T cells are the relevant target of the CD47 morpholino.
Granzyme B is secreted mainly by cytotoxic T cells and NK cells and can induce apoptosis of target cells by activation of caspase or directly by disrupting mitochondrial function (44, 45). Correspondingly, granzyme B expression in the tumor microenvironment is associated with a positive clinical outcome (44). In our three in vivo mouse models, deficiency of CD47 in combination with IR and functional T cell immunity results in increased granzyme B expression in the tumor. Moreover, the T cells that we used for our adoptive transfer model expressed increased granzyme B mRNA and protein levels when CD47 expression was suppressed using anti-sense morpholinos.
Our previous studies and some others have observed that blocking CD47 alone does not result in significant tumor ablation (24), but combining CD47 blockade with radiation or specific cytotoxic antibodies enhances the anti-tumor activity (24, 46). On the other hand, we observed in our fibrosarcoma model in immunocompetent mice that blockade of CD47 alone moderately reduced tumor burden. We cannot distinguish whether this is mediated by enhanced CTL activity or the enhancement of antitumor innate immunity induced by CD47 blockade that was observed in several xenograft models in Nod.SCID mice (47, 48). The relative enhancement of macrophage versus CTL killing following CD47 blockade could be tumor dependent and may be influenced by the level of CD47 expression on a given tumor. However, the importance of T cells in tumor responses to CD47 blockade cannot be studied in xenograft models. Additional functions of CD47 in the tumor microenvironment involving its interaction with thrombospondin-1 that regulate angiogenesis and tumor blood flow may also contribute to an anti-tumor response (49, 50).
In addition to enhancing functional CTL activation, CD47 blockade is expected to increase survival of tumor infiltrating lymphocytes due to the general radioprotective activity of CD47 depletion in normal cells and soft tissues (25, 34, 50, 51). Radiation elicits an initial activation of the immune system that can stimulate an anti-tumor immune response but can also stimulate immunosuppressive pathways (52). Thus, blockade with CD47 could sustain an anti-tumor response indirectly by inhibiting immunosuppressive signals activated by its expression in antigen presenting cells ((53, 54)) and directly by protecting tumor-infiltrating CTL from death caused by local irradiation of the tumor.
Systemic blockade of CD47 using antisense morpholinos did not result in any gross pathologies in this or previously reported mouse models (24, 34). Morpholinos targeting other transcripts have shown safety and efficacy in clinical studies and have shown efficacy as anti-cancer strategies (55). Therefore, CD47 antisense morpholinos could be an effective clinical adjuvant to improve immunotherapy. These could be employed alone or, preferably, combined with cytotoxic therapies such as IR. Importantly, targeting CD47 promotes ablation of tumor tissue while sparing normal cells from the off-target effects of IR therapy. Therefore, therapeutic CD47 blockade may improve patient quality of life while increasing the percentage of complete responses to current anti-cancer therapies.
Supplementary Material
Acknowledgments
This work was supported by the Intramural Research Program of the NIH/NCI (D.D.R., D.A.W., J.A.B.) D. R. S.-P., M. T., W.D.G and L.A.R. performed experiments, D.R.S.-P. and A.G. analyzed data. D.R.S.-P. M. T., J. A. B., D.A.W, and D.D.R. wrote the manuscript. We would like to thank Dr. Steven Rosenberg for providing human melanoma sections for our study.
Footnotes
Conflict of Interest Statement: The authors have no conflict of interest to report.
