Abstract
Mycophenolate mofetil (MMF) is an immunosuppressive agent that has been shown to be effective in transplant patients. It is also efficacious in the management of lupus nephritis and useful in the treatment of autoimmune conditions because its mechanisms of action target T- and B- lymphocytes, leading to suppression of the cell-mediated immune response and antibody formation. MMF has been used successfully to treat immune-mediated conditions like myasthenia gravis, autoimmune hepatitis and immune cytopenias. However, the conditions for its optimal use for non-renal manifestations (e.g., hematological, neuropsychiatric, myocardial, pulmonary or cutaneous symptoms) in lupus patients are unclear. There have yet to be any randomized, controlled trials to guide the optimal dose and duration of MMF treatment in such situations. MMF is well tolerated and safe to use, although there are reports of serious adverse effects including urticaria, myopathy, Epstein-Barr virus-associated B-cell lymphoma, cytomegalovirus infection and disseminated varicella zoster infection. Immunosuppressive treatment with MMF and supportive care over the past few decades have led to improved clinical outcomes in patients with severe lupus nephritis. A favorable long-term prognosis can be ensured provided that effective treatment is instituted early, before irreversible renal parenchymal damage occurs. Another area of concern for patients is the increased cost of long-term MMF use.
Keywords: Systemic lupus erythematosus, Mycophenolate mofetil, Treatment
Özet
Mikofenolat mofetil (MMF) transplant hastalarında etkinliği gösterilmiş bir immünsüpresif ajandır. Lupus nefriti ve diğer otoimmün hastalıkların tedavisinde; T ve B lenfositleri hedef alan etki mekanizmasına bağlı olarak, hücresel immün yanıtı ve antikor oluşumunu baskılayarak, etkinliği gösterilmiştir. Myastenia gravis, otoimmün hepatit, immün sitopeniler gibi otoimmün hastalıklarda başarıyla kullanılmaktadır. Fakat, lupus hastalarında böbrek dışı tutulumlarda (hematolojik, nöropsikiyatrik, miyokardiyal, pulmoner, kütanöz vb.) optimal kullanımı kesinlik kazanmamıştır. Bu durumlarda kullanımında optimal doz ve süreye dair yol gösterici randomize kontrollü çalışma henüz bulunmamaktadır. MMF iyi tolere edilmekte ve kullanımı güvenli olmasına rağmen; ürtiker, miyopati, Epstein-Barr virüs ilişkili B-hücreli lenfoma, sitomegalovirüs enfeksiyonları ve dissemine varisella zoster gibi birtakım yan etkiler bildirilmiştir. Ciddi lupus nefritli hastalarda MMF ile immünsüpresif tedavi ve destek bakımı sayesinde son yıllarda klinik sonuçlarda iyileşme sağlanmıştır. İrreversibl renal parankimal hasar oluşmadan önce etkin tedavinin başlanması halinde uzun dönem prognoz daha iyi olabilir. MMF’in uzun süreli kullanımında hastalar için önemli olan bir diğer noktada artan maliyetidir.
Introduction
Mycophenolate mofetil (MMF), a mycophenolic acid (MPA) prodrug, depletes guanosine nucleotides through the inhibition of inosine-5’-monophosphate dehydrogenase (IMPDH), acting preferentially on T- and B-lymphocytes [1]. IMPDH is the rate-limiting enzyme in the de novo synthesis of guanosine nucleotides, and T- and B-lymphocytes depend on this pathway more than other cell types. MPA is also a more potent inhibitor of the type II isoform of IMPDH, which is expressed in activated lymphocytes, than of the type I isoform of IMPDH, which is expressed in most other cell types [2]. Therefore, MPA exerts a more potent cytostatic effect on lymphocytes than on other cell types. This is the main mechanism by which MPA suppresses the cell-mediated immune response and antibody formation. Additionally, MPA also inhibits the glycosylation and expression of adhesion molecules and hinders the recruitment of lymphocytes and monocytes into sites of inflammation [3]. The production of nitric oxide (NO) by inducible NO synthase (iNOS) is also decreased, without affecting the activity of constitutive NO synthases. This effect is mediated by MPA through the depletion of tetrahydrobiopterin, a cofactor of the inducible form of iNOS. Through these mechanisms, MMF exerts anti-inflammatory and immunosuppressive activities.
In contrast to calcineurin inhibitors, MMF is not nephrotoxic. It does not induce the production of transforming growth factor (TGF)-β, a cytokine that is fibrogenic. Additionally, MMF has no adverse effects on blood pressure, cholesterol levels or triglyceride levels in recipients. It was also noted that MPA is not mutagenic and inhibits the proliferation of human B-lymphocytes that are transformed by Epstein-Barr virus. MPA also suppresses the proliferation of human arterial smooth muscle cells. These two properties of MPA may decrease the risk of lymphoma development and proliferative arteriopathy in recipients of MMF. Analyses of clinical trials show that MMF reduces the incidence of early and late rejection, is protective against long-term deterioration of renal function, and reduces late renal allograft loss independently of acute rejection and without increasing the risk for malignancies [4]. Apart from renal transplants, MMF has also been found to be useful in the management of pancreatic, hepatic and cardiac transplants [5–9].
