Abstract
Background
While transmission of the human Plasmodium species has declined, a significant increase in Plasmodium knowlesi/Plasmodium malariae cases was reported in Hulu Selangor, Selangor, Malaysia. Thus, a study was undertaken to determine the epidemiology and the vectors involved in the transmission of knowlesi malaria.
Methods
Cases of knowlesi/malariae malaria in the Hulu Selangor district were retrospectively reviewed and analyzed from 2009 to 2013. Mosquitoes were collected from areas where cases occurred in order to determine the vectors. Leucosphyrus group of mosquitoes were genetically characterized targeting the nuclear internal transcribed spacer 2 (ITS2) and mitochondrial cytochrome c oxidase subunit I (CO1). In addition, temporal and spatial analyses were carried out for human cases and vectors.
Results
Of the 100 microscopy diagnosed P. knowlesi/P. malariae cases over the 5 year period in the Hulu Selangor district, there was predominance of P. knowlesi/P. malariae cases among the young adults (ages 20–39 years; 67 cases; 67%). The majority of the infected people were involved in occupations related to agriculture and forestry (51; 51%). No death was recorded in all these cases.
Five hundred and thirty five mosquitoes belonging to 14 species were obtained during the study. Anopheles maculatus was the predominant species (49.5%) followed by Anopheles letifer (13.1%) and Anopheles introlatus (11.6%). Molecular and phylogenetic analysis confirmed the species of the Leucosphyrus group to be An. introlatus. In the present study, only An. introlatus was positive for oocysts. Kernel Density analysis showed that P. knowlesi hotspot areas overlapped with areas where the infected An. introlatus was discovered. This further strengthens the hypothesis that An. introlatusis is the vector for P. knowlesi in the Hulu Selangor district.
Unless more information is obtained on the vectors as well as macaque involved in the transmission, it will be difficult to plan effective control strategies. The utilization of modern analytical tools such as GIS (Geographic Information System) is crucial in estimating hotspot areas for targeted control strategies.
Conclusions
Anopheles introlatus has been incriminated as vector of P. knowlesi in Hulu Selangor. The cases of P. knowlesi are on the increase and further research using molecular techniques is needed.
Keywords: An. introlatus, Vector, P. knowlesi, Malaysia
Background
Malaria still remains a serious public health challenge in the tropics. However, with improved and effective treatment coupled with the use of long lasting insecticide treated nets, the burden of disease has decreased over the years [1]. In highly endemic countries, this disease is observed to be more preponderant in infants and young children [2]. Lately, with the decline in the number of cases, a contrasting observation noted that older children and adults are more susceptible to the disease.
Historically, malaria used to be a major health concern in Malaysia with >200,000 cases in the 1960s [3]. The introduction of a malaria eradication programme in the late 1960s, which continued to the late 80s has contributed to the significant reduction of malaria cases in Malaysia to approximately 43,000 cases in the early 1990s. During those years, the states of Kelantan, Pahang and Perak had the highest number of cases in peninsular Malaysia [4]. During the eradication period, DDT residual house spraying was the main method used to reduce malaria vectors. With economic development and the introduction of insecticide treated bednets, malaria was further reduced to approximately 12,000 cases in 2000. In 2001, 12,780 cases were reported and this was significantly scaled down to 4,725 cases in 2012 [5].
Four species of malaria parasites (namely Plasmodium falciparum, P. vivax, P. malariae and P.ovale) are known to affect humans. In 2004, Singh et al. [6] reported the presence of a large number of P. knowlesi cases in Sarawak, a state in Malaysian Borneo. Subsequently, P. knowlesi became known as the fifth malaria parasite species to infect humans [7]. Various studies have shown that P. knowlesi is not only present in Malaysia but has a wider presence in other parts of Southeast Asia as well, namely Singapore [8–10], Thailand [11–14], Myanmar [15, 16], Philippines [17], Indonesia [18, 19], Vietnam [20, 21] and Cambodia [22]. To date, only Laos and Timor-Leste are devoid of any reported P. knowlesi cases.
Lately, it was observed that as transmission of the human Plasmodium species declined, a significant increase in microscopy diagnosed P. knowlesi/P. malariae cases were reported [23]. A similar trend was also observed in peninsular Malaysia [24]. This was confirmed in the Ministry of Health Annual Report that in 2012, P. knowlesi was the predominant species comprising 38% of the malaria cases in Malaysia followed by P. vivax (31% of cases) [5]. In Malaysian Borneo, Sabah and Sarawak contributed most of the cases, whereas Pahang has always had a high number of P. knowlesi malaria cases [24, 25] in peninsular Malaysia.
Malaria is transmitted from one person to another by the bite of the female infected Anopheles mosquito. The main malaria vectors in Malaysia are Anopheles campestris and An. sundaicus (now known as An. epiroticus) in the coastal areas, and An. maculatus in the hilly regions of peninsular Malaysia, An. latens and An. balabacensis the predominant vectors in Sarawak and Sabah, respectively [26–28]. The malaria eradication programme worked very well in the coastal areas and the vectors were almost eliminated due to their resting behavior indoors [29]. However, in the hilly regions of peninsular Malaysia, An. maculatus remains the primary vector of human malaria. When the first case of P. knowlesi infection was reported in Pahang, Malaysia, the Leucosphyrus group of mosquitoes was incriminated as the vectors. The vectors incriminated were An. hackeri (for P. knowlesi) in the coastal area of Selangor [30] and An. cracens (then known as An. balabacensis) in Perlis (northern region of peninsular Malaysia) (for Plasmodium inui) [31]. However, An. hackeri was found to only biting monkeys, and was not reported to bite humans. In 2006, An. latens was incriminated as a vector of P. knowlesi in Kapit Sarawak [32, 33], An. cracens in Kuala Lipis Pahang [25, 34] and An. balabacensis in Sabah [35].
The state of Selangor consists of nine districts and the Hulu Selangor district had the highest number of knowlesi/malariae malaria cases (Records from State Health Department). For the past five years, the cases of knowlesi/malariae malaria have also increased in Hulu Selangor. However, the vector involved in the transmission of knowlesi malaria remained unknown in this district. The national malaria program strategy has been reoriented from control to elimination, and Malaysia is now working to eliminate malaria from the peninsular by 2015 and from Malaysian Borneo by 2020 [36].
In view of these recent events, it is imperative that a holistic approach is adopted in order to understand the transmission of P. knowlesi malaria better especially in this district. Thus, the objective of this study was to analyse the epidemiology of knowlesi/malariae malaria in Hulu Selangor in a more comprehensive manner, taking into consideration the human infection and vector distribution. This study actively collected mosquitoes from locations with human cases and in so doing, managed to determine the vectors in Hulu Selangor. Another interesting feature of the study involved the temporal and spatial analysis utilizing advanced tools such as GIS analysis to shed some light on the association of human infection and mosquito distribution in order to elucidate the transmission of P. knowlesi malaria in Hulu Selangor.
Methods
Ethical clearance
This study was approved by the Medical Ethics Committee of University of Malaya Medical Center (UMMC), Malaysia (Reference number: 890.12) and the National Medical Research Register (Reference number: NMRR-11-1050-10619). Prior to mosquito collection, volunteers who consented were provided antimalarial prophylaxis.
Epidemiology aspects
Study site
This study was carried out in the state of Selangor, the most developed state [23.5% of Malaysia’s total GDP (gross domestic product) in 2012; Department of Statistics, Malaysia] among the 13 states in Malaysia equipped with modern infrastructure. The state also has the most concentrated population in Malaysia, with high living standards and the lowest poverty rate in the country. Selangor state consists of 9 districts, namely, Gombak, Hulu Langat, Hulu Selangor, Klang, Kuala Langat, Kuala Selangor, Petaling, Sabak Bernam and Sepang.
