Abstract
Necrotic enteritis (NE) is an important intestinal infectious disease of commercial poultry flocks caused by Clostridium perfringens. Using an experimental model of NE involving co-infection with C. perfringens and Eimeria maxima, transcriptome profiling and functional genomics approaches were applied to identify the genetic mechanisms that might regulate the host response to this disease. Microarray hybridization identified 1,049 transcripts whose levels were altered (601 increased, 448 decreased) in intestinal lymphocytes from C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls. Five biological functions, all related to host immunity and inflammation, and 11 pathways were identified from this dataset. To further elucidate the role of host genetics in NE susceptibility, two inbred chicken lines, ADOL line 6 and line 7 which share an identical B2 major histocompatibility complex haplotype but differ in their susceptibility to virus infection, were compared for clinical symptoms and the expression levels of a panel of immune-related genes during experimental NE. Line 6 chickens were more susceptible to development of experimental NE compared with line 7, as revealed by decreased body weight gain and increased E. maxima oocyst shedding. Of 21 immune-related genes examined, 15 were increased in C. perfringens/E. maxima co-infected line 6 vs. line 7 chickens. These results suggest that immune pathways are activated in response to experimental NE infection and that genetic determinants outside of the chicken B complex influence resistance to this disease.
Introduction
Necrotic enteritis (NE) is an acute clostridial disease of economic importance to the poultry industry [1]. NE is caused by Clostridium perfringens, a Gram-positive, rod-shaped, spore-forming, and oxygen-tolerant anaerobe [2]. C. perfringens is a normal component of the chicken gut microbiota and the alterations in the host-pathogen relationship that regulate the development of NE remain to be determined [3]. A variety of predisposing factors, however, are known to promote disease, including diets containing high levels of wheat, barley, or poorly digestible proteins and co-infection by the apicomplexan protozoa, Eimeria, the etiologic agent of avian coccidiosis [4], [5]. Because of the risk of transmission to humans through the food chain, C. perfringens also is important for public health [4], [6]. Traditionally, prophylactic in-feed antibiotics have proven effective for control of clostridial field infections in chickens. However, NE has recently emerged as a significant problem as a result of decreased antibiotic usage amid concerns over the appearance of antibiotic-resistant human pathogens [4], [7].
Only limited information exists on the role of genetic factors in controlling the susceptibility of chickens to NE. Since many common host immune responses are elicited following infection by viral, bacterial, or parasitic pathogens [9], we hypothesized that two inbred chicken lines with dissimilar susceptibility to virus-induced diseases might provide insights into the genetic mechanisms regulating avian NE. White Leghorn USDA Avian Disease and Oncology Laboratory (ADOL) line 6 and line 7 chickens are 99% inbred and possess the major histocompatibility complex (MHC) B2 genotype, but differ in their immunoglobulin G allotype (G1E and G1A, respectively). Both lines were originally selected based on their susceptibility to avian leukosis retroviruses (ALV) and Marek's diseases herpesviruses (MDV), with line 6 being the more susceptible to both viruses [8]. Although susceptibility to Marek's disease has not been associated with co-susceptibility Eimeria infection [9], line 6 chickens do exhibit greater susceptibility to E. maxima infection compared with line 7 [10]. However, no published studies have examined NE susceptibility in these two chicken lines. Therefore, the objectives of the current study were (1) to determine whether ADOL line 6 and line 7 exhibit different susceptibilities to experimental NE using a C. perfringens/E. maxima co-infection model and (2) to compare transcriptional profiles in co-infected vs. uninfected Ross chickens in order to identify candidate genes that might be differentially expressed in NE-afflicted line 6 vs. line 7 birds.
Materials and Methods
Experimental animals
Commercial Ross x Ross broilers were obtained from Longenecker's Hatchery (Elizabethtown, PA). ADOL line 6 and line 7 chickens were hatched at the USDA Avian Disease and Oncology Laboratory (East Lansing, MI). All chickens were housed in Petersime starter brooder units in an Eimeria-free facility for 14 days post-hatch and provided with feed and water ad libitum. Chickens were transferred to larger hanging cages with 2 birds/cage at a separate location where they were co-infected with C. perfringens and E. maxima and kept until the end of the experimental period.
Experimental NE disease model
Chickens were infected with E. maxima strain 41A (1.0×104 oocysts/bird) by oral gavage on day 14 post-hatch followed by infection with C. perfringens strain Del-1 (1.0×109 colony forming units/bird) by oral gavage on day 18 [11]. To facilitate development of NE, birds were fed an antibiotic-free, certified organic starter diet containing 17% crude protein between days 1–18 followed by a standard grower diet containing 24% crude protein between days 18–20. Uninfected control animals were housed in neighboring cages and given the same diet. Body weight gains (16 birds/group) were measured and fecal samples were collected between days 14–20 post-hatch (days 0–9 post-infection with E. maxima). Oocyst numbers were determined using a McMaster chamber (HK Inc., Tokyo, Japan) [12]. Gut lesion scores were determined at day 20 post-hatch (day 2 post-infection with C. perfringens) on a scale from 0 (none) to 4 (high) in a blinded fashion by three independent observers as described [13], [14]. All protocols were approved by the USDA Beltsville Area Institutional Animal Care and Use Committee.
RNA extraction from intestinal intraepithelial lymphocytes (IELs)
Total RNA was isolated from intestinal IELs of Ross chickens as described [15]. Briefly, the intestinal jejunum was removed at day 20 post-hatch, cut longitudinally, and washed three times with ice-cold Hank's balanced salt solution (HBSS). Harvested tissues were incubated in HBSS containing 0.5 mM EDTA and 5% FBS for 20 min at 37°C with constant swirling. Cells released into the supernatant were passed through nylon wool (Robbins Scientific, Sunnyvale, CA), and washed twice with HBSS containing 5% FBS. IELs were purified by Percoll density gradient centrifugation and washed three times with HBSS containing 5% FBS. Total RNA was isolated using Trizol (Invitrogen, Grand Island, NY).
Microarray hybridization
RNA from 6 birds was pooled into 2 equal weight samples including RNA from 3 birds each. Each sample was labeled with cyanine 3 (Cy3)- or Cy5-CTP using the Two-Color Quick Amp Labeling Kit (Agilent Technologies, Santa Clara, CA) as described [16]. Labeled RNAs from uninfected and C. perfringens/E. maxima co-infected chickens were hybridized with the Chicken Gene Expression Microarray (Agilent Technologies) containing 43,803 elements. Four hybridizations were conducted with alternation of Cy3- and Cy5-labeled RNAs to prevent data distortion from sample labeling [17]. Microarray images were scanned and data extraction and analysis were performed using Feature Extraction software version 10.7.3.1 (Agilent Technologies).
Microarray data analysis
GeneSpring GX10 software (Agilent Technologies) was used to qualify and normalize image analysis data and to determine the fold changes in gene expression. Average signal intensities were corrected for background signals and normalized by block locally-weighted regression scatterplot smoothing (LOWESS). Flag information, the feature quality data from the signals on microarray, was applied to the spots without blank or invalid values from each sample asymptotic t-test analysis with P<0.05 was performed to analyze the significance of the differences between the uninfected and NE-afflicted groups. To generate signal ratios, signal channel values from NE-afflicted birds were divided by values from uninfected negative controls. All microarray information and data were deposited in the Gene Expression Omnibus database (series record number, GSE51154). Differentially expressed genes between NE-afflicted and uninfected control chickens were analyzed using Ingenuity Pathway Analysis (IPA) software (Ingenuity Systems, Redwood City, CA). Each identifier was mapped to its corresponding gene in IPA. These identified genes were superimposed onto the global molecular networks contained within IPA. IPA functional analysis was performed to identify the biological functions associated with the identified genes from the mapped datasets.
