Abstract
HER family has been implicated in a number of malignant tumors for predicting prognosis and potential targeted therapy purposes, however, the prognostic roles of HER family in GISTs have not been elaborated yet. Our study aims to fully evaluate the prognostic value of HER family in GIST patients and efficacy of imatinib adjuvant therapy. For HER family expression detection, qPCR were used in 33 flesh GIST specimens, and then, 453 GIST samples (405 GISTs with operation only and 48 with imatinib adjuvant therapy after radical surgery) were collected for tissue microarrays construction and immunohistochemistry (IHC). Clinicopathological data were confirmed by pathological diagnosis and clinical recorders, recurrence-free survivals (RFS) were evaluated in 453 GIST patients. With qPCR and IHC performed, EGFR, HER2 and HER4 are focused on examining prognostic value in remainder of our study by high positive expression rates in GISTs. In high-risk GISTs with or without imatinib adjuvant therapy, EGFR negative expression are associated with decreased RFS when compared to positive cases. HER2 present no relationship with GIST patients’ prognosis. HER4 positive expression significantly associated with disease recurrence in GISTs. Further subgroup studies revealed HER4 was an independent prognostic indicator especially for gastric GISTs, and also for gastric high-risk GISTs. In our study, detection of EGFR expression helps to precisely subdivide high-risk GISTs for different prognosis and probably predict outcomes for imatinib treatment. HER4 is a novel independent prognostic biomarker for gastric GISTs specifically, which could be potential therapeutic target in GISTs originated from stomach.
Keywords: Gastrointestinal stromal tumor, EGFR, HER2, HER4, prognosis
Introduction
Gastrointestinal stromal tumors (GISTs) accounts for more than 80% of all gastrointestinal mesenchymal tumors [1]. As it ranks below only gastric and colorectal cancers, GIST is among the most common types of gastrointestinal tumors with increased incidence in recent years [2-4].
Most of GISTs show abnormal activation of the tyrosine kinase proteins KIT or PDGFRa, which were transcribed by specific mutations in these genes. For evaluating the potential malignancy and predicting the prognosis of GISTs, several criteria were wildly accepted as risk-stratification schemes for GISTs with similar accuracy, such as NIH consensus criteria, Modified NIH criteria or AFIP criteria, the mitosis count, tumor size, tumor site were important prognostic indicators in these schemes [5]. Following the operation, a considerable amount of patients suffered disease recurrence especially in high-risk GISTs [5]. Although the majority of KIT or PDGFRa mutations predict response to the tyrosine kinase inhibitor imatinib, however, still quite a part of GISTs relapsed even with imatinib adjuvant therapy [6,7], and almost all advanced GIST patients eventually develop resistance to imatinib treatment [8]. On the other hand, with the same KIT or PDGFRa mutations as highly malignant GISTs, the observation on micro-GISTs (1-10 mm size), which were present high incidence from stomachs, suggested these mutations probably not sufficient for progression to an oncologically threatening lesion even kinase gene mutations occur very early in GISTs tumorigenesis [9]. All of these indicated some unrevealed biological characteristics that derive tumorigenesis and progression in GISTs.
HER (human epidermal growth factor receptor related) family includes four transmembrane tyrosine kinase receptors named EGFR (HER1, ErbB1), HER2 (ErbB2), HER3 (ErbB3) and HER4 (ErbB4), which shared highly conserved extracellular domain, transmembrane junction, and intracellular ATP-binding kinase domain [10]. HER family and their downstream signaling components had been implicated in a number of malignant tumors occurring in humans for predicting prognosis and potential targeted therapy purposes. Several monoclonal antibodies or small molecular agents for targeting these receptors have been identified and a significant number of them shown efficacy in clinical trials [10,11]. However, the prognostic roles of HER family in GISTs have not been elaborated yet. Here, we use quantitative real-time PCR and immunohistochemistry for determining the differences of HER family mRNA and protein expression levels in GISTs, and tissue microarrays (TMAs) containing tumor samples (405 GISTs with operation only and 48 high-risk GISTs received imatinib adjuvant therapy after radical surgery) with known clinical outcomes for predicting prognosis by HER family.
Materials and methods
Ethics statement
This project was approved by ethics committee of Ren Ji Hospital, Shanghai Jiao Tong University, School of Medicine for the use of samples. Informed consents were obtained from all patients before study inclusion.
Patients and specimens
33 flesh tumor tissues obtained from GIST patients during tumor resection between August 2012 to February 2014 were collected for detecting mRNA expression level of HER family by quantitative real-time PCR.
As for evaluation immunoreactivity and prognostic value of HER family in GISTs. GIST patients inclusion criteria were as follows: 1) a distinct pathologic diagnosis of GIST (CD117 positive in immunohistochemistry staining); 2) underwent radical surgical resection, radical surgical resection in our study defined as margin-free resection and no metastasis detected before and during the surgery; 3) no radiotherapy, chemotherapy, nor other anti-cancer therapies prior to the surgery; and 4) availability of complete clinicopathologic and follow-up data. High-risk GIST patients with imatinib adjuvant therapy should meet extra inclusion criteria which required at least 12 months uninterrupted drugs taking with 400 mg/day in our study. The parameters, including patient age, gender, tumor site, tumor size and number of mitoses/50 high-power fields (HPF) were recorded in the official pathology database. The risk of aggressive tumor behavior was calculated according to the modified NIH criteria [12], which classified GIST into very low, low, intermediate, and high-risk categories.
A total of 453 paraffin-embedded tissue samples met the criteria were collected from GIST patients (405 GISTs with operation only and 48 high-risk GISTs received imatinib adjuvant therapy after radical surgery) at Ren Ji Hospital, Shanghai Jiao Tong University School of Medicine from June 2004 to May 2013 for TMAs construction and immunohistochemistry staining. Complete follow-up data until May 2014 for GIST patients were available. Recurrence free survival (RFS) was calculated from the date of tumor resection until the detection of tumor recurrence or last observation. The median follow-up of 405 GISTs with operation only was 53 months (range, 8-113 months). In high-risk GISTs with imatinib adjuvant therapy, the median follow-up was 45 months (range 22-74 months). Computed tomography (CT) and/or magnetic resonance imaging (MRI) were used to verify tumor recurrence in suspected cases.
