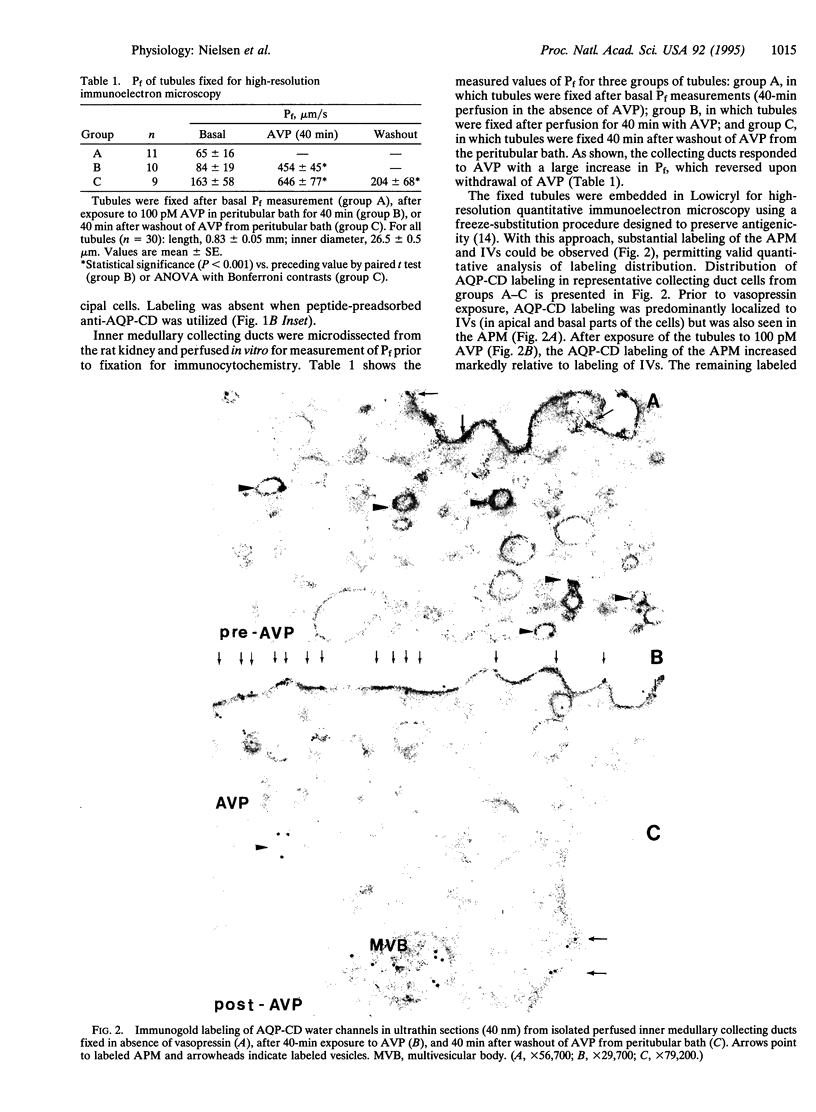
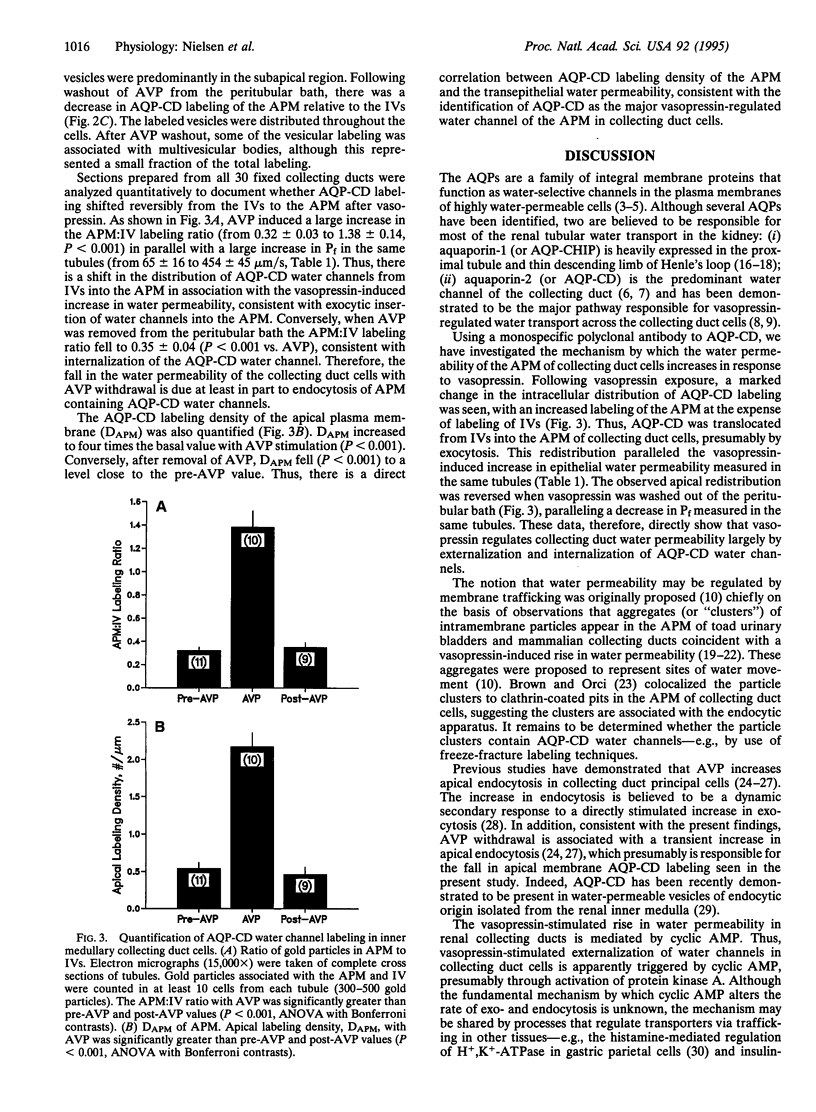
Abstract
Water excretion by the kidney is regulated by the peptide hormone vasopressin. Vasopressin increases the water permeability of the renal collecting duct cells, allowing more water to be reabsorbed from collecting duct urine to blood. Despite long-standing interest in this process, the mechanism of the water permeability increase has remained undetermined. Recently, a molecular water channel (AQP-CD) has been cloned whose expression appears to be limited to the collecting duct. Previously, we immunolocalized this water channel to the apical plasma membrane (APM) and to intracellular vesicles (IVs) of collecting duct cells. Here, we test the hypothesis that vasopressin increases cellular water permeability by inducing exocytosis of AQP-CD-laden vesicles, transferring water channels from IVs to APM. Rat collecting ducts were perfused in vitro to determine water permeability and subcellular distribution of AQP-CD in the same tubules. The collecting ducts were fixed for immunoelectron microscopy before, during, and after exposure to vasopressin. Vasopressin exposure induced increases in water permeability and the absolute labeling density of AQP-CD in the APM. In parallel, the APM:IV labeling ratio increased. Furthermore, in response to vasopressin withdrawal, AQP-CD labeling density in the APM and the APM:IV labeling ratio decreased in parallel to a measured decrease in osmotic water permeability. We conclude that vasopressin increases the water permeability of collecting duct cells by inducing a reversible translocation of AQP-CD water channels from IVs to the APM.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Agre P., Preston G. M., Smith B. L., Jung J. S., Raina S., Moon C., Guggino W. B., Nielsen S. Aquaporin CHIP: the archetypal molecular water channel. Am J Physiol. 1993 Oct;265(4 Pt 2):F463–F476. doi: 10.1152/ajprenal.1993.265.4.F463. [DOI] [PubMed] [Google Scholar]
- Agre P., Sasaki S., Chrispeels M. J. Aquaporins: a family of water channel proteins. Am J Physiol. 1993 Sep;265(3 Pt 2):F461–F461. doi: 10.1152/ajprenal.1993.265.3.F461. [DOI] [PubMed] [Google Scholar]
- Brown D., Orci L. Vasopressin stimulates formation of coated pits in rat kidney collecting ducts. Nature. 1983 Mar 17;302(5905):253–255. doi: 10.1038/302253a0. [DOI] [PubMed] [Google Scholar]