REFERENCES
- 1.Majeti R, Chao MP, Alizadeh AA, Pang WW, Jaiswal S, Gibbs KD, Jr, et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell. 2009;138:286–299. doi: 10.1016/j.cell.2009.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chao MP, Alizadeh AA, Tang C, Jan M, Weissman-Tsukamoto R, Zhao F, et al. Therapeutic antibody targeting of CD47 eliminates human acute lymphoblastic leukemia. Cancer Res. 2011;71:1374–1384. doi: 10.1158/0008-5472.CAN-10-2238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Edris B, Weiskopf K, Volkmer AK, Volkmer JP, Willingham SB, Contreras-Trujillo H, et al. Antibody therapy targeting the CD47 protein is effective in a model of aggressive metastatic leiomyosarcoma. Proc Natl Acad Sci U S A. 2012;109:6656–6661. doi: 10.1073/pnas.1121629109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jaiswal S, Jamieson CH, Pang WW, Park CY, Chao MP, Majeti R, et al. CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell. 2009;138:271–285. doi: 10.1016/j.cell.2009.05.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Willingham SB, Volkmer JP, Gentles AJ, Sahoo D, Dalerba P, Mitra SS, et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci U S A. 2012;109:6662–6667. doi: 10.1073/pnas.1121623109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Isenberg JS, Maxhimer JB, Powers P, Tsokos M, Frazier WA, Roberts DD. Treatment of liver ischemia-reperfusion injury by limiting thrombospondin-1/CD47 signaling. Surgery. 2008;144:752–761. doi: 10.1016/j.surg.2008.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Isenberg JS, Martin-Manso G, Maxhimer JB, Roberts DD. Regulation of nitric oxide signalling by thrombospondin 1: implications for anti-angiogenic therapies. Nat Rev Cancer. 2009;9:182–194. doi: 10.1038/nrc2561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ramanathan S, Mazzalupo S, Boitano S, Montfort WR. Thrombospondin-1 and angiotensin II inhibit soluble guanylyl cyclase through an increase in intracellular calcium concentration. Biochemistry. 2011;50:7787–7799. doi: 10.1021/bi201060c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li Z, He L, Wilson K, Roberts D. Thrombospondin-1 inhibits TCR-mediated T lymphocyte early activation. J Immunol. 2001;166:2427–2436. doi: 10.4049/jimmunol.166.4.2427. [DOI] [PubMed] [Google Scholar]
- 10.Li Z, Calzada MJ, Sipes JM, Cashel JA, Krutzsch HC, Annis DS, et al. Interactions of thrombospondins with alpha4beta1 integrin and CD47 differentially modulate T cell behavior. J Cell Biol. 2002;157:509–519. doi: 10.1083/jcb.200109098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kaur S, Kuznetsova SA, Pendrak ML, Sipes JM, Romeo MJ, Li Z, et al. Heparan sulfate modification of the transmembrane receptor CD47 is necessary for inhibition of T cell receptor signaling by thrombospondin-1. J Biol Chem. 2011;286:14991–15002. doi: 10.1074/jbc.M110.179663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Miller TW, Kaur S, Ivins-O'Keefe K, Roberts DD. Thrombospondin-1 is a CD47-dependent endogenous inhibitor of hydrogen sulfide signaling in T cell activation. Matrix Biol. 2013;32:316–324. doi: 10.1016/j.matbio.2013.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brooke G, Holbrook JD, Brown MH, Barclay AN. Human lymphocytes interact directly with CD47 through a novel member of the signal regulatory protein (SIRP) family. J Immunol. 2004;173:2562–2570. doi: 10.4049/jimmunol.173.4.2562. [DOI] [PubMed] [Google Scholar]
