Summary
L-fucose, a monosaccharide widely distributed in eukaryotes and certain bacteria, is a determinant of many functional glycans that play central roles in numerous biological processes. The molecular mechanism, however, by which fucosylation mediates these processes remains largely elusive. To study how changes in fucosylation impact embryonic development, we up-regulated N-linked fucosylation via over-expression of a key GDP-Fucose transporter, Slc35c1, in zebrafish. We show that Slc35c1 overexpression causes elevated N-linked fucosylation and disrupts embryonic patterning in a transporter activity dependent manner. We demonstrate that patterning defects associated with enhanced N-linked fucosylation are due to diminished canonical Wnt signaling. Chimeric analyses demonstrate that elevated Slc35c1 expression in receiving cells decreases the signaling range of Wnt8a during zebrafish embryogenesis. Moreover, we provide biochemical evidence that this decrease is associated with degradation of Wnt8 ligand and elevated Lrp6 coreceptor, which we show are both substrates for N-linked fucosylation in zebrafish embryos. Strikingly, slc35c1 expression is regulated by canonical Wnt signaling. These results suggest that Wnt limits its own signaling activity in part via up-regulation of a transporter, slc35c1 that promotes terminal fucosylation and thereby limits Wnt activity.
Keywords: Zebrafish patterning, fucosylation, GDP-Fucose transporter, slc35c1, Wnt signaling
Introduction
Fucosylation, the modification of proteins or glycans with a 6-deoxy monosaccharide, L-fucose, plays critical roles in many physiological and pathological processes. Tissue development, fertilization and cell adhesion rely on proper protein fucosylation. Aberrant fucosylation of membrane proteins, however, is characteristic of many pathological conditions, including cancer, neurological disorders, immune dysfunction, and developmental defects. (Becker & Lowe, 2003; Ma et al, 2006). For example, deficiency in fucosylation impairs leukocyte-vascular epithelium interactions, which leads to a congenital disorder known as leukocyte adhesion deficiency type II (LAD II). L-fucose can branch from the core N-Acetylglucosamine (GlcNAc) of N-linked glycans or from the peripheral GlcNAc and Galactose residues in both N- and O-linked glycans. In signaling receptors that contain the epidermal growth factor (EGF) motif (Shao & Haltiwanger, 2003; Wang & Spellman, 1998) or thrombospondin type repeats (TSR), fucose is directly linked to the hydroxyl group of serine or threonine residues (Becker & Lowe, 2003) (Fig. 1A). In vertebrates, the universal fucosyl donor, GDP-Fucose (GDP-Fuc), is produced in the cytoplasm through two independent pathways—the de novo biosynthesis pathway that uses GDP-mannose as the substrate and the salvage pathway that uses fucose directly (Becker & Lowe, 2003). GDP-Fuc serves as the donor substrate for fucosyltransferases (Futs), enzymes located in the endoplasmic reticulum and Golgi, which transfer fucose from GDP-Fuc to N- and O-linked glycans or to protein acceptors directly (Becker & Lowe, 2003; Ma et al, 2006) (Fig. 1A).
Figure 1. slc35c1 enhances the level of N-linked fucosylation expression in zebrafish embryos.
A) Schematic illustration of fucose biosynthetic pathways. Slc35c1 transports GDP-Fuc into the late secretary pathway (mainly the Golgi apparatus). B) slc35c1 mRNA expression during zebrafish development as quantified by qPCR. The blue bars show slc35c1 relative expression during development and the red dots show the qPCR cycle number of the control gene odc1. C) Western blot analysis of the expression of mouse Slc35c1-myc fusion protein of 5hpf zebrafish embryos. D) Western blot analysis of global fucosylation levels at shield stage (6hpf) in embryos microinjected with gmds MO or mouse slc35c1-myc mRNA in the presence or absence of GDP-Fuc before and after PNGase F digestion. Proteins were resolved by SDS-PAGE and IB with AAL followed by anti-biotin-HRP in D) or by streptavidin-Alexa488 for the quantification in E). N-fucosylation: N-linked fucosylation; O-fucosylation: fucose in mucin and O-fucosylated proteins.
As links between GDP-Fuc production and usage, GDP-Fuc transporters are critical regulators of the fucosylation level (Lu et al, 2010; Ma et al, 2006; Moriwaki et al, 2007). In vertebrates, Slc35c1 is the primary transporter for GDP-Fuc into the Golgi apparatus, where Futs modify glycosylated substrates, primarily N-linked glycans (Hellbusch et al, 2007; Ma et al, 2006) (Fig. 1A). Deficiency in fucose due to mutation in Slc35c1 leads to a congenital disorder of glycosylation type IIc in humans, which is characterized by immunodeficiency, developmental abnormalities, psychomotor difficulties, and intellectual disability (Lubke et al, 2001; Luhn et al, 2001).
Almost every cellular process in eukaryotes involves N-linked glycoproteins on some level. Recently, different levels of fucosylation have been associated with distinct receptor activities(Huang et al, 2013; Liu et al, 2011), suggesting potential regulatory functions of fucose modification. However, the nature of such regulation remains unknown. In this study, we found that expression of slc35c1, a rate-limiting step regulating fucosylation (Lu et al, 2010; Moriwaki et al, 2007), fluctuates dramatically during development. This suggests that besides functioning as a “housekeeping gene”, slc35c1 may also play some regulatory role through N-linked fucosylation in specific developmental processes.
We used the zebrafish system to dissect the consequence of over-expression (OE) of Slc35c1 during zebrafish embryonic patterning for two main reasons: first, a limited number of defined signaling pathways regulate axis patterning (Langdon & Mullins, 2011; Schier & Talbot, 2005); second, many of the components of these signaling pathways (e.g. Wnt ligand, Lrp6 and Frizzled in Wnt signaling) are modified with N-linked glycan, but the functional significance of these modifications remains unknown (Cheng et al, 2011; Janda et al, 2012; Joiner et al, 2013). Here we show that Slc35c1 OE triggers enhanced N-linked fucosylation and that elevating N-linked fucosylation in the early zebrafish embryo negatively regulates Wnt signaling at the level of the Wnt ligand. Furthermore, our findings show that Wnt promotes elevated expression of slc35c1. These results suggest that Wnt promotes its own inactivation via up-regulation of a transporter, slc35c1 that promotes terminal fucosylation and thereby limits Wnt activity.
Results
Expression of the GDP-Fuc transporter slc35c1 is dynamic in early zebrafish development
GDP-Fuc availability in cellular compartments is a limiting factor for fucosylation (Lu et al, 2010; Moriwaki et al, 2007). GDP-Fuc transporters play a key role in directing GDP-Fuc to cellular compartments, and thus limit the GDP-Fuc available for protein or glycan modification (Ma et al, 2006). If fucosylation is highly regulated, these transporters are likely targets for regulation. Thus, we reasoned that the expression of GDP-Fuc transporters should also be dynamic during development. In support of this notion, expression of the related slc35c2, which encodes a candidate GDP-Fuc transporter for the early secretory pathway, has been reported to be dynamic in microarray analysis (http://zf-espresso.tuebingen.mpg.de). However, slc35c1 expression has not been reported during zebrafish development. Therefore, we examined the abundance of slc35c1 mRNA during development using quantitative PCR (qPCR) at several developmental stages, including blastula (4hpf and 5hpf), segmentation (10, 13 and 17hpf), pharyngula [32hpf and 2dpf(day post fertilization)], at hatching (3dpf) and during larval stages (4dpf). slc35c1 mRNA was abundant in blastula, reduced during segmentation stages, and abundant again in hatching and larval stages (Fig. 1B). Our qPCR time course shows that zebrafish slc35c1 mRNA fluctuates in abundance during development (Fig. 1B), which suggests that GDP-Fuc transport is developmentally regulated rather than static, as would be expected for a constitutive housekeeping process. Fluctuation of slc35c1 expression levels during zebrafish development suggested that GDP-Fuc transportation to the Golgi, and the N-linked fucosylation that ensues are developmentally regulated.
The highly conserved GDP-Fucose transporter mSlc35c1 promotes N-linked fucosylation in zebrafish
Comparison of the predicted protein sequence of Slc35c1 among fishes including Danio rerio (zebrafish), Takifugu rubripes (Japanese pufferfish), Tetraodon nigroviridis (spotted green pufferfish) and Gasterosteus aculeatus (three-spined stickleback); amphibians Xenopus tropicalis (western clawed frog); birds Gallus gallus (chicken); and mammals including Mus musculus (mouse) and Homo sapiens (human) indicated zebrafish Slc35c1 was 70% identical and around 80% similar to Slc35c1 of other species (Table 1). For example, Slc35C1 proteins showed 69% identity and 81% similarity overall between zebrafish and mouse. Within functionally important regions (e.g. the trans-membrane domains), Slc35c1 was >95% identical among all the vertebrates examined.
Table 1.
Comparison of vertebrate Slc35c1
| Slc35c1 | Danio rerio (Zebrafish) | Takifugu rubripes (Japanese pufferfish) | Tetraodon nigroviridis (Spotted green pufferfish) | Gasterosteus aculeatus (Three-spined stickleback) | Xenopus tropicalis (Western clawed frog) | Gallus gallus (Chicken) | Mus musculus (Mouse) | Homosapiens (Human) |
|---|---|---|---|---|---|---|---|---|
| Danio rerio (Zebrafish) | 100.00 % | 69.00% | 69.00% | 75.00% | 70.00% | 71.00% | 69.00% | 69.00% |
| Takifugu rubripes (Japanese pufferfish) | 100.00% | 90.00% | 78.00% | 66.00% | 63.00% | 62.00% | 65.00% | |
| Tetraodon nigroviridis (Spotted green pufferfish) | 100.00% | 79.00% | 69.00% | 65.00% | 64.00% | 67.00% | ||
| Gasterosteus aculeatus (Three-spined stickleback) | 100.00% | 69.00% | 71.00% | 64.00% | 66.00% | |||
| Xenopus tropicalis (Western clawed frog) | 100.00% | 72.00% | 72.00% | 72.00% | ||||
| Gallus gallus (Chicken) | 100.00 % | 76.00% | 78.00% | |||||
| Mus musculus (Mouse) | 100.00 % | 89.00% | ||||||
| Homo sapiens (Human) | 100.00% |
The biological function of slc35c1 fluctuation can be examined through inhibition or through over-expression of Slc35c1. In previous work, knocking out an individual GDP-Fuc transporter only caused mild phenotypes in flies and in mice, likely due to functional redundancy between transporters (Geisler et al, 2012; Hellbusch et al, 2007; Ishikawa et al, 2010; Lu et al, 2010). We reasoned that over-expressing a transporter might provide a more robust means to perturb fucosylation. Based on the high degree of similarity between zebrafish and mouse Slc35c1, the fully characterized mouse Myc-tagged Slc35c1 (Lu et al, 2010) might similarly promote N-linked fucosylation in zebrafish embryos, as previously reported in cell culture (Lu et al, 2010). To test this notion we expressed mouse slc35c1-myc (mSlc35c1) mRNA in zebrafish and detected the Slc35c1-Myc protein produced using an anti-Myc antibody immuno-blot (IB). We determined that mSlc35c1-Myc was expressed in zebrafish embryos, and its abundance was not affected by GDP-Fuc co-injection (Fig. 1C). To investigate whether mSlc35c1 was biologically active in zebrafish embryos, we examined fucosylation by IB with Aleuria Aurantia Lectin (AAL), which specifically recognizes fucose modifications on glycan or protein (Fig. 1D). As anticipated, lysate of embryos injected with either GDP-Fuc or mSlc35c1 showed an increase in AAL staining, and fucosylation was further enhanced when GDP-Fuc and mSlc35c1 were co-injected (Fig. 1D).
To determine if the enhanced AAL staining was due to N-linked fucosylation on glycan we treated the injected embryos with PNGase F, an enzyme that selectively removes the N-linked glycan from glycoproteins (Maley et al, 1989). After PNGase F digestion, only mucin- type O-linked glycoproteins and O-fucosylated glycoproteins remain. The AAL signal of embryos injected with mSlc35c1 was less abundant after PNGase F digestion with or without co-injection of GDP-Fuc (Fig. 1D, E) as compared to wild-type or GDP-Fuc injected samples. These results indicate that mSlc35c1 enhances N-linked fucosylation.
Enhanced N-linked fucosylation disrupts dorsal-ventral patterning of zebrafish embryos
Previous studies have shown that fucosylation progressively accumulates through the first four days of early zebrafish development (Dehnert et al, 2011). Limiting fucosylation in mice is known to cause developmental defects including early embryonic death (Du et al, 2010; Smith et al, 2002), neuronal migration disorders (Ohata et al, 2009), skeletal abnormalities and other defects (Hayes et al, 2013; Ma et al, 2006). However, the consequence of excess fucosylation has not been examined. To test the effect of excess fucosylation on zebrafish development, we injected various doses of GDP-Fuc into early zebrafish embryos. At 26–30hpf (hours post fertilization) patterning defects were apparent in 5% (n=237) of GDP-Fuc injected embryos with a minority of these lacking identifiable structures. The infrequency of severe phenotypes in embryos injected with GDP-Fuc alone suggests that embryos possess a robust system to monitor fucose levels and balance fucosylation events (e.g. N-linked versus O-linked).
In contrast to GDP-Fuc injection, which affected less than 10% of injected embryos, mSlc35c1 caused dorsalization of the majority of injected embryos (Fig. 2A) in a dose dependent manner in terms of severity and penetrance (Fig. 2B). To determine if a larger GDP-Fuc pool would modulate the mSlc35c1 phenotype, we co-injected GDP-Fuc with mSlc35c1 mRNA. Co-injection of GDP-Fuc caused more severe dorsalization without changing the ratio of dorsalized embryos (Fig. 2B).
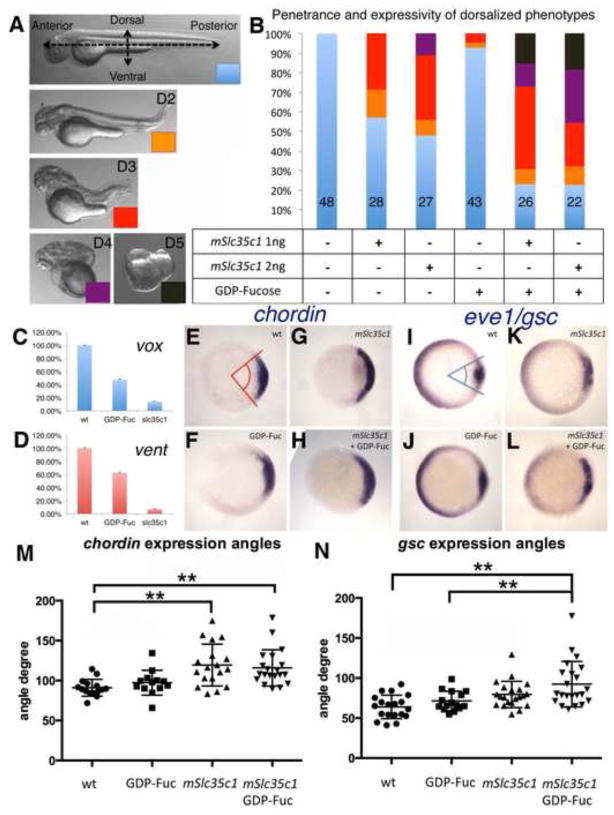
Figure 2. Elevated N-fucosylation dorsalizes zebrafish embryos.
