Summary
Activation of the gap gene hunchback (hb) by the maternal Bicoid gradient is one of the most intensively studied gene regulatory interactions in animal development. Most efforts to understand this process have focused on the classical Bicoid target enhancer located immediately upstream of the P2 promoter [1-12]. However, hb is also regulated by a recently identified distal shadow enhancer as well as a neglected “stripe” enhancer, which mediates expression in both central and posterior regions of cellularizing embryos [13,14]. Here, we employ BAC transgenesis and quantitative imaging methods to investigate the individual contributions of these different enhancers to the dynamic hb expression pattern. These studies reveal that the stripe enhancer is crucial for establishing the definitive border of the anterior Hb expression pattern, just beyond the initial border delineated by Bicoid. Removal of this enhancer impairs dynamic expansion of hb expression, and results in variable cuticular defects in the mesothorax (T2) due to abnormal patterns of segmentation gene expression. The stripe enhancer is subject to extensive regulation by gap repressors, including Kruppel, Knirps, and Hb itself. We propose that this repression helps ensure precision of the anterior Hb border in response to variations in the Bicoid gradient.
Results and Discussion
hunchback (hb) is the premiere gap gene of the segmentation regulatory network. It coordinates the expression of other gap genes, including Kruppel (Kr), knirps (kni), and giant (gt) in central and posterior regions of cellularizing embryos [15,16]. The gap genes encode transcriptional repressors that delineate the borders of pair-rule stripes of gene expression. hb is activated in the anterior half of the precellular embryo, within 20-30 min after the establishment of the Bicoid gradient during nuclear cleavage cycles 9 and 10 (~90 min following fertilization) [3,6,17,18]. This initial hb mRNA transcription pattern exhibits a reasonably sharp on/off border within the presumptive thorax [1-3,5,13]. This border depends on cooperative interactions of Bicoid monomers bound to linked sites in the proximal (“classical”) enhancer (Fig. 1A). However, past studies and recent computational modeling suggest that Bicoid cooperativity is not sufficient to account for this precision in hb expression [4-12].
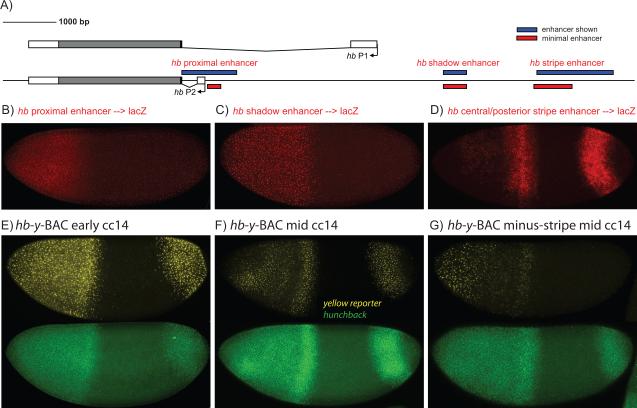
Fig. 1. Summary of Hb Cis-Regulatory DNAs.
(A) The Hb locus contains two promoters, P2 and P1, and three enhancers. The proximal and distal shadow enhancers are targets of the Bicoid gradient, while the stripe enhancer is regulated by gap repressors (see text). (B-D) lacZ antisense RNA in situ hybridization assays with transgenic embryos expressing proximal->lacZ (B), shadow->lacZ (C), or stripe->lacZ (D) transgenes. The proximal and shadow enhancers mediate broad expression in anterior regions, while the stripe enhancer produces central and posterior stripes of expression. (E-G) Transgenic embryos expressing a y-BAC transgene containing a 44 kb genomic DNA encompassing the Hb locus and associated regulatory DNAs. The Hb transcription unit was replaced with the yellow (y) reporter. The wild-type y-BAC transgene exhibits broad anterior expression and a posterior stripe (E,F), whereas a mutagenized y-BAC transgene containing an internal replacement of stripe enhancer sequences with a non-regulatory spacer exhibits reduced expression of the central and posterior stripes (G). The embryos were double stained for yellow nascent transcripts (y-BAC transgenes) (shown in yellow) and endogenous hb (shown in green).