- Brown D., Weyer P., Orci L. Vasopressin stimulates endocytosis in kidney collecting duct principal cells. Eur J Cell Biol. 1988 Jun;46(2):336–341. [PubMed] [Google Scholar]
- Burg M. B. Perfusion of isolated renal tubules. Yale J Biol Med. 1972 Jun-Aug;45(3-4):321–326. [PMC free article] [PubMed] [Google Scholar]
- Deen P. M., Verdijk M. A., Knoers N. V., Wieringa B., Monnens L. A., van Os C. H., van Oost B. A. Requirement of human renal water channel aquaporin-2 for vasopressin-dependent concentration of urine. Science. 1994 Apr 1;264(5155):92–95. doi: 10.1126/science.8140421. [DOI] [PubMed] [Google Scholar]
- Denker B. M., Smith B. L., Kuhajda F. P., Agre P. Identification, purification, and partial characterization of a novel Mr 28,000 integral membrane protein from erythrocytes and renal tubules. J Biol Chem. 1988 Oct 25;263(30):15634–15642. [PubMed] [Google Scholar]
- DiGiovanni S. R., Nielsen S., Christensen E. I., Knepper M. A. Regulation of collecting duct water channel expression by vasopressin in Brattleboro rat. Proc Natl Acad Sci U S A. 1994 Sep 13;91(19):8984–8988. doi: 10.1073/pnas.91.19.8984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flamion B., Spring K. R. Water permeability of apical and basolateral cell membranes of rat inner medullary collecting duct. Am J Physiol. 1990 Dec;259(6 Pt 2):F986–F999. doi: 10.1152/ajprenal.1990.259.6.F986. [DOI] [PubMed] [Google Scholar]
- Forte J. G., Hanzel D. K., Urushidani T., Wolosin J. M. Pumps and pathways for gastric HCl secretion. Ann N Y Acad Sci. 1989;574:145–158. doi: 10.1111/j.1749-6632.1989.tb25153.x. [DOI] [PubMed] [Google Scholar]
- Fushimi K., Uchida S., Hara Y., Hirata Y., Marumo F., Sasaki S. Cloning and expression of apical membrane water channel of rat kidney collecting tubule. Nature. 1993 Feb 11;361(6412):549–552. doi: 10.1038/361549a0. [DOI] [PubMed] [Google Scholar]
- Harmanci M. C., Stern P., Kachadorian W. A., Valtin H., DiScala V. A. Vasopressin and collecting duct intramembranous particle clusters: a dose-response relationship. Am J Physiol. 1980 Dec;239(6):F560–F564. doi: 10.1152/ajprenal.1980.239.6.F560. [DOI] [PubMed] [Google Scholar]
