Abstract
OBJECTIVES:
Despite its rising popularity, reports on the use of preoperative imatinib mesylate (IM) in patients with advanced gastrointestinal stromal tumor (GIST) are limited. This study aims to explore the clinical efficacy of preoperative IM in patients with primarily unresectable or metastatic/recurrent GIST.
METHODS:
Between September 2009 and February 2014, patients with primarily unresectable or metastatic/recurrent GIST treated by a single medical team were recruited and considered for preoperative IM therapy. Re-examination was conducted regularly and abdominal enhanced CT data, blood biochemistry and responses to IM were recorded.
RESULTS:
A total of 18 patients were enrolled, including 13 with a primary tumor (7 stomach, 3 small bowel, 2 rectal and 1 pelvic tumor) and 5 with recurrent or metastatic GIST (2 with liver metastasis, 2 with anastomotic recurrence and 1 with pelvic GIST). The median follow-up time was 9.5 months (range of 3-63). The median tumor sizes before and after initiation of IM treatment were 9.1 cm and 6.0 cm (p = 0.003) based on the CT findings, respectively. All patients showed a decrease in tumor burden and the median tumor size reduction was 35%. Sixteen of the 18 patients showed a partial response to IM and two possessed stable disease. Nine of the 18 patients (50%) underwent surgical resection of primary or metastatic/recurrent tumors, with a median of 7 months of IM therapy. One case each of multivisceral resection and tumor recurrence were noted.
CONCLUSIONS:
IM as a preoperative therapy is feasible and safe for unresectable or metastatic/recurrent GIST that can effectively decrease tumor size, facilitating resection.
Keywords: Gastrointestinal Stromal Tumor, Imatinib Mesylate, Preoperative Therapy
INTRODUCTION
Currently, the standard treatment for gastrointestinal stromal tumor (GIST) is radical surgery aimed at obtaining negative microscopic margins 1. However, GIST patients are at risk of recurrence and approximately 40-50% of those who undergo radical resections will develop recurrence or metastasis 2. In some patients, the lesion cannot be completely removed, which has been shown to be partially due to specific anatomic locations (esophagogastric junction, duodenum, or low rectum) and huge tumor diameters 3-4. Treatment of these patients can only be completed after extensive procedures are performed that are associated with increased risks for postoperative complications and mortality.
As understanding of the molecular pathogenesis of GIST has increased, imatinib mesylate (IM) (Glivec®/Gleevec®, Novartis Pharma AG, Basel, Switzerland) has emerged as a targeted therapy that represents the first effective, globally approved, nonsurgical treatment for GIST. Previously published research has suggested that IM considerably improves the prognosis of patients with locally advanced and metastatic GIST 5-6. A large, non-blinded trial of patients with unresectable or metastatic GIST treated with IM demonstrated a median time to progression of 24 months and a median overall survival time of 57 months 7. In addition, recent reports have suggested that preoperative IM use can increase the resectability rate and reduce procedure-related mortality, thereby improving the survival of GIST patients 8-9. However, few rigorous studies on preoperative IM therapy in GIST patients exist to date, the majority of which are retrospective.
Therefore, the purpose of present study was to prospectively analyze the clinical efficacy of preoperative IM therapy administered at a single institution in Chinese patients with unresectable or metastatic/recurrent GIST with the goal of standardizing the preoperative use of IM in the future.
MATERIALS AND METHODS
Patient selection
All included patients were treated by the same medical team at a single institution (Department of Gastrointestinal Surgery, West China Hospital, Sichuan University, China) between September 2009 and February 2014.