Total RNA extraction and quantitative real-time PCR
Total RNA was extracted from 33 fresh GIST tissues using Trizol reagent (Takara, Dalian, China) followed the manufacturer instructions. The reverse-transcription reactions were carried out with random primers and M-MLV Reverse Transcriptase (Takara, Dalian, China). The 33 cases of cDNA were used for templates of quantitative real-time PCR reaction in SYBR-Green method. All the qPCR reactions were performed on a StepOneTM real-time PCR System (Applied Biosystems, Foster City, CA, USA). The specific primer sequences of HER family and 18s were as follow: EGFR [forward: 5’-GCTGGATGATAGACGCAGATAG-3’; reverse: 5’-TGGGAACGGACTGGTTTATG-3’], HER2 [forward: 5’-CCCTGTTCTCCGATGTGTAAG-3’; reverse: 5’-AAGCAGAGGTGGGTGTTATG-3’], HER3 [forward: 5’-GGCGATGCTGAGAACCAATA-3’; reverse: 5’-GTGGCTGGAGTTGGTGTTATAG-3’], HER4 [forward: 5’-CCGAGGATGAGTATGTGAATGAG-3’; reverse: 5’-CAGTATTCCGGTGTCTGTAAGG-3’], and 18s [forward: 5’-TGCGAGTACTCAACACCAACA-3’; reverse: 5’-GCATATCTTCGGCCCACA-3’]. 18s was used as an internal control. The 2-∆Ct method was used to quantify the relative HER family expression levels.
Tissue microarrays construction
TMAs were constructed by Suzhou Xinxin Biotechnology Co., Ltd (Xinxin Biotechnology Co, Suzhou, China). Tissue paraffin blocks of GIST samples were stained with hematoxylin-eosin to confirm the diagnoses and marked at fixed points with most typical histological characteristics under a microscope. Two 1.6 mm cores per donor block were transferred into a recipient block tissue microarrayer, and each dot array contained fewer than one hundred and sixty dots. Three-micron-thick sections were cut from the recipient block and transferred onto glass slides using an adhesive tape transfer system for ultraviolet cross linkage.
Immunohistochemistry
The tissue microarray glass slides were baked at 55°C for one hour, and then de-paraffinized gradually through xylene, 50% xylene, and gradient concentrations of ethanol until immersed in tap water. Tissue sections were blocked for peroxidase activity with 0.3% Hydrogen peroxide at 37°C for 30 mins. Antigen retrieval was carried out via boiling in 10 mmol/L citrate buffer (pH 6.0) for fifteen mins. Then the tissues were incubated with EGFR antibody (mouse monoclonal antibody, Abcam), ErbB2 antibody (mouse monoclonal antibody, Abcam), ErbB3 antibody (rabbit monoclonal antibody, CST) or ErbB4 antibody (mouse monoclonal antibody, Abcam) overnight at 4°C. Next day, the tissues were washed with phosphate buffer solution (PBS) for three times and incubated with goat anti-rabbit IgG-HRP (HUABIO) or goat anti-mouse IgG-HRP (ABCAM) secondary antibody for one hour at room temperature. Immunostaining was carried out using diaminobenzidine substrate chromogen (Dako, Carpinteria, CA, USA) method and chromogenic reaction was controlled under microscope. After immunostaining, tissues were immersed into hematoxylin for nuclear staining. The slides were then dehydrated through gradient concentrations of ethanol, cleared with xylene, and coverslipped with neutral balsam.
Judgment criteria for immunoreactivity of HER family in GISTs was referred from Edris et al’s research [13]. TMAs were scored as follows: 0: absence of any staining; 1: weak staining whether diffusely or focally present in the tumor; 2: strong staining whether diffusely or focally present in the tumor. Score 0-1 was considered as negative (-) and a score of 2 was positive (+) for subsequent statistical analyses.
Statistical analysis
Statistical analyses were conducted using SPSS for Windows (version 21.0) and MedCalc (version 11.4.2.0). RFS was calculated according to Kaplan-Meier method and log-rank test was used for comparing the survival distributions. Univariate and multivariate analyses were based on the cox proportional hazards regression model. All statistical tests were two-sided. P value less than 0.05 was considered statistically significant.
Results
HER family mRNA and protein expression in GISTs
To determine the differences in expression of HER family in GISTs, we analyzed mRNA transcript levels of HER family by quantitative real-time PCR from 33 GISTs. Scatter dot plot for mRNA relative expression levels (2-∆Ct) of HER family in GISTs were shown in Figure 1, which HER3 showed a low levels of expression in GISTs. The clinicopathological characters of GISTs for quantitative real-time PCR were shown in Table 1.
Figure 1.
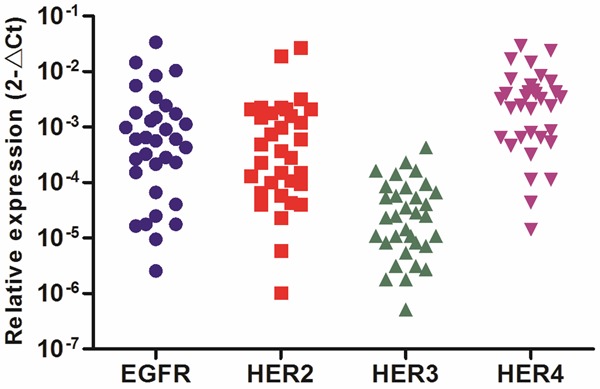
Relative expression levels of HER family in GISTs by quantitative real-time PCR.
Table 1.
Clinicopathological characters of GISTs for qantitative real-time PCR
| Number | % | |
|---|---|---|
| Age (years) | ||
| ≤ 50 | 5 | 15.2 |
| > 50 | 28 | 84.8 |
| Gender | ||
| Male | 17 | 51.5 |
| Female | 16 | 48.5 |
| Tumor site | ||
| Stomach | 21 | 63.6 |
| Small bowel | 12 | 36.4 |
| Tumor size (cm) | ||
| 2.1-5.0 | 13 | 39.4 |
| 5.1-10.0 | 13 | 39.4 |
| > 10.0 | 7 | 21.2 |
| Mitoses per 50 HPFs | ||
| ≤ 5 | 24 | 72.7 |
| 6-10 | 4 | 12.1 |
| > 10 | 5 | 15.2 |
| Modified NIH criteria | ||
| Low risk | 14 | 42.4 |
| Intermediate risk | 6 | 18.2 |
| High risk | 13 | 39.4 |
| Total | 33 | 100.0 |
To confirm HER family expression, we performed immunohistochemistry (IHC) study using TMAs that contained 453 GISTs (405 GISTs with operation only and 48 high-risk GISTs with imatinib adjuvant therapy). Similar to our quantitative real-time PCR findings, GISTs on TMAs fail to react with HER3 antibody (434 cases with score 0 and 19 cases with score 1), but EGFR, HER2 and HER4 showed strong IHC reactivity in GISTs (Table 2). Representative stains of HER family scored as 0, 1 and 2 were shown in Figure 2. Negative (score 0-1) or positive (score 2) expression were used for subsequent statistical analyses as aforementioned. Given the IHC reactivity of HER family observed, we focused remainder of our study on examining the prognostic value of EGFR, HER2 and HER4 in GISTs. The clinicopathological characters of GISTs with operation only and high-risk GISTs with imatinib adjuvant therapy could be referred from Tables 3 and 4.
