Abstract
The proteoglycan aggrecan is a prominent component of the extracellular matrix in growth plate cartilage. A naturally occurring, recessive, perinatally lethal mutation in the aggrecan core protein gene, cmdbc (Acancmd-Bc), that deletes the entire protein-coding sequence provided a model in which to characterize the phenotypic and morphologic effects of aggrecan deletion on skeletal development. We also generated a novel transgenic mouse, Tg(COL2A1-ACAN), that has the chick ACAN coding sequence driven by the mouse COL2A1 promoter, to enable production of cmdbc/cmdbc; Tg(COL2A1-ACAN) rescue embryos. These were used to assess the impact of aggrecan on growth plate organization, chondrocyte survival and proliferation, and the expression of mRNAs encoding chondrocyte differentiation markers and growth factors. Homozygous mutant (cmdbc/cmdbc) embryos exhibited severe defects in all skeletal elements with deformed and shortened (50%) limb elements. Expression of aggrecan in rescue embryos reversed the skeletal defects to varying degrees with a 20% increase in limb element length and near-full reversal (80%) of size and diameter of the ribcage and vertebrae. Aggrecan-null growth plates were devoid of matrix and lacked chondrocyte organization and differentiation, while those of the rescue embryos exhibited matrix production concomitant with partial zonation of chondrocytes having proliferative and hypertrophic morphologies. Deformation of the trachea, likely the cause of the mutation’s lethality, was reduced in the rescue embryos. Aggrecan-null embryos also had abnormal patterns of COL10A1, SOX9, IHH, PTCH1 and FGFR3 mRNA expression in the growth plate. Expression of chick aggrecan in the rescue embryos notably increased COLX expression, accompanied by the reappearance of a hypertrophic zone and IHH expression. Significantly, in transgenic rescue embryos the cell death and decreased proliferation phenotypes exhibited by the mutants were reversed; both were restored to wild-type levels. These findings suggest that aggrecan has a major role in regulating the expression of key growth factors and signaling molecules during development of cartilaginous tissue and is essential for proper chondrocyte organization, morphology and survival during embryonic limb development.
Keywords: Aggrecan, growth plate, chondrocyte morphology, extracellular matrix, cartilage matrix deficiency, proteoglycan
INTRODUCTION
The development of the skeleton involves a highly regulated, spatially organized series of events that requires the precise temporal control of a multitude of morphogens, growth factors, transcription factors, and mechanical signaling pathways. Chondrodysplasias and other cartilage defects, frequently caused by perturbations of the extracellular matrix (ECM), are a relatively rare group of genetic disorders with no treatment options thus far. An understanding of the mechanisms and signaling pathways involved in these disease processes is essential to finding potential therapeutic targets.
Long bone formation begins with the condensation of mesenchymal cells that undergo chondrogenic differentiation in the early limb bud under the appropriate signals; these primitive chondrocyte precursors create the scaffold upon which the long bones of the skeleton develop and elongate. The most rapid linear endochondral growth occurs prenatally and is dependent upon proper organization and maturation of chondrocytes, a process that occurs in multiple histologically distinct stages in the epiphyseal growth plates of long bones. Resting chondrocytes, found near the distal end of the epiphysis, have a small, round morphology, divide slowly, and serve as a stem-cell-like population for the growth plate. The resting chondrocyte population gives rise to and controls the orientation of the proliferative zone chondrocytes (Abad et al., 2002). Chondrocytes enter the proliferative state, assume a characteristic flattened morphology, begin to rapidly divide, and secrete abundant extracellular matrix components. Elongation of the limb element occurs predominantly during this phase as the chondrocytes form clonal columns parallel to the longitudinal axis of the bone. After several mitoses chondrocytes mature to prehypertrophic and then to large, spherical hypertrophic cells which increase their intracellular calcium levels, commencing the mineralization process of the surrounding extracellular matrix (ECM) (Iannotti J.P., 1989). Ultimately, hypertrophic chondrocytes undergo apoptosis and resorption at the vascular invasion front, where osteoblasts, osteoclasts and chondroclasts are recruited to complete the transformation of the extracellular matrix into mineralized bone tissue (Lewinson D., 1992). This series of events is tightly regulated by the interplay of systemic and local paracrine factors, growth regulators, and mechanical forces that together direct the differentiation and gene expression profile of growth plate chondrocytes (Wagner and Karsenty, 2001). Although most studies focus on limb development, the endochondral growth processes of other skeletal elements (i.e., rib cage) follow similar regulatory paradigms and can be severely affected by genetic mutations.
The ECM plays a critical role not only in the structural support of chondrocytes but also by serving as a medium in which signaling molecules and growth factors are able to diffuse through the avascular cartilage tissue to target cells (Cortes et al., 2009). The ECM is composed predominantly of collagens, proteoglycans, hyaluronan and link protein with smaller amounts of other matrix-specific proteins (Schwartz and Domowicz, 2002). Different proteoglycans have unique expression patterns during skeletal development that are regulated spatially and temporally (Ovadia M., 1980). Versican, a large chondroitin sulfate proteoglycan (CSPG) expressed in mesenchymal condensations of the pre-cartilaginous anlagen, is down-regulated as aggrecan, the predominant CSPG in cartilage, is significantly up-regulated upon chondrogenic differentiation (Kimata et al., 1986; Schwartz et al., 1993). As endochondral ossification of the cartilage elements ensues, the expression of aggrecan by differentiating chondrocytes predominates over other proteoglycans (Ovadia M., 1980).
Aggrecan has a variety of important functions in the growth plate aside from its structural role in the ECM. The numerous sulfated glycosaminoglycans (GAG), attached to the core protein of aggrecan create a large, highly negatively-charged molecule that enables hydration of the cartilage tissue as well as binding of growth factors and morphogens crucial to chondrocyte maturation and function (Cortes et al., 2009; Ruoslahti and Yamaguchi, 1991). In particular, our laboratory has provided evidence that proper sulfation patterns of the GAG chains attached to aggrecan are essential for postnatal morphogen gradient formation in the growth plate, further emphasizing the role of proteoglycans in regulation and modification of normal bone growth (Cortes et al., 2009). Mutations in the aggrecan gene are the cause of several chondrodysplasias and inherited skeletal disorders in humans (Schwartz and Domowicz, 2002; Schwartz and Domowicz, 2014), including spondyloepimetaphyseal dysplasia with premature and severe osteoarthritis and osteochondritis dissecans (Gleghorn et al., 2005; Stattin E.L., 2010; Tompson et al., 2009). Other cartilage disorders, such as pseudoachondroplasia and those involving impaired sulfation of proteoglycans, are caused in part by improper processing and intracellular trafficking of aggrecan, resulting in retarded bone growth (Kwak Y.H., 2009; Vissers L.E., 2011). Despite the increasing association of aggrecan with skeletal disorders, a mechanistic understanding of how aggrecan influences growth plate cytoarchitecture and organization during development remains limited.
We have previously reported on spontaneous autosomal recessive mutations in the chick and mouse aggrecan core protein genes that cause severe chondrodystrophies with perinatal lethality. The chick mutation nanomelia is a single-base substitution which introduces a premature stop codon in the aggrecan gene (Li et al., 1993), resulting in a severely truncated core protein that is not glycosylated or transported through the secretory pathway, as it does not possess the G3 domain required for translocation from the endoplasmic reticulum (Domowicz et al., 2000; Vertel et al., 1994). This mutation renders the nanomelic chick a functional aggrecan-null model, grossly characterized by severe reduction in the size of the head, trunk, and limbs. Growth plates of these embryos contain homogenous populations of small, round chondrocytes, are devoid of extracellular matrix and have significantly altered morphogen gradient distributions and gene expression profiles of critical chondrocyte regulators (Domowicz et al., 2009; Schwartz and Domowicz, 2002; Schwartz and Domowicz, 2014). Similarly, two different recessive mutations in the mouse aggrecan core protein each result in cartilage matrix deficiency (cmd) and produce consistent phenotypes; i.e. shortened trunk, cleft palate, extremely truncated limbs, and protruding abdomen (Bell et al., 1986; Brennan et al., 1983; Rittenhouse et al., 1978). One of these mutations, Acancmd, is a 7-base-pair deletion in exon 5 of the aggrecan gene, which results in a truncated protein product (Watanabe et al., 1994). Our laboratory previously identified and sequenced a separate recessive mutation, cmdbc (or Acancmd-Bc), which causes visible skeletal deformations beginning at embryonic day 15. The mutation is a deletion that begins in intron 1 of the mouse aggrecan gene and removes exons 2 through 18, the entire protein-coding sequence (Krueger et al., 1999). To date, the morphological characterization, gene expression analysis, and cellular consequences of these mutations in the aggrecan-null mouse embryonic growth plate are not well defined; the present study, using mice with the cmdbc mutation, provides that characterization.
Although our previous observations in the nanomelic chick demonstrated that loss of aggrecan is accompanied by significant abnormalities in chondrocyte maturation, proliferation and survival, it is not known which of these phenotypes are primary effects or secondary to perturbation of the ECM environment caused by the aggrecan deficiency. To understand how aggrecan influences cartilage growth plate organization and cytoarchitecture, we generated a novel, tissue-specific transgenic model, cmdbc/cmdbc; Tg(COL2A1-ACAN), referred in this paper as Agc/+, in which chick aggrecan is expressed under the control of the collagen type II, alpha 1(COL2A1) promoter. This transgenic mouse line was successively crossed with +/cmdbc mice to generate embryos lacking endogenous mouse aggrecan production while expressing chick aggrecan specifically in cartilage tissue at the onset of COL2A1 promoter activation during day 10 of development (i.e., cmdbc/cmdbc, Agc/+). Partial rescue of the cmd phenotype was obtained confirming aggrecan to be an essential ECM molecule that influences chondrocyte survival, promotes hypertrophic maturation, and contributes to the proper morphogen, growth factor, and transcription factor expression profile in the developing growth plate.
MATERIALS AND METHODS
Mouse Model and Husbandry
To generate cartilage-specific, aggrecan-expressing transgenic mice, the plasmid pBluescript KS+ (pBS) was first modified by inserting a bovine growth hormone poly A tail between PacI and XhoI restriction sites, and a 200 base pair (bp) splice acceptor was introduced between SpeI and HindIII. The chondrocyte-specific collagen type II, alpha 1 (COL2A1) promoter was digested from the vector p3000i3020Col2a-βgeo (kindly provided by Dr. Veronique Lefebvre at Cleveland Clinic) utilizing NotI and XbaI, and cloned into NotI and SpeI sites of the pBS vector to create the plasmid pBSCol2a1. The 6.3 kb chicken aggrecan cDNA was then cloned into the pBS-Col2a1 plasmid in 2 fragments using the In-fusion PCR system (Clontech)(Cortes et al., 2012). The construct was purified and utilized for pronuclear injection into one-cell mouse embryos to generate 3 heterozygous founder lines on the C57BL/6 background that express chick aggrecan driven by the COL2A1 promoter (Fig. 1).