- Harris H. W., Jr, Zeidel M. L., Jo I., Hammond T. G. Characterization of purified endosomes containing the antidiuretic hormone-sensitive water channel from rat renal papilla. J Biol Chem. 1994 Apr 22;269(16):11993–12000. [PubMed] [Google Scholar]
- Kachadorian W. A., Levine S. D., Wade J. B., Di Scala V. A., Hays R. M. Relationship of aggregated intramembranous particles to water permeability in vasopressin-treated toad urinary bladder. J Clin Invest. 1977 Mar;59(3):576–581. doi: 10.1172/JCI108673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kachadorian W. A., Wade J. B., DiScala V. A. Vasopressin: induced structural change in toad bladder luminal membrane. Science. 1975 Oct 3;190(4209):67–69. doi: 10.1126/science.809840. [DOI] [PubMed] [Google Scholar]
- Knepper M. A. The aquaporin family of molecular water channels. Proc Natl Acad Sci U S A. 1994 Jul 5;91(14):6255–6258. doi: 10.1073/pnas.91.14.6255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lencer W. I., Brown D., Ausiello D. A., Verkman A. S. Endocytosis of water channels in rat kidney: cell specificity and correlation with in vivo antidiuresis. Am J Physiol. 1990 Dec;259(6 Pt 1):C920–C932. doi: 10.1152/ajpcell.1990.259.6.C920. [DOI] [PubMed] [Google Scholar]
- Lolait S. J., O'Carroll A. M., McBride O. W., Konig M., Morel A., Brownstein M. J. Cloning and characterization of a vasopressin V2 receptor and possible link to nephrogenic diabetes insipidus. Nature. 1992 May 28;357(6376):336–339. doi: 10.1038/357336a0. [DOI] [PubMed] [Google Scholar]
- Nielsen S., DiGiovanni S. R., Christensen E. I., Knepper M. A., Harris H. W. Cellular and subcellular immunolocalization of vasopressin-regulated water channel in rat kidney. Proc Natl Acad Sci U S A. 1993 Dec 15;90(24):11663–11667. doi: 10.1073/pnas.90.24.11663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen S., Knepper M. A. Vasopressin activates collecting duct urea transporters and water channels by distinct physical processes. Am J Physiol. 1993 Aug;265(2 Pt 2):F204–F213. doi: 10.1152/ajprenal.1993.265.2.F204. [DOI] [PubMed] [Google Scholar]
- Nielsen S., Muller J., Knepper M. A. Vasopressin- and cAMP-induced changes in ultrastructure of isolated perfused inner medullary collecting ducts. Am J Physiol. 1993 Aug;265(2 Pt 2):F225–F238. doi: 10.1152/ajprenal.1993.265.2.F225. [DOI] [PubMed] [Google Scholar]
