Abstract
Purpose
The study was to estimate the likelihood of axillary downstaging and to identify the factors predicting a pathologically node negative status after neoadjuvant chemotherapy (NAC) with or without trastuzumab in HER2-positive breast cancer.
Methods
Patients with HER2-positive, stage IIa-IIIc breast cancer were enrolled. Axillary status was evaluated by palpation and fine needle aspiration (FNA) before NAC. All patients received 4–6 cycles of PCrb (paclitaxel 80 mg/m2 and carboplatin AUC = 2 d1, 8, and 15 of a 28-day cycle, or paclitaxel 175 mg/m2 and carboplatin AUC = 6 every-3-week) and were non-randomly administered trastuzumab (2 mg/kg weekly or 6 mg/kg every-3-week) or not. After NAC, each patient underwent standard axillary lymph node dissection and breast-conserving surgery or mastectomy. And some patients received sentinel lymph node biopsy (SLNB) before axillary dissection.
Results
Between November-2007 and June-2013, 255 patients were enrolled. Of them, 157 were confirmed as axillary node positive by FNA (group-A) and 98 as axillary node negative either by FNA or impalpable (group-B). After axillary dissection, the overall pathologically node negative rates (pNNR) were 52.9% in group-A and 69.4% in group-B. The ER-poor/HER2-positive subtype acquired the highest pNNR (79.6% in group-A and 87.9% in group-B, respectively) and the lowest rate of residual with ≥4 nodes involvement (1.9% and 3%, respectively) after PCrb plus trastuzumab. In multivariate analysis, trastuzumab added and ER-poor status were independent factors in predicting a higher pNNR in HER2-positive breast cancer. Forty-six tested patients showed that the ER-poor/HER2-positive subtype acquired a considerable high pNNR and axillary status with SLNB was well macthed with the axillary dissection.
Conclusions
ER-poor/HER2-positive subtype of breast cancer is a potential candidate for undergoing sentinel lymph node biopsy instead of regional node dissection for accurate axillary evaluation after effective downstaging by neoadjuvant chemo-trastuzumab therapy.
Introduction
Although sentinel lymph node biopsy (SLNB) is a good substitute for axillary dissection in some situations, axillary dissection currently remains the standard of care for axillary node positive operable breast cancer [1]. However, axillary dissection would significantly increase the risk of severe morbidity of the ipsilateral arm, such as lymphatic edema, pain and dyskinesia [2], [3].
Neoadjuvant chemotherapy (NAC) has become a standard treatment for locally advanced breast cancer to downstage tumors, which aids in surgical resection [4]. Additionally, downsizing the primary tumor can make breast-conservative surgery (BCS) possible for some patients who were initial candidates for mastectomy and therefore improve cosmetic outcomes [5], [6]. Furthermore, patients who achieved pathologic complete remission (pCR) after NAC would acquire better long-term survival than those who did not. The pCR occurs not only in the primary tumor but also in the involved axillary nodes [7]. Therefore, axillary dissection might be avoided or replaced by a more conservative procedure in a certain subtype of node positive breast cancer patients after NAC.
Amplification of human epidermal growth factor receptor-2 (HER2) is identified in approximately 20–25% of all human breast cancers [8]. Many studies have reported that breast cancers with HER2-positive can achieve a high pCR rate after NAC, especially using regimens including trastuzumab [9], [10]. However, the data about predicting axilla downstaging and pathologically node negative status after NAC are rare. The aim of our current study was to identify the likelihood of axillary downstaging and to seek the factors that may predict a pathologically node negative status after NAC in HER2-positive breast cancer.