For the present study, the focus area was on the Hulu Selangor district which is located in the northeastern part of Selangor. Its principal town is Kuala Kubu Bharu (KKB) (3°34′N 101°39′E / 3.567°N 101.650°E). The district of Hulu Selangor is blessed with natural beauty such as lush greenery, hilly terrains, scenic lakes and waterfalls. It provides an ideal place for nature activities especially for the city dwellers. The local people are mostly involved in agricultural activities, small businesses and factories. There are also communities of the indigenous people (locally known as the ‘Orang Asli’) living in this district.
Retrospective review of human malaria cases
Records of all human cases positive for P.knowlesi/P. malariae in the district of Hulu Selangor were meticulously retrieved from 1 January 2009 till 31 December 2013 from Kuala Kubu Bharu Health office in the district and cross-checked with data from the State Health Department, Selangor. These cases were microscopy diagnosed by experienced laboratory technicians and all positive cases and 10% of the negative cases were examined by the State Health Department, Selangor. Due to the limitatations of microscopy in distinguishing P. knowlesi and P. malariae species, it was decided to consider both these species as a single group (P. knowlesi/P. malariae) as practiced by William et al. [23] in their study.
Information regarding age, gender, race/nationality, occupation, work address, home address, location of source of infection (obtained during case investigation exercise) coupled with its latitudinal and longitudinal coordinates were gathered and entered into a database. To reduce any data entry error, entries were doubled checked by at least two data entry assistants.
Statistical analysis
Data analysis was carried out using the Statistical Package for the Social Sciences (SPSS) for Windows version 17 (SPSS Inc, Chicago, USA). Categorical variables such as infection rate was presented as frequencies and percentages.
Entomology
The study was carried out in the Hulu Selangor district from 2012 to 2013. Various locations were surveyed within the district where knowlesi malaria cases were reported. The sites surveyed were possible locations where the cases could have picked up the infection. Most of the sites were in secondary forested areas or in rubber plantations surrounded by scrub forest. The study sites included lowland areas and hilly terrains.
Mosquito collection, identification and dissection
Mosquito collections, using the bare-leg catch method [37] was carried out from 19:00 to 00:00. This is the only proven method of collection of Anopheles mosquitoes in this country. All mosquitoes collected were brought back to the laboratory for identification. The keys of Reid [26] and Sallum [38] were used for the identification of Anopheles mosquitoes. All Anopheles mosquitoes were dissected to extract the midguts, salivary glands and ovaries and these were examined for oocysts and sporozoites and parity status respectively.
DNA extraction and PCR
DNA was extracted from positive midguts and Leucosphyrus group mosquitoes using the DNeasy tissue kit (Qiagen, Germany), according to the manufacturer’s protocol. The extracted DNA was kept at -20°C until required. A hexaplex PCR detection system was employed to simultaneously detect the various Plasmodium species present according to a published protocol [39].
Molecular identification and sequencing targeting the internal transcribed spacer 2 (ITS2) and cytochrome c oxidase subunit I (CO1)mt DNA
To confirm the species of An. introlatus, PCR was carried on targeted at ITS2 and CO1 genes. The ITS2 was amplified using primers ITS2A (5′TGTGAACTGCAGGACA 3′) and ITS2B(5′TATGCTTAAATTCAGGGGGT 3′) [40]. For CO1, the primers used were UEA9.2 (5′CTA ACA TTT TTT CCT CAA CAT TTT TTA GG-3′) and UEAlO.2 (5′TTA TTAGTT AAT AAY GGT ART TCT G-3′) [41]. PCR reactions were performed as previously described [41] without modifications. Amplicons were subjected to electrophoresis on 1.5% agarose gels (Promega, Madison, WI). The amplified product from the gel was purified and sequenced bidirectionally on an ABI prism sequencer using the PCR primers (Genomics Bioscience and Technology Co Ltd. China).
Analysis of sequence data
Sequences were aligned and checked manually using BioEdit (version 7.1.11) (Applied Biosystem, UK). The sequences were aligned with other representative sequences obtained from the GenBank using ClustalW version 1.7. A Neighbor Joining (NJ) phylogenetic tree was constructed using the MEGA version 5.1 software with 1000 bootstrap replicates. Branches corresponding to partitions reproduced in less than 80% of bootstrap replicates were collapsed. All sequences were submitted to GenBank(KM032605;KM032606;KM032607;KM032608;KM032609;KM032610;KM032611;KM032612;KM032613;KM032614;KM032615;KM032616;KM032617;KM032618;KM032619;KM032620;KM032621).
Geo-positioning, temporal and spatial analysis
The geographic coordinate for P. knowlesi/P. malariae human cases from 2009 to 2013 with complete addresses of the source of infection were determined using handheld Garmin GPSMAP 60CSx and downloaded from the GPS memory card into the computer using the GPS Pathfinder software. These geographic coordinates were synchronized using World Geodetic System (WGS 1984), which serve the x (longitude or east–west) and y (latitude or north–south). The recorded coordinates of each location were imported into Microsoft Excel and documented as a vector program database. All the geo-positioned cases were then exported and stored into ArcGIS 9.3 software (ERSI, Redlands, CA, USA) as raster format for further spatial risk patterns analyses and exploration. These were then geo-referenced on a GIS map layer at a Hulu Selangor sub-district level as point features to create a new GIS layer representing the point location of each P. knowlesi/P. malariae case. The collected An. introlatus mosquitoes were also geo-referenced in the same layer as P. knowlesi/P. malariae cases to create a GIS database format.
The spatial distribution of P. knowlesi cases was analyzed using the Average Nearest Neighbor (ANN) method, a method that provides indication of whether the spatial distribution pattern of particular a case occurs randomly, is clustered or dispersed [42]. The ANN method measures the average nearest neighbor index (R) value of the particular distance between each feature location and its nearest neighbour’s location. If the R value is less than 1 (R < 1), the distribution of the cases being analyzed are considered clustered. If the R value is greater 1 (R > 1), the distribution patterns are considered dispersed, while if the R value is to equal 1 (R = 1), it indicates a random distribution pattern. The Z value indicates if the observed pattern is significantly different from a random pattern with a significance level of 0.001 [42]. The Average Nearest Neighbor (ANN) ratio was calculated as:
Where, Do is the observed average distance between each case and its nearest neighbours’ cases and DE is the expected average distance for the cases given in a random pattern.
A hot spot analysis using the Kernel Density estimation and interpolating technique was carried out to identify high-risk areas and density of P. knowlesi/P. malariae cases in neighborhood locations with reported infections. Kernel density estimation is an effective tool to identify high-risk areas within point patterns of disease incidence by producing a smooth, continuous surface that defines the level of risk for that area [43, 44]. The interpolation and dissemination pattern of P. knowlesi/P. malariae cases using Kernel Density is estimated based on the given formula:
Where, λ (s) is the estimated infected value by P. knowlesi per area; г is the smoothing factor, k is the kernel weighting function and s is the centre of the area and si is the location of the case.
Results
Overall malaria cases from 2009–2013 in Selangor state
From 2009 until 2013, there were a total of 1,284 cases of malaria reported in the state of Selangor, Malaysia. Of this total, 915 (71.3%) were caused by P.vivax, 175 (13.6%) by P. falciparum, 171(13.3%) by P. knowlesi/P. malariae, 22 (1.7%) mixed infections and 1 (0.1%) by P. ovale (Table 1). Based on the total samples according to year, there were 231 cases in 2009 with number of cases declining to 196 in 2010 before hovering around 286 (2011), 270 (2012) and 301 (2013) in recent years. However, only about 30% (379) of cases were locally acquired.
Table 1.