Table 2.
IHC scores for HER family in 453 GISTs (405 GISTs with operation only and 48 high-risk GISTs with imatinib adjuvant therapy)
| EGFR | % | HER2 | % | HER3 | % | HER4 | % | |
|---|---|---|---|---|---|---|---|---|
| Score | ||||||||
| 0 | 28 | 6.2 | 39 | 8.6 | 434 | 95.8 | 20 | 4.4 |
| 1 | 172 | 38.0 | 177 | 39.1 | 19 | 4.2 | 209 | 46.1 |
| 2 | 253 | 55.8 | 237 | 52.3 | 0 | 0.0 | 224 | 49.4 |
| Total | 453 | 100.0 | 453 | 100.0 | 453 | 100.0 | 453 | 100.0 |
Figure 2.
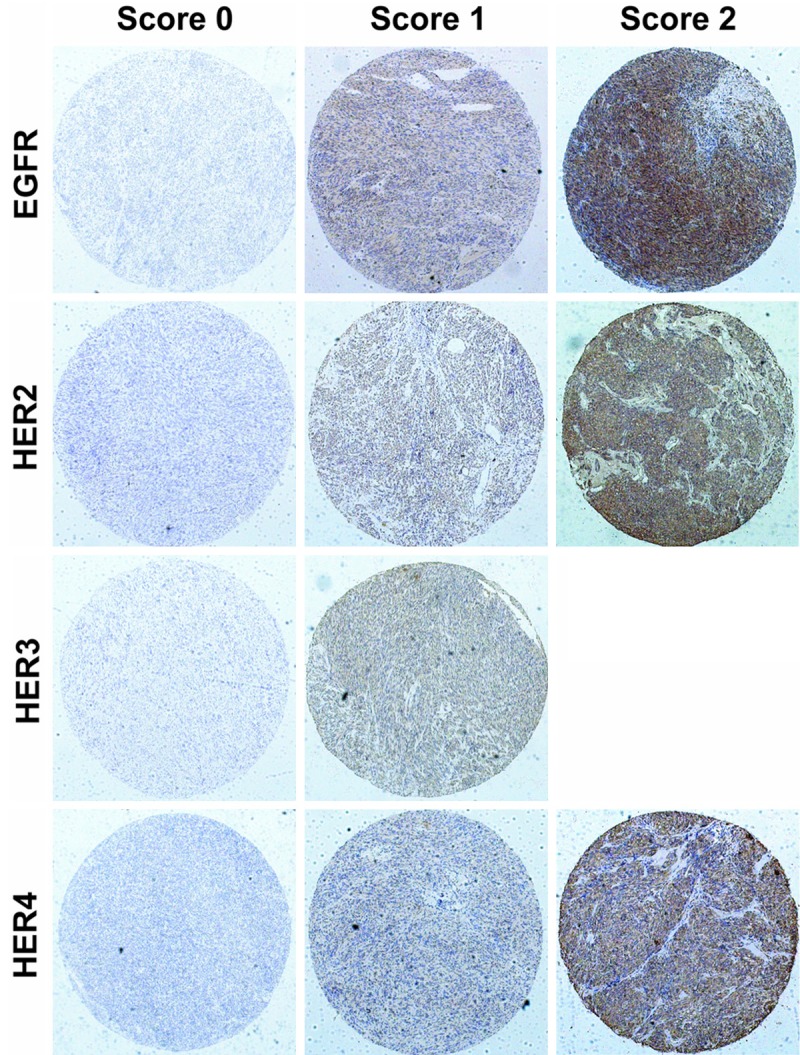
Representative immunohistochemical stains for HER family in GISTs.
Table 3.
IHC expression for HER family and clinicopathological characters in GISTs with operation only
| Total | % | EGFRs | HER2 | HER4 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||||||
| — | % | + | % | — | % | + | % | — | % | + | % | |||
| Age (years) | ||||||||||||||
| ≤ 50 | 81 | 20.0 | 38 | 9.4 | 43 | 10.6 | 42 | 10.4 | 39 | 9.6 | 45 | 11.1 | 36 | 8.9 |
| > 50 | 324 | 80.0 | 139 | 34.3 | 185 | 45.7 | 153 | 37.8 | 171 | 42.2 | 162 | 40.0 | 162 | 40.0 |
| Gender | ||||||||||||||
| Male | 215 | 53.1 | 101 | 24.9 | 114 | 28.1 | 109 | 26.9 | 106 | 26.2 | 113 | 27.9 | 102 | 25.2 |
| Female | 190 | 46.9 | 76 | 18.8 | 114 | 28.1 | 86 | 21.2 | 104 | 25.7 | 94 | 23.2 | 96 | 23.7 |
| Tumor site | ||||||||||||||
| Stomach | 238 | 58.8 | 111 | 27.4 | 127 | 31.4 | 126 | 31.1 | 112 | 27.7 | 128 | 31.6 | 110 | 27.2 |
| Small bowel | 129 | 31.9 | 45 | 11.1 | 84 | 20.7 | 44 | 10.9 | 85 | 21.0 | 55 | 13.6 | 74 | 18.3 |
| Colorectum | 19 | 4.7 | 14 | 3.5 | 5 | 1.2 | 15 | 3.7 | 4 | 1.0 | 11 | 2.7 | 8 | 2.0 |
| Others | 19 | 4.7 | 7 | 1.7 | 12 | 3.0 | 10 | 2.5 | 9 | 2.2 | 13 | 3.2 | 6 | 1.5 |
| Tumor size (cm) | ||||||||||||||
| ≤ 2.0 | 36 | 8.9 | 23 | 5.7 | 13 | 3.2 | 27 | 6.7 | 9 | 2.2 | 26 | 6.4 | 10 | 2.5 |
| 2.1-5.0 | 194 | 47.9 | 84 | 20.7 | 110 | 27.2 | 89 | 22.0 | 105 | 25.9 | 102 | 25.2 | 92 | 22.7 |
| 5.1-10.0 | 115 | 28.4 | 41 | 10.1 | 74 | 18.3 | 53 | 13.1 | 62 | 15.3 | 52 | 12.8 | 63 | 15.6 |
| > 10.0 | 60 | 14.8 | 29 | 7.2 | 31 | 7.7 | 26 | 6.4 | 34 | 8.4 | 27 | 6.7 | 33 | 8.1 |
| Mitoses per 50 HPFs | ||||||||||||||
| ≤ 5 | 327 | 80.7 | 145 | 35.8 | 182 | 44.9 | 162 | 40.0 | 165 | 40.7 | 174 | 43.0 | 153 | 37.8 |
| 6-10 | 43 | 10.6 | 17 | 4.2 | 26 | 6.4 | 17 | 4.2 | 26 | 6.4 | 15 | 3.7 | 28 | 6.9 |
| > 10 | 35 | 8.6 | 15 | 3.7 | 20 | 4.9 | 16 | 4.0 | 19 | 4.7 | 18 | 4.4 | 17 | 4.2 |
| Modified NIH criteria | ||||||||||||||
| Very low risk | 32 | 7.9 | 21 | 5.2 | 11 | 2.7 | 26 | 6.4 | 6 | 1.5 | 25 | 6.2 | 7 | 1.7 |
| Low risk | 187 | 46.2 | 83 | 20.5 | 104 | 25.7 | 85 | 21.0 | 102 | 25.2 | 98 | 24.2 | 89 | 22.0 |
| Intermediate risk | 62 | 15.3 | 24 | 5.9 | 38 | 9.4 | 34 | 8.4 | 28 | 6.9 | 31 | 7.7 | 31 | 7.7 |
| High risk | 124 | 30.6 | 49 | 12.1 | 75 | 18.5 | 50 | 12.3 | 74 | 18.3 | 53 | 13.1 | 71 | 17.5 |
| Total | 405 | 100.0 | 177 | 43.7 | 228 | 56.3 | 195 | 48.1 | 210 | 51.9 | 207 | 51.1 | 198 | 48.9 |
Table 4.