Quantitative (q)RT-PCR
Equivalent amounts of the same intestinal IEL RNA used for microarray hybridization from uninfected or C. perfringens/E. maxima co-infected Ross chickens, and fresh IEL RNA from uninfected or co-infected ADOL line 6 and line 7 chickens, were reverse-transcribed using the AffinityScript Multiple Temperature cDNA Synthesis Kit (Agilent Technologies) as described [20]. PCR amplification was performed using the oligonucleotide primers listed in Table 1 with the Mx3000P system and Brilliant SYBR Green qRT-PCR master mix (Agilent Technologies). Standard curves were generated using log10 diluted standard RNAs and the levels of individual transcripts were normalized to those of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) by the Q-gene program [18]. The cycle threshold (Ct) value of the target gene was normalized to GAPDH and calibrated to the relevant control value. Each analysis was performed in triplicate.
Table 1. Oligonucleotide primers used for qRT-PCR.
| Gene Symbol | Primer Sequence (5′→3′) | Entrez Gene Name | GenBank Accession No. | |
| LCP1 | Forward | GCCAGTAGACTGGAACAGAG | Lymphocyte cytosolic protein 1 (L-plastin) | NM_001008440.1 |
| Reverse | TTCTCTCCACCACCAATATC | |||
| MTTP | Forward | TTCCTATATGCCTGTGGATT | Microsomal triglyceride transfer protein | NM_001109784.1 |
| Reverse | AGGTACATCCTCACGTTGTC | |||
| CALB1 | Forward | GCTTGGACTTAACACCTGAA | Calbindin 1, 28 kDa | NM_205513.1 |
| Reverse | TCCTCAGAATCAATGAAACC | |||
| CXCL14 | Forward | CCAGTGCAGAAGGAGTAAAG | Chemokine (C-X-C motif) ligand 14 | NM_204712.2 |
| Reverse | TTCCATACTCGGTACCACTT | |||
| ANXA1 | Forward | CAATGATGCAAGGGCCTTAT | Annexin A1 | NM_206906.1 |
| Reverse | CTTCATTGCCAGGTGGAGTT | |||
| IL8 | Forward | GGCTTGCTAGGGGAAATGA | Interleukin 8 | AJ009800 |
| Reverse | AGCTGACTCTGACTAGGAAACTGT | |||
| APP | Forward | GGAAGCGATGATAAGGTGGTAGAAGAACAA | Amyloid beta (A4) precursor protein | NM_204308 |
| Reverse | CATCACCATCATCATCGTCATCATCATCAG | |||
| ARHGEF6 | Forward | ACTGCTGGGAAATGTGGAGGAAATC | Rac/Cdc42 guanine nucleotide exchange factor (GEF) 6 | NM_001006432 |
| Reverse | ACGTCAGGTACAGGGAGCGGAACT | |||
| BCL2 | Forward | GATGTGCGTCGAGAGCGTCAA | B-cell CLL/lymphoma 2 | NM_205339 |
| Reverse | GTGCAGGTGCCGGTTCAGGT | |||
| COL1A2 | Forward | CTCAGCTTTGTGGATACGCGGATTTTG | Collagen, type I, alpha 2 | NM_001079714 |
| Reverse | GCCCTGCAGATGCCTCACTCACA | |||
| CXCL14 | Forward | GCCTTGCTTCTGCTGGTCATC | Cchemokine (C-X-C motif) ligand 14 | NM_204712 |
| Reverse | ATCTTATTTTCGGCCCTTTCCTT | |||
| GJA1 | Forward | GTCTTCATGCTGGTAGTGTCTTTGGTGTCT | Gap junction protein, alpha 1, 43 kDa | NM_204586 |
| Reverse | CTGTGGGAGTAGGGGTCGGTTTTTC | |||
| HSP90B1 | Forward | CTGGCTCTGGCATGCACGCTTCT | Heat shock protein 90 kDa beta (Grp94), member 1 | NM_204289 |
| Reverse | CTTCATCATCAGTTCGGGACCCTTCTCTAC | |||
| NFKBIZ | Forward | CCAGGTCCTCCAGGCAATCCAAAAG | Nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, zeta | NM_001006254 |
| Reverse | AGTGCAGGGCTGTCAAACCATCGTAG | |||
| SERPINF1 | Forward | CGGCAGCAGACAAGGGGAAGGATT | Serpin peptidase inhibitor, clade F, member 1 | NM_001257289 |
| Reverse | TGAAGTAAGCAGCCCCAGCAAGGAG | |||
| SOCS3 | Forward | GACACCAGCCTGCGCCTCAAGA | Suppressor of cytokine signaling 3 | NM_204600 |
| Reverse | GCCCGTCACCGTGCTCCAGTAGA | |||
| SOCS6 | Forward | CAGATATCTTTGTGGACCAGGCAGTGAA | Suppressor of cytokine signaling 6 | NM_001127312 |
| Reverse | GGTAGCAAAGGTGAAAGTGGAGGGACATC | |||
| TAB3 | Forward | CACCGCAAAGACCTGGGACTG | TGF-beta activated kinase 1/MAP3K7 binding protein 3 | XM_416787 |
| Reverse | GTGGGTGCTGGTTTCGTTGAGATGGT | |||
| TCF12 | Forward | CTCGGGGAAACCTGGAACAACCTACTA | Transcription factor 12 | NM_205375 |
| Reverse | GGGGGCACCTTTCTTACTTTCTTTGTCT | |||
| TNFRSF11B | Forward | CATCTCGGCCAACCAAGTCTCACCT | Tumor necrosis factor receptor superfamily, member 11 b | NM_001033641 |
| Reverse | CGCTCGATATCTTCTTTTCCCACTTTCTTG | |||
| TRAF3 | Forward | CGTCTCGGCGCCACTTAGGA | TNF receptor-associated factor 3 | XM_421378 |
| Reverse | GGGCAGCCAGACGCAATGTTCA | |||
| VEGFA | Forward | GGCCTAGAATGTGTCCCTGTGG | Vascular endothelial growth factor A | NM_001110355 |
| Reverse | ATGTGCGCTATGTGCTGACTCTGA | |||
| LITAF | Forward | TGTGTATGTGCAGCAACCCGTAGT | Lipopolysaccharide-induced TNF factor | AY765397 |
| Reverse | GGCATTGCAATTTGGACAGAAGT | |||
| GAPDH | Forward | TGCTGCCCAGAACATCATCC | Glyceraldehyde-3-phosphate dehydrogenase | NM_204305 |
| Reverse | ACGGCAGGTCAGGTCAACAA |
Statistical analyses
Data from body weight gains, lesion scores, oocyst shedding, and mRNA expression levels were expressed as the mean ± SD values. Comparisons of the mean values were performed by ANOVA and the Student's t-test using SPSS software (SPSS 15.0 for Windows, Chicago, IL) and were considered significant at P<0.05. IPA biological function analysis was performed using the Fisher's exact test to calculate the probability of each biological function assigned to that dataset and were considered significant at P<0.05.
