Abstract
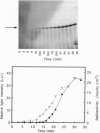
To compare the process of protein folding in the cell with refolding following denaturation in vitro, we have investigated and compared the kinetics of renaturation of a full-length protein upon dilution from concentrated urea with the rate of folding in the course of biosynthesis. Formation of enzymatically active bacterial luciferase, an alpha beta heterodimer, occurred 2 min after completion of beta-subunit synthesis in an Escherichia coli cell-free system. Renaturation of urea-denatured beta subunit, either in the presence of the cell-free protein synthesis system or in buffer solutions, proceeded more slowly. Cellular components present in the cell-free protein synthesis system slightly accelerated the rate of refolding of urea-unfolded beta subunit. The results indicate that the luciferase beta subunit begins the folding process cotranslationally and that cotranslational folding contributes to the rapid formation of the native structure in the cell.
Full text
PDF
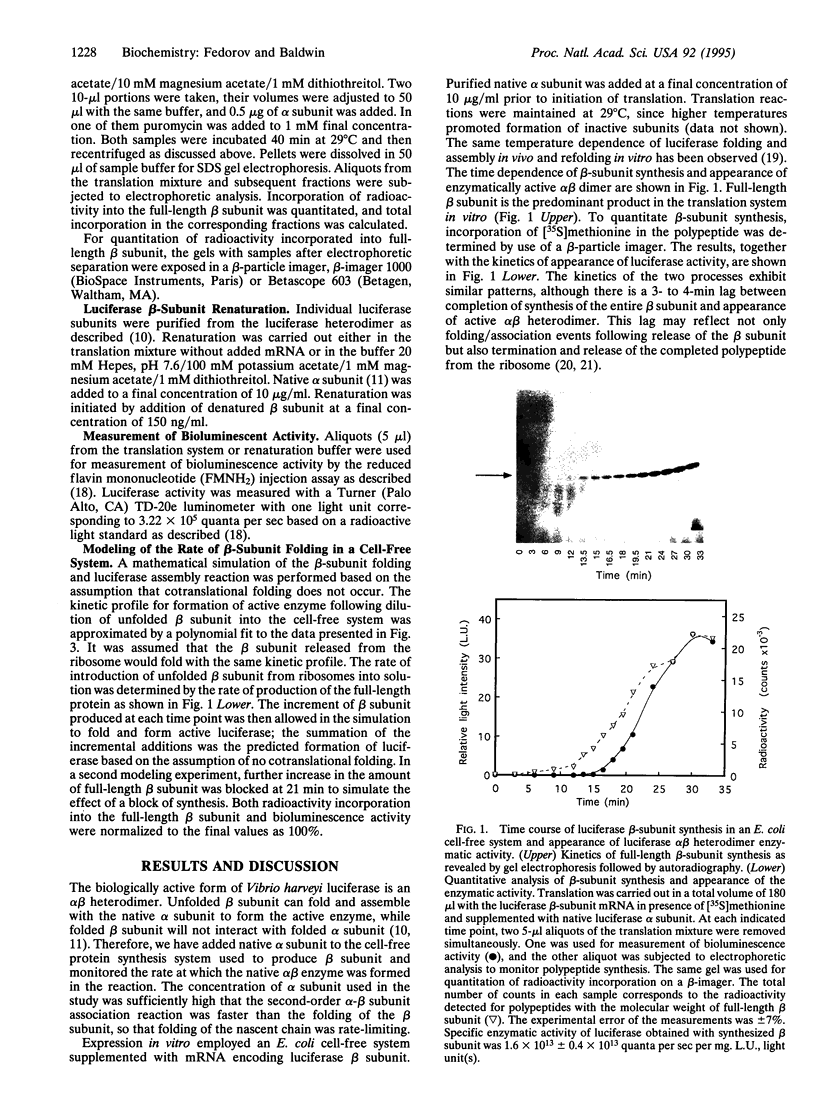
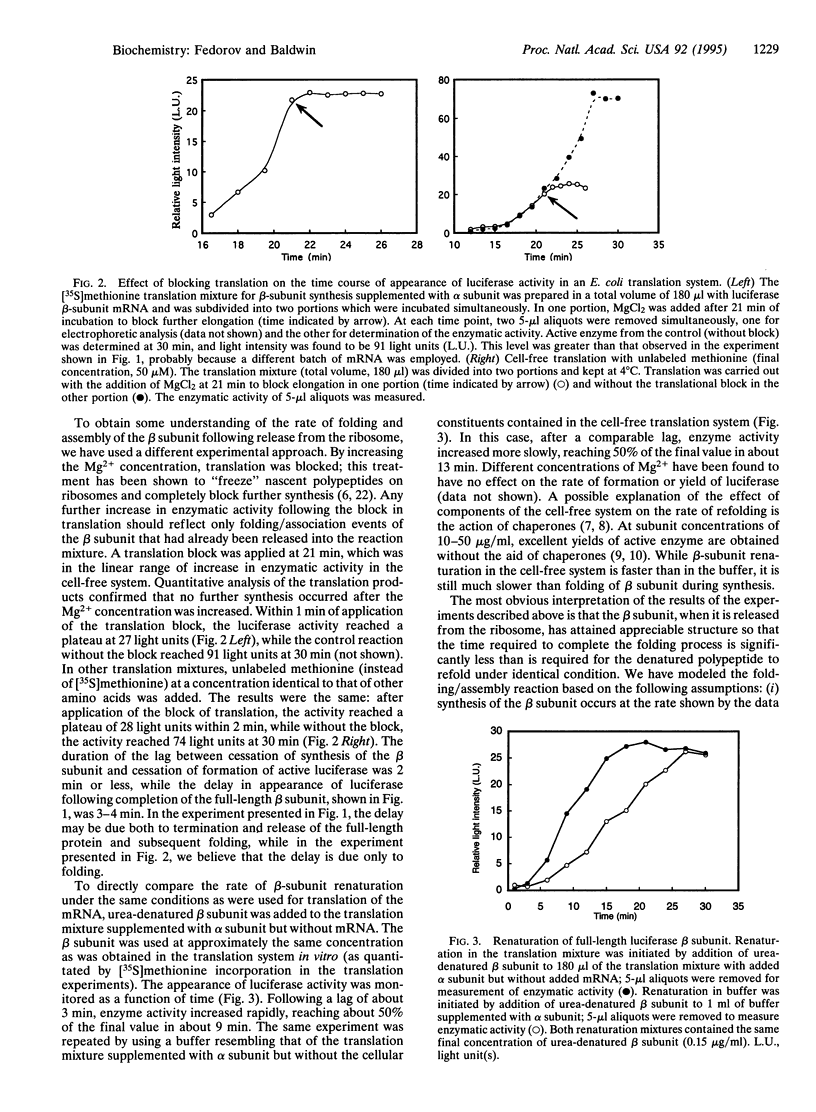
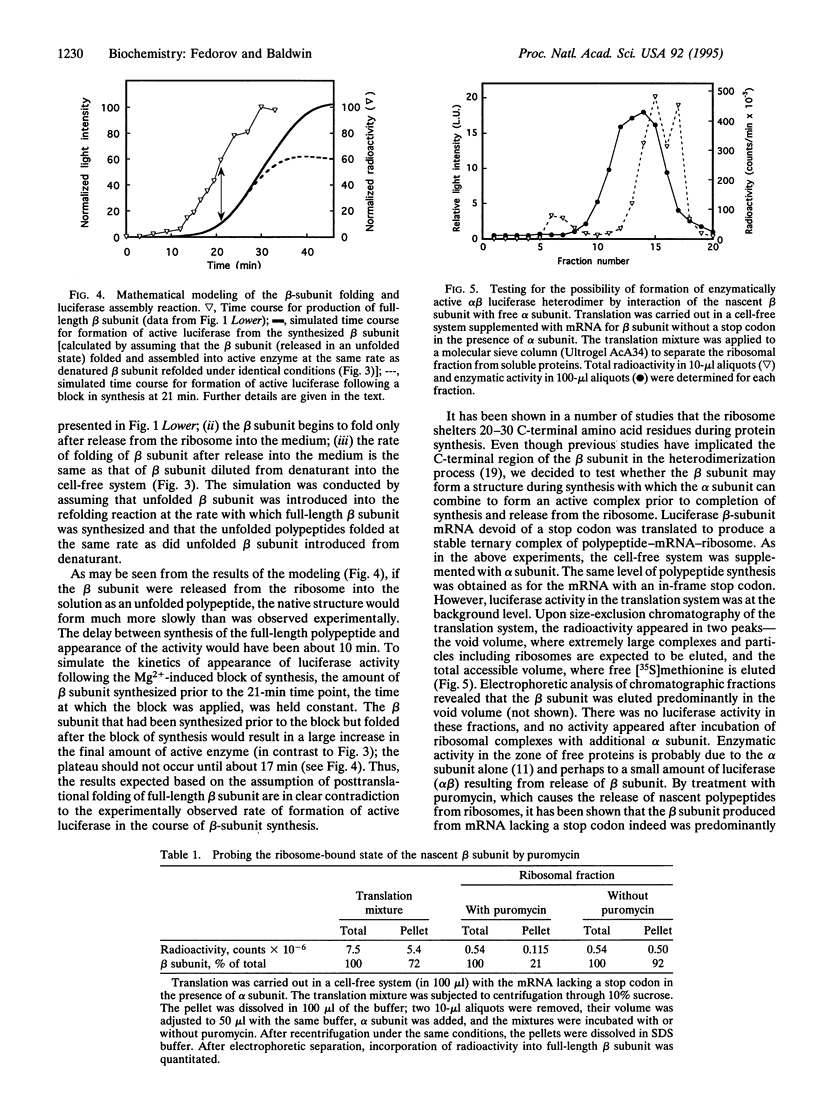