IHC expression for HER family and clinicopathological characters in high-risk GISTs with imatinib adjuvant therapy
| Total | % | EGFR | HER2 | HER4 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||||||
| — | % | + | % | — | % | + | % | — | % | + | % | |||
| Age (years) | ||||||||||||||
| ≤ 50 | 7 | 14.6 | 2 | 4.2 | 5 | 10.4 | 3 | 6.3 | 4 | 8.3 | 2 | 4.2 | 5 | 10.4 |
| > 50 | 41 | 85.4 | 21 | 43.8 | 20 | 41.7 | 18 | 37.5 | 23 | 47.9 | 20 | 41.7 | 21 | 43.8 |
| Gender | ||||||||||||||
| Male | 28 | 58.3 | 13 | 27.1 | 15 | 31.3 | 14 | 29.2 | 14 | 29.2 | 13 | 27.1 | 15 | 31.3 |
| Female | 20 | 41.7 | 10 | 20.8 | 10 | 20.8 | 7 | 14.6 | 13 | 27.1 | 9 | 18.8 | 11 | 22.9 |
| Tumor site | ||||||||||||||
| Stomach | 18 | 37.5 | 11 | 22.9 | 7 | 14.6 | 9 | 18.8 | 9 | 18.8 | 9 | 18.8 | 9 | 18.8 |
| Small bowel | 23 | 47.9 | 9 | 18.8 | 14 | 29.2 | 9 | 18.8 | 14 | 29.2 | 10 | 20.8 | 13 | 27.1 |
| Colorectum | 3 | 6.3 | 2 | 4.2 | 1 | 2.1 | 1 | 2.1 | 2 | 4.2 | 2 | 4.2 | 1 | 2.1 |
| Others | 4 | 8.3 | 1 | 2.1 | 3 | 6.3 | 2 | 4.2 | 2 | 4.2 | 1 | 2.1 | 3 | 6.3 |
| Tumor size (cm) | ||||||||||||||
| ≤ 2.0 | 1 | 2.1 | 0 | 0.0 | 1 | 2.1 | 0 | 0.0 | 1 | 2.1 | 1 | 2.1 | 0 | 0.0 |
| 2.1-5.0 | 3 | 6.3 | 0 | 0.0 | 3 | 6.3 | 0 | 0.0 | 3 | 6.3 | 1 | 2.1 | 2 | 4.2 |
| 5.1-10.0 | 24 | 50.0 | 15 | 31.3 | 9 | 18.8 | 12 | 25.0 | 12 | 25.0 | 10 | 20.8 | 14 | 29.2 |
| > 10.0 | 20 | 41.7 | 8 | 16.7 | 12 | 25.0 | 9 | 18.8 | 11 | 22.9 | 10 | 20.8 | 10 | 20.8 |
| Mitoses per 50 HPFs | ||||||||||||||
| ≤ 5 | 17 | 35.4 | 7 | 14.6 | 10 | 20.8 | 7 | 14.6 | 10 | 20.8 | 6 | 12.5 | 11 | 22.9 |
| 6-10 | 16 | 33.3 | 8 | 16.7 | 8 | 16.7 | 8 | 16.7 | 8 | 16.7 | 10 | 20.8 | 6 | 12.5 |
| > 10 | 15 | 31.3 | 8 | 16.7 | 7 | 14.6 | 6 | 12.5 | 9 | 18.8 | 6 | 12.5 | 9 | 18.8 |
| Total | 48 | 100.0 | 23 | 47.9 | 25 | 52.1 | 21 | 43.8 | 27 | 56.3 | 22 | 45.8 | 26 | 54.2 |
In 405 GISTs with operation only, subgroup studies were designed based on different tumor site and modified NIH criteria. GISTs from stomach (283/405, 58.8%) and small bowel (129/405, 31.9%) are majority positions that GISTs occur, with different clinical outcomes, were set as subgroups for further analyses. High-risk GIST patients which suffered worst prognosis than very low, low and intermediate-risk were also the most important population deserved attention in GISTs. Kaplan-Meier survival analyses with log-rank test for RFS of GISTs sorted by different tumor site and modified NIH criteria were shown in Figure 3.
Figure 3.
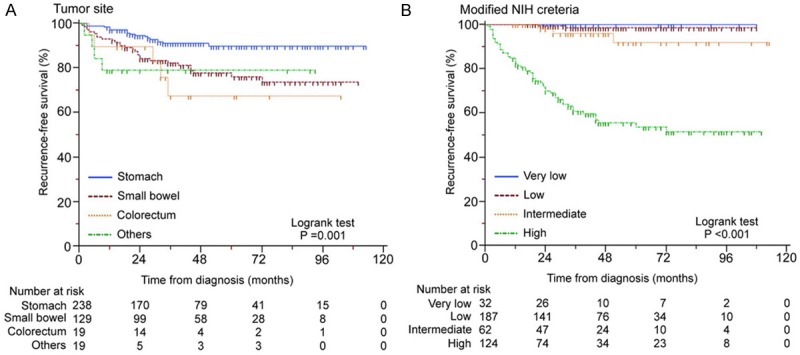
Kaplan-Meier survival analyses with log-rank test for RFS of GISTs sorted by different tumor site (A) and modified NIH criteria (B).