Results
Effects of experimental NE on body weight gain and lesion score
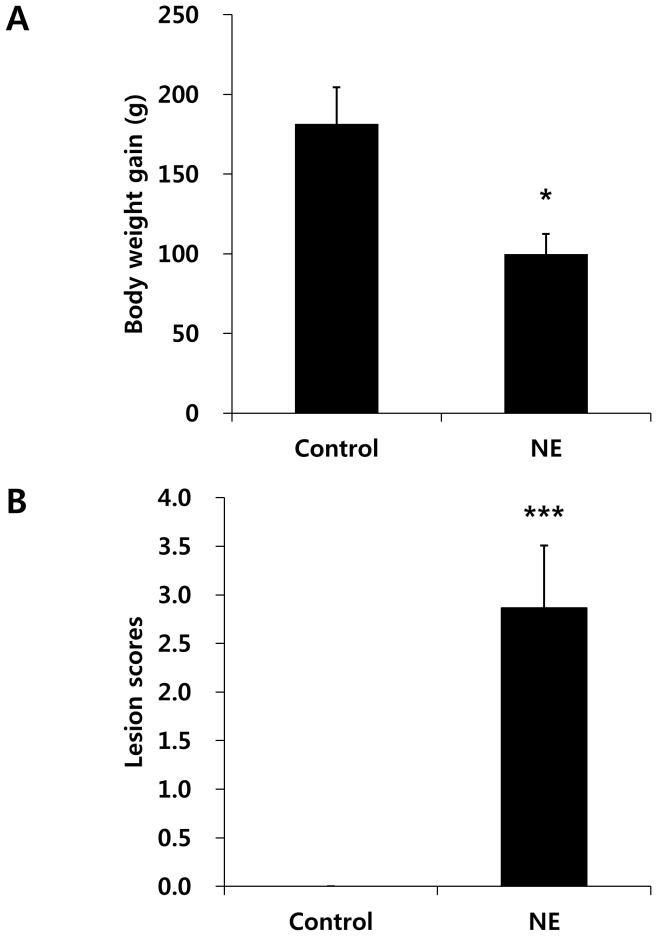
Co-infection of Ross chickens with C. perfringens and E. maxima reduced body weight gain between days 0–9 post-infection with E. maxima by 45% compared with uninfected birds (uninfected gain, 182±22 g; co-infected gain, 100±12 g) (Fig. 1A). Co-infected chickens also had higher gut lesion scores at day 2 post-infection with C. perfringens compared with uninfected chickens (uninfected score 0±0; co-infected score, 2.9±0.6) (Fig. 1B).
Figure 1. Effect of C. perfringens/E. maxima co-infection of Ross chickens on body weight gains and intestinal lesion scores.
(A) Chickens were uninfected (Control) or co-infected with C. perfringens and E. maxima (NE) and body weight gains were measured between days 0–9 post-infection with E. maxima. (B) Gut lesion scores were determined at day 2 post-infection with C. perfringens on a scale from 0 (none) to 4 (high) in a blinded fashion by three independent observers. Each bar represents the mean ± SD value (n = 8). *, P<0.05; ***, P<0.001.
Effects of NE on global gene expression in intestinal IELs
Microarray hybridization analysis identified 1,049 transcripts in intestinal IELs whose levels were significantly altered beyond a cutoff of>2.0-fold at day 2 post-infection with C. perfringens in C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls. Of these, 601 transcript levels were increased and 448 were decreased. This dataset was mapped to the human, mouse, rat, and chicken genomes using Ingenuity Knowledge Base software, leading to the identification and annotation of 462 chicken genes. The most 20 most up-regulated and 20 most down-regulated transcripts in co-infected birds compared with uninfected controls are listed in Table 2.
Table 2. The 20 most up-regulated and 20 most down-regulated intestinal IEL transcript levels in C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls.
| Symbol | Entrez Gene Name | Fold Change | Location | Type(s) |
| Up-regulated | ||||
| MMP7 | Matrix metallopeptidase-7 | 45.476 | Extracellular Space | Peptidase |
| LYG2 | Lysozyme G-like 2 | 35.509 | Extracellular Space | Enzyme |
| VCAM1 | Vascular cell adhesion molecule 1 | 14.508 | Plasma Membrane | Other |
| ATP13A4 | ATPase type 13A4 | 12.263 | Unknown | Transporter |
| BATF3 | Basic leucine zipper transcription factor, ATF-like 3 | 11.368 | Nucleus | Transcription Regulator |
| TAC1 | Tachykinin, precursor 1 | 9.837 | Extracellular Space | Other |
| C3 | Complement component 3 | 9.8 | Extracellular Space | Peptidase |
| SOCS1 | Suppressor of cytokine signaling 1 | 9.571 | Cytoplasm | Other |
| LAG3 | Lymphocyte-activation gene 3 | 9.217 | Plasma Membrane | Transmembrane Receptor |
| GLDC | Glycine dehydrogenase (decarboxylating) | 8.991 | Cytoplasm | Enzyme |
| IL21R | Interleukin 21 receptor | 8.101 | Plasma Membrane | Transmembrane Receptor |
| RRM2 | Ribonucleotide reductase M2 | 7.797 | Nucleus | Enzyme |
| SAMHD1 | SAM domain and HD domain 1 | 7.729 | Nucleus | Enzyme |
| MIS12 | MIS12, MIND kinetochore complex component, homolog (S. pombe) | 7.474 | Nucleus | Other |
| CSF2RA | Colony stimulating factor 2 receptor, alpha, low-affinity (granulocyte-macrophage) | 7.444 | Plasma Membrane | Transmembrane Receptor |
| IRG1 | Immunoresponsive 1 homolog (mouse) | 7.171 | Unknown | Other |
| MDFI | MyoD family inhibitor | 7.161 | Cytoplasm | Other |
| TRAIP | TRAF interacting protein | 7.09 | Cytoplasm | Other |
| SFTPA1 | Surfactant protein A1 | 7.003 | Extracellular Space | Transporter |
| SMC2 | Structural maintenance of chromosomes 2 | 6.872 | Nucleus | Transporter |
| Down-regulated | ||||
| C13orf15 | Chromosome 13 open reading frame 15 | −104.868 | Cytoplasm | Other |
| AGXT2L1 | Alanine-glyoxylate aminotransferase 2-like 1 | −70.677 | Unknown | Enzyme |
| CMBL | Carboxymethylenebutenolidase homolog (Pseudomonas) | −28.026 | Unknown | Enzyme |
| HES6 | Hairy and enhancer of split 6 (Drosophila) | −25.238 | Nucleus | Transcription Regulator |
| RBPMS2 | RNA binding protein with multiple splicing 2 | −22.282 | Unknown | Other |
| S100B | S100 calcium binding protein B | −17.322 | Cytoplasm | Other |
| SERPINF1 | Serpin peptidase inhibitor, clade F (alpha-2 antiplasmin, pigment epithelium derived factor), member 1 | −17.216 | Extracellular Space | Other |
| SERPINB2 | Serpin peptidase inhibitor, clade B (ovalbumin), member 2 | −16.241 | Extracellular Space | Other |
| BBOX1 | Butyrobetaine (gamma), 2-oxoglutarate dioxygenase (gamma-butyrobetaine hydroxylase) 1 | −15.873 | Cytoplasm | Enzyme |
| RHAG | Rh-associated glycoprotein | −13.518 | Plasma Membrane | Peptidase |
| CXCL14 | Chemokine (C-X-C motif) ligand 14 | −13.029 | Extracellular Space | Cytokine |
| MYL10 | Myosin, light chain 10, regulatory | −12.77 | Cytoplasm | Other |
| TMEM199 | Transmembrane protein 199 | −12.112 | Unknown | Other |
| ANXA1 | Annexin A1 | −11.609 | Plasma Membrane | Other |
| NRG4 | Neuregulin 4 | −11.399 | Extracellular Space | Growth Factor |
| ALDH1A1 | Aldehyde dehydrogenase 1 family, member A1 | −10.85 | Cytoplasm | Enzyme |
| RNF152 | Ring finger protein 152 | −10.558 | Cytoplasm | Enzyme |
| COL1A2 | Collagen, type I, alpha 2 | −10.196 | Extracellular Space | Other |
| SYNM | Synemin, intermediate filament protein | −9.059 | Cytoplasm | Other |
| CALB1 | Calbindin 1, 28 kDa | −8.966 | Cytoplasm | Other |
Effects of NE on biological function, pathway, and network analysis of differentially regulated IELs transcripts
Five significantly altered biological functions were identified from the differently altered genes in intestinal IELs from C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls (Table 3). The P value associated with a particular function in this analysis is a statistical measure of the likelihood that genes from the dataset under investigation participate in that function. All of the 5 identified biological functions were related to the immune response, particularly the quantity and movement of leukocytes. The most significant function annotated was “immune response” including a category of “inflammatory response” with 83 genes, followed by “cell movement of leukocytes” including the categories of “cellular movement,” “hematological system development and function,” and “immune cell trafficking” with 49 genes.