The hb locus contains two promoters, P2 and P1, and three enhancers (Fig. 1A) [1,14]. The “classical” proximal enhancer [1,3] and distal shadow enhancer [13] mediate activation in response to the Bicoid gradient. Expression is also regulated by a third enhancer, the “stripe” enhancer, which is located over 5 kb upstream of P2 [14]. Each of these enhancers was separately attached to a lacZ reporter gene and expressed in transgenic embryos. As shown previously, the Bicoid target enhancers mediate expression in anterior regions of cc12-13 embryos (Fig. 1B,C) [1,2,3,13], whereas the stripe enhancer mediates two stripes of gene expression at later stages, during cc14 (Fig. 1D) [14]. The anterior stripe is located immediately posterior to the initial hb border established by the proximal and distal Bicoid target enhancers (see below).
BAC transgenesis was used to determine the contribution of the stripe enhancer to the complex hb expression pattern. For some of the experiments, we replaced the hb transcription unit with the yellow (y) reporter gene, which contains a large intron permitting quantitative detection of nascent transcripts (see [19]). The resulting BAC mimics the endogenous expression pattern (Fig. 1E,F), including augmented expression at the Hb border. However, removal of the stripe enhancer from an otherwise intact y-BAC transgene leads to diminished expression at this border and in posterior regions (Fig. 1G).
The functional impact of removing the stripe enhancer was investigated by genetic complementation assays. A BAC transgene containing 44 kb of genomic DNA encompassing the entire hb locus and flanking regulatory DNAs fully complements deficiency homozygotes carrying a newly created deletion that cleanly removes the hb transcription unit (see Fig. S1). The resulting adults are fully viable, fertile, and indistinguishable from normal strains. Embryos obtained from these adults exhibit a normal Hb protein gradient, including a sharp border located between eve stripes 2 and 3 (Fig. 2B).
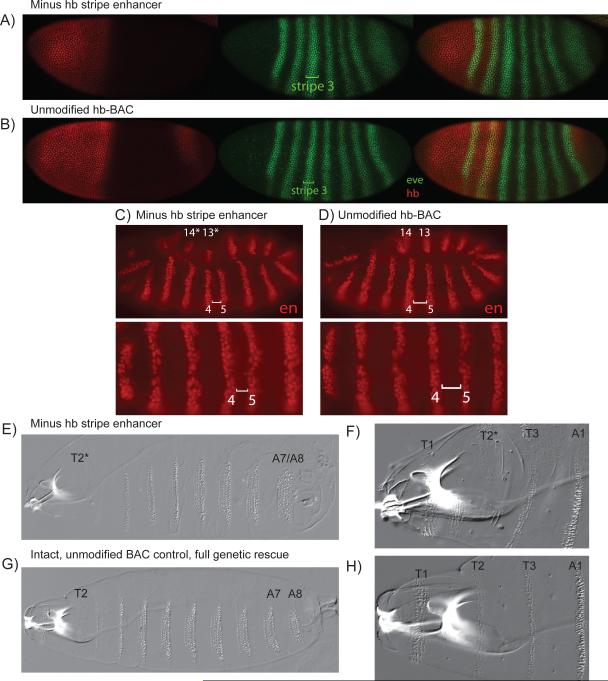
Fig. 2. Developmental consequences of removing the Hb stripe enhancer.
Hb-mutant embryos carrying either a mutant BAC transgene lacking the stripe enhancer (A, C, E, F) or wild-type BAC transgene (B, D, G, H). Both transgenes contain 44 kb of genomic DNA encompassing the Hb transcription unit and flanking sequences. cc14 embryos were stained with antibodies against Hb (red) and even-skipped (green). The wild-type BAC directs a normal Hb expression pattern, and even-skipped (eve) pattern (B). In contrast, the mutant BAC transgene lacking the stripe enhancer exhibits reduced Hb expression in anterior regions, and loss of the posterior stripe (A; compare with B). The eve pattern is also altered, with expanded patterns of stripes 3 and 7 (A). During germband elongation, the wild-type BAC transgene directs normal stripes of engrailed (en) expression (D), whereas the mutant BAC lacking the stripe enhancer exhibits irregular spacing between en stripes 4 and 5 (C). The wild-type BAC transgene also produces completely normal cuticles (G, H), whereas the mutant BAC results in the variable loss of ventral mesothoracic (T2) pattern elements (E, F) and fusion of A7/A8 (E). eve and en expression patterns were diagnostic of genotype, clearly discernible by eye, and this was confirmed by staining for the presence or absence of a labeled balancer.
