Abstract
While regulation of the activity of developmental control genes at the transcriptional level as well as by specific miRNA-based degradation are intensively studied, little is known whether general cellular mechanisms controlling mRNA decay may contribute to differential stability of mRNAs of developmental control genes. Here, we investigate whether a mutation in the deadenylation dependent mRNA decay pathway may reveal differential effects on developmental mechanisms, using dopaminergic differentiation in the zebrafish brain as model system. In a zebrafish genetic screen aimed at identifying genes controlling dopaminergic neuron development we isolated the m1061 mutation that selectively caused increased dopaminergic differentiation in the caudal hypothalamus, while other dopaminergic groups were not affected. Positional cloning revealed that m1061 causes a premature stop codon in the cnot8 open reading frame. Cnot8 is a component of the Ccr4-Not complex and displays deadenylase activity, which is required for removal of the poly (A) tail in bulk mRNA turnover. Analyses of expression of developmental regulators indicate that loss of Cnot8 activity results in increased mRNA in situ hybridization signal levels for a subset of developmental control genes. We show that in the area of caudal hypothalamic dopaminergic differentiation, mRNA levels for several components of the FGF signaling pathway, including Fgf3, FGF receptors, and FGF target genes, are increased. Pharmacological inhibition of FGF signaling or a mutation in the fgf3 gene can compensate the gain of caudal hypothalamic dopaminergic neurons in cnot8m1061 mutants, indicating a role for Fgf3 in control of development of this dopaminergic population. The cnot8m1061 mutant phenotype provides an in vivo system to study roles of the Cnot8 deadenylase component of the mRNA decay pathway in vertebrate development. Our data indicate that attenuation of Cnot8 activity differentially affects mRNA levels of developmental control genes.
Introduction
The regulation of mRNA stability is one of the mechanisms for major regulatory transitions during embryonic development, clearing mRNAs characteristic for an early phase of development and facilitating the control of the next developmental phase or differentiation state by newly transcribed mRNAs [1]–[4]. in vivo mRNA metabolism is determined by quantity and time period of transcription, but also by mRNA turnover mechanisms including deadenylation and decapping, defining the half life of an mRNA species during which functional protein can be generated. mRNA turnover is a highly sophisticated and carefully regulated mechanism evolved to establish and maintain the amount of functional protein required by a cell [5]. Recent interest has much focused on the roles of non-coding RNAs in controlling both mRNA translation and mRNA decay in embryogenesis [6], [7]. Whether and how enzymatic mRNA decay control mechanism [8] contribute to specific patterning or differentiation decisions during vertebrate embryogenesis is less well understood.
Deadenylation is thought to be the initial step in bulk mRNA turnover [9]–[11] and is first mediated by the Pab1p-dependent poly (A) nuclease (PAN2-PAN3) complex trimming the poly (A) tail to a length of 60 to 80 nucleotides [12], [13]. Subsequently the Ccr4-Not complex removes the remaining poly (A) tail finally exposing the mRNA to decapping and decay mechanisms. The Ccr4-Not complex is conserved from yeast to human [14]–[17]. In yeast the Ccr4-Not complex is the main deadenylase and comprises 9 core components [18]. Ccr4 associated factor 1 (Caf1) and Carbon catabolite repression factor 4 (Ccr4) are the only subunits of the Ccr4-Not complex involved in 3′ to 5′ deadenylase activity [19], [20]. Caf1 has a second function in associating Ccr4 to the Ccr4-Not complex [21]. cnot8 and cnot7 are homologs of the yeast Caf1 gene in zebrafish, mouse, and human. Instead, in Drosophila only one homolog (POP2) has been identified [15], [17], [19]. In vertebrates the Ccr4-Not complex contains the three proteins Ccr4, Cnot8 and Cnot7, dispatching deadenylase function (reviewed in [11]). Whereas the biochemical roles of cnot8 and cnot7 in the mRNA decay pathway are well studied, and functions have been determined in cell culture (for example, [22]) as well as invertebrate systems, it so far is not well understood whether they may also contribute to differential control of mRNA turnover during development and differentiation in vertebrates. Cnot7 has been shown to be required for normal spermatogenesis in mice [23], but for Cnot8 mouse phenotypes have not been reported.
Dopaminergic (DA) neurons are intensively studied both because of the medical relevance of DA neurons for many neurological diseases, and because DA neurons are an excellent paradigm for differentiation of neurons of one transmitter phenotype in diverse regions of the brain [24]–[26]. Zebrafish have become popular as animal model to study DA differentiation, because of ease of genetic and experimental access and excellent visualization of neuronal types in the transparent embryos and larvae [27]–[34]. Most zebrafish DA groups topologically correspond to those typically found in other vertebrates. Like in mammals, DA neurons develop in the olfactory bulb (OB; mammalian A16) and in the retina (mammalian A17) of zebrafish. The prethalamic group (numbered DC1 in larvae) is homologous to mammalian neurons of the prethalamic zona incerta (A13). All DA clusters located in the posterior tuberculum in zebrafish (groups DC 2, 4, 5, and 6) require the activity of the transcription factor Orthopedia (Otp) and are homologous to the OTP-depending A11 DA in the mammalian brain [35]. The DA groups of the preoptic region and the hypothalamus (groups DC 3 and 7) in zebrafish correlate with mammalian A12 and A14 DA groups. Zebrafish however lack ventral midbrain DA neurons homologous to the mammalian A8-10 group. Instead, an additional group of DA neurons exists in the zebrafish striatum [27], [29], [36]. Most studies in zebrafish have focused on transcriptional control of DA groups [29], especially of the A11-type DA neurons [37]–[42], and on signaling mechanisms [43],[44]. Studies on signaling mechanisms have provided insight into mechanisms controlling the number of DA neurons for the A11-type DA group [42], [44], [45]. In contrast, very little is known about mechanisms that control DA neuron number in other anatomical groups, including the hypothalamic ones.
In a zebrafish genetic screen for mutations affecting expression pattern and level of tyrosine hydroxylase (th) mRNA as marker for DA and noradrenergic (NA) neurons, we identified a mutation in the cnot8 gene. The mutant phenotype is attenuated during early developmental stages by maternally derived functional Cnot8 protein, which gradually decays as development proceeds. cnot8m1061 mutant embryos display increased th transcript levels and increased numbers of DA neurons particularly in the caudal hypothalamus. The hypothalamic DA phenotype may be caused by direct effects on th mRNA stability, or by changes in levels of developmental regulators controlling formation of caudal hypothalamic dopaminergic neurons. We find that fgf3 as well as FGF receptor genes are expressed at higher levels in the caudal hypothalamus in cnot8m1061 mutant embryos. Our data suggest that Cnot8, as Caf1 in yeast [20] and POP2 in Drosophila [17], may have a function in mRNA turnover in zebrafish. Rendering the Cnot8 protein non-functional may result in decreased mRNA decay rates for many but not all developmental regulators, and thus accumulation of those mRNAs. The detailed analysis of genes affected by Cnot8 deficiency in the caudal hypothalamus led us to identify FGF signaling, and specifically Fgf3, as pathway contributing to the specification of the proper number of DA neurons in the caudal hypothalamus.
Results
The zebrafish mutation m1061 causes increased catecholaminergic differentiation marker transcript levels
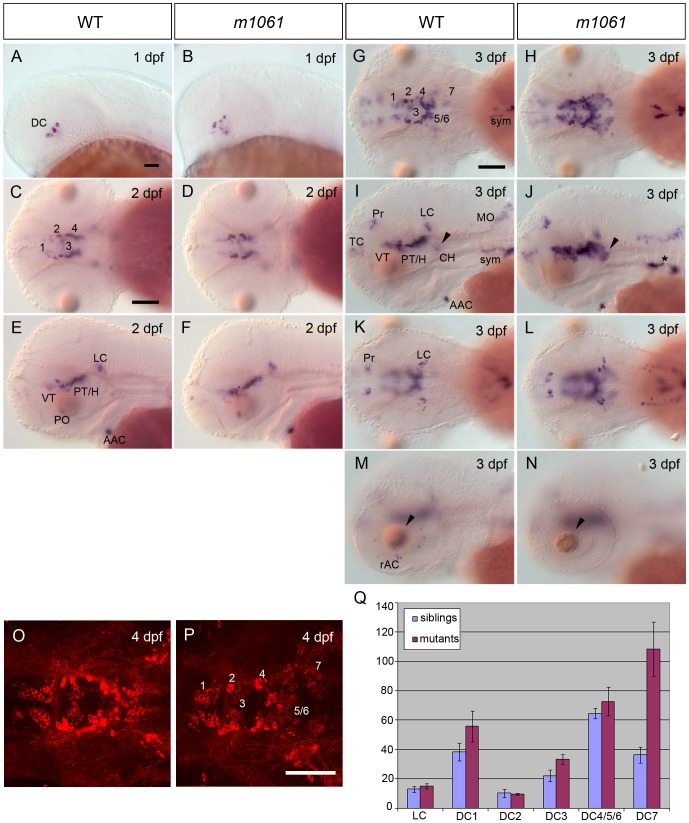
During a systematic genetic screen for mutations affecting development of catecholaminergic (CA) neurons in zebrafish, we identified the m1061 mutation, which was unique in that it was the only mutation isolated which appeared to cause enhanced differentiation of selected CA neuronal groups. In the screen, which was based on detecting tyrosine hydroxylase mRNA expression as marker for CA neurons, the m1061 mutation gave rise to a stronger and expanded whole mount in situ hybridization (WISH) signal for th expression, likely reflecting higher th mRNA levels and potentially also an elevated number of DA and NA neurons. At 1 and 2 days post fertilization (dpf), (Figure 1A–F) m1061 mutants display a th expression pattern indistinguishable from wild-type siblings. At 3 dpf, a stronger in situ hybridization signal for th expression was observed in the diencephalic DA cell clusters of m1061 mutants. This phenotype was especially pronounced in the caudal hypothalamic diencephalic dopaminergic cluster 7 (DC7; Figure 1G–J, arrowhead in I, J). Most other DA groups in the forebrain developed normally up to 4 dpf, with the exception of the pretectal cluster, which showed a minor increase in stain intensity (Figure 1K, L), and the retinal amacrine DA neurons, which appear to be severely reduced in m1061 mutant embryos (Figure 1M, N). We also analyzed NA neuronal clusters, which revealed that the th signal for medulla oblongata (MO) and sympathetic NA neurons appeared to be stronger in m1061 mutant embryos (Figure 1I and asterisk in J). The NA neurons of the locus coeruleus (LC) did not appear to be affected in m1061 mutants. Whole mount immunofluorescence detection of TH was performed and confocal stacks representing the whole ventral diencephalon and rostral hindbrain were analyzed for cell numbers of diencephalic DA cell clusters and the NA cells of the LC at 4 dpf (Figure 1O–Q). As DC 4, 5 and 6 are difficult to distinguish due to close anatomical location, neurons of these groups were combined for analysis. The cell counts were analyzed for statistical significant differences in mutant embryos using the Mann-Whitney test. In m1061 mutant embryos, a strong increase of DA cell numbers was only found for the caudal hypothalamic DC7 DA group (p = 0.008). DA neurons of DC7 showed a more than 2-fold increase in number in m1061 mutants when compared to wild-type siblings (Figure 1Q). Milder increases in cell number were found in DA DC1 (p = 0.032) and 3 (p = 0.008; Figure 2C). The cell numbers in other DA cell clusters like DC2, 4, 5, 6 and the LC were not significantly different between m1061 embryos and controls.
Figure 1. m1061 mutants show increased th expression and increase in number of hypothalamic DA neurons from 3 dpf onwards.