Patients and Methods
Patients Information
A retrospective analysis was carried out among patients from the phase II trial evaluating the activity and safety of a weekly paclitaxel plus carboplatin (PCrb) regimen as NAC in women with locally advanced breast cancer (ClinicalTrials.gov NCT01203267) [11] and the two-arm randomized phase II trial comparing the efficacy and safety between weekly scheduled PCrb with weekly herceptine and once-every-3-week schedule in women with HER2-positive aggressive breast cancer (ClinicalTrials.gov NCT01170143) [12]. Untreated patients at ages of 18–80 years old with primary histologically confirmed, clinical stage IIa-IIIc, HER2-positive, invasive breast cancer were included. Patients with distant metastasis or prior history of malignancy were excluded. Up to June 2013, 255 patients were included for main analysis. And another 46 newly diagnosed patients were enrolled to identify and confirm the previous results. They were all with Eastern Cooperative Oncology Group performance status ≤1 and left ventricular ejection fraction>55% by multiple gated acquisition scan or echocardiography. Besides, they all had adequate hematopoietic function (absolute neutrophil count ≥1.5*109/L, platelet count ≥100*109/L, and hemoglobin level ≥100 g/L) and appreciate hepatic and renal function (bilirubin level, aspartate aminotransferase, and alanine aminotransferase <1.5 times the normal upper limit and serum creatinine <110 umol/L). Women of childbearing potential were required to have a negative pregnancy test and to agree to take adequate contraceptive measures.Immunohistochemical (IHC) assessment of estrogen receptor (ER), progesterone receptor (PR) and HER2 expression were conducted in paraffin-embedded tumor samples biopsied before neoadjuvant treatment according to the guidelines from the American Society of Clinical Oncology and the College of American Pathologists. The intensity was graded as 0 (poor), 1 (weak), 2 (moderate) or 3 (strong). The abundance of positive cells was graded as 0, <10% positive cells; 1, 10–30%; 2, 30–50%; 3,>50%; the scoring system of IHC for ER and PR is automated. All of the immunostained slides were analyzed independently by two pathologists without any knowledge of the clinicopathological features. HER2 positivity was determined by IHC 3 (HerceptTest; Dako, Glostrup, Copenhagen, Denmark, http://www.dako.com) or fluorescence in situ hybridization (FISH) positive status (PathVysion HER-2 DNA probekit; Vysis Inc., Downers Grove, IL) [13]–[15].
This study was approved by the Ethical Committee of Shanghai Cancer Center, Fudan University. Every patient signed a written informed consent.
Procedures
We sorted patients to two study groups for both 255 and 46 patients according to the baseline axillary nodal status, determined by both palpation and fine-needle aspiration (FNA), before NAC (Fig. 1). Group-A and C included patients with an axillary node positive status confirmed by FNA (cN+FNA+). Group-B and D included patients with clinically enlarged axillary node(s) that were negative by FNA (cN+/FNA-) or patients with clinically impalpable node (cN-). We classified tumor deposits according to the Union for International Cancer Control TNM classification system [16].
Figure 1. The retrospective study and testing group designs.
HER2 = human epideminal growth factor receptor 2; cN+ = clinically axillary node positive; cN- = clinically axillary node negative; FNA = fine needle aspiration; P = paclitaxel; Crb = carboplatin; SLNB = sentinel lymph node biopsy. Some patients received SLNB before axillary dissection in the testing group for exploring the accuracy of SLNB after neoadjuvant therapy.
For main analysis, all patients were non-randomly administered PCrbH (weekly or every-3-week) or PCrb (weekly) for 4–6 cycles. In weekly schedule, patients received an 80 mg/m2 dose of paclitaxel (P) followed by carboplatin (Crb) dosed at an area under the concentration-time curve (AUC) of 2 on days 1, 8, and 15 of a 28-day cycle with or without trastuzumab given at a dose of 2 mg/kg (with a loading dose of 4 mg/kg) every week. In every-3-week schedule, patients received 175 mg/m2 paclitaxel followed by carboplatin at an AUC of 6 on day 1 of a 21-day cycle with or without trastuzumab at 6 mg/kg (with a loading dose of 8 mg/kg) every 3 weeks. 46 newly diagnosed patients all administered PCrbH (weekly or every-3-week) for 4-6 cycles. Breast surgery (BCS or mastectomy) and standard axillary dissection were performed 2–4 weeks after the last chemotherapy dose. 18 of 46 patients received SLNB just before axillary dissection. The surgery type was at the surgeon's discretion.
The primary end point was to evaluate the factors influencing the pathologically node negative rate (pNNR) of the axillary lymph nodes, which was defined as the absence of malignant cells in all lymph nodes removed by axillary dissection assessed microscopically with hematoxylin/eosin staining.
Statistical Analysis
Medians and ranges of demographic characteristics and distribution of clinical characteristics were presented. Clinical characteristics between pNNR and non-pNNR subgroups for both Group-A and Group-B patients were compared using Pearson's χ2test or Fisher's exact test where appropriate. Similar analyses were done between Group-C and Group-D patients to identify and confirm the previous results. Multivariate analysis was performed using logistic regression analysis. The regimen, menopausal status, baseline tumor stage and ER/PR status were taken into account in the multivariate analysis. The hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated. All tests were two-sided and P-value less than 0.05 was considered statistically significant. All statistical analyses were performed using SPSS v.19.0 (SPSS, Chicago, IL).
Results
Between November-2007 and June-2013, 255 patients with HER2-positive breast cancer were enrolled, with median age of 50 years. One hundred and fifty-seven 157(61.6%) patients with cN+FNA+ were included in group-A and 98 (38.4%) patients with cN+FNA- (n = 64) or cN- (n = 34) were included in group-B. The patients' major characteristics were shown in Table 1. The overall pCR rate of the primary site was 42.5%, with 39.5% in group-A and 45.9% in group-B.