The Hb BAC transgene lacking the stripe enhancer fails to complement hb−/hb− mutant embryos due to the absence of the posterior hb expression pattern (Fig. 2A), which results in the fusion of the 7th and 8th abdominal segments (Fig. 2E) (see [14]). In addition, the anterior Hb domain lacks the sharp “stripe” at its posterior limit, resulting in an anterior expansion of eve stripe 3 (Fig. 2A; compare with B) since the Hb repressor directly specifies this border [20-23]. There is also a corresponding shift in the position of engrailed stripe 5, which is regulated by eve stripe 3 (Fig. 2D; compare with C) [e.g., 24]. The narrowing of engrailed stripes 4 and 5, due to the anterior shift of stripe 5, correlates with patterning defects in the mesothorax (Fig. 2E,F; compare with G,H).
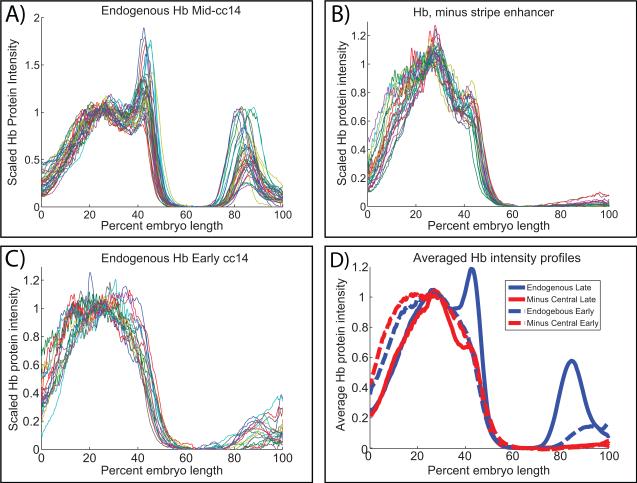
Quantitative measurements indicate significant alterations of the anterior Hb expression pattern upon removal of the stripe enhancer (Fig. 3). There is an anterior shift at the midpoint of the mature pattern, spanning 2-3 cell diameters. This boundary normally occurs at 47.2 % EL. In contrast, removal of the stripe enhancer shifts the boundary to 45.6% EL. The border also exhibits a significant diminishment in slope. Normally, there is a decrease in Hb protein concentration of 20% over 1% EL. Removal of the stripe enhancer diminishes this drop in concentration, with a reduction of just 10% over 1% EL. The most obvious qualitative change in the distribution of Hb protein is seen in regions where there are rapidly diminishing levels of the Bicoid gradient. Normally, the transition from maximum to minimal Hb levels occurs over a region of 10% EL (43% to 53% EL). Removal of the stripe enhancer causes a significant expansion of this transition, to 26% EL (27% to 53% EL). We therefore conclude that the stripe enhancer is essential for shaping the definitive Hb border.
Fig. 3. Quantitative analysis of Hb expression profiles.
Profile of Hb protein late for 31 wild type embryos (A) and for 23 transgenic recue embryos lacking the stripe enhancer (B). The average position of the Hb boundary (as defined by the %EL at which the Hb instensity reaches 0.5) in endogenous embryos is 47.2 % EL. In the minus stripe enhancer constructs the average position of the boundary is shifted anteriorly to 45.6% EL. In both cases the Hb profile sharpens during cc 14 (C). Removal of the stripe enhancer significantly affects the boundary region, as shown by comparing the average Hb profile for endogenous and the rescue constructs (D). A small amount of residual expression remains in central regions in some embryos. This expression may result from an incompletely knocked-out stripe enhancer, but more likely comes from a small amount of activation from the shadow enhancer, which drives some expression in this region in the enhancer->lacZ embryos (and which rapidly disappears).