The inclusion criteria were as follows:  patients with primarily unresectable or metastatic/recurrent GIST (unresectable tumors were defined as being huge in size or having already infiltrated the adjacent tissues and organs with unclear boundaries, according to radiological findings);
patients with primarily unresectable or metastatic/recurrent GIST (unresectable tumors were defined as being huge in size or having already infiltrated the adjacent tissues and organs with unclear boundaries, according to radiological findings);  GIST that was confirmed by immunohistochemical staining and/or KIT and platelet-derived growth factor receptor A (PDGFRA) mutation detection (samples were acquired by endoscopic ultrasound-guided fine-needle aspiration biopsy or needle biopsy guided by abdominal CT 10; and
GIST that was confirmed by immunohistochemical staining and/or KIT and platelet-derived growth factor receptor A (PDGFRA) mutation detection (samples were acquired by endoscopic ultrasound-guided fine-needle aspiration biopsy or needle biopsy guided by abdominal CT 10; and  patients with no history of taking IM who were over eighteen of age. The exclusion criteria were as follows:
patients with no history of taking IM who were over eighteen of age. The exclusion criteria were as follows:  patients with major contraindications to IM therapy (such as allergies);
patients with major contraindications to IM therapy (such as allergies);  pregnant or lactating women; and
pregnant or lactating women; and  absence of additional concurrent malignant tumors.
absence of additional concurrent malignant tumors.
IM treatment and surgery
All of the patients signed informed consent before treatment. The subjects generally received systemic therapy at a dosage of 400 mg/d. Upon disease progression, the dosage was to 600 mg or higher per day or surgical treatment was indicated. Clinical efficacy and drug toxicity were assessed biweekly during the first month of treatment and then each month the next three months. Thereafter, these parameters were assessed every three months through outpatient follow-up office visits or occasionally via email. Patient responses were evaluated according to the Choi criteria 11.
All surgical procedures were conducted by the same medical team at the West China Hospital. When disease progression was determined or surgery was deemed feasible by surgeons (namely, when the best response was considered to be obtained in the absence of further improvement as shown by 2 successive CT scans), surgical treatment was performed. The primary surgical aims were to maintain as many residual organ functions as possible and to achieve R0 resection as frequently as possible. To achieve a radical cure, additional debulking surgery was performed in some cases, mainly if the tumor had infiltrated adjacent tissues and organs. Frozen slices of the incisal margin and tumor were routinely collected during surgery.
Patient follow-up
All included patients were mainly followed up on an outpatient basis or through email. The clinical data presented here are up to date through May 2014 and include a median follow-up time of 9.5 months (range of 3-63) from the onset of medication administration. The follow-up assessments included abdominal enhanced CT, whole blood count and classification, evaluations of liver and kidney functions, clinical examination and medication compliance assessment.
Data and statistical analysis
The continuous variables are described using the median and range. The differences between the tumor sizes before and after administration of IM treatment were compared using an independent samples t-test. Because of the limited sample size and short-term follow up, no patient survival analysis was attempted. Statistical analyses were performed using SPSS 17.0 software package (SPSS, Chicago, IL, USA). Differences were considered statistically significant when the p-value was less than 0.05.
RESULTS
Demographic and Clinical Characteristics
As of February 2014, a consecutive series of 18 patients with unresectable or metastatic/recurrent GIST treated by the same medical team at a single institution were enrolled in the present study. The main clinical characteristics of these 18 patients are presented in Tables 1 and 2. Thirteen of the 18 cases included primary tumors (stomach in 7, small bowel in 3, rectum in 2 and pelvis in one case), and 5 possessed recurrent or metastatic GIST (liver metastasis in 2, anastomotic recurrences in 2 and pelvic cavity GIST in one case). In total, 6 patients presented with hepatic metastasis. One patient (no. 4) died due to disease progression at 12 months after initiation of IM treatment and 1 patient (no. 3) experienced disease recurrence during follow-up. The median age of the patients at diagnosis was 48 years (range of 30-87). The male to female ratio was 15∶3. Fourteen genetic mutation results were available (10 mutations were in exon 11, 3 were in exon 9 and 1 was in exon 13) and the genetic mutations in the four remaining patients were unavailable (they all refused to submit to genetic mutation assessments, mainly for economic reasons). Considering the paucity of data available on genetic mutations, no efficacy evaluation associated with genetic mutations was attempted.
Table 1.