- 14.Irandoust M, Alvarez Zarate J, Hubeek I, van Beek EM, Schornagel K, Broekhuizen AJ, et al. Engagement of SIRPalpha Inhibits Growth and Induces Programmed Cell Death in Acute Myeloid Leukemia Cells. PLoS One. 2013;8:e52143. doi: 10.1371/journal.pone.0052143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barazi HO, Li Z, Cashel JA, Krutzsch HC, Annis DS, Mosher DF, et al. Regulation of integrin function by CD47 ligands. Differential effects on alpha vbeta 3 and alpha 4beta1 integrin-mediated adhesion. J Biol Chem. 2002;277:42859–42866. doi: 10.1074/jbc.M206849200. [DOI] [PubMed] [Google Scholar]
- 16.Grimbert P, Bouguermouh S, Baba N, Nakajima T, Allakhverdi Z, Braun D, et al. Thrombospondin/CD47 interaction: a pathway to generate regulatory T cells from human CD4+ CD25− T cells in response to inflammation. J Immunol. 2006;177:3534–3541. doi: 10.4049/jimmunol.177.6.3534. [DOI] [PubMed] [Google Scholar]
- 17.Van VQ, Lesage S, Bouguermouh S, Gautier P, Rubio M, Levesque M, et al. Expression of the self-marker CD47 on dendritic cells governs their trafficking to secondary lymphoid organs. EMBO J. 2006;25:5560–5568. doi: 10.1038/sj.emboj.7601415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lamy L, Foussat A, Brown EJ, Bornstein P, Ticchioni M, Bernard A. Interactions between CD47 and thrombospondin reduce inflammation. J Immunol. 2007;178:5930–5939. doi: 10.4049/jimmunol.178.9.5930. [DOI] [PubMed] [Google Scholar]
- 19.Kim MJ, Lee JC, Lee JJ, Kim S, Lee SG, Park SW, et al. Association of CD47 with natural killer cell-mediated cytotoxicity of head-and-neck squamous cell carcinoma lines. Tumour Biol. 2008;29:28–34. doi: 10.1159/000132568. [DOI] [PubMed] [Google Scholar]
- 20.Bouguermouh S, Van VQ, Martel J, Gautier P, Rubio M, Sarfati M. CD47 expression on T cell is a self-control negative regulator of type 1 immune response. J Immunol. 2008;180:8073–8082. doi: 10.4049/jimmunol.180.12.8073. [DOI] [PubMed] [Google Scholar]
- 21.Van VQ, Darwiche J, Raymond M, Lesage S, Bouguermouh S, Rubio M, et al. Cutting edge: CD47 controls the in vivo proliferation and homeostasis of peripheral CD4+ CD25+ Foxp3+ regulatory T cells that express CD103. J Immunol. 2008;181:5204–5208. doi: 10.4049/jimmunol.181.8.5204. [DOI] [PubMed] [Google Scholar]
- 22.Azcutia V, Routledge M, Williams MR, Newton G, Frazier WA, Manica A, et al. CD47 plays a critical role in T-cell recruitment by regulation of LFA-1 and VLA-4 integrin adhesive functions. Mol Biol Cell. 2013 doi: 10.1091/mbc.E13-01-0063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tseng D, Volkmer JP, Willingham SB, Contreras-Trujillo H, Fathman JW, Fernhoff NB, et al. Anti-CD47 antibody-mediated phagocytosis of cancer by macrophages primes an effective antitumor T-cell response. Proc Natl Acad Sci U S A. 2013;110:11103–11108. doi: 10.1073/pnas.1305569110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Maxhimer JB, Soto-Pantoja DR, Ridnour LA, Shih HB, Degraff WG, Tsokos M, et al. Radioprotection in normal tissue and delayed tumor growth by blockade of CD47 signaling. Sci Transl Med. 2009;1:3ra7. doi: 10.1126/scitranslmed.3000139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Soto Pantoja DR, Miller TW, Pendrak ML, DeGraff WG, Sullivan C, Ridnour LA, et al. CD47 deficiency confers cell and tissue radioprotection by activation of autophagy. Autophagy. 2012;8:0–1. doi: 10.4161/auto.21562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Demaria S, Ng B, Devitt ML, Babb JS, Kawashima N, Liebes L, et al. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int J Radiat Oncol Biol Phys. 2004;58:862–870. doi: 10.1016/j.ijrobp.2003.09.012. [DOI] [PubMed] [Google Scholar]
- 27.Ludgate CM. Optimizing cancer treatments to induce an acute immune response: radiation Abscopal effects, PAMPs, and DAMPs. Clin Cancer Res. 2012;18:4522–4525. doi: 10.1158/1078-0432.CCR-12-1175. [DOI] [PubMed] [Google Scholar]