The preceding studies suggest that the proximal and distal Bicoid target enhancers are not sufficient to establish the definitive Hb border at the onset of segmentation during cc14. Instead, the initial border undergoes a dynamic posterior expansion encompassing several cell diameters due to the action of the stripe enhancer. This enhancer is similar to the eve stripe 3+7 enhancer [20-23]. Both enhancers mediate two stripes, one in central regions and the other in the posterior abdomen, and the two sets of stripes extensively overlap. Previous studies provide a comprehensive model for the specification of eve stripes 3 and 7, whereby the Hb repressor establishes the anterior border of stripe 3 and the posterior border of stripe 7 while the Kni repressor establishes the posterior border of stripe 3 and anterior border of stripe 7 [20-23]. Whole-genome ChIP binding assays [25] and binding site analysis identify numerous Hb and Kni binding sites in the hb stripe enhancer, along with several Kr sites (Fig. S2).
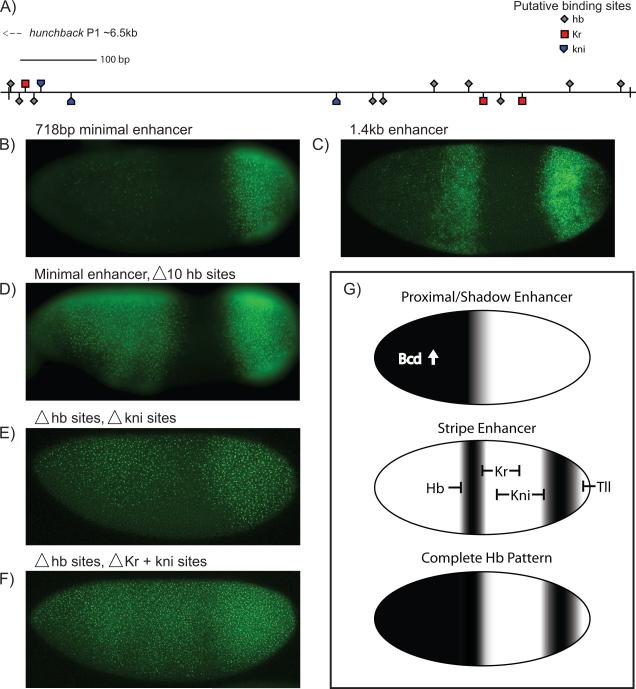
Site-directed mutagenesis was used to examine the function of gap binding sites in the hb stripe enhancer (Fig. 4). Since the full-length, 1.4 kb enhancer contains too many binding sites for systematic mutagenesis (Fig. 1D; Fig. 4C), we identified a 718 bp DNA fragment that mediates weak but consistent expression of both stripes, particularly the posterior stripe (Fig. 4B). Mutagenesis of all 10 Hb binding sites in this minimal enhancer resulted in a striking anterior expansion of the expression pattern (Fig. 4D). This observation suggests that the Hb repressor establishes the anterior border of the central stripe, as seen for eve stripe 3 [22,23]. There is no significant change in the posterior border of the central stripe or the anterior border of the posterior stripe, and repression persists in the presumptive abdomen (Fig. 4D).
Fig. 4. Analysis of the Hb stripe enhancer.
Transgenic embryos expressing different versions of the Hb stripe enhancer were attached to a yellow reporter gene. The 1.4 kb stripe enhancer contains >30 potential Hb sites, at least 8 potential Kr sites, and 6 potential kni sites, and produces strong stripes of expression in central and posterior regions (C). A minimal, 718 bp stripe enhancer produces a weak central stripe and strong posterior stripe (B), putative binding site locations shown in (A). There is augmented expression and an anterior expansion of the anterior stripe>yellow staining pattern upon site-specific mutations in 10 Hb binding sites (D). There is a further expansion of the staining pattern upon mutagenesis of 3 putative kni sites (E) and even more dramatic expansion upon mutagenesis of the 3 putative Kr and 3 putative Kni sites (F); see Fig S2 for details on enhancer structure and specific binding sites. A proposed model is shown in (G). The complete Hb pattern is a composite of three enhancer inputs; the proximal and shadow enhancers rely on activation by anterior Bcd, while the stripe enhancer is instead ubiquitously activated and carved out by gap repressors. The composite boundary is both steeper and shifted posteriorly with the addition of the stripe enhancer's input.