A) Variable degrees of dorsalization in embryos over-expressing mSlc35c1. B) Quantification of the severity and penetrance of dorsalization in each treatment. C–D) qPCR of the ventral transcriptional factors vox and vent in WT, GDP-Fuc injected or mSlc35c1 expressing embryos. E–L) in situ hybridization with dorsal (chordin and gsc) and ventral (eve1) specific markers at shield stage (6hpf). E–H) chordin expression in treated embryos I–L) eve1 and gsc expression. The expression angles of chordin and gsc of embryos with each treatment are summarized in M) and N) respectably. The sample sizes for each treatment are as follows: wt (N=15), GDP-Fuc(N=13), mSlc35c1(N=18) and mSlc35c1 plus GDP-Fuc (N=21) for M); and wt (N=19), GDP-Fuc (N=15), mSlc35c1(N=21) and mSlc35c1 plus GDP-Fuc (N=22) for N). For chordin angle, ordinary One-way ANOVA between sample group means, p=0.0002. For gsc angle, ordinary One-way ANOVA between sample group means, p=0.0001. Note: ** Tukey’s procedure after ANOVA p<0.01. Null hypothesis for χ2 between embryos injected with 1ng mSlc35c1 and with GDP-Fuc plus 1ng mSlc35c1 p=1.41E-04; for χ2 between embryos injected with 2ng mSlc35c1 and with GDP-Fuc plus 2ng mSlc35c1 p=8.99E-03.
To exclude the possibility that dorsalization was caused by a nonspecific overexpression effect that was independent of Slc35c1’s GDP-Fuc transporter function we expressed a mutant Slc35c1 protein, lacking the transporter activity. The Slc35c1 T308R single point mutation was first reported in a human patient with Leukocyte adhesion deficiency type II (Etzioni et al, 2002; Roos & Law, 2001). This mutant protein retains the cellular localization of the wild-type protein but lacks GDP-Fuc transportation activity (van de Vijver et al, 2012). Therefore, we mutated this conserved amino acid to create a Flag-tagged mouse version of the T308R mutant. We injected mSlc35c1T308R mRNA into zebrafish embryos and confirmed by western blotting that the injected mSlc35c1T308R mRNA produced a stable mutant protein (Fig. S1A). Consistent with a requirement for its transporter activity, mSlc35c1T308R OE did not cause significant dorsalization compared to injecting RNA coding for the wild-type protein (Fig. S1B).
Some transporters are known to transport multiple sugars (Ishikawa et al, 2010); thus, it was possible that Slc35c1 induced phenotypes were due to effects beyond fucose transport. To investigate whether mSlc35c1 induced dorsalization was specific for GDP-Fuc, we co-injected mSlc35c1 with UDP-sugars. In contrast to GDP-Fuc, neither UDP-Glucose nor UDP-Galactose enhanced or ameliorated the mSlc35c1 phenotypes (Fig. S1C). To independently assess whether mSlc35c1 induced dorsalization was through GDP-Fuc transporter function, we injected mSlc35c1 together with morpholino to knock down GDP-mannose 4,6-dehydratase (gmds) (Ohata et al, 2009) (Fig. S1D). As in previous studies (Ohata et al, 2009), knocking down gmds, the first enzyme in the de novo GDP-Fuc synthetic pathway decreased fucosylation in zebrafish embryos, as assessed by AAL (Fig. 1D). Whereas co-injection of mSlc35c1 with GDP-Fuc, which enhanced the penetrance and severity of dorsalization (Fig. 2B), co-injection of mslc35c1 with gmds MO shifted the distribution of dorsalized phenotypes toward weaker classes (Fig. S1D). Taken together, these results indicate the DV patterning defects were dependent on GDP-Fuc transport activity.
Expression of ventral transcriptional factors, vox/vent, declines when N-linked fucosylation is enhanced
Dorsal ventral patterning of the early zebrafish embryo is regulated by direct mutual antagonism between pathways that promote dorsal fate and those that promote ventral cell fates (Langdon & Mullins, 2011; Melby et al, 2000; Schier & Talbot, 2005). A maternal Wnt pathway specifies the dorsal organizer (Langdon & Mullins, 2011). During zygotic stages the activities of BMP and Wnt signaling pathways establish ventrolateral fates (Langdon & Mullins, 2011; Schier & Talbot, 2005). Thus, the patterning defects caused by elevated N-fucosylation could be due to enhanced maternal Wnt signaling. Alternatively, enhanced N-fucosylation could interfere with zygotic Bmp/Wnt signaling during ventrolateral patterning. To investigate these possibilities we examined embryos at sphere stage (3.5hpf), a stage when transcription factors promoting dorsal and ventral fates are first expressed and before reciprocal antagonism would confound interpretation of changes in expression. qPCR analysis revealed that GDP-Fuc or mSlc35c1 decreased the abundance of mRNAs encoding ventral transcriptional factors, vent and vox (Fig. 2C,D), suggesting that dorsalization in the presence of elevated N-fucosylation was due to inhibition of factors that promote ventral fates.
To further investigate the molecular basis of the patterning defect we used in situ hybridization to examine markers of dorsal and ventral fates in early gastrula (shield stage embryos). Consistent with the earlier disruption of vox and vent, the dorsal marker chordin (Fig. 2E–H and quantification in Fig. 2M) was quantitatively expanded from an average of 91 degrees in wild type (WT) (n=15; Fig. 2E,M), to 97 degrees in GDP-Fuc injected (n=13; Fig. 2F,M) and 115 degrees in mSlc35c1 injected (n=18; Fig. 2G,M) embryos. Similarly, the dorsal mesodermal marker gsc (goosecoid) (Fig. 2I–L and quantification in Fig. 2N) was expanded from 64 degrees in WT (n=19, Fig. 2I,N) to 79 degrees in mSlc35c1 injected (n=21; Fig. 2K,N) embryos. Statistical analysis showed that mSlc35c1 OE significantly expanded the chordin and gsc expression regions compared to untreated embryos [PostHoc Tukey’s HSD (honest significant difference) test, p<0.01]. Reciprocally, a ventral marker eve1 (even-skipped-like1) was reduced at 6hpf in embryos expressing GDP-Fuc (n=15; Fig. 2J) or mSlc35c1 (n=21; Fig. 2K). Consistent with the reduced ventral marker expression (Fig. 2C–D), eve1 mRNA was also less abundant (Compare Fig. 2K–L to 2I-J). These phenotypes were more severe in embryos injected with both GDP-Fuc and mSlc35c1 (Fig. 2H, L). Expansion of goosecoid, which is not observed in zebrafish BMP mutants strongly indicates that Wnt signaling rather than BMP signaling is the target of N-fucosylation.
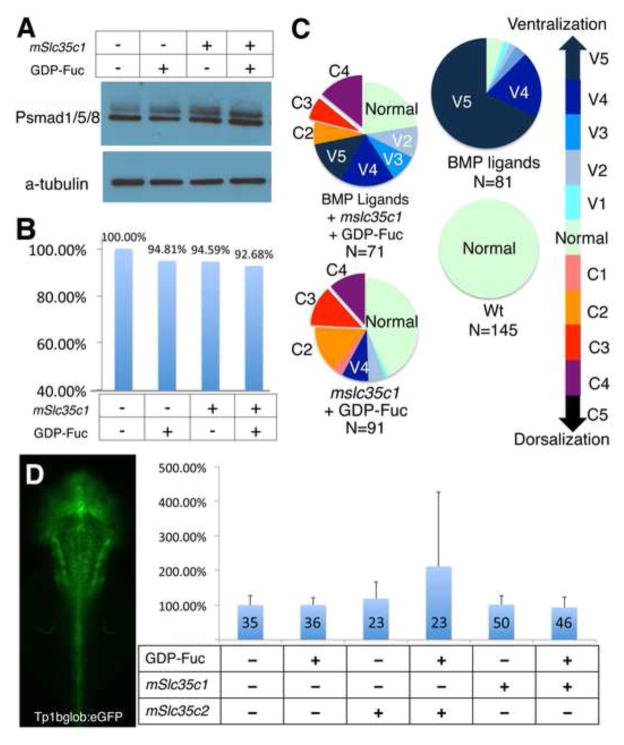
To exclude the possibility that BMP signaling is perturbed in mSlc35c1 OE embryos, we examined phosphorylated Smad1/5/8 (P-Smad) levels, which provide a direct indicator of Bmp2b/4/7 activity (Little & Mullins, 2009; Mintzer et al, 2001), in treated embryos. We found no significant change in P-Smad1/5/8 abundance in embryos treated with GDP-Fuc, mSlc35c1 or both (Fig. 3A,B), indicating that elevated fucosylation did not interfere with BMP activation at early stages. In addition, we investigated whether excess BMP signaling could suppress dorsalization associated with elevated N-fucosylation. As previously reported, over-expression of bmp2b/7 ventralized most wild-type embryos (Little & Mullins, 2009; Nikaido et al, 1997) and some embryos co-expressing mslc35c1. However, excess BMP did not suppress the strongest dorsalized phenotypes (Fig. 3C). Taken together, the expansion of axial mesoderm markers, P-Smad levels and Bmp2b/7-induced ventralization indicate that BMP signaling is intact in the presence of excess N-linked fucosylation.
Figure 3. BMP and Notch signaling are intact in mSlc35c1 expressing embryos.
A) Phospho-Smad1/5/8 antibody blot indicates BMP signaling activity in each treatment. B) Quantification of Phospho-Smad1/5/8 levels, normalized to α-Tubulin protein levels. C) Molecular interaction between BMP signaling and Slc35c1 function. D) Dorsal view of a transgenic zebrafish larva expressing a GFP reporter of Notch activity. Graph shows the GFP expression levels determined by the fluorescence intensities of transgenic notch reporter embryos injected with the specified molecules. The data represent the average of GFP reporter intensity from individual treated embryos, with WT set as 100%.
Notch signaling is the best-characterized signaling pathway regulated by fucose modification (Stanley, 2007; Stanley & Okajima, 2010). To investigate whether transporter overexpression alters Notch signaling, we examined Notch activity using a transgenic GFP-reporter line for Notch activity (Parsons et al, 2009). No significant changes in Notch signaling were observed in embryos expressing mslc35c1 or another GDP-Fuc transporter, slc35c2, that is responsible for the early secretory pathway (Fig. 3D), thus indicating that Notch signaling is also intact when N-linked fucosylation is elevated.
Diminished expression of vox and vent in blastula indicated Wnt signaling might be affected in mSlc35c1 expressing embryos. In zebrafish development, Wnt signaling is critical for patterning the dorsal-ventral (DV) embryonic axis, maternal Wnt8a promotes specification of the dorsal organizer and zygotic Wnts contribute to patterning of ventral-lateral mesoderm (Fekany et al, 1999; Kelly et al, 2000; Langdon & Mullins, 2011; Lu et al, 2011; Marlow, 2010; Schier & Talbot, 2005) and during later stages Wnt promotes posterior fates along the anterior posterior (AP) axis (Erter et al, 2001; Kim et al, 2000; Kimelman et al, 1992; Lekven et al, 2001; Schier & Talbot, 2005).
Wnt signaling is reduced when N-linked fucosylation is elevated
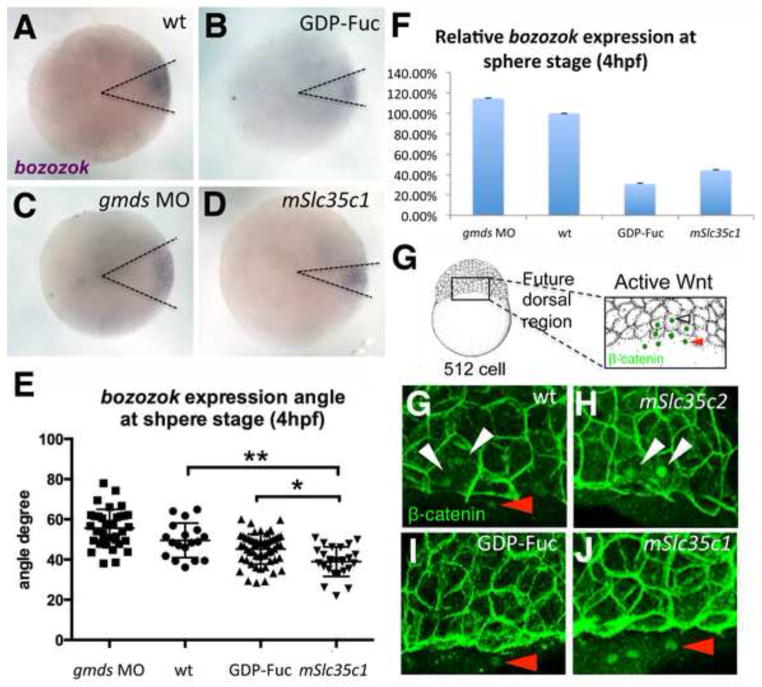
To examine maternal Wnt we assessed two of the earliest indicators of dorsal canonical Wnt activity: activation of an immediate target gene, bozozok (dharma) and nuclear β-catenin accumulation in cells at the core dorsal Wnt response region and the adjacent marginal region, which will become the future dorsal region (Illustrated as Fig. 4G) (Fekany-Lee et al, 2000; Kelly et al, 2000; Langdon & Mullins, 2011; Ryu et al, 2001; Shimizu et al, 2000; Sirotkin et al, 2000; Solnica-Krezel & Driever, 2001). As shown by in situ hybridization (Fig. 4A–D), the territory of the dorsal boz (bozozok) expression domain was significantly reduced in embryos injected with GDP-Fuc, or mSlc35c1 mRNA (Fig. 4B, D, E). Quantitative RT-PCR and analysis of the boz expression domain at sphere stage showed that both GDP-Fuc and mSlc35c1 led to decreased boz abundance and angle of expression (Fig. 4E,F), while gmds MO slightly expanded boz expression (Fig. 4E,F). To determine if reduced activation of boz was due to diminished Wnt signal, we examined the localization of β-catenin just before the onset of the zygotic gene transcription. In response to maternally provided Wnt, β-catenin accumulation within the yolk syncytial layer (YSL), a region adjacent to the margin characterized by the presence of nuclei without surrounding membranes, indicates the future dorsal region (Fig. 4G, H, K). β-catenin was also strongly enriched in the nuclei of marginal cells proximal to this region in normal embryos (Fig. 4H: n=6), and in embryos injected with mSlc35c2 (Fig. 4I; n=5). However, strong nuclear β-catenin was absent in the marginal blastoderm cells in GDP-Fuc and mSlc35c1 expressing embryos (Fig. 4I, K; n=7 for GDP-Fuc and n=9 for mSlc35c1). Together with the expression analyses these results suggest excess N-fucosylation disrupts maternal Wnt signaling and leads to diminished expression of immediate Wnt target genes.