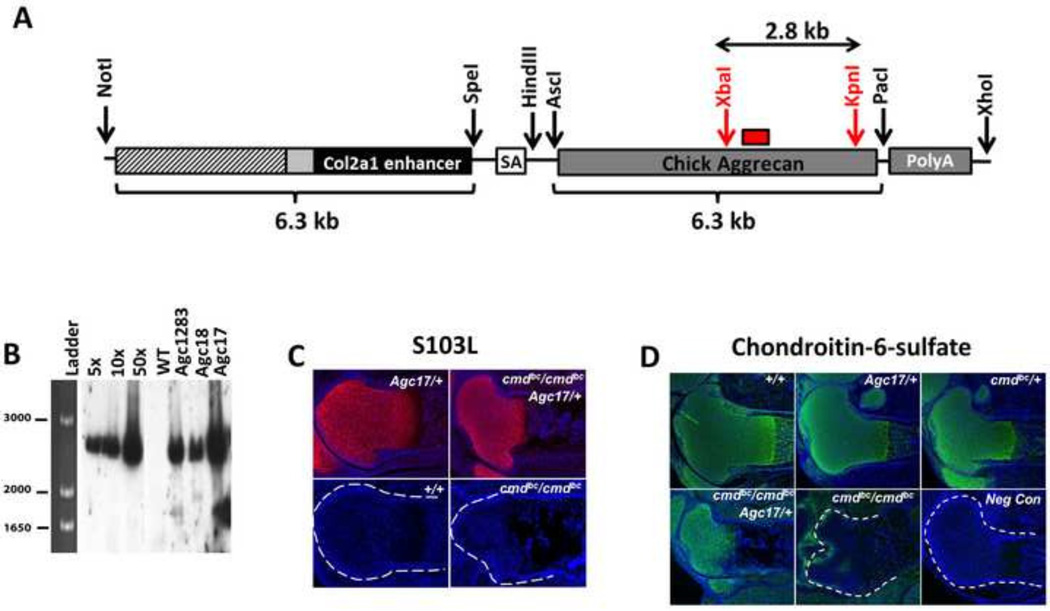
Figure 1. Generation of chick aggrecan transgenic mice.
(A) Schematic representation of the Col2a1-chick aggrecan DNA construct utilized for generation of Agc17/+, Agc18/+, and Agc1283/+ transgenic mice. The construct consists of a 3 kb Col2a1 promoter (hatched box), 237 bp exon 1 (gray box), a 3.02 kb segment of Col2a1 intron 1, and the 6.3 kb chick aggrecan gene. Restriction sites used in cloning are depicted and described in the Materials and Methods section. (B) Representative Southern blot using a 32P-dCTP labeled probe (panel A, red box) of genomic DNA from each of the Agc/+ transgenic lines following XbaI and KpnI digestion (panel A, red). Transgenic line Agc17/+ contained the highest copy number with greater than 50 copies. Representative immunohistochemistry experiments using monoclonal antibodies against chick aggrecan (S103L, C) and chondroitin-6-sulfate (D) are shown for distal femurs of 18.5 dpc embryos of each genotype from the same litter. SA=splice acceptor site
The cartilage matrix deficiency allele utilized in these studies, cmdbc, a mutation causing the deletion of exon 2 through exon 18 in the aggrecan genewhich is perinatally lethal (Krueger et al., 1999), was initially found on a BALB/c background, and was subsequently backcrossed more than 20 generations onto the C57BL/6 background prior to experimentation. To generate embryonic mice which are homozygous for the cmdbc mutation but express chicken aggrecan, heterozygous (+/cmdbc) females) were crossed with heterozygous males that also carried the chicken aggrecan transgene (+/cmdbc: Agc/+). Litters were harvested from pregnant females at 18.5 days postcoitum (dpc), which were sacrificed according to University of Chicago Institutional Animal Care and Use Committee protocol.
To identify chick-aggrecan-transgenic animals and mice hetero- or homozygous for the cmdbc mutation, the following specific primers were used to genotype the embryos from 18.5 dpc litters: Aggrecan transgene: Forward 5’-CTAGAGATCAGTGGACTGCCT-3’; Reverse 5’-TCTGGAGCTGTGCAGTCTAGTGG-3’ cmdbc allele: Forward (mutant) 5’-GGTAAGCCAGGAGCCAGAACTGTGCCCAGA-3’; Reverse (wild type) 5’-GACAATCCCTCTTACACCTCCCCTACTCAA-3’; Reverse (mutant) 5’-GCCCAGCCTCGTCTAAGAGACCCTGGCACT-3’
Southern blot analysis
Chick aggrecan transgenic animals were initially identified by Southern blot analysis according to standard protocol (Sambrook and Russell, 2001) with minor modifications. Briefly, restriction fragments from XbaI- and KpnI- digested tail DNA were separated on an agarose gel, denatured and transferred to a nylon membrane. Transferred DNA was UV cross-linked to the membrane, and a probe homologous to a 392 bp segment of the chick aggrecan coding sequence, shown in Fig. 1A, was generated by PCR using the following primers: forward, 5’-CTAGAGATCAGTGGACTGCCT-3’; and reverse, 5’-GGTCTGGAGCTGTGCAGTCTAGTGG-3’. The PCR product was purified and radioactively labeled with 32P-dCTP (using the Random Primed DNA labeling kit (Roche). The probe was hybridized to the membranes at 68°C for one hour in hybridization solution (QuickHyb, Stratagene) containing denatured salmon sperm DNA, the blots were washed at high stringency, and bound probe was detected on X-ray film. To infer transgene copy number it was assumed that the haploid content of a mammalian genome is 3 × 109 bp and that the mass of the one copy standard was represented by 26.6 pg of the original construct per 10 µg genomic DNA. This mass was used to calculate the copy standards by digesting 10 µg of genomic DNA from wildtype littermates with 5 copies, 10 copies, and 50 copies of the pBSCol2a1-chick aggrecan plasmid construct. Ten µg of tail DNA from the three transgenic founder lines Agc17, Agc18, and Agc1283 was digested and the expected 2.8 kb band’s density was compared with the standards to estimate transgenic copy number. These three colonies have been maintained for over 5 years with comparable levels of transgene expression and consistent Southern blot profiles, which supports the assumption that the transgene has a single integration site as has been reported for transgenic animals derived by similar methods (Brinster et al., 1981; Brinster et al., 1985; Lacy et al., 1983).
Skeletal Preparations
At 18.5 dpc, embryos were fixed in 95% ethanol for 24 hours, then eviscerated and placed in Alcian blue staining solution (20% glacial acetic acid/80% ethanol/0.015% Alcian blue 8GX) for one week. The embryos were further dehydrated in three changes of 95% ethanol over the course of one week, then cleared in 1% potassium hydroxide (KOH). Staining of mineralized tissue involved placing embryos in 0.001% Alizarin Red-0.5% KOH, followed by passing them through a graded series of KOH solutions (0.5% to 0.05%) for final clearing and storing in glycerol/PBS/formaldehyde 10:1:1. Limb elements were dissected free and imaged on a stereoscope in order to quantify. individual limb lengths (proximal epiphysis to distal epiphysis) using the measurement tool in Adobe Photoshop. Statistical significance across genotypes for each individual element was analyzed using a one-way ANOVA with Tukey’s post-hoc testing.
mRNA in situ Hybridization
Antisense RNA probes for the collagen type X, alpha 1 (Col10a1), collagen type II, alpha 1 (Col2a1), SRY (sex determining region Y)-box 9 (Sox9), Indian hedgehog (Ihh), patched homolog 1 (Ptch1), and fibroblast growth factor receptor 3 (Fgfr3) gene transcripts were transcribed from cDNA fragments using digoxygenin (DIG) labeled nucleotide mix and T7 or SP6 polymerase- (Roche) as previously described (Cortes et al., 2009; Domowicz et al., 2008). Mouse hind limbs (18.5 dpc) were dissected free, fixed in 4% paraformaldehyde/PBS at 4°C, sunk in 20% sucrose/10% formalin in PBS, embedded in gelatin, and 40 µm serial sections were cut, mounted onto silane-treated slides and dried overnight. Sections were hybridized with RNA antisense probes at 70°C for 16 hours followed by four post-hybridization stringent washes (50% formamide, 2× SSC, 1% SDS). Hybridization signals were detected by incubating sections with alkaline phosphatase-anti-DIG Fab fragments (Roche) overnight at 4°C. The colorimetric reaction was carried out using NBT-BCIP as substrates.
TUNEL Staining
Hind limb tissue sections from 18.5 dpc embryos were deparaffinized and permeabilized with 0.1% Triton-X. Apoptotic chondrocytes were detected in the sections using the In situ Cell Death Kit (Roche) following the manufacturer’s instructions. Sections were counterstained with DAPI and mounted. Negative control slides were treated with the incubation buffer in the absence of enzyme, as suggested by the manufacturer’s protocol. TUNEL-positive nuclei were counted in the distal femoral growth plates using ImageJ software (Schindelin J.S., 2012), and data were analyzed for statistical significance using a one-way ANOVA with Tukey’s post-hoc testing.
Immunohistochemistry
Limbs were dissected free and fixed in 4% paraformaldehyde overnight at 4°C. Paraffin sections (5 µm) were permeabilized with 0.1% Triton-X followed by digestion with 0.5 U/ml chondroitinase ABC (Seikagaku). Sections were stained with anti-S103L, a monoclonal antibody specific for chick aggrecan (Krueger et al., 1990), anti-phosphohistone H3 (Ser10) polyclonal antibody (Upstate Biotechnology), anti-chondroitin-4-sulfate antibobdy (2B6 clone) and anti-chondroitin-6-sulfate antibody (3B3 clone) (Seikagaku) in 5% serum overnight at 4°C. Sections were washed and incubated with AlexaFluor fluorescent secondary antibodies (Invitrogen), counterstained with DAPI and mounted. Positive phospho-histone H3 nuclei were quantified in the 18.5 dpc embryos’ distal femurs of using ImageJ software, and data were analyzed for statistical significance using a one-way ANOVA with Tukey’s post-hoc testing.