Table 3. The biological functions significantly associated with the differentially expressed transcripts in C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls.
| Functions Annotation | Category | P Value | No. of Genes |
| Immune response | Inflammatory Response | 5.93E-12 | 83 |
| Cell movement of leukocytes | Cellular Movement | 1.36E-09 | 49 |
| Hematological System Development and Function | |||
| Immune Cell Trafficking | |||
| Quantity of leukocytes | Hematological System Development and Function | 1.96E-09 | 45 |
| Tissue Morphology | |||
| Cell movement of blood cells | Cellular Movement | 2.21E-09 | 53 |
| Quantity of mononuclear leukocytes | Hematological System Development and Function | 2.52E-09 | 38 |
| Tissue Morphology |
Canonical pathway analysis of the differentially expressed genes in co-infected vs. uninfected chickens identified 11 pathways (Table 4). The most significant of these were “leukocyte extravasation signaling,” “relaxin signaling,” and “LPS/IL-1 mediated inhibition of RXR function”, which were associated with 14, 11, and 13 genes, respectively (Table 5).
Table 4. The IPA pathways significantly associated with the differentially expressed transcripts in C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls.
| Ingenuity Canonical Pathway | P Value | Ratio* |
| Leukocyte Extravasation Signaling | 1.58E-03 | 7.00E-02 |
| Relaxin Signaling | 1.82E-03 | 6.96E-02 |
| LPS/IL-1 Mediated Inhibition of RXR Function | 2.29E-03 | 5.83E-02 |
| Pyruvate Metabolism | 2.82E-03 | 5.19E-02 |
| Neuroprotective Role of THOP1 in Alzheimer's Disease | 2.88E-03 | 9.26E-02 |
| HIF1α Signaling | 2.95E-03 | 8.33E-02 |
| ATM Signaling | 7.08E-03 | 1.11E-01 |
| HMGB1 Signaling | 7.41E-03 | 8.00E-02 |
| Tight Junction Signaling | 8.32E-03 | 6.10E-02 |
| ILK Signaling | 9.33E-03 | 6.22E-02 |
| Hepatic Fibrosis/Hepatic Stellate Cell Activation | 9.77E-03 | 6.80E-02 |
*The ratio of the number of genes from the dataset mapped to the indicated pathway divided by the total number of genes within that particular pathway.
Table 5. Genes included in the three canonical pathways most significantly associated with the differentially expressed transcripts in C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls.
| Pathway | Gene Symbol | Entrez Gene Name | Fold Change | Location | Type(s) |
| Leukocyte Extravasation Signaling | CD99 | CD99 molecule | −4.524 | Plasma Membrane | Other |
| ACTC1 | Actin, alpha, cardiac muscle 1 | −3.201 | Cytoplasm | Enzyme | |
| CTTN | Cortactin | −2.679 | Plasma Membrane | Other | |
| MAPK9 | Mitogen-activated protein kinase 9 | −2.154 | Cytoplasm | Kinase | |
| ARHGAP12 | Rho GTPase activating protein 12 | −2.1 | Cytoplasm | Other | |
| CLDN3 | Claudin 3 | −2.098 | Plasma Membrane | Transmembrane Receptor | |
| FER | Fer (fps/fes related) tyrosine kinase | 2.279 | Cytoplasm | Kinase | |
| PIK3R5 | Phosphoinositide-3-kinase, regulatory subunit 5 | 2.584 | Cytoplasm | Kinase | |
| MMP9 | Matrix metallopeptidase 9 (gelatinase B, 92kDa gelatinase, 92kDa type IV collagenase) | 3.071 | Extracellular Space | Peptidase | |
| NCF2 | Neutrophil cytosolic factor 2 | 3.464 | Cytoplasm | Enzyme | |
| PIK3CD | Phosphoinositide-3-kinase, catalytic, delta polypeptide | 3.775 | Cytoplasm | Kinase | |
| RAC2 | Ras-related C3 botulinum toxin substrate 2 (rho family, small GTP binding protein Rac2) | 3.919 | Cytoplasm | Enzyme | |
| VCAM1 | Vascular cell adhesion molecule 1 | 14.508 | Plasma Membrane | Other | |
| MMP7 | Matrix metallopeptidase 7 (matrilysin, uterine) | 45.476 | Extracellular Space | Peptidase | |
| Relaxin Signaling | MPPE1 | Metallophosphoesterase 1 | 2.005 | Cytoplasm | Enzyme |
| PDE3B | Phosphodiesterase 3B, cGMP-inhibited | 2.18 | Cytoplasm | Enzyme | |
| PIK3CD | Phosphoinositide-3-kinase, catalytic, delta polypeptide | 3.775 | Cytoplasm | Kinase | |
| PIK3R5 | Phosphoinositide-3-kinase, regulatory subunit 5 | 2.584 | Cytoplasm | Kinase | |
| MMP9 | Matrix metallopeptidase 9 (gelatinase B, 92kDa gelatinase, 92kDa type IV collagenase) | 3.071 | Extracellular Space | Peptidase | |
| VEGFA | Vascular endothelial growth factor A | −2.245 | Extracellular Space | Growth Factor | |
| CREB1 | cAMP responsive element binding protein 1 | 2.05 | Nucleus | Transcription Regulator | |
| ADCY5 | Adenylate cyclase 5 | −2.53 | Plasma Membrane | Enzyme | |
| ADCY9 | Adenylate cyclase 9 | −3.136 | Plasma Membrane | Enzyme | |
| GNA11 | Guanine nucleotide binding protein (G protein), alpha 11 (Gq class) | −2.271 | Plasma Membrane | Enzyme | |
| GNG13 | Guanine nucleotide binding protein (G protein), gamma 13 | −3.434 | Plasma Membrane | Enzyme | |
| LPS/IL-1 Mediated Inhibition of RXR Function | ACSBG2 | Acyl-CoA synthetase bubblegum family member 2 | 2.046 | Cytoplasm | Enzyme |
| ACSL5 | Acyl-CoA synthetase long-chain family member 5 | 4.067 | Cytoplasm | Enzyme | |
| ALDH1A1 | Aldehyde dehydrogenase 1 family, member A1 | −10.85 | Cytoplasm | Enzyme | |