Figure 4. GDP-Fuc or mSlc35c1 overexpression inhibits maternal Wnt signaling.
A–D) in situ hybridization of a direct Wnt signal target gene bozozok(dharma) in sphere stage (4hpf) embryos; lateral views. E) Quantification of bozozok domain angle (e.g. dashed lines in A–D) in each group. The sample sizes for each treatment were as follows: gmds MO (N=35), wt (N=18), GDP-Fuc(N=54) and mSlc35c1(N=26). Ordinary One-way ANOVA between sample group means, p<0.0001. F) qPCR of bozozok, a direct Wnt target gene in WT, GDP-Fuc injected or mSlc35c1 expressing embryos. bozozok expression is normalized to expression of the eef1a1a house keeping gene. G) Schematic illustration of an embryo demonstrates the boxed regions shown in H–K) for β-catenin protein localization at 512 cell stage (3hpf). View of the dorsal margin, as indicated by the nuclei of the yolk syncitial layer, which lack membranes (red arrowhead) at the margin, and the strong nuclear label (white arrowhead) in cells within the “core” region (i.e. closest to the Wnt8a source). The nuclear localization of β-catenin in the core region of (H) WT (n=6) and (I) mSlc35c2 (n=5) injected embryo, but is faint in either (J) GDP-Fuc (n=7) or (K) mSlc35c1 (n=9) injected embryos. Note: ** Tukey’s procedure after ANOVA ** p<0.01; * p<0.05. Bar=100μm.
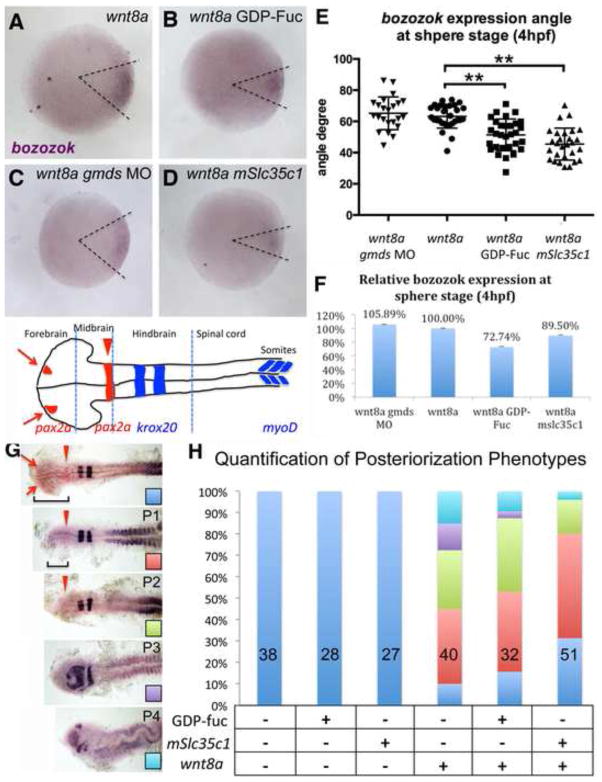
Wnt8a gain of function is known to cause an expansion of the immediate Wnt target boz during early dorsal-ventral patterning (Compare Figs. 4A and 5A). As shown by in situ hybridization (Fig. 5A–D), the territory of the boz expression domain was significantly reduced in embryos co-injected with GDP-Fuc, or mSlc35c1 mRNA (Fig. 5B, D and quantification shown in Fig. 5E). Quantitative RT-PCR analysis of boz mRNA at sphere stage showed that both GDP-Fuc and mSlc35c1 led to decreased boz abundance (Fig. 5F) in Wnt8a OE embryos, while coninjection of gmds MO did not significantly modulate boz expression (Fig. 5E,F). Together with the expression analyses these results suggest excess N-fucosylation blocks maternal Wnt signaling and leads to reduction of immediate Wnt target genes.
Figure 5. mSlc35c1 and GDP-Fuc can suppress Wnt8a induced patterning defects.
A–D) in situ hybridization of bozozok expression in injected embryos at sphere stage (4hpf). E) Quantification of bozozok angle (area outlined by dashed lines in A–D). F) qPCR of bozozok, a direct Wnt target gene in wnt8a alone or combined with GDP-Fuc injected, gmds MO injected or mSlc35c1 expressing embryos. bozozok expression was normalized to the expression of the house keeping gene eef1a1a. G) Posteriorization caused by over-expression of wnt8a. All embryos shown are 18hpf. P1–P4 denote the degree of posterioization, as determined by the marker gene pax2a-krox20-myoD expression patterns. The P1 class lacks the telencephalon as indicated by loss of anterior pax2a expression, (red arrow). P2 embryos lack regions anterior to the MHB domain of pax2a, (red arrowhead). P3 embryos lack MHB expression of pax2a. In the P4 class krox20 expression indicates loss of anterior hindbrain segments. H) Quantification of each treatment. Note: ** Tukey’s procedure after ANOVA p<0.01. Null hypothesis for χ2 between embryos injected with wnt8a and with wnt8a plus mSlc35c1 p= 6.05E-08.
We further investigated whether excess N-linked fucosylation affected the later zygotic roles of Wnt in patterning ventrolateral and posterior fates. Specifically, we determined if elevating N-fucosylation with mSlc35c1 and GDP-Fuc could suppress posteriorization induced by Wnt8a gain of function. As reported previously (Kelly et al, 1995), Wnt8a gain of function caused loss of anterior brain structures concomitant with expansion of posterior structures at late stages (Fig. 5G–H). Co-injection of mSlc35c1 or GDP-Fuc partially suppressed posteriorization by Wnt8 (Fig. 5H). Taken together, these results suggest that excess N-fucosylation can block both endogenous and exogenous Wnt signaling.
As an independent assessment of N-linked fucosylation effects on Wnt signaling, we performed Top-flash assays using CHO (Chinese hamster ovary) cell lines. We choose CHO cells because there are well-established engineered CHO cell lines with different repertoires of glycan modification (Patnaik & Stanley, 2006; Stanley et al, 1996). Normally, CHO cells do not express 1–3,4 Fucosyltransferases. The main N-linked glycan structure is represented by structure 1 (Fig. S2A). The LEC30 cell line is a CHO cell derivative in which forced expression of FUT IV and FUT IX has been introduced (Patnaik et al, 2000; Potvin & Stanley, 1991). In these cells, the predominant N-linked glycan structure is represented by structure 2 (Fig. S2A). Fucose modification can also be achieved by directly adding fucose using in vitro fucosylation assays (Zheng et al, 2011)(Fig. S2A).
We performed TOP-flash assays as reported in (Mao et al, 2002; Mao et al, 2001) and found that the LEC30 line, which has alpha 1–3/1–4 fucosylation on the N-linked glycan, had less Wnt activity (Fig. S2B). To control for potential differential responses between cell lines, we performed in vitro fucosylation assays. Since both CHO and LEC30 lines have endogenous Wnt activity, we normalized all data to untreated CHO or LEC30 lines. The dual luciferase assay revealed decreased Wnt response after fucosylation in CHO cells, whereas the LEC30 cells, which already had 1–3/1–4 fucosylation, showed no difference in Wnt activity (Fig. S2C). Therefore, terminal N-fucosylation can also block Wnt signaling in CHO cells.
Elevated N-fucosylation diminishes the range of Wnt signaling
We used a chimeric approach to distinguish between an effect on ligand secretion versus receptor and cofactor mediated reception of the Wnt signal. Briefly, donor embryos were injected with a tracer dye and wnt8a mRNA, with or without mSlc35c1 mRNA and transplanted into dome-stage wild-type host embryos (Fig. 6A). At one-hour post transplantation, the intensity of nuclear β-catenin in donor cells and neighboring host cells was quantified (Fig. 6A). Briefly donor cells were identified based on the fluorescent signal of the tracer dye, and nearby host cells were assigned based on their distance from the donor cells as N1 (the cells in direct contact with donor cells), N2 (one cell away from donor cells), N3, etc. and all cells four cell diameter away from the donor were defined as B (background) cells. The nuclear region was defined based on DAPI, and the β-catenin intensity in the nuclear region was measured (Fig. 6B).
Figure 6. mSlc35c1 non-cell-autonomously limits Wnt8a activity.
A–B) Schematic depicts the procedure for making chimeras and the quantification of the Wnt responses in host embryos. Donor embryos were injected with tracer dye and wnt8a mRNA, with or without mSlc35c1 mRNA. At the 1000-cell stage, cells were extracted from donors and transplanted into dome-stage host embryos. Confocal images of chimeric embryo were taken with the same setting for all embryos. Then 8 sections (equal to 2.4μm of embryo volume) were projected to a single image, as shown in B–C). B) Merged images (Red: donor; blue: nuclei and green: β-catenin staining) regions to be quantified were determined in Photoshop CS5 by manually circling the nuclear region of target cells in nuclei channel and measure in the β-catenin channel. Cells are divided accordingly: D, donor cells; N1, cells directly adjacent to donors; N2, cells 1- cell away from donors; B, background cells (those at least 4 cell diameters away from donor cells). C–D) nuclear β-Catenin abundance in the chimeras, normalized to the staining in background cells. The numbers in each column indicate the number of cells analyzed. C) The response of WT embryos to donor cells expressing mSlc35c1 mRNA. D) The response of WT embryos to donor cells expressing wnt8a or wnt8a plus mSlc35c1 mRNA. F) The response of WT or mSlc35c1 expressing embryos to donor cells expressing wnt8a mRNA. Note: ** Tuckey’s procedure after ANOVA p<0.01; * p<0.05.
As anticipated, the Wnt secreted by these donor cells activated β-catenin nuclear translocation in neighboring cells within a certain range of the ligand-expressing donors (Fig. 6B). However, given the small amount of wnt8a mRNA introduced into donor cells, the brief one-hour incubation time and the limited donor cell numbers, β-catenin accumulation was used as a direct readout of the response of wild-type host cells to wnt8a expressing donor cells (Fig. 6D). Donor cells also responded to the wnt8 they secreted and the response of the adjacent host cells directly correlated with the distance of the cell from the donors (Fig. 6C–D). The cells in direct contact with donors (N1) showed the strongest response, N2 was weaker, and so on. For all experiments examined, N4 was nearly identical to the background cells, including in the experiment shown in Fig. 6C, in which N3 cells were also not different relative to background (data not shown).
If mSlc35c1 affected the process of Wnt8a secretion, then less Wnt8 would be released from donor cells that co-express mSlc35c1. This would result in a reduced Wnt active region surrounding donor cells as determined by assessing nuclear β-catenin accumulation in the neighboring host cells. If secretion was not impaired in donor cells co-expressing mSlc35c1 and Wnt, then the Wnt activity range would resemble that induced by donors expressing Wnt8 alone. Conversely, the ability of cells to respond to Wnt ligand in the presence of Slc35c1 can be determined by transplanting Wnt expressing donor cells into host embryos that express mSlc35c1. If mSlc35c1 decreased the ability of host cells to respond to Wnt ligand, then the Wnt8 active region (neighbors with nuclear β-catenin) would be reduced in host cells that express mSlc35c1 compared to WT hosts. We observed no significant difference in the Wnt activity region formed by donor cells that expressed Wnt8a alone or together with mSlc35c1 (Fig. 6C–D). In contrast, the response to Wnt8a expressing donor cells was diminished when the host cells expressed mSlc35c1 (Fig. 6D). Taken together, these results indicate that Wnt8a secretion is intact, but the ability of neighboring cells to respond to Wnt8a signaling is attenuated in the presence of mSlc35c1.
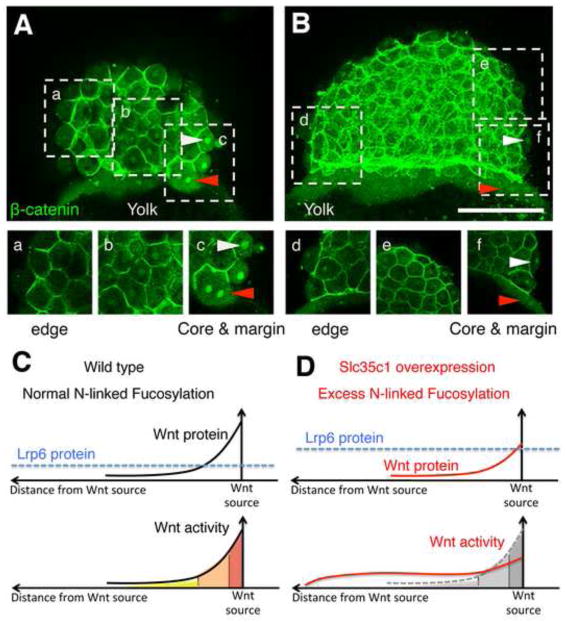
In light of the transplantation results, we closely examined the Wnt activity gradients established by maternal Wnt in wild-type and slc35c1 OE embryos (Fig. 7A–B; n=3 for WT and n=7 for slc35c1 OE). As previously reported (Lim et al, 2012; Lu et al, 2011), three regions of distinct Wnt activity were observed: the region closest to the Wnt source, referred to here as the core and margin region, had the highest Wnt activity as indicated by nuclear β-Catenin (white arrowhead) (Fig. 7A, c); the region adjacent to the core had weaker activity (Fig. 7A, b); and regions distal to the source lacked nuclear β-Catenin (Fig. 7A,a). In slc35c1OE embryos, the region of the embryo with the intermediate Wnt response based on nuclear β-catenin staining (regions adjacent to and distal to the core region) was expanded. Although the region with nuclear β-catenin staining was expanded, the highest levels normally found on dorsal were not attained (Fig. 7B, d-f). This flattened gradient suggests that Slc35C1 interferes with formation of distinct domains of high and low Wnt activity.
Figure 7. Excess fucosylation changes the response pattern of Wnt signaling.
AB) Projections of stacks of individual confocal image sections show the nuclear localization of β-catenin in early zebrafish embryos (512 cell stage). a-f) Single confocal sections. A) β-catenin in a representative wild-type embryo. a) Peripheral blastomeres at the animal pole. b) medial region of the blastoderm. c) The “core” region of highest Wnt activity at the dorsal margin. B) The Wnt gradient of a representative mSlc35c1 expressing embryo. d) The edge, e) the medial and f) the core and margin regions. C–D) Schematic illustration shows Wnt activities based on β-catenin nuclear localization. In wt embryos, with normal level of N-fucosylaiton, Wnt protein creates a morphogen gradient. In Slc35c1 over-expressing embryos, with excess N-fucosylation, Wnt protein levels are diminished, which reduces the highest Wnt activity response region; Lrp6 protein level is increased, which increases the basal level of the Wnt response, creating an extended zone of intermediate Wnt response. Note: Bar=200μm.