Malaria cases according to species from 2009 till 2013 for the state of Selangor
| Parasite species | 2009 | 2010 | 2011 | 2012 | 2013 | Total |
|---|---|---|---|---|---|---|
| P. vivax | 179 (77.5%) | 131 (66.8%) | 193 (67.5%) | 199 (73.7%) | 213 (70.8%) | 915 (71.3%) |
| P. falciparum | 31 (13.4%) | 28 (14.3%) | 42 (14.7%) | 28 (10.4%) | 46 (15.3%) | 175 (13.6%) |
| P. knowlesi /P. malariae | 21 (9.1%) | 25 (12.8%) | 43 (15%) | 41 (15.2%) | 41 (13.6%) | 171 (13.3%) |
| Mixed infections | 0 | 11 (5.6%) | 8 (2.8%) | 2 (0.7%) | 1 (0.3%) | 22 (1.7%) |
| P. ovale | 0 | 1 (0.5%) | 0 | 0 | 0 | 1 (0.1%) |
| Total | 231 (79 [34.2%])* | 196 (23[11.7%])* | 286 (116[40.6%])* | 270 (115 [42.6])* | 301 (46 [15.3])* | 1284 (379[29.5%])* |
*Locally acquired cases.
The P. knowlesi/P. malariae cases in Selangor rose from 21 cases (9.1% of 231) in 2009 to 25 cases (12.8% of 196) in 2010, then peaked at 43 cases (15% of 286) in 2011 and 41 cases both in 2012 (15.2% of 270) and 2013 (13.6% of 301).
Plasmodium knowlesi/Plasmodium malariaecases from 2009–2013 in Hulu Selangor district
Among the nine districts in Selangor, the Hulu Selangor district charted the most cumulative number of P. knowlesi/P. malariae cases consisting of 100 (58.48% of 171) cases over the five years (i.e., 2009–2013) period. There were 4 cases of P. knowlesi/P. malariae notified in 2009, 5 in 2010, then an increase of 7 fold to 35 cases in 2011, declining to 30 cases in 2012 and 26 cases in 2013.
Of these 100 P. knowlesi/P. malariae cases, 91 (91%) cases involved male versus 9 (9%) female cases (Table 2). Cases of P. knowlesi/P. malariae were more predominant among the young adults (ages 20–39 years; 67 cases; 67%) followed by those above 40 years (23 cases; 23%). Based on nationality, 51 cases (51%) were Malaysians followed by Indonesians (35 cases; 35%). The majority of the infected people were involved in occupation related to agriculture and forestry (51; 51%). No death was recorded in all these cases.
Table 2.
Characteristics of Plasmodium knowlesi/Plasmodium malariae cases from 2009 till 2013 in Hulu Selangor district (Total cases = 100)
| Variable | 2009 | 2010 | 2011 | 2012 | 2013 | Total |
|---|---|---|---|---|---|---|
| (Total case = 4) | (Total case = 5) | (Total case = 35) | (Total case = 30) | (Total case = 26) | ||
| Gender | ||||||
| Male | 4 | 5 | 33 | 25 | 24 | 91 |
| Female | 0 | 0 | 2 | 5 | 2 | 9 |
| Age (years) | ||||||
| <1-9 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10-19 | 1 | 0 | 2 | 4 | 3 | 10 |
| 20-29 | 2 | 2 | 6 | 9 | 7 | 26 |
| 30-39 | 0 | 3 | 19 | 10 | 9 | 41 |
| 40-49 | 0 | 0 | 4 | 2 | 5 | 11 |
| >50 | 1 | 0 | 4 | 5 | 2 | 12 |
| Nationality | ||||||
| Malaysian | 3 | 0 | 17 | 15 | 16 | 51 |
| Indonesian | 0 | 3 | 14 | 10 | 8 | 35 |
| Myanmarese | 0 | 1 | 1 | 1 | 0 | 3 |
| Cambodian | 0 | 0 | 2 | 0 | 0 | 2 |
| Filipino | 0 | 0 | 1 | 0 | 0 | 1 |
| Nepalese | 0 | 0 | 0 | 1 | 2 | 3 |
| Indian | 1 | 0 | 0 | 2 | 0 | 3 |
| Bangladeshi | 0 | 1 | 0 | 1 | 0 | 2 |
| Occupation | ||||||
| Agriculture | 0 | 3 | 22 | 13 | 10 | 48 |
| Construction | 0 | 1 | 0 | 4 | 2 | 7 |
| Forestry | 0 | 0 | 2 | 1 | 0 | 3 |
| Industry | 1 | 1 | 1 | 3 | 5 | 11 |
| Security/Soldier/Police | 1 | 0 | 3 | 3 | 2 | 9 |
| Civil servant | 0 | 0 | 0 | 1 | 0 | 1 |
| Hotel/Tourism | 1 | 0 | 1 | 0 | 1 | 3 |
| Self employed/Housewife/Maid | 0 | 0 | 5 | 2 | 3 | 10 |
| Student | 1 | 0 | 1 | 3 | 3 | 8 |
| Outcome | ||||||
| Alive | 4 | 5 | 35 | 30 | 26 | 100 |
| Dead | 0 | 0 | 0 | 0 | 0 | 0 |
Species composition of Anophelesmosquitoes
Five hundred and thirty five mosquitoes belonging to 14 species were obtained during the study as shown in Table 3. Anopheles maculatus was the predominant species (49.5%) followed by An. letifer (13.1%) and An. introlatus (11.6%). However, An. maculatus and An. letifer were collected in large numbers only once in a particular site. Anopheles latens was the only other mosquito belonging to the Leucosphyrus group besides An. introlatus.
Table 3.
Anopheles species collected from Hulu Selangor district between 2012 to 2013
| Mosquito species | Numbers collected | Percentage (%) |
|---|---|---|
| An. donaldi | 2 | 0.4 |
| An. hyrcanus gr. | 26 | 4.9 |
| An. introlatus | 62 | 11.6 |
| An. karwari | 39 | 7.3 |
| An. latens | 2 | 0.4 |
| An. letifer | 70 | 13.1 |
| An. maculatus | 265 | 49.5 |
| An. peditaniatus | 2 | 0.4 |
| An. philippinensis | 10 | 1.9 |
| An. separatus | 15 | 2.8 |
| An. sinensis | 19 | 3.6 |
| An. tesselatus | 1 | 0.2 |
| An. umbrosus gr | 2 | 0.4 |
| Total | 535 | 100.00 |
Molecular analysis confirmed the species of Leucosphyrus group to be An. introlatus. Phylogenetic analysis inferred from the NJ method showed that the ITS2 region of nine adult An. introlatus (KM032613-KM032621) caught from various areas where knowlesi malaria cases occurred formed a monophyletic clade supported by a strong bootstrap value (Figure 1). Further phylogenetic analysis based on the CO1 gene found that six isolates (KM032605-KM032609; KM032611) also clustered in a monophyletic clade (Figure 2).
Figure 1.
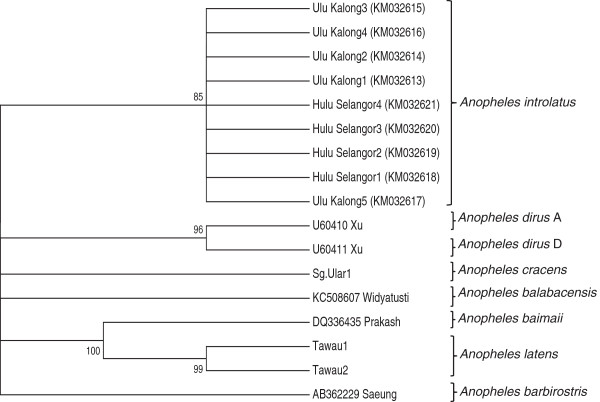
Phylogenetic tree based on the ITS2 gene produced by the neighbor-joining method. Figures on the branches are bootstrap percentages based on 1000 replicates.
Figure 2.
