Abstract
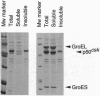
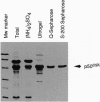
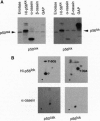
An Escherichia coli expression system overproducing the bacterial chaperones GroES and GroEL was engineered and has been successfully used to produce large quantities of the recombinant human protein-tyrosine kinase p50csk. The co-overproduction of the two chaperones with p50csk results in increased solubility of the kinase and allows purification of milligram amounts of active enzyme. Analysis of the purified protein by SDS/polyacrylamide gel electrophoresis reveals a single band with an apparent molecular mass of 50 kDa, indicating that recombinant human p50csk has been purified to near homogeneity. The purified enzyme displays tyrosine kinase activity as measured by both autophosphorylation and phosphorylation of exogenous substrates. Biochemical properties, including in vitro substrate specificity and enzymatic characteristics of the enzyme, have been assessed and compared with those of members of the Src family of protein-tyrosine kinases. Results indicate that p50csk and p56lck have different substrate specificities and that p50csk and p60c-src have similar kinetic parameters. The successful production and purification of an enzymatically active form of p50csk will enable further characterization of this important kinase and allow clarification of its physiological role. In addition, the results suggest that the approach described may be generally applicable to improve the solubility of recombinant proteins which otherwise are produced in an insoluble form in E. coli.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amrein K. E., Flint N., Panholzer B., Burn P. Ras GTPase-activating protein: a substrate and a potential binding protein of the protein-tyrosine kinase p56lck. Proc Natl Acad Sci U S A. 1992 Apr 15;89(8):3343–3346. doi: 10.1073/pnas.89.8.3343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergman M., Mustelin T., Oetken C., Partanen J., Flint N. A., Amrein K. E., Autero M., Burn P., Alitalo K. The human p50csk tyrosine kinase phosphorylates p56lck at Tyr-505 and down regulates its catalytic activity. EMBO J. 1992 Aug;11(8):2919–2924. doi: 10.1002/j.1460-2075.1992.tb05361.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blum P., Velligan M., Lin N., Matin A. DnaK-mediated alterations in human growth hormone protein inclusion bodies. Biotechnology (N Y) 1992 Mar;10(3):301–304. doi: 10.1038/nbt0392-301. [DOI] [PubMed] [Google Scholar]
- Bougeret C., Rothhut B., Jullien P., Fischer S., Benarous R. Recombinant Csk expressed in Escherichia coli is autophosphorylated on tyrosine residue(s). Oncogene. 1993 May;8(5):1241–1247. [PubMed] [Google Scholar]
- Caspers P., Stieger M., Burn P. Overproduction of bacterial chaperones improves the solubility of recombinant protein tyrosine kinases in Escherichia coli. Cell Mol Biol (Noisy-le-grand) 1994 Jul;40(5):635–644. [PubMed] [Google Scholar]
- Cooper J. A., Esch F. S., Taylor S. S., Hunter T. Phosphorylation sites in enolase and lactate dehydrogenase utilized by tyrosine protein kinases in vivo and in vitro. J Biol Chem. 1984 Jun 25;259(12):7835–7841. [PubMed] [Google Scholar]
- Craig E. A., Gambill B. D., Nelson R. J. Heat shock proteins: molecular chaperones of protein biogenesis. Microbiol Rev. 1993 Jun;57(2):402–414. doi: 10.1128/mr.57.2.402-414.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dale G. E., Broger C., Langen H., D'Arcy A., Stüber D. Improving protein solubility through rationally designed amino acid replacements: solubilization of the trimethoprim-resistant type S1 dihydrofolate reductase. Protein Eng. 1994 Jul;7(7):933–939. doi: 10.1093/protein/7.7.933. [DOI] [PubMed] [Google Scholar]
- Ellis J. Proteins as molecular chaperones. 1987 Jul 30-Aug 5Nature. 328(6129):378–379. doi: 10.1038/328378a0. [DOI] [PubMed] [Google Scholar]
- Feder D., Bishop J. M. Purification and enzymatic characterization of pp60c-src from human platelets. J Biol Chem. 1990 May 15;265(14):8205–8211. [PubMed] [Google Scholar]
- Flint N. A., Amrein K. E., Jascur T., Burn P. Purification and characterization of an activated form of the protein tyrosine kinase Lck from an Escherichia coli expression system. J Cell Biochem. 1994 Jul;55(3):389–397. doi: 10.1002/jcb.240550317. [DOI] [PubMed] [Google Scholar]
- Gething M. J., Sambrook J. Protein folding in the cell. Nature. 1992 Jan 2;355(6355):33–45. doi: 10.1038/355033a0. [DOI] [PubMed] [Google Scholar]
- Gilmer T. M., Erikson R. L. Rous sarcoma virus transforming protein, p60src, expressed in E. coli, functions as a protein kinase. Nature. 1981 Dec 24;294(5843):771–773. doi: 10.1038/294771a0. [DOI] [PubMed] [Google Scholar]
- Hartl F. U., Martin J., Neupert W. Protein folding in the cell: the role of molecular chaperones Hsp70 and Hsp60. Annu Rev Biophys Biomol Struct. 1992;21:293–322. doi: 10.1146/annurev.bb.21.060192.001453. [DOI] [PubMed] [Google Scholar]