-Main clinical characteristic of patients with primary GIST.
| Patient no. | Sex | Age (years) | Primary tumor site | Metastatic location | Operative treatment | Preoperative medication duration (months) | Medication duration for nonsurgical patients (months) | Mutation status |
| 1 | M | 59 | Stomach | None | Yes | 15 | - | Exon 11 |
| 2 | F | 68 | Stomach | None | Yes | 2 | - | Exon 11 |
| 3 | M | 40 | Small bowel | None | Yes | 9 | - | Exon 11 |
| 4 | M | 46 | Stomach | Liver | No | - | 12 | Exon 9 |
| 5 | M | 40 | Pelvis | None | No | - | 57 | Exon 13 |
| 6 | M | 74 | Stomach | Liver | No | - | 6 | Exon 11 |
| 7 | M | 40 | Small bowel | Liver | No | - | 6 | Unknown |
| 8 | M | 70 | Stomach | Liver | No | - | 21 | Exon 11 |
| 9 | F | 51 | Rectum | None | Yes | 4.5 | - | Exon 11 |
| 10 | M | 87 | Stomach | None | No | - | 7 | Unknown |
| 11 | M | 77 | Stomach | None | No | - | 7 | Unknown |
| 12 | M | 55 | Small bowel | None | Yes | 9 | - | Exon 11 |
| 13 | M | 46 | Rectum | None | Yes | 7 | - | Exon 11 |
Table 2.
-Main clinical characteristic of patients with recurrent/metastatic GIST.
| Patient no. | Sex | Age (years) | Previous tumor site | Recurrent/metastatic location | Operative treatment | Preoperative medication duration (months) | Medication duration of nonsurgical patients (months) | Mutation status |
| 14 | M | 45 | Rectum | Anastomosis | No | - | 8 | Unknown |
| 15 | M | 30 | Small bowel | Anastomosis | Yes | 1 | - | Exon 11 |
| 16 | M | 42 | Rectum | Liver | No | - | 35 | Exon 11 |
| 17 | M | 39 | Small bowel | Liver | Yes | 9 | - | Exon 9 |
| 18 | F | 50 | Small bowel | Pelvis | Yes | 1 | - | Exon 9 |
Preoperative IM Treatment
A total of 9 patients (50.0%) underwent surgery after the IM treatment. The median durations of preoperative medication use in the patients that did and did not receive surgical intervention were 7 months (range of 1-15) and 8 months (range of 6-57), respectively. Seventeen patients received a standard dosage of 400 mg daily, with the exception of patient no. 18, who was administered a dosage of 600 mg/d (because tumor recurrence occurred at the dosage of 400 mg/d). The IM dose was also raised to 600 mg/d for patient no. 4 due to a decrease and subsequent increase in tumor size. No other patients discontinued or reduced their IM dosages. No treatment-related serious adverse effects were documented during the follow-up period. The median tumor size before and after initiation of IM treatment (considering the best response obtained or that reported at the end of follow-up) were 9.1 cm (range of 3.0-16.8) and 6.0 cm (range of 2.5-11.1) (p = 0.003) based on the CT findings, respectively. The median tumor size reduction after IM therapy was 35% (range of 1-62%). However, the sizes of the hepatic metastases decreased in six cases, although not significantly (p = 0.614), with a median reduction of 20.1%. No complete responses were observed; however, 16 patients (88.9%) showed a partial response and two (11.1%) possessed stable disease according to the Choi criteria. One patient with a partial response experienced disease progression and another suffered from postoperative tumor recurrence.