Mutagenesis of the Kni binding sites resulted in expanded expression in the presumptive abdomen (Fig. 4E), similar to that seen for the eve 3+7 enhancer [22,23]. More extensive depression was observed upon mutagenesis of both the Kni and Kr binding sites (Fig. 4F). These results suggest that the Kr and Kni repressors establish the posterior border of the central Hb stripe and the anterior border of the posterior stripe. This depressed pattern is virtually identical to the late hb expression pattern observed in Kr1;kni10 double mutants [22]. The reliance on Kr could explain why the Hb central stripe is shifted anterior of eve stripe 3, which is regulated solely by Kni.
The dynamic regulation of the zygotic Hb expression pattern can be explained by the combinatorial action of the proximal, shadow, and stripe enhancers (summarized in Fig. 4G). The proximal and distal shadow enhancers mediate activation of hb transcription in response to the Bicoid gradient in anterior regions of cc10-13 embryos. The initial border of Hb transcription is rather sharp, but the protein that is synthesized from this early pattern is distributed in a broad and shallow gradient, extending from 30 to 50% EL (see Fig. 3C). During cc14 the stripe enhancer mediates transcription in a domain that extends just beyond the initial Hb border. Gap repressors, including Hb itself, restrict this second wave of zygotic hb transcription to the region when there are rapidly diminishing levels of the Bicoid gradient, in a stripe that encompasses 44-47% EL. The protein produced from the stripe enhancer is distributed in a sharp and steep gradient in the anterior thorax. It has been previously suggested that the steep Hb protein gradient is a direct readout of the broad Bicoid gradient [e.g.,4,6,7]. However, our studies indicate that this is not the case. It is the combination of the Bicoid target enhancers and the hb stripe enhancer that produces the definitive pattern.
It has been proposed that Hb positive autofeedback is an important feature of the dynamic expression pattern [2,10,14]. However, the mutagenesis of the hb stripe enhancer (e.g., Fig. 4) is consistent with past studies suggesting that hb primarily functions as a repressor [20-23]. The only clear-cut example of positive regulation is seen for the eve stripe 2 enhancer. Mutagenesis of the lone Hb-3 binding site results in diminished expression from a minimal enhancer [26]. It was suggested that Hb somehow facilitates neighboring Bicoid activator sites, and we sought to determine whether a similar mechanism might apply to the proximal Bicoid target enhancer. The two Hb binding sites contained in this enhancer were mutagenized, but the resulting fusion gene mediates an expression pattern that is indistinguishable from the normal enhancer (Fig. S3). It is therefore likely that the reduction of the central Hb stripe in hb-/hb- embryos is the indirect consequence of expanded expression of other gap repressors, particularly Kr and Kni (Fig. S4).
The Hb stripe enhancer mediates expression in a central domain spanning 44-47% EL, which coincides with the region exhibiting population variation in the distribution of the Bicoid gradient [e.g., 4]. Despite this variability, the definitive Hb border was shown to be relatively constant among different embryos. Previous studies suggest that the Kr and Kni repressors function in a partially redundant fashion to ensure the reliability of this border [9,10,22]. We have presented evidence for direct interactions of these repressors with the hb stripe enhancer, and suggest that a major function of the enhancer is to “dampen” the variable Bicoid gradient. Indeed, removal of this enhancer from an otherwise normal Hb BAC transgene results in variable patterning defects in the mesothorax, possibly reflecting increased noise in the Hb border.
Experimental Procedures
Drosophila Genetics
A new deletion of the hb coding region was generated using DrosDel collection lines as in [27–29] and Harvard Exelixis lines f07611 and f00586. Flies positive for hb-BACs (carrying w+) were balanced over labeled balancers carrying either TM3hb-lacZ or TM3actin-GFP. The stable full genetic rescue line hb-BAC/hb-BAC; hbdel10/hbdel10 was used as a control and compared to embryos from hb-minus-stripe_enhancer-BAC/hb-minus-stripe_enhancer-BAC; hbdel10/labeled-TM3 balancers. One in four embryos lost the balancer, indicating appropriate genotype, as defined by the absence of the label or by posterior A7/A8 segmental defects (also seen in [14]).
Recombineering and Transgenesis
Recombineering was performed as described previously [13,19,30-34] with modifications described in the Supplemental Experimental Procedures. Primers used for recombineering and screening can be found in Table S1. All constructs were integrated into landing site VK33 on chromosome 3 [29], Bloomington Stock Center number 24871.