RESULTS
Generation of chicken aggrecan transgenic and aggrecan-null mutant mouse embryos
In order to investigate the contribution of the cartilage-specific proteoglycan aggrecan in growth plate maturation and skeletal development, a chick-aggrecan expressing transgenic mouse was created by cloning the full-length chicken aggrecan cDNA (Cortes et al., 2012) into a vector containing a 3 kb Col2a1 promoter and a 3 kb segment of intron 1, previously shown to be required for efficient cartilage-specific expression (Zhou et al., 1995). The construct (Fig. 1A) was microinjected into fertilized single cell mouse embryos, generating three heterozygous founder lines: Agc17/+, Agc18/+, and Agc1283/+. All Col2a1-chick aggrecan transgenic mice, as well as mice heterozygous for the cmdbc mutation, were phenotypically indistinguishable from wildtype littermates at 18.5 dpc and postnatally, and no gross or quantitative skeletal abnormalities were observed (data not shown). Each genotype was analyzed for potential differences in mouse aggrecan expression by in situ hybridization and quantitative RT-PCR (Supplemental Figs. 1 and 2). In situ hybridization revealed no major differences in the expression pattern of endogenous mouse aggrecan between +/+, cmdbc/+, and Agc/+, embryos at 18.5 dpc, (Supplemental Fig 1); as well no significant difference in the relative expression of mouse aggrecan across genotypes was demonstrated by quantitative RT-PCR (Supplemental Fig 1).
Southern blot analysis demonstrated that founder line 17 retained the highest copy number of the transgene with greater than 50 copies (Fig. 1B), and therefore this line was utilized for all remaining experiments. Successive crossing of Agc17/+ mice with cmdbc/+ generated mice concurrently heterozygous for the cmdbc mutation and Agc17 transgene. These mice were crossed with cmdbc/+ mice to generate litters including “mutant” embryos (cmdbc/cmdbc), which completely lack endogenous aggrecan due to a mutation that results in the deletion of exons 2 through 18 (Krueger et al., 1999), and “rescue” embryos (cmdbc/cmdbc;Agc/+) which lack endogenous mouse aggrecan but express the chick aggrecan transgene. Cartilage-specific protein expression of the chick aggrecan transgene was verified in the limbs of founder line 17 (Fig. 1C, left panel) and in the rescue animals derived from line 17 (Fig. 1C, right panel) using the S103L monoclonal antibody, which recognizes a specific epitope on the core protein of chick aggrecan exclusively (Krueger et al., 1990). Limbs from 18.5 dpc rescue embryos show robust extracellular S103L staining comparable to that for the transgenic founder line, indicating that efficient expression and secretion of chick aggrecan protein was achieved in embryos lacking mouse aggrecan. We also performed qRT-PCR for relative quantitation of chick aggrecan and found no differences in chick aggrecan expression levels between Agc17/+ and rescue embryos (Supplemental Fig. 2). However, when analyzing the gene expression ratio of chick vs. mouse aggrecan in transgenic line 17 cartilage, we found that there is 5-fold less chick aggrecan mRNA expressed compared to endogenous mouse aggrecan mRNA at 18.5 dpc, indicating that the aggrecan transgene does not achieve the same levels of expression as endogenous mouse aggrecan.
Although chick aggrecan core protein was synthesized and secreted, it was necessary to demonstrate that the protein undergoes extensive post-translational addition of chondroitin sulfate side chains. Therefore, we examined overall levels of chondroitin-6-sulfate and chondroitin-4-sulfate in the growth plates of founder line 17 (Fig. 1D, upper left panel and Supplemental Fig 3, respectively), in rescue embryos, mutant embryos, and representative embryos of different genotypes from the same litter. Compared to all other genotypes, mutant growth plates exhibit very low chondroitin-6- and chondroitin-4-sulfate staining in growth plate cartilage, as expected due to the lack of aggrecan in the matrix. Patterns of chondroitin-6-sulfate detected in the rescue embryos lacking endogenous aggrecan were comparable to that of the wildtype lines and the staining intensity of chondroitin 4-sulfate increased in rescue growth plates compared to that of mutants, indicating that the chick aggrecan protein expressed by transgenic rescue embryos contains chondroitin sulfated moieties.
Chick aggrecan transgene expression in cmdbc/cmdbc embryos increases limb length and partially restores skeletal element Alizarin red staining patterns
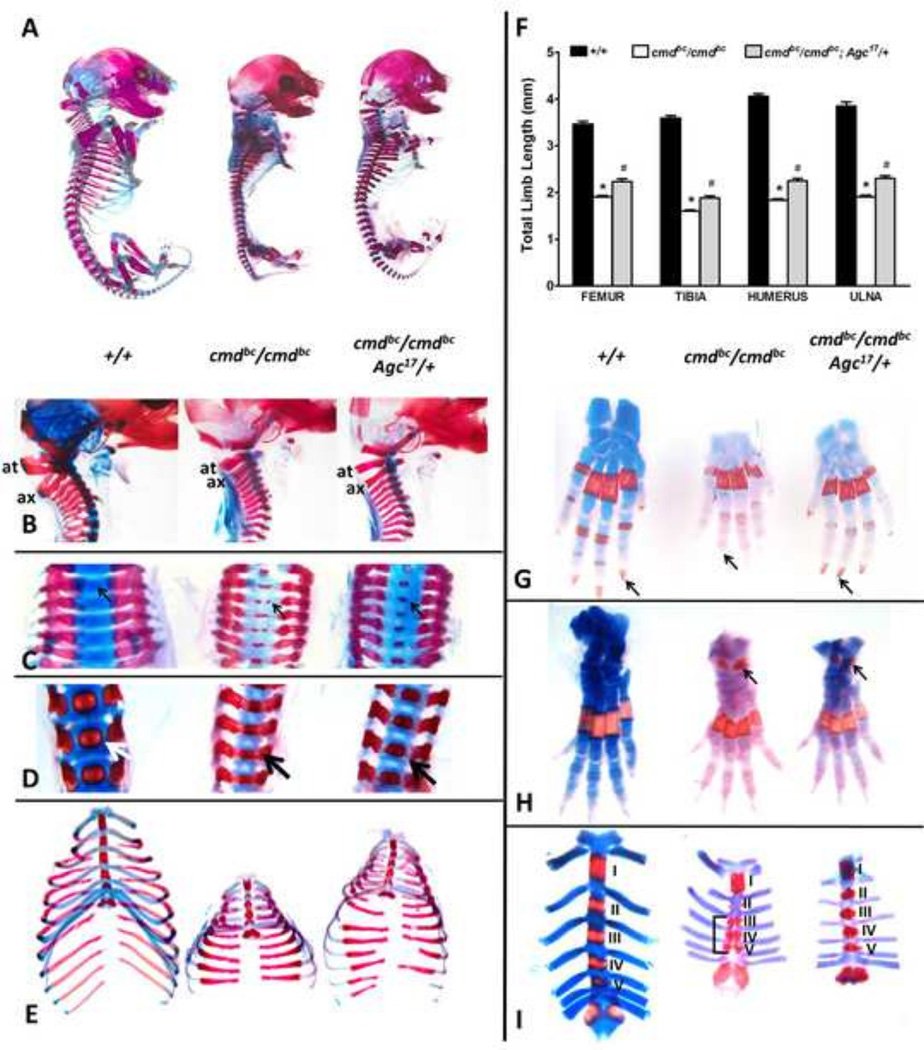
Dysregulation of several genes that affect the formation of appendicular and axial skeletal elements has been described; few of these have been verified by transgenerescue approaches. Analysis of skeletal preparations from 18.5 dpc embryos was carried out using Alcian blue and Alizarin red to stain cartilage and calcified tissue, respectively. Examination of individual axial and appendicular skeletal elements revealed improved skeletal element formation compared to mutants. The cmdbc/cmdbc mutant embryos had a 45–55% decrease in the length of the long-bone elements of the forelimb and hindlimb (Fig. 2A, F) compared to wild-type (+/+) embryos, while in the cmdbc/cmdbc;Agc17/+ rescue embryos, the overall length of the femur and tibia increased 18% and the length of the humerus and ulna increased by 21% compared to mutant embryos (Fig. 2F).
Figure 2. Skeletal element analysis in aggrecan null cmdBc/cmdBc embryos and transgenic cmdBc/cmdBc;Ag17/+ rescue mice.
(A) Comparison between whole skeletons of wildtype (+/+), mutant (cmdBc/cmdBc), and rescue (cmdBc/cmdBc;Agc17/+) embryos harvested 18.5 dpc and stained with Alizarin red (presumed ossified areas) and Alcian blue (cartilage). Individual skeletal element dissections were analyzed for staining patterns and overall size for the cervical spine (B–C), lumbar spine (D), ribcage (E), forelimb digits (G), hindlimb digits (H) and sternum (I). Arrows demonstrate Alizarin red staining differences in the spine and digits, and sternum segments are labeled to demonstrate abnormal staining patterns of the third through fifth sternebrae in mutants (I). Quantification of long bone deficiencies found a 50% decrease in length of mutant limbs, with a 20% improvement in rescue mice (F). n=6–18 embryos/genotype, *p<0.001 vs. +/+, #p<0.05 vs. cmdBc/cmdBc, one-way ANOVA with Tukey’s post-hoc testing.
Abnormal cervical spine alignment and positioning of the atlas and axis (Fig. 2A, B) and was observed in mutant embryos; these defects were largely corrected in rescue embryos. In E18.5 dpc wild-type embryos, the centra of the cervical vertebrae are cartilaginous (Fig. 2C), while mutant embryos possess dual Alizarin red-positive sites in the same regions (Fig. 2C, arrows). The cartilaginous phenotype is largely restored in the rescue mice, which exhibit only a single premature ossification center within the centra of the cervical vertebrae. Normal staining patterns are also restored in the centra of the lumbar spine by transgene expression compared to mutant embryos, which display aberrant Alizarin red staining patterns within the centrum and neural arch (Fig. 2D, arrow) and decreased vertebral body size.
The digits of the forelimbs of rescue embryos are lengthened compared to mutants and display enhanced Alcian blue staining intensity, indicative of the increased proteoglycan content of the ECM (Fig. 2G). Ossification centers in the distal phalanx of the forelimb that are lost in aggrecan-null mutants are restored in rescue embryos (Fig. 2G, arrows). Transgene expression did not rescue the premature bone deposition observed in the tarsals of the hindlimb (Fig. 2H, arrows), however increased Alcian blue stain intensity is again observed.