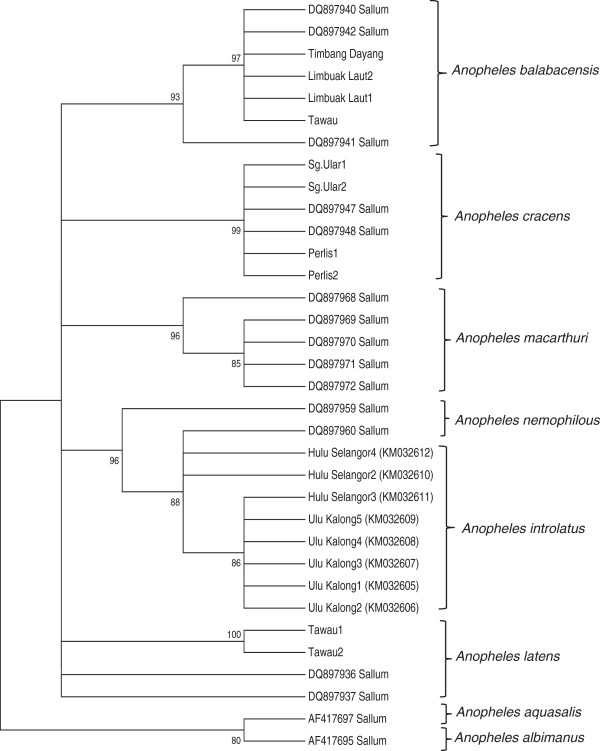
Phylogenetic tree based on the CO 1 genes produced by the neighbor-joining method. Figures on the branches are bootstrap percentages based on 1000 replicates.
Vector efficiency of Anopheles introlatus
The efficiency of a vector depends on how long it lives and how frequently it bites humans. The gonotrophic cycle of An. introlatus was taken as three days (this was based on observation in the laboratory where the blood fed mosquito laid eggs on the 3rd day after a blood meal). The daily survival rate [45], life expectancy [46] and vectorial capacity [47] of An. introlatus are shown in Table 4. The parous rate of An. introlatus was 65.5%. This showed that more than half of the mosquitoes were potentially dangerous. Of these, 25% of the An. introlatus would be expected to live the 10 days for the P. knowlesi sporozoites to be formed. Those surviving the 10 days would have a further life expectancy of 7.2 days. However, the vectorial capacity was only 1.94 since the man biting rate was low.
Table 4.
Parous rate, life expectancy and vectorial capacity of Anopheles introlatus
| Number dissected | 55 |
| Parous rate | 65.5 |
| Probability of survival (p)1 | 0.87 |
| P10 (%) | 25 |
| p10/-loge p (days)2 | 7.2 |
| Vectorial capacity 3 | 1.94 |
However, the most important index of transmission is the number of mosquitoes infected with sporozoites. In this study none of the mosquitoes were infected with sporozoites. Only two An. introlatus were positive for oocysts. One was found positive with 56 occysts in the midgut (Figure 3). The PCR results showed it was positive for P. knowlesi. The other had only two oocysts and PCR failed to detect any species. The infected rate was 1.82 (1/55 dissected). In addition, An. introlatus was found biting in the early part of the night. The peak biting rate was from 1900 to 2100 as shown in Figure 4. Most of the An. introlatus was obtained biting humans in the forest.
Figure 3.
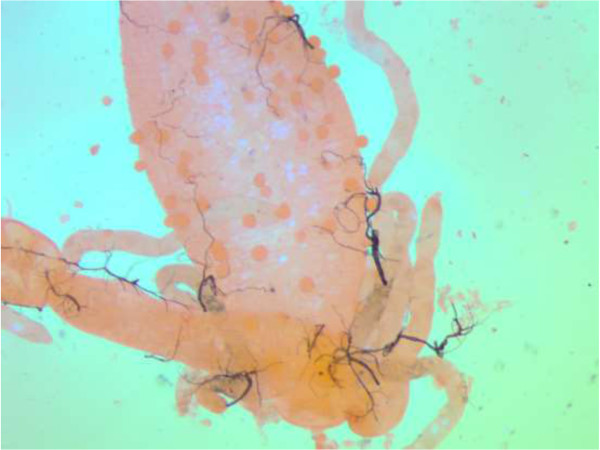
Midgut of the An. introlatus positive with 56 oocysts.
Figure 4.
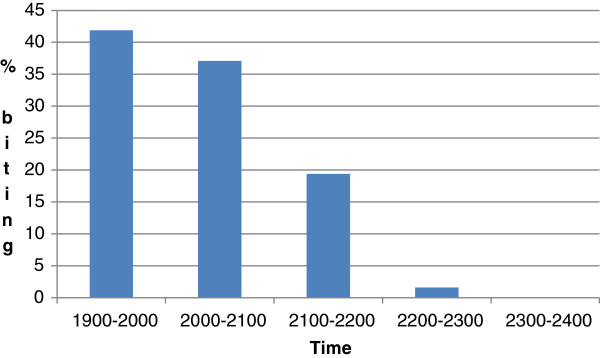
Biting times of Anopheles introlatus in Hulu Selangor district.
Temporal and spatial analysis of Plasmodium knowlesi/Plasmodium malariaecases
The ANN analysis of P. knowlesi/P. malariae cases demonstrated several dissemination patterns on a yearly basis (Figure 5). Generally, the distribution pattern of P. knowlesi/P. malariae cases from 2009 to 2013 in Hulu Selangor sub-districts were statistically clustered with an R value of less than 1 (R = 0.77; Z = -4.55; p < 0.01). The P. knowlesi/P. malariae cases in 2012 were clustered in certain sub-districts with an R value of 0.82 (Z = -186; p < 0.1). In contrast, a dispersed distribution pattern was observed for cases in 2011 (R = 1.16; z = 198; p < 0.05), 2010 (R = 1.40; z = 187; p < 0.1) and 2009 (R = 4.27; z = 12.52; p < 0.001), while a random dissemination pattern was reported for P. knowlesi/P. malariae cases that occurred in 2013 (R = 102; z = 021; p = 1).
Figure 5.
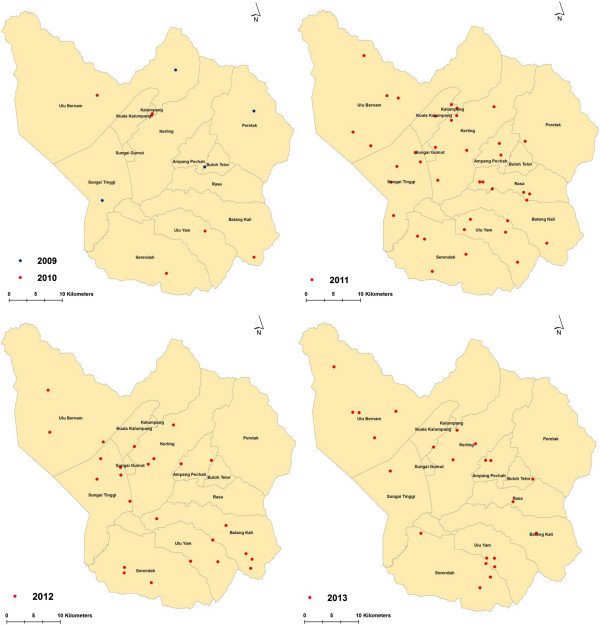
Temporal distribution pattern of P. knowlesi/P. malariae cases in Hulu Selangor sub-district, Selangor by year (2009–2013). The map demonstrates a clear spatial pattern of P. knowlesi/P. malariae cases that was mostly spread in all sub-districts in Hulu Selangor.