MMF is a suppressor of both T- and B-cell lymphocyte proliferation and has been used successfully for the prevention of acute and chronic rejection of renal allografts [10–13]. MMF has a selective antiproliferative effect on lymphocytes and inhibits antibody production by B-lymphocytes. MMF also induces deoxyguanosine nucleotide depletion and inhibits the transfer of fucose and mannose to glycoproteins including glycoprotein adhesion molecules. In view of the functions of adhesion molecules (facilitating the attachment of leukocytes to endothelial cells, playing a role in the initial interaction between leukocytes and endothelial cells, and involvement in the interactions between antigen-presenting cells and lymphocytes as well as effector lymphocytes and target cells), MMF should reduce the inflammatory process in its early stages.
At higher concentrations, which may be reached in the clinical setting, MMF has effects on cells that are unrelated to the immune system. It has an antiproliferative effect on vascular smooth muscle cells, even when pro-proliferative stimuli (e.g., angiotensin II and TGF-β) are present. This effect is not shared by other immunosuppressive drugs such as cyclosporine or tacrolimus. This antiproliferative effect on vascular smooth muscle cells may be of relevance concerning the effect of MMF on chronic allograft dysfunction. Because some glomerulopathies are associated with vascular lesions and microthrombus formation, which resemble vascular rejection, MMF might be of use in advanced stages of chronic glomerulopathies. Other documented chronic activities of MMF include reduction of glomerular hypertrophy and hyperfiltration, reduction of myofibroblast formation and collagen III deposition and reduction of tubular cell proliferation and interstitial fibrosis [10–14].
T- and B-lymphocytes are involved in the pathogenesis of autoimmune conditions. Hence, interference with their function or proliferation will be beneficial for the management of these conditions. Suppression of the cell-mediated immune response and antibody production are key elements in the successful treatment of many immune-mediated conditions. Therefore, MMF has a role to play in the management of such conditions. MMF has been a useful drug in the treatment of recurrent glomerulonephritis in allografts and all forms of primary glomerulonephritides, especially nephrotic syndrome, lupus nephritis and vasculitis.
Zandman-Goddard and Shoenfeld reviewed the evidence for the contribution of MMF in autoimmunity in animal models of systemic lupus erythematosus (SLE), mercury-induced auto-immune glomerulonephritis, diabetes mellitus, experimental autoimmune uveoretinitis and experimental allergic encephalitis [15]. Clinically, MMF has been used as a monotherapy or adjunct therapy for myasthenia gravis [16–17], chronic immune demyelinating polyneuropathy [18], chronic autoimmune hepatitis [19–20], immune cytopenias [21–23], autoimmune inflammatory myopathy [24], psoriatic arthritis [25], non-lupus glomerulopathies (IgA nephropathy, membranous nephropathy, focal segmental glomerulosclerosis, membranoproliferative glomerulonephritis or hepatitis C-associated glomerulonephritis) [26], autoimmune HCV-associated hematological disorders [27], systemic vasculitis [28] and inflammatory skin diseases (pemphigus vulgaris, pemphigus foliaceus and bullous pemphigoid) [29].
Treatment
Treatment of Non-renal Manifestations of SLE:
A. Hematological Manifestations
Hematological manifestations are common among lupus patients [30–32], and immune leukopenia, thrombocytopenia and hemolytic anemia are commonly encountered in the clinic. Most of these lupus-related cytopenias respond well to higher doses of corticosteroids or immunosuppressive drugs. Patients with refractory immune cytopenias are uncommon, but very high doses of corticosteroids and/or immunosuppressive agents are often necessary to control the disease. Therapy-related complications (for example, avascular necrosis associated with long-term high-dose corticosteroids or recurrent infections associated with high-dose cytotoxic drugs) have become significant issues that need to be addressed. In 2003, Vasoo et al. reported the successful use of MMF in the treatment of a lupus patient with refractory thrombocytopenia [33]. Prior treatment with high-dose corticosteroids, pulse methylprednisolone and intravenous immunoglobulin therapy had failed. The patient’s platelet counts returned to normal when MMF was instituted into the drug regimen. More recently, Chang [34] described another successful outcome with lupus-related refractory thrombocytopenia. Alba [35] and Mak et al. [36] have reported the use of MMF to treat lupus patients with hemolytic anemia refractory to conventional treatment and observed good responses. In the rare occurrence of pure red-cell aplasia in a lupus patient, a successful outcome was obtained with the administration of cyclosporine and MMF [37]. These cases represent a small series of successfully treated patients, but their promising outcomes give us an insight into the usefulness of MMF as a second- or third-line therapy for lupus patients with refractory immune cytopenias.
B. Neuropsychiatric Manifestations
Neuropsychiatric abnormalities, ranging from psychosis to cognitive deficits, occur frequently in lupus patients with active disease. Frequently, the severity of the neurological involvement requires chronic administration of high doses of corticosteroids and/or cytotoxic drugs, with their attendant complications. However, there are no randomized controlled trials assessing the efficacy of MMF in such lupus patients reported to date. Most of the relevant literature consists of anectodal reports. Jose and co-workers successfully treated and maintained a lupus patient with psychotic manifestations with MMF [38]. Another reported success involved a case of cerebral vasculitis in a patient with hereditary complete C4 deficiency and SLE. She was treated with a combination of immunoadsorption and MMF [39]. Additionally, Mok et al. have reported their preliminary experience with MMF in the treatment of a patient with a lupus-related myelopathy in the spinal cord [40].