Comparison of wild-type and mutant embryo skeletons demonstrates the severely shortened (by 50%), decreased in diameter, and bell-shaped rib cage in mutant embryos (Fig. 2E). Transgene expression in the rescue embryos significantly increased the intercostal spaces, overall size (80%), and diameter of the rib cage compared to mutant embryos. Compared to wild-type, mutant embryonic sterna have highly irregular patterns of staining. Increased Alizarin red staining between the third, fourth and fifth sternebrae, and absence of Alcian blue-positive cartilage, suggested possible premature bone formation (Fig. 2I). In order to verify whether these staining patterns were indicative of precocious ossification of the sternebrae, hematoxylin and eosin (H&E) staining and von Kossa staining was performed on serial sections of sterna from wild-type, mutant, and rescue embryos (Supplemental Fig. 4). Interestingly, the Alizarin red-positive areas in the mutant are not predominantly calcified as seen in the wild-type and rescue sterna, but rather consist of irregular-appearing cartilage. Aberrant chondrocyte morphology, organization, and matrix deposition are also seen in mutant sterna. These anomalies are improved in the sternum of rescue embryos, which exhibit the expected patterns of sternebrae ossification found in wild-type embryos, despite the sustained reduction in total sternum length.
Chondrocyte cytoarchitecture and growth plate morphology is improved in rescue embryos
To investigate the impact of reintroducing aggrecan into the extracellular matrix on chondrocyte cytoarchitecture and growth plate organization, histological sections of 18.5 dpc femurs from wild-type, mutant and rescue embryos were H&E-stained to detect the organization of distinct chondrocyte populations within each growth plate (Fig. 3A–C). In wild-type embryos, immature resting (R) chondrocytes are located closest to the articular surface of the bone (Fig. 3A), and display the typical small, round morphology and homogenous distribution within the extracellular matrix. As these chondrocytes enter the cell cycle and begin to proliferate (P), they assume a discoid shape and align into the characteristic columnar organization (Fig. 3A’, red outline) with abundant extracellular matrix production that is required for growth plate elongation (arrows). Finally, the chondrocytes reach the post-mitotic hypertrophic stage of maturation (H), and are identified by their enlarged volume, spherical shape, increased cytoplasmic to nuclear ratio, and decreased interterritorial matrix (Fig. 3A’, outlined by black box).
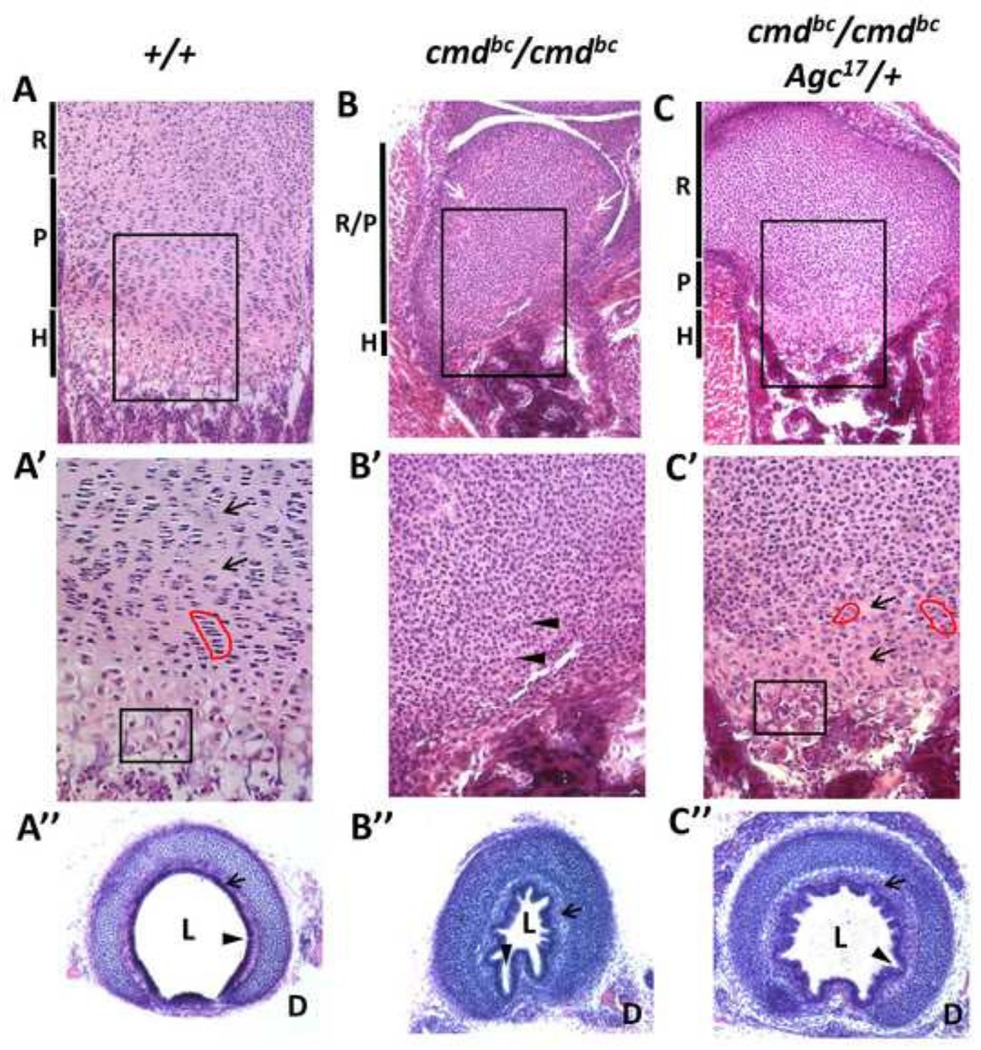
Figure 3. Histological examination of mutant and rescue growth plate cytoarchitecture.
Representative sections of 18.5 dpc femoral growth plates stained with hematoxylin and eosin demonstrating chondrocyte morphology within each growth plate zone (A–C) for wild-type, mutant, and rescue embryos. (A’–C’) Higher magnification of the proliferative and hypertrophic regions outlined in black boxes in top row. Red outlines highlight the columnar formation of proliferative chondrocytes, and the black box denotes chondrocytes with hypertrophic morphology, both of which are absent in aggrecan-null growth plates. Note the hypercellularity and lack of extracellular matrix in mutant embryos (arrow heads) compared to wild type and rescue (arrows). (A’’–C’’) Sections of cartilaginous rings from the upper third of the trachea, showing differences in airway patency with and without aggrecan. R= resting, P= proliferative, H= hypertrophic, L= lumen, D= dorsal aspect.
While the chondrocyte zones within growth plates of wild-type embryos are clearly defined, the growth plates of mutant embryos (Fig. 3B) appear homogenous and devoid of zone organization and cytoarchitecture. The chondrocytes are small, round cells dispersed randomly throughout the markedly truncated epiphyseal growth plates, and are indistinguishable from one zone to the next. Mutants display small areas of hypocellularity in arbitrary regions of the resting zone (Fig. 3B, white arrows), while the rest of the growth plate exhibits a severe deficiency in matrix production, evident by the markedly increased cell density (Fig 3B’, arrow heads). In addition, mutant cartilage exhibits very few cells that can be distinguished as “hypertrophic” chondrocytes at the chondro-osseous junction.
Rescue embryonic growth plates have improved chondrocyte zonation and partial restoration of the associated cytoarchitecture of the proliferative and hypertrophic zones. As shown in Fig. 3C, there is an increase in the overall size of the epiphyseal growth plate of the femur compared to the mutant samples, and a decrease in cell density. Though smaller and less defined than in wild-type embryos, a proliferative zone is evident in rescue growth plate cartilage, as noted by formation of small columns of cells and flattened chondrocyte morphology (Fig. 3C’, red outline) in addition to increased matrix secretion between the columns (arrows). Importantly, expression of the chick transgene in aggrecan-null embryos resulted in the development of a distinct hypertrophic zone, in which the chondrocytes have assumed the characteristic enlarged, spherical cell shape (Fig. 3C’, outlined by black box).
The initial studies on the cmd/cmd embryos postulated that lethality in the mutants was due to inability to respire resulting from tracheal collapse (Bell et al., 1986; Rittenhouse et al., 1978). The cartilaginous rings of the trachea provide rigid structural support to ensure airway patency, and chondrocytes in this type of cartilage do not progress to hypertrophy and apoptosis, as seen in growth plates (Fig. 3A–C). We examined the tracheas of wild-type, mutant and rescue embryos (Fig. 3A’’–C’’) at 18.5 dpc. The tracheal cartilage, as shown in a transverse section from the upper third of the trachea, consists of a C-shaped ring of hyaline cartilage connected at the dorsal aspect (D) by the trachealis muscle. The lumen (L) of a wild-type trachea appears round and the epithelial lining is smooth (Fig. 3A’’, arrow head), demonstrating airway patency. In contrast, the airway of a mutant embryo has extensive epithelial folding (Fig. 3B’’, arrow head), resulting in a star-shaped, narrowed lumen, features that are consistent with severe tracheomalacia and stenosis which often cause collapse of the trachea during expiration. Rescue embryos show significantly improved diameter of both the tracheal lumen and cartilaginous ring structure, and a dramatic decrease in the degree of epithelial folding, though some evidence of tracheomalacia remains (Fig. 3C’’, arrow head). In both mutant and rescue embryos, these findings are associated with hypercellularity within the hyaline cartilage and thickening of the submucosal layer (arrows).