Table 1. The demographic characteristics and clinicopathological information of 255 patients for the main analysis.
| Characteristics (n, %) | Group-A,cN+FNA+ N = 157 | Group-B,cN+FNA- N = 64 | Group-B,cN- N = 34 | Total N = 255 |
| Median age* yr(range) | 51(21–71) | 49.5(26–79) | 51.5(27–63) | 50(21–79) |
| NAC regimen | ||||
| PCrbH | 95(60.5%) | 39(60.9%) | 15(44.1%) | 149(58.4%) |
| PCrb | 62(39.5%) | 25(39.1%) | 19(55.9%) | 106(41.6%) |
| Median BSA, m2(range) | 1.60(1.31–1.96) | 1.58(1.37–2.00) | 1.58(1.40–2.26) | 1.60(1.31–2.26) |
| Median BMI(range) | 23.2(19.9–32.1) | 23.2(15.9–33.5) | 23.7(16.9–40.4) | 23.2(15.9–23.7) |
| Menopausal status | ||||
| Pre- | 82(52.2%) | 32(50.0%) | 21(61.8%) | 135(52.9%) |
| Post- | 75(47.8%) | 32(50.0%) | 13(38.2%) | 120(47.1%) |
| T | ||||
| 1 | 10(6.4%) | 2(3.1%) | 1(2.9%) | 13(5.1%) |
| 2 | 49(31.2%) | 20(31.3%) | 13(38.2%) | 82(32.2%) |
| 3 | 22(14.0%) | 12(18.8%) | 7(20.6%) | 41(16.1%) |
| 4 | 76(48.4%) | 30(46.9%) | 13(38.2%) | 119(46.7%) |
| N | ||||
| 0 | 0(0.0%) | 56(87.5%) | 0(0.0%) | 56(22.0%) |
| 1 | 117(74.5%) | 0(0.0%) | 0(0.0%) | 117(45.6%) |
| 2 | 14(8.9%) | 0(0.0%) | 0(0.0%) | 14(5.5%) |
| 3 | 26(16.6%) | 8(12.5%) | 4(11.8%) | 38(14.9%) |
| unknown | 0(0.0%) | 0(0.0%) | 30(88.2%) | 30(11.8%) |
| Estrogen receptor | ||||
| Positive | 67(42.7%) | 20(31.3%) | 16(47.1%) | 103(40.4%) |
| Poor | 90(57.3%) | 44(68.7%) | 18(52.9%) | 152(59.6%) |
| Progesterone receptor | ||||
| Positive | 66(42.0%) | 21(32.8%) | 18(52.9%) | 105(41.2%) |
| Poor | 91(58.0%) | 43(67.2%) | 16(47.1%) | 150(58.8%) |
| Breast Surgery | ||||
| Mastectomy | 147(93.6%) | 62(96.9%) | 34(100%) | 243(95.3%) |
| BCS | 10(6.4%) | 2(3.1%) | 0(0.0%) | 12(4.7%) |
Data was given as the number and percent (n/N, %) of patients, unless otherwise stated.
*Median and full range.
FNA = fine-needle aspiration; NAC = neoadjuvant chemotherapy; P = paclitaxel; Crb = carboplatin; H = Trastuzumab (Herceptin); T = primary tumor site of TNM classification system; N = the regional lymph node involvement of TNM classification system; BCS = breast conserving surgery; ALND = axillary lymph node dissection.
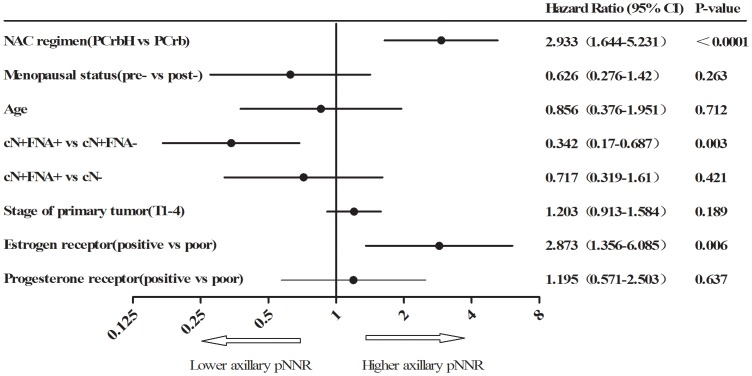
After axillary dissection, the overall pNNR were 52.7% and 69.4% in group-A and group-B, respectively. Axillary pNNR was highly correlated with the pCR rate of the primary site (p<0.0001). With the PCrbH regimen, 61.6% patients achieved pNNR in group-A and 81.5% in group-B, which were significantly higher than those treated with the PCrb regimen (p = 0.011 in group-A and p = 0.004 in group-B). The outcomes of axillary dissection in different patient subgroups were listed in Table 2. The subtype of ER-poor and HER2-positive breast cancer treated with neoadjuvant PCrbH showed the highest pNNR (79.6% in group-A and 87.9% in group-B) and the least residual (≥4 nodes) involvement (1.9% in group-A and 3% in group-B). These findings were also confirmed by multivariate analysis which indicated that trastuzumab addition to NAC (HR = 2.933) and an ER-poor status (HR = 2.873) may be related factors for predicting a higher pNNR after NAC in HER2-positive breast cancer (Fig. 2).