| CYP3A4 | Cytochrome P450, family 3, subfamily A, polypeptide 4 | 2.265 | Cytoplasm | Enzyme | |
| FABP5 | Fatty acid binding protein 5 (psoriasis-associated) | −2.642 | Cytoplasm | Transporter | |
| GSTT1 | Glutathione S-transferase theta 1 | −2.622 | Cytoplasm | Enzyme | |
| HMGCS2 | 3-hydroxy-3-methylglutaryl-CoA synthase 2 (mitochondrial) | −6.489 | Cytoplasm | Enzyme | |
| MAPK9 | Mitogen-activated protein kinase 9 | −2.154 | Cytoplasm | Kinase | |
| SMOX | Spermine oxidase | −2.372 | Cytoplasm | Enzyme | |
| HS6ST1 | Heparan sulfate 6-O-sulfotransferase 1 | 2.632 | Plasma Membrane | Enzyme | |
| SLC27A4 | Solute carrier family 27 (fatty acid transporter), member 4 | 2.839 | Plasma Membrane | Transporter | |
| TNFRSF11B | Tumor necrosis factor receptor superfamily, member 11b | −4.19 | Plasma Membrane | Transmembrane Receptor | |
| TNFRSF1B | Tumor necrosis factor receptor superfamily, member 1B | 4.62 | Plasma Membrane | Transmembrane Receptor |
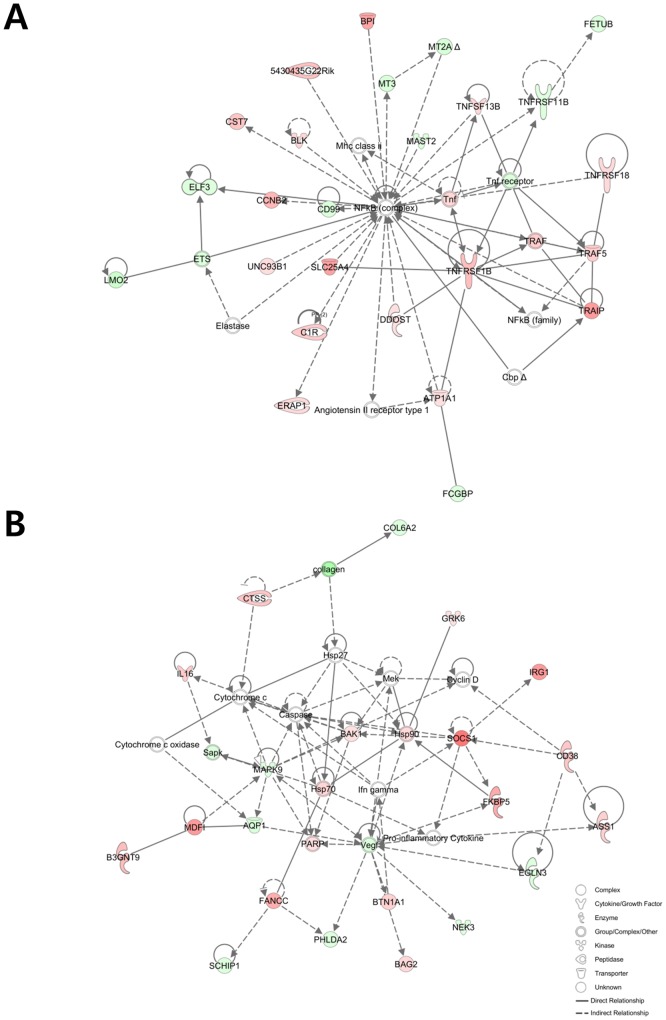
Twenty-five networks related to the differentially expressed genes in co-infected vs. uninfected chickens were identified by IPA network analysis (S1 Table). Two of these networks were related to the immune response, including the most significantly associated network containing 25 focused genes and associated with the biological functions “cellular function and maintenance”, “inflammatory response”, and “cell-to-cell signaling and interaction” (Fig. 2A). The seventh most significantly associated network also was related to the immune response and contained 21 focused genes associated with the biological functions “cell death,” “inflammatory response,” and “organismal injury and abnormalities” (Fig. 2B).
Figure 2. The first (A) and seventh (B) networks of genes most significantly associated with the differentially expressed transcripts in C. perfringens/E. maxima co-infected Ross chickens compared with uninfected controls.
Up- and down-regulated genes are illustrated by red and green, respectively. The color intensity directly correlates with the difference in the expression level of the corresponding gene compared between line 6 and line 7 chickens.
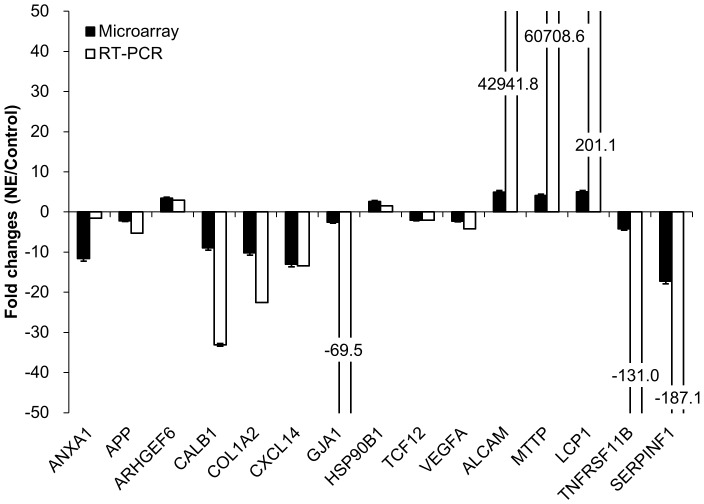
Validation of microarray analysis by qRT-PCR
The transcript expression patterns observed by microarray analysis between C. perfringens/E. maxima co-infected Ross chickens and uninfected controls were validated by qRT-PCR for 15 immune-related mRNAs. The levels of all transcripts were consistently increased or decreased by each method of quantification (Fig. 3). The some cases, differences in the magnitude of the changes observed by the two techniques might be related to differences in the normalization methods used and/or the different fluorescent dyes used [19].
Figure 3. Comparison of the fold changes in the expression levels of 15 immune-related gene transcripts in intestinal IELs of C. perfringens/E. maxima co-infected Ross chickens (NE) compared with uninfected controls (Control).
Transcript levels were analyzed by microarray hybridization and qRT-PCR in both groups. Each bar represents the mean ± SD value of the fold change in the transcript level (n = 3). ANXA1, Annexin A1; APP, Amyloid precursor protein; ARHGEF6, Rho guanine nucleotide exchange factor 6; CALB1, Calbindin 1; COL1A2, Collagen, type 1, alpha 2; CXCL14, Chemokine (C-X-C motif) ligand 14; GJA1, Gap junction alpha 1; HSP90B1, Heat shock protein 90kDa beta 1; VEGFA, Vascular endothelial growth factor A; ALCAM, Activated leukocyte cell adhesion molecule; MTTP, Microsomal triglyceride transfer protein; LCP1, Lymphocyte cytosolic protein 1; TNFSF11B, Tumor necrosis factor superfamily 11; SERPINE1, Serpin peptidase inhibitor, clade F1.
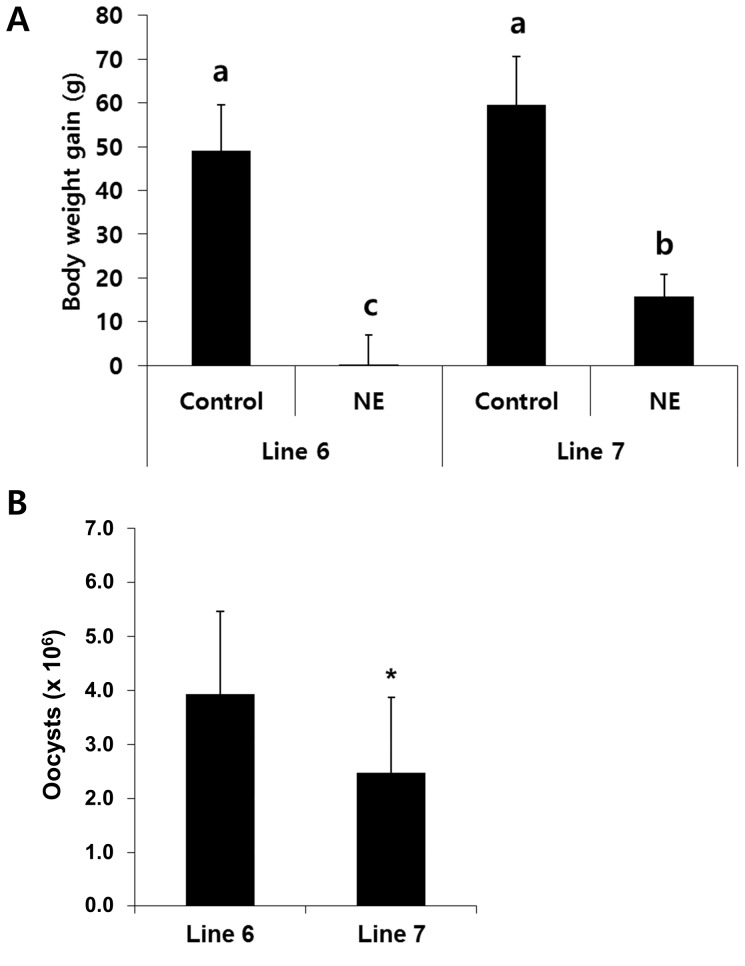
Body weight gain, oocyst shedding, and immune-related gene expression during experimental NE in ADOL line 6 and line 7 chickens
Line 6 chickens had reduced body weight gains between days 0–9 post-infection with E. maxima (line 6 gain, 0.5±7.0 g; line 7 gain, 16±5.0 g) (Fig. 4A). Body weight gains between these same time points were equal in the two lines in uninfected birds. Co-infected line 6 chickens had greater fecal oocyst shedding between days 0–9 post-infection with E. maxima compared with line 7 chickens (line 6 oocyts, 3.9±1.5×106; line 7 oocysts, 2.5±1.4×106) (Fig. 4B). These results suggest that line 6 chickens have increased susceptibility for the development of experimental NE compared with line 7 chickens. To characterize the pattern of immune-related gene expression that might be related to this observed difference in disease susceptibility, the levels of transcripts for 21 selected genes were analyzed by qRT-PCR in uninfected and C. perfringens/E. maxima co-infected line 6 and line 7 chickens. Of these, 15 transcripts were expressed at greater levels in co-infected line 6 chickens compared with line 7 birds, 5 were equal in the two lines, and 1 was decreased in co-infected line 6 vs. line 7 chickens (Table 6).