Altered gene expression patterns in aggrecan-null growth plates are partially restored with chick aggrecan transgene expression
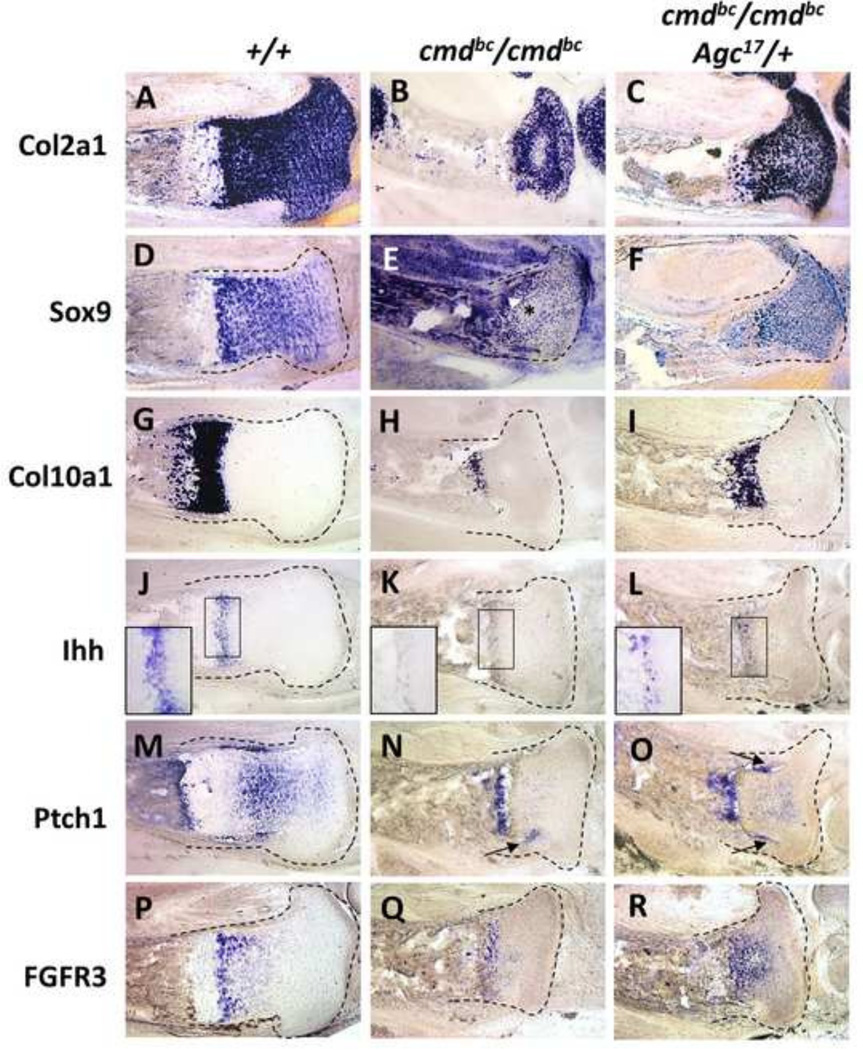
In order to assess whether the histological changes observed in mutant and rescue growth plates are associated with temporal and/or spatial differences in gene expression patterns of key embryonic growth plate regulatory components, mRNA in situ hybridization was performed on serial sections of 18.5 dpc proximal tibias, probing for several chondrocyte growth factors and markers. As shown in Figure 4G, wild-type embryos exhibit robust Col10a1 expression restricted to hypertrophic chondrocytes (left panel). However, the region of Col10a1 expression in mutants is drastically decreased near the chondro-osseous junction (Fig. 4H), implying that the loss of aggrecan has severely impacted the normal hypertrophic differentiation of chondrocytes at this developmental stage. Expression of the chick aggrecan transgene in rescue mice (Fig. 4I) results in a notable increase in the area and intensity of Col10a1 expression in the hypertrophic zone compared to mutants, congruent with the reappearance of enlarged, hypertrophic chondrocytes in this region (Fig. 3C’, box). The expression pattern of Col2a1, the common cartilage marker expressed by all committed chondrocytes, is not significantly altered among wild-type, mutant, and rescue cartilage tissue despite the variation in growth plate size and chondrocyte morphology (Fig. 4A–C), as was previously reported (Kimata et al., 1981). For this reason, the Col2a1 promoter construct was used to drive expression of the chick aggrecan transgene.
Figure 4. Expression pattern of growth plate transcripts by mRNA in situ hybridization.
Representative expression analyses of Col2a1 (A–C), Sox9 (D–F), Col10a1 (G–I), Ihh (J–L), Ptch1 (M–O), and Fgfr3 (P–R) transcripts in 18.5 dpc tibia sections from wild-type, mutant, and rescue embryos. The panels demonstrating Ihh data contain insets that enlarge the prehypertrophic/hypertrophic zone of expression. Results were observed consistently in five independent experiments with at least four embryos for each mRNA probed. In situ hybridizations with DIG-labeled RNA probes were performed on 40 µm sections; cartilage elements are outlined for clarity.
SOX9, a critical transcription factor controlling chondrocyte differentiation, is expressed concomitantly with COL2A1 during cartilage development (Bi et al., 1999; Zhao et al., 1997). Similar to the wild-type Col2a1 gene expression pattern, Sox9 transcripts in the wild-type growth plate appear throughout the resting zone, and peak expression is observed in the late-proliferative/prehypertrophic chondrocytes, followed by an abrupt decline in expression in the hypertrophic zone (Fig. 4D). However, mutant growth plates exhibit diffuse, patchy expression of Sox9 with indiscriminate regions of intense expression (Fig. 4E, arrow head) adjacent to areas of very low levels of Sox9 (asterisk), unlike the pattern of Col2a1 expression. Transgene expression in rescue mice restores the Sox9 expression pattern in the resting and proliferative zone similar to that seen in the wild type (Fig. 4F), although the presumptive zone of hypertrophy in rescue embryos does not demonstrate the typical decline in Sox9 intensity seen in wildtype cartilage.
Gene expression patterns of Ihh, its receptor Ptch1, and Fgfr3, all of which encode central factors controlling the rate of chondrocyte differentiation to hypertrophy, are shown in Fig. 4 J–R. In wild-type growth plates expression of Ihh is restricted to the prehypertrophic and early hypertrophic chondrocytes, as expected (Fig. 4J). Mutants express extremely low levels of Ihh (Fig. 4K) in a single fragmented row of weakly positive chondrocytes (inset), while expression of the aggrecan transgene results in an increase in IHH mRNA production, in a wider band with more chondrocytes, in the prehypertrophic/hypertrophic region (Fig. 4L, inset), indicating that the presence of aggrecan in the growth plate is imperative for normal Ihh expression. Ptch1 encodes the receptor for IHH, is also a downstream target of IHH pathway activation (Ingham, 1998), and Ptch1 transcipts are normally found in the proliferative zone and in the perichondrium flanking this region, as shown in wild-type sections (Fig. 4M, arrows). Although mutant growth plates display very little Ihh expression, some Ptch1 transcripts are present in growth plate cartilage near the perichondrium (Fig. 4N, arrow) and faint expression is detected within the mid-growth plate. Unlike the expression observed in growth plate cartilage, there appears to be relatively normal expression of Ptch1 within the bone marrow near the chondro-osseous junction. Rescue growth plates display specific localization of Ptch1 transcripts in both the perichondrium and the proliferative zone, similar to wild-type embryos (Fig. 4O, arrows), as well as within the vascular invasion/ bone marrow regions.
The expression of Fgfr3, which encodes the receptor for the negative regulators of chondrocyte proliferation in the growth plate such as FGF9 (Garofalo S., 1999) and FGF18 (Davidson et al., 2005), is expressed by the proliferative zone chondrocytes in the wild-type growth plate (Fig 4P). In mutant growth plates, cells centrally located near the presumptive hypertrophic region express Fgfr3, however the intensity of expression is notably decreased compared to wild type (Fig. 4Q). In contrast, rescue growth plates exhibit strong Fgfr3 signals in the proliferative and early hypertrophic zones, an expression pattern that more closely resembles that found in wild-type embryos (Fig. 4R). Taken together, the data obtained from mRNA in situ hybridization analysis suggest that the presence of aggrecan in the growth plate extracellular matrix is fundamental to maintaining normal expression and spatial localization of essential regulators of chondrocyte differentiation.
The apoptotic and proliferative phenotypes in the mutant growth plate are ameliorated by aggrecan transgene expression
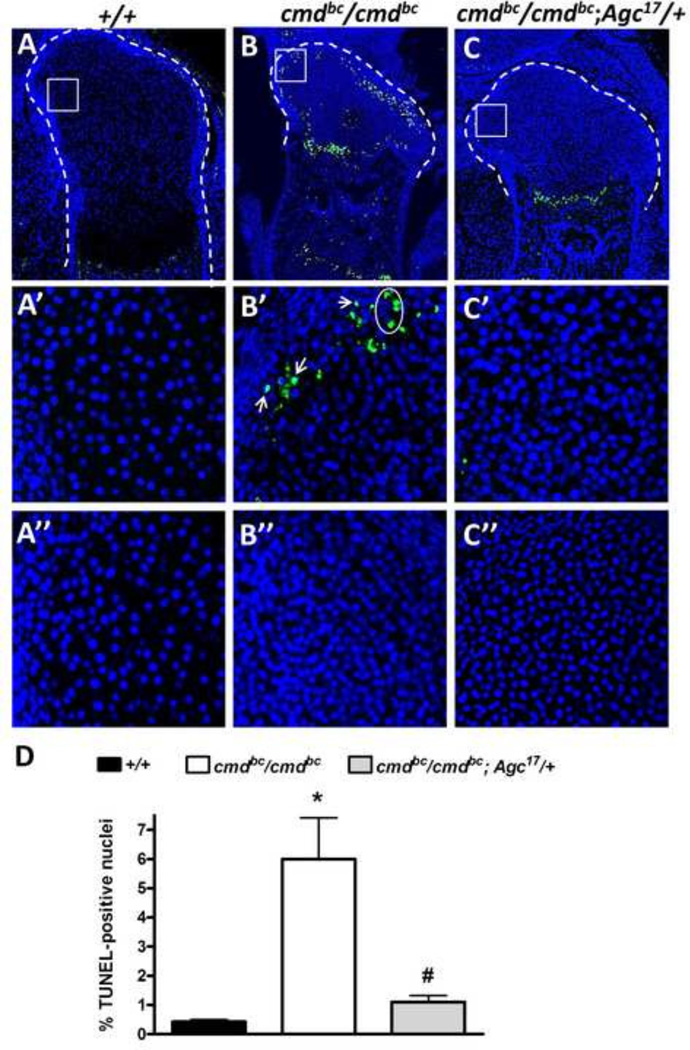
To further examine the cellular consequences associated with aggrecan deletion in growth plate cartilage, we performed terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling (TUNEL) staining and phospho-histone H3 (Ser10) staining, which specifically labels cells entering the M phase of mitosis, to examine apoptosis and proliferation, respectively. Wild-type embryos exhibited very low overall levels of apoptosis at 18.5 dpc (Fig. 5A, D), while growth plates of mutant embryos displayed a significant 14-fold increase in cell death, with most of the apoptotic cells appearing in the presumptive resting zone adjacent to the perichondrium (Fig. 5B, D). The total percentage of apoptotic chondrocytes in transgenic rescue mice was the normal basal level found in wild-type mice (Fig. 5D); few apoptotic chondrocytes were detected in the resting zone of rescue mice, and TUNEL positivity was localized to the chondro-osseous junction (Fig. 5C). Upon closer examination of the resting zone mutant cartilage (Fig. 5B’), many of the apoptotic cells are nucleated, as indicated by the colocalization with DAPI (white arrows). There are also clusters of “ghost” cells that retain no visible nuclear structure yet exhibit strong TUNEL positivity, signifying the presence of released nuclear contents within the chondrocyte lacunae (Fig. 5B’, circle). The extranuclear staining observed was not due to nonspecific staining or autofluorescence, as indicated by the negative control (Fig. 5B’’).