- 28.Demaria S, Formenti SC. Role of T lymphocytes in tumor response to radiotherapy. Front Oncol. 2012;2:95. doi: 10.3389/fonc.2012.00095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Matsui S, Ahlers JD, Vortmeyer AO, Terabe M, Tsukui T, Carbone DP, et al. A model for CD8+ CTL tumor immunosurveillance and regulation of tumor escape by CD4 T cells through an effect on quality of CTL. J Immunol. 1999;163:184–193. [PubMed] [Google Scholar]
- 30.Terabe M, Ambrosino E, Takaku S, O'Konek JJ, Venzon D, Lonning S, et al. Synergistic enhancement of CD8+ T cell-mediated tumor vaccine efficacy by an anti-transforming growth factor-beta monoclonal antibody. Clin Cancer Res. 2009;15:6560–6569. doi: 10.1158/1078-0432.CCR-09-1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Izhak L, Ambrosino E, Kato S, Parish ST, O'Konek JJ, Weber H, et al. Delicate balance among three types of T cells in concurrent regulation of tumor immunity. Cancer Res. 2013;73:1514–1523. doi: 10.1158/0008-5472.CAN-12-2567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Takahashi H, Houghten R, Putney SD, Margulies DH, Moss B, Germain RN, et al. Structural requirements for class I MHC molecule-mediated antigen presentation and cytotoxic T cell recognition of an immunodominant determinant of the human immunodeficiency virus envelope protein. J Exp Med. 1989;170:2023–2035. doi: 10.1084/jem.170.6.2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yokosuka T, Takase K, Suzuki M, Nakagawa Y, Taki S, Takahashi H, et al. Predominant role of T cell receptor (TCR)-alpha chain in forming preimmune TCR repertoire revealed by clonal TCR reconstitution system. J Exp Med. 2002;195:991–1001. doi: 10.1084/jem.20010809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Soto-Pantoja DR, Ridnour LA, Wink DA, Roberts DD. Blockade of CD47 increases survival of mice exposed to lethal total body irradiation. Sci Rep. 2013;3:1038. doi: 10.1038/srep01038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chou J, Voong LN, Mortales CL, Towlerton AM, Pollack SM, Chen X, et al. Epigenetic modulation to enable antigen-specific T-cell therapy of colorectal cancer. J Immunother. 2012;35:131–141. doi: 10.1097/CJI.0b013e31824300c7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Henle AM, Erskine CL, Benson LM, Clynes R, Knutson KL. Enzymatic discovery of a HER-2/neu epitope that generates cross-reactive T cells. J Immunol. 2013;190:479–488. doi: 10.4049/jimmunol.1201264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Peper JK, Schuster H, Loffler MW, Schmid-Horch B, Rammensee HG, Stevanovic S. An impedance-based cytotoxicity assay for real-time and label-free assessment of T-cell-mediated killing of adherent cells. J Immunol Methods. 2014;405:192–198. doi: 10.1016/j.jim.2014.01.012. [DOI] [PubMed] [Google Scholar]
- 38.Zhao XW, van Beek EM, Schornagel K, Van der Maaden H, Van Houdt M, Otten MA, et al. CD47-signal regulatory protein-alpha (SIRPalpha) interactions form a barrier for antibody-mediated tumor cell destruction. Proc Natl Acad Sci U S A. 2011;108:18342–18347. doi: 10.1073/pnas.1106550108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jaiswal S, Chao MP, Majeti R, Weissman IL. Macrophages as mediators of tumor immunosurveillance. Trends Immunol. 2010;31:212–219. doi: 10.1016/j.it.2010.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tsubaki K, Kuriyama T, Shimizu H, Matsuyama S, Yokota S, Takahashi K, et al. Detection and analysis of IgG antibodies in sera of patient with atopic dermatitis (AD) to individual wheat polypeptides by immunoblotting techniques. Arerugi. 1991;40:521–528. [PubMed] [Google Scholar]
