Abstract
Essential oils are widely used in pharmaceutical, sanitary, cosmetic, agriculture and food industries for their bactericidal, virucidal, fungicidal, antiparasitical and insecticidal properties. Their anticancer activity is well documented. Over a hundred essential oils from more than twenty plant families have been tested on more than twenty types of cancers in last past ten years. This review is focused on the activity of essential oils and their components on various types of cancers. For some of them the mechanisms involved in their anticancer activities have been carried out.
Keywords: Essential oils, anticancer activity, chemical composition
Introduction
Recognized since ancient times for their medicinal value, but often considered as a relic of medieval medical practice by representatives of modern medicine, essential oils (EOs) are currently receiving therapeutic interest fully renewed. Thus, during recent years, plant EOs have come more into the focus of phytomedicine [1,2]. Their widespread use has raised the interest of scientists in basic research of EOs. Especially, anti-microbial and anti-oxidant activities as well as potential anti-cancer activity have been investigated in recent years [3,4].
Cancer is the second largest single cause of death claiming over six million lives every year worldwide [5]. There has been a recent upsurge in the use of natural products to supersede current treatment in patients that develop multidrug resistance. Scientific studies of plants used in various types of ethnic medicine has led to the discovery of many valuable drugs, including taxol, camptothecin, vincristine and vinblastine [6,7]. Many studies pointed out anticancer properties of other plants [8-11]. Over five hundred papers have been published on anticancer activity of EOs. The first publications on the anticancer activity of essential oils dated to 1960s. So far, the effects of EOs have been investigated on glioblastoma, melanoma, leukemia and oral cancers, as well as on bone, breast, cervix, colon, kidney, liver, lung, ovary, pancreas, prostate, and uterus cancers.
The aim of this review is to state the work carried out on the anticancer properties of EOs, their mode of action and the types of cancers targeted.
Essential oils, a mix of complex molecules
EOs are natural, complexe, volatile, and odorous molecules synthesized by the secretory cells of aromatic plants [12]. Also known as volatile oils, EO could be considered as a generic term for the liquid and highly volatile components of plants, with a strong and characteristic odor. Altogether EOs are the concentration of hydrophobic liquid containing multiple volatile aroma compounds found in glands located in various parts of the aromatic plants: leaves, flowers, fruit, seeds, barks and roots.
Even though various methods could be used for their extraction, hydrodistillation remains the most used extraction method to obtain EOs, especially for commercial and medicinal purposes [13]. EOs can also be obtained by cold [14], liquid carbon dioxide at low temperature and high pressure, or ultrasound-assisted extraction or microwave [15]. Usually color less or pale yellow, EOs are volatile, flammable and odorous, and their density is generally less than 1 [16], except for cinnamon, cloves and sassafras. Insoluble in water and soluble in alcohols, oils and petrolatum, which explains the term “oil”, they could be rapidly oxidized and isomerized by light [16].
Even though over 300 different compounds could be identified, three main groups of compounds have been described [17]. While the main group is composed of terpenes and terpenoids, the others include aromatic (phenolic) components, and in a lower extent aliphatic (alkanes and alkenes) compounds are generally in trace. All compounds are characterized by a low molecular weight [18].
Terpenes and terpenoids
About 30 000 terpenes have been described in the literature [19,20]. Some of the most common have been represented in Figures 1, 2. Their basic structure follows a general principle: 2-methylbutane, also refered as isoprene residues build up the carbon skeleton of terpenes (C5)n, as described by Ruzicka [21]. They are subdivided according to the number of isoprene units in monoterpenes (C10H16), sesquiterpenes (C15H24), diterpenes (C20H32) and tetraterpenes, which contain eight units of isoprene such as carotenoids [22]. Terpenoids (Figure 3) are terpenes-derived compounds with one or more chemical functional groups (alcohol, aldehyde, ketone, acid…).
Figure 1.
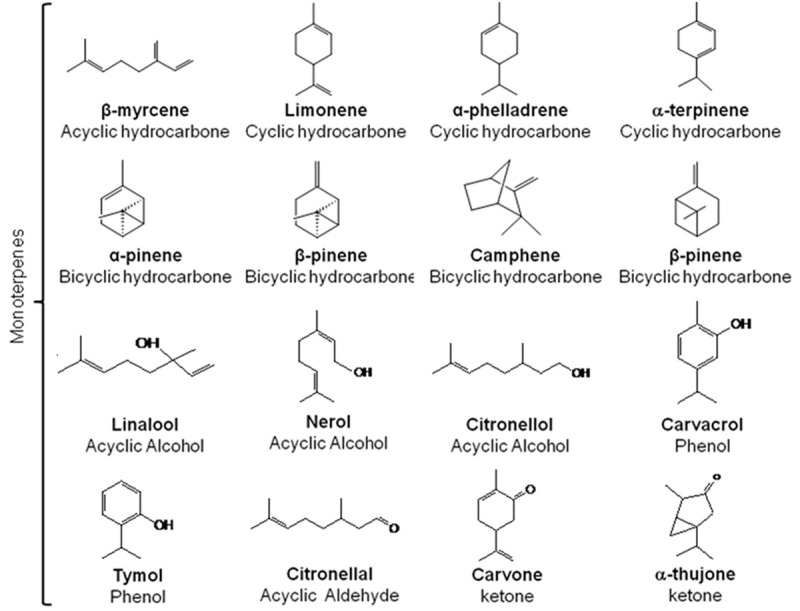
Examples of some monoterpenes compounds found in essential oils of plants.
Figure 2.
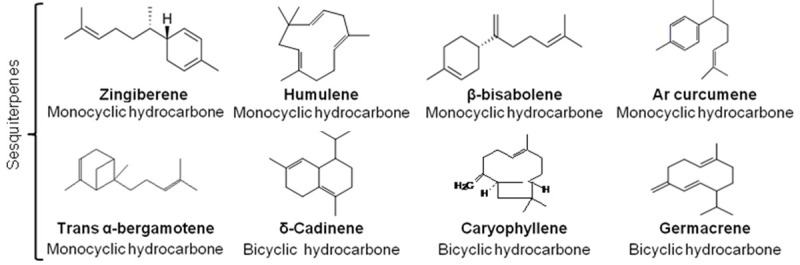
Examples of some sesquiterpenes compounds found in essential oils of plants.
Figure 3.
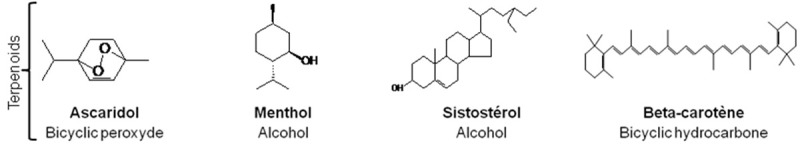
Examples of some terpenoids compounds found in essential oils of plants.
Aromatic compounds
Aromatic compounds (Figure 4) are phenyl propane derivatives. They are less abundant than terpenoids. Two classes of aromatic compounds can been distinguished: the nuclear substituted compounds and derivatives of benzene in which the substituent is directly attached to the benzene ring; the side chain substituted compounds.
Figure 4.
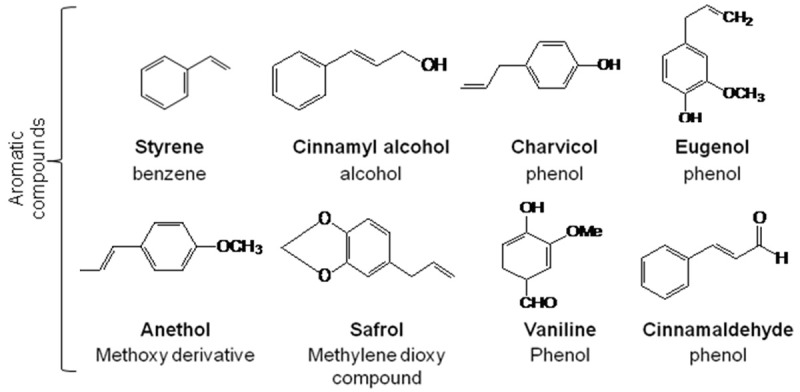
Examples of some aromatic compounds found in essential oils of plants.
Plant essential composition varies according to its environmental and living conditions
According to environmental and living conditions, the same species may show intraspecific chemical differences in its EO compositions [23,24]. These intraspecific differences are defined as chemotypes.
Effects of EOs on various types of cancer
Most of EOs have been first identified and used for the treatment of inflammatory and oxidative diseases. It appeared that these EOs could also have anticancer effects as there is a relationship between the production of reactive oxygen species to the origin of oxidation and inflammation that can lead to cancer [25]. Initial experiments assumed that oxidative stress could act as a DNA-damaging agent, effectively increasing the mutation rate within cells and thus promoting oncogenic transformation. Besides, reactive oxygen species could also specifically activate signaling pathways and thus contribute to tumor development through the regulation of cellular proliferation, angiogenesis, and metastasis [26]. Hence chronic inflammation has been linked to various steps involved in carcinogenesis, such as cellular transformation, promotion, survival, proliferation, invasion, angiogenesis, and metastasis [27]. Several studies have thus shown that EOs and their components therein could be active against various cancer cells (Table 1).
Table 1.