Surgery Information
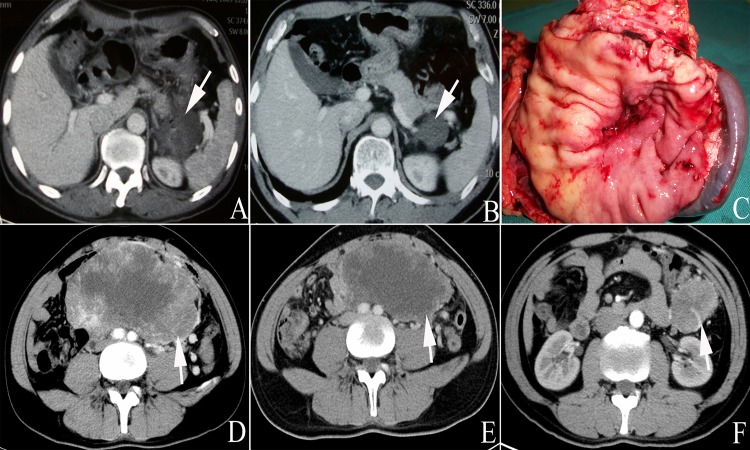
Some patients refused surgery due to a partial radiological response, fear of surgery or other reasons. All decisions to proceed with surgery were made collectively by three senior surgeons from our medical team. In total, 9 patients eventually agreed to receive surgical treatment after the preoperative IM treatment. Two underwent bowel resection (nos. 3 and 12); however, patient no. 3 had a positive margin due to severe tumor invasion. One patient underwent radical distal gastrectomy (no. 2), one received rectal tumor resection and sigmoid colostomy (no. 9), one underwent recurrent tumor resection (no. 14), one received right liver metastasis resection and radiofrequency ablation (no. 16) and one received multi-organ resection and radical proximal gastrectomy with splenectomy (no. 1, as shown in Figures 1A, 1B and 1C). Two additional patients underwent pelvic tumor resection and another 2 received rectal tumor resection. No perioperative deaths or blood transfusions occurred. All operated patients received IM treatment after recovering their gastrointestinal functions and drug tolerance. The median duration of adjuvant therapy with IM was 3 months (range of 2-13) until the deadline for follow-up of May 2014.
Figure 1.
Evaluation of Tumor Imaging
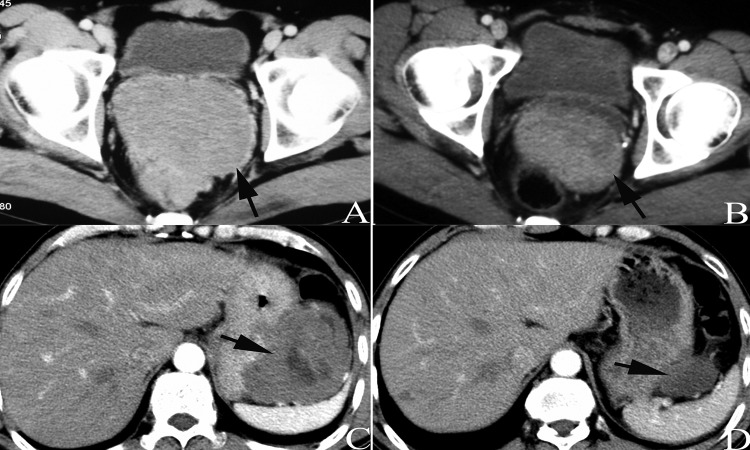
All patients underwent abdominal enhanced CT examination before IM treatment and during subsequent hospital visits. The CT results characterized the metastases and tumor sizes and tumor density was shown to be reduced in most patients, indicating relatively well-defined tumors (Figures 2 and 3). However, two patients experienced very subtle changes bordering on slight increases in tumor size after the IM treatment (nos. 2 and 17). A partial response was noted in the remaining 16 patients, which was defined as a complete or mainly central liquefactive necrosis of the lesion (as depicted in Figure 4). A lesion with a huge mass originating from the jejunum that nearly occupied the entire abdominal cavity was observed in one case; however, the tumor size was reduced by approximately one-third after 9 months of medication (Figures 1D and 1E). Still, this patient experienced postoperative tumor recurrence at 14 months (one month after interruption of IM therapy, Figure 1F).
Figure 2.
This CT scan obtained after medical treatment depicts tumors located in the pelvic cavity (AB) and greater curvature (CD) that significantly decreased in size and density (indicated by arrows). Patient no. 4 (CD) died at 25 months after diagnosis due to disease progression.