- Hendrick J. P., Hartl F. U. Molecular chaperone functions of heat-shock proteins. Annu Rev Biochem. 1993;62:349–384. doi: 10.1146/annurev.bi.62.070193.002025. [DOI] [PubMed] [Google Scholar]
- Imamoto A., Soriano P. Disruption of the csk gene, encoding a negative regulator of Src family tyrosine kinases, leads to neural tube defects and embryonic lethality in mice. Cell. 1993 Jun 18;73(6):1117–1124. doi: 10.1016/0092-8674(93)90641-3. [DOI] [PubMed] [Google Scholar]
- Lindberg R. A., Pasquale E. B. Isolation of cDNA clones that encode active protein-tyrosine kinases using antibodies against phosphotyrosine. Methods Enzymol. 1991;200:557–564. doi: 10.1016/0076-6879(91)00171-r. [DOI] [PubMed] [Google Scholar]
- McGrath J. P., Levinson A. D. Bacterial expression of an enzymatically active protein encoded by RSV src gene. Nature. 1982 Feb 4;295(5848):423–425. doi: 10.1038/295423a0. [DOI] [PubMed] [Google Scholar]
- Mendoza J. A., Lorimer G. H., Horowitz P. M. Chaperonin cpn60 from Escherichia coli protects the mitochondrial enzyme rhodanese against heat inactivation and supports folding at elevated temperatures. J Biol Chem. 1992 Sep 5;267(25):17631–17634. [PubMed] [Google Scholar]
- Mizobata T., Akiyama Y., Ito K., Yumoto N., Kawata Y. Effects of the chaperonin GroE on the refolding of tryptophanase from Escherichia coli. Refolding is enhanced in the presence of ADP. J Biol Chem. 1992 Sep 5;267(25):17773–17779. [PubMed] [Google Scholar]
- Mustelin T., Burn P. Regulation of src family tyrosine kinases in lymphocytes. Trends Biochem Sci. 1993 Jun;18(6):215–220. doi: 10.1016/0968-0004(93)90192-p. [DOI] [PubMed] [Google Scholar]
- Nada S., Okada M., MacAuley A., Cooper J. A., Nakagawa H. Cloning of a complementary DNA for a protein-tyrosine kinase that specifically phosphorylates a negative regulatory site of p60c-src. Nature. 1991 May 2;351(6321):69–72. doi: 10.1038/351069a0. [DOI] [PubMed] [Google Scholar]
- Nada S., Yagi T., Takeda H., Tokunaga T., Nakagawa H., Ikawa Y., Okada M., Aizawa S. Constitutive activation of Src family kinases in mouse embryos that lack Csk. Cell. 1993 Jun 18;73(6):1125–1135. doi: 10.1016/0092-8674(93)90642-4. [DOI] [PubMed] [Google Scholar]
- Okada M., Nada S., Yamanashi Y., Yamamoto T., Nakagawa H. CSK: a protein-tyrosine kinase involved in regulation of src family kinases. J Biol Chem. 1991 Dec 25;266(36):24249–24252. [PubMed] [Google Scholar]
- Okada M., Nakagawa H. A protein tyrosine kinase involved in regulation of pp60c-src function. J Biol Chem. 1989 Dec 15;264(35):20886–20893. [PubMed] [Google Scholar]
- Partanen J., Armstrong E., Bergman M., Mäkelä T. P., Hirvonen H., Huebner K., Alitalo K. cyl encodes a putative cytoplasmic tyrosine kinase lacking the conserved tyrosine autophosphorylation site (Y416src). Oncogene. 1991 Nov;6(11):2013–2018. [PubMed] [Google Scholar]
- Piwnica-Worms H., Williams N. G., Cheng S. H., Roberts T. M. Regulation of pp60c-src and its interaction with polyomavirus middle T antigen in insect cells. J Virol. 1990 Jan;64(1):61–68. doi: 10.1128/jvi.64.1.61-68.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramer S. E., Winkler D. G., Carrera A., Roberts T. M., Walsh C. T. Purification and initial characterization of the lymphoid-cell protein-tyrosine kinase p56lck from a baculovirus expression system. Proc Natl Acad Sci U S A. 1991 Jul 15;88(14):6254–6258. doi: 10.1073/pnas.88.14.6254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reuter C., Findik D., Presek P. Characterization of purified pp60c-src protein tyrosine kinase from human platelets. Eur J Biochem. 1990 Jun 20;190(2):343–350. doi: 10.1111/j.1432-1033.1990.tb15581.x. [DOI] [PubMed] [Google Scholar]
- Sabe H., Knudsen B., Okada M., Nada S., Nakagawa H., Hanafusa H. Molecular cloning and expression of chicken C-terminal Src kinase: lack of stable association with c-Src protein. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2190–2194. doi: 10.1073/pnas.89.6.2190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schröder H., Langer T., Hartl F. U., Bukau B. DnaK, DnaJ and GrpE form a cellular chaperone machinery capable of repairing heat-induced protein damage. EMBO J. 1993 Nov;12(11):4137–4144. doi: 10.1002/j.1460-2075.1993.tb06097.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Superti-Furga G., Fumagalli S., Koegl M., Courtneidge S. A., Draetta G. Csk inhibition of c-Src activity requires both the SH2 and SH3 domains of Src. EMBO J. 1993 Jul;12(7):2625–2634. doi: 10.1002/j.1460-2075.1993.tb05923.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watts J. D., Wilson G. M., Ettenhadieh E., Clark-Lewis I., Kubanek C. A., Astell C. R., Marth J. D., Aebersold R. Purification and initial characterization of the lymphocyte-specific protein-tyrosyl kinase p56lck from a baculovirus expression system. J Biol Chem. 1992 Jan 15;267(2):901–907. [PubMed] [Google Scholar]