Analysis of th gene expression in cnot8m1061 mutants and WT siblings as indicated in header. (A–F) th transcript levels are not altered in m1061/m1061 mutants at 1 and 2 dpf. Embryos were genotyped by PCR. (G–N) m1061 mutants at 3 dpf display stronger WISH signal indicating increased th mRNA levels in DA groups 1 to 7 (arrowhead) and NA sympathetic cells (asterisk). (K, L) NA locus coeruleus (LC) and DA pretectal cells (Pre) do not display altered th expression levels in m1061 mutant embryos. (M, N) The number of DA retinal amacrine cells (rAC) is reduced in m1061 mutant embryos. Additionally m1061 mutants display a lens defect (arrowheads). Genotypes were inferred by th WISH analysis. (O, P) anti-TH immunohistochemistry of m1061 mutant embryo and WT sibling at 4 dpf document DA neurons in the ventral diencephalon. Data confirm a higher cell number of DC7 DA neurons in the caudal hypothalamus of m1061 mutants. Dorsal view z-projections of partial confocal stacks representing the ventral diencephalon are shown. (A, B, E, F, G, H, M, N) lateral views, anterior at left; (C, D, G, H, K.L, O, P) dorsal views, anterior at left. Scale bars 100 µm in A for A, B; in C for C–F; in G for G–N; in P for O, P. Abbreviations: DC - early diencephalic DA group (DC2, DC4), 1 - ventral thalamic DA group, 2,4,5,6 posterior tubercular and hypothalamic Orthopedia-dependent DA groups, 3 - medial hypothalamic DA group, 7 - caudal hypothalamic DA group, VT - ventral thalamus, PT/H - posterior tuberculum/hypothalamus, PO - preoptic area, AAC - arch associated catecholaminergic neurons/carotid body, sym - sympathetic NA neurons, Pr - pretectum, TC - telencephalon, CH - caudal hypothalamus, LC locus coeruleus, MO - medulla oblongata, rAC - retinal amacrine cells. (Q) Quantification of CA cell numbers in m1061 embryos at 4 dpf. Cell counts of DC1-7 and LC TH-expressing cells. Bars show the average number of CA neurons in five m1061 mutants and five WT sibling embryos. Error bars indicate standard deviation. Significance was evaluated using Mann-Whitney test (see text and Table S1).
Figure 2. The live phenotype and positional cloning of the cnot8m1061 mutation.
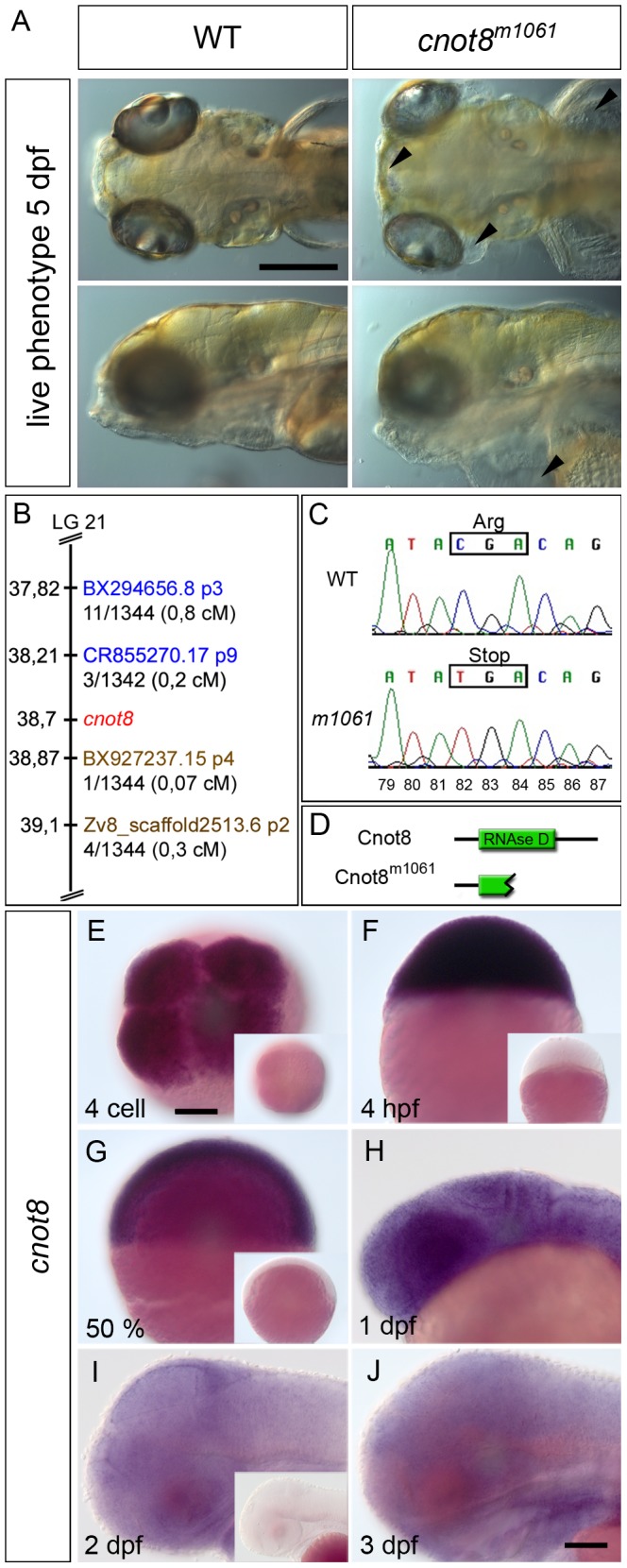
(A) Comparison of cnot8m1061 mutant to wild-type sibling embryos at 5 dpf; cnot8m1061 mutants display edema formation in the eyes and brain (top row, arrowheads) as well as cardiac and yolk sac (bottom row, arrowhead). Top row dorsal view, bottom row lateral view, anterior to the left. Scale bar 100 µm. (B) Scheme of the m1061 genetic interval. Gene and marker positions were obtained from Ensembl Zv8. CR855270.17 p9 and BX927237 p4 define the interval which comprises the genes zgc:77151, MRP L22, gemin5, cnot8, kibra/WWC1, ARL10 and Nop16 (data not shown). Proximal SSLP markers are highlighted in blue. Distal SSLP markers are highlighted in brown. Numbers provided with SSLP markers reflect recombination events identified per number of meioses analyzed. (C) Sequencing of genomic DNA amplified by PCR from individual m1061 homozygous WT and mutant embryos. The C-to-T mutation at bp 82 of the ORF results in the formation of a premature stop codon in m1061 mutant cnot8 ORF. Numbers (79–87) indicate ORF bp position. (D) Zebrafish Cnot8 comprises 286 amino acids and contains an RNAse D domain which is truncated in cnot8m1061 mutants. (E–J) Expression analysis by WISH using cnot8 antisense probes. Sense controls processed exactly in parallel with the antisense reactions are shown as insets in E, F, G and H to evaluate background stain intensities. (E) Maternal mRNA is detected at 4 cell stage (dorsal view). (F) Ubiquitous expression of cnot8 mRNA at 4 hpf (lateral view). (G, H, I) cnot8 continues to be expressed ubiquitously at 50% epiboly, 1, 2 and 3 dpf (lateral views). Scale bars 100 µm.
Given the pronounced changes in CA differentiation, we determined whether living m1061 mutant larvae also develop morphological abnormalities. On the first and second day of development, the overall live morphology of m1061 mutant embryos appear indistinguishable from wild-type siblings (data not shown). From 3 dpf on, m1061 mutant embryos have smaller heads with smaller brain and eyes, as well as impaired branchial arch development. In 5 dpf old m1061 mutant larvae, the eyes are significantly smaller than in wild-type siblings, and edema formation was observed in several tissues including the eyes, brain, heart sack, yolk sack and the intestine (Figure 2A). In addition, m1061 mutants have defective swim bladder formation and reduced heart beat frequency. Further, m1061 mutant larvae from 4 dpf on have a reduced motility in comparison to wild-type sibling larvae (data not shown). Mutant larvae die soon after 5 dpf and cannot be raised to feeding larvae.
The m1061 mutation affects the cnot8 genetic locus
To identify the gene affected by the m1061 mutation, we mapped it using bulked segregant analysis and Simple Sequence Length Polymorphism (SSLP) genetic markers [46], and identified linkage to chromosome 21. Fine mapping of the m1061 mutation required the generation of additional SSLP markers based on published sequence (Materials and Methods). The m1061 critical genetic interval was defined by the proximal marker CR855270.17 p9 (genetic distance of 0.2 cM) and the distal marker BX927237 p4 (genetic distance of 0.07 cM) (Figure 2B). At the approximate position of 38.7 Mb of the linkage group 21 (assembly Zv8; www.ensembl.org), we identified cnot8 and other adjacent genes as candidate genes for the m1061 mutation. The sequencing of cDNA from homozygous mutant m1061 larvae identified a base change at bp 82 within the cnot8 ORF, which resulted in changing an arginine codon to a stop codon, causing premature termination of the peptide after 27 amino acids. This result was confirmed by amplification and sequencing of the corresponding genomic DNA from m1061 mutant and wild-type sibling larvae (Figure 2C). The zebrafish Cnot8 protein consists of 286 amino acids and has one predicted functional domain which exerts exonuclease activity and comprises amino acids 13 to 240 (Figure 2D). The premature stop codon causes the formation of a truncated Cnot8 peptide which lacks most of its RNAse domain and should lack its poly (A) deadenylation function. Thus, the m1061 allele is expected to represent a complete loss of function or null allele of cnot8.
cnot8 has been reported to be broadly expressed (www.ZFIN.org ZDB-IMAGE-050809-54; [47]). To confirm whether cnot8 is globally expressed and not in a tissue specific manner, we analyzed its expression throughout embryonic and early larval stages. cnot8 mRNA was already detected at 4-cell stage (Figure 2E). Since transcription from the zebrafish zygotic genome starts only at midblastula transition (MBT), shortly after 3 hpf [48], this reveals maternally transcribed cnot8 mRNA deposited into the oocyte. Therefore functional maternal mRNA derived Cnot8 protein is likely to enable proper development at early gastrula stages in cnot8m1061 mutant embryos. During later embryonic and early larval stages cnot8 was ubiquitously expressed (Figure 2F–J; insets show sense control WISH).
Based on the maternal and zygotic expression, cnot8m1061 mutant embryos are expected to contain about half the amount of wild-type functional maternal protein, but no functional zygotic mRNA derived Cnot8 protein. Therefore, the mutant phenotype represents the gradual loss of functional maternally derived protein during embryonic stages, as most maternal mRNAs appear to be degraded during blastula and gastrulation stages [1]. Stable maternal derived Cnot8 protein may therefore persist well through the first two to three days of development. Therefore, we investigated whether translational block of cnot8 mRNA by antisense Morpholino oligonucleotides directed against sequence including the start Cnot8 ATG (MOcnot8ATG) would cause a phenotype stronger than cnot8m1061. Injection of a low amount of MOcnot8ATG (1 ng) lead to a more severe morphological phenotype, while 2–8 ng amounts caused early lethality (Figure S1). The morphant phenotype was not further investigated because Morpholino-induced broadly lethal phenotypes are difficult to analyze. We also did not attempt to generate maternal and zygotic mutant embryos based on mutant germline clones, because we reasoned that Cnot deficient germline might be cell-lethal. We also investigated whether microinjection of mRNA encoding the wild-type Cnot8 protein into one-cell stage embryos would rescue the cnot8m1061 mutant phenotype, but did not achieve a significant rescue (data not shown). We interpret this as injected mRNA similar to maternal mRNA being degraded during early development, and not being able to contribute to a rescue of the phenotype on the third day of development and later.