EGFR negative expression predict poor prognosis of high-risk GISTs and imatinib treatment outcome
In GISTs with operation only, we found no significant association between EGFR expression and disease recurrence by univariate and multivariate cox proportional hazards model analyses (Table 5). But in high-risk GISTs, tumors with EGFR negative expression were associated with decreased RFS when compared to cases expressed EGFR positively by Kaplan-Meier survival analysis with log-rank test (P = 0.040, Figure 4A) and univariate analysis (P = 0.037, Table 6).
Table 5.
Univariate & multivariate cox proportional hazards model to predict factors associated with RFS in GISTs with operation only
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
|
|
|
|||
| Hazard Ratio (95% Cl) | P value | Hazard Ratio (95% Cl) | P value | |
| Total | ||||
| Age | 1.414 (0.692-2.887) | 0.342 | 0.905 (0.422-1.943) | 0.799 |
| Gender | 0.413 (0.231-0.738) | 0.003** | 0.523 (0.287-0.954) | 0.034* |
| Tumor site | 1.728 (1.309-2.281) | < 0.001** | 1.392 (1.034-1.875) | 0.029* |
| Tumor size | 4.280 (2.990-6.127) | < 0.001** | 2.812 (1.900-4.161) | < 0.001** |
| Mitosis count | 4.251 (3.180-5.681) | < 0.001** | 2.762 (1.980-3.850) | < 0.001** |
| EGFR | 0.703 (0.415-1.188) | 0.188 | 0.648 (0.360-1.166) | 0.148 |
| HER2 | 1.063 (0.628-1.799) | 0.819 | 0.949 (0.515-1.749) | 0.867 |
| HER4 | 1.870 (1.088-3.213) | 0.023* | 2.370 (1.269-4.423) | 0.007** |
| Stomach sub-group | ||||
| Age | 1.507 (0.348-6.527) | 0.583 | 1.099 (0.229-5.273) | 0.906 |
| Gender | 0.339 (0.129-0.892) | 0.028* | 0.942 (0.313-2.838) | 0.915 |
| Tumor size | 4.452 (2.407-8.235) | < 0.001** | 2.225 (1.101-4.499) | 0.026* |
| Mitosis count | 7.181 (4.207-12.256) | < 0.001** | 6.337 (3.302-12.159) | < 0.001** |
| EGFR | 0.745 (0.302-1.836) | 0.522 | 0.730 (0.245-2.174) | 0.572 |
| HER2 | 1.298 (0.527-3.198) | 0.571 | 1.453 (0.498-4.242) | 0.494 |
| HER4 | 3.745 (1.348-10.410) | 0.011* | 5.207 (1.567-17.297) | 0.007** |
| Small bowel sub-group | ||||
| Age | 2.114 (0.803-5.565) | 0.129 | 0.743 (0.228-2.419) | 0.622 |
| Gender | 0.651 (0.287-1.480) | 0.306 | 0.508 (0.209-1.232) | 0.134 |
| Tumor size | 3.847 (2.325-6.365) | < 0.001** | 3.141 (1.687-5.847) | < 0.001** |
| Mitosis count | 3.452 (2.234-5.334) | < 0.001** | 2.282 (1.359-3.832) | 0.002** |
| EGFR | 0.490 (0.232-1.032) | 0.060 | 0.539 (0.232-1.249) | 0.149 |
| HER2 | 0.653 (0.308-1.382) | 0.265 | 1.003 (0.397-2.535) | 0.995 |
| HER4 | 0.977 (0.461-2.067) | 0.951 | 1.217 (0.485-3.053) | 0.675 |
P < 0.05;
P < 0.01.
Figure 4.
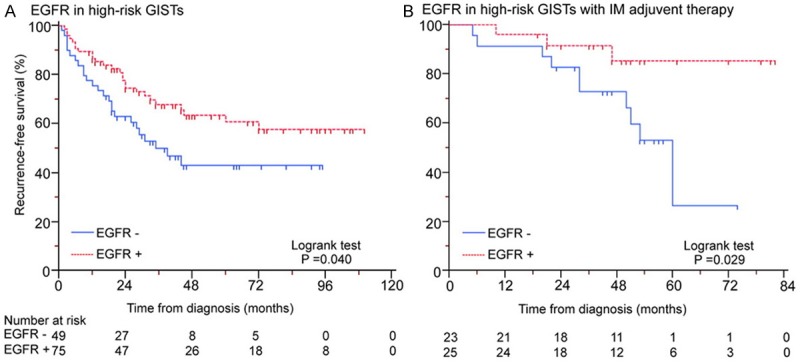
EGFR negative expression predicts poor prognosis in high-risk GISTs (A) and imatinib treatment outcome (B).
Table 6.
Univariate & multivariate cox proportional hazards model to predict factors associated with RFS in high-risk GISTs with operation only
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
|
|
|
|||
| Hazard Ratio (95% Cl) | P value | Hazard Ratio (95% Cl) | P value | |
| Total | ||||
| Age | 1.120 (0.524-2.393) | 0.770 | 0.800 (0.357-1.794) | 0.588 |
| Gender | 0.584 (0.315-1.081) | 0.087 | 0.582 (0.310-1.093) | 0.092 |
| Tumor site | 1.062 (0.770-1.466) | 0.714 | 1.163 (0.845-1.600) | 0.354 |
| Tumor size | 1.582 (1.001-2.500) | 0.049* | 1.726 (1.061-2.808) | 0.028* |
| Mitosis count | 2.060 (1.480-2.868) | < 0.001** | 2.159 (1.518-3.072) | < 0.001** |
| EGFR | 0.567 (0.327-0.984) | 0.044* | 0.608 (0.319-1.159) | 0.131 |
| HER2 | 0.804 (0.464-1.394) | 0.438 | 0.958 (0.505-1.817) | 0.895 |
| HER4 | 1.301 (0.742-2.284) | 0.359 | 1.967 (1.028-3.761) | 0.041* |
| Stomach sub-group | ||||
| Age | 1.173 (0.265-5.183) | 0.833 | 0.579 (0.106-3.150) | 0.527 |
| Gender | 0.694 (0.224-2.154) | 0.528 | 0.725 (0.204-2.572) | 0.618 |
| Tumor size | 1.060 (0.557-2.019) | 0.859 | 1.450 (0.608-3.458) | 0.402 |
| Mitosis count | 3.590 (1.626-7.928) | 0.002** | 4.641 (1.690-12.740) | 0.003** |
| EGFR | 0.533 (0.197-1.440) | 0.215 | 0.563 (0.145-2.192) | 0.408 |
| HER2 | 0.856 (0.320-2.285) | 0.756 | 1.268 (0.386-4.166) | 0.696 |
| HER4 | 3.345 (1.076-10.394) | 0.037* | 4.632 (1.269-16.904) | 0.020* |
| Small bowel sub-group | ||||
| Age | 1.039 (0.357-3.025) | 0.944 | 0.804 (0.237-2.731) | 0.727 |
| Gender | 0.495 (0.215-1.136) | 0.097 | 0.513 (0.217-1.212) | 0.128 |
| Tumor size | 1.933 (0.978-3.821) | 0.058 | 1.678 (0.799-3.524) | 0.172 |
| Mitosis count | 2.061 (1.296-3.279) | 0.002** | 1.984 (1.199-3.285) | 0.008** |
| EGFR | 0.452 (0.210-0.974) | 0.043* | 0.551 (0.232-1.306) | 0.176 |
| HER2 | 0.689 (0.321-1.476) | 0.337 | 0.959 (0.366-2.515) | 0.933 |
| HER4 | 0.742 (0.346-1.589) | 0.442 | 1.048 (0.400-2.742) | 0.924 |
P < 0.05;
P < 0.01.