Table 2. Axillary nodal status after neo-adjuvant therapy according to different subgroups.
| Group-A (cN+/FNA+) N = 157 | Group-B (cN+/FNA- or cN-) N = 98 | |||||||
| pNNR | non-pNNR | pNNR | non-pNNR | |||||
| No. of involved nodes (n, %) | 0 | 1–3 | ≥4 | P value | 0 | 1–3 | ≥4 | P value |
| Overall | 83(52.9%) | 43(27.4%) | 31(19.7%) | 68(69.4%) | 15(15.3%) | 15(15.3%) | ||
| NAC regimen | 0.011 | 0.004 | ||||||
| PCrbH | 58(61.1%) | 23(24.2%) | 14(14.7%) | 44(81.5%) | 6(11.1%) | 4(7.4%) | ||
| PCrb | 25(40.3%) | 20(32.3%) | 17(27.4%) | 24(54.5%) | 9(20.5%) | 11(25%) | ||
| Menopausal Status | 0.597 | 0.328 | ||||||
| Pre- | 38(50.7%) | 25(33.3%) | 12(16.0%) | 29(64.4%) | 9(20.0%) | 7(15.6%) | ||
| Post- | 45(54.9%) | 18(22%) | 19(23.2%) | 39(73.6%) | 6(11.3%) | 8(15.1%) | ||
| T stage | 0.661 | 0.418 | ||||||
| 1 | 6(60.0%) | 2(20.0%) | 2(20.0%) | 3(100.0%) | 0(0.0%) | 0(0.0%) | ||
| 2 | 29(59.2%) | 9(18.4%) | 11(22.4%) | 20(60.6%) | 7(21.2%) | 6(18.2%) | ||
| 3 | 11(50.0%) | 7(31.8%) | 4(18.2%) | 14(73.7%) | 3(15.8%) | 2(10.5%) | ||
| 4 | 37(48.7%) | 25(32.9%) | 14(18.4%) | 31(72.1%) | 5(11.6%) | 7(16.3%) | ||
| ER | 0.002 | 0.024 | ||||||
| Positive | 26(38.8%) | 19(28.4%) | 22(32.8%) | 20(55.6%) | 7(19.4%) | 9(25.0%) | ||
| Poor | 57(63.3%) | 24(26.7%) | 9(10.0%) | 48(77.4%) | 8(12.9%) | 6(9.7%) | ||
| PgR | 0.113 | 0.007 | ||||||
| Positive | 30(45.5%) | 18(27.3%) | 18(27.3%) | 21(53.8%) | 8(20.5%) | 10(25.6%) | ||
| Poor | 53(58.2%) | 25(27.5%) | 13(14.3%) | 47(79.7%) | 7(11.9%) | 5(8.5%) | ||
| NAC regimen & ER status | ||||||||
| PCrbH | <0.0001 | 0.129 | ||||||
| ER Positive | 15(36.6%) | 13(31.7%) | 13(31.7%) | 15(71.4%) | 3(14.3%) | 3(14.3%) | ||
| ER-poor | 43(79.6%) | 10(18.5%) | 1(1.9%) | 29(87.9%) | 3(9.1%) | 1(3.0%) | ||
| PCrb | 0.787 | 0.042 | ||||||
| ER Positive | 11(42.3%) | 6(23.1%) | 9(34.6%) | 5(33.3%) | 4(26.7%) | 6(40.0%) | ||
| ER-poor | 14(38.9%) | 14(38.9%) | 8(22.2%) | 19(65.5%) | 5(17.2%) | 5(17.2%) | ||
Data was given as the number and percent (n/N, %) of patients.
FNA = fine needle aspiration; pNNR = pathologically node negative rate; NAC = neoadjuvant chemotherapy; P = paclitaxel; Crb = carboplatin; H = Trastuzumab (Herceptin); ER = estrogen receptor; PgR = progesterone receptor; T = primary tumor site of TNM classification system.
P values were calculated from χ2 test to compare pNNR and non-pNNR proportions of patients for different subgroups.
Figure 2. Multivariate regression analysis for axillary pNNR.