C. Myocardial and Pulmonary Manifestations
Pericarditis and serositis (e.g., pleurisy) occur in lupus patients and are usually treated adequately with corticosteroids. However, more serious involvements, such as myocarditis with pulmonary hemorrhage, are uncommon but can be fatal. Treatment of severe lupus-related pulmonary hemorrhage is often difficult. Samad reported on the use of MMF to treat a patient with childhood SLE with recurrent pulmonary hemorrhage [41]. Regarding another pulmonary disorder, Swigris and co-workers performed a retrospective survey of 28 patients with connective tissue-related interstitial lung disease, one of whom had SLE, and found that MMF preserves lung function and is safe and well tolerated [42]. In animal studies, MMF has been shown to prevent the development of experimental autoimmune myocarditis [43]. Hence, MMF may be useful in the management of lupus-related myocarditis. In a review of 12 lupus-related hemophagocytic syndrome patients with high prevalance of pericarditis and/or myocarditis. MMF was successfully used as an adjunct in the long-term treatment regimens for two of the patients [44].
D. Cutaneous Manifestations
Cutaneous lesions are common manifestations in lupus patients, and discoid rashes often lead to scarring of the involved skin. In 2001, Goyal reported the successful treatment of recalcitrant palmoplantar lesions in two lupus patients [45], and a year later, Schanz described the resolution of extensive and refractory subacute cutaneous lupus erythematosus with MMF in another two patients. These patients had unfortunately developed severe complications while on high-dose corticosteroid therapy [46]. Hanjani and Nousari expanded the use of MMF for the treatment of four lupus patients with cutaneous lupus (lupus tumidus, lupus panniculitis, discoid LE with perniosis and subacute LE) [47]. These reports collectively testify to the promise that MMF holds for the treatment of lupus-related cutaneous manifestations. However, Pisoni recently reported poor results from the use of MMF to treat refractory cutaneous lesions. Of the seven patients with SLE and refractory skin involvement (including acute cutaneous lupus, subacute cutaneous lupus, discoid lupus erythematosus, vasculitis, urticarial rash and chilblain lupus), five did not respond, one demonstrated only a partial response and one showed an initial response, with subsequent relapse while still on MMF [48]. Hence, the usefulness of MMF in the management of lupus with refractory cutaneous involvement is yet to be fully understood.
E. Treatment Strategies
MMF seems to have promise as a second- or third-line agent for the treatment of refractory non-renal lupus manifestations. It is relatively well tolerated [49–51], safe to use and shows no nephrotoxicity or adverse effects on blood pressure, cholesterol levels or triglyceride levels. It is not associated with significant risk of ovarian toxicity. Despite its clinical safety and relatively mild, common side-effects, such as nausea, vomiting and diarrhea, there are other concerns associated with MMF use. Urticaria [52], myopathy [53], Epstein-Barr virus-associated B-cell lymphoma [54], cytomegalovirus (CMV) infections [55,56] and disseminated varicella zoster infections [57,58] have been reported.
Anectodal reports of the successful treatment of refractory non-renal lupus manifestations cannot allow us to definitively determine the optimal dose or duration of treatment. Hence, the lack of randomized, double-blind controlled trials presents a drawback in the effort to optimize the use of MMF in such situations. In the renal setting, the Tricontinental Mycophenolate Mofetil Renal Transplantation Study Group [59] determined that a dosage of 2–3 g of MMF daily is efficacious and that the lower, 2-g daily dose regimen is associated with fewer side effects and CMV infections. Another drawback to the wide use of MMF is the high cost of the drug. Its cost can sometimes be prohibitive, especially to those not covered by comprehensive medical insurance and those residing in developing countries.
Lupus Nephritis Treatment
The management of severe proliferative lupus nephritis can be divided into an initial induction phase followed by a prolonged maintenance phase (Figure 1). Immunosuppressive treatment during the two phases has the respective aims of achieving remission and preventing relapse. Immunosuppressive medications, tailored according to disease activity, remain the mainstay of treatment for severe lupus nephritis. Commonly adopted treatment regimens include combinations of corticosteroids and anti-proliferative agents, such as cyclophosphamide, azathioprine or MMF.
Fig. 1.
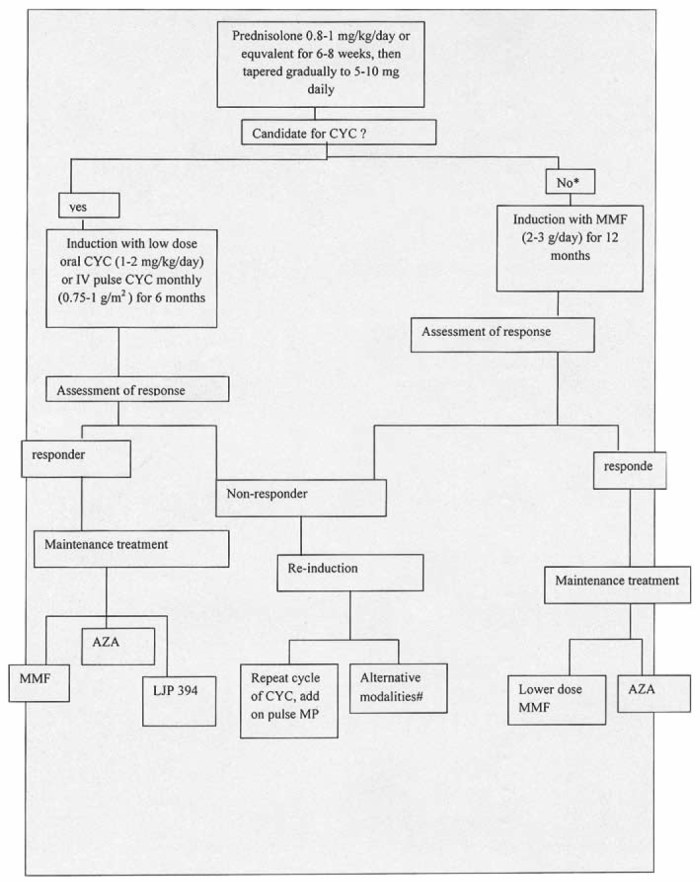
Treatment algorithm for diffuse proliferative lupus nephritis. *: Prior serious toxicities due to CYC, severe cytopenia, patient reluctance, etc. #: Alternative treatments including MMF and cyclosporine, A: immunoadsorption, intravenous immunoglobulin, AZA: azathioprine, MP: methylprednisolone, CYC: cyclophosphamide, MMF: mycophenolate mofetil [75].