In high-risk GISTs with imatinib adjuvant therapy, both univariate and multivariate analyses showed EGFR negative expression significantly associated with unsatisfied imatinib treatment outcome (P = 0.043 and 0.027, Table 7). Kaplan-Meier survival analysis with log-rank test for RFS also present a decreased curve in EGFR negative expression tumors compared with positive cases (P = 0.029, Figure 4B).
Table 7.
Univariate & multivariate cox proportional hazards model to predict factors associated with RFS in high-risk GISTs with imatinib adjuvant therapy
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
|
|
|
|||
| Hazard Ratio (95% Cl) | P value | Hazard Ratio (95% Cl) | P value | |
| Age | 0.684 (0.188-2.498) | 0.566 | 0.582 (0.131-2.594) | 0.478 |
| Gender | 0.993 (0.332-2.974) | 0.990 | 0.828 (0.223-3.071) | 0.777 |
| Tumor site | 1.340 (0.701-2.561) | 0.375 | 2.230 (1.065-4.669) | 0.033* |
| Tumor size | 1.811 (0.704-4.659) | 0.218 | 1.778 (0.499-6.330) | 0.374 |
| Mitosis count | 1.514 (0.749-3.059) | 0.248 | 2.243 (0.942-5.345) | 0.068 |
| EGFR | 0.260 (0.070-0.957) | 0.043* | 0.195 (0.046-0.827) | 0.027* |
| HER2 | 0.578 (0.192-1.735) | 0.328 | 0.871 (0.244-3.113) | 0.832 |
| HER4 | 0.891 (0.294-2.706) | 0.839 | 1.216 (0.353-4.193) | 0.756 |
P < 0.05.
Expression of HER2 present no relationship with prognosis in GISTs
Although quite a part of GISTs performed HER2 positive expression, no significant association for prognosis was found between HER2 and GISTs by univariate and multivariate analyses (Tables 5 and 6). The results were same in high-risk GISTs with imatinib adjuvant therapy (Table 7).
HER4 acts as an independent prognostic indicator for gastric GISTs
Compared with other members from HER family, HER4 was less notable in current cancer research, but present its specific value for GISTs in our study. In GISTs with operation only, we found HER4 positive expression significantly associated with disease recurrence by univariate and multivariate cox proportional hazards model analyses (P = 0.023 and 0.007, Table 5). Further subgroup univariate and multivariate studies revealed HER4 was an independent prognostic indicator specifically for gastric GISTs (P = 0.011 and 0.007, Table 5), and also for gastric high-risk GISTs (P = 0.037 and 0.020, Table 6). Kaplan-Meier survival analyses with log-rank test for RFS showed decreased curves in HER4 positive expression tumors compared with negative cases in gastric GISTs (Figure 5).
Figure 5.
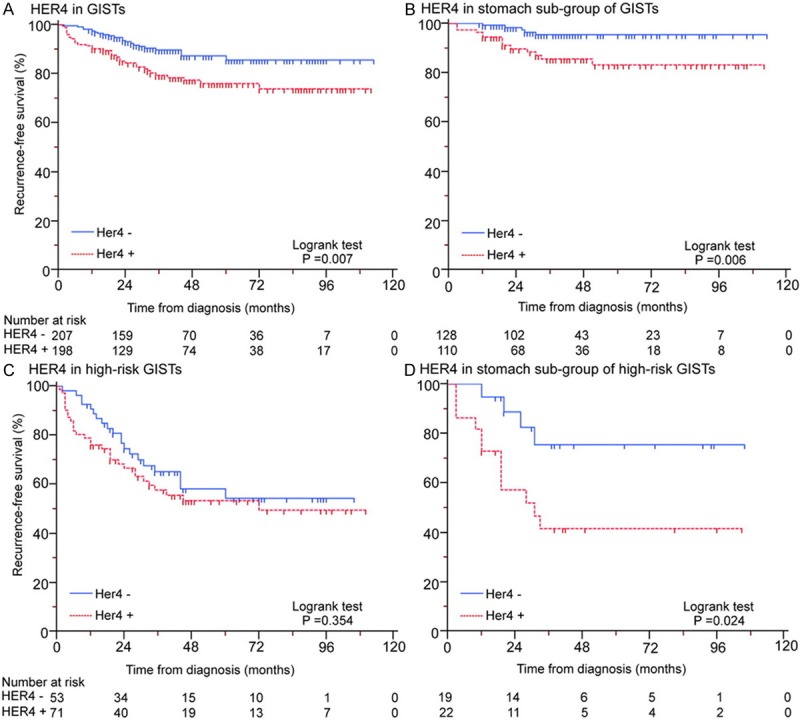
Expression of HER4 for predicting prognosis in GISTs (A), stomach subgroup of GISTs (B), high-risk GISTs (C) and stomach subgroup of high-risk GISTs (D) with operation only.