Pattern formation and global organization of the brain appear normal in cnot8m1061 mutant embryos
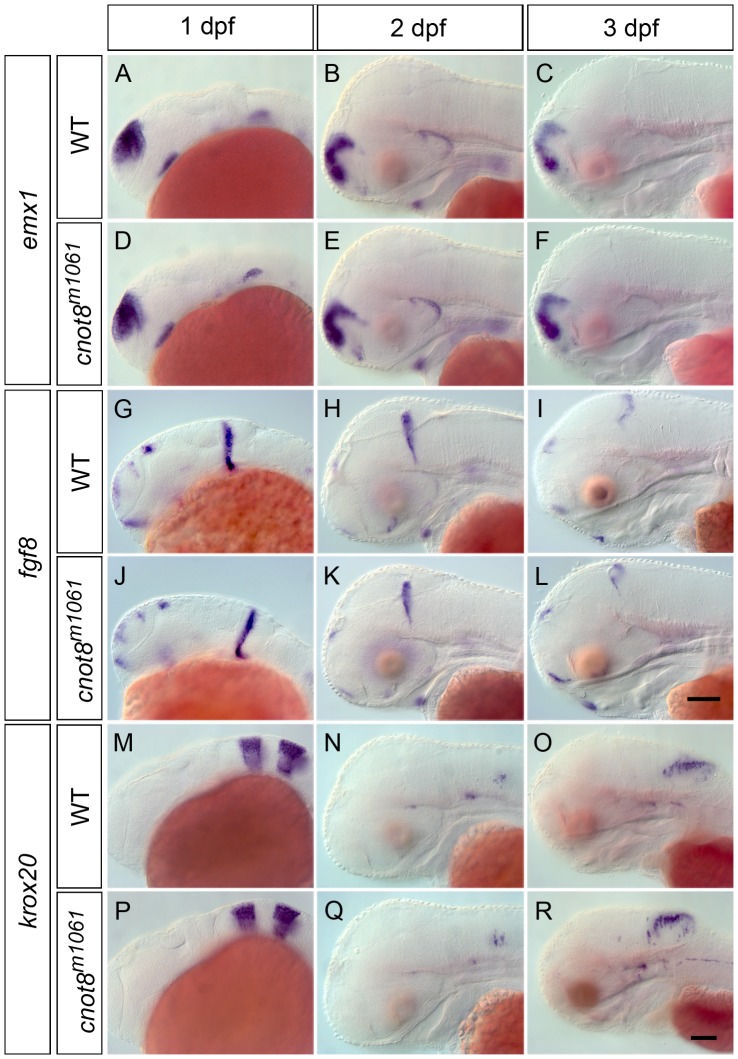
To identify whether pattern formation and morphogenesis of the brain proceed normally during the first three days of development, we investigated the expression of genes involved in pattern formation and regionalization of the brain in cnot8m1061 mutant embryos. To determine whether changes in WISH signal may be significant, we measured mean intensities of WISH signal within defined anatomical domains of marker gene expression in five or more images of control and experimental (mutant) embryos, and analyzed data for significant differences in mean intensities using the Mann-Whitney test (see Methods; all measurements and statistical analyses are documented in Table S1). We used emx1 for telencephalic development [49], krox20/egr2b as a marker for rhombomeres 3 and 5 [50] and fgf8a as a marker for prominent signaling centers like the mid-hindbrain boundary, anterior neural ridge and optic stalk [51]. In 1 and 2 dpf old cnot8m1061 mutant and wild-type sibling embryos the expression pattern of these genes was indistinguishable (Figure 3). In contrast, at 3 dpf a slightly stronger WISH stain was observed for emx1 and krox20/egr2b in cnot8m1061 mutant embryos compared to wild-type, while fgf8a expression appeared not affected. Measurements revealed that the WISH signal was significantly stronger in the telencephalic emx1 domain at 3 dpf (p = 0.032), and at 2 dpf (p = 0.017) and 3 dpf (p = 0.009) for the hindbrain krox20/egr2b domain.
Figure 3. cnot8m1061 mutant embryos are not generally delayed in development.
Gene expression analysis of emx1, fgf8 and krox20/egr2b in cnot8m1061 mutants and wild-type siblings at 1, 2 and 3 dpf. (A–F) emx1, (G–L) fgf8 and (M–R) krox20/egr2b. Embryos were genotyped by PCR. All pictures show lateral views. Scale bars 100 µm.
Given that the most severe dopaminergic phenotype was observed in the caudal hypothalamus, we analyzed expression of transcription factors involved in dopaminergic differentiation and in hypothalamic development. otpa and sim1a have been shown to encode transcription factors required for the specification and differentiation of a subset of DA neurons in the ventral diencephalon in zebrafish [41], [52]. At 2 dpf, otpa is expressed in several ventral diencephalic domains within the dorsal posterior tuberculum, hypothalamus, and ventral pituitary [52]. In cnot8m1061 mutant embryos otpa is expressed in the same spatial pattern but at stronger WISH signal intensity as compared to wild-type siblings (Figure 4A, B; preoptic domain not significantly different, but hindbrain WISH signal significantly stronger, p = 0.008). At 3 dpf expression levels of otpa decline and the corresponding domains in the posterior tuberculum and hypothalamus are very faint in wild-type siblings, while otpa WISH signal in these domains is detected at higher levels in cnot8m1061 mutants (Figure 4C, D; preoptic area p = 0–004). The otpa expression domain in the hindbrain also has a stronger WISH stain in cnot8m1061 mutants embryos compared to wild-type siblings (hindbrain p = 0.002). Expression analysis of sim1a at 2 dpf also revealed a slightly stronger staining in cnot8m1061 mutant embryos in comparison to wild-type siblings, while the expression pattern was normal (Figure 4E, F; posterior tuberculum p = 0.004). We further analyzed nkx2.1a expression as a marker for the hypothalamus [53]. The analysis revealed that at 3 dpf the hypothalamus in cnot8m1061 mutant embryos and wild-type siblings are of equal size (Figure 4G–H). However, we observed a slightly stronger nkx2.1a WISH signal in cnot8m1061 mutant embryos as compared to wild-type siblings (hypothalamus p = 0.008). In summary, it appears that patterning and regionalization of the brain occur normally in cnot8m1061 mutant embryos, while, as judged from WISH stain intensities, the transcript levels of some genes, including the transcription factors sim1a, otpa, and nkx2.1a, appear to be increased.
Figure 4. sim1a, otpa and nkx2.1a expression in cnot8m1061 embryos and WT siblings.
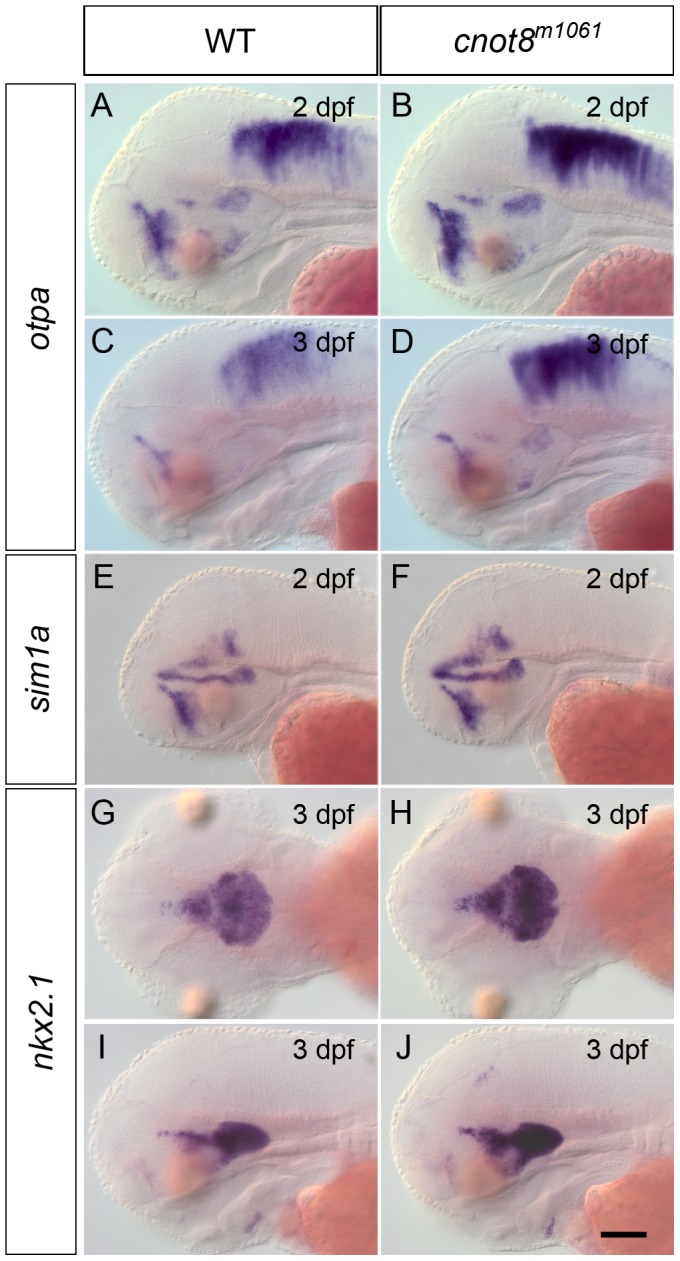
Analysis of otpa (A–D), sim1a (E, F) and nkx2.1a (G–J) gene expression in cnot8m1061 mutant embryos and wild-type siblings, stages as indicated. (A–F, I, J) lateral views. (G, H) dorsal views. Embryos were genotyped by PCR. Scale bar 100 µm.
Selected neuronal cell types are affected in cnot8m1061 mutant embryos
We analyzed whether also other neuronal cell types in addition to DA cells were affected in cnot8m1061 mutants. We focused on neurons in the hypothalamus with regulatory links to dopaminergic specification. The transcription factors Sim1 and Otp have been shown to be required for the development of dopamine, CRH, and Oxytocin secreting neurons in mammals [52], [54]–[56] and zebrafish [41], [52], [57], [58]. In addition we analyzed serotonergic neurons for comparison.
Oxtl/Isotocin is the homolog of oxytocin In zebrafish [59]. oxtl neurons have been shown to form in two distinct areas in the preoptic region of the hypothalamus [59]. The comparison of cnot8m1061 mutant embryos and wild-type siblings and mutants revealed that the oxtl expression pattern is not altered at 3 dpf in mutant embryos, but the WISH signal was slightly darker (Figure 5A–B; p = 0.016).
Figure 5. oxtl, tphd2 and crh gene expression in cnot8m1061 embryos at 3 dpf.