- Nielsen S., Smith B. L., Christensen E. I., Knepper M. A., Agre P. CHIP28 water channels are localized in constitutively water-permeable segments of the nephron. J Cell Biol. 1993 Jan;120(2):371–383. doi: 10.1083/jcb.120.2.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenthal W., Seibold A., Antaramian A., Lonergan M., Arthus M. F., Hendy G. N., Birnbaumer M., Bichet D. G. Molecular identification of the gene responsible for congenital nephrogenic diabetes insipidus. Nature. 1992 Sep 17;359(6392):233–235. doi: 10.1038/359233a0. [DOI] [PubMed] [Google Scholar]
- Sabolić I., Valenti G., Verbavatz J. M., Van Hoek A. N., Verkman A. S., Ausiello D. A., Brown D. Localization of the CHIP28 water channel in rat kidney. Am J Physiol. 1992 Dec;263(6 Pt 1):C1225–C1233. doi: 10.1152/ajpcell.1992.263.6.C1225. [DOI] [PubMed] [Google Scholar]
- Sasaki S., Fushimi K., Saito H., Saito F., Uchida S., Ishibashi K., Kuwahara M., Ikeuchi T., Inui K., Nakajima K. Cloning, characterization, and chromosomal mapping of human aquaporin of collecting duct. J Clin Invest. 1994 Mar;93(3):1250–1256. doi: 10.1172/JCI117079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slot J. W., Geuze H. J., Gigengack S., Lienhard G. E., James D. E. Immuno-localization of the insulin regulatable glucose transporter in brown adipose tissue of the rat. J Cell Biol. 1991 Apr;113(1):123–135. doi: 10.1083/jcb.113.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strange K., Spring K. R. Cell membrane water permeability of rabbit cortical collecting duct. J Membr Biol. 1987;96(1):27–43. doi: 10.1007/BF01869332. [DOI] [PubMed] [Google Scholar]
- Strange K., Willingham M. C., Handler J. S., Harris H. W., Jr Apical membrane endocytosis via coated pits is stimulated by removal of antidiuretic hormone from isolated, perfused rabbit cortical collecting tubule. J Membr Biol. 1988 Jul;103(1):17–28. doi: 10.1007/BF01871929. [DOI] [PubMed] [Google Scholar]
- Wade J. B., Stetson D. L., Lewis S. A. ADH action: evidence for a membrane shuttle mechanism. Ann N Y Acad Sci. 1981;372:106–117. doi: 10.1111/j.1749-6632.1981.tb15464.x. [DOI] [PubMed] [Google Scholar]
- Wall S. M., Han J. S., Chou C. L., Knepper M. A. Kinetics of urea and water permeability activation by vasopressin in rat terminal IMCD. Am J Physiol. 1992 Jun;262(6 Pt 2):F989–F998. doi: 10.1152/ajprenal.1992.262.6.F989. [DOI] [PubMed] [Google Scholar]