Summary of literature review (2004-2014)
| Cancer | Essential oil or compound tested | Plant family | Cell line used | Reference |
|---|---|---|---|---|
| Brain | Croton regelianus and ascaridole compound | Euphorbiaceae | SF-295 (IC50 = 48.0 μg/ml and 8.4 μg/ml respectively) | [97] |
| Brain (glioblastoma) | Afrostyrax lepidophyllus and Scorodophloeus zenkeri | Huaceae and Caesalpiniaceae (Fabaceae) | T98G (IC50 = 15.4 μg/ml and 12.4 μg/ml respectively) | [10] |
| Brain (glioblastoma) | α-Bisabolol | --- | T67 and C6: 50% of cell death after 24 h treatment with 2.5 µM | [94] |
| Brain (glioblastoma) | Casearia sylvestris | Salicaceae | U87 (IC50 = 27.1 μg/ml) | [42] |
| Brain (glioblastoma) | O. basilicum, L. multiflora, A. conizoides, and Z. officiniale | Lamiaceae, Verbenaceae, Asteraceae and Zingiberaceae respectively | SF-767 (0.30, 0.31, 0.43 and 0.48 mg/ml respectively) and SF-763 (0.43, 0.47, 0.38 and 0.44 respectively) | [36] |
| Brain (glioma) | Malus domestica | Rosaceae | C-6 (1 mg/ml = 58.5% inhibition) | [51] |
| Breast | Afrostyrax lepidophyllus and Scorodophloeus zenkeri | Huaceae and Caesalpiniaceae (Fabaceae) | MDA-MB 231 (IC50 = 10.9 μg/ml and 8.0 μg/ml, respectively) | [10] |
| Breast | Satureja khuzistanica | Lamiaceae | MCF7 (IC50 = 125 μg/ml) | [8] |
| Breast | Casearia sylvestris | Salicaceae | MCF-7 (IC50 = 42.2 μg/ml) | [42] |
| Breast | Cedrelopsis grevei | Rutaceae | MCF-7 (IC50 = 21.5 mg/L) | [43] |
| Breast | Solanium spirale Roxb. | Solanaceae | MCF-7 (IC50 = 19.69 μg/ml) | [44] |
| Breast | carbazole alkaloids | --- | MCF-7 (IC50 = 2.12 μg/ml) | [98] |
| Breast | Helichrysum gymnocephalum | Asteraceae | MCF-7 (IC50 = 16 μg/ml) | [99] |
| Breast | Pituranthos tortuosus (Desf.) | Apiaceae | MCF-7 (IC50 = 3.38 μg/ml) | [54] |
| Breast | Melaleuca armillaris | Myrtaceae | MCF-7 (IC50 = 12 μg/ml) | [100] |
| Breast | Rosmarinus officinalis | Lamiaceae | MCF-7 (IC50 = 190.1 μg/ml) | [101] |
| Breast | Schinus molle L. and Schinus terebinthifolius Raddi | Anacardiaceae | MCF-7 (IC50 = 54 mg/ml and 47 mg/ml, respectively) | [102] |
| Breast | Erigeron acris L. | Asteraceae | MCF-7 (IC50 = 14.5 μg/ml) | [103] |
| Breast | Aquilaria sinensis (Lour.) Gilg. | Thymelaeaceae | MCF-7 (99.6% inhibition at 500 μg/ml) | [104] |
| Breast | Thymus vulgaris L. | Lamiaceae | MCF-7 (IC50 = 0.030% (v/v)) | [32] |
| Breast | Aristolochia mollissima rhizome and the aerial part | Aristolochiaceae | MCF-7 (IC50 = 20.6 and 21.1 μg/ml respectively) and MDA-MB-435S (IC50 = 22.1 and 20.3 μg/ml respectively) | [59] |
| Breast | Schefflera heptaphylla (L.) | Araliaceae | MCF-7 (IC50 = 7.3 μg/ml) | [72] |
| Breast | β-caryophyllene oxide | --- | MDA-MB-231 | [78] |
| Breast (mouse) | Angelica archangelica fruits from separate locations A, B and C | Apiaceae | Crl (IC50 = 47.7; 91.8 and 63.6 μg/ml respectively) | [71] |
| Cervix | Liquidambar styraciflua leaf and stem | Hamamelidaceae | HeLa (IC50 = 136.27 and 119.78 μg/ml respectively) | [56] |
| Cervix | Ocimum basilicum Linn. | Lamiaceae | HeLa (IC50 = 90.5 μg/ml) | [105] |
| Cervix | carbazole alkaloids | --- | HeLa (IC50 = 1.98 μg/ml) | [98] |
| Cervix | Aristolochia mollissima rhizome and the aerial part | Aristolochiaceae | HeLa (IC50 = 38.6 and 50.6 μg/ml respectively) | [59] |
| Ovary | D-Limonene | --- | V79 | [77] |
| Ovary | Malus domestica | Rosaceae | CHOK1 (1000 µg/ml = 68.3% inhibition) | [51] |
| Colon | Kadsura longipedunculata | Schisandraceae | SW-480 (IC50 = 136.62 µg/ml) | [70] |
| Colon | Comptonia peregrina (L.) | Myricaceae | DLD-1 (IC50 = 46 μg/ml) | [106] |
| Colon | Satureja khuzistanica | Lamiaceae | SW480 (IC50 = 62.5 μg/ml) | [8] |
| Colon | 1,8 cineol | --- | HCT116 and RKO | [9] |
| Colon | Artemisia indica | Asteraceae | Caco-2 (IC50 = 19.5 μg/ml) | [11] |
| Colon | Pituranthos tortuosus (Desf.) | Apiaceae | HCT116 (IC50 = 1.34 μg/ml) | [54] |
| Colon | Croton regelianus and ascaridole compound | Euphorbiaceae | HCT-8 (IC50 = 40.0 μg/ml and 18.4 μg/ml respectively) | [97] |
| Colon | Cymbopogon flexuosus | Poaceae | 502713 (IC50 = 4.2 µg/ml) | [107] |
| Colon | Eugenol | --- | SNU-C5 (IC50 = 129.4 μM) | [89] |
| Colon | Geraniol and 5-fluorouracil | --- | Caco-2 (IC50 = 250 and 0.4 µM respectively) and SW620 (IC50 = 330 and 2.0 µM respectively) | [96] |
| Colon | Afrostyrax lepidophyllus and Scorodophloeus zenkeri | Huaceae and Caesalpiniaceae (Fabaceae) | HCT116 (IC50 = 12.4 μg/ml and 8.5 μg/ml respectively) | [10] |
| Colon | Athanasia brownii Hochr. | Asteraceae | HCT 116 (IC50 = 29.53 μg/ml) | [40] |
| Kidney | Platycladus orientalis and Prangos asperula | Cupressaceae and Apiaceae | ACHN (IC50 = 121.93 and 139.17 µg/ml respectively) | [5] |
| Kidney | Laurus nobilis | Lauraceae | ACHN (IC50 = 78.24 μg/ml) | [67] |
| Kidney | Aristolochia mollissima rhizome and the aerial part | Aristolochiaceae | ACHN (IC50 = 22.3 and 33.8 μg/ml respectively) | [59] |
| Kidney | Satureja khuzistanica | Lamiaceae | Vero (IC50 = 31.26 μg/ml) | [8] |
| Leukaemia | Cymbopogon flexuosus and isointermedeol | Poaceae | HL-60 (IC50 = 30 μg/ml and 20 μg/ml, respectively) | [80] |
| Leukaemia | Casearia sylvestris | Salicaceae | HL-60 (IC50 = 29 μg/ml) | [42] |
| Leukaemia | Artemisia indica | Asteraceae | THP-1 (IC50 = 10 μg/ml) | [11] |
| Leukaemia | Malus domestica | Rosaceae | THP-1 (1000 µg/ml = 68.3% inhibition) | [51] |
| Leukaemia | carbazole alkaloids | --- | P388 (IC50 = 5.00 μg/ml) | [98] |
| Leukaemia | Croton regelianus and ascaridole compound | Euphorbiaceae | HL-60 (IC50 = 22.2 μg/ml and 6.32 μg/ml respectively) | [97] |
| Leukaemia (Promyelocytic) | Eugenol | --- | HL-60 (IC50 = 23.7 μM) | [89] |
| Leukaemia (mouse) | Ocimum basilicum L | Lamiaceae | P388 (IC50 = 0.0362 mg/ml) | [108] |
| Liver | Schefflera heptaphylla (L.) Frodin | Araliaceae | HepG2 (IC50 = 6.9 μg/ml) | [72] |
| Liver | Curcuma wenyujin | Zingiberaceae | HepG2 (IC50 = 70 μg/ml) | [109] |
| Liver | Curcuma zedoaria (Berg.) Rosc. | Zingiberaceae | SMMC-7721 (IC50 = 30.7 μg/ml) | [110] |
| Liver | Patrinia scabra Bunge | Caprifoliaceae | Bel-7402 (IC50 = 16 μg/ml) | [52] |
| Liver | Eugenol | --- | HepG2 (IC50 = 118.6 μM) and U-937 (IC50 = 39.4 μM) | [89] |
| Liver | Thymus citriodorus | Lamiaceae | HepG2 (IC50 = 0.34% v/v) | [53] |
| Liver | Artemisia indica | Asteraceae | HEP-2 (IC50 = 15.5 μg/ml) | [11] |
| Liver | Pituranthos tortuosus (Desf.) | Apiaceae | HEPG2 (IC50 = 1.67 μg/ml) | [54] |
| Liver | Kadsura longipedunculata | Schisandraceae | HepG2 (IC50 = 136.96 µg/ml) | [70] |
| Liver | Aristolochia mollissima rhizome and the aerial part | Aristolochiaceae | Bel-7402 (IC50 = 33.1 and 49.5 μg/ml respectively) and Hep G2 (IC50 = 33.2 and 40.7 μg/ml respectively) | [59] |
| Lung | Artemisia indica | Asteraceae | A-549 (IC50 = 25 μg/ml) | [11] |
| Lung | Tridax procumbens | Asteraceae | B16F-10 in vitro (70.2% of inhibition for 50 µg) and in vivo | [61] |
| Lung (small cell) | Solanium spirale Roxb. | Solanaceae | NCI-H187 (IC50 = 24.02 μg/ml) | [44] |
| Lung | Malus domestica | Rosaceae | A549 (1000 µg/ml = 60.7% inhibition) | [51] |
| Lung | Thymus vulgaris L. | Lamiaceae | A549 (IC50 = 0.011% (v/v)) | [32] |
| Lung | Comptonia peregrina (L.) | Myricaceae | A-549 (IC50 = 66 μg/ml) | [106] |
| Lung | Xylopia frutescens Aubl. | Annonaceae | NCI-H358M (IC50 = 24.6 μg/ml) | [60] |
| Mouth epidermal carcinoma | Psidium guajava L | Myrtaceae | KB (IC50 = 0.0379 mg/ml) | [108] |
| Multiple myeloma | β-caryophyllene oxide | --- | U266 and MM1.S | [78] |
| Nasopharyngeal cancer | Centipeda minima | Asteraceae | CNE (IC50 = 5.2 μg/ml after 72 hrs) | [111] |
| Neuroblastoma | Cymbopogon flexuosus | Poaceae | IMR-32 (IC50 = 4.7 µg/ml) | [107] |
| Oral cancer | Solanium spirale Roxb. | Solanaceae | KB (IC50 = 26.42 μg/ml) | [44] |
| Oral cancer | Salvia officinalis | Lamiaceae | UMSSC1 (IC50 = 135 μg/ml) | [112] |
| Oral cancer | Levisticum officinale | Apiaceae | HNSCC (IC50 = 292.6 μg/ml) | [65] |
| Ovary | Patrinia scabra Bunge | Caprifoliaceae | HO-8910 (IC50 = 21 μg/ml) | [52] |
| Ovary | Cymbopogon citratus | Poaceae | Chinese Hamster Ovary (CHO) | [50] |
| Pancreas | Angelica archangelica fruits from separate locations A, B and C | Apiaceae | PANC-1 (IC50 = 58.4; 108.3 and 48.6 μg/ml respectively) | [71] |
| Pancreas | Kadsura longipedunculata | Schisandraceae | MIA PaCa-2 (IC50 = 133.53 µg/ml) | [70] |
| Prostate | Xylopia frutescens Aubl | Annonaceae | PC-3M (IC50 = 40 μg/ml) and in vivo at 37.5% of inhibition | [60] |