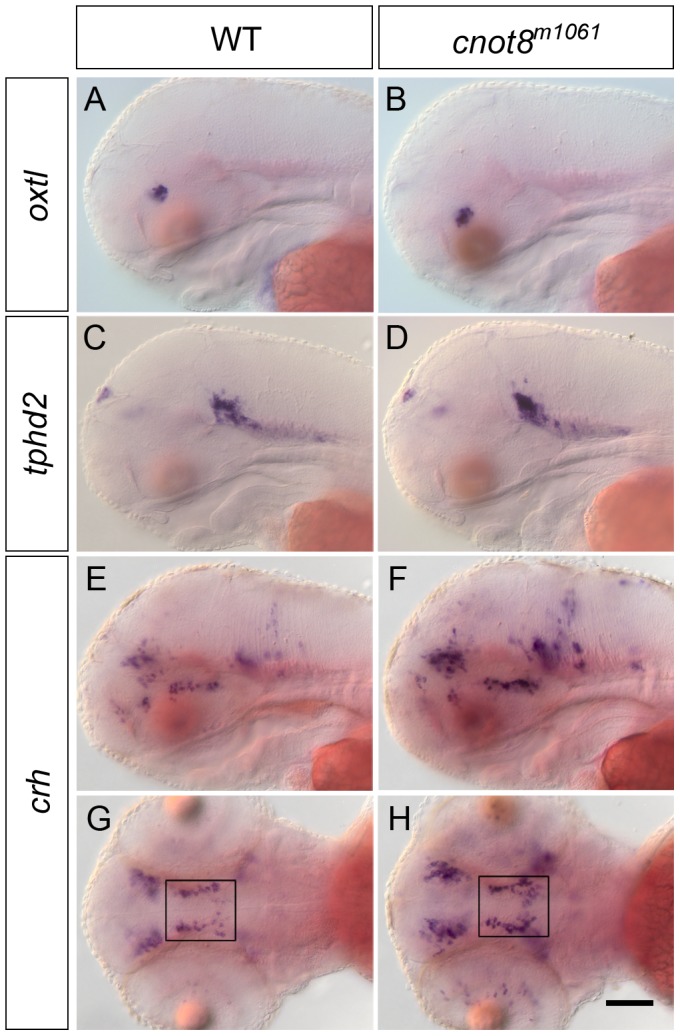
(A, B) oxtl, (C, D) tphd2 and (E–H) crh WISH expression analysis in wild-type siblings and cnot8m1061 mutants. (A–F) lateral views. (G, H) dorsal views. Boxes indicate areas of cell counts. Sibling WT embryos developed on average 34.6 crh neurons and cnot8m1061 mutants developed on average 59.4 crh neurons. Significance was evaluated by Mann-Whitney test (p = 0.008). Embryos were genotyped by PCR. Scale bar 100 µm.
crh expression in early zebrafish brain development has been analyzed in detail in comparison to th and oxtl gene expression [60]. At 3 dpf crh is expressed in the telencephalon, posterior tuberculum, hypothalamus, thalamus, epiphysis, midbrain tegmentum, rostral hindbrain and retina. We performed in situ hybridization to analyze the development of crh expressing neurons in cnot8m1061 mutants at 3 dpf. The analysis revealed that crh WISH stain intensity was increased in cnot8m1061 mutant embryos in all cell clusters (Figure 5E–H; p = 0.004). To address whether the stronger signal may be caused by an increase in mRNA levels or the formation of additional CRH neurons, we counted crh expressing cells in proximity to DA neurons in the posterior tuberculum and hypothalamus (boxes in Figure 5G, H). In this region, we identified approximately twice the number of crh neurons in cnot8m1061 mutant embryos in comparison to wild-type siblings (Figure 5 legend; Table S1; p = 0.008).
Serotonergic neurons are characterized by Tryptophan hydroxylase expression, the rate-limiting enzyme in neurotransmitter synthesis. In zebrafish two genes encoding Tryptophan hydroxylase, tphd1 and tphd2, have been identified and their expression analyzed [61], [62]. tphd2 is a marker for serotonergic neurons in the raphe nucleus and epiphysis. The analysis of tphd1 and tphd2 expression in cnot8m1061 mutant embryos did not reveal any significant differences in expression compared to wild-type siblings. (Figure 5C, D and data not shown).
In summary, we conclude that different neuronal cell types are differentially affected in cnot8m1061 mutant embryos: While serotonergic (tphd2) neurons form normally, CRH and dopaminergic neurons in selected anatomical areas are enhanced in number, and oxtl WISH signal appears enhanced in oxytocinergic neurons.
Increased fgf3 and fgf receptor 1, 2, 3 and 4 expression levels in cnot8m1061 mutants
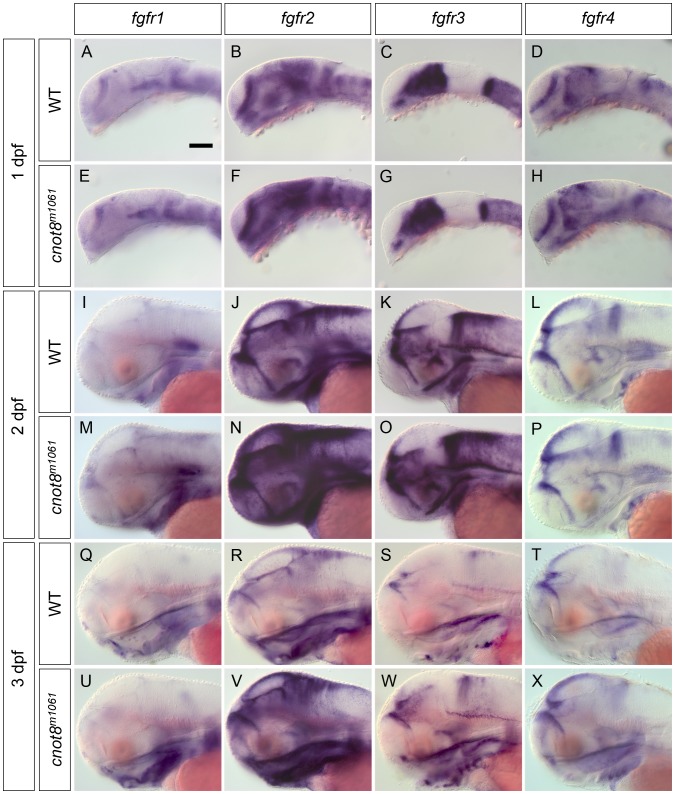
The DA phenotype in the caudal hypothalamus was qualitatively different from other CA groups in that not just the th expression level was increased, but also the number of caudal hypothalamic neurons appeared increased. Therefore signaling pathways controlling DA differentiation in this anatomical region may be affected. FGF signaling namely through Fgf3 has been demonstrated to be important in caudal hypothalamus development [63]–[65]. Expression analysis of fgf3 by WISH and quantification of signal intensity revealed that cnot8m1061 mutants have a mild increase of fgf3 WISH intensity already at 2 dpf (Figure 6A, B), both in the mid-hindbrain boundary expression domain (p = 0.008) and in the caudal hypothalamus (p = 0.008). fgf3 WISH signal continues to be enhanced in both domains at 3 dpf (Figure 6C, D; MHB p = 0.008. cHyp p = 0.004). We also analyzed expression of FGF receptor encoding genes fgfr1, 2, 3 and 4 in cnot8m1061 embryos. While at 1 dpf WISH signal intensities of fgfr1, 2, 3 and 4 are not significantly altered in cnot8m1061 mutants (Figure 7A–H), at 2 dpf fgfr1 and fgfr2 showed increased WISH stain intensities in mutants (Figure 7I–P; fgfr1 ventral hindbrain domain p = 0.008; fgfr2 dorsal forebrain and midbrain p = 0.008), while fgfr3 and fgfr4 WISH signal levels were not altered in comparison to WT siblings. WISH signals of fgfr1, 2, 3, and 4 were up-regulated in cnot8m1061 mutants at 3 dpf (Figure 7Q–X; fgfr1 lower jaw p = 0.031; fgfr2 dorsal forebrain/midbrain p = 0.016; fgfr3 hindbrain p = 0.016; fgfr4 dorsal midbrain p = 0.008).
Figure 6. fgf3 gene expression is increased in cnot8m1061 mutants.
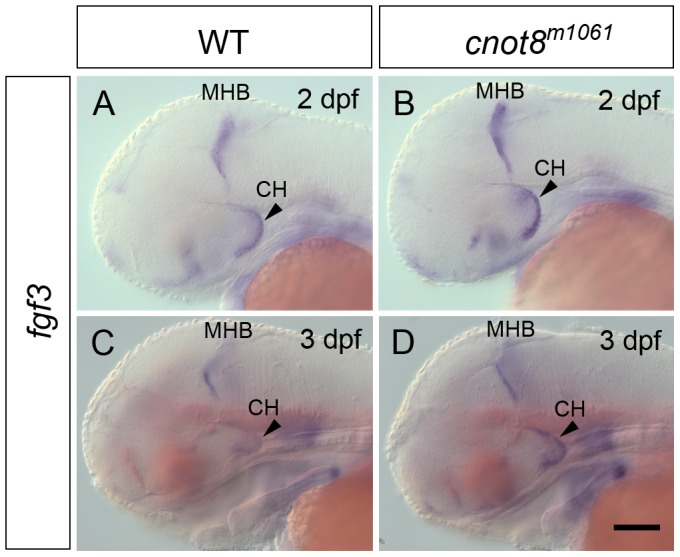
Analysis of fgf3 gene expression in cnot8m1061 mutants and WT siblings 2 dpf (A, B), and 3 dpf (C, D). All pictures show lateral views. Embryos were genotyped by PCR. Scale bar 100 µm.
Figure 7. The cnot8m1061 mutation affects FGF receptor expression levels.
The expression of the four zebrafish FGF receptor genes fgfr1, 2, 3 and 4 was analyzed by WISH in cnot8m1061 mutants and WT siblings at 1, 2 and 3 dpf. All pictures show lateral views. Embryos were genotyped by PCR. Scale bar 100 µm in A for A–X.
FGF target genes pea3 and erm show elevated WISH signals in cnot8m1061 mutants
The increase in fgf3 as well as FGF receptor mRNA WISH signal in cnot8m1061 mutants suggests that FGF signaling levels may also be increased. To address this issue we analyzed expression of the FGF downstream target genes pea3 and erm by WISH in cnot8m1061 embryos. Expression of ETS related protein (erm) was analyzed at 3 dpf (Figure 8A, B), and of polyoma enhancer activator 3 (pea3) at 1, 2 and 3 dpf (Figure 8C–H and data not shown). The experiments revealed that WISH signals for mRNA levels of pea3 and erm were stronger in cnot8m1061 mutants at 2 and 3 dpf (erm MHB, telencephalon, hypothalamus each p = 0.028; pea3 cHyp p = 0.008, lens p = 0.029). pea3 WISH signal was not increased at 1 dpf (data not shown). Increased expression of downstream targets of the FGF signaling pathway suggest increased FGF signaling levels in cnot8m1061 mutants, although due to the nature of the cnot8m1061 mutation in a gene affecting transcript stability, we cannot exclude that the increased pea3 and erm signals may be caused by increased transcript stability.
Figure 8. erm and pea3 expression in cnot8m1061 mutants and WT siblings.
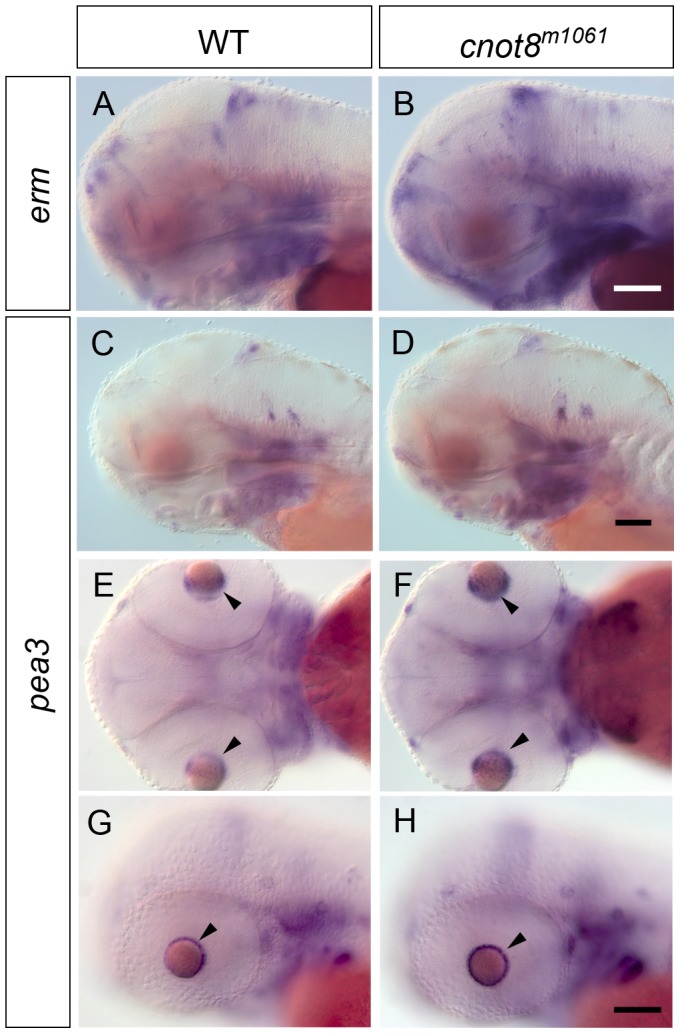
(A, B) erm expression in WT siblings and cnot8m1061 mutants at 3 dpf. (C–H) pea3 expression in WT siblings and cnot8m1061 mutants at (C, D) 3 dpf and (E–H) 2 dpf. (E–G) cnot8m1061 mutants show higher pea3 mRNA levels in cells surrounding the lens in comparison to WT siblings (arrowheads). (A–D, G, H) lateral views. (E, F) dorsal views, anterior at left. Embryos were genotyped by PCR. Scale bars 100 µm in B for (A, B), in D for (C, D) and in H for (E–H).