Figure 4. Effect of C. perfringens/E. maxima co-infection of ADOL line 6 and line 7 chickens on body weight gains and oocyst shedding.
(A) Chickens were uninfected (Control) or co-infected with C. perfringens and E. maxima (NE) and body weight gains were measured between days 0–9 post-infection with E. maxima. Bars with different letters are significantly different according to the Duncan's multiple range test (P<0.05). (B) Fecal oocyst shedding was measured between days 0–9 post-infection with E. maxima in C. perfringens/E. maxima co-infected chickens. Each bar represents the mean ± SD value (n = 8). *, P<0.05.
Table 6. Immune-related intestinal IEL transcript levels in uninfected and in C. perfringens/E. maxima co-infected ADOL line 6 and line 7 chickens.
| Gene | Treatment | ADOL Line 6 | ADOL Line 7 |
| ANXA1 | Uninfected | 3.6E-03±2.5E-04c | 6.5E-03±4.5E-04b |
| NE | 1.0E-02±7.4E-05a | 1.6E-03±9.4E-05bc | |
| ARHGEF6 | Uninfected | 1.9E-03±7.8E-06c | 2.4E-03±6.9E-05b |
| NE | 3.3E-03±1.0E-04a | 2.6E-03±9.9E-05b | |
| LITAF | Uninfected | 2.0E+00±3.6E-01b | 6.7E-01±2.6E-02c |
| NE | 4.8E+00±2.7E-01a | 8.3E-01±3.7E-02c | |
| SOCS3 | Uninfected | 1.3E-08±1.9E-09b | 4.0E-07±1.6E-08b |
| NE | 3.0E-06±1.1E-07a | 9.2E-07±3.5E-08b | |
| BCL2 | Uninfected | 2.7E-04±1.6E-05b | 5.1E-04±2.1E-05a |
| NE | 2.2E-04±7.4E-06b | 1.3E-04±9.3E-06c | |
| GJA1 | Uninfected | 5.5E-05±3.5E-06a | 1.8E-05±3.4E-06b |
| NE | 1.2E-05±3.7E-07b | 1.6E-05±9.7E-07b | |
| IL8 | Uninfected | 1.5E+00±1.0E-01a | 7.2E-01±6.3E-02b |
| NE | 5.7E-01±1.1E-02b | 4.4E-01±6.2E-02b | |
| SOCS6 | Uninfected | 7.5E-04±3.9E-05a | 2.6E-04±6.7E-06b |
| NE | 2.6E-04±9.7E-06b | 2.3E-04±8.1E-06b | |
| HSP90B1 | Uninfected | 1.2E-02±2.1E-04b | 1.3E-02±3.0E-04b |
| NE | 1.6E-02±3.8E-04a | 3.9E-03±8.1E-05c | |
| VEGFA | Uninfected | 2.6E-02±3.6E-04b | 2.8E-02±6.0E-04b |
| NE | 4.1E-02±4.0E-04a | 1.6E-02±3.1E-04c | |
| MTTP | Uninfected | 6.1E-06±1.7E-07bc | 3.1E-06±9.4E-08c |
| NE | 2.9E-05±6.2E-07a | 9.4E-06±7.2E-07b | |
| SERPINF1 | Uninfected | 3.8E-04±2.2E-05b | 1.5E-03±2.7E-05a |
| NE | 7.7E-05±2.1E-06c | 1.3E-04±8.0E-06c | |
| CALB1 | Uninfected | 2.6E+00±7.5E-02a | 7.3E-01±3.5E-02b |
| NE | 3.7E-03±6.0E-05c | 5.7E-02±9.2E-03c | |
| LCP1 | Uninfected | 3.4E-06±2.8E-07c | 2.8E-06±3.5E-07c |
| NE | 3.2E-05±3.8E-07a | 4.6E-05±3.9E-06b | |
| TNFRSF11B | Uninfected | 1.9E-07±9.5E-09a | 1.3E-07±2.3E-08b |
| NE | 1.0E-07±9.2E-09b | 3.3E-08±5.4E-09c | |
| COL1A2 | Uninfected | 2.9E-04±2.8E-05ab | 3.7E-04±2.6E-05a |
| NE | 1.2E-04±7.3E-06c | 2.4E-04±2.7E-05b | |
| TCF12 | Uninfected | 2.3E-04±1.4E-05a | 1.1E-04±1.2E-05b |
| NE | 1.6E-04±2.7E-05b | 6.1E-05±3.5E-06c | |
| APP | Uninfected | 1.9E-02±1.2E-04a | 1.2E-02±3.0E-04b |
| NE | 1.1E-02±3.0E-04b | 5.1E-03±1.4E-04c | |
| CXCL14 | Uninfected | 9.6E-04±5.4E-05a | 2.6E-04±1.5E-05b |
| NE | 3.1E-04±7.7E-05b | 5.2E-05±4.8E-06c | |
| NFKBIZ | Uninfected | 2.4E-03±9.9E-05b | 6.0E-03±1.6E-04a |
| NE | 1.8E-03±4.6E-05c | 1.3E-03±1.5E-05d | |
| TAB3 | Uninfected | 1.9E-03±1.2E-05a | 1.2E-03±1.2E-05b |
| NE | 4.4E-04±1.6E-05c | 2.4E-04±1.1E-05d | |
| TRAF3 | Uninfected | 1.9E-03±3.4E-05a | 6.8E-04±5.1E-06b |
| NE | 6.0E-04±1.9E-05b | 2.5E-04±9.7E-06c |
Each value represents the mean ± SD (n = 3). Within each gene group, values with different superscripts are significantly different according to the Duncan's multiple range test (P<0.05).
Discussion
This study was conducted to investigate the host response to avian NE, and the role of host genetics in this response, using an experimental model of C. perfringens/E. maxima co-infection. Microarray hybridization identified 1,049 transcripts whose levels were altered in intestinal IELs of co-infected Ross chickens compared with uninfected controls, the majority of which (57.3%) were increased. From these differentially expressed genes, 5 biological functions, all related to host immunity, and 11 pathways were identified. ADOL line 6 chickens were more susceptible to NE compared with the MHC-identical line 7, as revealed by decreased body weight gain and increased E. maxima oocyst shedding. Of note, body weight gains of the experimentally inbred ADOL chickens are lower compared with those of Ross broilers which have been specifically bred for rapid, high efficiency growth rate. Of 21 pro- and anti-inflammatory genes examined, 15 were increased in line 6 vs. line 7 chickens. It is interesting to note that most of immune-related transcripts examined (71%) were increased in the more disease susceptible line 6 chickens.