The role of MMF in improving long-term outcomes of lupus nephritis patients remains unknown. An ongoing, large-scale multicenter randomized controlled trial will determine the effectiveness of MMF compared to intravenous cyclophosphamide during the induction stage and MMF compared to azathioprine during the maintenance phase. The optimal treatment regimen for lupus nephritis varies according to several factors: class, activity and chronicity indices and other prognostic factors (Tables 1, 2).
Table 1.
Activity and chronicity indices.
| Activity index | Chronicity index | |
|---|---|---|
| Glomerular abnormalities | Cellular proliferation | Glomerular sclerosis |
| Fibrinoid necrosis, karyorrhexis | Fibrous crescents | |
| Cellular crescents | ||
| Hyaline thrombi, wire loops | ||
| Leukocyte infiltration | ||
| Tubulointerstitial abnormalities | Mononuclear cell infiltrates | Interstitial fibrosis |
| Tubular atrophy |
Table 2.
Prognostic factors of lupus nephritis.
| Renal factors | Non-renal factors |
|---|---|
| Abnormal renal function at presentation | Male sex |
| Delay in starting immunosuppressive therapy | Hematologic features (thrombocytopenia and leukopenia) |
| Renal response during treatment | |
| Presence of renal flares | Younger age at diagnosis |
| Persistent hypocomplementemia | |
| Raised anti-dsDNA antibodies pre-treatment | |
| Raised anti-dsDNA antibodies after treatment | |
| Anti-phospholipid antibodies |
MMF has been widely used to prevent renal allograft rejection. Many case series and small controlled trials have suggested the effectiveness of MMF in the treatment of lupus nephritis. In early trials, cyclophosphamide (CYC) in combination with glucocorticoids (GC) led to improved renal survival compared with GC therapy alone and achieved lower rates of recurrence. Intravenous CYC became preferred over oral CYC due to perceived lower levels of toxicity (i.e., increased risk of infection, ovarian failure, reversible alopecia and bladder toxicity, particularly with the use of pre-treatment hydration).
Subsequent studies have shown that a longer duration of therapy during the maintenance phase improved remission rates [60]. A recent randomized, open-label, non-inferiority trial supports the notion that MMF is as effective as intravenous CYC in inducing short-term remission of lupus nephritis with a better safety profile [61]. Regarding the management of lupus nephritis, the role of MMF and its safety profile and cost-effectiveness have been analyzed in several clinical trials by different groups [62–75].
Conclusion
The issues of optimal dose, duration of treatment and cost-effectiveness of MMF can only be adequately addressed with randomized, controlled trials. Advances in immunosuppression and supportive care over the past few decades have led to improved clinical outcomes in patients with severe lupus nephritis, with increased efficacy and fewer complications. However, based on anectodal reports, MMF is a useful addition to the armamentarium available for treatment of lupus patients with refractory non-renal manifestations. Another drawback to the wide use of MMF is the high cost of the drug. Its cost can sometimes be prohibitive, especially to those not covered by comprehensive medical insurance and those residing in developing countries.
Footnotes
Conflict interest statement The authors declare that they have no conflict of interest to the publication of this article.