Excess Slc35C1 reduces Wnt8a, but not Lrp6 protein levels
Because our chimeric analysis indicated that Wnt8 signaling was likely disrupted at the level of ligand/receptor/cofactor interactions we investigated which components were modified by Fucosylation. Previous reports indicate that Wnt8a is modified by N-linked glycan modification based on the PDB crystal structure and detection of fucose on Xenopus Wnt8 (Janda et al, 2012). Since the N-linked glycan modification site is conserved among vertebrates, we predicted that zebrafish Wnt8a was a likely candidate for fucosylation modification. To examine Wnt8a protein, we generated Flag-tagged versions of wild-type and dominant negative zebrafish Wnt8a. Through overexpression studies we determined that tagged Wnt retained biological activity, but was less potent than its untagged counterpart (Fig. S3). Moreover, as with the untagged Wnt8, co-expression with mSlc35c1 inhibited the activity of the Flag-tagged Wnt8a (Fig. S3B). However, we were unable to achieve expression levels of Wnt8a-flag that were sufficient to detect any fucosylation with AAL IB.
Dominant negative Xenopus Wnt8 protein, which lacks the C-terminal region, competes with wild-type Wnt8a for receptors and cofactors when expressed in Xenopus or zebrafish embryos (Erter et al, 2001; Hoppler et al, 1996). This dominant negative form of Wnt8 (DN-Wnt8) is thought to bind to the receptor without activating it or inducing its internalization (Hikasa & Sokol, 2011). Since this DN-Wnt8 retains the N-linked glycosylation sites and can be highly expressed (Hoppler et al, 1996), we reasoned that fucosylation of Wnt8 ligand could be examined using DN-Wnt8-Flag. First we determined if DN-Wnt8 had biological activity. As previously reported in Xenopus (Cheyette et al, 2002), DN-Wnt8 over-expression caused dorsal-ventral patterning defects but was less potent than its untagged counterpart (Fig. S3). As anticipated, the DN-Wnt8-Flag was recognized by the fucose specific lectin AAL (Fig. 8A), indicating that Wnt8 is likely modified with N-linked fucosylation in vivo.
Figure 8. Excess fucosylation is associated with decreased Wnt8 and enhanced Lrp6.

A) AAL Immuno-blot (IB) DN-Wnt8-Flag is modified by fucosylation. Top panel AAL IB; Lower panel, α-Flag IB shows DN-Wnt8-Flag input. Each lane represents pull-down of DN-Wnt8-Flag from a pool of 33 embryos. B) IB shows Lrp6 is fucosylated, which is recognized by bioorthogonal labeling. Top panel: GDP-FucAl incorporated bands. FucAl was detected by linking to Azido-biotin through a click reaction, which is recognized by anti-biotin IB. Lower panel: α-Lrp6 IB shows the endogenous Lrp6 input. Each lane represents Lrp6 pull-down from a pool of 45 embryos. C) anti-Flag IB showing Wnt8a-Flag protein in the presence or absence of mSlc35c1. Each lane represents an IP from a pool of 67 embryos. D) anti-Flag IB shows the DN-Wnt8-Flag (DNW-F) protein level in the presence or absence of mSlc35c1. Each lane represents pull-down of DN-Wnt8-Flag from a pool of 27 embryos. The lower panel is α-Actin IB (2 embryo equivalents for each treatment). E) IB shows mSlc35c1 expression has opposite effects on DN-Wnt8-Flag and Flag-Lrp6 protein abundance. Top panel: α-Flag IB reveals reduced DN-Wnt8-Flag in mSlc35c1 expressing embryos. Middle panel: α-Flag IB shows enhanced Flag-Lrp6 in mSlc35c1 expressing embryos. Each lane represents pull-down of Flag tagged protein from a pool of 64 embryos. The lower panel is the loading control with each lane representing the lysate from 2 embryo equivalents. Blue arrowhead indicates DN-Wnt8-Flag; red arrowhead marks Wnt8-Flag-Mid; orange arrowhead indicates Lrp6-Flag or endogenous Lrp6. F) Quantification of the penetrance and severity of dorsalization in embryos injected with mSlc35c1, lrp6 or both, reveals interaction between mslc35c1 and lrp6. 500pg of mslc35c1 and 300pg lrp6 mRNA was injected per embryo. Null hypothesis for χ2 between embryos injected with mSlc35c1 and with lrp6 plus mSlc35c1 p= 9.29E-09.
We also investigated whether endogenous Lrp6 was a substrate for fucosylation using a metabolic labeling system to introduce an alkyne-tagged GDP-Fuc analog, GDP-L-6-ethynylfucose (GDP-FucAl) (Wang et al, 2009b). The alkyne-bearing fucose incorporated into fucosylated proteins can then be labeled by biotin-azide via Copper (I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC) (Jiang et al, 2011; Soriano Del Amo et al, 2010) and detected with anti-biotin IB (Fig. 8B). We found that endogenous LRP6 was fucosylated in wild-type and mSlc35c1 expressing embryos (Fig. 8B), indicating that Lrp6 is also likely substrate for N-linked fucosylation.
Our chimeric analysis showed that mSlc35c1 co-expression did not impair Wnt secretion from donor cells. In contrast, mSlc35c1 diminished the response to Wnt ligand when expressed in receiving cells, indicating diminished activation of or interaction with receptors/cofactors on receiving cells or increased clearance of Wnt. To test the possibility that excess N-Fucosylation altered Wnt ligand abundance, we examined Wnt8a-Flag abundance in mSlc35c1 expressing embryos (Fig. 8C). We injected embryos with wnt8a-Flag in the presence or absence of mSlc35c1, and extracted protein at shield stage for IB. Wnt8a-flag expression was very weak in wild type, and was undetectable in the presence of mSlc35c1. We also examined the abundance of DN-Wnt8a protein and found that it too was reduced in embryos co-expressing mSlc35c1 (Fig. 8D, S4A). Thus, excess terminal fucose led to a decrease in Wnt ligand abundance.
Wnt ligand is strictly regulated at multiple levels (Gross & Boutros, 2013; Strigini & Cohen, 2000). However, the major known mechanism to clear Wnt ligand is through degradation mediated by Lrp6:Wnt binding, which limits both the Wnt ligand and Lrp6 receptor protein levels (Li et al, 2010; Park et al, 2013). Lrp6 mediates canonical Wnt signaling activity through a mechanism thought to involve regulated endocytosis (Pinson et al, 2000; Tamai et al, 2000; Wehrli et al, 2000). Once Wnt8a ligand binds to Lrp6 and Frizzled receptors, the Lrp6-Fz-Wnt complex is endocytosed via either Caveolin- or Clathrin-dependent pathways, which activates or inhibits β-catenin signaling by degrading or recycling the protein complex, respectively. (Yamamoto et al, 2008a). Our chimeric analysis and IP results showed that mSlc35c1 over-expression affected Wnt ligand abundance after its secretion; thus, it was possible that Wnt8a signaling was limited through Lrp6 mediated degradation.
If Lrp6 mediated degradation were responsible for diminished Wnt8a in mSlc35c1 expressing embryos, then both Lrp6 and Wnt8a protein levels should similarly decline in the presence of mSlc35c1. To investigate this possibility we expressed a tagged human Lrp6 (Mao et al, 2001). However, when expressed on its own Flag-Lrp6 was too weak to detect (Fig. 8E, S4B), likely due to DKK-Kremen mediated degradation (Mao et al, 2002; Mao et al, 2001). Previous studies have shown that ligand, when co-expressed can compete with DKK1 for Lrp6 binding (Li et al, 2010). Accordingly, we were able to detect Flag-Lrp6 when it was co-expressed with the DN-Wnt8a ligand (Fig. 8E, S4B). As before, DN-Wnt8 protein was less abundant in the presence of mSlc35c1(Fig. 8E, S4A). Surprisingly, the abundance of tagged Lrp6 did not decline, but instead increased in the presence of mSlc35c1. These results indicate that Lrp6 degradation and Wnt degradation are not coupled in the Slc35c1 OE environment.
Excess Lrp6 enhances N-fucosylation mediated dorsalization
Previous studies demonstrate direct linkage between Lrp6 glycosylation and its stability and function (Jung et al, 2011); therefore, it was possible that excess N-fucosylation stabilized Lrp6. However, the ratios of fucosylated Lrp6 between wild type and embryos expressing mSlc35c1 were not significantly different when normalized to the Lrp6 input for each sample (Fig. 8B). To further investigate the relationship between elevated Lrp6 and Slc35c1 induced dorsalization we tested whether embryos expressing mSlc35c1 were more sensitive to Lrp6 OE. Wild-type embryos injected with 300pg of lrp6 were phenotypically normal, embryos injected with 500pg mSlc35c1 showed patterning defects in less than 20% of embryos (Fig. 8F). By comparison combined injection of both caused dorsalization of more than 70% of embryos (Fig. 8F); thus, mSlc35c1 expressing embryos are more sensitive to elevated Lrp6 protein.
slc35c1 expression is associated with Wnt activity
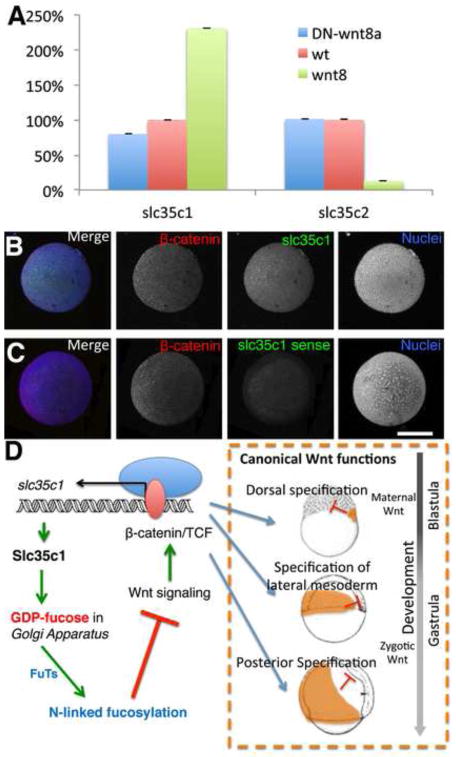
Given the developmental fluctuations in slc35c1 expression, Slc35c1’s inhibitory effect on Wnt8a signaling, recent reports that slc35c1 can be induced by TGF-β signaling (Xu et al, 2013) and the known function of TGF-β family members, BMPs, in antagonism of maternal Wnt activity during early development, we hypothesized that slc35c1 could be activated by BMP. Therefore we used qPCR to test the responsiveness of slc35c1 to these early signaling pathways. Surprisingly, among the pathways examined we found that Wnt8a OE led to a nearly twofold increase of slc35c1 mRNA levels compared to control embryos, when expression was normalized to that of the house keeping gene odc1 (Fig. 9A). This enhancement was specific as slc35c2, a candidate GDP-Fuc transporter for the early secretory pathway, showed diminished levels rather than enhanced expression in Wnt8a OE embryos when compared to wild type (Fig. 9A). On the other hand, when canonical Wnt signaling was inhibited by injecting DN-wnt8 mRNA, slc35c1 mRNA abundance declined, while slc35c2 mRNA abundance did not change (Fig. 9A).
Figure 9. The interplay of Wnt8a signaling, slc35c1-mediated GDP-Fuc transport and N-linked fucosylation.
A) qPCR results show the relative expression of slc35c1 and slc35c2 in zebrafish embryos treated with wnt8a or DN-wnt8a mRNA and untreated control sphere stage (4hpf) embryos:. All results are normalized to odc1. Expression in WT is defined as 100%. B–C) Confocal images of slc35c1 fluorescent in situ shows the positive correlation of slc35c1 expression to β-catenin richness. Red: β-catenin protein staining. Green: slc35c1 probe in situ hybridization in B) or slc35c1 sense probe in situ hybridization in C). Blue: nuclei. D) A schematic model for the relationship between Slc35c1 and Wnt8a. Wnt8a activated canonical Wnt signaling enhances slc35c1 expression. Increased slc35c1 enhances N-inked fucosylation, which inhibits Wnt/beta-catein/TCF signaling during both blastula and gastrula stages. Therefore, these components comprise a negative regulatory loop to control Wnt signaling through N-fucosylation. Note: Bar =300μm.
To further examine the relationship between slc35c1 expression and Wnt activity, we performed fluorescent in situ hybridization for zebrafish slc35c1 at shield stage and co-stained with β-catenin to label the Wnt active region. Confocal images showed that β-catenin staining was enriched in one side of the embryo (Fig 9B–C), consistent with reported Wnt activity regions(Erter et al, 2001; Itoh & Sokol, 1999; Lekven et al, 2001; Schier & Talbot, 2005; Sumoy et al, 1999). Fluorescent in situ hybridization for zebrafish slc35c1 showed that the slc35c1 mRNA was broadly distributed, as reported previously, and appeared to be enriched in regions with strong β-catenin staining (Fig. 9B; n=8). No signal was detected with slc35c1 sense probe (Fig. 9C; n=7). Taken together these findings suggest that canonical Wnt signaling can modulate slc35c1 expression.
Discussion
Fucosylated glycans are known to be involved in early development (Ma et al, 2006). However, single or double gene knockouts of the components of fucosylation pathways, mainly fucosyltransferases in mouse cause embryonic lethality (Du et al, 2010; Luhn et al, 2001; Okamura & Saga, 2008; Smith et al, 2002), compromise overall health of the embryo. (Domino et al, 2001; Homeister et al, 2001; Kudo et al, 2004; Maly et al, 1996). Thus, with the exception of FucT8 (Gu et al, 2013; Kamio et al, 2012; Wang et al, 2006) loss of function studies have been hampered by early lethality and redundancy among enzymes. Here we utilized an overexpression (OE) strategy to study the function of one specific fucose modification, N-linked fucosylation in zebrafish. We identified a regulatory function of N-linked fucosylation on Wnt signaling that is mediated through the GDP-Fuc transporter, Slc35c1. Furthermore, we identified Wnt8a ligand and the Lrp6 coreceptor as substrates of N-fucose modification. Interestingly, although both Wnt8a and Lrp6 are modified, Lrp6 abundance increases whereas based on the OE phenotypes and reduced DN-Wnt8 we infer that Wnt8a abundance similarly decreases in slc35C1 overexpressing embryos. We propose that the combined effect of reduced ligand and sustained Lrp6 dampens the Wnt activity gradient in slc35C1 overexpressing embryos and thereby disrupts patterning. Surprisingly, we found that Wnt promotes slc35c1 expression, thereby forming a negative feedback loop for Wnt regulation.