The prevalence of P. knowlesi/P. malariae cases reported in 2009 to 2013 was further analyzed using Kernel Density to identify high risk areas at sub-district level (Figure 6). The P. knowlesi density map can assist in precisely identifying the location, spatial extent and intensity of P. knowlesi/P. malariae case hotspots. The result showed that the red colored area was identified as a hotspot area with high P. knowlesi/P. malariae cases. In contrast, the location with yellow color indicates low density of hot spots areas. The Kernel Density map demonstrated the most affected area with P. knowlesi/P. malariae cases. Generally, most of the hot spot locations of P. knowlesi/P. malariae cases were mostly spread in all sub-districts of Hulu Selangor. It was demonstrated that the highest density hot spot of P. knowlesi/P. malariae cases were located in between the border of Sungai Tinggi and Sungai Gemut and the border of Kalumpang and Kerling. Likewise, Rasa and Serendah sub-districts were also identified as an area with high P. knowlesi/P. malariae cases.
Figure 6.
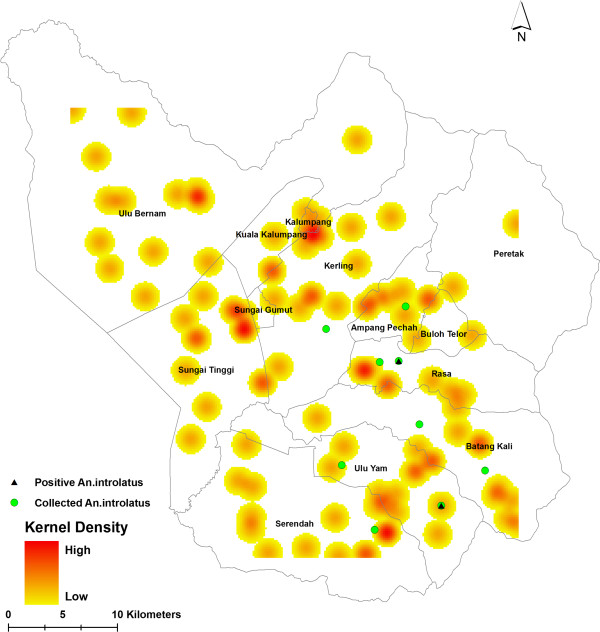
Hot spot location of P. knowlesi/P. malariae cases in Hulu Selangor sub-districts assessed using Kernel Density.
Discussion
In the state of Selangor, only 29.5% of malaria cases were locally acquired from 2009 until 2013. Throughout this period, although malaria cases were predominantly caused by P. vivax (71.3%) followed by P. falciparum (13.6%), cases of P. knowlesi/P. malariae (13.3%) is trailing very close to falciparum infections and showing an upward trend. The single grouping of P. knowlesi/P. malariae was adopted as the primary method of diagnosis of these malaria cases and was based on microscopy examination of peripheral blood smears with a small subset of specimens confirmed via PCR. In order to reduce death cases and heighten recognition and management of potentially fatal malaria species, Rajahram et al. [48]. suggested the need for microscopically diagnosed P. malariae to be reported as P. knowlesi in Sabah. Thus, it is highly recommended that future studies incorporate molecular techniques to decipher confirmed cases of P. knowlesi in order to arrive to a better conclusion with regards to the trend of P. knowlesi cases.
Now that knowlesi malaria is occurring in many areas, it is important to determine the vectors so that appropriate control measures can be instituted. Anopheles maculatus was the predominant mosquito obtained in this study and it is also the known vector of human malaria in peninsular Malaysia [26]. However, one needs to be careful in the interpretation of the data. Currently, only the Leucosphyrus group of mosquitoes have been incriminated as vectors of simian malaria [49]. Studies in Kuala Lipis also showed that An. cracens, which belongs to the Leucosphyrus group was the vector. Although An. maculatus was obtained in that study, none were positive [25, 34]. Laboratory studies have also shown that it is only the Leucosphyrus group of mosquitoes that are vectors [49]. The other mosquitoes for instance, An. freeborni can produce sporozoites but it cannot invade the salivary glands [50].
In the 1960s studies carried out in the lowland swamp forest using monkey bait and human bait traps, obtained four species of mosquitoes (not belonging to the Leucosphyrus group) namely An. donaldi, An. letifer, An. umbrosus, and An. roperi, which were positive for sporozoites. These sporozoites were inoculated into rhesus monkeys and long-tailed macaques but none turned positive [51]. Four decades later studies have shown that only Leucosphyrus group of mosquitoes are able to transmit simian malaria [25, 32–34].
In the present study, only An. introlatus was positive for oocysts. The mosquito must be able to develop sporozoites before it can be considered a vector. However, it is highly possible that An. introlatus will be able to produce sporozoites. The oocysts were not melanised and with such large numbers of oocysts being present in one mosquito, it may be the vector in the area. Moreover, Kernel Density analysis showed that P. knowlesi hotspot areas overlapped with areas where the infected An. introlatus was discovered. This further strengthens the hypothesis that An. introlatus is the vector for the P. knowlesi cases in the Hulu Selangor district. This is the first report of An. introlatus in Hulu Selangor. It has been previously reported from Gombak, Selangor, peninsular Malaysia [51], Thailand and Indonesia [38].
Studies in the 1960s have also incriminated An. introlatus as a vector of P. cynomolgi and P. fieldi [52]. Anopheles introlatus is known to be found only in small numbers in jungle areas [26]. In studies in the forest canopy using human and monkey bait traps at canopy level (15 meters), it was found that more An. introlatus were attracted to monkeys at canopy level compared to humans [51]. The An. introlatus in this study may be biting humans by chance and that could also be one reason why the number of cases was not very high as compared to Sabah and Sarawak in Malaysian Borneo. It has been demonstrated that An. introlatus are exophilic and early biters, which makes vector control very challenging. Thus, the current vector control measures like indoor residual spraying and insecticide treated nets will not be able break the chain of transmission and control the vectors. More innovative approaches will be required.
From this study, it is obvious that since mosquito populations are found only in small numbers, it is not possible to determine the vectors by carrying out surveys only once or twice in each site. A large number of field workers will have to be distributed throughout the sites in order to select the best sites for the collection of mosquitoes. This is also reflected in an earlier study [53], where they were not able to incriminate the vector. It is also known that the Asia Pacific Region has the highest number of dominant vector species for human malaria compared to other regions and is important to determine the correct species for control activities to succeed [54]. Thus, it shows that more rigorous sampling has to be carried out to determine the vectors of simian malaria. It has been noted that entomological studies are scanty and more areas need to be covered [55, 56]. However, one of the limitations for these studies is the involvement of humans for collection of mosquitoes.
With deforestation, the long tail macaques (Macaca fascicularis) have come to the forest fringes and perhaps these mosquitoes may have followed the macaques and colonized forest fringes. It is also possible that An. introlatus may be attracted more to monkeys than humans as shown in studies carried out four decades ago [51]. Studies should be carried out with monkey baited traps to determine if these mosquitoes are attracted to monkeys. Due to logistic problems, it was not possible to set up monkey baited traps.
In 2013, 15 M. fascicularis were trapped at Ulu Bernam, Kuala Kubu Bharu (Figure 7). Of this total, none were positive with P. knowlesi. However, the presence of other simian Plasmodium such as P. inui, P. coatneyi, P. fieldi and P cynomolgi were high and a majority had mixed infections (unpublished data, personal communication with Reuben Sharma). Although the macaques were not infected with P. knowlesi, the high infection rates (53.3% - 80%) with other simian malaria is of public health concern.
Figure 7.
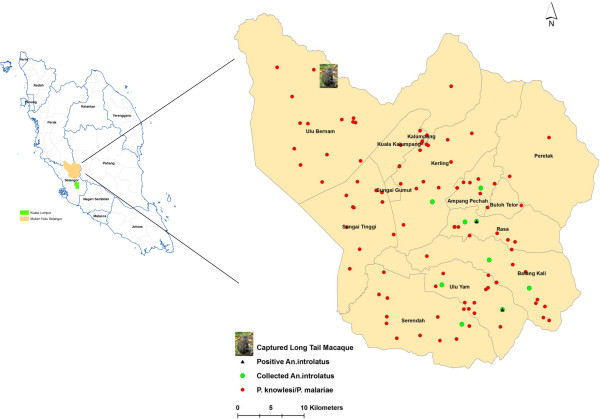
Spatial dissemination pattern of overall P. knowlesi/P. malariae cases, An. introlatus mosquitoes and long tail macaque in Hulu Selangor sub-districts.