References
- 1.McMurray RW, Harisdangkul V. Mycophenolate mofetil: selective T cell inhibition. Am J Med Sci. 2002;323:94–6. doi: 10.1097/00000441-200204000-00005. [DOI] [PubMed] [Google Scholar]
- 2.Allison AC, Eugui EM. Mycophenolate mofetil and in mechanisms of action. Immunopharmacology. 2000;47:85–118. doi: 10.1016/s0162-3109(00)00188-0. [DOI] [PubMed] [Google Scholar]
- 3.Allison AC. Mechanisms of action of mycophenolate mofetil. Lupus. 2005;14:2–8. doi: 10.1191/0961203305lu2109oa. [DOI] [PubMed] [Google Scholar]
- 4.Lang P, Pardon A, Audard V. Long-term benefit of mycophenolate mofetil in renal transplantation. Transplantation. 2005;79:47–8. doi: 10.1097/01.tp.0000153303.53900.83. [DOI] [PubMed] [Google Scholar]
- 5.Garcia VD, Keitel E, Santos AF, et al. Immunosuppression in pancreas transplantation: mycophenolate mofetil versus sirolimus. Transplant Proc. 2004;36:975–7. doi: 10.1016/j.transproceed.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 6.Pierini A, Mirabella S, Brunati A, Ricchiuti A, Franchello A, Salizzoni M. Mycophenolate mofetil monotherapy in liver transplantation. Transplant Proc. 2005;37:2614–5. doi: 10.1016/j.transproceed.2005.06.005. [DOI] [PubMed] [Google Scholar]
- 7.Klupp J, Pfitzmann R, Langrehr JM, Neuhaus P. Indications of mycophenolate mofetil in liver transplantation. Transplantation. 2005;80:5142–6. doi: 10.1097/01.tp.0000187133.53916.8f. [DOI] [PubMed] [Google Scholar]
- 8.Kobashigawa JA, Meiser BM. Review of major clinical trials with mycophenolate mofetil in cardiac transplantation. Transplantation. 2005;80:235–43. doi: 10.1097/01.tp.0000186383.22264.b3. [DOI] [PubMed] [Google Scholar]
- 9.Sanchez V, Delgado JF, Morales JM, et al. Chronic cyclosporine-induced nephrotoxicity in heart transplant patients long-term benefits of treatment with mycophenolate mofetil and low-dose cyclosporine. Transplant Proc. 2004;36:2823–5. doi: 10.1016/j.transproceed.2004.09.035. [DOI] [PubMed] [Google Scholar]
- 10.Badid C, Desmouliere A, Laville M. Mycophenolate mofetil: implications for the treatment of glomerular disease. Nephrol Dial Transplant. 2001;16:1752–6. doi: 10.1093/ndt/16.9.1752. [DOI] [PubMed] [Google Scholar]
- 11.Choi MJ, Eustace JA, Gimenez LF, et al. Mycophenolate mofetil treatment for primary glomerular diseases. Kidney Int. 2002;61:1098–114. doi: 10.1046/j.1523-1755.2002.00214.x. [DOI] [PubMed] [Google Scholar]
- 12.Harzallah K, Badid C, Fouque D, et al. Efficacy of mycophenolate mofetil on recurrent glomerulonephritis after renal transplantation. Clin Nephrol. 2003;59:212–6. doi: 10.5414/cnp59212. [DOI] [PubMed] [Google Scholar]
- 13.Briggs WA, Choi MJ, Scheel PJ. Successful mycophenolate mofetil treatment of glomerular disease. Am J Kidney Dis. 1998;31:213–7. doi: 10.1053/ajkd.1998.v31.pm9469489. [DOI] [PubMed] [Google Scholar]
- 14.Romero F, Rodriguez B-Iturbe, Parra G, et al. Mycophenolate mofetil prevents the progressive renal failure induced by 5/6 renal ablation in rats. Kidney Int. 1999;55:945–55. doi: 10.1046/j.1523-1755.1999.055003945.x. [DOI] [PubMed] [Google Scholar]
- 15.Zandman-Goddard G, Shoenfeld Y. Mycophenolate mofetil in animal models of auto-immune disease. Lupus. 2005;14:12–6. doi: 10.1191/0961203305lu2112oa. [DOI] [PubMed] [Google Scholar]
- 16.Cahoon WD, Jr, Kockler DR. Mycophenolate mofetil treatment of myasthenia gravis. Ann Pharmacother. 2006;40:295–8. doi: 10.1345/aph.1G501. [DOI] [PubMed] [Google Scholar]
- 17.Prakash KM, Ratnagopal P, Puvanendran K, Lo YL. Mycophenolate mofetil – as an adjunctive immunosuppressive therapy in refractory myasthenia gravis: The Singapore experience. J Clin Neurosci. 2006 doi: 10.1016/j.jocn.2005.12.008. [DOI] [PubMed] [Google Scholar]
- 18.Gorson KC, Amato AA, Ropper AH. Efficacy of Mycophenolate mofetil in patients with chronic immune demyelinating polyneuropathy. Neurology. 2004;63:715–7. doi: 10.1212/01.wnl.0000134676.05850.c0. [DOI] [PubMed] [Google Scholar]
- 19.Chatur N, Ramji A, Bain VG, et al. Transplant immunosuppressive agents in non-transplant chronic autoimmune hepatitis: the Canadian association for the study of liver (CASL) experience with Mycophenolate mofetil and tacrolimus. Liver Int. 2005;25:723–7. doi: 10.1111/j.1478-3231.2005.01107.x. [DOI] [PubMed] [Google Scholar]