NAC = neoadjuvant chemotherapy; pNNR = pathologically node negative rate; P = paclitaxel; Crb = carboplatin; H = Trastuzumab (Herceptin). In multivariate analysis, trastuzumab added and ER-poor status showed higher pNNR in HER2-positive breast cancer.
From June 2013 to March 2014, another 46 patients were enrolled to identify and confirm the previous results, 36 (78.3%) with positive axillary nodes (group-C) and 10 (21.7%) negative both by FNA (group-D). The patients' major characteristics were similar with the main study set. They all had a NAC regimen of PCrbH. After axillary dissection, the overall pNNR was 61.1% and 60.0% for group-C and group-D, respectively. The ER-poor/HER2-positive subtype acquired considerable high pNNRs (77.8% in group-C and 71.4% in group-D, seen in S1 Table).
Among 18 patients received SLNB before axillary dissection, SLN was successfully detected in 16 (88.9%). As shown in S2 Table, all 5 ER-poor patients with negative SLN showed pathologically negative node (100%) after axillary dissection.
Discussion
Immunohistochemical phenotype of ER-poor and HER2-positive breast cancer accounts for approximately 10-15% of early breast cancers. They have been found more sensitive to chemo-trastuzumab therapy in the neoadjuvant setting with a higher pCR rate in the primary site than that of the ER-positive/HER2-positive phenotype, as shown in Table 3 [9], [17]–[20]. In our current study, we observed optimal pathological axillary node downstaging of HER2-positive breast cancer after an NAC regimen of PCrbH. This result also indicated that the therapeutic sensitivity of breast cancer metastasis to the axilla is at least equal to or even higher than that of the primary site.
Table 3. pCR rate after neoadjuvant chemotherapy plus trastuzumab in breast cancer.
| Studies | Phase | Clinical stage | Neoadjuvant regimen | pCR (%) | pCR(%) for HR-positive | pCR(%) for HR-negative |
| MD Anderson [9] | III | II–III | P+T→FEC+T | 65.0 | 61.5 | 70.0 |
| NOAH [17] | III | III | AP+T→P+T→CMF+T | 38.0 | 18.0 | 38.0 |
| NeoALTTO [18] | III | II–III | Weekly P+T | 28.0 | 22.7 | 36.5 |
| NeoSphere [19] | III | II–III | D+T | 23.0 | 20.0 | 36.8 |
| NSABP B-41 [20] | III | II–III | AC→weekly P+T | 49.4 | 46.7 | 65.5 |
A = doxorubicin; CMF = cyclophosphamide, methotrexate and 5- fluorouracil; D = docetaxel; FEC = 5-fluorouracil, epirubicin and cyclophosphamide; P = paclitaxel; T = trastuzumab.
pCR was defined as no invasive tumor in the breast and axilla.
For clinically node negative early breast cancer, SLNB is now routinely used as a standard method for pathological staging of the axilla. SLNB has little morbidity and high accuracy that is comparable with traditional axillary dissection [21]. However, the limitations of SLNB for evaluating a downstaged breast cancer after NAC should be considered, because of the increased false negative rate (FNR: 5.6–35.5%) and decreased detection rate (77.6–98.0%) [22]–[26]. SENTINA (SENTinel NeoAdjuvant) trial is thus far the largest prospective, multicenter cohort study to investigate the feasibility and accuracy of SLNB after NAC [27]. In this study, 592 breast cancer patients who converted from a clinically axillary node positive to negative status after NAC received SLNB just before axillary dissection. A relatively lower sentinel node detection rate of 80.1% (compared with 99.1% in SLNB before NAC) and a FNR of 14.2% were observed. The ACOSOG Z1071 trial was designed to determine the FNR of SLN surgery after chemotherapy in women initially presenting with cN1 disease [28]. In this trial, the sentinel node detection (≥2 SLN) rate was 80.9% and the FNR was 12.6%. According to these reports, the missing rate of SLNB after NAC far exceeded the acceptable extension of <5% recommended by the ASCO guideline [29]. However, if there was a subgroup of patients who were more likely to have a pathologically node negative status in the axilla after NAC, the false negative error from SLNB would decrease and most of the axillary dissection could be avoided for them. For example, in group-A from our study, a subgroup (n = 54) of FNA confirmed node positive patients with the ER-poor and HER2-positive phenotype achieved a pNNR of 79.6% after neoadjuvant PCrbH. Supposing that these tumors had the same FNR and detection rate of SLNB as reported in Z1071 trial [28], axillary dissection could safely be avoided in approximately two thirds of the patients from a successful and true negative SLNB (pNNR×Detection Rate [79.6%*80.9%] = 64.40%) with an estimated acceptable false negative error rate of about 2 cases per 100 patients ([1-pNNR]×FNR×Detection Rate = 2.08%).