Negative feedback of canonical Wnt signal through Slc35c1 mediated N-fucosylation
Canonical Wnt signaling is required for multiple aspects of early embryonic patterning. In zebrafish, a maternal Wnt pathway specifies the dorsal organizer (Langdon & Mullins, 2011). During gastrula stages the activity of zygotic Wnt and BMP signaling pathways establishes ventrolateral fates and limits axial (organizer) fates (Langdon & Mullins, 2011; Schier & Talbot, 2005). During segmentation stages, Wnt activity is required to pattern the anterior-posterior axis (McGrew et al, 1995) and for somitogenesis (Takada et al, 1994). Significantly, the domains of Wnt activity shift as the embryo is undergoing morphogenesis (Langdon & Mullins, 2011; Schier & Talbot, 2005), but the mechanisms that facilitate redeployment of Wnt for its maternal and zygotic functions are not fully understood.
Here we found that mRNA encoding slc35c1, a GDP-fucose transporter that facilitates N-Fucosylation, fluctuates in abundance during zebrafish embryogenesis, increases with elevated Wnt and decreases when Wnt is inhibited, suggesting modulation of slc35c1 expression may be important for Wnt regulated developmental processes. We show that BMP and Notch signaling pathways are intact in the presence of elevated fucosylation associated with excess Slc35C1. In contrast, canonical Wnt signaling is diminished. As has been recently reported for other Wnt inhibitors (Flowers et al, 2012; Hayes et al, 2013; Liu et al, 2013), the reduced expression of direct Wnt targets in slc35c1 expressing embryos is consistent with an inhibitory function of Slc35C1 on Wnt signaling. Reduced intensity of nuclear β-Catenin in the “core” region (future dorsal) and the immediate Wnt target boz prior to formation of the dorsal organizer are consistent with inhibition of maternal Canonical Wnt signaling. Although Wnt is initially required to induce the organizer, it subsequently acts in lateral mesoderm to limit the size of the organizer reviewed in (Fig. 9D) (Langdon & Mullins, 2011; Schier & Talbot, 2005). In bozozok mutants, loss of anterior head structures occurs due to expanded Wnt activity (Erter et al, 2001; Fekany-Lee et al, 2000; Gonzalez, 2000). In constrast, the expanded organizer based on chordin and gsc expression and subsequent morphological phenotypes of Slc35c1 OE embryos are consistent with sustained attenuation of Wnt. Therefore, these seemingly contradictory results at different stages suggest that N-fucosylation continuously modulates Wnt signaling during early morphogenesis and gastrulation: Slc35c1 OE embryos have reduced Wnt during both blastula and gastrula stages, when they display reduced and expanded dorsal structures, respectively. Notably, because negative regulation of Wnt activity by Slc35C1 is not confined to early zygotic stages before the formation of the previously defined transcription-dependent Wnt signaling feedback loop (Logan & Nusse, 2004), but was also observed in later AP patterning events, as shown by the inhibition of the early patterning defects and posteriorization induced by exogenous wnt8a our findings suggest the existence of a negative feedback loop, by which Wnt signaling controls Slc35c1-mediated N-fucosylation, which in turn inhibits Wnt8 signaling (Fig. 9D). Negative feedback regulation such as the N-fucosylation mediated feedback loop discovered in this work could provide a robust mechanism to clear the maternal Wnt signal and allow for reutilization of Wnt in later processes, such as patterning of the zygotic anterior posterior axis.
Wnt ligand and Lrp6 receptor modification and patterning
Many Wnt signaling components, including Wnt (Tang et al, 2012), Frizzled receptor (Janda et al, 2012), Lrp6 co-receptor(Khan et al, 2007), and antagonists such as DKK (Haniu et al, 2011), Wif1 (Malinauskas et al, 2011), and sFRPs (Chong et al, 2002) are known to be modified by N-linked glycans in species ranging from fruit flies to humans. Other studies have shown that N-glycosylation of Lrp6 is indispensable for its folding and normal function in cultured cells (Jung et al, 2011; Singh et al, 2013). Among the potential Wnt signaling candidates for regulation via N-Fucosylation, RNAseq data (Sanger Zebrafish Sequencing Project) and their published expression patterns indicate that wif1, and the secreted frizzled-related proteins sfrp1b, sfrp 2, sfrp2l and sfrp5 (Lu et al, 2011; Tendeng & Houart, 2006) are not expressed at stages corresponding to the earliest phenotypes caused by excess Slc35c1. Thus, fucosylation of these proteins cannot account for the observed Wnt regulation. In contrast, Frizzled receptors, Lrp6 and two early antagonists, Sfrp1a and Frzb (Lu et al, 2011) are expressed during this developmental period, and thus are potential substrates for fucosylation-mediated regulation of Wnt signaling. In this study, we have confirmed that at least two such components, Wnt8a and Lrp6, are fucosylated in the zebrafish embryo.
Importantly, we discovered that elevating N-fucosylation by OE of slc35c1 decreases the abundance of Wnt8a without impairing its secretion. Instead N-fucosylation attenuates Wnt signaling in the responding cells. Similarly, studies in Drosophila that indicate that Wnt ligands devoid of N-glycan modification have no apparent effect on ligand secretion, or short-range target activation indicating receptor binding, but show reduced rescue capacity in paracrine rescue assays (Tang et al, 2012). The authors concluded that N-glycosylation was dispensable for Wg/Wnt activity; however, Wnt abundance and forced fucosylation were not examined in that study.
The major known mechanism to degrade Wnt protein is through Lrp6:Wnt binding (Yamamoto et al, 2008a); thus, we were initially surprised that slc35c1 OE did not diminish Lrp6 abundance, but rather increased it. However, reduced Wnt8a and sustained Lrp6 could account for the elevated basal but flattened Wnt activity gradient observed in Slc35C1 expressing embryos, as Lrp6 has been previously shown to activate canonical Wnt signaling (Tamai et al, 2000). Consistent with this notion, slc35c1 OE are more sensitive to elevated Lrp6 than wild-type embryos. Previous studies of Lrp6 mediated regulation of Wnt signaling indicate that the presence of Lrp6 does not necessarily result in Wnt activation because Lrp6 must be phosphorylated and internalized to activate Wnt signaling (Liang et al, 2011) and reviewed in (Cruciat & Niehrs, 2013). In addition, Lrp6 is known to form higher order complexes with multiple factors that can influence the composition of the higher order complexes as well as whether or not the Wnt signal is transduced. For example, several Wnt inhibitors interact with Lrp6 and block Wnt signaling (e.g. Dkk1 (Sakane et al, 2010; Yamamoto et al, 2008b), IGFBP-4 (Zhu et al, 2008), and Sclerostin domain-containing 1 (Wise/SOST)(Lintern et al, 2009)). Thus, it is possible that Wnt signaling is attenuated despite the excess Lrp6 because it is not in the proper state (e.g. phosphorylation or higher order complex) to activate high levels of Wnt signaling. Another Low-density lipoprotein receptor, LRP-1 depends on core fucosylation for its function in endocytosis (Lee et al, 2006). Previous studies indicate that whether LRP6 functions in the canonical Wnt versus the non-canonical Wnt pathway is regulated at the level of endocytic sorting(Yamamoto et al, 2006; Yamamoto et al, 2008a), an exciting possibility for future exploration is that fucosylation differentially regulates the abundance of Lrp and Wnt ligands via modulation of endocytosis.
Dorsal-ventral patterning in vertebrates requires input from multiple coordinated pathways. A delicate balance between the major pathways, Wnt and TGF-β (BMP and Nodals) controls cell proliferation, differentiation and migration during this early developmental process. We provide evidence that enhanced fucosylation of N-linked glycans disrupts embryonic patterning by inhibiting the canonical Wnt signaling pathway. Although knock down of GDP-mannose 4,6-dehydratase (gmds) the first enzyme in the de novo GDP-Fuc synthetic pathway decreased fucosylation in zebrafish embryos, it did not fully suppress the mSlc35c1 OE phenotype. Because the Gmds protein is maternally loaded (Ohata et al, 2009), it is likely that the doses of gmds MO used only partially disrupted gmds and did not completely eliminate endogenous GDP-Fuc production or deplete the maternal pool of GDP-Fuc. Alternatively, Slc35c1 exerts its effects on Wnt signaling by a mechanism independent of gmds. Notably, the expression of human GDP-fucose transporter Slc35c1 was recently reported to be up-regulated by TGF-β (Xu et al, 2013). Moreover, TGF-beta1 signaling deficiency occurs in alpha1,6-fucosyltransferase (Fut8) null mice, which lack the enzyme that catalyzes the core fucose modification of N-glycans under the direct control of slc35c1(Wang et al, 2005). This work, together with our findings suggests that negative regulatory loops involving slc35c1 may be a common mechanism utilized by diverse signaling pathways to limit signaling activity spatially and temporally; thus, allowing the same factors to be redeployed during multiple stages of morphogenesis such that the embryo can be patterned with a relatively small number of signaling pathways.
Materials and methods
Animal welfare
The animal work performed in this study was approved by the Institutional Review Board of the Albert Einstein College of Medicine.
Fish strains
Notch reporter lines (Parsons et al, 2009), AB, and casper (mitfaw2/w2;roya9/a9) (White et al, 2008) lines were used in this study. All fish were maintained under standard conditions and were staged by morphological characteristics and hours post fertilization (hpf) as described (Kimmel et al, 1995).
Nucleotide sugar treatments
GDP-Fucose and GDP-FucAl were synthesized and purified as previously described (Wang et al, 2009a). UDP-Galactose and UDP-Glucose were gifts from the Wang lab (Ban et al, 2012; Woodward et al, 2010). All nucleotide sugars were resuspended in nuclease free water at a concentration of 30nM. 1nl of solution was injected into 1–4 cell stage embryos unless specifically mentioned.
The dose of GDP-Fuc injected was determined as follows: 10pmol per embryo GDP-Fuc rescues the gmds mutant phenotype (Ohata et al, 2009). Considering maternal contribution, three different doses were tested: 30, 60 and 90pmol of GDP-Fuc per embryo. All three doses had similar survival and abnormality ratios. Therefore, 30pmol was used for all GDP-Fuc injections.
RNA isolation, RNA reverse transcription (RT) and quantitative real-time PCR (qPCR)
RNA was extracted using TRIzol® RNA isolation reagents (Life Technologies, CT, USA). 20 embryos were pooled for RNA isolation for each stage or treated condition examined. RT was carried out as previously described (Feng et al, 2010).
Quantitative PCR was carried out using an Eppendorf Mastercycler® pro system (Eppendorf, NY, USA) with SYBR green fluorescent label. For housekeeping gene loading controls, ornithine decarboxylase 1 (odc1), eukaryotic translation elongation factor 1 alpha 1a (eef1a1a) and glyceraldehyde-3-phosphate dehydrogenase, spermatogenic (gapdhs) genes were selected and compared. odc1 showed the least fluctuation as reported (http://zf-espresso.tuebingen.mpg.de), thus was used as the control amplicon for the slc35c1 and slc35c2 time course. eef1a1a was the most abundant thus was used as the control amplicon for bozozok (dharma), vent and vox. Detailed sample preparation, cycle setting and data analysis were performed as described (Feng et al, 2010). The primer sequences used in this study were as follows: zebrafish dharma (left, 5′-TGGTGAACTTTCCTTGTCCA; right, 5′-TTGCCCTGCATAGTAAGTCG), slc35c1 (left, 5′-CCGGTCTTCATCACCTTCTT; right, 5′-CAGGACACAAGCTTGAGAGG), slc35c2 (left, 5′-CTCATGTTCCTGGGCCTATT; right, 5′-CCACCCACAGACAGAGTGAC); vent (left 5′-ACCGATGTGGAGAGTGATGA, right 5′-GAGATCTGATCGCAGGTGAA); vox (left 5′-TGATGCCTCTTCATCCTCAC; right 5′-ATATTGCACACCAGCGTGAT).
Notch reporter GFP quantification
To measure Notch activity, treated embryos were sorted into 96-well plate with one embryo per well. Quantification of GFP signales was carried out using an Eppendorf Mastercycler® pro system (Eppendorf, NY, USA) using the SYBR green fluorescent channel. All data were normalized to the signal detected in negative embryos, then analyzed using Excel (Microscoft, USA). Post-hoc analysis after ANOVA was used to determine the differences among sample groups.
Constructs, mRNA preparation and injection
The following plasmids were obtained from and used according to the original sources: Mouse slc35c1 (Lu et al, 2010), zebrafish bmp2 and bmp7(Little & Mullins, 2009), zebrafish wnt8a (Lu et al, 2011), Dominant-negative wnt8 (McGrew et al, 1997) and Human Lrp6 (Mao et al, 2001).
The mouse slc35c1 T308R mutation construct was generated using QuikChange Lightning Site-Directed Mutagenesis Kit (Agilent Technologies, Santa Clara, CA,) to change nucleotide 920C to 920G.
To avoid disrupting the three dimensional structure of Wnt8a, we applied an in silico design approach to compare the predicted Flag inserted Wnt8a with wt Wnt8a 3-D structure based on the Xenopus Wnt8 structure (PDB4F0A) (Janda et al, 2012) using Phyre2 online software. Based on the in silico candidates, we opted to insert the Flag tag DNA sequence GACTACAAGGATGACGATGACAAA between 274A and 275R of Wnt8a protein to generate Wnt8a-Flag. The DN-wnt8 Flag construct was created by inserting the Flag tag sequence 5′ to the TGA stop codon. All insertions were introduced using the QuikChange Kit. All changes were confirmed by sequencing.
mRNA was synthesized from linearized DNA using the mMessenger Machine kit (Ambion, TX, USA). The amount of mRNA injected per embryo was as following: mouse slc35c1 1ng, unless specifically mentioned otherwise, 15 pg zebrafish bmp2b, 50 pg bmp7(Little & Mullins, 2009), 50pg wnt8a (Lu et al, 2011), 500pg Dominant-negative wnt8 (McGrew et al, 1997); 300 pg Lrp6.
Immunofluorescence
Antibody staining of whole-mounted embryos with β-catenin was performed as described (Lyman Gingerich et al, 2005) with the following exceptions. Embryos were incubated with a 1:500 dilution of either alexa 488, or alexa 533-labeled rabbit anti-mouse secondary antibody (Molecular Probes, USA). Nuclei were stained with 0.5μg/ml bisBenzimide H 33342 trihydrochloride (Hoechst 33342) in PBS (Sigma-Aldrich, Germany) overnight. Images were taken using Leica TCS SP5 confocal microscope (Leica Microsystems, Wetzlar, Germany) and projected using ImageJ software.
Chimeric analysis and nuclear β-catenin quantification
To determine the autonomy of Slc35c1 function on Wnt signaling, donor embryos were injected with Dextran–Alexa 488 (Molecular Probes, Eugene, OR) and RNA encoding wnt8a with or without slc35c1, host embryos were injected with/without slc35c1 mRNA. Chimeras were made using standard methods(Westerfield, 1995). After 1 hour, embryos were fixed and β-catenin staining was performed as previously described (Tang et al, 2012).