| Prostate | Nagami kumquats | Rutaceae | LNCaP (200 ppm = 55, 61 and 63.4 % inhibition at 24, 48, 72 h | [113] |
| Prostate | Rosmarinus officinalis | Lamiaceae | LNCaP (IC50 = 180.9 μg/ml) | [101] |
| Prostate | a-humulene | --- | LNCaP (IC50 = 11.24 μg/ml) | [67] |
| Prostate | β-caryophyllene oxide | --- | DU145 | [78] |
| Prostate | Thymus vulgaris L. | Lamiaceae | PC-3 (IC50 = 0.010% (v/v)) | [32] |
| Prostate | O. basilicum, L. multiflora, A. conizoides, and Z. officiniale | Lamiaceae, Verbenaceae, Asteraceae and Zingiberaceae respectively | LNCaP (0.46, 0.58, 0.35 and 0.38 mg/ml respectively) and PC3 (0.45, 0.30, 0.49 and 0.42 respectively) | [36] |
| Skin (melanoma) | Athanasia brownii Hochr. | Asteraceae | A375 (IC50 = 19.85 μg/ml) | [40] |
| Skin (melanoma) | Afrostyrax lepidophyllus and Scorodophloeus zenkeri | Huaceae and Caesalpiniaceae (Fabaceae) | A375 (IC50 =20.6 μg/ml and 17.7 μg/ml respectively) | [10] |
| Skin (melanoma) | Casearia sylvestris | Salicaceae | A2058 (IC50 = 41.1 μg/ml) | [42] |
| Skin (melanoma) | Curcuma zedoaria (Berg.) Rosc. | Zingiberaceae | B16BL6 (IC50 = 41.8 μg/ml) | [110] |
| Skin (melanoma) | Croton regelianus and ascaridole compound | Euphorbiaceae | MDA-MB-435 (IC50 = 47.3 μg/ml and 10.5 μg/ml respectively) | [97] |
| Skin (melanoma) | Schefflera heptaphylla (L.) | Araliaceae | A375 (IC50 = 7.5 μg/ml) | [72] |
| Skin (amelanotic) | Cupressus sempervirens ssp. pyramidalis | Cupressaceae | C32 (IC50 = 104.90 µg/ml) | [5] |
| Skin (amelanotic) | Laurus nobilis | Lauraceae | C32 (IC50 = 75.45 μg/ml) | [67] |
| Stomach | Nigella sativa seeds | Ranunculaceae | SCL, SCL-6, SCL-37’6, NUGC-4 and Kato-3 (IC50 = 155.02; 185.77; 120.40; 384.53 and 286.83 respectively) | [114] |
| Uterus | Casearia sylvestris | Salicaceae | Siha (IC50 = 23.9 μg/ml) | [42] |
Prostate cancer
EO of Hypericum hircinum L. subsp. Majus revealed antiproliferative activity on human prostatic adenocarcinoma (PC3) [28]. Jacaric acid and four of its octadecatrienoic geoisomers selectively induced apoptosis in hormone-dependent (LNCaP) and -independent (PC-3) human prostate cancer cells, whilst not affecting the viability of normal human prostate epithelial cells (RWPE-1) [29]. Pinus wallichiana EO showed significant anti-proliferative activity on prostate cancer cells [30]. Solanum erianthum leaf volatile oil demonstrated potent inhibitory activity against PC-3 cells [31]. Thymus vulgaris L. EO exhibited the strongest cytotoxicity towards three human cancer cells. Its half inhibitory concentration (IC50) value on PC-3 tumor cell line was 0.010% (v/v) [32]. EO of Mentha arvensis showed cytotoxic activity on LNCaP cells [33]. Oxidative stress results from an imbalance in the production of reactive oxygen species (ROS) and cell own antioxidant defenses that in part leads to numerous carcinogenesis. Kim et al. [34] have shown that saponins contained in EO of ginger reduces the incidence of prostate cancer by exerting antimutagenic activity and also inhibits tumor metastasis. Guatteria pogonopus leaves showed significant in vitro and in vivo antitumor activity on PC-3M metastatic prostate carcinoma [35]. EOs from Ageratum conyzoides Linnæus and Lippia multiflora Moldenk were the most active on LNCaP and PC-3 cell lines [36].
Glioblastoma
EO of Hypericum hircinum had antiproliferative activity on human glioblastoma tumor cells T98G [28]. It increases cytosolic Ca2+ concentrations and alters the viability of human glioblastoma cells by inducing apoptosis [37]. Zanthoxylum tinguassuiba EO contains α-bisabolol, a known antiglioma sesquiterpene, among other potentially active substances [38]. It was observed that thermal-oxidative stability of the liposomal Z. tinguassuiba EO was enhanced when compared to its free form. The liposomal form also presented significant apoptotic-inducing activity for glioma cells. These results show that this EO could be a potential alternative for gliobastoma treatment [38]. The results of the studies whose the objective was to examine the augmentation of the therapeutic activity in human glioblastoma cells with combination of paclitaxel (PTX) and the apoptotic signaling molecule, C6-ceramide (CER), show that PTX and CER can be used together to enhance therapeutic activity, especially in aggressive tumor models such as glioblastoma [39]. A recent study has showed that SF-767 glioblastoma cell line was the most sensitive to Ocimum basilicum Linnæus and Lippia multiflora Moldenk EOs, while essential oil of Ageratum conyzoides Linnæus showed the highest antitumoral activity on SF-763 cells [36].
Melanoma
EOs of Afrostyrax lepidophyllus and Scorodophloeus zenkeri exhibited a strong growth-inhibitory effect on human malignant melanoma A375 cell line [10]. The EO obtained from hydrodistillation of flowering aerial parts of Athanasia brownii also showed significant effect on A375 cells [40]. EOs from the leaves of Neolitsea variabillima [41] and Casearia sylvestris [42] had cytotoxic activity against human melanoma cancer.
Breast cancer
EOs of A. lepidophyllus and S. zenkeri inhibited the growth of human breast adenocarcinoma MDA-MB 231 cell line [10]; likewise, EOs extracted from leaves of Satureja khuzistanica Jamzad [8], Casearia sylvestris [42], Cedrelopsis grevei [43] and Solanium spirale Roxb [44] significantly reduced cell viability and/or increased cytotoxicity of MCF7 cells in a dose-dependent manner. Human breast cancer cell lines T47D, MCF7, MDA-MB-231 were sensitive to the treatment with Boswellia sacra EO with reduced cell viability and elevated cell death [45]. S. erianthum leaf volatile oil demonstrated potent inhibitory activity against human breast Hs 578T tumor cells [31].
Colon cancer
Geraniol, a monoterpene found in EOs of various fruits and herbs has been proposed to represent a new class of agents for cancer chemoprevention, as it has antiproliferative activity on Caco-2 colon cancer cells [46]. EOs of A. lepidophyllus, S. zenkeri [10] and Athanasia brownii exhibited a strong growth-inhibitory effect on human colon carcinoma HCT116 cell line [40]. EO isolated from the leaf of Neolitsea variabillima exhibited cytotoxic activity against human colon cancer [41]. EO from Satureja khuzistanica significantly reduced cell viability of SW480 cell line in a dose-dependent manner [8]. Volatile oil was obtained from blood oranges showed pro-apoptotic and anti-angiogenesis potential on colon cancer cells [47]. EO of Artemisia campestris exhibited significant antitumor activity against the HT-29 cells of colon cancer deserve further research into the chemoprevention and treatment [48]. Thymoquinone inhibited the proliferation of a panel of human colon cancer cells (Caco-2, HCT-116, LoVo, DLD-1 and HT-29), without exhibiting cytotoxicity on normal human intestinal FHs74Int cells [49].
Ovary cancer
EO of Cymbopogon citratus was toxic against Chinese Hamster Ovary cells [50]. Guatteria pogonopus leaves EO showed significant in vitro and in vivo antitumor activity on ovarian adenocarcinoma OVCAR-8 [35]. The essential oil of leaves of Malus domestica at 1000 µg/ml has resulted 68.3% of inhibition of CHOK1 cells [51]. The volatile oil from the roots of Patrinia scabra Bunge showed the strongest inhibitory effect on human ovarian carcinoma cells HO-8910 [52].
Liver cancer
EOs from Thymus citriodorus [53], Artemisia indica [11] and Pituranthos tortuosus (Desf.) [54] leaves have strong toxic effects on liver cancer cells HepG2. Su et al. [41] showed that EO isolated from the leaf of N. variabillima exhibited cytotoxic activity against human liver cancer. Zanthoxylum schinifolium essential oils induced apoptosis of human hepatoma HepG2 cell line is dependent of the production of ROS but not on caspase activation [55].
Uterus and cervix cancers
Leaves of Casearia sylvestris showed cytotoxic activity on uterus carcinoma Siha cell line [42]. The leaf and stem EOs of Liquidambar styraciflua L. induce low cytotoxic activity on cervix cancer cells HeLa [56]. An identical effect was observed with Schinus terebinthifolius Raddi [57]. Furanodiene, a sesquiterpene extracted from the essential oil of the rhizome of Curcuma wenyujin, inhibited the growth of uterine cervical (U14) tumors in mice [58]. The rhizome and the aerial part of Aristolochia mollissima has a significantly stronger cytotoxicity on human cervix carcinoma cell line HeLa [59].