Immunohistochemistry and in Situ Hybridization
For antibody staining, embryos were collected in tightly timed ~2-3.5 hour collections for Hb and eve, and 6-12 hour collections for en. We used mouse anti-hb monoclonal antibody 1G10 at a concentration of 1:10; rabbit anti-eve polyclonal #10900 at 1:2000, and mouse anti-en monoclonal 4D9 at 1:40, antibodies kindly provided by Nipam Patel, and commercial AlexaFluor secondaries (Molecular Probes). Embryos were fixed and stained using standard protocols. Fluorescent in situ hybridization was performed as described in [36]. Probes were generated from plasmids made using primers in Table S1 and in vitro transcription. Nuclei were counterstained with DAPI.
Cuticle Preparations
Cuticles were prepared by pipetting late stage embryos/1st instar larvae onto a slide and removing excess fluid. These were mounted in a mixture of glacial acetic acid mixed 1:1 with Hoyer's solution and dried for several days in an oven at 65C for clearing. After 24 hours, cover slips were weighted to flatten the preps. These were imaged on an upright Zeiss Axiophot microscope with brightfield illumination, and grayscale images were later inverted and oversaturated for increased contrast using Adobe Photoshop.
Mutagenesis and Enhancer Testing
Mutagenesis was performed either by using a Stratagene QuikChange Site-Directed mutagenesis kit or by direct synthesis of the 718bp enhancer fragment using Integrated DNA Technologies custom gene synthesis service, followed by subcloning into the enhancer testing vector. This vector is a modified version of the nE2G plasmid described previously [13], used with phiC31 targeted integration after injection into line BSC24871. Primers used can be found in Table S1; specific sites mutagenized are detailed in Supplemental Materials (Figs. S2 and S3).
Imaging and Hb Protein Quantification
High-resolution digital images (1024x1024, 12 bits per pixel) of fixed embryos were obtained on a Carl Zeiss LSM 700 confocal microscope with a Plan Apochromat 20x/0.8 NA objective lens. Embryos were mounted in Prolong Gold (Invitrogen) and placed under a coverslip. The image focal plane of the embryos was chosen at the mid-sagittal plane for protein profile extraction. All images were taken with the same microscope settings. Hb protein profiles were extracted from confocal images of stained embryos by using software routines written in Matlab (See supplement for details). Updated versions of the code used can be found at https://github.com/JacquesBothma. The source codes used to compute and plot the results from this publication are available at https://github.com/JacquesBothma/Hb_Stripe_Enhancer
Supplementary Material
Highlights.
- The Hb embryonic pattern is produced via the action of three enhancers
- The “stripe” enhancer is critical for production of the final, refined Hb border
- This enhancer is regulated via general activation and carved out by gap repressors
- It is unexpectedly auto-repressed, which restricts expression to the boundary region
Acknowledgements
The authors thank Vivek Chopra, Valerie Hilgers, and Alistair Boettiger, and other members of the Levine laboratory, as well as Nipam Patel and Thomas Cline, for helpful discussion and suggestions. We also thank Nipam Patel for providing monoclonal mouse anti-Hb 1G10 and anti-en 4D9 antibodies and Hoyer's solution; we thank Mark Biggin for rabbit polyclonal anti-eve antibody #10900. M.W.P. is the recipient of a National Science Foundation predoctoral fellowship and J.P.B. is the recipient of a Berkeley Fellowship. This work was funded by the National Institutes of Health (grant GM34431 to M.L.).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Driever W, Nüsslein-Volhard C. The bicoid protein is a positive regulator of hunchback transcription in the early Drosophila embryo. Nature. 1989;337:138–43. doi: 10.1038/337138a0. [DOI] [PubMed] [Google Scholar]
- 2.Treisman J, Desplan C. The products of the Drosophila gap genes hunchback and Krüppel bind to the hunchback promoters. Nature. 1989;341:335–7. doi: 10.1038/341335a0. [DOI] [PubMed] [Google Scholar]