SU5402 treatment of cnot8m1061 embryos results in reduced numbers of DC7 DA neurons but not DC1-6 DA neurons
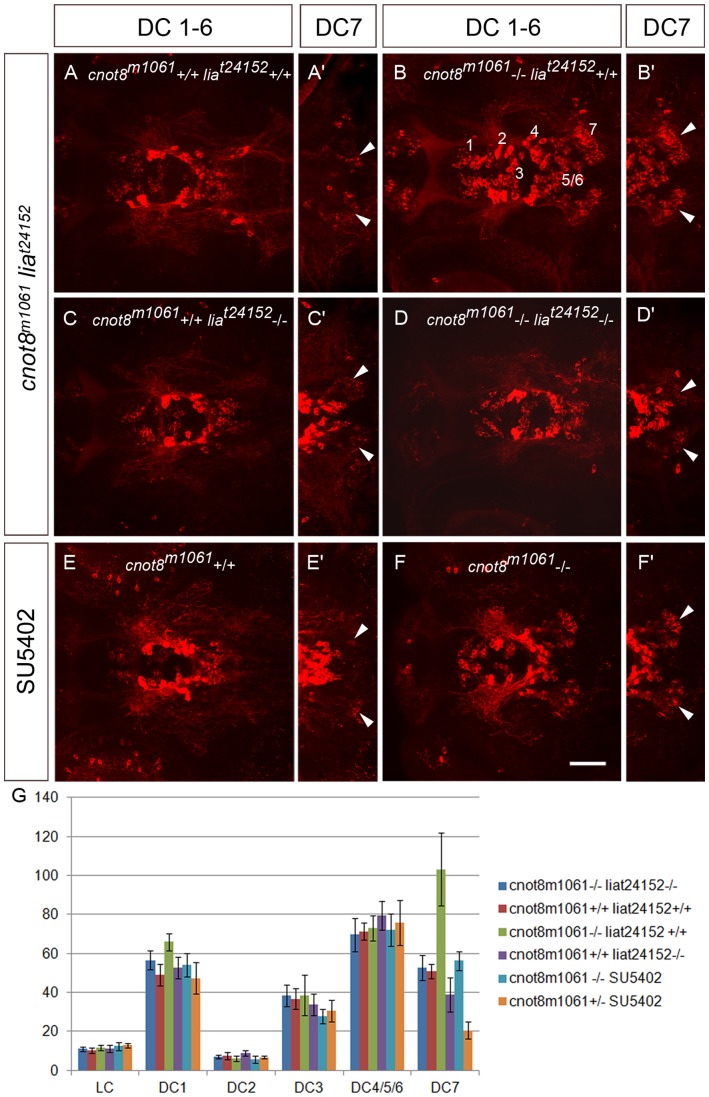
To determine whether enhanced FGF signaling pathway activity may contribute to formation of supernumerary DC7 DA neurons, we treated cnot8m1061 embryos with SU5402, an inhibitor of FGF signaling [66]. SU5402 was applied to embryos for 6 hours from 42 to 48 hpf, the developmental period during which a significant amount of DC7 DA neurons becomes postmitotic [44]. SU5402 treatment did not result in any significant changes in cell numbers of DC1-6 (compare Figure 9A and E). cnot8m1061 mutants had an average of 103.2 DC7 DA neurons, which is a 2-fold increase in DC7 cells in comparison to WT siblings (compare Figure 9A' and B'; p = 0.008). SU5402 treated cnot8m1061 mutant embryos developed an average of 56.2 DC7 neurons, while SU5402 treated WT siblings contained an average of 20.6 DC7 neurons (compare Figure 9B' and F'; p = 0.008). Thus cnot8m1061 mutants treated with SU5402 had only half the amount of DC7 neurons in comparison to non-treated m1061 mutants (p = 0.008). However, cnot8m1061 mutants treated with SU5402 still showed a 2.6 fold increase in cell number in comparison to SU5402 treated WT siblings (compare Figure 9E' and F'; p = 0.008). The significant reduction of DA neurons in both cnot8m1061 and WT embryos by SU5402 treatment by about 50% indicates that FGF signaling may be involved in formation of DC7 DA neurons. The increase in DA neuron number in cnot8m1061 may in part be mediated by enhanced FGF signaling.
Figure 9. fgf3/lia FGF signaling mediates the increase in caudal hypothalamic DC7 DA neurons in cnot8m1061 mutant embryos.
(A–F) Analysis of DA neurons in cnot8m1061 mutant embryos combined with a mutation in the fgf3 locus (liat24152) or pharmacological suppression of FGF signaling by SU5402. Embryos were fixed at 4 dpf, stained by anti TH immunofluorescence and DA neurons documented by confocal microscopy. Shown are Z-projections of confocal stacks representing the ventral diencephalic DA groups 1 to 7 (dorsal views). Scale bar 50 µm. (A–D) DA neurons in WT, cnot8m1061 or liat24152 single mutant, and cnot8m1061, liat24152 double mutant embryos. Double mutant embryos show loss of cnot8m1061-mediated increase of DC7 caudal hypothalamic DA neurons. (E, F) DA neurons in WT and cnot8m1061 mutant embryos treated with SU5402 from 42 to 48 hpf. Inhibition of FGF signaling by SU5402 reduces the number of DC7 caudal hypothalamic DA neurons in cnot8m1061 mutants below WT levels. (G) Quantification of effects on CA neurons by cell counting of forebrain DA neuronal clusters and locus coeruleus NA neurons in genetic and experimental conditions as indicated in the index at right. Each bar shows the average number of CA neurons in five independent embryos for each experimental condition. Error bars indicate standard deviation. Average DC7 cell numbers: cnot8+/+ lia +/+ 50.8 (WT control); cnot8-/- lia +/+ 103.2; cnot8-/- lia -/- 52.6; cnot8+/+ lia -/- (38.8); cnot8+/+ SU5402 20.6; cnot8-/-SU5402 56.2. Significance was evaluated by Mann-Whitney test. The cell count in single mutant cnot8m1061 (-/-)liat24152 (+/+) is significantly different from single mutant cnot8m1061 (+/+) liat24152 (-/-) embryos (p = 0.008). Comparison of cnot8m1061, liat24152 double mutant and WT embryos reveal no significant difference (p = 0.45). For SU5402 treatments, the number of DC7 DA neurons differs significantly between WT controls and SU5402 treated WT (p = 0.008) and between cnot8m1061 and SU5402-treated cnot8m1061 embryos (p = 0.008). For all other catecholaminergic groups, no significant differences were observed when WT was compared to single mutants, double mutants, or SU5402-treated embryos.
Loss of Fgf3 signaling in cnot8m1061 lia(fgf3)t24152 double mutants reduces DC7 cell numbers almost to WT levels
To specifically investigate if Fgf3 signaling is involved in the formation of the DC7 DA cnot8m1061 mutant phenotype we used the lia mutation, which eliminates Fgf3 activity [64], and generated cnot8m1061 lia(fgf3)t24152 double mutants. Embryos were analyzed at 4 dpf by anti-TH immunofluorescene and confocal stacks were recorded. Using the confocal data, DA neurons were counted in embryos of WT, single mutant, and double mutant backgrounds (Figure 9G). In lia(fgf3)t24152 mutant embryos DA neurons of DC1-6 develop normally (Figure 9A and C) indicating that the development of these neurons does not depend on Fgf3 signaling. DC7 neurons are about 20% reduced in number in lia(fgf3)t24152 mutants in comparison to WT siblings (p = 0.024). Most interestingly cnot8m1061 lia(fgf3)t24152 double mutants have on average of 52.6 DC7 DA neurons, and thus a significant reduction can be observed in comparison to cnot8m1061 double mutants having an average of 103.4 DC7 DA neurons (p = 0.008). Loss of Fgf3 signaling in a cnot8m1061 mutant background results in a reduction of DC7 DA neurons almost restoring DA cell numbers counted in WT genetic background (Figure 9G; cell numbers not significantly different; p = 0.45). These findings indicate that Fgf3 signaling, although not strictly required for differentiation of the caudal hypothalamic DA group DC7, has an important role in determining the number of these dopaminergic neurons.
Discussion
In a forward mutagenesis screen we have identified a mutant which eliminates the activity of the zebrafish cnot8 gene. We show that cnot8 is expressed maternally and uniformly zygotically. The cnot8m1061 zygotic mutant phenotype becomes progressively more severe as maternal Cnot8 activity declines. In situ expression analysis reveals that the mRNAs for a subset of neuronal differentiation markers, developmental transcription factors, and signals can be detected with elevated signal intensities in cnot8m1061 mutant embryos. th as marker for dopaminergic neurons is enhanced in several DA neuronal groups, and the number of caudal hypothalamic DA neurons is significantly increased in cnot8m1061 mutants. Analysis of the FGF signaling pathway in cnot8m1061 mutants reveals that stabilization of fgf3 mRNA and FGF receptors may control DA neuron number in the caudal hypothalamus. Using Fgf3 loss-of-function experiments, we confirm that Fgf3 contributes to control of caudal hypothalamic DA neuron number.
Cnot8 is a component of the Ccr4-Not complex which is conserved from yeast to human and considered to be a platform to regulate gene expression at different levels, including bulk mRNA degradation, protein ubiquitination, and transcription [67], [68]. While biochemical and cellular functions of the complex have been extensively characterized in cell lines and invertebrate model organisms, little is known whether Ccr4-Not or its subunits may contribute to tissue specific mRNA turnover or regulatory mechanisms in vertebrates. In mice, Cnot8 has been shown to contribute to proper spermatogenesis [23], revealing a potential for selective Cnot8 activity in development and differentiation. In this work we characterized the zebrafish cnot8m1061 mutant allele which contains a premature stop codon in the ORF after amino acid 27, likely resulting in a null allele. The Cnot8 protein contains only one functional domain which is required for exonuclease activity and a truncation of the protein likely impedes deadenylation of bulk mRNA resulting in accumulation of transcripts. Proper Ccr4-Not complex function is substantial and necessary as only completely deadenylated transcripts are degraded and no longer translated. Our data suggest that Cnot8, similar to Caf1 in Saccharomyces cerevisiae and POP2 in Drosophila melanogaster, has a function in mRNA turnover in zebrafish.
Analysis of cnot8 expression revealed that maternal cnot8 mRNA is deposited in the embryo and cnot8 is expressed zygotically at early zebrafish embryonic and larval stages in a ubiquitous fashion. Thus, Cnot8 may function in all cells of the embryo. cnot8 and other components of the Ccr4-Not complex were also reported to be expressed ubiquitously during all developmental stages at least until 3 dpf in zebrafish (www.zfin.org data base search).