To measure Wnt8a activity, we set nuclear β-catenin abundance as the direct readout. For the nuclear β-catenin quantification, 8 image sections (0.3μm each to make 2.4μm section) were projected into a single figure for all individual color channels. All three channels were combined together using Photoshop CS5 (Adobe, San Jose, CA, USA). Donor cells were identified by the Dextran–Alexa 488, cells were divided into groups according to proximity (cell diameters from) the donor cells: the cells directly adjacent to donor cells were named N1 cells; then, N2, N3 etc. Background cells were at least 4 cells away from donor cells. Nuclear β-catenin abundance was measured manually, by circling the nuclei area (as shown in Fig. 5B). Only the nuclei at the S to G stage were used for quantification and all data were normalized to the background cell intensity from the same picture, which was set to be 100%. All data were pooled according to their location and grouped as donor cells, N1 cells and N2 cell. The average and standard deviation were measured from the pools. Data analysis was performed as described (Feng et al, 2010), except after one-way analysis of variance (ANOVA), Tukey’s HSD test was used to determine which groups differed from each other. Analysis of mSlc35c1 OE donor cells transplanted to wild-type hosts showed no changes in nuclear β-Catenin abundance in donor or host cells after transplantation, indicating the transplantation procedure does not perturb activity (Fig. S5).
Morpholino
The injection of gmds MO (5′-ACCCATCCTGAAACCACATAGAGGG-3′) was 2ng per embryo as published before (Ohata et al, 2009).
RNA in situ hybridization
RNA in situ hybridization was performed as described (Feng et al, 2010; Prince et al, 1998). The probes used in this study: bozozok, chordin, eve1, gsc, egr2b (krox20), myoD, pax2a and squint (ndr1) were obtained from Dr. Solnica-Krezel (Washington University, Saint Louis, USA). Unless otherwise stated, images of whole mount embryos were acquired using an Olympus SZ61 dissecting microscope equipped with a high-resolution digital camera (model S97809, Olympus America) and Picture Frame 2.0 or 3.0 software (Optronics). Two-color in situ hybridization embryos were flat mounted as described and images were acquired using an AxioPlan2 microscope equipped with an AxioCam CCD camera (Zeiss). Images of fluorescent in situ hybridization embryos were acquired using Leica SP5 confocal. GFP reporter expression of the Tp1bglob:egfp line was acquired using an Olympus SZ16 fluorescent dissecting microscope and Microfire digital camera (Olympus). Images were processed in ImageJ, Adobe Photoshop, and Adobe Illustrator.
Immuno-precipitation (IP) and western blot
Antibody IP and western blot for Flag tagged proteins were generally performed as described (Little & Mullins, 2009). For Wnt8a-Flag detection, 67 embryos were pooled for lysate and IP; for DN-Wnt8-Flag detection, 33 embryos were pooled for IP for both the DN-Wnt8 injected and WT control samples; for the DN-Wnt8-Flag and Lrp6-Flag interaction experiment, 64 embryos were pooled for IP. Protein equal to two embryo equivalents was used for each sample.
Antibody IP and western blot for Myc tagged mSlc35c1 and mSlc35c1 T308R proteins were performed as described (Lu et al, 2010). Except zebrafish embryo lysates rather than cell lysates were used for analysis. For mSlc35c1-Myc and mSlc35c1 T308R protein detection, 20 embryos were pooled. Protein equal to two embryo equivalents was used for western analysis.
Western blot for P-Smad1/5/8 was performed as described (Little & Mullins, 2009), except 20 shield stage embryos per condition were manually deyolked, frozen immediately in liquid nitrogen after deyolking, and lysed in SDS loading buffer just before running the gel.
To analyze endogenous Lrp6 fucosylation, GDP-FucAl was injected into 1–4 cell stage embryos and 45 embryos were collected at sphere stage (4hpf) for protein. MBS buffer was used to deyolk as described (Cousin et al, 2012). Next a click reaction to link biotin to the Alkyne group containing fucose was performed as previously described (Besanceney-Webler et al, 2011). A mouse monoclonal antibody for Lrp6 [1C10] (2μg per IP, Abcam,) followed by Protein G resin (3μl per IP, Genscript) was used to enrich for zebrafish Lrp6 IP. 1C10 (1:1,000) was also used for western blot. FucAl was detected through the linked biotin using a mouse anti-biotin HRP (1:10,000, Invitrogen). For Aleuria Aurantia Lectin (AAL) staining, we manually deyolked embryos to circumvent yolk protein interference with the detection of fucose with AAL. 20 embryos per treatment were manually deyolked at shield stage, then pooled and lysed in 2 x SDS sampling buffer. Protein equal to 1.5 embryo equivalents per sample were separated using SDS-PAGE and detected by IB with biotinylated AAL (1:200, Vector lab B-1395) and mouse anti-biotin HRP (1:10,000). Rabbit anti-actin or anti-α-tubulin antibodies (1:500, Millipore, ABT170) were used as loading controls for the western blot as described (Tucker et al, 2008). For all loading control IB, each lane represents the protein equivalent of one embryo, except where stated otherwise.
PNGase F digestion and AAL quantification
Peptide-N-Glycosidase F(PNGase F) (New England Biolabs, USA) digestion was performed on nitro-cellulose membranes after transfer of proteins. For an 8X6 cm2 membrane, 5μl of enzyme was used in a total volume of 10ml in a buffer as described in the manufactures protocol. For PNGase F digestion, duplicate samples were loaded and SDS-PAGE was conducted. After transferring to nitro-cellulose film, one sample was digested with PNGase F and the duplicate was incubated with only digestion buffer. Digestion was performed in a sealed plastic bag on a shaker at 37°C for 16 hours. The digestion time was determined by comparing the AAL staining signal on the same sample for 6, 12, 16, 24 and 48-hour periods. Results showed no difference for any samples with digestion over 12-hour. After digestion, the PNGase F was washed 3 times with PBST buffer for 3 times and AAL IB was simultaneously performed on both membranes.
AAL immune-blot was carried out as follows: Membranes were blocked with 1 X Western blotting reagent solution (Roche, USA) in PBST, incubated with Biotin-AAL, which was then incubated with HRP-mouse anti-Biotin. AAL detection and quantification were performed using Streptavidin Alexa-488 (1:300, Invitrogen) to detect Biotin signals. AAL abundance was scanned using Storm 840 (GE, Healthcare) and quantified using ImageJ software.
TOP-flash assays
Top-flash assays were performed as reported in (Mao et al, 2002; Mao et al, 2001) using well-established engineered CHO cell lines with different repertoires of glycan modification (Patnaik & Stanley, 2006; Stanley et al, 1996). To control for potential differential responses between cell lines, we performed in vitro fucosylation assays as described (Zheng et al, 2011). Since both CHO and LEC30 lines have endogenous Wnt activity, we normalized all data to untreated CHO or LEC30 lines, which we set as 100%. After a 20 minute in vitro fucosylation treatment, the cells were washed once with Dulbecco’s Phosphate Buffered Saline (DPBS), then treated with the Wnt8a containing serum produced by CHO or LEC30 cells for 6 hours before performing the dual luciferase assay.
Supplementary Material
Highlights.
Enhanced N-linked fucosylation disrupts zebrafish dorsal-ventral patterning
Wnt signaling is reduced when N-linked fucosylation is enhanced
Elevated N-fucosylation in receiving cells diminishes the range of Wnt signaling
Wnt8 and LRP6 are targets for N-fucosylation in zebrafish
Excess Wnt8 increases slc35c1 expression
Acknowledgments
We are grateful to Lilianna Solnica-Krezel, William S. Talbot, David Vocadlo, Pamela Stanley, and members of the Marlow lab for discussions and manuscript evaluation. We thank S. Kalinin and C. Depaolo for fish care and Dr. P. Guo for technical assistance with confocal imaging. Confocal images were acquired using the Einstein Analytical Imaging Facility. This work was supported in part by NIHRO1GM089979 and start-up funds to FLM and NIHR01GM093282 and The Mizutani Foundation for Glycoscience funds to PW.
Footnotes
Author contributions
Conceived and designed the experiments: L.F, H.J., P.W. and F.L.M. Performed the experiments: L.F and H.J. Analyzed the data: L.F, H.J., P.W. and F.L.M. All authors discussed the data and the manuscript. L.F. P.W. and F.L.M. wrote the manuscript.
Conflict of Interest
All the authors agreed to the manuscript’s contents. The authors do not have any financial conflict of interest that might be construed to influence the results or interpretation of the manuscript.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ban L, Pettit N, Li L, Stuparu AD, Cai L, Chen W, Guan W, Han W, Wang PG, Mrksich M. Discovery of glycosyltransferases using carbohydrate arrays and mass spectrometry. Nat Chem Biol. 2012;8:769–773. doi: 10.1038/nchembio.1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker DJ, Lowe JB. Fucose: biosynthesis and biological function in mammals. Glycobiology. 2003;13:41R–53R. doi: 10.1093/glycob/cwg054. [DOI] [PubMed] [Google Scholar]
- Besanceney-Webler C, Jiang H, Zheng T, Feng L, Soriano del Amo D, Wang W, Klivansky LM, Marlow FL, Liu Y, Wu P. Increasing the efficacy of bioorthogonal click reactions for bioconjugation: a comparative study. Angew Chem Int Ed Engl. 2011;50:8051–8056. doi: 10.1002/anie.201101817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Z, Biechele T, Wei Z, Morrone S, Moon RT, Wang L, Xu W. Crystal structures of the extracellular domain of LRP6 and its complex with DKK1. Nat Struct Mol Biol. 2011;18:1204–1210. doi: 10.1038/nsmb.2139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheyette BN, Waxman JS, Miller JR, Takemaru K, Sheldahl LC, Khlebtsova N, Fox EP, Earnest T, Moon RT. Dapper, a Dishevelled-associated antagonist of beta-catenin and JNK signaling, is required for notochord formation. Dev Cell. 2002;2:449–461. doi: 10.1016/s1534-5807(02)00140-5. [DOI] [PubMed] [Google Scholar]
- Chong JM, Uren A, Rubin JS, Speicher DW. Disulfide bond assignments of secreted Frizzled-related protein-1 provide insights about Frizzled homology and netrin modules. J Biol Chem. 2002;277:5134–5144. doi: 10.1074/jbc.M108533200. [DOI] [PubMed] [Google Scholar]
- Cousin H, Abbruzzese G, McCusker C, Alfandari D. ADAM13 function is required in the 3 dimensional context of the embryo during cranial neural crest cell migration in Xenopus laevis. Dev Biol. 2012;368:335–344. doi: 10.1016/j.ydbio.2012.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruciat CM, Niehrs C. Secreted and transmembrane wnt inhibitors and activators. Cold Spring Harbor perspectives in biology. 2013;5:a015081. doi: 10.1101/cshperspect.a015081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dehnert KW, Beahm BJ, Huynh TT, Baskin JM, Laughlin ST, Wang W, Wu P, Amacher SL, Bertozzi CR. Metabolic labeling of fucosylated glycans in developing zebrafish. ACS Chem Biol. 2011;6:547–552. doi: 10.1021/cb100284d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domino SE, Zhang L, Gillespie PJ, Saunders TL, Lowe JB. Deficiency of reproductive tract alpha(1,2)fucosylated glycans and normal fertility in mice with targeted deletions of the FUT1 or FUT2 alpha(1,2)fucosyltransferase locus. Mol Cell Biol. 2001;21:8336–8345. doi: 10.1128/MCB.21.24.8336-8345.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du J, Takeuchi H, Leonhard-Melief C, Shroyer KR, Dlugosz M, Haltiwanger RS, Holdener BC. O-fucosylation of thrombospondin type 1 repeats restricts epithelial to mesenchymal transition (EMT) and maintains epiblast pluripotency during mouse gastrulation. Dev Biol. 2010;346:25–38. doi: 10.1016/j.ydbio.2010.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erter CE, Wilm TP, Basler N, Wright CV, Solnica-Krezel L. Wnt8 is required in lateral mesendodermal precursors for neural posteriorization in vivo. Development. 2001;128:3571–3583. doi: 10.1242/dev.128.18.3571. [DOI] [PubMed] [Google Scholar]
- Etzioni A, Sturla L, Antonellis A, Green ED, Gershoni-Baruch R, Berninsone PM, Hirschberg CB, Tonetti M. Leukocyte adhesion deficiency (LAD) type II/carbohydrate deficient glycoprotein (CDG) IIc founder effect and genotype/phenotype correlation. Am J Med Genet. 2002;110:131–135. doi: 10.1002/ajmg.10423. [DOI] [PubMed] [Google Scholar]
- Fekany K, Yamanaka Y, Leung T, Sirotkin HI, Topczewski J, Gates MA, Hibi M, Renucci A, Stemple D, Radbill A, Schier AF, Driever W, Hirano T, Talbot WS, Solnica-Krezel L. The zebrafish bozozok locus encodes Dharma, a homeodomain protein essential for induction of gastrula organizer and dorsoanterior embryonic structures. Development. 1999;126:1427–1438. doi: 10.1242/dev.126.7.1427. [DOI] [PubMed] [Google Scholar]
- Fekany-Lee K, Gonzalez E, Miller-Bertoglio V, Solnica-Krezel L. The homeobox gene bozozok promotes anterior neuroectoderm formation in zebrafish through negative regulation of BMP2/4 and Wnt pathways. Development. 2000;127:2333–2345. doi: 10.1242/dev.127.11.2333. [DOI] [PubMed] [Google Scholar]