Lung cancer
The Xylopia frutescens leaf EO of displayed in vitro and in vivo cytotoxicity on bronchoalveolar lung carcinoma cell line NCI-H358M [60]. In vivo activity was shown by EOs of X. frutescens [60], Guatteria pogonopus [35] and Neolitsea variabillima [41]. Investigations showed significant effects of the EO of Tridax procumbens L in preventing lung metastasis by B16F-10 cell line in C57BL/6 mice [61]. EO from the aerial parts of A. indica had concentration dependent growth inhibition of A-549 cell line [11]. Vapor of volatile oil compounds obtained from Litsea cubeba seeds killed human NSCLC cells, A549, through the induction of apoptosis and cell cycle arrest [62]. Vapor generated from the combined oils deactivated Akt, a key player in cancer cell survival and proliferation, and Mdm2, which induced overexpression of p53 which in turn upregulated p21 expression [62]. EO of Solanium spirale Roxb. leaves showed significant cytotoxicity against NCI-H187 cells [44].
Oral cancer
EO isolated from the leaf of N. variabillima exhibited cytotoxic activity against human oral cancer [63]. EO of the leaves of Solanium spirale Roxb. showed significant cytotoxicity against KB cell line [44]. Treatment with Pinus densiflora leaf EO at 60 µg/ml strongly inhibited proliferation and survival of YD-8 oral squamous cell carcinoma by apoptosis [64]. Indeed, this treatment led to the activation of caspase-9, PARP cleavage, down-regulation of Bcl-2, and phosphorylation of ERK-1/2 and JNK-1/2 in YD-8 cells [64]. Salvia officinalis EO reduced UMSSC1 cell viability by regulating the aryl hydrocarbon receptor signaling, cell cycle (G1/S checkpoint) transition, and p53 signaling [65]. Levisticum officinale EO inhibits human HNSCC growth by modulating extracellular signal-regulated kinase 5 (ERK5), integrin-linked kinase (ILK), virus entry via endocytic pathways and p53 pathway [65].
Leukemia
Leaves of N. variabillima [41] and Casearia sylvestris [42] showed cytotoxic activity on leukemia HL-60 cell line while EO from A. indica exhibited concentration dependent growth inhibition of THP-1 cell line [11]. Juniperus excelsa fruit essential oil as well as Juniperus oxycedrus, Cedrus libani, and Pinus pinea wood EOs showed cytotoxic activity against drug-sensitive CCRF-CEM and multidrug-resistant P-glycoprotein-expressing CEM/ADR5000 leukemia [66]. EO from Malus domestica leaves at 1000 µg/ml has resulted 65.7% of inhibition of human acute monocytic leukemia cell THP-1 [51].
Kidney cancer
Satureja khuzistanica significantly reduced cell viability of Vero cell line in a dose-dependent manner [8]. EO of Platycladus orientalis, Prangos asperula [5] and Sideritis perfoliata [67] exerted cytotoxic activity on renal adenocarcinoma cell line ACHN. Rhizome and aerial parts of Aristolochia mollissima showed cytotoxicity activity on ACHN cells [59].
Bone cancer
Volatile oil from Pyrolae herba demonstrated potent antitumor activity against SW1353 cells in dose- and time-dependent manner. Furthermore, these EOs decreased the number of cells entering the S phase and caused a reduction in the expression of cyclin D1, cyclin-dependent kinase (CDK)4 and CDK6, whereas it caused an increase of the expression of p21 [68].
Pancreas cancer
Human pancreas cancer cells were sensitive to EO fractions prepared from Boswellia species gum resins treatment with suppressed cell viability and increased cell death. In fact, EO activates the caspase-dependent apoptotic pathway, induces a rapid and transient activation of Akt and Erk1/2, and suppresses levels of cyclin D1 cdk4 expression in cultured cancer cells [69]. EO from stem bark of Kadsura longipedunculata exhibited cytotoxic activity against MIA PaCa-2 cell line of human pancreas carcinoma [70]. EOs from Angelica archangelica fruits collected in Reykjavik, from various locations showed cytotoxic activity independent of the quantity of their main components on PANC-1 human pancreas cancer cells [71].
Skin cancer
EO of Schefflera heptaphylla (L.) Frodin and its major compound beta-pinene ((-)-beta-pinene and (+)-beta-pinene) showed significant antiproliferative activity against A375 cancer cell lines [72].
Mode of action
Due to their high heterogeneous compositions, it is difficult to define a unique mechanism of action for EOs. Indeed, a molecule could have an effect on one type of tumor and not on others. For example, Murata et al. [9] showed that 1,8-cineole/eucalyptol induces apoptosis of human colon cancer cells. Conversely, this molecule has no effect on prostate cancer and glioblastoma cell survival [36]. Moreover, depending on the enrichment of the active compounds, various mechanisms could be observed, such as an effect on the cell cycle, cell growth, and/or apoptosis.
Indeed, generally biological activity of an EO is related to its chemical composition, to the major functional groups of compounds (alcohols, phenols, terpene compounds and ketone). However the less present compounds could also be of importance as the various molecules could synergistically act with the major compounds [73]. For exemple the single exposure to limonene or linalyl acetate found enriched in bergamot (Citrus bergamia Risso et Poiteau) does not replicate the effect of bergamot EO on caspase-3 activation, PARP cleavage, DNA fragmentation, cell shrinkage, cytoskeletal alterations, together with necrotic and apoptotic cell death [74]. Despite that fact and for a didactic aspect, it has seemed interesting however to present the molecular effects of some example of isolated compounds from EOs.
Terpenes and terpenoids
Edris [75] reported that β-elemene, a sesquiterpene from Nigella sativa, could inhibit the growth of laryngeal cancer cells by activating caspase-3 cleavage and decreasing the accumulation of eukaryotic initiation factors eIF-4E and 4G, basic fibroblast growth factor (bFGF) and vascular epithelial growth factor (VEGF).
In ginger, gingerol down-regulates the antiapoptotic protein Bcl-2 and enhances the pro-apoptotic protein Bax, while gingerdione is an effective anti-tumor agent in human leukemia cells by inducing G1 arrest, through the down-regulation of cyclin D2, cyclin E and cdc25A and the up-regulation of CDK1 and p15. Gingerdione also decreases Bcl-2 accumulation and activates caspase-3 cleavage [76]. The monoterpene 1,8-cineole/eucalyptol induces specific apoptosis, and not necrosis, on human colon cancer cell lines HCT116 and RKO. The treatment with 1,8-cineole was associated with the inactivation of surviving, and Akt and activation of p38. These molecules induce the cleavage of PARP and caspase-3, finally causing apoptosis [9]. D-Limonene is toxic on V79 cells in a dose-dependent manner [77]. In fact, this drug has a direct effect on dividing cells, preventing assembly of mitotic spindle microtubules. This affects both chromosome segregation and cytokinesis, resulting in aneuploidy that in turn can lead to cell death or genomic instability [77].
β-caryophyllene oxide, a sesquiterpene isolated primarily from the EOs of medicinal plants such as guava (Psidium guajava) and oregano (Origanum vulgare L.), suppresses constitutive STAT3 activation in multiple myeloma, breast and prostate cancer cell lines, with a significant dose- and time-dependent effects observed in multiple myeloma cells [78]. The suppression was mediated through the inhibition of activation of upstream kinases c-Src and JAK1/2. Indeed, β-caryophyllene oxide induces the expression of tyrosine phosphatase SHP-1 that correlates with the down-regulation of constitutive STAT3 activation [78].
Geraniol, present in the EOs of many aromatic plants, has in vitro and in vivo antitumor activity against several cell lines [79]. In fact, geraniol alters several lipid metabolic pathways of HepG2 cells such as the mevalonate pathway and the phosphatidylcholine biosynthesis, which results in cell growth inhibition, cell cycle arrest occurring at the G0/G1 interphase, and increased apoptosis [79].
Isointermedeol, a major sesquiterpene found in EO extracted from Cymbopogon flexuosus, induces apoptosis in human leukaemia HL-60 cells [80]. Indeed, isointermedeol activates apical death receptors TNFR1, DR4 and caspase-8 activity. Simultaneously, both increase the expression of mitochondrial cytochrome c protein with its concomitant release to cytosol leading to caspase-9 activation. Further, Bax translocation and decrease in nuclear NF-kappaB expression predict multi-target effects of isointermedeol while both appeared to follow similar signaling apoptosis pathways [80].
Furanodiene, a sesquiterpene extracted from Curcuma wenyujin, enhances mitochondrial transmembrane depolarization, release of mitochondrial cytochrome c, activation of caspases-3 and cleavage of PARP [81]. Furanodiene mediated mitochondria-caspase apoptotic pathway also involves activation of p38 and inhibition of ERK mitogen-activated protein kinase (MAPK) signaling [81].
Collectively, thymol induces a cytosolic Ca2+ rise by inducing phospholipase C- and protein kinase C-dependent Ca2+ release from the endoplasmic reticulum and Ca2+ entry. Likewise Liang and Lu [82] showed that carvacrol has the same effect on Ca2+ and cell viability as thymol, even though carvacrol effect could also involve ROS-mediated apoptosis.
Terpenoids thymoquinone, which is the major compound of black seed (Nigella sativa) oil, traditionally used in Mediterranean and arab medicine, possesses significant anticancer effects in various cancer models [83]. It was previously shown that thymoquinone induces apoptosis through p53-dependent pathways in human colon cancer cells and animal models [84,85]. β-Elemene arrests the cell cycle and induces apoptosis of lung cancer cells [86].
Aromatic compounds
Carvacrol induces apoptosis and activation of ROS and caspase-3 [87]. Indeed this monoterpenoid raises intracellular concentration of Ca2+ by activating a PKC-sensitive, non store-operated Ca2+ channels. Carvacrol also induces ROS- and caspase-3-associated apoptosis of OC2 human oral cancer cells [87].
Eugenol is an essential oil mainly found in buds and leaves of clove Syzygium aromaticum (L.) inhibits the cell proliferation and induces the apoptosis in human MCF-7 breast cancer cells [88]. This biological activity is correlated to its activity as an estrogen receptor antagonist. In this article, we present the construction and validation of structure-based virtual screening. This biological property has been correlated to its activity as an estrogen receptor antagonist [88]. Found also in EO of Eugenia caryophyllata, eugenol induces apoptosis of human promyelocytic leukemia HL-60 cells [89]. This phenomenon has been explained by the translocation of Bax from the cytosol to the mitochondria, the reduction of Bcl-2 protein level and cytochrome c release into cytosol, thus leading to sequential activation of caspase-9 and caspase-3 [89].
Indolizine derivatives could also be anticancer agents [90]. For example, 4-(3,4)-dihydroxyphenyl)-2-phenylpyrido[2,3-b]indolizine-10-carbonitrile has a strong cytotoxicity on all tested colorectal cancer cell lines, and this at concentrations that have no effect on fibroblasts [90]. Cell-cycle analysis indicated that pyrido [2,3-b]indolizines could affect cell-cycle progression by accumulating treated cells in S- and G2/M-phases [90].