Identification of antibiotic-free disease control strategies against NE has been hindered due, in large part, to the difficulty of experimentally reproducing the disease by C. perfringens infection alone [20], [21]. Nevertheless, several prior studies have profiled chicken transcriptome changes following infection by C. perfringens alone [22], [23]. Functional genomics using the C. perfringens/E. maxima co-infection method offers one possibility to investigate the host response to experimental disease, and to the best of our knowledge, this study is the first report of such an approach. We chose to analyze transcriptome alterations using intestinal IELs because they are the primary immune effector cells of the gut-associated lymphoid tissues which recognize and destroy pathogens that breach the intestinal epithelial barrier [24]. Chicken intestinal IELs are composed of two phenotypically and functionally distinct subpopulations, natural killer cells and T-lymphocytes [25], and both cell types are major effectors against C. perfringens and Eimeria spp. [26].
In addition to infection of the chicken gut, C. perfringens infection also can be manifested as gangrenous dermatitis, a necrotizing skin disease accompanied by severe cellulitis of the subcutaneous tissues [27]. In a previous study, we characterized gene expression profiles in chickens with gangrenous dermatitis and identified “inflammatory response” as the most significantly affected biological functions associated with this disease [27]. Therefore, it was not unexpected to find in the current study that the differentially regulated genes in intestinal IELs from chickens with experimental NE also were associated with biological functions and networks related to “inflammatory response” and “immune response”. Interestingly, all of the remaining significantly annotated biological functions in C. perfringens/E. maxima co-infected Ross birds were related to movement or quantity of leukocytes. Further, the most reliable canonical pathway identified was “leukocytes extravasation signaling”. Leukocyte extravasation from the vasculature into infected interstitial tissues constitutes a key component of the host inflammatory response. Inflammatory cytokines and proteases, particularly matrix metalloproteinases (MMPs), are released by extravasating cells to modify the extracellular matrix [28]–[30]. Our results now establish that both matrix MMP-7 and MMP-9 were up-regulated in intestinal IELs of co-infected birds compared with uninfected controls. Collectively, the results suggest that the movement and/or quantity of leukocytes regulate host resistance to experimental avian NE.
ADOL lines 6 and 7 are homozygous for the MHC B2 haplotype, but differ in MDV and ALV resistance [31], [32]. Line 6 chickens are susceptible to both MDV and ALV infection, whereas line 7 chickens are relatively resistant to infection by these viruses. The current study now extends the differential response of these two lines to experimental NE, where line 6 chickens showed greater susceptibility to C. perfringens/E. maxima co-infection compared with line 7 birds. Of the 21 immune-related transcripts analyzed in line 6 and line 7 birds, 15 were increased in the more disease susceptible line 6 chickens. The levels of 9 of these up-regulated transcripts also were examined in Ross chickens, where 8 were increased following C. perfringens/E. maxima co-infection compared with uninfected controls (ANXA1, HSP90B1, VEGFA, MTTP, TNFSF11B, TCF12, APP, and CXCL14). In humans, annexin A1 (ANXA1) regulates tumor necrosis factor-α (TNF-α)-induced cell proliferative and inflammatory responses [33]. In other human studies, annexin A1 protein acted as a positive regulator of MMP-9 expression, as well as metastatic invasion of breast cancer cells, through activation of NF-κB signaling [34]. The expression of LPS-induced TNF-α factor (LITAF), a transcription factor that regulates inflammation through transcriptional activation of TNF-α [35], [36], also was increased in C. perfringens/E. maxima co-infected line 6 chickens compared with line 7 birds. Counteracting these proinflammatory effects, suppressors of cytokine signaling (SOCS) proteins act in an anti-inflammatory manner by providing a negative feedback loop to attenuate cytokine signaling. Mouse TNF-α, and by inference chicken LITAF, were responsible for the maintenance of SOCS3 expression in vitro and TNF-α deficiency in murine macrophages accelerated the degradation of SOCS3 protein [37]. In the current study, LITAF and SOCS3 were increased in co-infected ADOL line 6 chickens compared with line 7 birds. Therefore, the susceptibility to experimental avian NE might be determined, at least in part, by interactions among ANXA1, LITAF, SOCS, and/or other proteins encoded by genes that are differentially regulated in line 6 vs. line 7 chickens during experimental NE. Future studies are needed to provide additional insights into the host-pathogen interactions in avian NE that might facilitate the development of effective control strategies against field infections by clostridial bacteria.
Supporting Information
Significant networks from the differentially expressed transcripts (>2.0 fold changes, P<0.05) following NE infection compared with uninfected controls.
(XLSX)
Acknowledgments
The authors thank Margie Nichols and Stacy O′Donnell for their significant contribution to this research and Dr. Song Ki Duk for his helpful comments and Dr. Erik P. Lillehoj for his considerable time for revising this manuscript.
Data Availability
The authors confirm that all data underlying the findings are fully available without restriction. All microarray information and data were deposited in GEO (series record number, GSE51154).
Funding Statement
This work was supported by ARS CRIS, partially by grants from the Next-Generation BioGreen 21 Program (No. PJ008084 and No. PJ008196) and the project ‘Investigation of the functional activity and the development of functional foods of Allium hookeri (PJ010490)’, Rural Development Administration, Republic of Korea. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Hermans PG, Morgan KL (2007) Prevalence and associated risk factors of necrotic enteritis on broiler farms in the United Kingdom; a cross-sectional survey. Avian Pathol 36:43–51. [DOI] [PubMed] [Google Scholar]
- 2. Lee K, Lillehoj HS, Li G, Park MS, Jang SI, et al. (2011) Identification and cloning of two immunogenic Clostridium perfringens proteins, elongation factor Tu (EF-Tu) and pyruvate:ferredoxin oxidoreductase (PFO) of C. perfringens. Res Vet Sci 91:e80–86. [DOI] [PubMed] [Google Scholar]
- 3. Songer JG (1996) Clostridial enteric diseases of domestic animals. Clin Microbiol Rev 9:216–234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Van Immerseel F, De Buck J, Pasmans F, Huyghebaert G, Haesebrouck F, et al. (2004) Clostridium perfringens in poultry: an emerging threat for animal and public health. Avian Pathol 33:537–549. [DOI] [PubMed] [Google Scholar]
- 5. Timbermont L, Lanckriet A, Gholamiandehkordi AR, Pasmans F, Martel A, et al. (2009) Origin of Clostridium perfringens isolates determines the ability to induce necrotic enteritis in broilers. Comp Immunol Microbiol Infect Dis 32:503–512. [DOI] [PubMed] [Google Scholar]
- 6. Olkowski AA, Wojnarowicz C, Chirino-Trejo M, Laarveld B, Sawicki G (2008) Sub-clinical necrotic enteritis in broiler chickens: novel etiological consideration based on ultra-structural and molecular changes in the intestinal tissue. Res Vet Sci 85:543–553. [DOI] [PubMed] [Google Scholar]