- 3.Struhl G, Struhl K, Macdonald PM. The gradient morphogen bicoid is a concentration-dependent transcriptional activator. Cell. 1989;57:1259–73. doi: 10.1016/0092-8674(89)90062-7. [DOI] [PubMed] [Google Scholar]
- 4.Houchmandzadeh B, Wieschaus E, Leibler S. Establishment of developmental precision and proportions in the early Drosophila embryo. Nature. 2002;415:798–802. doi: 10.1038/415798a. [DOI] [PubMed] [Google Scholar]
- 5.Porcher A, Abu-Arish A, Huart S, Roelens B, Fradin C, Dostatni N. The time to measure positional information: maternal hunchback is required for the synchrony of the Bicoid transcriptional response at the onset of zygotic transcription. Development. 2010;137:2795–804. doi: 10.1242/dev.051300. [DOI] [PubMed] [Google Scholar]
- 6.Gregor T, Tank DW, Wieschaus EF, Bialek W. Probing the limits to positional information. Cell. 2007;130:153–64. doi: 10.1016/j.cell.2007.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gregor T, Wieschaus EF, McGregor AP, Bialek W, Tank DW. Stability and nuclear dynamics of the bicoid morphogen gradient. Cell. 2007;130:141–52. doi: 10.1016/j.cell.2007.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.He F, Wen Y, Deng J, Lin X, Lu LJ, Jiao R, Ma J. Probing intrinsic properties of a robust morphogen gradient in Drosophila. Dev Cell. 2008;15:558–67. doi: 10.1016/j.devcel.2008.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Manu, Surkova S, Spirov AV, Gursky VV, Janssens H, Kim A, Radulescu O, Vanario-Alonso CE, Sharp DH, Samsonova M, Reinitz J. Canalization of gene expression in the Drosophila blastoderm by gap gene cross regulation. PLoS Bio. 2009;7:e1000049. doi: 10.1371/journal.pbio.1000049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Holloway DM, Lopes FJP, da Fontoura Costa L, Travençolo BAN, Golyandina N, Usevich K, Spirov AV. Gene expression noise in spatial patterning: hunchback promoter structure affects noise amplitude and distribution in Drosophila segmentation. PLoS Comp Bio. 2011;7:e1001069. doi: 10.1371/journal.pcbi.1001069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lopes FJP, Vieira FMC, Holloway DM, Bisch PM, Spirov AV. Spatial bistability generates hunchback expression sharpness in the Drosophila embryo. PLoS Comp Bio. 2008;4:e1000184. doi: 10.1371/journal.pcbi.1000184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Manu, Surkova S, Spirov AV, Gursky VV, Janssens H, Kim A, Radulescu O, Vanario-Alonso CE, Sharp DH, Samsonova M, Reinitz J. Canalization of gene expression and domain shifts in the Drosophila blastoderm by dynamical attractors. PLoS Comp Bio. 2009;5:e1000303. doi: 10.1371/journal.pcbi.1000303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Perry MW, Boettiger AN, Levine M. Multiple enhancers ensure precision of gap gene-expression patterns in the Drosophila embryo. PNAS. 2011;108:13570–5. doi: 10.1073/pnas.1109873108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Margolis JS, Borowsky ML, Steingrímsson E, Shim CW, Lengyel JA, Posakony JW. Posterior stripe expression of hunchback is driven from two promoters by a common enhancer element. Development. 1995;121:3067–77. doi: 10.1242/dev.121.9.3067. [DOI] [PubMed] [Google Scholar]
- 15.Hülskamp M, Pfeifle C, Tautz D. A morphogenetic gradient of hunchback protein organizes the expression of the gap genes Krüppel and knirps in the early Drosophila embryo. Nature. 1990;346:577–80. doi: 10.1038/346577a0. [DOI] [PubMed] [Google Scholar]
- 16.Struhl G, Johnston P, Lawrence PA. Control of Drosophila body pattern by the hunchback morphogen gradient. Cell. 1992;69:237–249. doi: 10.1016/0092-8674(92)90405-2. [DOI] [PubMed] [Google Scholar]
- 17.Driever W, Nüsslein-Volhard C. A gradient of bicoid protein in Drosophila embryos. Cell. 1988;54:83–93. doi: 10.1016/0092-8674(88)90182-1. [DOI] [PubMed] [Google Scholar]
- 18.Driever W, Nüsslein-Volhard C. The bicoid protein determines position in the Drosophila embryo in a concentration-dependent manner. Cell. 1988;54:95–104. doi: 10.1016/0092-8674(88)90183-3. [DOI] [PubMed] [Google Scholar]