- Feng L, Hernandez RE, Waxman JS, Yelon D, Moens CB. Dhrs3a regulates retinoic acid biosynthesis through a feedback inhibition mechanism. Dev Biol. 2010;338:1–14. doi: 10.1016/j.ydbio.2009.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flowers GP, Topczewska JM, Topczewski J. A zebrafish Notum homolog specifically blocks the Wnt/beta-catenin signaling pathway. Development. 2012;139:2416–2425. doi: 10.1242/dev.063206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisler C, Kotu V, Sharrow M, Rendic D, Poltl G, Tiemeyer M, Wilson IB, Jarvis DL. The Drosophila neurally altered carbohydrate mutant has a defective Golgi GDP-fucose transporter. J Biol Chem. 2012;287:29599–29609. doi: 10.1074/jbc.M112.379313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez E, Fekany-Lee K, Carmany-Rampey A, Erter C, Topczewski J, Wright CVE, Solnica-Krezel L. Head and trunk development in zebrafish requires inhibition of Bmp signaling by bozozok and chordino. Genes and Development. 2000 doi: 10.1101/gad.852400. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross JC, Boutros M. Secretion and extracellular space travel of Wnt proteins. Curr Opin Genet Dev. 2013;23:385–390. doi: 10.1016/j.gde.2013.02.017. [DOI] [PubMed] [Google Scholar]
- Gu W, Fukuda T, Isaji T, Hashimoto H, Wang Y, Gu J. alpha1,6-Fucosylation regulates neurite formation via the activin/phospho-Smad2 pathway in PC12 cells: the implicated dual effects of Fut8 for TGF-beta/activin-mediated signaling. Faseb J. 2013 doi: 10.1096/fj.12-225805. [DOI] [PubMed] [Google Scholar]
- Haniu M, Horan T, Spahr C, Hui J, Fan W, Chen C, Richards WG, Lu HS. Human Dickkopf-1 (huDKK1) protein: characterization of glycosylation and determination of disulfide linkages in the two cysteine-rich domains. Protein Sci. 2011;20:1802–1813. doi: 10.1002/pro.705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayes M, Naito M, Daulat A, Angers S, Ciruna B. Ptk7 promotes non-canonical Wnt/PCP-mediated morphogenesis and inhibits Wnt/beta-catenin-dependent cell fate decisions during vertebrate development. Development. 2013;140:1807–1818. doi: 10.1242/dev.090183. [DOI] [PubMed] [Google Scholar]
- Hellbusch CC, Sperandio M, Frommhold D, Yakubenia S, Wild MK, Popovici D, Vestweber D, Grone HJ, von Figura K, Lubke T, Korner C. Golgi GDP-fucose transporter-deficient mice mimic congenital disorder of glycosylation IIc/leukocyte adhesion deficiency II. J Biol Chem. 2007;282:10762–10772. doi: 10.1074/jbc.M700314200. [DOI] [PubMed] [Google Scholar]
- Hikasa H, Sokol SY. Phosphorylation of TCF proteins by homeodomain-interacting protein kinase 2. J Biol Chem. 2011;286:12093–12100. doi: 10.1074/jbc.M110.185280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homeister JW, Thall AD, Petryniak B, Maly P, Rogers CE, Smith PL, Kelly RJ, Gersten KM, Askari SW, Cheng G, Smithson G, Marks RM, Misra AK, Hindsgaul O, von Andrian UH, Lowe JB. The alpha(1,3)fucosyltransferases FucT-IV and FucT-VII exert collaborative control over selectin-dependent leukocyte recruitment and lymphocyte homing. Immunity. 2001;15:115–126. doi: 10.1016/s1074-7613(01)00166-2. [DOI] [PubMed] [Google Scholar]
- Hoppler S, Brown JD, Moon RT. Expression of a dominant-negative Wnt blocks induction of MyoD in Xenopus embryos. Genes Dev. 1996;10:2805–2817. doi: 10.1101/gad.10.21.2805. [DOI] [PubMed] [Google Scholar]
- Huang HW, Chen CH, Lin CH, Wong CH, Lin KI. B-cell maturation antigen is modified by a single N-glycan chain that modulates ligand binding and surface retention. Proc Natl Acad Sci U S A. 2013;110:10928–10933. doi: 10.1073/pnas.1309417110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishikawa HO, Ayukawa T, Nakayama M, Higashi S, Kamiyama S, Nishihara S, Aoki K, Ishida N, Sanai Y, Matsuno K. Two pathways for importing GDP-fucose into the endoplasmic reticulum lumen function redundantly in the O-fucosylation of Notch in Drosophila. J Biol Chem. 2010;285:4122–4129. doi: 10.1074/jbc.M109.016964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh K, Sokol SY. Axis determination by inhibition of Wnt signaling in Xenopus. Genes Dev. 1999;13:2328–2336. doi: 10.1101/gad.13.17.2328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janda CY, Waghray D, Levin AM, Thomas C, Garcia KC. Structural basis of Wnt recognition by Frizzled. Science. 2012;337:59–64. doi: 10.1126/science.1222879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang H, Feng L, Soriano del Amo D, Seidel RD, III, Marlow F, Wu P. Imaging glycans in zebrafish embryos by metabolic labeling and bioorthogonal click chemistry. J Vis Exp. 2011 doi: 10.3791/2686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joiner DM, Ke J, Zhong Z, Xu HE, Williams BO. LRP5 and LRP6 in development and disease. Trends Endocrinol Metab. 2013;24:31–39. doi: 10.1016/j.tem.2012.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung H, Lee SK, Jho EH. Mest/Peg1 inhibits Wnt signalling through regulation of LRP6 glycosylation. Biochem J. 2011;436:263–269. doi: 10.1042/BJ20101512. [DOI] [PubMed] [Google Scholar]
- Kamio K, Yoshida T, Gao C, Ishii T, Ota F, Motegi T, Kobayashi S, Fujinawa R, Ohtsubo K, Kitazume S, Angata T, Azuma A, Gemma A, Nishimura M, Betsuyaku T, Kida K, Taniguchi N. alpha1,6-Fucosyltransferase (Fut8) is implicated in vulnerability to elastase-induced emphysema in mice and a possible non-invasive predictive marker for disease progression and exacerbations in chronic obstructive pulmonary disease (COPD) Biochem Biophys Res Commun. 2012;424:112–117. doi: 10.1016/j.bbrc.2012.06.081. [DOI] [PubMed] [Google Scholar]
- Kelly C, Chin AJ, Leatherman JL, Kozlowski DJ, Weinberg ES. Maternally controlled (beta)-catenin-mediated signaling is required for organizer formation in the zebrafish. Development. 2000;127:3899–3911. doi: 10.1242/dev.127.18.3899. [DOI] [PubMed] [Google Scholar]
- Kelly GM, Greenstein P, Erezyilmaz DF, Moon RT. Zebrafish wnt8 and wnt8b share a common activity but are involved in distinct developmental pathways. Development. 1995;121:1787–1799. doi: 10.1242/dev.121.6.1787. [DOI] [PubMed] [Google Scholar]
- Khan Z, Vijayakumar S, de la Torre TV, Rotolo S, Bafico A. Analysis of endogenous LRP6 function reveals a novel feedback mechanism by which Wnt negatively regulates its receptor. Mol Cell Biol. 2007;27:7291–7301. doi: 10.1128/MCB.00773-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim CH, Oda T, Itoh M, Jiang D, Artinger KB, Chandrasekharappa SC, Driever W, Chitnis AB. Repressor activity of Headless/Tcf3 is essential for vertebrate head formation. Nature. 2000;407:913–916. doi: 10.1038/35038097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimelman D, Christian JL, Moon RT. Synergistic principles of development: overlapping patterning systems in Xenopus mesoderm induction. Development. 1992;116:1–9. doi: 10.1242/dev.116.Supplement.1. [DOI] [PubMed] [Google Scholar]
- Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF. Stages of embryonic development of the zebrafish. Dev Dyn. 1995;203:253–310. doi: 10.1002/aja.1002030302. [DOI] [PubMed] [Google Scholar]
- Kudo T, Kaneko M, Iwasaki H, Togayachi A, Nishihara S, Abe K, Narimatsu H. Normal embryonic and germ cell development in mice lacking alpha 1,3-fucosyltransferase IX (Fut9) which show disappearance of stage-specific embryonic antigen 1. Mol Cell Biol. 2004;24:4221–4228. doi: 10.1128/MCB.24.10.4221-4228.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langdon YG, Mullins MC. Maternal and zygotic control of zebrafish dorsoventral axial patterning. Annu Rev Genet. 2011;45:357–377. doi: 10.1146/annurev-genet-110410-132517. [DOI] [PubMed] [Google Scholar]
- Lee SH, Takahashi M, Honke K, Miyoshi E, Osumi D, Sakiyama H, Ekuni A, Wang X, Inoue S, Gu J, Kadomatsu K, Taniguchi N. Loss of core fucosylation of low-density lipoprotein receptor-related protein-1 impairs its function, leading to the upregulation of serum levels of insulin-like growth factor-binding protein 3 in Fut8−/− mice. J Biochem. 2006;139:391–398. doi: 10.1093/jb/mvj039. [DOI] [PubMed] [Google Scholar]
- Lekven AC, Thorpe CJ, Waxman JS, Moon RT. Zebrafish wnt8 encodes two wnt8 proteins on a bicistronic transcript and is required for mesoderm and neurectoderm patterning. Dev Cell. 2001;1:103–114. doi: 10.1016/s1534-5807(01)00007-7. [DOI] [PubMed] [Google Scholar]
- Li Y, Lu W, King TD, Liu CC, Bijur GN, Bu G. Dkk1 stabilizes Wnt co-receptor LRP6: implication for Wnt ligand-induced LRP6 down-regulation. PLoS One. 2010;5:e11014. doi: 10.1371/journal.pone.0011014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang J, Fu Y, Cruciat CM, Jia S, Wang Y, Tong Z, Tao Q, Ingelfinger D, Boutros M, Meng A, Niehrs C, Wu W. Transmembrane protein 198 promotes LRP6 phosphorylation and Wnt signaling activation. Molecular and cellular biology. 2011;31:2577–2590. doi: 10.1128/MCB.05103-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim S, Kumari P, Gilligan P, Quach HN, Mathavan S, Sampath K. Dorsal activity of maternal squint is mediated by a non-coding function of the RNA. Development. 2012;139:2903–2915. doi: 10.1242/dev.077081. [DOI] [PubMed] [Google Scholar]
- Lintern KB, Guidato S, Rowe A, Saldanha JW, Itasaki N. Characterization of wise protein and its molecular mechanism to interact with both Wnt and BMP signals. The Journal of biological chemistry. 2009;284:23159–23168. doi: 10.1074/jbc.M109.025478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Little SC, Mullins MC. Bone morphogenetic protein heterodimers assemble heteromeric type I receptor complexes to pattern the dorsoventral axis. Nat Cell Biol. 2009;11:637–643. doi: 10.1038/ncb1870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu JX, Zhang D, Xie X, Ouyang G, Liu X, Sun Y, Xiao W. Eaf1 and Eaf2 negatively regulate canonical Wnt/beta-catenin signaling. Development. 2013;140:1067–1078. doi: 10.1242/dev.086157. [DOI] [PubMed] [Google Scholar]
- Liu YC, Yen HY, Chen CY, Chen CH, Cheng PF, Juan YH, Khoo KH, Yu CJ, Yang PC, Hsu TL, Wong CH. Sialylation and fucosylation of epidermal growth factor receptor suppress its dimerization and activation in lung cancer cells. Proc Natl Acad Sci U S A. 2011;108:11332–11337. doi: 10.1073/pnas.1107385108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logan CY, Nusse R. The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol. 2004;20:781–810. doi: 10.1146/annurev.cellbio.20.010403.113126. [DOI] [PubMed] [Google Scholar]
- Lu FI, Thisse C, Thisse B. Identification and mechanism of regulation of the zebrafish dorsal determinant. Proc Natl Acad Sci U S A. 2011;108:15876–15880. doi: 10.1073/pnas.1106801108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu L, Hou X, Shi S, Korner C, Stanley P. Slc35c2 promotes Notch1 fucosylation and is required for optimal Notch signaling in mammalian cells. J Biol Chem. 2010;285:36245–36254. doi: 10.1074/jbc.M110.126003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lubke T, Marquardt T, Etzioni A, Hartmann E, von Figura K, Korner C. Complementation cloning identifies CDG-IIc, a new type of congenital disorders of glycosylation, as a GDP-fucose transporter deficiency. Nat Genet. 2001;28:73–76. doi: 10.1038/ng0501-73. [DOI] [PubMed] [Google Scholar]
- Luhn K, Wild MK, Eckhardt M, Gerardy-Schahn R, Vestweber D. The gene defective in leukocyte adhesion deficiency II encodes a putative GDP-fucose transporter. Nat Genet. 2001;28:69–72. doi: 10.1038/ng0501-69. [DOI] [PubMed] [Google Scholar]
- Lyman Gingerich J, Westfall TA, Slusarski DC, Pelegri F. hecate, a zebrafish maternal effect gene, affects dorsal organizer induction and intracellular calcium transient frequency. Dev Biol. 2005;286:427–439. doi: 10.1016/j.ydbio.2005.07.031. [DOI] [PubMed] [Google Scholar]
- Ma B, Simala-Grant JL, Taylor DE. Fucosylation in prokaryotes and eukaryotes. Glycobiology. 2006;16:158R–184R. doi: 10.1093/glycob/cwl040. [DOI] [PubMed] [Google Scholar]
- Maley F, Trimble RB, Tarentino AL, Plummer TH., Jr Characterization of glycoproteins and their associated oligosaccharides through the use of endoglycosidases. Anal Biochem. 1989;180:195–204. doi: 10.1016/0003-2697(89)90115-2. [DOI] [PubMed] [Google Scholar]
- Malinauskas T, Aricescu AR, Lu W, Siebold C, Jones EY. Modular mechanism of Wnt signaling inhibition by Wnt inhibitory factor 1. Nat Struct Mol Biol. 2011;18:886–893. doi: 10.1038/nsmb.2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maly P, Thall A, Petryniak B, Rogers CE, Smith PL, Marks RM, Kelly RJ, Gersten KM, Cheng G, Saunders TL, Camper SA, Camphausen RT, Sullivan FX, Isogai Y, Hindsgaul O, von Andrian UH, Lowe JB. The alpha(1,3)fucosyltransferase Fuc-TVII controls leukocyte trafficking through an essential role in L-, E-, and P-selectin ligand biosynthesis. Cell. 1996;86:643–653. doi: 10.1016/s0092-8674(00)80137-3. [DOI] [PubMed] [Google Scholar]
- Mao B, Wu W, Davidson G, Marhold J, Li M, Mechler BM, Delius H, Hoppe D, Stannek P, Walter C, Glinka A, Niehrs C. Kremen proteins are Dickkopf receptors that regulate Wnt/beta-catenin signalling. Nature. 2002;417:664–667. doi: 10.1038/nature756. [DOI] [PubMed] [Google Scholar]
- Mao B, Wu W, Li Y, Hoppe D, Stannek P, Glinka A, Niehrs C. LDL-receptor-related protein 6 is a receptor for Dickkopf proteins. Nature. 2001;411:321–325. doi: 10.1038/35077108. [DOI] [PubMed] [Google Scholar]