Conclusion and future callenges
According to the World Health Organization [91], incidence and mortality of cancer is increasing worlwide. The various treatments require a careful selection of one or more of existing modalities such as surgery, radiotherapy and systemic therapy. This selection should be based on evidence of the best existing treatment given the resources available. However in developing countries such as Burkina Faso, it is not always possible to have a correct access to anticancer molecules. Plants are hence a potential source of drug discovery and development of cancer chemoprevention or treatment [92]. They could thus provide a hope for finding anticancer molecules available and efficient for the treatment of persons with cancer (Figure 5). This review shows that more and more studies are necessary to carry out on the anti-cancer activity of EOs as nature is a rich source of biological and chemical diversity. The unique and complex structures of natural products cannot be obtained easily by chemical synthesis. Interest in medicinal plant research has increased in recent years, especially for the treatment of cancer [6,93]. Cytotoxicity has been reported for many EOs [94,95]. However, very few studies have been done on the combination of EOs and their major compounds to find putative synergistic beneficial effects, as the association of 5-fluorouracil and geraniol in both SW620 and Caco-2 cells of human colonic carcinoma [96].
Figure 5.
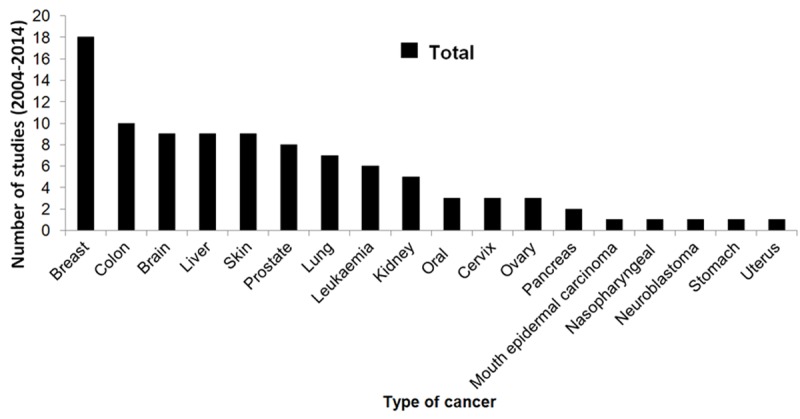
Total of studies with EOs and compounds from 2004 to 2014 on various types of cancer.
However, when EOs are extracted from aromatic plants, there is a long road before using them as a drug. The pharmaceutical research phase is mandatory to identify new molecular targets, both in cell culture and in animal models, and to engineer more efficient molecules from the natural compound. To help in accelerating the molecule identification it is important to remember that all ancient civilizations have developed alongside agriculture, herbal medicine and eminent physicians of the past were usually also herbalists. Nobody could deny that chemotypes of EOs have a wide spectrum of action on various therapies, even though the molecular mechanisms and events need to be identified as well.
The joint activity between chemistry, biochemistry, biology, medicine, pharmacy and botany to correctly identify these chemical constituents is thus fundamental. Finding new active EOs for the treatment of cancers is a challenge. Using it in Human to treat is promise.
Acknowledgements
Research supports were provided by Conférence Episcopale Italienne (CEI), Union Economique Monétaire Ouest Africaine (UEMOA) and Campus France for BB, IHNB, CG, JS; Erasmus Exchange Programm for RS; Fondation BNP-Paris, Région Auvergne, Fond Européen de Développement Régional (FEDER), Association de Recherche sur les Tumeurs Prostatiques (ARTP), Fondation ARC, Ligue contre le Cancer and Plan National de Recherche -Perturbateur Endocrinien (13-MRES-PNRPE-1-CVS-043) for LM and JMAL.
Disclosure of conflict of interest
None.
References
- 1.Buckle J. Use of aromatherapy as a complementary treatment for chronic pain. Altern Ther Health Med. 1999;5:42–51. [PubMed] [Google Scholar]
- 2.Sylvestre M, Pichette A, Longtin A, Nagau F, Legault J. Essential oil analysis and anticancer activity of leaf essential oil of Croton flavens L. from Guadeloupe. J Ethnopharmacol. 2006;103:99–102. doi: 10.1016/j.jep.2005.07.011. [DOI] [PubMed] [Google Scholar]
- 3.Mimica-Dukic N, Bozin B, Sokovic M, Simin N. Antimicrobial and antioxidant activities of Melissa officinalis L. (Lamiaceae) essential oil. J Agric Food Chem. 2004;52:2485–9. doi: 10.1021/jf030698a. [DOI] [PubMed] [Google Scholar]
- 4.Sylvestre M, Legault J, Dufour D, Pichette A. Chemical composition and anticancer activity of leaf essential oil of Myrica gale L. Phytomedicine. 2005;12:299–304. doi: 10.1016/j.phymed.2003.12.004. [DOI] [PubMed] [Google Scholar]
- 5.Loizzo MR, Tundis R, Menichini F, Saab AM, Statti GA, Menichini F. Antiproliferative effects of essential oils and their major constituents in human renal adenocarcinoma and amelanotic melanoma cells. Cell Prolif. 2008;41:1002–12. doi: 10.1111/j.1365-2184.2008.00561.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Heinrich M, Bremner P. Ethnobotany and ethnopharmacy--their role for anti-cancer drug development. Curr Drug Targets. 2006;7:239–45. doi: 10.2174/138945006776054988. [DOI] [PubMed] [Google Scholar]
- 7.Newman DJ, Cragg GM. Natural products as sources of new drugs over the last 25 years. J Nat Prod. 2007;70:461–77. doi: 10.1021/np068054v. [DOI] [PubMed] [Google Scholar]
- 8.Yousefzadi M, Riahi-Madvar A, Hadian J, Rezaee F, Rafiee R, Biniaz M. Toxicity of essential oil of Satureja khuzistanica: In vitro cytotoxicity and anti-microbial activity. J Immunotoxicol. 2014;11:50–5. doi: 10.3109/1547691X.2013.789939. [DOI] [PubMed] [Google Scholar]
- 9.Murata S, Shiragami R, Kosugi C, Tezuka T, Yamazaki M, Hirano A, Yoshimura Y, Suzuki M, Shuto K, Ohkohchi N, Koda K. Antitumor effect of 1, 8-cineole against colon cancer. Oncol Rep. 2013;30:2647–52. doi: 10.3892/or.2013.2763. [DOI] [PubMed] [Google Scholar]
- 10.Fogang HPD, Maggi F, Tapondjou LA, Womeni HM, Papa F, Quassinti L, Bramucci M, Vitali LA, Petrelli D, Lupidi G, Vittori S, Barboni L. In vitro Biological Activities of Seed Essential Oils from the Cameroonian Spices Afrostyrax lepidophyllus Mildbr. and Scorodophloeus zenkeri Harms Rich in Sulfur-Containing Compounds. Chem Biodivers. 2014;11:161–9. doi: 10.1002/cbdv.201300237. [DOI] [PubMed] [Google Scholar]
- 11.Rashid S, Rather MA, Shah WA, Bhat BA. Chemical composition, antimicrobial, cytotoxic and antioxidant activities of the essential oil of Artemisia indica Willd. Food Chem. 2013;138:693–700. doi: 10.1016/j.foodchem.2012.10.102. [DOI] [PubMed] [Google Scholar]
- 12.Duquénois P, Anton R. [Search for derivatives of anthracene in 2 African Cassia: Cassia nigricans Vahl et Cassia podocarpa Guill. et Perr] . Ann Pharm Fr. 1968;26:607–14. [PubMed] [Google Scholar]
- 13.Burt S. Essential oils: their antibacterial properties and potential applications in foods--a review. Int J Food Microbiol. 2004;94:223–53. doi: 10.1016/j.ijfoodmicro.2004.03.022. [DOI] [PubMed] [Google Scholar]
- 14.Santoyo S, Cavero S, Jaime L, Ibañez E, Señoráns FJ, Reglero G. Chemical composition and antimicrobial activity of Rosmarinus officinalis L. essential oil obtained via supercritical fluid extraction. J Food Prot. 2005;68:790–5. doi: 10.4315/0362-028x-68.4.790. [DOI] [PubMed] [Google Scholar]
- 15.Kimbaris AC, Siatis NG, Daferera DJ, Tarantilis PA, Pappas CS, Polissiou MG. Comparison of distillation and ultrasound-assisted extraction methods for the isolation of sensitive aroma compounds from garlic (Allium sativum) Ultrason Sonochem. 2006;13:54–60. doi: 10.1016/j.ultsonch.2004.12.003. [DOI] [PubMed] [Google Scholar]
- 16.Charpentier B. [New therapeutic targets for antibodies and recombinant proteins in organ transplantation] . Bull Acad Natl Med. 2008;192:883–893. discussion 893-894. [PubMed] [Google Scholar]
- 17.Pichersky E, Noel JP, Dudareva N. Biosynthesis of plant volatiles: nature’s diversity and ingenuity. Science. 2006;311:808–11. doi: 10.1126/science.1118510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bakkali F, Averbeck S, Averbeck D, Idaomar M. Biological effects of essential oils--a review. Food Chem Toxicol. 2008;46:446–75. doi: 10.1016/j.fct.2007.09.106. [DOI] [PubMed] [Google Scholar]
- 19.Connolly JD, Hill RA. Dictionary of Terpenoids. Vol. 1: Mono- and Sesquiterpenoids, Vol. 2: Di- and higher Terpenoids, Vol. 3: Indexes. New York, Tokyo, Melbourne, Madras: Chapman & Hall London; 1991. [Google Scholar]
- 20.Pinder AR. The Chemistry of Terpenes. New York: Chapman & Hall London; 1960. [Google Scholar]
- 21.Ruzicka L. Proc Chem Soc (London) 1959:341. [Google Scholar]
- 22.Hernandez Ochoa LR. Substitution de solvants et matieres actives de synthese par un combine “solvant/actif” d’origine vegetale [Internet] 2005. [cited 2013 Jul 27]. Available from: http://www.youscribe.com/catalogue/ressources-pedagogiques/education/etudes-superieures/n-d-ordre-these-1667992.