For patients with clinically node negative disease who require NAC (i.e patients in group-B and group-D of the study), the timing of SLNB is still controversial. Conducting SLNB before NAC can provide an accurate histological evaluation of the initial axillary nodal status, which might be essential in the prognosis estimation and for guidance of the following loco regional treatment, including axillary dissection and radiation. However, growing evidences have suggested that the nodal status after NAC reflects the prognosis more accurately than the initial axillary status [30]. Thus, a reliable estimation of an excellent regional node response to the NAC could also change the current strategy of axillary dissection to a more conservative approach. Unfortunately, according to the data from the SENTINA trial, if SLNB was performed before NAC and identified as positive, a second SLNB after NAC was far less reliable than axillary dissection, with a detection rate of only 60.8% and a FNR of 51.6% [27]. For example, in group-B of the current study, a subgroup (n = 33) of clinically node negative patients with the ER-poor and HER2-positive phenotype achieved the highest pNNR (87.9%) after neoadjuvant PCrbH. According to the results from the SENTINA trial, if SLNB is scheduled before NAC, the repeat SLNB for those sentinel node positive patients after NAC might result in a false negative error rate of 3.79%. Alternatively, a single procedure of SLNB scheduled after NAC could be more feasible for this group of patients and the axillary dissection could be safely avoided in at least 70% of the patients with a lower false negative error rate that is expected to be 1.38%.
Recently, data from some prospective studies indicated that if there is not palpable adenopathy breast cancer with positive axillary sentinel nodes (micro-metastatic size<2 mm or 1–3 macro-metastatic nodes) has no significant difference in regional recurrence rate, disease free survival or overall survival for complete axillary dissection versus axillary nodal irradiation or observation except that the complications were reduced in the axilla conservation groups [31]–[33]. In the current study, very few patients (1.9% in group-A and 3% in group-B) with the ER-poor and HER2-positive phenotype had more than 3 macro-metastatic nodes remaining in the axilla after neoadjuvant PCrbH. Therefore, with the replacement of axillary irradiation, avoidance of axillary dissection in the ER-poor and HER2-positive subtype might be extended to patients with 1–3 positive sentinel nodes including those who failed to achieve a successful SLNB after NAC. Further studies are needed to verify these results.
Conclusions
In conclusion, patients with ER-poor and HER2-positive subtype of breast cancer are potential candidates for avoiding unnecessary axillary dissection due to its higher axillary node negative status rate after neoadjuvant chemo-trastuzumab therapy. Further prospective trials should be focused on SLNB and axillary irradiation after neoadjuvant chemo-trastuzumab therapy to take the place of traditional axillary dissection in the ER-poor and HER2-positive subtype of breast cancer.
Supporting Information
Axillary nodal status after neo-adjuvant therapy according to ER status among 46 newly patients. Data was given as the number and percent (n/N, %) of patients. FNA = fine needle aspiration; pNNR = pathologically node negative rate; ER = estrogen receptor.
(DOC)
Axillary nodal status after neo-adjuvant therapy according to ER status among patients received sentinel lymph node biopsy before axillary dissection. Data was given as the number and percent (n/N, %) of patients. SLNB = sentinel lymph node biopsy; pNNR = pathologically node negative rate; ER = estrogen receptor.
(DOC)
Data Availability
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper and its Supporting Information files.
Funding Statement
This research is supported by the Shanghai Committee of Science and Technology, China (grant 12DZ2260100), and the Shanghai United Developing Technology Project of Municipal Hospitals (SHDC12010116), the Key Clinical Program of the Ministry of Health (2010–2012). The research funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. No other potential confict of interest relevant to this article was reported.