Figure 5. Patterns of cell death in the growth plate of mutant and rescue embryos.
Paraffin sections of 18.5 dpc embryonic femurs were analyzed for apoptotic cells using TUNEL staining. Cartilaginous element limits are outlined in white. (A–C) Increased numbers of apoptotic cells are present in mutants, an occurrence not found in rescue embryos. (A’–C’) Higher magnification of the white boxes outlined in A–C, demonstrating the aberrant cell death occurring in the resting zone of mutants but not in wild-type or rescue embryos. (A’’–C’’) Negative control images of resting zone areas outlined. (D) Quantification of total TUNEL-positive cells in the growth plate. n=3–6 embryos/genotype, *p<0.001 vs. +/+, #p<0.001 vs. cmdBc/cmdBc, one-way ANOVA with Tukey’s post-hoc testing.
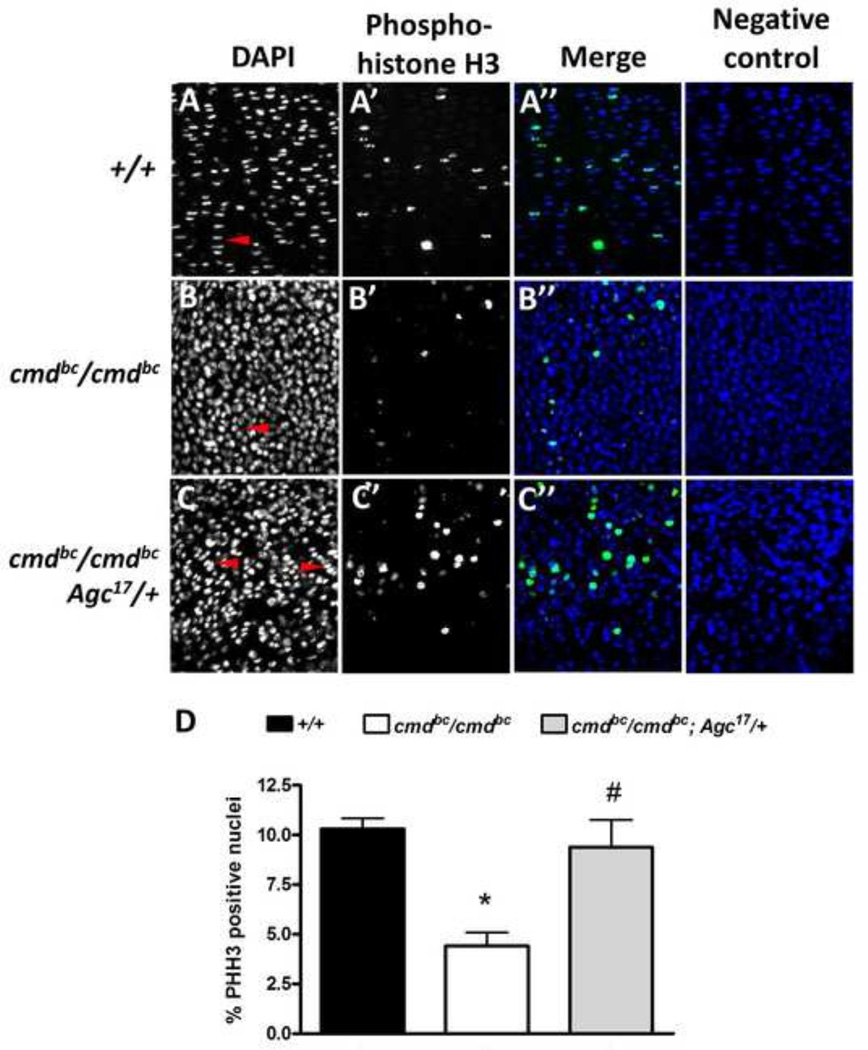
Concomitant with increased cell-death levels, mutant growth plates (Fig. 6B’’) display a significant 57% decrease in proliferation compared to wild-type cartilage (Fig. 6A’’), as seen by phospho-histone H3 (PHH3) staining, which specifically identifies cells undergoing mitosis. Expression of the chick aggrecan transgene in rescue mice significantly increased the percentage of proliferating chondrocytes by more than 2-fold (Fig. 6C’’), which restored proliferation to levels observed in wild-type embryos (Fig. 6D). Additionally, the DAPI nuclear stain in the proliferative zone of the rescue embryos again demonstrates the change in morphology of the chondrocytes from the round, immature appearance seen in mutants (Fig. 6B, arrow head) to a flattened morphology, including columnar formation (Fig. 6C, arrow heads), which more resembles wild-type embryonic growth plate cartilage (Fig. 6A, arrow head). These results implicate a role for aggrecan in maintaining chondrocyte survival and proliferation in the developing growth plate.
Figure 6. Proliferation in mutant and rescue embryonic growth plates.
Embryonic femurs 18.5 dpc stained with DAPI and anti-phospho-histone H3 (PHH3) for analysis of mitotic chondrocytes within the proliferative zone. (A–C) DAPI-stained proliferative chondrocytes demonstrate partial columnar formation in rescue embryos (red arrow heads) compared to mutants. (A’’–C’’) Merged images show that rescue proliferative chondrocytes include double the percentage of pHH3-staining cells in the growth plate compared to mutants, which is comparable to wild-type levels. (D) Quantification of pHH3-positive proliferative chondrocytes in the growth plate. n=3–6 embryos/genotype, *p<0.001 vs. +/+, #p<0.01 vs. cmdBc/cmdBc, one-way ANOVA with Tukey’s post-hoc testing.
DISCUSSION
Specific ECM components are critical to skeletal development, as they define the milieu for chondrocyte differentiation and maturation, provide a scaffold to support the chondrocytes, and constitute a medium in which signaling molecule gradients can differentially affect fields of cells. Evidence that proper ECM composition is essential to skeletal development emanates from the impact of ECM-component-altering genetic defects, mutations which affect cartilage formation and endochondral ossification. Since the pathogenesis is often complex and only partially explained by examining the mutant phenotypes, we have taken the approach of attempting correction of certain phenotypes which result from absence of aggrecan, the predominant proteoglycan in cartilage ECM.
Our model uses the aggrecan cmdbc allele in the C57BL/6background to explore the molecular and morphological consequences of aggrecan deletion on embryonic skeletal development in a mammalian system, and a novel transgenic mouse line that overexpresses the full-length chick aggrecan gene in chondrocytes under the control of the mouse Col2a1 promoter (Zhou et al., 1995) that mimics the temporal and spatial patterns of aggrecan expression and partially rescues the cmdbc/cmdbc phenotype. Since Col2a1 transcripts co-localize with aggrecan transcripts during the same stage of limb bud development (Mallein-Gerin F., 1988), and the expression pattern of the cartilage-specific Col2a1 mRNA transcripts is highly similar to that of aggrecan (Kosher R.A., 1986), our choice of the Col2a1 promoter to drive transgene expression is appropriate.
To our knowledge, this Col2a1-chick aggrecan transgenic mouse possesses the largest transgene insert successfully cloned (12.6 kb) and functionally expressed. Use of the rescue embryos, which are hemizygous for the chick aggrecan transgene while lacking mouse aggrecan, complements and expands upon our previous studies in the chick investigating the critical roles of chondroitin sulfate proteoglycans during development (Domowicz et al., 2009). This approach also allows direct immunological detection of the transgene product in mouse embryos. While some differences exist between mouse and chick aggrecan (Li et al., 1993), it is unknown to what extent these differences may contribute to rescuing the cmdbc/cmdbc phenotype. As well, the rate of aggrecan synthesis and turnover in chondrocytes differs between cartilage zones (Shapses S.A., 1994), and this pattern may not precisely be duplicated in Col2a1-driven aggrecan expression. Nonetheless, the presence of abundant S103L-positive chick aggrecan protein in the ECM of transgenic mice and rescue embryonic growth plates, as well as abundant levels of chondroitin sulfate throughout the matrix provide evidence that chick aggrecan protein is produced by transgenic mouse embryos, is posttranslationally modified and is secreted to the ECM.
In analyzing the morphological and functional phenotypes, it became clear that only certain of these were restored to wild-type levels while others were only partially corrected, a result that has proved to be as informative as a complete rescue would have been. The observed outcome reflects the existence of many likely sources of phenotypic variance, including: cross-species differences in aggrecan core protein primary structure with attendant possible variations in GAG content; the cDNA source of the rescue aggrecan altering/eliminating potential alternative splicing of exons; the nonnative regulation of the transgene’s expression affecting the rate and level of aggrecan production; although designed to produce chick aggrecan in the appropriate cells at the desired time, aggrecan expression may differ in subtle yet important ways from the native pattern; and possible differences in interactions with other ECM components, which could arise from one or more of the aforementioned factors.
Aggrecan modulates chondrocyte differentiation
Rescue embryos demonstrate that considerable changes occurred in both skeletal elements that develop through the process of endochondral ossification as well as permanent tracheal cartilage. Distinct changes in chondrocyte morphology and gene expression in the growth plate hypertrophic zone were associated with the absence of aggrecan, most strikingly the diminished zone of Col10a1-positive cells and lack of enlarged chondrocytes in cmdbc/cmdbc embryos. Chick aggrecan transgene expression in such mutants resulted in an expanded zone of Col10a1 expression and enlarged chondrocyte morphology typical of the hypertrophic zone, indicating that aggrecan plays a major role in modulating chondrocyte hypertrophic maturation in the developing growth plate, although not fully establishing the morphology characteristic of a wild-type hypertrophic zone. A recent hypothesis that the degree of hypertrophic chondrocyte enlargement within a growth plate is relative to the rate and extent of the linear growth of the element (Cooper K.L., 2013) also could explain why more substantial element elongation was not observed in the rescue embryos.