- 19.Perry MW, Boettiger AN, Bothma JP, Levine M. Shadow Enhancers Foster Robustness of Drosophila Gastrulation. Curr Bio. 2010;20:1562–1567. doi: 10.1016/j.cub.2010.07.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Small S, Blair A, Levine M. Regulation of two pair-rule stripes by a single enhancer in the Drosophila embryo. Dev Bio. 1996;175:314–24. doi: 10.1006/dbio.1996.0117. [DOI] [PubMed] [Google Scholar]
- 21.Yan R, Small S, Desplan C, Dearolf CR, Darnell JE. Identification of a Stat gene that functions in Drosophila development. Cell. 1996;84:421–30. doi: 10.1016/s0092-8674(00)81287-8. [DOI] [PubMed] [Google Scholar]
- 22.Clyde DE, Corado MSG, Wu X, Paré A, Papatsenko D, Small S. A self-organizing system of repressor gradients establishes segmental complexity in Drosophila. Nature. 2003;426:849–53. doi: 10.1038/nature02189. [DOI] [PubMed] [Google Scholar]
- 23.Struffi P, Corado M, Kaplan L, Yu D, Rushlow C, Small S. Combinatorial activation and concentration-dependent repression of the Drosophila even skipped stripe 3+7 enhancer. Development. 2011;138:4291–9. doi: 10.1242/dev.065987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Frasch M, Warrior R, Tugwood J, Levine M. Molecular analysis of even-skipped mutants in Drosophila development. Genes and Dev. 1998;2:1824–1838. doi: 10.1101/gad.2.12b.1824. [DOI] [PubMed] [Google Scholar]
- 25.Li X.-yong, et al. Transcription factors bind thousands of active and inactive regions in the Drosophila blastoderm. PLoS Bio. 2008;6:e27. doi: 10.1371/journal.pbio.0060027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Small S, Blair A, Levine M. Regulation of the even-skipped stripe 2 in the Drosophila embryo. EMBO J. 1992;11:4047–4057. doi: 10.1002/j.1460-2075.1992.tb05498.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ryder E, et al. The DrosDel deletion collection: a Drosophila genomewide chromosomal deficiency resource. Genetics. 2007;177:615–29. doi: 10.1534/genetics.107.076216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Thibault ST, et al. A complementary transposon tool kit for Drosophila melanogaster using P and piggyBac. Nat Gen. 2004;36:283–7. doi: 10.1038/ng1314. [DOI] [PubMed] [Google Scholar]
- 29.Parks AL, et al. Systematic generation of high-resolution deletion coverage of the Drosophila melanogaster genome. Nat Gen. 2004;36:288–92. doi: 10.1038/ng1312. [DOI] [PubMed] [Google Scholar]
- 30.Venken KJT, He Y, Hoskins RA, Bellen HJ. P[acman]: a BAC transgenic platform for targeted insertion of large DNA fragments in D. melanogaster. Science. 2006;314:1747–51. doi: 10.1126/science.1134426. [DOI] [PubMed] [Google Scholar]
- 31.Venken KJT, et al. Versatile P[acman] BAC libraries for transgenesis studies in Drosophila melanogaster. Nat Meth. 2009;6:431–4. doi: 10.1038/nmeth.1331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee EC, Yu D, Martinez de Velasco J, Tessarollo L, Swing DA, Court DL, Jenkins NA, Copeland NG. A highly efficient Escherichia coli-based chromosome engineering system adapted for recombinogenic targeting and subcloning of BAC DNA. Genomics. 2001;73:56–65. doi: 10.1006/geno.2000.6451. [DOI] [PubMed] [Google Scholar]
- 33.Liu P, Jenkins NA, Copeland NG. A highly efficient recombineering-based method for generating conditional knockout mutations. Genome Res. 2003;13:476–84. doi: 10.1101/gr.749203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Warming S, Costantino N, Court DL, Jenkins NA, Copeland NG. Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res. 2005;33:e36. doi: 10.1093/nar/gni035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Datsenko KA, Wanner BL. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. PNAS. 2000;97:6640–5. doi: 10.1073/pnas.120163297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kosman D, Mizutani CM, Lemons D, Cox WG, McGinnis W, Bier E. Multiplex detection of RNA expression in Drosophila embryos. Science. 2004;305:846. doi: 10.1126/science.1099247. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.