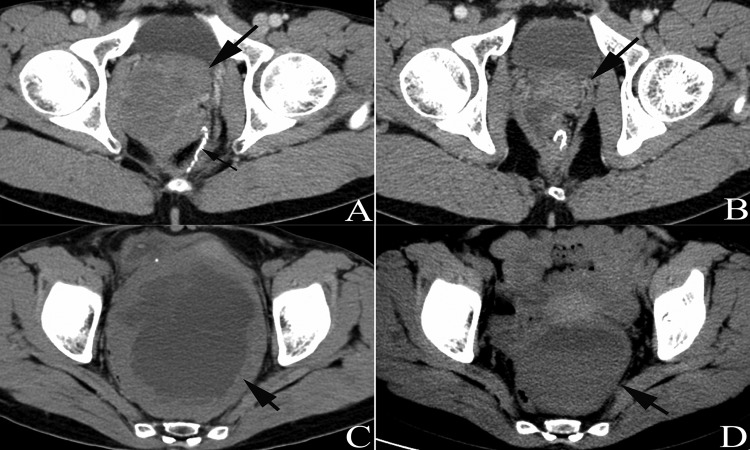
Figure 3.
Patient no. 13 underwent resection of rectal GIST eight years prior (the thin arrow in A shows the anastomotic nail) and the tumor was reduced by 61% after six months of IM treatment at a dosage of 400 mg per day (indicated by thick arrows in AB). CT scans showed a huge mass located in the pelvic cavity, presenting as mixed cystic lesions (C). The tumor size decreased after 4 months of IM treatment (D) and the patient received surgical treatment at 4.5 months after initiation of IM treatment.
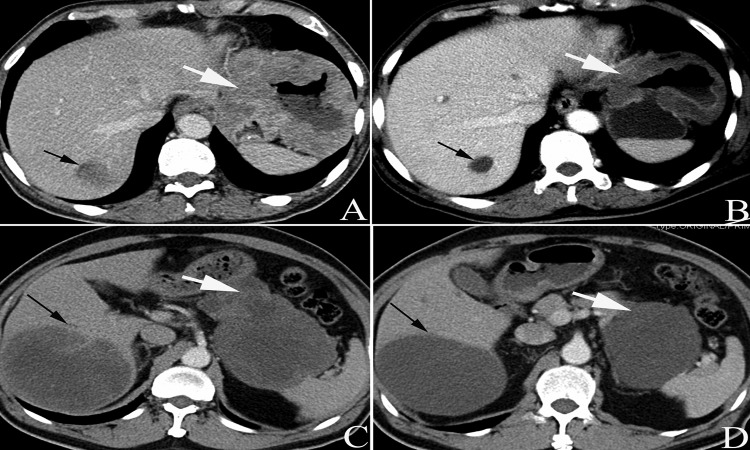
Figure 4.
Based on the CT images, the IM responses were rated as partial. Patients no. 6 (AB) and no. 8 (CD) were first diagnosed with combined liver metastases (black arrows in A and C) and signs of liquefactive necrosis in the metastases and tumors were both observed (white/black arrows in B and D) at two months after initiation of IM treatment.
DISCUSSION
GIST is the most common mesenchymal tumor of the digestive tract 6. This tumor is considered to originate from a common precursor of the interstitial Cajal cells or its precursor cells and are characterized by the presence of activating mutations in KIT and platelet-derived growth factor receptor (PDGFR) 12-13. GIST diagnoses have increased over the past two decades, which is partially due to an increase in relevant information available and improved reclassification guidelines 14. IM, which was originally designed as an inhibitor of Bcr-Abl but was later also demonstrated to effectively inhibit the tyrosine kinase activities of KIT and PDGFR, was shown to markedly increase the recurrence-free and overall survival rates of GIST patients, suggesting that this therapy is effective as a standard of care in the adjuvant treatment of these patients 15.