Discussion
Many studies had shown that deregulated HER family could play very important roles in cancer tumorigenesis and progression [10,14,15]. Furthermore, HER family had been proven as therapeutic targets evidenced by several FDA-approved antibodies such as cetuximab (Erbitux) and trastuzumab (Herceptin), and a series of small molecule drugs for targeting HER family. These therapeutics have already shown clinical efficacy in a wide range of cancer types [10,11]. However, the studies about relationship between HER family and GISTs were very limited, EGFR was once reported high immunohistochemical overexpression level but with no exhibition of gene amplification by fluorescence in situ hybridization (FISH) in 82 GIST cases [16]. Here, we present the first large-scale characterization of HER family expression in GISTs. The initial mRNA expression detection were conducted by quantitative realtime PCR and then confirmed by a larger number of GISTs on 453 cases (405 cases with operation only and 48 high-risk cases received imatinib adjuvant therapy after radical surgery) by IHC on TMAs. HER3 was excluded after mRNA and protein level detection with negative expression in GISTs. EGFR, HER2 and HER4, which present high positive expression proportion (55.8%, 52.3% and 49.4%, respectively) in GISTs, were focused on examining the prognostic value in remainder of our study.
Although EGFR overexpression might predict poor prognosis in many cancer types [10,11], EGFR negative expression cases in high-risk GISTs in our study presented a poor outcome for prognosis, and more important, EGFR negative expression might be an independent adverse indicator for efficacy of imatinib adjuvant therapy through multivariate cox proportional hazards model analysis. GIST patients classified as high-risk by modified NIH criteria faced worst prognosis than other risk grades, but even in the same high-risk GISTs, clinical outcomes were various no matter with or without imatinib treatment. Detection expression of EGFR may help to precisely subdivide high-risk GISTs for different prognosis and imatinib treatment outcome.
HER2 has already been a very important biomarker and therapeutic target in breast and gastric cancer [17,18]. Although positive expression of HER2 was high in our study, no significant association for prognosis was found between HER2 and GISTs, and as for anti-HER2 therapeutic strategies in GISTs still need further evaluation.
HER4 is frequently upregulated in various tumor tissues [19-21] and established as having critical roles with tumor progression and metastasis by activation on PI3K-AKT cascade and focal adhesion kinase [22,23], experimental blockade or down-regulation of HER4 can suppress tumor cell proliferation and growth [24,25], which indicate that HER4 is not only a valuable biomarker but also a potential target for anticancer therapy. With absence of HER3 in GISTs, HER4 play an interesting role in our study. By univariate and multivariate cox proportional hazards model analyses in GISTs with operation only and subgroups, HER4 was revealed as an independent prognostic indicator specifically for gastric GISTs, and also for gastric high-risk GISTs. Stomach was the most common primary site of GISTs, which occupied more than 50% incidence rate in GIST patients [5]. Although diverse biological behaviors and various clinical outcomes from different tumor site had been noticed by researchers [5,12], there are still no biomarkers in distinguishing or predicting prognosis for specific site of GISTs now. The results of our study indicated HER4 was a novel prognostic biomarker especially for gastric GISTs, which implied HER4 could be an important molecular target participated in tumorigenesis and progression of GISTs originated from stomach.
The limitations of our study are mainly in follow-up time and sample size for high-risk GIST patients with imatinib adjuvant therapy. By prolonging the duration of imatinib treatment can apparently improve 5 years’ RFS, and quite a part of GIST patients only present tumor recurrence after stopping imatinib therapy [7,26], which implied we need more sufficient time for following-up especially after patients stopping drugs taking for imatinib treatment evaluation purpose. In consideration of above-mentioned, the result that EGFR negative expression was an adverse indicator for efficacy of imatinib adjuvant therapy in our study can not be considered as fully confidential evidence, but providing suggestion for further confirmation. HER2 and HER4 showed no relationship in predicting imatinib treatment outcome in our current study.
In summary, detection of EGFR expression help to precisely subdivide high-risk GISTs for different prognosis and probably predict outcomes of imatinib treatment. HER4 is a novel independent prognostic biomarker for gastric GISTs specifically, which could be potential therapeutic target in GISTs originated from stomach.
Acknowledgements
National Natural Science Foundation of China (No. 81272743); Shanghai Committee of Science and Technology, China (No. 13XD1402500 and 13411950902).
Disclosure of conflict of interest
None.
References
- 1.Steigen SE, Eide TJ. Gastrointestinal stromal tumors (GIST): a review. APMIS. 2009;117:73–86. doi: 10.1111/j.1600-0463.2008.00020.x. [DOI] [PubMed] [Google Scholar]
- 2.Nilsson B, Bümming P, Meis-Kindblom JM, Odén A, Dortok A, Gustavsson B, Sablinska K, Kindblom LG. Gastrointestinal stromal tumors: the incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate era- a population-based study in western Sweden. Cancer. 2005;103:821–829. doi: 10.1002/cncr.20862. [DOI] [PubMed] [Google Scholar]
- 3.Mucciarini C, Rossi G, Bertolini F, Valli R, Cirilli C, Rashid I, Marcheselli L, Luppi G, Federico M. Incidence and clinicopathologic features of gastrointestinal stromal tumors. A population-based study. Bmc Cancer. 2007;7:230. doi: 10.1186/1471-2407-7-230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sandvik OM, Søreide K, Kvaløy JT, Gudlaugsson E, Søreide JA. Epidemiology of gastrointestinal stromal tumours: single-institution experience and clinical presentation over three decades. Cancer Epidemiol. 2011;35:515–520. doi: 10.1016/j.canep.2011.03.002. [DOI] [PubMed] [Google Scholar]