References
- 1. Carlson RW, Allred DC, Anderson BO, Burstein HJ, Carter WB, et al. (2009) Breast cancer. Clinical practice guidelines in oncology. J Natl Compr Canc Netw 7:122–192. [DOI] [PubMed] [Google Scholar]
- 2. Kuehn T, Klauss W, Darsow M, Regele S, Flock F, et al. (2000) Long-term morbidity following axillary dissection in breast cancer patients–clinical assessment, significance for life quality and the impact of demographic, oncologic and therapeutic factors. Breast Cancer Res Treat 64:275–286. [DOI] [PubMed] [Google Scholar]
- 3. Ashikaga T, Krag DN, Land SR, Julian TB, Anderson SJ, et al. (2010) Morbidity results from the NSABP B-32 trial comparing sentinel lymph node dissection versus axillary dissection. J Surg Oncol 102:111–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mauri D, Pavlidis N, Ioannidis JP (2005) Neoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysis. J Natl Cancer Inst 97:188–194. [DOI] [PubMed] [Google Scholar]
- 5. Mieog JS, van der Hage JA, van de Velde CJ (2007) Neoadjuvant chemotherapy for operable breast cancer. Br J Surg 94:1189–1200. [DOI] [PubMed] [Google Scholar]
- 6. Fitzal F, Riedl O, Mittlbock M, Dubsky P, Bartsch R, et al. (2011) Oncologic safety of breast conserving surgery after tumour downsizing by neoadjuvant therapy: a retrospective single centre cohort study. Breast Cancer Res Treat 127:121–128. [DOI] [PubMed] [Google Scholar]
- 7. Bear HD, Anderson S, Smith RE, Geyer CE Jr, Mamounas EP, et al. (2006) Sequential preoperative or postoperative docetaxel added to preoperative doxorubicin plus cyclophosphamide for operable breast cancer: National Surgical Adjuvant Breast and Bowel Project Protocol B-27. J Clin Oncol 24:2019–2027. [DOI] [PubMed] [Google Scholar]
- 8. Reese DM, Slamon DJ (1997) HER-2/neu signal transduction in human breast and ovarian cancer. Stem Cells 15:1–8. [DOI] [PubMed] [Google Scholar]
- 9. Buzdar AU, Ibrahim NK, Francis D, Booser DJ, Thomas ES, et al. (2005) Significantly higher pathologic complete remission rate after neoadjuvant therapy with trastuzumab, paclitaxel, and epirubicin chemotherapy: results of a randomized trial in human epidermal growth factor receptor 2-positive operable breast cancer. J Clin Oncol 23:3676–3685. [DOI] [PubMed] [Google Scholar]
- 10. Untch M, Fasching PA, Konecny GE, Hasmuller S, Lebeau A, et al. (2011) Pathologic complete response after neoadjuvant chemotherapy plus trastuzumab predicts favorable survival in human epidermal growth factor receptor 2-overexpressing breast cancer: results from the TECHNO trial of the AGO and GBG study groups. J Clin Oncol 29:3351–3357. [DOI] [PubMed] [Google Scholar]
- 11. Chen XS, Nie XQ, Chen CM, Wu JY, Wu J, et al. (2010) Weekly paclitaxel plus carboplatin is an effective nonanthracycline-containing regimen as neoadjuvant chemotherapy for breast cancer. Ann Oncol 21:961–967. [DOI] [PubMed] [Google Scholar]
- 12. Yu KD, Liu GY, Chen CM, Li JW, Lu JS, et al. (2013) Weekly paclitaxel/carboplatin/trastuzumab therapy improves pathologic complete remission in aggressive HER2-positive breast cancers, especially in luminal-B subtype, compared with a once-every-3-weeks schedule. Oncologist 18:511–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, Allred DC, et al. (2007) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25:118–145. [DOI] [PubMed] [Google Scholar]
- 14. Regan MM, Viale G, Mastropasqua MG, Maiorano E, Golouh R, et al. (2006) Re-evaluating adjuvant breast cancer trials: assessing hormone receptor status by immunohistochemical versus extraction assays. J Natl Cancer Inst 98:1571–1581. [DOI] [PubMed] [Google Scholar]
- 15. Phillips T, Murray G, Wakamiya K, Askaa J, Huang D, et al. (2007) Development of standard estrogen and progesterone receptor immunohistochemical assays for selection of patients for antihormonal therapy. Appl Immunohistochem Mol Morphol 15:325–331. [DOI] [PubMed] [Google Scholar]
- 16.Sobin L, Gospodarowicz M, Wittekind C (2009) UICC TNM classification of malignant tumours, 7th edn. New York: Wiley-Liss.