Preoperative IM therapy for primary unresectable or recurrent/metastatic GIST can shrink the tumor size, increase the resectability rate and achieve a potentially better clinical outcome 8,16. Fiore et al. have shown a median tumor size reduction of 34% after preoperative medication administered for a median duration of 9 months at a standard dose of 400 mg/d 17. These findings are consistent with those of the present study, in which the median tumor size reduction was 35%. Of the tumors that were initially considered to be unresectable, half eventually underwent surgical resection and only one was observed with a positive incisal margin. The use of surgery following IM treatment for patients with advanced GIST remains controversial in this era of molecular targeted therapy because such good therapeutic results were obtained with IM. The vast majority of unresectable GIST is due to extremely large tumor sizes or to excessive tumor infiltration. Huge GIST is typically supplied by multi-source arteries and it sometimes exhibit plexiform-like layers attached to a meager envelope. Uncontrollable bleeding may occur despite careful surgery.
The effects of IM on surgical intervention and outcome after surgical resection in 37 patients with GIST were previously investigated 18. The results showed that approximately 89% of the patients underwent complete resection. In the present study, 8 cases (88.9%) underwent involved the complete removal of the tumor and achievement of a negative incisal margin after preoperative IM treatment in accordance with the aforementioned previous results. Enhanced abdominal CT is routinely used to monitor the responses of GIST to IM 19. Changes in tumor density, vessels and size as shown by CT scan may provide information regarding the biological behaviors and effects of the treatment to a certain extent. Evident changes in tumor size and density were observed in our study and our radiographic assessment showed that approximately 89% of the patients had a partial response to IM, similar to previous findings 20. It is possible that multivisceral resection may occur in huge or locally advanced GIST. Therefore, the NCCN guidelines state that for multivisceral resection huge, poorly positioned or recurrent/metastatic GIST, preoperative IM treatment may be beneficial 21. The separation and removal of tumors to maintain increased organ function is contradictory to the no-tumor principle. If patient quality of life is not significantly influenced, multivisceral resection should be completed within the tumor to achieve a negative margin. In this study, one patient who received 9 months of preoperative medication underwent multivisceral resection (proximal gastrectomy with+ splenectomy). However, although the mass was removed, R0 resection was not achieved due to the huge tumor size.
The optimal duration of preoperative IM treatment is still unknown and under debate. The European Society for Medical Oncology (ESMO) guidelines recommend a period of 6 to 12 months from the initiation of IM treatment until a maximal response is noted in GIST patients and following surgical intervention 22. It is well known that excessive prolongation of preoperative medication may lead to secondary resistance and disease progression. Previous data collected have shown a preoperative IM time range of 7.3 to 12.9 months as well as a significant reduction in tumor size following IM treatment 17,20,23. We performed surgery after a median time of 7 months (range of 1-15). Seven of the 9 operated patients experienced a reduction in tumor size and achieved a maximal response, whereas the two remaining patients showed almost no change after initiation of IM treatment. One patient remained sensitive to IM after 57 months of treatment. Hence, the optimal duration of preoperative therapy may vary from person to person. GIST patients taking preoperative medications should continue tyrosine kinase inhibitor (TKI) adjuvant therapy postoperatively because this practice has been proven to be beneficial 24. Compared with 1 year of adjuvant IM treatment, three years of this treatment have been shown to improve the recurrence-free survival and overall survival rates of GIST patients at a high risk of GIST recurrence 5. Among the 18 patients evaluated in our study, the median time of adjuvant therapy was three months until the deadline for follow-up. All of these patients continued with their use of this drug.
The present study has some limitations. Our series is a small, non-randomized controlled trial, precluding any definitive conclusions. Further studies should include large sample sizes, rigorous sample assessments and multi-center randomized controlled experiments. Our findings suggest that preoperative IM therapy provides clinical benefits for Chinese patients with unresectable or metastatic/recurrent GIST.
In conclusion, application of IM as a preoperative therapy is feasible and safe for primarily unresectable or metastatic/recurrent GISTs in Chinese patients and this treatment can effectively decrease tumor size, facilitating successful tumor resection.