- 41.Kaur S, Chang T, Singh SP, Lim L, Mannan P, Garfield SH, Pendrak ML, Soto-Pantoja DR, Rosenberg AZ, Jin S, Roberts DD. CD47 signaling regulates the immunosuppressive activity of VEGF in T cells. J Immunol. doi: 10.4049/jimmunol.1303116. (in press). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Fogh J, Giovanella BC. The Nude mouse in experimental and clinical research. New York: Academic Press; 1978. [Google Scholar]
- 43.Van VQ, Raymond M, Baba N, Rubio M, Wakahara K, Susin SA, et al. CD47(high) expression on CD4 effectors identifies functional long-lived memory T cell progenitors. J Immunol. 2012;188:4249–4255. doi: 10.4049/jimmunol.1102702. [DOI] [PubMed] [Google Scholar]
- 44.Costa NL, Alencar Rde C, Valadares MC, Silva TA, Mendonca EF, Batista AC. The clinicopathological significance of the expression of Granzyme B in oral squamous cell carcinoma. Oral Oncol. 2010;46:185–189. doi: 10.1016/j.oraloncology.2009.11.016. [DOI] [PubMed] [Google Scholar]
- 45.Heibein JA, Goping IS, Barry M, Pinkoski MJ, Shore GC, Green DR, et al. Granzyme B-mediated cytochrome c release is regulated by the Bcl-2 family members bid and Bax. J Exp Med. 2000;192:1391–1402. doi: 10.1084/jem.192.10.1391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vermeer DW, Spanos WC, Vermeer PD, Bruns AM, Lee KM, Lee JH. Radiation-induced loss of cell surface CD47 enhances immune-mediated clearance of human papillomavirus-positive cancer. Int J Cancer. 2013;133:120–129. doi: 10.1002/ijc.28015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kim D, Wang J, Willingham SB, Martin R, Wernig G, Weissman IL. Anti-CD47 antibodies promote phagocytosis and inhibit the growth of human myeloma cells. Leukemia. 2012 doi: 10.1038/leu.2012.141. [DOI] [PubMed] [Google Scholar]
- 48.Zhao X, Kong D, Jozsef G, Chang J, Wong EK, Formenti SC, et al. Automated beam placement for breast radiotherapy using a support vector machine based algorithm. Med Phys. 2012;39:2536–2543. doi: 10.1118/1.3700736. [DOI] [PubMed] [Google Scholar]
- 49.Isenberg JS, Ridnour LA, Dimitry J, Frazier WA, Wink DA, Roberts DD. CD47 is necessary for inhibition of nitric oxide-stimulated vascular cell responses by thrombospondin-1. J Biol Chem. 2006;281:26069–26080. doi: 10.1074/jbc.M605040200. [DOI] [PubMed] [Google Scholar]
- 50.Isenberg JS, Maxhimer JB, Hyodo F, Pendrak ML, Ridnour LA, DeGraff WG, et al. Thrombospondin-1 and CD47 limit cell and tissue survival of radiation injury. Am J Pathol. 2008;173:1100–1112. doi: 10.2353/ajpath.2008.080237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Soto-Pantoja DR, Isenberg JS, Roberts DD. Therapeutic Targeting of CD47 to Modulate Tissue Responses to Ischemia and Radiation. J Genet Syndr Gene Ther. 2011;2 doi: 10.4172/2157-7412.1000105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Demaria S, Formenti SC. Radiation as an immunological adjuvant: current evidence on dose and fractionation. Front Oncol. 2012;2:153. doi: 10.3389/fonc.2012.00153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mittal R, Gonzalez-Gomez I, Prasadarao NV. Escherichia coli K1 promotes the ligation of CD47 with thrombospondin-1 to prevent the maturation of dendritic cells in the pathogenesis of neonatal meningitis. J Immunol. 2010;185:2998–3006. doi: 10.4049/jimmunol.1001296. [DOI] [PubMed] [Google Scholar]
- 54.Fortin G, Raymond M, Van VQ, Rubio M, Gautier P, Sarfati M, et al. A role for CD47 in the development of experimental colitis mediated by SIRPalpha+CD103− dendritic cells. J Exp Med. 2009;206:1995–2011. doi: 10.1084/jem.20082805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Iversen PL, Arora V, Acker AJ, Mason DH, Devi GR. Efficacy of antisense morpholino oligomer targeted to c-myc in prostate cancer xenograft murine model and a Phase I safety study in humans. Clin Cancer Res. 2003;9:2510–2519. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