This is especially so as a recent article has highlighted that besides P. knowlesi, another simian species, P. cynomolgi has also been found to infect humans naturally. This first known case was observed in a 39-year-old woman from a malaria-free area in Terengganu (east coast state of Malaysia). Although the initial diagnosis was P. malariae/P. knowlesi by microscopy, subsequent PCR assay with sequencing analysis confirmed that the patient was infected with P. cynomolgi [57]. As P. cynomolgi is morphologically indistinguishable from P. vivax, the true cases may be under-diagnosed [57]. In view of this limitation, it is crucial that novel molecular-based diagnostic tools are developed particularly those with multiplex systems to include the identification of these simian malaria species.
In terms of elimination of malaria it refers to only human malaria [58]. However, here is a situation where human malaria has been reduced or is in the process of being eliminated and simultaneously knowlesi malaria is on the rise in affecting humans. Thus, from the public health point of view, it is difficult to explain to the community that malaria has been eliminated when cases are still occurring in larger numbers than before.
Thus for elimination of malaria to be successful, the vector(s) need to be determined. With changing landscape and ecology, there will be changes to vector distribution and thus new maps for vector distribution is needed so as to enable an efficient control strategy.
As illustrated by the epidemiological data, the knowlesi malaria is also to some extent related to the migrant workers working in the forest fringe late at night tapping rubber. These people live in the forest fringe and the houses are just a bare minimum with temporary walls. Anopheles introlatus mosquitoes have been collected close by to their dwellings. In Southeast Asia, it has also been documented that the migrant population living in forest fringe villages, and also the ethnic minority groups in villages surrounded by forest are at risk of malaria [59]. In Thailand, Myanmar, eastern Bangladesh, western Cambodia, southern Laos and Vietnam, there is usually a close association between forests and the An. dirus complex. This vector may also be present in fruit orchards, but at lower density than in forests [60–62].
In peninsular Malaysia, it was always the An. maculatus that was the predominant vector transmitting malaria to the aborigine population in the forest area [37]. However, the situation has changed and now like the neighbouring countries the Leucosphyrus group mosquitoes are involved in malaria transmission [25, 34]. Thus, vector ecology and transmission patterns of knowlesi malaria present a unique challenge for vector control management, which is pivotal for malaria elimination.
In addition, based on the results of this study, utilization of tools such as GIS analysis should be highly recommended as a surveillance tool particularly in areas where there are high incidence rates. Spatial analysis has highlighted that in 2013, cases were highly clustered in certain sub-districts, however in 2012, a random dissemination pattern was observed. Having this knowledge at hand will enable control and prevention strategies to be more targeted, efficient and more cost effective. Finally, it also shows that the utilization of molecular techniques is crucial in the detection of the various malaria species so as to avoid mortality due to malaria species misidentification.
Conclusion
From this study, it can be concluded that more effort is warranted in research and development for control of knowlesi malaria cases. Unless more information is obtained on the vectors as well as macaques involved in the transmission, it will be difficult to plan any effective control strategies. The utilization of modern analytical tools such as GIS is crucial in estimating hotspot areas for targeted control strategies. With the elimination of human malaria, cases of knowlesi malaria will be on the increase in coming years as evident from this study. Thus, there is a greater need for multi disciplinary research to combat simian malaria affecting humans.
Acknowledgment
This study was supported by University of Malaya High Impact Research Grant UM.C/625/1/HIR/099 J-20011-73822; UM-MOHE (UM.C/HIR/MOHE/MED/18,UMRG (RG503/13HTM) and PPP Grant (PG056/2013A). The authors like to thank the staff of vector team Hulu Selangor for all their help in collection of mosquitoes and staff of the laboratory for data collection. Prof Chua Kek Heng for the hexaplex primers for PCR. Lee Col Lin, Anna Louisa John and Priya Mahalingam from the Parasitology Laboratory, Faculty of Veterinary Medicine, Universiti Putra Malaysia for providing unpublished data on the prevalence of Plasmodium in Macaca fascicularis from UluBernam, Kuala Kubu Bharu. The Director General of Health, Ministry of Health Malaysia for permission to publish.
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
Study design – IV, YALL, BV, RM. Sample collection – MLW, CSL, LTK, WYWS, JJ, LYL, CTZAG, NA, RSKS. Sample analysis – MLW, CSL, LTK, JJ, XTG, NJY. Statistical analysis – RN, XTG. Manuscript preparation – IV, YALL, RN, RM, LYL. All authors read and approved the final version of the manuscript.
Contributor Information
Indra Vythilingam, Email: indra.vythilingam@gmail.com.
Yvonne AL Lim, Email: limailian@um.edu.my.
Balan Venugopalan, Email: drbvenu@moh.gov.my.
Romano Ngui, Email: skyromano@gmail.com.
Cherng Shii Leong, Email: qqshiiqq@gmail.com.
Meng Li Wong, Email: mushy.mengli5298@gmail.com.
LokeTim Khaw, Email: khaw@um.edu.my.
XiangTing Goh, Email: gohxt88@gmail.com.
NanJiun Yap, Email: ynj1203@gmail.com.
Wan Yusoff Wan Sulaiman, Email: wanyus@um.edu.my.
John Jeffery, Email: johnjeffery4@gmail.com.
Ab Ghani CT Zawiah, Email: ctzawiah@moh.gov.my.
Ismail Nor Aszlina, Email: aszlina79@yahoo.com.
Reuben SK Sharma, Email: reuben@upm.edu.my.
Lau Yee Ling, Email: yllau@ummc.edu.my.
Rohela Mahmud, Email: rohela@ummc.edu.my.