- 20.Devlin SM, Swain MG, Urbanski SJ, Burak KW. Mycophenolate mofetil for the treatment of autoimmune hepatitis in patients refractory to Standard therapy. Can J Gastroenterol. 2004;18:321–6. doi: 10.1155/2004/504591. [DOI] [PubMed] [Google Scholar]
- 21.Hou M, Peng J, Shi Y, et al. Mycophenolate mofetil (MMF) for the treatment of steroid–resistant idiopathic thrombocytopenic purpura. Eur J Hematol. 2003;70:353–7. doi: 10.1034/j.1600-0609.2003.00076.x. [DOI] [PubMed] [Google Scholar]
- 22.Lin JT, Wang WS, Yen CC, et al. Myelodisplastic syndrome complicated by autoimmune hemolytic anemia: remission of refractory anemia following Mycophenolate mofetil. Ann Hematol. 2002;81:723–6. doi: 10.1007/s00277-002-0539-3. [DOI] [PubMed] [Google Scholar]
- 23.Zimmer-Molsberger B, Knauf W, Thief E. Mycophenolate mofetil for severe autoimmune hemolytic anemia. Lancet. 1997;350:1003–4. doi: 10.1016/S0140-6736(05)64068-8. [DOI] [PubMed] [Google Scholar]
- 24.Majithia V, Harisdangkul V. Mycophenolate mofetil (Cellcept): an alternative therapy for autoimmune inflammatory myopathy. Rheumatology. 2005;44:386–9. doi: 10.1093/rheumatology/keh499. [DOI] [PubMed] [Google Scholar]
- 25.Schrader P, Mooser G, Peter RU, Puhl W. Preliminary results in the therapy of psoriatic arthritis with Mycophenolate mofetil. Z Rheumatol. 2002;61:545–50. doi: 10.1007/s00393-002-0369-7. [DOI] [PubMed] [Google Scholar]
- 26.Karim MY, Abbs IC. Mycophenolate mofetil in nonlupus glomerulonephropathy. Lupus. 2005;14:39–41. doi: 10.1191/0961203305lu2116oa. [DOI] [PubMed] [Google Scholar]
- 27.Ierardi E, Rendina M, Francavilla R, et al. Mycophenolate mofetil in the treatment of autoimmune HCV-associated hematological disorders showing steroid resistance or dependence. J Viral Hepat. 2003;10:390–3. doi: 10.1046/j.1365-2893.2003.00438.x. [DOI] [PubMed] [Google Scholar]
- 28.Koukoulaki M, Jayne DR. Mycophenolate mofetil in anti-neutrophil cytoplasm antibodies-associated systemic vasculitis. Nephron Clin Pract. 2006;102:100–7. doi: 10.1159/000089667. [DOI] [PubMed] [Google Scholar]
- 29.Nousari HC, Sragovich A, Kimyasi-Asadi A, Orlinsky D, Anhalt GJ. Mycophenolate mofetil in autoimmune and inflammatory skin disorders. J Am Acad Dermatol. 1999;40:265–8. doi: 10.1016/s0190-9622(99)70203-3. [DOI] [PubMed] [Google Scholar]
- 30.Fong KY, Thumboo J, Koh ET, et al. Systemic lupus erythematosus: initial manifestations and clinical features after 10 years of disease. Ann Acad Med Singapore. 1997;26:278–81. [PubMed] [Google Scholar]
- 31.Alsaeid K, Kamal H, Haider MZ, Al-Enezi HM, Malaviya AN. Systemic lupus erythematosus in Kuwaiti children: organ system involvement and serological findings. Lupus. 2004;13:613–7. doi: 10.1191/0961203304lu1075xx. [DOI] [PubMed] [Google Scholar]
- 32.Mok CC, Lau CS. Lupus in Hong Kong Chinese. Lupus. 2003;12:717–22. doi: 10.1191/0961203303lu451xx. [DOI] [PubMed] [Google Scholar]
- 33.Vasoo S, Thumboo J, Fong KY. Refractory immune thrombocytopenia in systemic lupus erythematosus: response to mycophenolate mofetil. Lupus. 2003;12:603–2. doi: 10.1191/0961203303lu417cr. [DOI] [PubMed] [Google Scholar]
- 34.Chang HK. Successful tretament of refractory thrombocytopenia with mycophenolate mofetil in a patient with systemic lupus erythematosus. J Korean Med Sci. 2005;20:883–5. doi: 10.3346/jkms.2005.20.5.883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Alba P, Karim MY, Hunt BJ. Mycophenolate mofetil as a treatment for autoimmune hemolytic anaemia in patients with systemic lupus erythematosus and antiphospholipid syndrome. Lupus. 2003;12:633–5. doi: 10.1191/0961203303lu419cr. [DOI] [PubMed] [Google Scholar]
- 36.Mak A, Mok CC. Mycophenolate mofetil for refractory hemolytic anaemia in systemic lupus erythematosus. Lupus. 2005;14:856–8. doi: 10.1191/0961203305lu2163cr. [DOI] [PubMed] [Google Scholar]
- 37.Arcasoy MO, Chao NJ. T-cell mediated pure red-cell aplasia in systemic lupus erythematosus: response to cyclosporine A and Mycophenolate mofetil. Am J Hematol. 2005;78:161–3. doi: 10.1002/ajh.20237. [DOI] [PubMed] [Google Scholar]
- 38.Jose J, Paulose BK, Vasuki Z, Danda D. Mycophenolate mofetil in neuropsychiatric systemic lupus erythematosus. Indian J Med Sci. 2005;59:353–6. [PubMed] [Google Scholar]
- 39.Lhotta K, Wurzner R, Rosenkranz AR, et al. Cerebral vasculitis in a patient with hereditary complete C4 deficiency and systemic lupus erythematosus. Lupus. 2004;13:139–41. doi: 10.1191/0961203304lu489cr. [DOI] [PubMed] [Google Scholar]