- 23.Lahlou M, Berrada R. Composition and niticidal activity of essential oils of three chemotypes of Rosmarinus officinalis L. acclimatized in Morocco. J Flavour Fragr. 2003;18:124–7. [Google Scholar]
- 24.Lahlou M. Essential oils and fragrance compounds: bioactivity and mechanisms of action. J Flavour Fragr. 2004;19:159–65. [Google Scholar]
- 25.Jackson AL, Loeb LA. The contribution of endogenous sources of DNA damage to the multiple mutations in cancer. Mutat Res. 2001;477:7–21. doi: 10.1016/s0027-5107(01)00091-4. [DOI] [PubMed] [Google Scholar]
- 26.Storz P. Reactive oxygen species in tumor progression. Front Biosci. 2005;10:1881–96. doi: 10.2741/1667. [DOI] [PubMed] [Google Scholar]
- 27.Mantovani A. Cancer: inflammation by remote control. Nature. 2005;435:752–3. doi: 10.1038/435752a. [DOI] [PubMed] [Google Scholar]
- 28.Quassinti L, Lupidi G, Maggi F, Sagratini G, Papa F, Vittori S, Bianco A, Bramucci M. Antioxidant and antiproliferative activity of Hypericum hircinum L. subsp. majus (Aiton) N. Robson essential oil. Nat Prod Res. 2013;27:862–8. doi: 10.1080/14786419.2012.677044. [DOI] [PubMed] [Google Scholar]
- 29.Gasmi J, Thomas Sanderson J. Jacaric acid and its octadecatrienoic acid geoisomers induce apoptosis selectively in cancerous human prostate cells: a mechanistic and 3-D structure–activity study. Phytomedicine. 2013;20:734–42. doi: 10.1016/j.phymed.2013.01.012. [DOI] [PubMed] [Google Scholar]
- 30.Yousuf Dar M, Shah WA, Mubashir S, Rather MA. Chromatographic analysis, anti-proliferative and radical scavenging activity of Pinus wallichina essential oil growing in high altitude areas of Kashmir, India. Phytomedicine. 2012;19:1228–33. doi: 10.1016/j.phymed.2012.07.015. [DOI] [PubMed] [Google Scholar]
- 31.Essien EE, Ogunwande IA, Setzer WN, Ekundayo O. Chemical composition, antimicrobial, and cytotoxicity studies on S. erianthum and S. macranthum essential oils. Pharm Biol. 2012;50:474–80. doi: 10.3109/13880209.2011.614623. [DOI] [PubMed] [Google Scholar]
- 32.Zu Y, Yu H, Liang L, Fu Y, Efferth T, Liu X, Wu N. Activities of ten essential oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 cancer cells. Molecules. 2010;15:3200–10. doi: 10.3390/molecules15053200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hussain AI, Anwar F, Nigam PS, Ashraf M, Gilani AH. Seasonal variation in content, chemical composition and antimicrobial and cytotoxic activities of essential oils from four Mentha species. J Sci Food Agric. 2010;90:1827–36. doi: 10.1002/jsfa.4021. [DOI] [PubMed] [Google Scholar]
- 34.Kim HS, Lee EH, Ko SR, Choi KJ, Park JH, Im DS. Effects of ginsenosides Rg3 and Rh2 on the proliferation of prostate cancer cells. Arch Pharm Res. 2004;27:429–35. doi: 10.1007/BF02980085. [DOI] [PubMed] [Google Scholar]
- 35.Do N Fontes JE, Ferraz RPC, Britto ACS, Carvalho AA, Moraes MO, Pessoa C, Costa EV, Bezerra DP. Antitumor effect of the essential oil from leaves of Guatteria pogonopus (Annonaceae) Chem Biodivers. 2013;10:722–9. doi: 10.1002/cbdv.201200304. [DOI] [PubMed] [Google Scholar]
- 36.Bayala B, Bassole IHN, Gnoula C, Nebie R, Yonli A, Morel L, Figueredo G, Nikiema JB, Lobaccaro JMA, Simpore J. Chemical composition, antioxidant, anti-inflammatory and anti-proliferative activities of essential oils of plants from burkina faso. PLoS One. 2014;9:e92122. doi: 10.1371/journal.pone.0092122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hsu SS, Lin KL, Chou CT, Chiang AJ, Liang WZ, Chang HT, Tsai JY, Liao WC, Huang FD, Huang JK, Chen IS, Liu SI, Kuo CC, Jan CR. Effect of thymol on Ca2+ homeostasis and viability in human glioblastoma cells. Eur J Pharmacol. 2011;670:85–91. doi: 10.1016/j.ejphar.2011.08.017. [DOI] [PubMed] [Google Scholar]
- 38.Detoni CB, de Oliveira DM, Santo IE, Pedro AS, El-Bacha R, da Silva Velozo E, Ferreira D, Sarmento B, de Magalhães Cabral-Albuquerque EC. Evaluation of thermal-oxidative stability and antiglioma activity of Zanthoxylum tingoassuiba essential oil entrapped into multi- and unilamellar liposomes. J Liposome Res. 2012;22:1–7. doi: 10.3109/08982104.2011.573793. [DOI] [PubMed] [Google Scholar]
- 39.Desai A, Vyas T, Amiji M. Cytotoxicity and apoptosis enhancement in brain tumor cells upon coadministration of paclitaxel and ceramide in nanoemulsion formulations. J Pharm Sci. 2008;97:2745–56. doi: 10.1002/jps.21182. [DOI] [PubMed] [Google Scholar]
- 40.Rasoanaivo P, Fortuné Randriana R, Maggi F, Nicoletti M, Quassinti L, Bramucci M, Lupidi G, Petrelli D, Vitali LA, Papa F, Vittori S. Chemical composition and biological activities of the essential oil of Athanasia brownii Hochr. (Asteraceae) endemic to Madagascar. Chem Biodivers. 2013;10:1876–86. doi: 10.1002/cbdv.201300147. [DOI] [PubMed] [Google Scholar]
- 41.Su YC, Hsu KP, Wang EIC, Ho CL. Composition and in vitro anticancer activities of the leaf essential oil of Neolitsea variabillima from Taiwan. Nat Prod Commun. 2013;8:531–2. [PubMed] [Google Scholar]
- 42.Bou DD, Lago JH, Figueiredo CR, Matsuo AL, Guadagnin RC, Soares MG, Sartorelli P. Chemical composition and cytotoxicity evaluation of essential oil from leaves of Casearia sylvestris, its main compound α-zingiberene and derivatives. Molecules. 2013;18:9477–87. doi: 10.3390/molecules18089477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Afoulous S, Ferhout H, Raoelison EG, Valentin A, Moukarzel B, Couderc F, Bouajila J. Chemical composition and anticancer, antiinflammatory, antioxidant and antimalarial activities of leaves essential oil of Cedrelopsis grevei. Food Chem Toxicol. 2013;56:352–62. doi: 10.1016/j.fct.2013.02.008. [DOI] [PubMed] [Google Scholar]
- 44.Keawsa-ard S, Liawruangrath B, Liawruangrath S, Teerawutgulrag A, Pyne SG. Chemical constituents and antioxidant and biological activities of the essential oil from leaves of Solanum spirale. Nat Prod Commun. 2012;7:955–8. [PubMed] [Google Scholar]
- 45.Suhail MM, Wu W, Cao A, Mondalek FG, Fung KM, Shih PT, Fang YT, Woolley C, Young G, Lin HK. Boswellia sacra essential oil induces tumor cell-specific apoptosis and suppresses tumor aggressiveness in cultured human breast cancer cells. BMC Complement Altern Med. 2011;11:129. doi: 10.1186/1472-6882-11-129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Carnesecchi S, Schneider Y, Ceraline J, Duranton B, Gosse F, Seiler N, Raul F. Geraniol, a component of plant essential oils, inhibits growth and polyamine biosynthesis in human colon cancer cells. J Pharmacol Exp Ther. 2001;298:197–200. [PubMed] [Google Scholar]
- 47.Chidambara Murthy KN, Jayaprakasha GK, Patil BS. D-limonene rich volatile oil from blood oranges inhibits angiogenesis, metastasis and cell death in human colon cancer cells. Life Sci. 2012;91:429–39. doi: 10.1016/j.lfs.2012.08.016. [DOI] [PubMed] [Google Scholar]
- 48.Akrout A, Gonzalez LA, El Jani H, Madrid PC. Antioxidant and antitumor activities of Artemisia campestris and Thymelaea hirsuta from southern Tunisia. Food Chem Toxicol. 2011;49:342–7. doi: 10.1016/j.fct.2010.11.003. [DOI] [PubMed] [Google Scholar]
- 49.El-Najjar N, Chatila M, Moukadem H, Vuorela H, Ocker M, Gandesiri M, Schneider-Stock R, Gali-Muhtasib H. Reactive oxygen species mediate thymoquinone-induced apoptosis and activate ERK and JNK signaling. Apoptosis. 2010;15:183–95. doi: 10.1007/s10495-009-0421-z. [DOI] [PubMed] [Google Scholar]
- 50.Kpoviessi S, Bero J, Agbani P, Gbaguidi F, Kpadonou-Kpoviessi B, Sinsin B, Accrombessi G, Frédérich M, Moudachirou M, Quetin-Leclercq J. Chemical composition, cytotoxicity and in vitro antitrypanosomal and antiplasmodial activity of the essential oils of four Cymbopogon species from Benin. J Ethnopharmacol. 2014;151:652–9. doi: 10.1016/j.jep.2013.11.027. [DOI] [PubMed] [Google Scholar]
- 51.Walia M, Mann TS, Kumar D, Agnihotri VK, Singh B. Chemical Composition and In Vitro Cytotoxic Activity of Essential Oil of Leaves of Malus domestica Growing in Western Himalaya (India) Evid Based Complement Alternat Med. 2012;2012:649727. doi: 10.1155/2012/649727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sun H, Sun C, Pan Y. Cytotoxic activity and constituents of the volatile oil from the roots of Patrinia scabra Bunge. Chem Biodivers. 2005;2:1351–7. doi: 10.1002/cbdv.200590107. [DOI] [PubMed] [Google Scholar]
- 53.Wu S, Wei FX, Li HZ, Liu XG, Zhang JH, Liu JX. [Chemical composition of essential oil from Thymus citriodorus and its toxic effect on liver cancer cells] . Zhong Yao Cai. 2013;36:756–9. [PubMed] [Google Scholar]