References
- 1.O’Meara WP, Mangeni JN, Steketee R, Greenwood B. Changes in the burden of malaria in sub-Saharan Africa. Lancet Infect Dis. 2010;10(8):545–555. doi: 10.1016/S1473-3099(10)70096-7. [DOI] [PubMed] [Google Scholar]
- 2.Carneiro I, Roca-Feltrer A, Griffin JT, Smith L, Tanner M, Schellenberg JA, Greenwood B, Schellenberg D. Age-patterns of malaria vary with severity, transmission intensity and seasonality in sub-Saharan Africa: a systematic review and pooled analysis. PLoS One. 2010;5(2):e8988. doi: 10.1371/journal.pone.0008988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lim E. Current status of malaria in Malaysia. Southeast Asian J Trop Med Public Health. 1992;23:43–49. [PubMed] [Google Scholar]
- 4.Hafiz H, NorRasidah H. Spatial and temporal distribution of malaria in Peninsular Malaysia from 1998–2010. Health Enviro J. 2012;3(3):46–50. [Google Scholar]
- 5.Annual report ministry of health [ http://vlib.moh.gov.my/cms/content.jsp?id=com.tms.cms.section.Section_cc7aebb6-c0a81049-18ac8000-11671d74]
- 6.Singh B, Sung LK, Matusop A, Radhakrishnan A, Shamsul SSG, Cox-Singh J, Thomas A, Conway DJ. A large focus of naturally acquired Plasmodium knowlesi infections in human beings. Lancet. 2004;363(9414):1017–1024. doi: 10.1016/S0140-6736(04)15836-4. [DOI] [PubMed] [Google Scholar]
- 7.White N. Plasmodium knowlesi: the fifth human malaria parasite. Clin Infect Dis. 2008;46(2):172. doi: 10.1086/524889. [DOI] [PubMed] [Google Scholar]
- 8.Ong CW, Lee SY, Koh WH, Ooi E-E, Tambyah PA. Monkey malaria in humans: a diagnostic dilemma with conflicting laboratory data. Am J Trop Med Hyg. 2009;80(6):927–928. [PubMed] [Google Scholar]
- 9.Jeslyn WPS, Huat TC, Vernon L, Irene LMZ, Sung LK, Jarrod LP, Singh B, Ching NL. Molecular epidemiological investigation of Plasmodium knowlesi in humans and macaques in Singapore. Vector-Borne Zoo Dis. 2011;11(2):131–135. doi: 10.1089/vbz.2010.0024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ng OT, Ooi EE, Lee CC, Lee PJ, Ng LC, Pei SW, Tu TM, Loh JP, Leo YS. Naturally acquired human Plasmodium knowlesi infection, Singapore. Emerg Infect Dis. 2008;14(5):814. doi: 10.3201/eid1405.070863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jongwutiwes S, Putaporntip C, Iwasaki T, Sata T, Kanbara H. Naturally acquired Plasmodium knowlesi malaria in human, Thailand. Emerg Infect Dis. 2004;10:2211–2213. doi: 10.3201/eid1012.040293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jongwutiwes S, Buppan P, Kosuvin R, Seethamchai S, Pattanawong U, Sirichaisinthop J, Putaporntip C. Plasmodium knowlesi Malaria in Humans and Macaques, Thailand. Emerg Infect Dis. 2011;17(10):1799. doi: 10.3201/eid1710.110349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Putaporntip C, Hongsrimuang T, Seethamchai S, Kobasa T, Limkittikul K, Cui L, Jongwutiwes S. Differential prevalence of Plasmodium infections and cryptic Plasmodium knowlesi malaria in humans in Thailand. J Infect Dis. 2009;199(8):1143. doi: 10.1086/597414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sermwittayawong N, Singh B, Nishibuchi M, Sawangjaroen N, Vuddhakul V. Human Plasmodium knowlesi infection in Ranong province, southwestern border of Thailand. Malar J. 2012;11(1):36. doi: 10.1186/1475-2875-11-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jiang N, Chang Q, Sun X, Lu H, Yin J, Zhang Z, Wahlgren M, Chen Q. Co-infections with Plasmodium knowlesi and other malaria parasites, Myanmar. Emerg Infect Dis. 2010;16:1476–1478. doi: 10.3201/eid1609.100339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhu H, Li J, Zheng H. Human natural infection of Plasmodium knowlesi. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2006;24(1):70. [PubMed] [Google Scholar]
- 17.Luchavez J, Espino F, Curameng P, Espina R, Bell D, Chiodini P, Nolder D, Sutherland C, Lee KS, Singh B. Human infections with Plasmodium knowlesi, the Philippines. Emerg Infect Dis. 2008;14(5):811–813. doi: 10.3201/eid1405.071407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Figtree M, Lee R, Bain L, Kennedy T, Mackertich S, Urban M, Cheng Q, Hudson BJ. Plasmodium knowlesi in Human, Indonesian Borneo. Emerg Infect Dis. 2010;14(4):672–674. doi: 10.3201/eid1604.091624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sulistyaningsih E, Fitri LE, Löscher T, Berens-Riha N. Diagnostic difficulties with Plasmodium knowlesi infection in humans. Emerg Infect Dis. 2010;16(6):1033. doi: 10.3201/eid1606.100022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Eede P, Van H, Van Overmeir C, Vythilingam I, Duc T, Hung L, Manh H, Anné J, D’Alessandro U, Erhart A. Human Plasmodium knowlesi infections in young children in central Vietnam. Malar J. 2009;8(1):249. doi: 10.1186/1475-2875-8-249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Marchand RP, Culleton R, Maeno Y, Quang NT, Nakazawa S. Co-infections of Plasmodium knowlesi, P. falciparum, and P. vivax among humans and Anopheles dirus mosquitoes, southern Vietnam. Emerg Infect Dis. 2011;17(7):1232–1239. doi: 10.3201/eid1707.101551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Khim N, Siv S, Kim S, Mueller T, Fleischmann E, Singh B, Divis P, Steenkeste N, Duval L, Bouchier C. Plasmodium knowlesi infection in humans, Cambodia, 2007–2010. Emerg Infect Dis. 2011;17(10):1900. doi: 10.3201/eid1710.110355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.William T, Rahman HA, Jelip J, Ibrahim MY, Menon J, Grigg MJ, Yeo TW, Anstey NM, Barber BE. Increasing incidence of Plasmodium knowlesi malaria following control of P. falciparum and P. vivax Malaria in Sabah, Malaysia. PLoS Neg Trop Dis. 2013;7(1):e2026. doi: 10.1371/journal.pntd.0002026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yusof R, Lau YL, Mahmud R, Fong MY, Jelip J, Ngian HU, Mustakim S, Hussin HM, Marzuki N, Ali MM. High proportion of knowlesi malaria in recent malaria cases in Malaysia. Malar J. 2014;13(1):168. doi: 10.1186/1475-2875-13-168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vythilingam I, NoorAzian YM, Huat TC, Jiram AI, Yusri YM, Azahari AH, NorParina I, NoorRain A, LokmanHakim S. Plasmodium knowlesi in humans, macaques and mosquitoes in peninsular Malaysia. Parasit Vector. 2008;1:26. doi: 10.1186/1756-3305-1-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Reid JA. Anopheline Mosquitoes Of Malaya And Borneo. vol 31. Malaysia: Inst Medical Res Malaysia; 1968. [Google Scholar]
- 27.Chang MS, Doraisingam P, Hardin S, Nagum N. Malaria and filariasis transmission in a village/forest setting in Baram District, Sarawak, Malaysia. J Trop Med Hyg. 1995;98(3):192. [PubMed] [Google Scholar]
- 28.Hii JKL, Kan S, Pereria M, Parmar SS, Campos RL, Chan MKC. Transmission dynamics and estimates of malaria vectorial capacity for An. balabacensis and An. flavirostris on Bangii island, Sabah, Malaysia. Ann Tropl Med Parasitol. 1988;82:91–101. doi: 10.1080/00034983.1988.11812214. [DOI] [PubMed] [Google Scholar]
- 29.Vythilingam I, Chiang GL, Lee HL, Singh KI. Bionomics of important mosquito vectors in Malaysia. Southeast Asian J Trop Med Public Health. 1992;23(4):587–603. [PubMed] [Google Scholar]
- 30.Wharton RH, Eyles DE. Anopheles hackeri, a vector of Plasmodium knowlesi in Malaya. Science. 1961;134(3474):279. doi: 10.1126/science.134.3474.279. [DOI] [PubMed] [Google Scholar]
- 31.Cheong WH, Warren MW, Omar AH, Mahadevan S. Anopheles balabacensis balabacensis identified as vector of simian malaria in Malaysia. Science. 1965;150(3701):1314. doi: 10.1126/science.150.3701.1314. [DOI] [PubMed] [Google Scholar]