The analysis of expression of several embryonic patterning genes as well as neural differentiation markers by in situ hybridization analysis revealed increased transcript levels in cnot8m1061 mutant embryos for some but not all analyzed genes in embryos beginning from the second day of development. The lack of an early phenotype of cnot8m1061 mutant embryos is likely caused by maternal rescue during the first day of development. At 3 dpf, in cnot8m1061 mutants elevated WISH stain intensities and thus likely mRNA levels were detected for th, crh, krox20, nkx2.1a, sim1, otpa, fgf3, pea3, fgf3 and FGF receptor genes, while gene expression levels of fgf8 and tphd2 were indistinguishable from WT siblings. These observations raised the question why only a subset of mRNA species accumulated in cnot8m1061 mutants. Gene expression levels initially are defined by the rate of transcription. First, the amount of generated mRNA depends on the gene and developmental stage. Second, the half-life of each mRNA species is different as e.g. the removal of longer poly (A) requires more time [13]. Third, mRNA decay involves the function of deadenylating and decapping protein complexes (reviewed in [5]). The recruitment processes of mRNAs targeted for decay to these complexes remain poorly understood [69]. Different mRNA species may display different affinities toward protein complexes involved in mRNA decay. In vertebrates Cnot7 and Cnot8 are paralogs of yeast Caf1 [15], [16], [19]. In yeast, Ccr4 is associated to the Ccr4-Not complex via Caf1 [21], [70], [71]. Cnot7 may compensate loss of Cnot8 function. Addressing the functions of both Caf1 paralogs, Aslam et al. [22] performed siRNA mediated knockdown of Cnot8 and Cnot7 in MCF7 breast cancer cells. Subsequent microarray analysis revealed that single knock down of either component resulted in alteration in expression of few genes only, while combined knockdown caused altered expression in more than two hundred genes. Together, this argues that Cnot7 and Cnot8 may function redundantly in MCF7 cells. This notion is further supported by the finding that Cnot7 knockout mice are viable with a spermatogenesis defect [23]. Expression analysis in mouse neural tissues also revealed differential expression of CNOT7 and CNOT8 in different tissues and downregulation of CNOT8 during differentiation [72]. Our finding of multiple changes in gene expression levels in cnot8m1061 mutants combined with lethality of the mutation indicate that both paralogs, Cnot8 and Cnot7, are not entirely redundant in function during development.
We further investigated potential differential functins of Cnot8 in development using DA neural differentiation as model system. Early differentiating DA neurons of the DC2, 4, and 5 groups, and noradrenergic neurons of the locus coeruleus form during the first 36 hours of development, and appear normal in cnot8m1061 mutant embryos. During these time periods sufficient functional Cnot8 may be present in cnot8m1061 mutants as a result of maternally deposited cnot8 mRNA. In contrast, cell counts of DA cells in the ventral diencephalon and hypothalamus showed that DA cell number was significantly increased in the caudal hypothalamic DC7 group in cnot8m1061 mutants compared to WT siblings. An increase in the number of scored cells was also observed for CRH neurons, but not for serotonergic neurons located adjacent to DC7 DA neurons. Therefore, the effect of Cnot8 depletion is not selective for DA neurons, but also does not globally affect all neuronal types in this region and at this stage. The increase in number of counted cells may be caused by a true increase in number of DC7 DA neurons in mutant embryos, but may alternatively also be explained by increased th mRNA levels due to less decay, which may facilitate detection of newly specified DC7 DA neurons which in wildtype controls express too little th to be detected by the technique. However, based on the fact that selective interference with Fgf3 signaling can compensate the formation of supernumerary DC7 DA neurons, we think that indeed additional DA neurons are observed in cnot8m1061 mutant embryos.
Our findings reveal elevated fgf3 mRNA levels in cnot8m1061 mutant embryos, along with elevated levels of expression for the FGF responsive transcription factor genes pea3 and erm. Given the expression domain of fgf3 in the posterior wall of the caudal hypothalamus, we hypothesized that enhanced Fgf3 signaling may be the cause for formation of supernumerary DC7 DA neurons. To test this hypothesis, we blocked FGF signaling using the FGF receptor inhibitor SU5402. While DC7 DA neurons become postmitotic during an extended developmental period [44], we restricted our analysis to a shorter time window, because permanent global inhibition of FGF signaling affect pattern formation and embryonic survival. However, even when we restricted SU5402 application to 42 to 48 hpf only, we could reduce the wildtype and cnot8m1061 mutant DC7 DA population by about half each. More specifically, when we eliminated Fgf3 activity in lia mutant embryos, we observed almost complete compensation of the supernumerary DC7 DA neurons in cnot8m1061 mutant embryos. These data reveal that Fgf3 signaling contributes to specification of the number of DA neurons in the caudal hypothalamus. However, Fgf3 may not provide the only FGF activity, because residual DC7 DA neurons form in lia/fgf3 homozygous mutant embryos. Other FGFs, including Fgf8a [63], may act redundantly with Fgf3. Fgf8 from the mid-hindbrain boundary has previously been linked to midbrain DA differentiation [73]. An alternative explanation would be that Fgf3 is not directly involved in controlling DC7 DA differentiation, but may rather control the size of the DC7 precursor pool size or proliferation of the precursor pool. A link for Fgf3 signaling to cell cycle control and differentiation in the hypothalamus has also been established for serotonergic neuron differentiation in zebrafish [63]. A regulation of DA neuron number by control of precursor pool size has also been shown for the more rostral posterior tubercular DA neurons in zebrafish, involving WNT signaling [45].
In summary, our data indicate that zebrafish Cnot8 contributes to regulation of proper transcript levels for a subset of developmental control and differentiation genes during embryogenesis, confirming conserved functions of Caf1, Pop2, and Cnot7/8 from yeast to vertebrates. The enhanced activity of Fgf3 signaling in the cnot8 mutant helped us to uncover a role for Fgf3 in controlling the number of DA neurons developing in the zebrafish caudal hypothalamus. The cnot8m1061 mutantion may be a useful model to further study the contribution and mechanisms of the Ccr4-Not complex towards control of transcript levels during development.
Methods
Ethics statement
This study was performed with the approval of the State of Baden-Württemberg Regierungspraesidium Freiburg Animal Protection Authorities in accordance with the German Animal Protection Act under permission number 35-9185.81/G-12/40.
Fish maintenance, strains and genetics
Zebrafish were crossed and eggs maintained under standard conditions at 28,5°C [74]. Embryos were treated with 200 mM phenylthiourea to inhibit melanin pigmentation.
The m1061 allele (Tübingen isolation number t-03-0366) was isolated in an ENU mutagenesis screen in a Tübingen wildtype strain genetic background [41]. Map crosses were set up between a heterozygous carrier of the m1061 mutation and wild-type fish of the AB or HK strain.
The lia(fgf3)t24152 mutant line was previously described [64]. Embryos were genotyped by PCR using the following primers: forward 5′ CAACCGAGAGTGTGAGTTTC 3′ and reverse 5′ CGTCCCTTTCCATTGATGGACAGATA 3′. PCR conditions: 94°C 2 min; 94°C 1 min; 56°C 1 min; 72°C 1 min; 35 cycles. The PCR product had a length of 200 bp. The lia(fgf3)t24152 mutation generates an EcoRV site: PCR product digestion from yielded a 180 bp fragment indicative of the mutant allele.
Mapping and cloning of cnot8m1061
We established a high resolution genetic mapping pannel consisting of 672 F2 embryos representing 1344 meioses from crosses of the original isolate t-03-0366 into AB and HK strain polymorphic strains, providing approximately 0.07 cM resolution. Whole genome scan analysis using SSLP markers [46] linked the m1061 mutation to chromosome 21. To yield additional polymorphic markers for fine mapping, genomic sequences in the critical region were obtained from Ensembl (Zv6, 7 and 8) genome browser and primer pairs for candidate SSLP markers were generated using the Zebrafish SSR search website, Massachusetts General Hospital, Charlestown USA. (http://danio.mgh.harvard.edu/markers/ssr.html). Fine mapping was performed using DNA from single embryos. BX294656.8 3p (forward, 5′-ATGTGCACCTGCAAAAGACA-3′; reverse, 5′-AATCAACCTCGTCATCCTCA-3′); CR855270.17 p9 (forward, 5′-TCATTATGCAGACTACATTTGAAAG-3′; reverse, 5-CCGTCCATTTGTTCATTCCT-3′); BX927237 p4 (forward, 5′-GAATGCAGGCGAATAGAACC-3′; reverse, 5-GCGAGACGCTCTAGGCTAGTT-3′); Zv8_scaffold2513.6 p2 (forward, 5′-TGTTGTTTGTGAGGAACTTAATGA-3′; reverse, 5-TCTCTCTTTTCTCAGCTGTGTTG-3′). Listed SSLP markers were also used to genotype m1061 embryos. The full length cnot8 ORF was amplified from cDNA of 3 dpf cnot8m1061 mutant embryos and wild-type siblings (cnot8 p4 forward, 5′-GTTCCTCTGCCTTCATCATC-3′; reverse, 5′-AACTGCCTCGGTCAACAGAT-3′). PCR conditions: 94°C 2 min; 94°C 1 min; 60°C 1 min, 72°C 2 min, 30 cycles. Partial cnot8 ORF was amplified from genomic DNA confirming the premature stop codon in m1061 mutants (cnot8stop p1 forward, 5′-ATCCTTCAATAATCGCCATGT-3′; reverse, 5′-AACACAAAAGTAAGAAATGCTATTTGG-3′). PCR conditions: 94°C 2 min; 94°C 1 min; 60°C 1 min, 72°C 2 min, 30 cycles.
Genotyping of cnot8m1061 embryos
Following WISH, tail clips for DNA extraction were performed and the tightly linked BX294656.8 3p, CR855270.17 and p9 BX927237 p4 SSLP markers were used for genotyping comparing cnot8m1061 mutant and wild-type embryos respectively.
Plasmids and probes
cnot8 digoxigenin-labeled sense probe was generated by linearizing the pCRIITOPO-cnot8 plasmid with BamHI. cnot8 digoxigenin-labeled antisense probe was generated linearizing the pCRIITOPO-cnot8 plasmid with EcoRV. In addition, the following digoxigenin-labeled antisense probes were used: th [34], fgf8 [51], krox20/egr2b [75], emx1 [49], nkx2.1a [53], otpa [52], sim1a, oxtl, and crh [41], tphd2 [61], fgfr1-4 [76], pea3 [77], erm [78], fgf3 [79].
Whole-mount in situ hybridization and fluorescent immunohistochemistry
Both methods were performed as previously described [80].
Morpholinos
Morpholinos were obtained from Gene Tools LLC. Knock down of maternal and zygotic Cnot8 was achieved by coinjection of 1 ng of both MOcnot8ATG (5′–ATGATGAAGGCAGAGGAACCAATTC–3′) and p53ATG Morpholino at 1 cell stage in wild-type embryos. p53ATG Morpholino was previously described [81].
Cell counts and statistical analysis
Counts of crh and DA cells were performed visually using DIC transmitted light image stacks documenting the regions of the brain in which dopaminergic neurons are located. Cell counts were performed using ImageJ or Zeiss ZEN software. For all cell count analyses (Figures 1, 5, 9), cell numbers of 5 embryos for each experimental condition were determined. The numbers reported in the bar graphs are average numbers determined using the Microsoft Excel "Average" function, and plotted with standard deviation calculated by Microsoft Excel. Statistical analyses were performed using the nonparametric Mann-Whitney test by the statistical analysis software GraphPad Prism version 6.0d for MacOS, GraphPad Software, La Jolla California USA, www.graphpad.com.
Image analysis
Images of WISH gene expression data were analyzed to determine whether significant differences in WISH signal intensities exist when wildtype control and mutant or experimental embryos were compared. To compare WISH signal intensities, control and experimental embryos were fixed and processed together. For mutant and WT embryos, both were processed together in one reaction tube, and genotypes determined by PCR after alkaline phosphatase stain reaction. Thus, WISH and stain procedures were under exactly identical conditions for samples to be compared. For each analysis, control and experimental WISH embryos were photographed in one session under identical imaging conditions (same orientation of embryo, comparable focal plane, same lens, DIC, and light setting on microscope, same exposure setting on ZEISS Axiocam MRc camera utilizing Axiovision software). For control and experimental embryos, typically five to six each were documented (see number of embryos N reported with experiments).