- Marlow FL. Maternal Control of Development in Vertebrates: My Mother Made Me Do It! San Rafael (CA): 2010. [PubMed] [Google Scholar]
- McGrew LL, Hoppler S, Moon RT. Wnt and FGF pathways cooperatively pattern anteroposterior neural ectoderm in Xenopus. Mech Dev. 1997;69:105–114. doi: 10.1016/s0925-4773(97)00160-3. [DOI] [PubMed] [Google Scholar]
- McGrew LL, Lai CJ, Moon RT. Specification of the anteroposterior neural axis through synergistic interaction of the Wnt signaling cascade with noggin and follistatin. Dev Biol. 1995;172:337–342. doi: 10.1006/dbio.1995.0027. [DOI] [PubMed] [Google Scholar]
- Melby AE, Beach C, Mullins M, Kimelman D. Patterning the early zebrafish by the opposing actions of bozozok and vox/vent. Dev Biol. 2000;224:275–285. doi: 10.1006/dbio.2000.9780. [DOI] [PubMed] [Google Scholar]
- Mintzer KA, Lee MA, Runke G, Trout J, Whitman M, Mullins MC. Lost-a-fin encodes a type I BMP receptor, Alk8, acting maternally and zygotically in dorsoventral pattern formation. Development. 2001;128:859–869. doi: 10.1242/dev.128.6.859. [DOI] [PubMed] [Google Scholar]
- Moriwaki K, Noda K, Nakagawa T, Asahi M, Yoshihara H, Taniguchi N, Hayashi N, Miyoshi E. A high expression of GDP-fucose transporter in hepatocellular carcinoma is a key factor for increases in fucosylation. Glycobiology. 2007;17:1311–1320. doi: 10.1093/glycob/cwm094. [DOI] [PubMed] [Google Scholar]
- Nikaido M, Tada M, Saji T, Ueno N. Conservation of BMP signaling in zebrafish mesoderm patterning. Mech Dev. 1997;61:75–88. doi: 10.1016/s0925-4773(96)00625-9. [DOI] [PubMed] [Google Scholar]
- Ohata S, Kinoshita S, Aoki R, Tanaka H, Wada H, Tsuruoka-Kinoshita S, Tsuboi T, Watabe S, Okamoto H. Neuroepithelial cells require fucosylated glycans to guide the migration of vagus motor neuron progenitors in the developing zebrafish hindbrain. Development. 2009;136:1653–1663. doi: 10.1242/dev.033290. [DOI] [PubMed] [Google Scholar]
- Okamura Y, Saga Y. Pofut1 is required for the proper localization of the Notch receptor during mouse development. Mech Dev. 2008;125:663–673. doi: 10.1016/j.mod.2008.04.007. [DOI] [PubMed] [Google Scholar]
- Park DS, Seo JH, Hong M, Choi SC. Role of the Rap2/TNIK kinase pathway in regulation of LRP6 stability for Wnt signaling. Biochem Biophys Res Commun. 2013;436:338–343. doi: 10.1016/j.bbrc.2013.05.104. [DOI] [PubMed] [Google Scholar]
- Parsons MJ, Pisharath H, Yusuff S, Moore JC, Siekmann AF, Lawson N, Leach SD. Notch-responsive cells initiate the secondary transition in larval zebrafish pancreas. Mechanisms of development. 2009;126:898–912. doi: 10.1016/j.mod.2009.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patnaik SK, Stanley P. Lectin-resistant CHO glycosylation mutants. Methods Enzymol. 2006;416:159–182. doi: 10.1016/S0076-6879(06)16011-5. [DOI] [PubMed] [Google Scholar]
- Patnaik SK, Zhang A, Shi S, Stanley P. alpha(1,3)fucosyltransferases expressed by the gain-of-function Chinese hamster ovary glycosylation mutants LEC12, LEC29, and LEC30. Arch Biochem Biophys. 2000;375:322–332. doi: 10.1006/abbi.1999.1693. [DOI] [PubMed] [Google Scholar]
- Pinson KI, Brennan J, Monkley S, Avery BJ, Skarnes WC. An LDL-receptor-related protein mediates Wnt signalling in mice. Nature. 2000;407:535–538. doi: 10.1038/35035124. [DOI] [PubMed] [Google Scholar]
- Potvin B, Stanley P. Activation of two new alpha(1,3)fucosyltransferase activities in Chinese hamster ovary cells by 5-azacytidine. Cell Regul. 1991;2:989–1000. doi: 10.1091/mbc.2.12.989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prince VE, Moens CB, Kimmel CB, Ho RK. Zebrafish hox genes: expression in the hindbrain region of wild-type and mutants of the segmentation gene, valentino. Development. 1998;125:393–406. doi: 10.1242/dev.125.3.393. [DOI] [PubMed] [Google Scholar]
- Roos D, Law SK. Hematologically important mutations: leukocyte adhesion deficiency. Blood Cells Mol Dis. 2001;27:1000–1004. doi: 10.1006/bcmd.2001.0473. [DOI] [PubMed] [Google Scholar]
- Ryu SL, Fujii R, Yamanaka Y, Shimizu T, Yabe T, Hirata T, Hibi M, Hirano T. Regulation of dharma/bozozok by the Wnt pathway. Dev Biol. 2001;231:397–409. doi: 10.1006/dbio.2000.0150. [DOI] [PubMed] [Google Scholar]
- Sakane H, Yamamoto H, Kikuchi A. LRP6 is internalized by Dkk1 to suppress its phosphorylation in the lipid raft and is recycled for reuse. Journal of cell science. 2010;123:360–368. doi: 10.1242/jcs.058008. [DOI] [PubMed] [Google Scholar]
- Schier AF, Talbot WS. Molecular genetics of axis formation in zebrafish. Annu Rev Genet. 2005;39:561–613. doi: 10.1146/annurev.genet.37.110801.143752. [DOI] [PubMed] [Google Scholar]
- Shao L, Haltiwanger RS. O-fucose modifications of epidermal growth factor-like repeats and thrombospondin type 1 repeats: unusual modifications in unusual places. Cell Mol Life Sci. 2003;60:241–250. doi: 10.1007/s000180300019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu T, Yamanaka Y, Ryu SL, Hashimoto H, Yabe T, Hirata T, Bae YK, Hibi M, Hirano T. Cooperative roles of Bozozok/Dharma and Nodal-related proteins in the formation of the dorsal organizer in zebrafish. Mech Dev. 2000;91:293–303. doi: 10.1016/s0925-4773(99)00319-6. [DOI] [PubMed] [Google Scholar]
- Singh R, Smith E, Fathzadeh M, Liu W, Go GW, Subrahmanyan L, Faramarzi S, McKenna W, Mani A. Rare Nonconservative LRP6 Mutations Are Associated with Metabolic Syndrome. Hum Mutat. 2013;34:1221–1225. doi: 10.1002/humu.22360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sirotkin HI, Dougan ST, Schier AF, Talbot WS. bozozok and squint act in parallel to specify dorsal mesoderm and anterior neuroectoderm in zebrafish. Development. 2000;127:2583–2592. doi: 10.1242/dev.127.12.2583. [DOI] [PubMed] [Google Scholar]
- Smith PL, Myers JT, Rogers CE, Zhou L, Petryniak B, Becker DJ, Homeister JW, Lowe JB. Conditional control of selectin ligand expression and global fucosylation events in mice with a targeted mutation at the FX locus. J Cell Biol. 2002;158:801–815. doi: 10.1083/jcb.200203125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solnica-Krezel L, Driever W. The role of the homeodomain protein Bozozok in zebrafish axis formation. Int J Dev Biol. 2001;45:299–310. [PubMed] [Google Scholar]
- Soriano Del Amo D, Wang W, Jiang H, Besanceney C, Yan AC, Levy M, Liu Y, Marlow FL, Wu P. Biocompatible copper(I) catalysts for in vivo imaging of glycans. J Am Chem Soc. 2010;132:16893–16899. doi: 10.1021/ja106553e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanley P. Regulation of Notch signaling by glycosylation. Curr Opin Struct Biol. 2007;17:530–535. doi: 10.1016/j.sbi.2007.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanley P, Okajima T. Roles of glycosylation in Notch signaling. Curr Top Dev Biol. 2010;92:131–164. doi: 10.1016/S0070-2153(10)92004-8. [DOI] [PubMed] [Google Scholar]
- Stanley P, Raju TS, Bhaumik M. CHO cells provide access to novel N-glycans and developmentally regulated glycosyltransferases. Glycobiology. 1996;6:695–699. doi: 10.1093/glycob/6.7.695. [DOI] [PubMed] [Google Scholar]
- Strigini M, Cohen SM. Wingless gradient formation in the Drosophila wing. Curr Biol. 2000;10:293–300. doi: 10.1016/s0960-9822(00)00378-x. [DOI] [PubMed] [Google Scholar]
- Sumoy L, Kiefer J, Kimelman D. Conservation of intracellular Wnt signaling components in dorsal-ventral axis formation in zebrafish. Dev Genes Evol. 1999;209:48–58. doi: 10.1007/s004270050226. [DOI] [PubMed] [Google Scholar]
- Takada S, Stark KL, Shea MJ, Vassileva G, McMahon JA, McMahon AP. Wnt-3a regulates somite and tailbud formation in the mouse embryo. Genes Dev. 1994;8:174–189. doi: 10.1101/gad.8.2.174. [DOI] [PubMed] [Google Scholar]
- Tamai K, Semenov M, Kato Y, Spokony R, Liu C, Katsuyama Y, Hess F, Saint-Jeannet JP, He X. LDL-receptor-related proteins in Wnt signal transduction. Nature. 2000;407:530–535. doi: 10.1038/35035117. [DOI] [PubMed] [Google Scholar]
- Tang X, Wu Y, Belenkaya TY, Huang Q, Ray L, Qu J, Lin X. Roles of N-glycosylation and lipidation in Wg secretion and signaling. Dev Biol. 2012;364:32–41. doi: 10.1016/j.ydbio.2012.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tendeng C, Houart C. Cloning and embryonic expression of five distinct sfrp genes in the zebrafish Danio rerio. Gene Expr Patterns. 2006;6:761–771. doi: 10.1016/j.modgep.2006.01.006. [DOI] [PubMed] [Google Scholar]
- Tucker JA, Mintzer KA, Mullins MC. The BMP signaling gradient patterns dorsoventral tissues in a temporally progressive manner along the anteroposterior axis. Dev Cell. 2008;14:108–119. doi: 10.1016/j.devcel.2007.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van de Vijver E, Maddalena A, Sanal O, Holland SM, Uzel G, Madkaikar M, de Boer M, van Leeuwen K, Koker MY, Parvaneh N, Fischer A, Law SK, Klein N, Tezcan FI, Unal E, Patiroglu T, Belohradsky BH, Schwartz K, Somech R, Kuijpers TW, Roos D. Hematologically important mutations: leukocyte adhesion deficiency (first update) Blood Cells Mol Dis. 2012;48:53–61. doi: 10.1016/j.bcmd.2011.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang W, Hu T, Frantom PA, Zheng T, Gerwe B, Del Amo DS, Garret S, Seidel RD, 3rd, Wu P. Chemoenzymatic synthesis of GDP-L-fucose and the Lewis X glycan derivatives. Proc Natl Acad Sci U S A. 2009a;106:16096–16101. doi: 10.1073/pnas.0908248106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang W, Hu T, Frantom PA, Zheng T, Gerwe B, Del Amo DS, Garret S, Seidel RD, 3rd, Wu P. Chemoenzymatic synthesis of GDP-L-fucose and the Lewis X glycan derivatives. Proc Natl Acad Sci U S A. 2009b;106:16096–16101. doi: 10.1073/pnas.0908248106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Gu J, Miyoshi E, Honke K, Taniguchi N. Phenotype changes of Fut8 knockout mouse: core fucosylation is crucial for the function of growth factor receptor(s) Methods Enzymol. 2006;417:11–22. doi: 10.1016/S0076-6879(06)17002-0. [DOI] [PubMed] [Google Scholar]
- Wang X, Inoue S, Gu J, Miyoshi E, Noda K, Li W, Mizuno-Horikawa Y, Nakano M, Asahi M, Takahashi M, Uozumi N, Ihara S, Lee SH, Ikeda Y, Yamaguchi Y, Aze Y, Tomiyama Y, Fujii J, Suzuki K, Kondo A, Shapiro SD, Lopez-Otin C, Kuwaki T, Okabe M, Honke K, Taniguchi N. Dysregulation of TGF-beta1 receptor activation leads to abnormal lung development and emphysema-like phenotype in core fucose-deficient mice. Proc Natl Acad Sci U S A. 2005;102:15791–15796. doi: 10.1073/pnas.0507375102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Spellman MW. Purification and characterization of a GDP-fucose:polypeptide fucosyltransferase from Chinese hamster ovary cells. J Biol Chem. 1998;273:8112–8118. doi: 10.1074/jbc.273.14.8112. [DOI] [PubMed] [Google Scholar]
- Wehrli M, Dougan ST, Caldwell K, O’Keefe L, Schwartz S, Vaizel-Ohayon D, Schejter E, Tomlinson A, DiNardo S. arrow encodes an LDL-receptor-related protein essential for Wingless signalling. Nature. 2000;407:527–530. doi: 10.1038/35035110. [DOI] [PubMed] [Google Scholar]
- Westerfield M. The zebrafish book. Eugene: University Oregon Press; 1995. [Google Scholar]
- White RM, Sessa A, Burke C, Bowman T, LeBlanc J, Ceol C, Bourque C, Dovey M, Goessling W, Burns CE, Zon LI. Transparent adult zebrafish as a tool for in vivo transplantation analysis. Cell Stem Cell. 2008;2:183–189. doi: 10.1016/j.stem.2007.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodward R, Yi W, Li L, Zhao G, Eguchi H, Sridhar PR, Guo H, Song JK, Motari E, Cai L, Kelleher P, Liu X, Han W, Zhang W, Ding Y, Li M, Wang PG. In vitro bacterial polysaccharide biosynthesis: defining the functions of Wzy and Wzz. Nat Chem Biol. 2010;6:418–423. doi: 10.1038/nchembio.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu YX, Ma A, Liu L. Transforming growth factor beta signaling upregulates the expression of human GDP-fucose transporter by activating transcription factor Sp1. PLoS One. 2013;8:e74424. doi: 10.1371/journal.pone.0074424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto H, Komekado H, Kikuchi A. Caveolin is necessary for Wnt-3a-dependent internalization of LRP6 and accumulation of beta-catenin. Dev Cell. 2006;11:213–223. doi: 10.1016/j.devcel.2006.07.003. [DOI] [PubMed] [Google Scholar]
- Yamamoto H, Sakane H, Michiue T, Kikuchi A. Wnt3a and Dkk1 regulate distinct internalization pathways of LRP6 to tune the activation of beta-catenin signaling. Dev Cell. 2008a;15:37–48. doi: 10.1016/j.devcel.2008.04.015. [DOI] [PubMed] [Google Scholar]
- Yamamoto H, Sakane H, Yamamoto H, Michiue T, Kikuchi A. Wnt3a and Dkk1 regulate distinct internalization pathways of LRP6 to tune the activation of beta-catenin signaling. Developmental cell. 2008b;15:37–48. doi: 10.1016/j.devcel.2008.04.015. [DOI] [PubMed] [Google Scholar]
- Zheng T, Jiang H, Gros M, del Amo DS, Sundaram S, Lauvau G, Marlow F, Liu Y, Stanley P, Wu P. Tracking N-acetyllactosamine on cell-surface glycans in vivo. Angew Chem Int Ed Engl. 2011;50:4113–4118. doi: 10.1002/anie.201100265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu W, Shiojima I, Ito Y, Li Z, Ikeda H, Yoshida M, Naito AT, Nishi J, Ueno H, Umezawa A, Minamino T, Nagai T, Kikuchi A, Asashima M, Komuro I. IGFBP-4 is an inhibitor of canonical Wnt signalling required for cardiogenesis. Nature. 2008;454:345–349. doi: 10.1038/nature07027. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.