- 7. Lee KW, Lillehoj HS, Jeong W, Jeoung HY, An DJ (2011) Avian necrotic enteritis: experimental models, host immunity, pathogenesis, risk factors, and vaccine development. Poult Sci 90:1381–1390. [DOI] [PubMed] [Google Scholar]
- 8. Bacon LD, Hunt HD, Cheng HH (2000) A review of the development of chicken lines to resolve genes determining resistance to diseases. Poult Sci 79:1082–1093. [DOI] [PubMed] [Google Scholar]
- 9. Bumstead N, Millard B (1987) Genetics of resistance to coccidiosis: response of inbred chicken lines to infection by Eimeria tenella and Eimeria maxima. Br Poult Sci 28:705–715. [DOI] [PubMed] [Google Scholar]
- 10. Bumstead JM, Bumstead N, Rothwell L, Tomley FM (1995) Comparison of immune responses in inbred lines of chickens to Eimeria maxima and Eimeria tenella. Parasitology 111 (Pt 2):143–151. [DOI] [PubMed] [Google Scholar]
- 11. Jang SI, Lillehoj HS, Lee SH, Lee KW, Lillehoj EP, et al. (2012) Vaccination with Clostridium perfringens recombinant proteins in combination with Montanide ISA 71 VG adjuvant increases protection against experimental necrotic enteritis in commercial broiler chickens. Vaccine 30:5401–5406. [DOI] [PubMed] [Google Scholar]
- 12. Yoo J, Jang SI, Kim S, Cho JH, Lee HJ, et al. (2009) Molecular characterization of duck interleukin-17. Vet Immunol Immunopathol 132:318–322. [DOI] [PubMed] [Google Scholar]
- 13. Lee SH, Lillehoj HS, Park DW, Jang SI, Morales A, et al. (2009) Protective effect of hyperimmune egg yolk IgY antibodies against Eimeria tenella and Eimeria maxima infections. Vet Parasitol 163:123–126. [DOI] [PubMed] [Google Scholar]
- 14. Johnson J, Reid WM (1970) Anticoccidial drugs: lesion scoring techniques in battery and floor-pen experiments with chickens. Exp Parasitol 28:30–36. [DOI] [PubMed] [Google Scholar]
- 15. Min W, Lillehoj HS, Ashwell CM, van Tassell CP, Dalloul RA, et al. (2005) Expressed sequence tag analysis of Eimeria-stimulated intestinal intraepithelial lymphocytes in chickens. Mol Biotechnol 30:143–150. [DOI] [PubMed] [Google Scholar]
- 16. Kim DK, Lillehoj HS, Lee SH, Lillehoj EP, Bravo D (2012) Improved resistance to Eimeria acervulina infection in chickens due to dietary supplementation with garlic metabolites. Br J Nutr 109:76–88. [DOI] [PubMed] [Google Scholar]
- 17. McShane LM, Shih JH, Michalowska AM (2003) Statistical issues in the design and analysis of gene expression microarray studies of animal models. J Mammary Gland Biol Neoplasia 8:359–374. [DOI] [PubMed] [Google Scholar]
- 18.Muller PY, Janovjak H, Miserez AR, Dobbie Z (2002) Processing of gene expression data generated by quantitative real-time RT-PCR. Biotechniques 32 : 1372–1374, 1376, 1378–1379. [PubMed] [Google Scholar]
- 19. Lee PD, Sladek R, Greenwood CM, Hudson TJ (2002) Control genes and variability: absence of ubiquitous reference transcripts in diverse mammalian expression studies. Genome Res 12:292–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Collier CT, Hofacre CL, Payne AM, Anderson DB, Kaiser P, et al. (2008) Coccidia-induced mucogenesis promotes the onset of necrotic enteritis by supporting Clostridium perfringens growth. Vet Immunol Immunopathol 122:104–115. [DOI] [PubMed] [Google Scholar]
- 21. Park SS, Lillehoj HS, Allen PC, Park DW, FitzCoy S, et al. (2008) Immunopathology and cytokine responses in broiler chickens coinfected with Eimeria maxima and Clostridium perfringens with the use of an animal model of necrotic enteritis. Avian Dis 52:14–22. [DOI] [PubMed] [Google Scholar]
- 22. Sarson AJ, Wang Y, Kang Z, Dowd SE, Lu Y, et al. (2009) Gene expression profiling within the spleen of Clostridium perfringens-challenged broilers fed antibiotic-medicated and non-medicated diets. BMC Genomics 10:260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Zhou H, Gong J, Brisbin J, Yu H, Sarson AJ, et al. (2009) Transcriptional profiling analysis of host response to Clostridium perfringens infection in broilers. Poult Sci 88:1023–1032. [DOI] [PubMed] [Google Scholar]
- 24. Neutra MR, Mantis NJ, Kraehenbuhl JP (2001) Collaboration of epithelial cells with organized mucosal lymphoid tissues. Nat Immunol 2:1004–1009. [DOI] [PubMed] [Google Scholar]
- 25. Gobel TW, Kaspers B, Stangassinger M (2001) NK and T cells constitute two major, functionally distinct intestinal epithelial lymphocyte subsets in the chicken. Int Immunol 13:757–762. [DOI] [PubMed] [Google Scholar]
- 26. Lillehoj HS, Trout JM (1996) Avian gut-associated lymphoid tissues and intestinal immune responses to Eimeria parasites. Clin Microbiol Rev 9:349–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kim DK, Lillehoj HS, Lee KW, Jang SI, Neumann AP, et al. (2012) Genome-wide differential gene expression profiles in broiler chickens with gangrenous dermatitis. Avian Dis 56:670–679. [DOI] [PubMed] [Google Scholar]
- 28. Feng ZY, Mo XL, Shao CK, Su ZL (2007) [Expressions of matrix metalloproteinase 9 in mucosal natural killer/T cell and mature T cell lymphomas and its relation to Epstein-Barr virus infection]. Nan Fang Yi Ke Da Xue Xue Bao 27:1338–1340. [PubMed] [Google Scholar]
- 29. Langers AM, Sier CF, Hawinkels LJ, Kubben FJ, van Duijn W, et al. (2008) MMP-2 geno-phenotype is prognostic for colorectal cancer survival, whereas MMP-9 is not. Br J Cancer 98:1820–1823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Wilson CL, Schmidt AP, Pirila E, Valore EV, Ferri N, et al. (2009) Differential Processing of {alpha}- and {beta}-Defensin Precursors by Matrix Metalloproteinase-7 (MMP-7). J Biol Chem 284:8301–8311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Liu HC, Cheng HH, Tirunagaru V, Sofer L, Burnside J (2001) A strategy to identify positional candidate genes conferring Marek's disease resistance by integrating DNA microarrays and genetic mapping. Anim Genet 32:351–359. [DOI] [PubMed] [Google Scholar]
- 32. Yu Y, Zhang H, Tian F, Bacon L, Zhang Y, et al. (2008) Quantitative evaluation of DNA methylation patterns for ALVE and TVB genes in a neoplastic disease susceptible and resistant chicken model. PLoS One 3:e1731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Jia Y, Morand EF, Song W, Cheng Q, Stewart A, et al. (2013) Regulation of lung fibroblast activation by annexin A1. J Cell Physiol 228:476–484. [DOI] [PubMed] [Google Scholar]
- 34. Kang H, Ko J, Jang SW (2012) The role of annexin A1 in expression of matrix metalloproteinase-9 and invasion of breast cancer cells. Biochem Biophys Res Commun 423:188–194. [DOI] [PubMed] [Google Scholar]
- 35. Ji ZZ, Dai Z, Xu YC (2011) A new tumor necrosis factor (TNF)-alpha regulator, lipopolysaccharides-induced TNF-alpha factor, is associated with obesity and insulin resistance. Chin Med J (Engl) 124:177–182. [PubMed] [Google Scholar]
- 36. Zhou J, Yang Z, Tsuji T, Gong J, Xie J, et al. (2011) LITAF and TNFSF15, two downstream targets of AMPK, exert inhibitory effects on tumor growth. Oncogene 30:1892–1900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Dagvadorj J, Naiki Y, Tumurkhuu G, Noman AS, Iftakhar EKI, et al. (2010) Tumor necrosis factor-a augments lipopolysaccharide-induced suppressor of cytokine signalling 3 (SOCS-3) protein expression by preventing the degradation. Immunology 129:97–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Significant networks from the differentially expressed transcripts (>2.0 fold changes, P<0.05) following NE infection compared with uninfected controls.
(XLSX)
Data Availability Statement
The authors confirm that all data underlying the findings are fully available without restriction. All microarray information and data were deposited in GEO (series record number, GSE51154).