For quantification of stain intensities, images from experimental and control embryos to be compared were all assembled into one composite Photoshop (typically five controls on left side and five experimental embryos on right side) file, merged into one single layer separate from the background layer. Using the Photoshop "Levels" tool composite layers that did not fully use the 8-bit intensity range were adjusted linearly taking great care not to saturate pixels. The composite was flattened, and converted to grayscale (8 bit). The grayscale image was then inverted using the Image - Adjustments - Invert tool. These composite images were saved as TIFF files. TIFF files were opened using ImageJ 1.48o (64bit). For each individual image in the composite, the equivalent anatomical areas of WISH signal were marked using the freehand selection tool, drawn using a graph tablet (Bamboo Fun CTH-461; WACOM). Using the ImageJ Analyze - Measure command, the area as well as mean, minimum and maximum grey values were determined. Data were controlled such that maximum grey values did not exceed 254 (to exclude saturation). Data were transferred into Excel:mac2011.
For statistical analysis, data were transferred to GraphPad Prism version 6.0d for MacOS. Mann-Whitney tests (unpaired non-Gaussian data, nonparametric test) were performed to calculate two-tailed P values. P<0.05 was considered significant.
Supporting Information
Evaluation of Cnot8 ATG Morpholino knockdown phenotype. Analysis of fgfr1 expression at 1 dpf in wildtype embryos injected with cnot8 ATG Morpholino. (A, C) non-injected WT sibling. (B) injection of 1 ng cnot8 ATG Morpholino and 1 ng p53 Morpholino. (D) Injection of 2 ng cnot8 ATG Morpholino and 1 ng p53 Morpholino (A and B) lateral views.
(TIF)
Summary of statistical evaluations of cell count experiments and quantificational analysis of WISH signals for Figure 3 – 9 . The File contains the following sheets: Sheet 1 - Summary of results from image quantifications (green = significant differences using Mann-Whitney test; red = no significant differences). Sheet 2 - Results of Mann-Whitney tests for analysis of stain intensities in Figures 3 to 8. Sheet 3 - Image measurements Figure 3. Sheet 4 - Image measurements Figure 4. Sheet 5 - Image measurements Figure 5. Sheet 6 - Image measurements Figure 6. Sheet 7 - Image measurements Figure 7. Sheet 8 - Image measurements Figure 8. Sheet 9 - Summary of cell counts Figures 1 and 9. Sheet 10 - Statistical analysis of cell counts in Figure 1. Sheet 11 - Cell counts and statistical analysis Figure 5. Sheet 12 - Cell counts and statistical analysis Figure 9.
(XLSX)
Acknowledgments
We thank C. Nüsslein-Volhard, B. Walderich and their laboratory for the mutagenesis screen in which we isolated the m1061 allele, as well as S. Götter for expert zebrafish care. The authors thank Prof. Dr. Matthias Hammerschmidt for kindly providing the lia(fgf3)t24152 line. The authors also thank Dr. Kyo Yamasu (Saitama University, Saitama City, Saitama 338-8570, Japan) for kindly providing the plasmids fgfr1-4 for antisense probe generation. This work was supported by DFG - GRK 843 “Mechanisms of Neuronal Signal Transduction - from Protein to Network” (PK; www.dfg.de), by the EC programs EU-FP6 IP ZF-MODELS (LSHG-CT-2003-503496; http://cordis.europa.eu), EU-FP7 IP ZF-HEALTH (EC Grant Agreement HEALTH-F4-2010-242048 http://cordis.europa.eu), and FP7 IP mesDANEURODEV (Grant number 222999; http://cordis.europa.eu), by GIF 926-183.1/2006, and the Excellence Initiative of the German Federal and State Governments (BIOSS - EXC 294; www.dfg.de).
Funding Statement
This work was supported by DFG - GRK 843 “Mechanisms of Neuronal Signal Transduction - from Protein to Network” (PK), by the EC programs EU-FP6 IP ZF-MODELS, EU-FP7 IP ZF-HEALTH, and FP7 IP mesDANEURODEV, by GIF 926-183.1/2006, and the Excellence Initiative of the German Federal and State Governments (BIOSS - EXC 294).
References
- 1. Schier AF (2007) The Maternal-Zygotic Transition: Death and Birth of RNAs. 316:406–407. [DOI] [PubMed] [Google Scholar]
- 2. Giraldez AJ (2010) microRNAs, the cell's Nepenthe: clearing the past during the maternal-to-zygotic transition and cellular reprogramming. Curr Opin Genet Dev 20:369–375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bashirullah A, Cooperstock RL, Lipshitz HD (2001) Spatial and temporal control of RNA stability. 98:7025–7028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Tadros W, Lipshitz HD (2005) Setting the stage for development: mRNA translation and stability during oocyte maturation and egg activation inDrosophila. 232:593–608. [DOI] [PubMed] [Google Scholar]
- 5. Garneau NL, Wilusz J, Wilusz CJ (2007) The highways and byways of mRNA decay. 8:113–126. [DOI] [PubMed] [Google Scholar]
- 6. Pauli A, Rinn JL, Schier AF (2011) Non-coding RNAs as regulators of embryogenesis. Nat Rev Genet 12:136–149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Huntzinger E, Izaurralde E (2011) Gene silencing by microRNAs: contributions of translational repression and mRNA decay. Nat Rev Genet 12:99–110. [DOI] [PubMed] [Google Scholar]
- 8. Schoenberg DR, Maquat LE (2012) Regulation of cytoplasmic mRNA decay. 13:246–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Fritz DT, Bergman N, Kilpatrick WJ, Wilusz CJ, Wilusz J (2004) Messenger RNA decay in mammalian cells: the exonuclease perspective. 41:265–278. [DOI] [PubMed] [Google Scholar]
- 10. Meyer S, Temme C, Wahle E (2004) Messenger RNA turnover in eukaryotes: pathways and enzymes. 39:197–216. [DOI] [PubMed] [Google Scholar]
- 11. Parker R, Song H (2004) The enzymes and control of eukaryotic mRNA turnover. 11:121–127. [DOI] [PubMed] [Google Scholar]
- 12. Brown CE, Tarun SZ, Boeck R, Sachs AB (1996) PAN3 encodes a subunit of the Pab1p-dependent poly(A) nuclease in Saccharomyces cerevisiae. 16:5744–5753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Yamashita A, Chang T-C, Yamashita Y, Zhu W, Zhong Z, et al. (2005) Concerted action of poly(A) nucleases and decapping enzyme in mammalian mRNA turnover. 12:1054–1063. [DOI] [PubMed] [Google Scholar]
- 14. Albert TK, Lemaire M, van Berkum NL, Gentz R, Collart MA, et al. (2000) Isolation and characterization of human orthologs of yeast CCR4-NOT complex subunits. 28:809–817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Draper MP, Salvadore C, Denis CL (1995) Identification of a mouse protein whose homolog in Saccharomyces cerevisiae is a component of the CCR4 transcriptional regulatory complex. 15:3487–3495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Dupressoir A, Morel AP, Barbot W, Loireau MP, Corbo L, et al. (2001) Identification of four families of yCCR4- and Mg2+-dependent endonuclease-related proteins in higher eukaryotes, and characterization of orthologs of yCCR4 with a conserved leucine-rich repeat essential for hCAF1/hPOP2 binding. 2:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Temme C, Zaessinger S, Meyer S, Simonelig M, Wahle E (2004) A complex containing the CCR4 and CAF1 proteins is involved in mRNA deadenylation in Drosophila. 23:2862–2871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Collart M (2003) Global control of gene expression in yeast by the Ccr4-Not complex. 313:1–16. [DOI] [PubMed] [Google Scholar]
- 19. Daugeron MC, Mauxion F, Séraphin B (2001) The yeast POP2 gene encodes a nuclease involved in mRNA deadenylation. 29:2448–2455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Tucker M, Staples RR, Valencia-Sanchez MA, Muhlrad D, Parker R (2002) Ccr4p is the catalytic subunit of a Ccr4p/Pop2p/Notp mRNA deadenylase complex in Saccharomyces cerevisiae. 21:1427–1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Liu HY, Badarinarayana V, Audino DC, Rappsilber J, Mann M, et al. (1998) The NOT proteins are part of the CCR4 transcriptional complex and affect gene expression both positively and negatively. 17:1096–1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Aslam A, Mittal S, Koch F, Andrau JC, Winkler GS (2009) The Ccr4-NOT deadenylase subunits CNOT7 and CNOT8 have overlapping roles and modulate cell proliferation. Mol Biol Cell 20:3840–3850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Nakamura T, Yao R, Ogawa T, Suzuki T, Ito C, et al. (2004) Oligo-astheno-teratozoospermia in mice lacking Cnot7, a regulator of retinoid X receptor beta. Nat Genet 36:528–533. [DOI] [PubMed] [Google Scholar]
- 24. Flames N, Hobert O (2011) Transcriptional control of the terminal fate of monoaminergic neurons. Annu Rev Neurosci 34:153–184. [DOI] [PubMed] [Google Scholar]
- 25. Prakash N, Wurst W (2006) Development of dopaminergic neurons in the mammalian brain. Cell Mol Life Sci 63:187–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Smidt MP, Burbach JP (2007) How to make a mesodiencephalic dopaminergic neuron. Nat Rev Neurosci 8:21–32. [DOI] [PubMed] [Google Scholar]
- 27. Tay TL, Ronneberger O, Ryu S, Nitschke R, Driever W (2011) Comprehensive catecholaminergic projectome analysis reveals single-neuron integration of zebrafish ascending and descending dopaminergic systems. Nat Commun 2:171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Filippi A, Mahler J, Schweitzer J, Driever W (2010) Expression of the paralogous tyrosine hydroxylase encoding genes th1 and th2 reveals the full complement of dopaminergic and noradrenergic neurons in zebrafish larval and juvenile brain. 518:423–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Filippi A, Jainok C, Driever W (2012) Analysis of transcriptional codes for zebrafish dopaminergic neurons reveals essential functions of Arx and Isl1 in prethalamic dopaminergic neuron development. Dev Biol 369:133–149. [DOI] [PubMed] [Google Scholar]
- 30. Panula P, Chen YC, Priyadarshini M, Kudo H, Semenova S, et al. (2010) The comparative neuroanatomy and neurochemistry of zebrafish CNS systems of relevance to human neuropsychiatric diseases. Neurobiol Dis 40:46–57. [DOI] [PubMed] [Google Scholar]
- 31. Yamamoto K, Vernier P (2011) The evolution of dopamine systems in chordates. Front Neuroanat 5:21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. McLean DL, Fetcho JR (2004) Ontogeny and innervation patterns of dopaminergic, noradrenergic, and serotonergic neurons in larval zebrafish. J Comp Neurol 480:38–56. [DOI] [PubMed] [Google Scholar]
- 33. Rink E, Wullimann MF (2002) Development of the catecholaminergic system in the early zebrafish brain: an immunohistochemical study. Brain Res Dev Brain Res 137:89–100. [DOI] [PubMed] [Google Scholar]
- 34. Holzschuh J, Ryu S, Aberger F, Driever W (2001) Dopamine transporter expression distinguishes dopaminergic neurons from other catecholaminergic neurons in the developing zebrafish embryo. 101:237–243. [DOI] [PubMed] [Google Scholar]
- 35. Ryu S, Mahler J, Acampora D, Holzschuh J, Erhardt S, et al. (2007) Orthopedia homeodomain protein is essential for diencephalic dopaminergic neuron development. Curr Biol 17:873–880. [DOI] [PubMed] [Google Scholar]
- 36. Filippi A, Mahler J, Schweitzer J, Driever W (2010) Expression of the paralogous tyrosine hydroxylase encoding genes th1 and th2 reveals the full complement of dopaminergic and noradrenergic neurons in zebrafish larval and juvenile brain. J Comp Neurol 518:423–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ryu S, Holzschuh J, Mahler J, Driever W (2006) Genetic analysis of dopaminergic system development in zebrafish. 61–66. [DOI] [PubMed]
- 38. Blechman J, Borodovsky N, Eisenberg M, Nabel-Rosen H, Grimm J, et al. (2007) Specification of hypothalamic neurons by dual regulation of the homeodomain protein Orthopedia. Development 134:4417–4426. [DOI] [PubMed] [Google Scholar]
- 39. Borodovsky N, Ponomaryov T, Frenkel S, Levkowitz G (2009) Neural protein Olig2 acts upstream of the transcriptional regulator Sim1 to specify diencephalic dopaminergic neurons. Dev Dyn 238:826–834. [DOI] [PubMed] [Google Scholar]
- 40. Levkowitz G, Zeller J, Sirotkin HI, French D, Schilbach S, et al. (2003) Zinc finger protein too few controls the development of monoaminergic neurons. Nat Neurosci 6:28–33. [DOI] [PubMed] [Google Scholar]
- 41. Löhr H, Ryu S, Driever W (2009) Zebrafish diencephalic A11-related dopaminergic neurons share a conserved transcriptional network with neuroendocrine cell lineages. 136:1007–1017. [DOI] [PubMed] [Google Scholar]
- 42. Jeong JY, Einhorn Z, Mercurio S, Lee S, Lau B, et al. (2006) Neurogenin1 is a determinant of zebrafish basal forebrain dopaminergic neurons and is regulated by the conserved zinc finger protein Tof/Fezl. Proc Natl Acad Sci U S A 103:5143–5148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Holzschuh J, Hauptmann G, Driever W (2003) Genetic analysis of the roles of Hh, FGF8, and nodal signaling during catecholaminergic system development in the zebrafish brain. 23:5507–5519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Mahler M, Filippi A, Driever W (2010) DeltaA/DeltaD regulate multiple and temporally distinct phases of Notch signaling during dopaminergic neurogenesis in zebrafish. 30:16621–16635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Russek-Blum N, Gutnick A, Nabel-Rosen H, Blechman J, Staudt N, et al. (2008) Dopaminergic neuronal cluster size is determined during early forebrain patterning. Development 135:3401–3413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Knapik EW, Goodman A, Ekker M, Chevrette M, Delgado J, et al. (1998) A microsatellite genetic linkage map for zebrafish (Danio rerio). 18:338–343. [DOI] [PubMed] [Google Scholar]
- 47.Thisse B, Pfumio S, Fürthauer M, B L, Heyer V, et al. (2001) Expression of the zebrafish genome during embryogenesis.