- 40.Mok CC, Mak A, To CH. Mycophenolate mofetil for lupus related myelopathy. Ann Rheum Dis. 2006;5:971–3. doi: 10.1136/ard.2005.046615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Samad AS, Lindsley CB. Treatment of pulmonary hemorrhage in childhood systemic lupus erythematosus with Mycophenolate mofetil. South Med J. 2003;96:705–7. doi: 10.1097/01.SMJ.0000054424.20202.7D. [DOI] [PubMed] [Google Scholar]
- 42.Swigris JJ, Olson AL, Fischer A, et al. Mycophenolate mofetil is afe, well tolarated,and preserves lung function in patients with connective tissue disease-related interstitial lung disease. Chest. 2006;130:30–6. doi: 10.1378/chest.130.1.30. [DOI] [PubMed] [Google Scholar]
- 43.Kamiyoshi Y, Takahashi M, Yokoseki O, et al. Mycophenolate mofetil prevents the development of experimental autoimmune myocarditis. J Mol Cell Cardiol. 2005;39:467–77. doi: 10.1016/j.yjmcc.2005.04.004. [DOI] [PubMed] [Google Scholar]
- 44.Lambotte O, Khellaf M, Harmouche H, et al. Characteristics and long-term outcome of 15 episodes of systemic lupus erythematosus-associated hemophagocytic syndrome. Medicine. 2006;85:169–82. doi: 10.1097/01.md.0000224708.62510.d1. [DOI] [PubMed] [Google Scholar]
- 45.Goyal S, Nousari HC. Treatment of resistant discoid lupus erythematosus of the palms and soles with Mycophenolate mofetil. J Am Acad Dermatol. 2001;45:142–4. doi: 10.1067/mjd.2001.114297. [DOI] [PubMed] [Google Scholar]
- 46.Schanz S, Ulmer A, Rassner G, Fierlbeck G. Successful treatment of subacute cutaneous lupus erythematosus with Mycophenolate mofetil. Br J Dermatol. 2002;147:174–8. doi: 10.1046/j.1365-2133.2002.04875.x. [DOI] [PubMed] [Google Scholar]
- 47.Hanjani NM, Nousari HC. Mycophenolate mofetil for the treatment of cutaneous lupus erythematosus with smoldering systemic involvement. 2002;138:1616–8. doi: 10.1001/archderm.138.12.1616. [DOI] [PubMed] [Google Scholar]
- 48.Pisoni CN, Obermoser G, Cuadrado MJ, et al. Skin manifestations of systemic lupus erythematosus refractory to multiple treatment modalities: poor results with Mycophenolate mofetil. Clin Exp Rheumatol. 2005;23:393–6. [PubMed] [Google Scholar]
- 49.Riskalla MM, Somers EC, Fatica RA, McCune WJ. Tolerability of Mycophenolate mofetil in patients with systemic lupus erythematosus. J Rheumatol. 2003;30:1508–12. [PubMed] [Google Scholar]
- 50.Pisoni CN, Sanchez FJ, Karim Y, et al. Mycophenolate mofetil in systemic lupus erythematosus: efficacy and tolerability in 86 patients. J Rheumatol. 2005;32:1047–52. [PubMed] [Google Scholar]
- 51.Karim MY, Alba P, Cuadrado MJ. Mycophenolate mofetil for systemic lupus erythematosus refractory to other immunosuppressive agents. Rheumatology. 2002;41:876–82. doi: 10.1093/rheumatology/41.8.876. [DOI] [PubMed] [Google Scholar]
- 52.Szyper-Kravitz M, Sheinberg P, Sidi Y, Schiffenbauer Y, Trubniykov E, Shoenfeld Y. Hypersensitivity to Mycophenolate mofetil in systemic lupus erythematosus: diagnostic measures and successful desensitization. Int Arch Allergy Immunol. 2005;138:334–6. doi: 10.1159/000088873. [DOI] [PubMed] [Google Scholar]
- 53.Galindo M, Cabello A, Joven B. Mycophenolate mofetil induced myopathy in a patient with lupus nephritis. J Rheumatol. 2005;32:188–90. [PubMed] [Google Scholar]
- 54.Waldman MA, Callen JP. Self-resolution of Epstein-Barr virus-associated B-cell lymphoma in a patient with dermatomyositis following withdrawal of Mycophenolate mofetil and methotrexate. J Am Acad Dermatol. 2004;51:124–30. doi: 10.1016/j.jaad.2004.03.020. [DOI] [PubMed] [Google Scholar]
- 55.Woywodt A, Choi M, Schneider W, Kettritz R, Gobel U. Cytomegalovirus colitis during Mycophenolate mofetil therapy for Wegener’s granulomatosis. Am J Nephrol. 2000;20:468–72. doi: 10.1159/000046201. [DOI] [PubMed] [Google Scholar]
- 56.Song AT, Abdala E, Bonazzi PR, Bacchella T, Machado MC. Does Mycophenolate mofetil increase the risk of cytomegalovirus infection in solid organ transplant recipients?-A mini-review. Braz J Infect Dis. 2006;10:132–8. doi: 10.1590/s1413-86702006000200011. [DOI] [PubMed] [Google Scholar]
- 57.Rothwell WS, Gloor JM, Morgerstern BZ, Milliner DS. Disseminated varicella infection in pediatric renal transplant recipients treated with Mycophenolate mofetil. Transplantation. 1999;68:158–61. doi: 10.1097/00007890-199907150-00030. [DOI] [PubMed] [Google Scholar]