An aggrecan-containing milieu is essential for chondrocyte survival and proliferation
Growth plate elongation is dependent upon the proliferation and maintenance of columnar chondrocytes and their intercalation into columns, events which are absent in aggrecan-null cartilage. These processes entail the production and secretion by prehypertrophic growth plate chondrocytes of IHH, which ultimately acts with other factors to slow the rate of hypertrophic maturation (Vortkamp et al., 1996), regulate the length of column growth (Kobayashi et al., 2005), and control the rate of proliferation (Minina et al., 2001). IHH is expressed at very low levels by a single, fragmented layer of growth plate chondrocytes in mutant tibial cartilage, and this low level of IHH expression appears to be insufficient to maintain normal proliferation rates in mutants. In contrast, even minimal levels of aggrecan enhance the number of IHH-expressing cells and double the rate of cell divison, effectively restoring proliferation rates to wildtype levels. These results indicate that aggrecan transgene expression influenced the survival or differentiation of IHH-producing cells concomitant with restoration of proliferation levels. However, the IHH-positive zone of chondrocytes remained superimposed on the hypertrophic Col10a1-positive region, as reported previously in the nanomelic chick (Domowicz et al., 2009), suggesting that rapid conversion of IHHproducing chondrocytes to COL10A1-positive hypertrophic chondrocytes is still occurring in rescue embryos and may be contributing to the incomplete rescue phenotype. Although chick-aggrecan transgene expression in rescue embryos did not fully restore normal Ihh expression, these data support a role for aggrecan in promoting chondrocyte survival and proliferation in the growth plate.
Lack of aggrecan leads to signaling defects
The absence of aggrecan in the growth plate may also influence the signaling range of IHH in the growth plate. The transmembrane receptor Ptch1 is a downstream target of IHH-mediated transcriptional activation that negatively regulates the propagation of IHH signaling (Chen Y., 1996), and is expressed in IHH target cells located predominantly in the proliferative zone. Mutant growth plates displayed very low levels of IHH mRNA, however the presence of Ptch1 transcripts suggests that IHH stimulation may have occurred at an earlier time point or that the low amount of IHH produced reaches alternative targets in the developing element. This is supported by an observed shift in the localization of Ptch1 expression to the trabecular bone, where robust staining is present while only weak staining is visualized in the growth plate cartilage, indicating that the range of IHH signaling in growth plate cartilage is perturbed in the absence of aggrecan. In contrast, rescue growth plates demonstrate PTCH1 signaling in the perichondrium and presumptive proliferative zone in addition to the bone marrow, suggesting improved IHH signaling range in cartilage accompanies aggrecan transgene expression. Collectively, these data build upon our previous findings that normal IHH gradient formation requires properly sulfated aggrecan (Cortes et al., 2009). The decrease in FGFR3 mRNA observed in cmdbc/cmdbc growth plates also suggests a loss of control of hypertrophic differentiation. Increased expression of FGFR3, which negatively regulates the rate of hypertrophic maturation (Garofalo S., 1999), within the proliferative zone of rescue embryos suggests that some control over the maturation rate of growth plate chondrocytes occurred, partially accounting for the appearance of IHH-expressing cells and re-establishment of a distinct hypertrophic zone.
SOX9 is an essential chondrocyte transcription factor directing differentiation and columnar organization in the proliferative zone (Dy P., 2012). Mutant growth plate chondrocytes mis-express SOX 9 throughout the growth plate, perturbing multiple stages of chondrocyte growth and leading to loss of columnar organization, early terminal maturation, and significantly increased chondrocyte apoptosis in resting zone cells. Interestingly, previous studies have shown that mutants with Sox9 conditionally inactivated in resting chondrocytes during limb development display increased apoptosis (Ikegami D., 2011), similar to what we observe in aggrecan-null mutant embryos. Since SOX9 is one of the transcription factors controlling aggrecan expression in chondrocytes (Sekiya et al., 2000), it is possible that the apoptotic phenotype in conditionally inactivated Sox9 mutants may be due in part to reduced aggrecan synthesis. The mis-expression and downregulation of Sox9 in the mutant is notably reduced in rescue embryos, which display very low levels of chondrocyte apoptosis. These observations may indicate a feedback mechanism in which proper matrix composition is required to maintain normal expression of SOX9. The Col2a1 enhancer element within the transgene constructs in rescue embryos contains SOX9 binding sites that drive efficient transcription (Lefebvre et al., 1997); however, SOX9 expression in the rescue embryos is not completely restored to wild-type levels, contributing to the incomplete rescue phenotype. Further studies that highlight the temporal expression patterns of IHH, PTCH1, SOX9, and FGFR3 expression at earlier time points during limb development are needed to comprehensively assess whether the gene expression changes observed are primary or secondary to aggrecan deletion, which will allow a more thorough understanding of the role of ECM proteins on the production and distribution of these important chondrocyte factors.
In summary, we present evidence that the combined effects of decreased proliferation, increased cell death, and altered gene expression of growth plate regulators in aggrecan-deficient mice are ameliorated to varying degrees by the expression of a novel Col2a1-driven chicken aggrecan transgene. These findings show that aggrecan is an essential ECM component that affects chondrocyte survival, morphological transition, and gene expression in growth plate cartilage, which may aid in elucidating therapeutic targets for a variety of chondrodysplasias, arthritic diseases, and other cartilage-related disorders associated with altered levels of aggrecan.
Supplementary Material
Highlights.
The rate of element ossification is in part controlled by aggrecan embryonically.
Aggrecan influences chondrocyte columnar orientation and hypertrophic maturation.
Ihh and Sox9 production by chondrocytes depends on proper aggrecan expression.
Chondrocyte survival and proliferation are contingent upon aggrecan expression.
ACKNOWLEDGEMENTS
The authors would like to thank Dr. Véronique Lefebvre (Cleveland Clinic) for kindly providing the Col2a1 construct, Linda Degenstein (University of Chicago Transgenic Core) for her technical assistance in creating the transgenic mice, James Mensch for editorial assistance and Natalie Rupp for technical assistance and animal care.
Supported by: U.S. Public Health Service Grant HD-17332
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Abad V, Meyers JL, Weise M, Gafni RI, Barnes KM, Nilsson O, Bacher JD, Baron J. The Role of the Resting Zone in Growth Plate Chondrogenesis. Endocrinology. 2002;143:1851–1857. doi: 10.1210/endo.143.5.8776. [DOI] [PubMed] [Google Scholar]
- Bell L, Juriloff M, Harris MJ. A new mutation at the cmd locus in the mouse. Journal of Heredity. 1986;77:205–206. doi: 10.1093/oxfordjournals.jhered.a110215. [DOI] [PubMed] [Google Scholar]
- Bi W, Deng JM, Zhang Z, Behringer RR, de Crombrugghe B. Sox9 is required for cartilage formation. Nature genetics. 1999;22:85–89. doi: 10.1038/8792. [DOI] [PubMed] [Google Scholar]
- Brennan MJ, Oldberg A, Ruoslahti E, Brown K, Schwartz NB. Immunological evidence for two distinct chondroitin sulfate proteoglycan core proteins: differential expression in cartilage matrix deficient mice. Dev. Biol. 1983;98:139–147. doi: 10.1016/0012-1606(83)90342-1. [DOI] [PubMed] [Google Scholar]
- Brinster RL, Chen HY, Trumbauer M, Senear AW, Warren R, Palmiter RD. Somatic expression of herpes thymidine kinase in mice following injection of a fusion gene into eggs. Cell. 1981;27:223–231. doi: 10.1016/0092-8674(81)90376-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brinster RL, Chen HY, Trumbauer ME, Yagle MK, Palmiter RD. Factors affecting the efficiency of introducing foreign DNA into mice by microinjecting eggs. Proc Natl Acad Sci U S A. 1985;82:4438–4442. doi: 10.1073/pnas.82.13.4438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, S G. Dual roles for patched in sequestering and transducing Hedgehog. Cell. 1996;87:553–563. doi: 10.1016/s0092-8674(00)81374-4. [DOI] [PubMed] [Google Scholar]
- Cooper KL, O S, Sung Y, Dasari RR, Kirschner MW, Tabin CJ. Multiple phases of chondrocyte enlargement underlie differences in skeletal proportions. Nature. 2013;495:375–378. doi: 10.1038/nature11940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cortes M, Baria AT, Schwartz NB. Sulfation of chondroitin sulfate proteoglycans is necessary for proper Indian hedgehog signaling in the developing growth plate. Development. 2009;136:1697–1706. doi: 10.1242/dev.030742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cortes M, Mensch JR, Domowicz M, Schwartz NB. Proteoglycans: gene cloning. Methods Mol Biol. 2012;836:3–21. doi: 10.1007/978-1-61779-498-8_1. [DOI] [PubMed] [Google Scholar]
- Davidson D, Blanc A, Filion D, Wang H, Plut P, Pfeffer G, Buschmann MD, Henderson JE. Fibroblast growth factor (FGF) 18 signals through FGF receptor 3 to promote chondrogenesis. J Biol Chem. 2005;280:20509–20515. doi: 10.1074/jbc.M410148200. [DOI] [PubMed] [Google Scholar]
- Domowicz MS, Cortes M, Henry JG, Schwartz NB. Aggrecan modulation of growth plate morphogenesis. Dev Biol. 2009;329:242–257. doi: 10.1016/j.ydbio.2009.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domowicz MS, Pirok EW, III, Novak TE, Schwartz NB. Role of the Cterminal G3 domain in sorting and secretion of aggrecan core protein and ubiquitinmediated degradation of accumulated mutant precursors. J Biol Chem. 2000;275:35098–35105. doi: 10.1074/jbc.275.45.35098. [DOI] [PubMed] [Google Scholar]