- 54.Abdallah HM, Ezzat SM. Effect of the method of preparation on the composition and cytotoxic activity of the essential oil of Pituranthos tortuosus. Z Naturforsch C. 2011;66:143–8. doi: 10.1515/znc-2011-3-408. [DOI] [PubMed] [Google Scholar]
- 55.Paik SY, Koh KH, Beak SM, Paek SH, Kim JA. The essential oils from Zanthoxylum schinifolium pericarp induce apoptosis of HepG2 human hepatoma cells through increased production of reactive oxygen species. Biol Pharm Bull. 2005;28:802–7. doi: 10.1248/bpb.28.802. [DOI] [PubMed] [Google Scholar]
- 56.El-Readi MZ, Eid HH, Ashour ML, Eid SY, Labib RM, Sporer F, Wink M. Variations of the chemical composition and bioactivity of essential oils from leaves and stems of Liquidambar styraciflua (Altingiaceae) J Pharm Pharmacol. 2013;65:1653–63. doi: 10.1111/jphp.12142. [DOI] [PubMed] [Google Scholar]
- 57.Santana JS, Sartorelli P, Guadagnin RC, Matsuo AL, Figueiredo CR, Soares MG, da Silva AM, Lago JHG. Essential oils from Schinus terebinthifolius leaves - chemical composition and in vitro cytotoxicity evaluation. Pharm Biol. 2012;50:1248–53. doi: 10.3109/13880209.2012.666880. [DOI] [PubMed] [Google Scholar]
- 58.Sun XY, Zheng YP, Lin DH, Zhang H, Zhao F, Yuan CS. Potential anti-cancer activities of Furanodiene, a Sesquiterpene from Curcuma wenyujin. Am J Chin Med. 2009;37:589–96. doi: 10.1142/S0192415X09007077. [DOI] [PubMed] [Google Scholar]
- 59.Yu JQ, Liao ZX, Cai XQ, Lei JC, Zou GL. Composition, antimicrobial activity and cytotoxicity of essential oils from Aristolochia mollissima. Environ Toxicol Pharmacol. 2007;23:162–7. doi: 10.1016/j.etap.2006.08.004. [DOI] [PubMed] [Google Scholar]
- 60.Ferraz RPC, Cardoso GMB, da Silva TB, Fontes JE do N, Prata AP do N, Carvalho AA, Moraes MO, Pessoa C, Costa EV, Bezerra DP. Antitumour properties of the leaf essential oil of Xylopia frutescens Aubl. (Annonaceae) Food Chem. 2013;141:196–200. doi: 10.1016/j.foodchem.2013.02.114. [DOI] [PubMed] [Google Scholar]
- 61.Manjamalai A, Kumar MJM, Grace VMB. Essential oil of Tridax procumbens L induces apoptosis and suppresses angiogenesis and lung metastasis of the B16F-10 cell line in C57BL/6 mice. Asian Pac J Cancer Prev. 2012;13:5887–95. doi: 10.7314/apjcp.2012.13.11.5887. [DOI] [PubMed] [Google Scholar]
- 62.Seal S, Chatterjee P, Bhattacharya S, Pal D, Dasgupta S, Kundu R, Mukherjee S, Bhattacharya S, Bhuyan M, Bhattacharyya PR, Baishya G, Barua NC, Baruah PK, Rao PG, Bhattacharya S. Vapor of volatile oils from Litsea cubeba seed induces apoptosis and causes cell cycle arrest in lung cancer cells. PLoS One. 2012;7:e47014. doi: 10.1371/journal.pone.0047014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Su YC, Hsu KP, Wang EIC, Ho CL. Composition and in vitro anticancer activities of the leaf essential oil of Neolitsea variabillima from Taiwan. Nat Prod Commun. 2013;8:531–2. [PubMed] [Google Scholar]
- 64.Jo JR, Park JS, Park YK, Chae YZ, Lee GH, Park GY, Jang BC. Pinus densiflora leaf essential oil induces apoptosis via ROS generation and activation of caspases in YD-8 human oral cancer cells. Int J Oncol. 2012;40:1238–45. doi: 10.3892/ijo.2011.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sertel S, Eichhorn T, Plinkert PK, Efferth T. Chemical Composition and antiproliferative activity of essential oil from the leaves of a medicinal herb, Levisticum officinale, against UMSCC1 head and neck squamous carcinoma cells. Anticancer Res. 2011;31:185–91. [PubMed] [Google Scholar]
- 66.Saab AM, Guerrini A, Sacchetti G, Maietti S, Zeino M, Arend J, Gambari R, Bernardi F, Efferth T. Phytochemical analysis and cytotoxicity towards multidrug-resistant leukemia cells of essential oils derived from Lebanese medicinal plants. Planta Med. 2012;78:1927–31. doi: 10.1055/s-0032-1327896. [DOI] [PubMed] [Google Scholar]
- 67.Loizzo MR, Tundis R, Menichini F, Saab AM, Statti GA, Menichini F. Cytotoxic activity of essential oils from labiatae and lauraceae families against in vitro human tumor models. Anticancer Res. 2007;27:3293–9. [PubMed] [Google Scholar]
- 68.Cai L, Ye H, Li X, Lin Y, Yu F, Chen J, Li H, Liu X. Chemical constituents of volatile oil from Pyrolae herba and antiproliferative activity against SW1353 human chondrosarcoma cells. Int J Oncol. 2013;42:1452–8. doi: 10.3892/ijo.2013.1816. [DOI] [PubMed] [Google Scholar]
- 69.Ni X, Suhail MM, Yang Q, Cao A, Fung KM, Postier RG, Woolley C, Young G, Zhang J, Lin HK. Frankincense essential oil prepared from hydrodistillation of Boswellia sacra gum resins induces human pancreatic cancer cell death in cultures and in a xenograft murine model. BMC Complement Altern Med. 2012;12:253. doi: 10.1186/1472-6882-12-253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mulyaningsih S, Youns M, El-Readi MZ, Ashour ML, Nibret E, Sporer F, Herrmann F, Reichling J, Wink M. Biological activity of the essential oil of Kadsura longipedunculata (Schisandraceae) and its major components. J Pharm Pharmacol. 2010;62:1037–44. doi: 10.1111/j.2042-7158.2010.01119.x. [DOI] [PubMed] [Google Scholar]
- 71.Sigurdsson S, Ogmundsdottir HM, Gudbjarnason S. The cytotoxic effect of two chemotypes of essential oils from the fruits of Angelica archangelica L. Anticancer Res. 2005;25:1877–80. [PubMed] [Google Scholar]
- 72.Li YL, Yeung CM, Chiu LCM, Cen YZ, Ooi VEC. Chemical composition and antiproliferative activity of essential oil from the leaves of a medicinal herb, Schefflera heptaphylla. Phytother Res. 2009;23:140–2. doi: 10.1002/ptr.2567. [DOI] [PubMed] [Google Scholar]
- 73.Bakkali F, Averbeck S, Averbeck D, Zhiri A, Baudoux D, Idaomar M. Antigenotoxic effects of three essential oils in diploid yeast (Saccharomyces cerevisiae) after treatments with UVC radiation, 8-MOP plus UVA and MMS. Mutat Res. 2006;606:27–38. doi: 10.1016/j.mrgentox.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 74.Russo R, Ciociaro A, Berliocchi L, Cassiano MGV, Rombolà L, Ragusa S, Bagetta G, Blandini F, Corasaniti MT. Implication of limonene and linalyl acetate in cytotoxicity induced by bergamot essential oil in human neuroblastoma cells. Fitoterapia. 2013;89:48–57. doi: 10.1016/j.fitote.2013.05.014. [DOI] [PubMed] [Google Scholar]
- 75.Edris AE. Anti-cancer properties of Nigella spp. essential oils and their major constituents, thymoquinone and beta-elemene. Curr Clin Pharmacol. 2009;4:43–6. doi: 10.2174/157488409787236137. [DOI] [PubMed] [Google Scholar]
- 76.Shukla Y, Singh M. Cancer preventive properties of ginger: a brief review. Food Chem Toxicol. 2007;45:683–90. doi: 10.1016/j.fct.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 77.Mauro M, Catanzaro I, Naselli F, Sciandrello G, Caradonna F. Abnormal mitotic spindle assembly and cytokinesis induced by D-Limonene in cultured mammalian cells. Mutagenesis. 2013;28:631–5. doi: 10.1093/mutage/get040. [DOI] [PubMed] [Google Scholar]
- 78.Kim C, Cho SK, Kapoor S, Kumar A, Vali S, Abbasi T, Kim SH, Sethi G, Ahn KS. β-Caryophyllene oxide inhibits constitutive and inducible STAT3 signaling pathway through induction of the SHP-1 protein tyrosine phosphatase. Mol Carcinog. 2014;53:793–806. doi: 10.1002/mc.22035. [DOI] [PubMed] [Google Scholar]
- 79.Crespo R, Montero Villegas S, Abba MC, de Bravo MG, Polo MP. Transcriptional and posttranscriptional inhibition of HMGCR and PC biosynthesis by geraniol in 2 Hep-G2 cell proliferation linked pathways. Biochem Cell Biol. 2013;91:131–9. doi: 10.1139/bcb-2012-0076. [DOI] [PubMed] [Google Scholar]
- 80.Kumar A, Malik F, Bhushan S, Sethi VK, Shahi AK, Kaur J, Taneja SC, Qazi GN, Singh J. An essential oil and its major constituent isointermedeol induce apoptosis by increased expression of mitochondrial cytochrome c and apical death receptors in human leukaemia HL-60 cells. Chem Biol Interact. 2008;171:332–47. doi: 10.1016/j.cbi.2007.10.003. [DOI] [PubMed] [Google Scholar]
- 81.Xiao Y, Yang FQ, Li SP, Gao JL, Hu G, Lao SC, Conceição EL, Fung KP, Wang lYT, Lee SM. Furanodiene induces G2/M cell cycle arrest and apoptosis through MAPK signaling and mitochondria-caspase pathway in human hepatocellular carcinoma cells. Cancer Biol Ther. 2007;6:1044–50. doi: 10.4161/cbt.6.7.4317. [DOI] [PubMed] [Google Scholar]
- 82.Liang WZ, Lu CH. Carvacrol-induced [Ca2+] i rise and apoptosis in human glioblastoma cells. Life Sci. 2012;90:703–11. doi: 10.1016/j.lfs.2012.03.027. [DOI] [PubMed] [Google Scholar]
- 83.Worthen DR, Ghosheh OA, Crooks PA. The in vitro anti-tumor activity of some crude and purified components of blackseed, Nigella sativa L. Anticancer Res. 1998;18:1527–32. [PubMed] [Google Scholar]