- 32.Vythilingam I, Tan CH, Asmad M, Chan ST, Lee KS, Singh B. Natural transmission of Plasmodium knowlesi to humans by Anopheles latens in Sarawak, Malaysia. Trans Roy Soc Trop Med Hyg. 2006;100(11):1087–1088. doi: 10.1016/j.trstmh.2006.02.006. [DOI] [PubMed] [Google Scholar]
- 33.Tan CH, Vythilingam I, Matusop A, Chan ST, Singh B. Bionomics of Anopheles latens in Kapit, Sarawak, Malaysian Borneo in relation to the transmission of zoonotic simian malaria parasite Plasmodium knowlesi. Malar J. 2008;7:52. doi: 10.1186/1475-2875-7-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jiram AI, Vythilingam I, NoorAzian YM, Yusof YM, Azahari AH, Fong MY. Entomologic investigation of Plasmodium knowlesi vectors in Kuala Lipis, Pahang, Malaysia. Malar J. 2012;11(1):213. doi: 10.1186/1475-2875-11-213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vythilingam I. Review Paper Plasmodium knowlesi in humans: a review on the role of its vectors in Malaysia. Trop Biomed. 2010;27(1):1–12. [PubMed] [Google Scholar]
- 36.Sanders KC, Rundi C, Jelip J, Rashman Y, Gueye CS, Gosling RD. Eliminating malaria in Malaysia: the role of partnerships between the public and commercial sectors in Sabah. Malar J. 2014;13(1):24. doi: 10.1186/1475-2875-13-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vythilingam I, Foo LC, Chiang GL, Chan ST, Eng KL, Mahadevan S, Mak JW, Singh KI. The impact of permethrin impregnated bednets on the malaria vector Anopheles maculatus (Diptera: Culicidae) in aboriginal villages of Pos Betau Pahang, Malaysia. Southeast Asian J Trop Med Public Health. 1995;26(2):354–358. [PubMed] [Google Scholar]
- 38.Sallum MAM, Peyton EL, Harrison BA, Wilkerson RC. Revision of the Leucosphyrus group of Anopheles (Cellia) (Diptera, Culicidae) Rev Bras de Entomolo. 2005;49:01–152. doi: 10.1590/S0085-56262005000500001. [DOI] [Google Scholar]
- 39.Chew CH, Lim YAL, Lee PC, Mahmud R, Chua KH. Hexaplex PCR detection system for identification of five human Plasmodium species with an internal control. J Clin Microbiol. 2012;50(12):4012–4019. doi: 10.1128/JCM.06454-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Walton C, Handley J, Kuvangkadilok C, Collins F, Harbach R, Baimai V, Butlin R. Identification of five species of the Anopheles dirus complex from Thailand, using allele‒specific polymerase chain reaction. Med Vet Entomol. 1999;13(1):24–32. doi: 10.1046/j.1365-2915.1999.00142.x. [DOI] [PubMed] [Google Scholar]
- 41.Sallum MAM, Peyton EL, Wilkerson RC. Six new species of the Anopheles leucosphyrus group, reinterpretation of An. elegans and vector implications. Med Vet Entomol. 2005;19(2):158–199. doi: 10.1111/j.0269-283X.2005.00551.x. [DOI] [PubMed] [Google Scholar]
- 42.Moore DA, Carpenter TE. Spatial analytical methods and geographic information systems: use in health research and epidemiology. Epidemiolo Rev. 1999;21(2):143–161. doi: 10.1093/oxfordjournals.epirev.a017993. [DOI] [PubMed] [Google Scholar]
- 43.Silverman BW. Density Estimation For Statistics And Data Analysis. vol. 26. London: CRC press; 1986. [Google Scholar]
- 44.Bithell J. An application of density estimation to geographical epidemiology. Stat Med. 1990;9(6):691–701. doi: 10.1002/sim.4780090616. [DOI] [PubMed] [Google Scholar]
- 45.Davidson G. Estimation of survival rate of anopheline mosquitoes in nature. Nature. 1954;174:792–793. doi: 10.1038/174792a0. [DOI] [PubMed] [Google Scholar]
- 46.Garrett-Jones C, Grab B. The assessment of insecticidal impact on the malaria mosquito’s vectorial capacity, from data on the proportion of parous females. Bull World Health Organ. 1964;31(1):71. [PMC free article] [PubMed] [Google Scholar]
- 47.Garret-Jones C, Shidrawi GR. Malaria vectorial capacity of a population of Anopheles gambiae, an exercise in epidemiological entomology. Bull World Health Organ. 1969;40:531–545. [PMC free article] [PubMed] [Google Scholar]
- 48.Rajahram GS, Barber BE, William T, Menon J, Anstey NM, Yeo TW. Deaths due to Plasmodium knowlesi malaria in Sabah, Malaysia: association with reporting as Plasmodium malariae and delayed parenteral artesunate. Malar J. 2012;11(1):284. doi: 10.1186/1475-2875-11-284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Collins WE. Plasmodium knowlesi: A malaria parasite of monkeys and humans. Ann Rev Entomol. 2012;57(1):107–121. doi: 10.1146/annurev-ento-121510-133540. [DOI] [PubMed] [Google Scholar]
- 50.Rosenberg R. Inability of plasmodium knowlesi sporozoites to invade Anopheles freeborni salivary glands. Am J Trop Med Hyg. 1985;34(4):687–691. doi: 10.4269/ajtmh.1985.34.687. [DOI] [PubMed] [Google Scholar]
- 51.Wharton RH, Eyles DE, Warren M, Cheong WH. Studies to determine the vectors of monkey malaria in Malaya. Ann Trop Med Parasitol. 1964;58:56. doi: 10.1080/00034983.1964.11686215. [DOI] [PubMed] [Google Scholar]
- 52.Warren MW, Wharton RH. The vectors of simian malaria: identity, biology, and geographical distribution. J Parasitol. 1963;49:892–904. doi: 10.2307/3275715. [DOI] [PubMed] [Google Scholar]
- 53.Braima KA, Sum J-S, Ghazali A-RM, Muslimin M, Jeffery J, Lee W-C, Shaker MR, Elamin A-EM, Jamaiah I, Lau Y-L. Is there a risk of suburban transmission of malaria in Selangor, Malaysia? PLoS One. 2013;8(10):e77924. doi: 10.1371/journal.pone.0077924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sinka ME, Bangs MJ, Manguin S, Chareonviriyaphap T, Patil AP, Temperley WH, Gething PW, Elyazar IRF, Kabaria CW, Harbach RE. The dominant Anopheles vectors of human malaria in the Asia-Pacific region: occurrence data, distribution maps and bionomic précis. Parasite Vectors. 2011;4(1):1–46. doi: 10.1186/1756-3305-4-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Singh B, Daneshvar C. Human infections and detection of Plasmodium knowlesi. Clin Microbiol Rev. 2013;26(2):165–184. doi: 10.1128/CMR.00079-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Moyes CL, Henry AJ, Golding N, Huang Z, Singh B, Baird JK, Newton PN, Huffman M, Duda KA, Drakeley CJ. Defining the Geographical Range of the Plasmodium knowlesi Reservoir. PLoS Neg Trop Dis. 2014;8(3):e2780. doi: 10.1371/journal.pntd.0002780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ta TH, Hisam S, Lanza M, Jiram AI, Ismail N, Rubio JM. First case of a naturally acquired human infection with Plasmodium cynomolgi. Malar J. 2014;13(1):68. doi: 10.1186/1475-2875-13-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Aregawi M, Cibulskis RE, Otten M, Williams R. World Malaria Report 2009. Geneva: World Health Organ; 2009. [Google Scholar]
- 59.Erhart A, Thang ND, Hung NQ, Toi V, Hung X, Tuy TQ, Cong D, Speybroeck N, Coosemans M, D’Alessandro U. Forest malaria in Vietnam: a challenge for control. Am J Trop Med Hyg. 2004;70(2):110–118. [PubMed] [Google Scholar]
- 60.Oo TT, Storch V, Becker N. Anopheles dirus and its role in malaria transmission in Myanmar. J Vector Ecol. 2003;28:175–183. [PubMed] [Google Scholar]
- 61.Rosenberg R, Maheswary N. Forest malaria in Bangladesh. II. Transmission by Anopheles dirus. Am J Trop Med Hyg. 1982;31(2):183–191. doi: 10.4269/ajtmh.1982.31.183. [DOI] [PubMed] [Google Scholar]
- 62.Obsomer V, Defourny P, Coosemans M. The Anopheles dirus complex: spatial distribution and environmental drivers. Malar J. 2007;6(1):26. doi: 10.1186/1475-2875-6-26. [DOI] [PMC free article] [PubMed] [Google Scholar]