- Domowicz MS, Sanders TA, Ragsdale CW, Schwartz NB. Aggrecan is expressed by embryonic brain glia and regulates astrocyte development. Dev Biol. 2008;315:114–124. doi: 10.1016/j.ydbio.2007.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dy P, W W, Bhattaram P, Wang Q, Wang L, Ballock RT, Lefebvre V. Sox 9 directs hypertrophic maturation and blocks osteoblast differentiation of growth plate chondrocytes. Dev Cell. 2012;22:597–609. doi: 10.1016/j.devcel.2011.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garofalo SK-SM, Cooke JL, Wolstin O, Lunstrum GP, Moshkovitz SM, Horton WA, Yayon A. Skeletal dysplasia and defective chondrocyte differentiation by targeted overexpression of fibroblast growth factor 9 in transgenic mice. J Bone Miner Res. 1999;14:1909–1915. doi: 10.1359/jbmr.1999.14.11.1909. [DOI] [PubMed] [Google Scholar]
- Gleghorn L, Ramesar R, Beighton P, Wallis G. A mutation in the variable repeat region of the aggrecan gene (AGC1) causes a form of spondyloepiphyseal dysplasia associated with severe, premature osteoarthritis. American journal of human genetics. 2005;77:484–490. doi: 10.1086/444401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iannotti JP, B CT. Cytosolic ionized calcium concentration in isolated chondrocytes from each zone of the growth plate. J Orthop Res. 1989;7:511–518. doi: 10.1002/jor.1100070408. [DOI] [PubMed] [Google Scholar]
- Ikegami D, A H, Suzuki A, Nakamura T, Nakano T, Yoshikawa H, Tsumaki N. Sox9 sustains chondrocyte survival and hypertrophy in part through Pik3ca-Akt pathways. Development. 2011;138:1507–1519. doi: 10.1242/dev.057802. [DOI] [PubMed] [Google Scholar]
- Ingham PW. Transducing Hedgehog: the story so far. Embo J. 1998;17:3505–3511. doi: 10.1093/emboj/17.13.3505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimata K, Barrach H-J, Brown KS, Pennypacker JP. Absence of proteoglycan core protein in cartilage from cmd/cmd (cartilage matrix deficiency) mice. J. Biol. Chem. 1981;256:6961–6968. [PubMed] [Google Scholar]
- Kimata K, Oike Y, Tani K, Shinomura T, Yamagata M. A large chondroitin sulfate proteoglyan (PG-M) synthesized before chondrogenesis in the limb bud of chick embryo. J. Biol. Chem. 1986;261:13517–13525. [PubMed] [Google Scholar]
- Kobayashi T, Soegiarto DW, Yang Y, Lanske B, Schipani E, McMahon AP, Kronenberg HM. Indian hedgehog stimulates periarticular chondrocyte differentiation to regulate growth plate length independently of PTHrP. J Clin Invest. 2005;115:1734–1742. doi: 10.1172/JCI24397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kosher RA, K WM, Gay SW. Collagen gene expression during limb cartilage differentiation. The Journal of cell biology. 1986;102:1151–1156. doi: 10.1083/jcb.102.4.1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krueger RC, Fields TA, Mensch JR, Schwartz NB. Chick cartilage chondroitin sulfate proteoglycan core protein II. Nucleotide sequence of cDNA clone and localization of the S103L epitope. J Biol Chem. 1990;265:12088–12097. [PubMed] [Google Scholar]
- Krueger RC, Kurima K, Schwartz NB. Completion of the mouse aggrecan structure and identification of the defect in the cmd-Bc as a near complete deletion of the murine aggrecan. Mamm. Genome. 1999;10:1119–1125. doi: 10.1007/s003359901176. [DOI] [PubMed] [Google Scholar]
- Kwak YH, R JY, Lee KS, Park HW, Kim HW. Altered synthesis of cartilage-specific proteoglycans by mutant cartilage oligomeric matrix protein. Clin Orthop Surg. 2009;1:181–187. doi: 10.4055/cios.2009.1.4.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacy E, Roberts S, Evans EP, Burtenshaw MD, Costantini FD. A foreign beta-globin gene in transgenic mice: integration at abnormal chromosomal positions and expression in inappropriate tissues. Cell. 1983;34:343–358. doi: 10.1016/0092-8674(83)90369-0. [DOI] [PubMed] [Google Scholar]
- Lefebvre V, Huang W, Harley VR, Goodfellow PN, de Crombrugghe B. SOX9 is a potent activator of the chondrocyte-specific enhancer of the pro alpha1(II) collagen gene. Mol Cell Biol. 1997;17:2336–2346. doi: 10.1128/mcb.17.4.2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewinson D, S M. Chondroclasts and endothelial cells collaborate in the process of cartilage resorption. Anat Rec. 1992;233:504–514. doi: 10.1002/ar.1092330403. [DOI] [PubMed] [Google Scholar]
- Li H, Schwartz NB, Vertel BM. cDNA cloning of chick cartilage chondroitin sulfate (aggrecan) core protein and identification of a stop codon in the aggrecan gene associated with the chondrodystrophy, nanomelia. J Biol Chem. 1993;268:23504–23511. [PubMed] [Google Scholar]
- Mallein-Gerin F, K RA, Upholt WB, Tanzer ML. Temporal and spatial analysis of cartilage proteoglycan core protein gene expression during limb development by in situ hybridization. Dev Biol. 1988;126:337–345. doi: 10.1016/0012-1606(88)90144-3. [DOI] [PubMed] [Google Scholar]
- Minina E, Wenzel HM, Kreschel C, Karp S, Gaffield W, McMahon AP, Vortkamp A. BMP and Ihh/PTHrP signaling interact to coordinate chondrocyte proliferation and differentiation. Development. 2001;128:4523–4534. doi: 10.1242/dev.128.22.4523. [DOI] [PubMed] [Google Scholar]
- Ovadia M, P CH, Lash JW. Changing patterns of proteoglycan synthesis during chondrogenic differentiation. J Embryol Exp Morphol. 1980;56:59–70. [PubMed] [Google Scholar]
- Rittenhouse E, Dunn LC, Cookingham J, Calo C, Spiegelman M, Dooher GB, Bennett D. Cartilage matrix deficiency (cmd): a new autosomal recessive lethal mutation in the mouse. J. Embryol. Exp. Morph. 1978;43:71–84. [PubMed] [Google Scholar]
- Ruoslahti E, Yamaguchi Y. Proteoglycans as modulators of growth factor activities. Cell. 1991;64:867–869. doi: 10.1016/0092-8674(91)90308-l. [DOI] [PubMed] [Google Scholar]
- Sambrook J, Russell DW. Molecular Cloning: A Laboratory Manual. Third ed. Cold Spring Harbor: Cold Spring Harbor Laboratory Press; 2001. [Google Scholar]
- Schindelin JS, A-C I, Frise E, Kaynig V, Longair M, Pietzsch T, Prebisch S, Rueden C, Saalfeld S, Scmid B, Tinevez J, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A. Fiji: an open-source platform for biological-image analysis. Nature Methods. 2012;9:7. doi: 10.1038/nmeth.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz NB, Domowicz M. Chondrodysplasias due to proteoglycan defects. Glycobiology. 2002;12:57R–68R. doi: 10.1093/glycob/12.4.57r. [DOI] [PubMed] [Google Scholar]
- Schwartz NB, Domowicz MS. Chondrodysplasias, Encyclopedia of Endocrine Disorders. San Diego, CA: Elsevier Science Inc; 2014. in press. [Google Scholar]
- Schwartz NB, Hennig AK, Krueger RC, Krzystolik M, Li H, Mangoura D. Developmental expression of S103L cross-reacting proteoglycans in embryonic chick. In: Fallon JF, Goetinck PF, Kelley RO, Stocum DL, editors. Limb Development and Regeneration. New York: Wiley-Liss, Inc.; 1993. pp. 505–514. [PubMed] [Google Scholar]
- Sekiya I, Tsuji K, Koopman P, Watanabe H, Yamada Y, Shinomiya K, Nifuji A, Noda M. SOX9 enhances aggrecan gene promoter/enhancer activity and is upregulated by retinoic acid in a cartilage-derived cell line, TC6. J Biol Chem. 2000;275:10738–10744. doi: 10.1074/jbc.275.15.10738. [DOI] [PubMed] [Google Scholar]
- Shapses SA, S LJ, Ratcliffe A. Differential rates of aggrecan synthesis and breakdown in different zones of the bovine growth plate. Matrix biology : journal of the International Society for Matrix Biology. 1994;14:77–86. doi: 10.1016/0945-053x(94)90031-0. [DOI] [PubMed] [Google Scholar]
- Stattin EL, W F, Lindblom K, Onnerfjord P, Jonsson BA, Tegner Y, Sasaki T, Struglics A, Lohmander S, Dahl N, Heinegard D, Aspberg A. A missense mutation in the aggrecan C-type lectin domain disrupts extracellular matrix interactions and causes dominant familial osteochondritis dissecans. American journal of human genetics. 2010;86:126–137. doi: 10.1016/j.ajhg.2009.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tompson SW, Merriman B, Funari VA, Fresquet M, Lachman RS, Rimoin DL, Nelson SF, Briggs MD, Cohn DH, Krakow D. A recessive skeletal dysplasia, SEMD aggrecan type, results from a missense mutation affecting the C-type lectin domain of aggrecan. American journal of human genetics. 2009;84:72–79. doi: 10.1016/j.ajhg.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vertel BM, Grier BL, Li H, Schwartz NB. The chondrodystrophy, nanomelia: biosynthesis and processing of the defective aggrecan precursor. Biochem. J. 1994;301:211–216. doi: 10.1042/bj3010211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vissers LE, L E, Unger S, Campos-Xavier AB, Gilissen C, Rossi A, Del Rosario M, Venselaar H, Knoll U, Nampoothiri S, Nair M, Spranger J, Brunner HG, Bonafe L, Veltman JA, Zabel B, Superti-Furga A. Chondrodysplasia and abnormal joint development associated with mutations in IMPAD1, encoding the Golgi-resident nucleotide phosphatase gPAPP. American journal of human genetics. 2011;88:608–615. doi: 10.1016/j.ajhg.2011.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vortkamp A, Lee K, Lanske B, Segre GV, Kronenberg HM, Tabin CJ. Regulation of rate of cartilage differentiation by Indian hedgehog and PTH-related protein. Science. 1996;273:613–622. doi: 10.1126/science.273.5275.613. [DOI] [PubMed] [Google Scholar]
- Wagner EF, Karsenty G. Genetic control of skeletal development. Current opinion in genetics & development. 2001;11:527–532. doi: 10.1016/s0959-437x(00)00228-8. [DOI] [PubMed] [Google Scholar]
- Watanabe H, Kimata K, Line S, Strong D, Gao L, Kozak CA, Yamada Y. Mouse cartilage matrix deficiency (cmd) caused by a 7 bp deletion in the aggrecan gene. Nat. Genet. 1994;7:154–158. doi: 10.1038/ng0694-154. [DOI] [PubMed] [Google Scholar]
- Zhao Q, Eberspaecher H, Lefebvre V, De Crombrugghe B. Parallel expression of Sox9 and Col2a1 in cells undergoing chondrogenesis. Developmental dynamics : an official publication of the American Association of Anatomists. 1997;209:377–386. doi: 10.1002/(SICI)1097-0177(199708)209:4<377::AID-AJA5>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- Zhou G, Garofalo S, Mukhopadhyay K, Lefebvre V, Smith CN, Eberspaecher H, de Crombrugghe B. A 182 bp fragment of the mouse pro alpha 1(II) collagen gene is sufficient to direct chondrocyte expression in transgenic mice. Journal of cell science. 1995;108:3677–3684. doi: 10.1242/jcs.108.12.3677. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.