- 48. Kane DA, Kimmel CB (1993) The zebrafish midblastula transition. Development 119:447–456. [DOI] [PubMed] [Google Scholar]
- 49. Morita T, Nitta H, Kiyama Y, Mori H, Mishina M (1995) Differential expression of two zebrafish emx homeoprotein mRNAs in the developing brain. 198:131–134. [DOI] [PubMed] [Google Scholar]
- 50. Oxtoby E, Jowett T (1993) Cloning of the zebrafish krox-20 gene (krx-20) and its expression during hindbrain development. Nucleic Acids Res 21:1087–1095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Reifers F, Böhli H, Walsh EC, Crossley PH, Stainier DY, et al. (1998) Fgf8 is mutated in zebrafish acerebellar (ace) mutants and is required for maintenance of midbrain-hindbrain boundary development and somitogenesis. 125:2381–2395. [DOI] [PubMed] [Google Scholar]
- 52. Ryu S, Mahler J, Acampora D, Holzschuh J, Erhardt S, et al. (2007) Orthopedia homeodomain protein is essential for diencephalic dopaminergic neuron development. 17:873–880. [DOI] [PubMed] [Google Scholar]
- 53. Rohr KB, Concha ML (2000) Expression of nk2.1a during early development of the thyroid gland in zebrafish. 95:267–270. [DOI] [PubMed] [Google Scholar]
- 54. Acampora D, Postiglione MP, Avantaggiato V, Di Bonito M, Simeone A (2000) The role of Otx and Otp genes in brain development. 44:669–677. [PubMed] [Google Scholar]
- 55. Michaud JL, Rosenquist T, May NR, Fan CM (1998) Development of neuroendocrine lineages requires the bHLH-PAS transcription factor SIM1. Genes Dev 12:3264–3275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Wang W, Lufkin T (2000) The murine Otp homeobox gene plays an essential role in the specification of neuronal cell lineages in the developing hypothalamus. Dev Biol 227:432–449. [DOI] [PubMed] [Google Scholar]
- 57. Eaton JL, Glasgow E (2007) Zebrafish orthopedia (otp) is required for isotocin cell development. Dev Genes Evol 217:149–158. [DOI] [PubMed] [Google Scholar]
- 58. Eaton JL, Glasgow E (2006) The zebrafish bHLH PAS transcriptional regulator, single-minded 1 (sim1), is required for isotocin cell development. Dev Dyn 235:2071–2082. [DOI] [PubMed] [Google Scholar]
- 59. Unger JL, Glasgow E (2003) Expression of isotocin-neurophysin mRNA in developing zebrafish. Gene Expr Patterns 3:105–108. [DOI] [PubMed] [Google Scholar]
- 60. Chandrasekar G, Lauter G, Hauptmann G (2007) Distribution of corticotropin-releasing hormone in the developing zebrafish brain. 505:337–351. [DOI] [PubMed] [Google Scholar]
- 61. Bellipanni G, Rink E, Bally-Cuif L (2002) Cloning of two tryptophan hydroxylase genes expressed in the diencephalon of the developing zebrafish brain. 119 Suppl 1S215–220. [DOI] [PubMed] [Google Scholar]
- 62. Lillesaar C, Tannhäuser B, Stigloher C, Kremmer E, Bally-Cuif L (2007) The serotonergic phenotype is acquired by converging genetic mechanisms within the zebrafish central nervous system. 236:1072–1084. [DOI] [PubMed] [Google Scholar]
- 63. Bosco A, Bureau C, Affaticati P, Gaspar P, Bally-Cuif L, et al. (2013) Development of hypothalamic serotoninergic neurons requires Fgf signalling via the ETS-domain transcription factor Etv5b. Development 140:372–384. [DOI] [PubMed] [Google Scholar]
- 64. Herzog W, Sonntag C, von dHS, Roehl HH, Varga ZM, et al. (2004) Fgf3 signaling from the ventral diencephalon is required for early specification and subsequent survival of the zebrafish adenohypophysis. Development 131:3681–3692. [DOI] [PubMed] [Google Scholar]
- 65. Wolf A, Ryu S (2013) Specification of posterior hypothalamic neurons requires coordinated activities of Fezf2, Otp, Sim1a and Foxb1.2. Development 140:1762–1773. [DOI] [PubMed] [Google Scholar]
- 66. Mohammadi M, McMahon G, Sun L, Tang C, Hirth P, et al. (1997) Structures of the tyrosine kinase domain of fibroblast growth factor receptor in complex with inhibitors. Science 276:955–960. [DOI] [PubMed] [Google Scholar]
- 67. Collart MA, Panasenko OO (2012) The Ccr4–not complex. Gene 492:42–53. [DOI] [PubMed] [Google Scholar]
- 68. Miller JE, Reese JC (2012) Ccr4-Not complex: the control freak of eukaryotic cells. Crit Rev Biochem Mol Biol 47:315–333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Funakoshi Y, Doi Y, Hosoda N, Uchida N, Osawa M, et al. (2007) Mechanism of mRNA deadenylation: evidence for a molecular interplay between translation termination factor eRF3 and mRNA deadenylases. 21:3135–3148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Bai Y, Salvadore C, Chiang YC, Collart MA, Liu HY, et al. (1999) The CCR4 and CAF1 proteins of the CCR4-NOT complex are physically and functionally separated from NOT2, NOT4, and NOT5. Mol Cell Biol 19:6642–6651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Liu HY, Chiang YC, Pan J, Chen J, Salvadore C, et al. (2001) Characterization of CAF4 and CAF16 reveals a functional connection between the CCR4-NOT complex and a subset of SRB proteins of the RNA polymerase II holoenzyme. J Biol Chem 276:7541–7548. [DOI] [PubMed] [Google Scholar]
- 72. Chen C, Ito K, Takahashi A, Wang G, Suzuki T, et al. (2011) Distinct expression patterns of the subunits of the CCR4–NOT deadenylase complex during neural development. 411:360–364. [DOI] [PubMed] [Google Scholar]
- 73. Ye W, Shimamura K, Rubenstein JL, Hynes MA, Rosenthal A (1998) FGF and Shh signals control dopaminergic and serotonergic cell fate in the anterior neural plate. Cell 93:755–766. [DOI] [PubMed] [Google Scholar]
- 74.Westerfield M (2000) The zebrafish book: a guide to the laboratory use of zebrafish.
- 75. Woo K, Fraser SE (1998) Specification of the hindbrain fate in the zebrafish. 197:283–296. [DOI] [PubMed] [Google Scholar]
- 76. Tonou-Fujimori N, Takahashi M, Onodera H, Kikuta H, Koshida S, et al. (2002) Expression of the FGF receptor 2 gene (fgfr2) during embryogenesis in the zebrafish Danio rerio. Mech Dev 119 Suppl 1S173–178. [DOI] [PubMed] [Google Scholar]
- 77. Brown LA, Amores A, Schilling TF, Jowett T, Baert JL, et al. (1998) Molecular characterization of the zebrafish PEA3 ETS-domain transcription factor. Oncogene 17:93–104. [DOI] [PubMed] [Google Scholar]
- 78. Munchberg SR, Ober EA, Steinbeisser H (1999) Expression of the Ets transcription factors erm and pea3 in early zebrafish development. Mech Dev 88:233–236. [DOI] [PubMed] [Google Scholar]
- 79. Kiefer P, Strahle U, Dickson C (1996) The zebrafish Fgf-3 gene: cDNA sequence, transcript structure and genomic organization. Gene 168:211–215. [DOI] [PubMed] [Google Scholar]
- 80. Filippi A, Durr K, Ryu S, Willaredt M, Holzschuh J, et al. (2007) Expression and function of nr4a2, lmx1b, and pitx3 in zebrafish dopaminergic and noradrenergic neuronal development. 7:135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Langheinrich U, Hennen E, Stott G, Vacun G (2002) Zebrafish as a model organism for the identification and characterization of drugs and genes affecting p53 signaling. Curr Biol 12:2023–2028. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Evaluation of Cnot8 ATG Morpholino knockdown phenotype. Analysis of fgfr1 expression at 1 dpf in wildtype embryos injected with cnot8 ATG Morpholino. (A, C) non-injected WT sibling. (B) injection of 1 ng cnot8 ATG Morpholino and 1 ng p53 Morpholino. (D) Injection of 2 ng cnot8 ATG Morpholino and 1 ng p53 Morpholino (A and B) lateral views.
(TIF)
Summary of statistical evaluations of cell count experiments and quantificational analysis of WISH signals for Figure 3 – 9 . The File contains the following sheets: Sheet 1 - Summary of results from image quantifications (green = significant differences using Mann-Whitney test; red = no significant differences). Sheet 2 - Results of Mann-Whitney tests for analysis of stain intensities in Figures 3 to 8. Sheet 3 - Image measurements Figure 3. Sheet 4 - Image measurements Figure 4. Sheet 5 - Image measurements Figure 5. Sheet 6 - Image measurements Figure 6. Sheet 7 - Image measurements Figure 7. Sheet 8 - Image measurements Figure 8. Sheet 9 - Summary of cell counts Figures 1 and 9. Sheet 10 - Statistical analysis of cell counts in Figure 1. Sheet 11 - Cell counts and statistical analysis Figure 5. Sheet 12 - Cell counts and statistical analysis Figure 9.
(XLSX)