ACKNOWLEDGMENTS
We would like to thank the Department of Gastrointestinal Surgery who generously provided assistance in the collection of data throughout the duration of the study.
Footnotes
No potential conflict of interest was reported.
REFERENCES
- 1.Hohenberger P, Eisenberg B. Role of surgery combined with kinase inhibition in management of gastrointestinal Stromal tumor (GIST) Ann Surg Oncol. 2010;17(10):2585–600. doi: 10.1245/s10434-010-1053-9. [DOI] [PubMed] [Google Scholar]
- 2.Rutkowshi P, Debiec-Rychter M, Ruka W. Gastrointestinal stromal tumors: key to diagnosis and choice of therapy. Mol Diagn Ther. 2008;12(3):131–43. doi: 10.1007/BF03256278. [DOI] [PubMed] [Google Scholar]
- 3. Bosman F T, Carneiro F, Hruban R H, Theise N D, edWHO classification of tumours of the digestive system 4th ed.: IARC, Lyon Press. 201074–6.(ed) [Google Scholar]
- 4.Corless CL, Feltcher JA, Heinrich MC. Biology of gastrointestinal Stromal tumors. J Clin Oncol. 2004;22(18):3813–25. doi: 10.1200/JCO.2004.05.140. [DOI] [PubMed] [Google Scholar]
- 5.Joensuu H, Eriksson M, Sundby Hall K, Hartmann JT, Pink D, Schütte J, et al. One vs Three Years of Adjuvant Imatinib for Operable Gastrointestinal Stromal Tumor A Randomized Trial 2012. JAMA. 2012;307(12):1265–72. doi: 10.1001/jama.2012.347. [DOI] [PubMed] [Google Scholar]
- 6.Joensuu H, Hohenberger P, Corless CL. Gastrointestinal stromal tumour. Lancet. 2013;382(9896):973–83. doi: 10.1016/S0140-6736(13)60106-3. [DOI] [PubMed] [Google Scholar]
- 7.Blanke CD, Demetri GD, von Mehren M, Heinrich MC, Eisenberg B, Fletcher JA, et al. Long-term results from a randomized phase II trial of standard-versus higher-dose imatinib mesylate for patients with unresectable or metastastic gastrointestinal stromal tumors expressing KIT. J Clin Oncol. 2008;26(4):620–5. doi: 10.1200/JCO.2007.13.4403. [DOI] [PubMed] [Google Scholar]
- 8.Wang C, Zheng B, Chen Y, Cao X, Zhang R, Shi Y. Imatinib as preoperative therapy in Chinese patients with recurrent or metastatic GISTs. Chin J Cancer Res. 2013;25(1):63–70. doi: 10.3978/j.issn.1000-9604.2012.12.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rutkowski P, Gronchi A, Hohenberger P, Bonvalot S, Schöffski P, Bauer S, et al. Neoadjuvant Imatinib in Locally Advanced Gastrointestinal Stromal Tumors (GIST): The EORTC STBSG Experience. Ann Surg Oncol. 2013;20(9):2937–43. doi: 10.1245/s10434-013-3013-7. [DOI] [PubMed] [Google Scholar]
- 10.Ando N, Goto H, Niwa Y, Hirooka Y, Ohmiya N, Nagasaka T, et al. The diagnosis of GIST with EUS guide fined needle aspiration with immunohistochemical analysis. Gastrointest Endosc. 2002;55(1):37–43. doi: 10.1067/mge.2002.120323. [DOI] [PubMed] [Google Scholar]
- 11.Choi H. Response evaluation of gastrointestinal stromal tumor. Oncologist. 2008;13(Suppl 2):4–7. doi: 10.1634/theoncologist.13-S2-4. [DOI] [PubMed] [Google Scholar]
- 12.Cassier PA, Ducimetie′re F, Lurkin A, et al. A prospective epidemiological study of new incident GISTs during two consecutive years in Rhône Alpes region: incidence and molecular distribution of GIST in a European region. Br J Cancer. 2010;103(2):165–70. doi: 10.1038/sj.bjc.6605743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liegl-Atzwanger B, Fletcher JA, Flethcher CD. Gastrointestinal stromal tumors. Virchows Arch. 2010;456(2):111–27. doi: 10.1007/s00428-010-0891-y. [DOI] [PubMed] [Google Scholar]