- 58.Lauzurica R, Bayes B, Frias C, et al. Disseminated varicella infection in adult renal allograft recipients: role of Mycophenolate mofetil. Transplant Proc. 2003;35:1758–9. doi: 10.1016/s0041-1345(03)00684-5. [DOI] [PubMed] [Google Scholar]
- 59.A blinded, randomized clinical trial of Mycophenolate mofetil for the prevention of acute rejection in cadaveric renal transplantation. The Tricontinental Mycophenolate mofetil Renal Transplantation Study Group. Transplantation. 1996;61:1029–37. [No authors listed] [PubMed] [Google Scholar]
- 60.Boumpas DT, Austin HA, III, Vaughn EM, et al. Controlled trial of pulse methylprednisolone versus two regimens of cyclophosphamide in severe lupus nephritis. Lancet. 1992;340:741–5. doi: 10.1016/0140-6736(92)92292-n. [DOI] [PubMed] [Google Scholar]
- 61.Ginzler E, Dooley M, Aranow C, et al. Mycophenolate mofetil or intravenous cyclophosphamide for lupus nephritis. N Engl J Med. 2005;353:2219–28. doi: 10.1056/NEJMoa043731. [DOI] [PubMed] [Google Scholar]
- 62.Halloran P, Mathew T, Tomlanovich S, Groth C, Hooftman L, Barker C. Mycophenolate mofetil in renal allograft recipients: a pooled efficacy analysis of three randomized, double-blind, clinical studies in prevention of rejection. Transplantation. 1997;63:39–47. doi: 10.1097/00007890-199701150-00008. [DOI] [PubMed] [Google Scholar]
- 63.Jonsson CA, Svensson L, Carlsten H. Beneficial effect of the inosine monophosphate dehydrogenase inhibitor Mycophenolate mofetil on survival and severity of glomerulonephritis in systemic lupus erythematosus (SLE)-prone MRLIpr/Ipr mice. Clin Exp Immunol. 1999;116:534–41. doi: 10.1046/j.1365-2249.1999.00901.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Van Bruggen MC, Walgreen B, Rijke TP, Berden JH. Attenuation of murine lupus nephritis by Mycophenolate mofetil. J Am Soc Nephrol. 1998;9:1407–15. doi: 10.1681/ASN.V981407. [DOI] [PubMed] [Google Scholar]
- 65.Dooley MA, Cosio FG, Nachman PH, et al. Mycophenolate mofetil therapy in lupus nephritis: clinical observations. J Am Soc Nephrol. 1999;10:833–9. doi: 10.1681/ASN.V104833. [DOI] [PubMed] [Google Scholar]
- 66.Glicklich D, Acharya A. Mycophenolate mofetil therapy for lupus nephritis refractory to intravenous cyclophosphamide. Am J Kidney Dis. 1998;32:318–22. doi: 10.1053/ajkd.1998.v32.pm9708620. [DOI] [PubMed] [Google Scholar]
- 67.Chan TM, Li FK, Tang CSO, et al. For the Hong Kong Guangzhou Nephrology Study Group Efficacy of Mycophenolate mofetil in patients with diffuse proliferative lupus nephritis. N Engl J Med. 2000;343:1156–62. doi: 10.1056/NEJM200010193431604. [DOI] [PubMed] [Google Scholar]
- 68.Chan TM, Wong WS, Lau CS, et al. Follow-up of patients with diffuse proliferative lupus nephritis treated with prednisolone and Mycophenolate mofetil. J Am Soc Nephrol. 2001;12:195A. [Google Scholar]
- 69.Contreras G, Pardo V, Leclercq B, et al. Sequential therapies for proliferative lupus nephritis. N Engl J Med. 2004;350:971–80. doi: 10.1056/NEJMoa031855. [DOI] [PubMed] [Google Scholar]
- 70.Grcevska L, Milovanceva M. Role of Mycophenolate mofetil in the treatment of lupus nephritis. Ann NY Acad Sci. 2007;1110:433–8. doi: 10.1196/annals.1423.045. [DOI] [PubMed] [Google Scholar]
- 71.Mak S, Lo K. Efficacy of enteric-coated Mycophenolate mofetil sodium in patients with active lupus nephritis. Nephrology. 2008;13:331–6. doi: 10.1111/j.1440-1797.2007.00862.x. [DOI] [PubMed] [Google Scholar]
- 72.Falcini F, Cappannini S. Mycophenolate mofetil for the treatment of Juvenile onset SLE: a multicenter study. Lupus. 2009;18:139–43. doi: 10.1177/0961203308094999. [DOI] [PubMed] [Google Scholar]
- 73.Sinclair A, Appel G. Mycophenolate mofetil as induction and maintenance therapy for lupus nephritis: rationale and protocol for the randomized, controlled Aspreva Lupus Management Study (ALMS) Lupus. 2007;16:972–80. doi: 10.1177/0961203307084712. [DOI] [PubMed] [Google Scholar]
- 74.Chan TM. Mycophenolate mofetil in the treatment of lupus nephritis: an appraisal of recent data. APLAR Jorunal of Rheumatology. 2006;9:403–7. [Google Scholar]
- 75.Lai KN, Tang Sydney CW, Mok CC. Treatment for lupus nephritis: A revisit. Nephrology. 2005;10:180–8. doi: 10.1111/j.1440-1797.2005.00372.x. [DOI] [PubMed] [Google Scholar]