- 17. Gianni L, Eiermann W, Semiglazov V, Manikhas A, Lluch A, et al. (2010) Neoadjuvant chemotherapy with trastuzumab followed by adjuvant trastuzumab versus neoadjuvant chemotherapy alone, in patients with HER2-positive locally advanced breast cancer (the NOAH trial): a randomised controlled superiority trial with a parallel HER2-negative cohort. Lancet 375:377–384. [DOI] [PubMed] [Google Scholar]
- 18. Baselga J, Bradbury I, Eidtmann H, Di Cosimo S, de Azambuja E, et al. (2012) Lapatinib with trastuzumab for HER2-positive early breast cancer (NeoALTTO): a randomised, open-label, multicentre, phase 3 trial. Lancet 379:633–640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Gianni L, Pienkowski T, Im YH, Roman L, Tseng LM, et al. (2012) Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial. Lancet Oncol 13:25–32. [DOI] [PubMed] [Google Scholar]
- 20. Robidoux A, Tang G, Rastogi P, Geyer CE Jr, Azar CA, et al. (2013) Lapatinib as a component of neoadjuvant therapy for HER2-positive operable breast cancer (NSABP protocol B-41): an open-label, randomised phase 3 trial. Lancet Oncol 14:1183–1192. [DOI] [PubMed] [Google Scholar]
- 21. Krag DN, Anderson SJ, Julian TB, Brown AM, Harlow SP, et al. (2007) Technical outcomes of sentinel-lymph-node resection and conventional axillary-lymph-node dissection in patients with clinically node-negative breast cancer: results from the NSABP B-32 randomised phase III trial. Lancet Oncol 8:881–888. [DOI] [PubMed] [Google Scholar]
- 22. Lee S, Kim EY, Kang SH, Kim SW, Kim SK, et al. (2007) Sentinel node identification rate, but not accuracy, is significantly decreased after pre-operative chemotherapy in axillary node-positive breast cancer patients. Breast Cancer Res Treat 102:283–288. [DOI] [PubMed] [Google Scholar]
- 23. Newman EA, Sabel MS, Nees AV, Schott A, Diehl KM, et al. (2007) Sentinel lymph node biopsy performed after neoadjuvant chemotherapy is accurate in patients with documented node-positive breast cancer at presentation. Ann Surg Oncol 14:2946–2952. [DOI] [PubMed] [Google Scholar]
- 24. Shen J, Gilcrease MZ, Babiera GV, Ross MI, Meric-Bernstam F, et al. (2007) Feasibility and accuracy of sentinel lymph node biopsy after preoperative chemotherapy in breast cancer patients with documented axillary metastases. Cancer 109:1255–1263. [DOI] [PubMed] [Google Scholar]
- 25. Alvarado R, Yi M, Le-Petross H, Gilcrease M, Mittendorf EA, et al. (2012) The role for sentinel lymph node dissection after neoadjuvant chemotherapy in patients who present with node-positive breast cancer. Ann Surg Oncol 19:3177–3184. [DOI] [PubMed] [Google Scholar]
- 26. Classe JM, Bordes V, Campion L, Mignotte H, Dravet F, et al. (2009) Sentinel lymph node biopsy after neoadjuvant chemotherapy for advanced breast cancer: results of Ganglion Sentinelle et Chimiotherapie Neoadjuvante, a French prospective multicentric study. J Clin Oncol 27:726–732. [DOI] [PubMed] [Google Scholar]
- 27. Kuehn T, Bauerfeind I, Fehm T, Fleige B, Hausschild M, et al. (2013) Sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy (SENTINA): a prospective, multicentre cohort study. Lancet Oncol 14:609–618. [DOI] [PubMed] [Google Scholar]
- 28. Boughey JC, Suman VJ, Mittendorf EA, Ahrendt GM, Wilke LG, et al. (2013) Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (Alliance) clinical trial. JAMA 310:1455–1461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lyman GH, Giuliano AE, Somerfield MR, Benson AB 3rd, Bodurka DC, et al. (2005) American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early-stage breast cancer. J Clin Oncol 23:7703–7720. [DOI] [PubMed] [Google Scholar]
- 30. Rouzier R, Extra JM, Klijanienko J, Falcou MC, Asselain B, et al. (2002) Incidence and prognostic significance of complete axillary downstaging after primary chemotherapy in breast cancer patients with T1 to T3 tumors and cytologically proven axillary metastatic lymph nodes. J Clin Oncol 20:1304–1310. [DOI] [PubMed] [Google Scholar]
- 31. Straver ME, Meijnen P, van Tienhoven G, van de Velde CJ, Mansel RE, et al. (2010) Role of axillary clearance after a tumor-positive sentinel node in the administration of adjuvant therapy in early breast cancer. J Clin Oncol 28:731–737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Galimberti V, Cole BF, Zurrida S, Viale G, Luini A, et al. (2013) Axillary dissection versus no axillary dissection in patients with sentinel-node micrometastases (IBCSG 23-01): a phase 3 randomised controlled trial. Lancet Oncol 14:297–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Giuliano AE, Hunt KK, Ballman KV, Beitsch PD, Whitworth PW, et al. (2011) Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA 305:569–575. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Axillary nodal status after neo-adjuvant therapy according to ER status among 46 newly patients. Data was given as the number and percent (n/N, %) of patients. FNA = fine needle aspiration; pNNR = pathologically node negative rate; ER = estrogen receptor.
(DOC)
Axillary nodal status after neo-adjuvant therapy according to ER status among patients received sentinel lymph node biopsy before axillary dissection. Data was given as the number and percent (n/N, %) of patients. SLNB = sentinel lymph node biopsy; pNNR = pathologically node negative rate; ER = estrogen receptor.
(DOC)
Data Availability Statement
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper and its Supporting Information files.