- 14.Perez EA, Livingstone AS, Franceschi D, Rocha-Lima C, Lee DJ, Hodgson N, et al. Current incidence and outcomes of gastrointestinal mesenchymal tumors including gastrointestinal stromal tumors. J Am Coll Surg. 2006;202(4):623–9. doi: 10.1016/j.jamcollsurg.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 15.Essat M, Cooper K. Imatinib as adjuvant therapy for gastrointestinal stromal tumors: a systematic review. Int J Cancer. 2011;128(9):2202–14. doi: 10.1002/ijc.25827. [DOI] [PubMed] [Google Scholar]
- 16.Kitamural Y, Hirota S, Nishida T. Gastrointestinal stromal tumors (GIST): a model for molecule-based diagnosis and treatment of solid tumors. Cancer Sci. 2003;94(4):315–20. doi: 10.1111/j.1349-7006.2003.tb01439.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fiore M, Palassini E, Fumagalli E, Pilotti S, Tamborini E, Stacchiotti S, et al. Preoperative imatinib mesylate for unresectable or locally advanced primary gastrointestinal stromal tumors (GIST) Eur J Surg Oncol. 2009;35(7):739–45. doi: 10.1016/j.ejso.2008.11.005. [DOI] [PubMed] [Google Scholar]
- 18.Goh BK, Chow P, Chuah KL, Yap WM, Wong WK. Pathologic, radiologic and PET scan response of gastrointestinal stromal tumors after neoadjuvant treatment with imatinib mesylate. Eur J Surg Oncol. 2006;32(9):961–3. doi: 10.1016/j.ejso.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 19.Antoch G, Kanja J, Bauer S, et al. Comparison of PET, CT, and dual-modality PET/CT imaging for monitoring of imatinib (STI571) therapy in patients with gastrointestinal stromal tumors. J Nucl Med. 2004;45(3):357–65. [PubMed] [Google Scholar]
- 20.Andtbacka RH, Ng CS, Scaife CL, Cormier JN, Hunt KK, Pisters PW, et al. Surgical resection of gastrointestinal stromal tumors after treatment with imatinib. Ann Surg Oncol. 2007;14(1):14–24. doi: 10.1245/s10434-006-9034-8. [DOI] [PubMed] [Google Scholar]
- 21.Demetri GD, Mehren MV, Antonescu CR, DeMatteo RP, Ganjoo KN, Maki RG, et al. NCCN task force report: update on the management of patients with gastrointestinal stromal tumors. J Natl Compr Canc Netw. 2010;8(Suppl 2):S1–41. doi: 10.6004/jnccn.2010.0116. quiz S42-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Casali PG, Jost L, Reichardt P, Schlemmer M Blay JY; ESMO Guidelines Working Group. Gastrointestinal stromal tumours: ESMO clinical recommendations for diagnosis treatment and follow up. Ann Oncol. 2009;20(Suppl 4):64–7. doi: 10.1093/annonc/mdp131. [DOI] [PubMed] [Google Scholar]
- 23.Blesius A, Cassier PA, Bertucci F, Fayette J, Ray-Coquard I, Bui B, et al. Neoadjuvant imatinib in patients with locally advanced non metastatic GIST in the prospective BFR14 trial. BMC Cancer. 2011;11:72. doi: 10.1186/1471-2407-11-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rutkowski P, Nowecki Z, Nyckowski P, Dziewirski W, Grzesiakowska U, Nasierowska-Guttmejer A, et al. Surgical treatment of patients with initially inoperable and/or metastatic gastrointestinal stromal tumors (GIST) during therapy with imatinib mesylate. J Surg Oncol. 2006;93(4):304–11. doi: 10.1002/jso.20466. [DOI] [PubMed] [Google Scholar]