Bipolar disorder (BP) is a progressive, life-threatening condition characterized by alternating episodes of severe depression and mania, ranked by the WHO as among the leading causes of lifetime disability. The heritability of BP is estimated at 85–95%, and genetic susceptibility loci, each with small effects, are emerging. Accumulating evidence suggests a developmental origin for BP: neuroanatomical abnormalities are often present at the first episode; there are organizational and neuronal migration alterations, with minimal astrogliosis. In addition, BP is typically diagnosed at adolescence when there is a shift from relying on earlier-developing brain regions to later-maturing prefrontal structures—a period when the brain may be particularly vulnerable to pre-existing neuropathologies (Strakowski, 2012). Since subtle changes in differentiation can produce neurological consequences that only become apparent much later in life, a developmental model to study BP is required.
The ability to reprogram adult somatic cells to induced pluripotent stem cells (iPSC) by expressing four transcription factors found in the blastocyst inner cell mass and embryonic stem cells (Takahashi et al, 2007) now makes it possible to derive BP patient-specific stem cells. iPSC are well suited to identify alterations in cell behavior, examine gene expression, and identify novel signaling pathways in the affected cell type(s). In BP, they provide a robust source of cells from individuals with longitudinal observations, individuals on or off medications, and patients who have comorbid diagnoses, multiple episodes and treatments, unlike embryonic stem cells. iPSC have been derived for other neuropsychiatric disorders (e.g., Brennand et al, 2011), and are now being reported for BP.
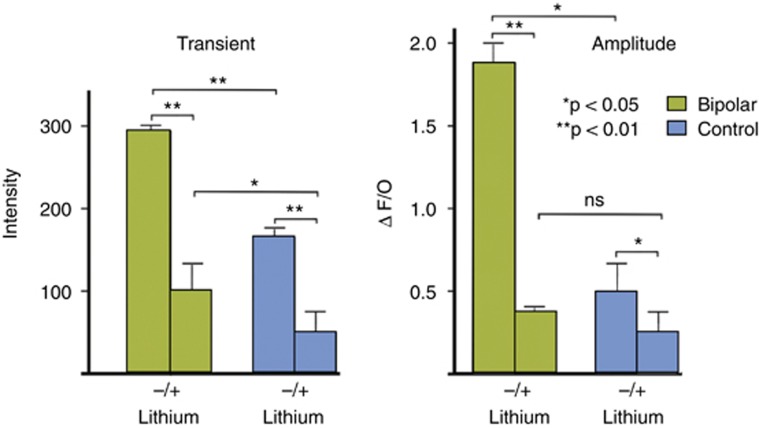
We have derived iPSC lines from six BPI patients and six healthy control (C) individuals and differentiated them into neurons to study their morphology, signaling characteristics, and transcriptome. We have shown that calcium signaling is altered in BP neurons (Chen et al, 2014). Importantly, these cells can be used to examine responsiveness to pharmacological interventions, as lithium pre-treatment reverted wave amplitudes to control levels (Figure 1).
Figure 1.
Changes in Flo-4AM fluorescence intensity and wave amplitude in control and bipolar neurons+Li.
These studies also indicated that BP neurons expressed transcripts characteristic of early ventral CNS fate: NKX2-2, FOXP2, ASCL1, LHX6, while control neurons expressed genes associated with dorsal telencephalic patterning: EMX2, FEZF2, PAX6, TBR2, TCF3, VGLUT1. Consistent with these results, prior to differentiation, control iPSC expressed significantly higher levels of LEFTY1,2 (which inhibit Nodal signaling) and of transcripts that regulate Hedgehog signaling, HHIP, and KIF7 (Cheung et al, 2009), which would promote dorsal telencephalic fate. Comparison of BP and C neurons also identified alterations in key components of the microRNA processing pathway, DICER and DROSHA, and of the mTOR pathway, RICTOR.
iPSC offer the first opportunity to study viable patient-derived neurons with the goal of understanding the molecular mechanisms underlying BP. We hypothesize that in BP neuronal cell fate determination is altered, increasing susceptibility to later epigenetic modifications. While iPSC provide important opportunities to model BP, much work remains to be carried out in larger family studies, to determine when during differentiation alterations may occur, and the cell types involved. This approach is beginning to identify novel, targetable signaling pathways in BP, and should provide the opportunity to identify and test effective medications for individual patients.
FUNDING AND DISCLOSURE
Dr O'Shea serves as a consultant for Genentech, Inc. Dr McInnis declares no conflict of interest.
Acknowledgments
This work is supported by the HC Prechter Bipolar Research Fund and Steven Schwartzberg Memorial Fund at the University of Michigan.
References
- Brennand KJ, Simone A, Jou J, Gelboin-Burkhart C, Tran N, Sangar S et al (2011). Modelling schizophrenia using human induced pluripotent stem cells. Nature 473: 221–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen HM, DeLong CJ, Bame M, Rajapakse I, Herron TJ, McInnis MG et al (2014). Transcripts involved in calcium signaling and telencephalic neuronal fate are altered in induced pluripotent stem cells from bipolar disorder patients. Transl Psychiatry 4: e375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheung HO-L, Zhang X, Ribeiro A, Mo R, Makino S, Puvlindan V et al (2009). The kinesin protein Kif7 is a critical regulator of Gli transcription factors in mammalian hedgehog signaling. Sci Signal 2: ra29. [DOI] [PubMed] [Google Scholar]
- Strakowski SM (2012). A neurophysiological model of bipolar disorder. In: Strakowski SM (ed). The Bipolar Brain. Integrating Neuroimaging and Genetics. Oxford University Press: New York. pp 253–274. [Google Scholar]
- Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K et al (2007). Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131: 861–872. [DOI] [PubMed] [Google Scholar]