- 5.Joensuu H, Vehtari A, Riihimäki J, Nishida T, Steigen SE, Brabec P, Plank L, Nilsson B, Cirilli C, Braconi C, Bordoni A, Magnusson MK, Linke Z, Sufliarsky J, Federico M, Jonasson JG, Dei Tos AP, Rutkowski P. Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13:265–274. doi: 10.1016/S1470-2045(11)70299-6. [DOI] [PubMed] [Google Scholar]
- 6.Dematteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, Blackstein ME, Blanke CD, von Mehren M, Brennan MF, Patel S, McCarter MD, Polikoff JA, Tan BR, Owzar K. American College of Surgeons Oncology Group (ACOSOG) Intergroup Adjuvant GIST Study Team: Adjuvant imatinib mesylate after resection of localized, primary gastrointestinal stromal tumour: a randomized, double-blind, placebo-controlled trial. Lancet. 2009;373:1097–1104. doi: 10.1016/S0140-6736(09)60500-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Joensuu H, Eriksson M, Sundby Hall K, Hartmann JT, Pink D, Schütte J, Ramadori G, Hohenberger P, Duyster J, Al-Batran SE, Schlemmer M, Bauer S, Wardelmann E, Sarlomo-Rikala M, Nilsson B, Sihto H, Monge OR, Bono P, Kallio R, Vehtari A, Leinonen M, Alvegård T, Reichardt P. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307:1265–1272. doi: 10.1001/jama.2012.347. [DOI] [PubMed] [Google Scholar]
- 8.Van Glabbeke M, Verweij J, Casali PG, Le Cesne A, Hohenberger P, Ray-Coquard I, Schlemmer M, van Oosterom AT, Goldstein D, Sciot R, Hogendoorn PC, Brown M, Bertulli R, Judson IR. Initial and late resistance to imatinib in advanced gastrointestinal stromal tumors are predicted by different prognostic factors: a European Organization for Research and Treatment of Cancer-Italian Sarcoma Group-Australasian Gastrointestinal Trials Group study. J. Clin. Oncol. 2005;23:5795–5804. doi: 10.1200/JCO.2005.11.601. [DOI] [PubMed] [Google Scholar]
- 9.Corless CL, Barnett CM, Heinrich MC. Gastrointestinal stromal tumours: origin and molecular oncology. Nat Rev Cancer. 2011;11:865–78. doi: 10.1038/nrc3143. [DOI] [PubMed] [Google Scholar]
- 10.Tebbutt N, Pedersen MW, Johns TG. Targeting the ERBB family in cancer: couples therapy. Nat Rev Cancer. 2013;13:663–73. doi: 10.1038/nrc3559. [DOI] [PubMed] [Google Scholar]
- 11.Montemurro F, Scaltriti M. Biomarkers of drugs targeting HER-family signalling in cancer. J Pathol. 2014;232:219–29. doi: 10.1002/path.4269. [DOI] [PubMed] [Google Scholar]
- 12.Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411–1419. doi: 10.1016/j.humpath.2008.06.025. [DOI] [PubMed] [Google Scholar]
- 13.Edris B, Espinosa I, Mühlenberg T, Mikels A, Lee CH, Steigen SE, Zhu S, Montgomery KD, Lazar AJ, Lev D, Fletcher JA, Beck AH, West RB, Nusse R, van de Rijn M. ROR2 is a novel prognostic biomarker and a potential therapeutic target in leiomyosarcoma and gastrointestinal stromal tumour. J Pathol. 2012;227:223–33. doi: 10.1002/path.3986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235:177–82. doi: 10.1126/science.3798106. [DOI] [PubMed] [Google Scholar]
- 15.Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008;19:1523–9. doi: 10.1093/annonc/mdn169. [DOI] [PubMed] [Google Scholar]
- 16.Lopes LF, Bacchi CE. EGFR and gastrointestinal stromal tumor: an immunohistochemical and FISH study of 82 cases. Mod Pathol. 2007;20:990–4. doi: 10.1038/modpathol.3800932. [DOI] [PubMed] [Google Scholar]
- 17.Pinto AC, Ades F, de Azambuja E, Piccart-Gebhart M. Trastuzumab for patients with HER2 positive breast cancer: delivery, duration and combination therapies. Breast. 2013;22(Suppl 2):S152–5. doi: 10.1016/j.breast.2013.07.029. [DOI] [PubMed] [Google Scholar]
- 18.Yan M, Parker BA, Schwab R, Kurzrock R. HER2 aberrations in cancer: Implications for therapy. Cancer Treat Rev. 2014;40:770–780. doi: 10.1016/j.ctrv.2014.02.008. [DOI] [PubMed] [Google Scholar]
- 19.Ljuslinder I, Malmer B, Isaksson-Mettävainio M, Oberg A, Henriksson R, Stenling R, Palmqvist R. ErbB 1-4 expression alterations in primary colorectal cancers and their corresponding metastases. Anticancer Res. 2009;29:1489–94. [PubMed] [Google Scholar]
- 20.Rickman OB, Vohra PK, Sanyal B, Vrana JA, Aubry MC, Wigle DA, Thomas CF Jr. Analysis of ErbB receptors in pulmonary carcinoid tumors. Clin Cancer Res. 2009;15:3315–24. doi: 10.1158/1078-0432.CCR-08-2549. [DOI] [PubMed] [Google Scholar]
- 21.Gilmour LM, Macleod KG, McCaig A, Gullick WJ, Smyth JF, Langdon SP. Expression of erbB-4/HER-4 growth factor receptor isoforms in ovarian cancer. Cancer Res. 2001;61:2169–76. [PubMed] [Google Scholar]
- 22.Mendoza-Naranjo A, El-Naggar A, Wai DH, Mistry P, Lazic N, Ayala FR, da Cunha IW, Rodriguez-Viciana P, Cheng H, Tavares Guerreiro Fregnani JH, Reynolds P, Arceci RJ, Nicholson A, Triche TJ, Soares FA, Flanagan AM, Wang YZ, Strauss SJ, Sorensen PH. ERBB4 confers metastatic capacity in Ewing sarcoma. EMBO Mol Med. 2013;5:1019–34. doi: 10.1002/emmm.201202343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yu T, Li J, Yan M, Liu L, Lin H, Zhao F, Sun L, Zhang Y, Cui Y, Zhang F, Li J, He X, Yao M. MicroRNA-193a-3p and -5p suppress the metastasis of human non-small-cell lung cancer by downregulating the ERBB4/PIK3R3/mTOR/S6K2 signaling pathway. Oncogene. 2014 doi: 10.1038/onc.2013.574. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 24.Hollmén M, Määttä JA, Bald L, Sliwkowski MX, Elenius K. Suppression of breast cancer cell growth by a monoclonal antibody targeting cleavable ErbB4 isoforms. Oncogene. 2009;28:1309–19. doi: 10.1038/onc.2008.481. [DOI] [PubMed] [Google Scholar]
- 25.Starr A, Greif J, Vexler A, Ashkenazy-Voghera M, Gladesh V, Rubin C, Kerber G, Marmor S, Lev-Ari S, Inbar M, Yarden Y, Ben-Yosef R. ErbB4 increases the proliferation potential of human lung cancer cells and its blockage can be used as a target for anti-cancer therapy. Int J Cancer. 2006;119:269–74. doi: 10.1002/ijc.21818. [DOI] [PubMed] [Google Scholar]
- 26.Dematteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, Blackstein ME, Blanke CD, von Mehren M, Brennan MF, Patel S, McCarter MD, Polikoff JA, Tan BR, Owzar K. American College of Surgeons Oncology Group (ACOSOG) Intergroup Adjuvant GIST Study Team. Adjuvant imatinib mesylate after resection of localized, primary gastrointestinal stromal tumour: a randomized, double-blind, placebo-controlled trial. Lancet. 2009;373:1097–1104. doi: 10.1016/S0140-6736(09)60500-6. [DOI] [PMC free article] [PubMed] [Google Scholar]