- 84.Gali-Muhtasib H, Kuester D, Mawrin C, Bajbouj K, Diestel A, Ocker M, Habold C, Foltzer-Jourdainne C, Schoenfeld P, Peters B, Diab-Assaf M, Pommrich U, Itani W, Lippert H, Roessner A, Schneider-Stock R. Thymoquinone triggers inactivation of the stress response pathway sensor CHEK1 and contributes to apoptosis in colorectal cancer cells. Cancer Res. 2008;68:5609–18. doi: 10.1158/0008-5472.CAN-08-0884. [DOI] [PubMed] [Google Scholar]
- 85.Gali-Muhtasib H, Diab-Assaf M, Boltze C, Al-Hmaira J, Hartig R, Roessner A, Schneider-Stock R. Thymoquinone extracted from black seed triggers apoptotic cell death in human colorectal cancer cells via a p53-dependent mechanism. Int J Oncol. 2004;25:857–66. [PubMed] [Google Scholar]
- 86.Jiang H, Sólyom AM, Timmermann BN, Gang DR. Characterization of gingerol-related compounds in ginger rhizome (Zingiber officinale Rosc. ) by high-performance liquid chromatography/ electrospray ionization mass spectrometry. Rapid Commun Mass Spectrom. 2005;19:2957–64. doi: 10.1002/rcm.2140. [DOI] [PubMed] [Google Scholar]
- 87.Liang WZ, Chou CT, Lu T, Chi CC, Tseng LL, Pan CC, Lin KL, Kuo CC, Jan CR. The mechanism of carvacrol-evoked [Ca2+] i rises and non-Ca2+-triggered cell death in OC2 human oral cancer cells. Toxicology. 2013;303:152–61. doi: 10.1016/j.tox.2012.10.026. [DOI] [PubMed] [Google Scholar]
- 88.Anita Y, Radifar M, Kardono LB, Hanafi M, Istyastono EP. Structure-based design of eugenol analogs as potential estrogen receptor antagonists. Bioinformation. 2012;8:901–6. doi: 10.6026/97320630008901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Yoo CB, Han KT, Cho KS, Ha J, Park HJ, Nam JH, Kil UH, Lee KT. Eugenol isolated from the essential oil of Eugenia caryophyllata induces a reactive oxygen species-mediated apoptosis in HL-60 human promyelocytic leukemia cells. Cancer Lett. 2005;225:41–52. doi: 10.1016/j.canlet.2004.11.018. [DOI] [PubMed] [Google Scholar]
- 90.Boot A, Brito A, VAN Wezel T, Morreau H, Costa M, Proença F. Anticancer activity of novel pyrido[2,3-b] indolizine derivatives: the relevance of phenolic substituents. Anticancer Res. 2014;34:1673–7. [PubMed] [Google Scholar]
- 91.WHO. World Cancer Report. In: Bernard S, Christopher PW, editors. IARC Nonserial Publication. Geneva: WHO Press; 2014. p. 630. [Google Scholar]
- 92.Newell DR. How to develop a successful cancer drug--molecules to medicines or targets to treatments? Eur J Cancer. 2005;41:676–82. doi: 10.1016/j.ejca.2004.12.024. [DOI] [PubMed] [Google Scholar]
- 93.Heinrich M, Gibbons S. Ethnopharmacology in drug discovery: an analysis of its role and potential contribution. J Pharm Pharmacol. 2001;53:425–32. doi: 10.1211/0022357011775712. [DOI] [PubMed] [Google Scholar]
- 94.Cavalieri E, Mariotto S, Fabrizi C, de Prati AC, Gottardo R, Leone S, Berra LV, Lauro GM, Ciampa AR, Suzuki H. alpha-Bisabolol, a nontoxic natural compound, strongly induces apoptosis in glioma cells. Biochem Biophys Res Commun. 2004;315:589–94. doi: 10.1016/j.bbrc.2004.01.088. [DOI] [PubMed] [Google Scholar]
- 95.Lampronti I, Saab AM, Gambari R. Antiproliferative activity of essential oils derived from plants belonging to the Magnoliophyta division. Int J Oncol. 2006;29:989–95. [PubMed] [Google Scholar]
- 96.Carnesecchi S, Bras-Gonçalves R, Bradaia A, Zeisel M, Gossé F, Poupon MF, Raul F. Geraniol, a component of plant essential oils, modulates DNA synthesis and potentiates 5-fluorouracil efficacy on human colon tumor xenografts. Cancer Lett. 2004;215:53–9. doi: 10.1016/j.canlet.2004.06.019. [DOI] [PubMed] [Google Scholar]
- 97.Bezerra DP, Marinho Filho JDB, Alves APNN, Pessoa C, de Moraes MO, Pessoa ODL, Torres MCM, Silveira ER, Viana FA, Costa-Lotufo LV. Antitumor activity of the essential oil from the leaves of Croton regelianus and its component ascaridole. Chem Biodivers. 2009;6:1224–31. doi: 10.1002/cbdv.200800253. [DOI] [PubMed] [Google Scholar]
- 98.Nagappan T, Ramasamy P, Wahid MEA, Segaran TC, Vairappan CS. Biological Activity of Carbazole Alkaloids and Essential Oil of Murraya koenigii Against Antibiotic Resistant Microbes and Cancer Cell Lines. Molecules. 2011;16:9651–64. doi: 10.3390/molecules16119651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Afoulous S, Ferhout H, Raoelison EG, Valentin A, Moukarzel B, Couderc F, Bouajila J. Helichrysum gymnocephalum Essential Oil: Chemical Composition and Cytotoxic, Antimalarial and Antioxidant Activities, Attribution of the Activity Origin by Correlations. Molecules. 2011;16:8273–91. doi: 10.3390/molecules16108273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Chabir N, Romdhane M, Valentin A, Moukarzel B, Marzoug HNB, Brahim NB, Mars M, Bouajila J. Chemical study and antimalarial, antioxidant, and anticancer activities of Melaleuca armillaris (Sol Ex Gateau) Sm essential oil. J Med Food. 2011;14:1383–8. doi: 10.1089/jmf.2010.0168. [DOI] [PubMed] [Google Scholar]
- 101.Hussain AI, Anwar F, Chatha SA, Jabbar A, Mahboob S, Nigam PS. Rosmarinus officinalis essential oil: antiproliferative, antioxidant and antibacterial activities. Braz J Microbiol. 2010;41:1070–8. doi: 10.1590/S1517-838220100004000027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Bendaoud H, Romdhane M, Souchard JP, Cazaux S, Bouajila J. Chemical composition and anticancer and antioxidant activities of Schinus molle L. and Schinus terebinthifolius Raddi berries essential oils. J Food Sci. 2010;75:C466–472. doi: 10.1111/j.1750-3841.2010.01711.x. [DOI] [PubMed] [Google Scholar]
- 103.Nazaruk J, Karna E, Wieczorek P, Sacha P, Tryniszewska E. In vitro antiproliferative and antifungal activity of essential oils from Erigeron acris L. and Erigeron annuus (L.) Pers. Z Naturforsch C. 2010;65:642–6. doi: 10.1515/znc-2010-11-1202. [DOI] [PubMed] [Google Scholar]
- 104.Xu WN, Gao XX, Guo XL, Chen YC, Zhang WM, Luo YS. [Study on volatile components from peel of Aquilaria sinensis and the anti-tumor activity] . Zhong Yao Cai. 2010;33:1736–40. [PubMed] [Google Scholar]
- 105.Kathirvel P, Ravi S. Chemical composition of the essential oil from basil (Ocimum basilicum Linn. ) and its in vitro cytotoxicity against HeLa and HEp-2 human cancer cell lines and NIH 3T3 mouse embryonic fibroblasts. Nat Prod Res. 2012;26:1112–8. doi: 10.1080/14786419.2010.545357. [DOI] [PubMed] [Google Scholar]
- 106.Sylvestre M, Pichette A, Lavoie S, Longtin A, Legault J. Composition and cytotoxic activity of the leaf essential oil of Comptonia peregrina (L. ) Coulter. Phytother Res. 2007;21:536–40. doi: 10.1002/ptr.2095. [DOI] [PubMed] [Google Scholar]
- 107.Sharma PR, Mondhe DM, Muthiah S, Pal HC, Shahi AK, Saxena AK, Qazi GN. Anticancer activity of an essential oil from Cymbopogon flexuosus. Chem Biol Interact. 2009;179:160–8. doi: 10.1016/j.cbi.2008.12.004. [DOI] [PubMed] [Google Scholar]
- 108.Manosroi J, Dhumtanom P, Manosroi A. Anti-proliferative activity of essential oil extracted from Thai medicinal plants on KB and P388 cell lines. Cancer Lett. 2006;235:114–20. doi: 10.1016/j.canlet.2005.04.021. [DOI] [PubMed] [Google Scholar]
- 109.Xiao Y, Yang FQ, Li SP, Hu G, Lee SM, Wang YT. Essential oil of Curcuma wenyujin induces apoptosis in human hepatoma cells. World J Gastroenterol. 2008;14:4309–18. doi: 10.3748/wjg.14.4309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chen W, Lu Y, Gao M, Wu J, Wang A, Shi R. Anti-angiogenesis effect of essential oil from Curcuma zedoaria in vitro and in vivo. J Ethnopharmacol. 2011;133:220–6. doi: 10.1016/j.jep.2010.09.031. [DOI] [PubMed] [Google Scholar]
- 111.Su M, Wu P, Li Y, Chung HY. Antiproliferative effects of volatile oils from Centipeda minima on human nasopharyngeal cancer CNE cells. Nat Prod Commun. 2010;5:151–6. [PubMed] [Google Scholar]
- 112.Sertel S, Eichhorn T, Plinkert PK, Efferth T. [Anticancer activity of Salvia officinalis essential oil against HNSCC cell line (UMSCC1)] . HNO. 2011;59:1203–8. doi: 10.1007/s00106-011-2274-3. [DOI] [PubMed] [Google Scholar]
- 113.Jayaprakasha G, Murthy K, Demarais R, Patil B. Inhibition of Prostate Cancer (LNCaP) Cell Proliferation by Volatile Components from Nagami Kumquats. Planta Med. 2012;78:974–80. doi: 10.1055/s-0031-1298619. [DOI] [PubMed] [Google Scholar]
- 114.Islam SN, Begum P, Ahsan T, Huque S, Ahsan M. Immunosuppressive and cytotoxic properties of Nigella sativa. Phytother Res. 2004;18:395–8. doi: 10.1002/ptr.1449. [DOI] [PubMed] [Google Scholar]