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baldwin T. O., Berends T., Bunch T. A., Holzman T. F., Rausch S. K., Shamansky L., Treat M. L., Ziegler M. M. Cloning of the luciferase structural genes from Vibrio harveyi and expression of bioluminescence in Escherichia coli. Biochemistry. 1984 Jul 31;23(16):3663–3667. doi: 10.1021/bi00311a014. [DOI] [PubMed] [Google Scholar]
- Baldwin T. O., Ziegler M. M., Chaffotte A. F., Goldberg M. E. Contribution of folding steps involving the individual subunits of bacterial luciferase to the assembly of the active heterodimeric enzyme. J Biol Chem. 1993 May 25;268(15):10766–10772. [PubMed] [Google Scholar]
- Baranov V. I., Morozov IYu, Ortlepp S. A., Spirin A. S. Gene expression in a cell-free system on the preparative scale. Gene. 1989 Dec 14;84(2):463–466. doi: 10.1016/0378-1119(89)90521-0. [DOI] [PubMed] [Google Scholar]
- Bergman L. W., Kuehl W. M. Formation of intermolecular disulfide bonds on nascent immunoglobulin polypeptides. J Biol Chem. 1979 Jul 10;254(13):5690–5694. [PubMed] [Google Scholar]
- Clark A. C., Sinclair J. F., Baldwin T. O. Folding of bacterial luciferase involves a non-native heterodimeric intermediate in equilibrium with the native enzyme and the unfolded subunits. J Biol Chem. 1993 May 25;268(15):10773–10779. [PubMed] [Google Scholar]
- Denisenko O. N. Reguliatsiia sinteza belkov v beskletochnoi sisteme pri teplovom shoke. Dokl Akad Nauk SSSR. 1987;297(3):725–727. [PubMed] [Google Scholar]
- Fedorov A. N., Friguet B., Djavadi-Ohaniance L., Alakhov Y. B., Goldberg M. E. Folding on the ribosome of Escherichia coli tryptophan synthase beta subunit nascent chains probed with a conformation-dependent monoclonal antibody. J Mol Biol. 1992 Nov 20;228(2):351–358. doi: 10.1016/0022-2836(92)90825-5. [DOI] [PubMed] [Google Scholar]
- Fischer G., Schmid F. X. The mechanism of protein folding. Implications of in vitro refolding models for de novo protein folding and translocation in the cell. Biochemistry. 1990 Mar 6;29(9):2205–2212. doi: 10.1021/bi00461a001. [DOI] [PubMed] [Google Scholar]
- Gething M. J., Sambrook J. Protein folding in the cell. Nature. 1992 Jan 2;355(6355):33–45. doi: 10.1038/355033a0. [DOI] [PubMed] [Google Scholar]
- Gurevich V. V., Pokrovskaya I. D., Obukhova T. A., Zozulya S. A. Preparative in vitro mRNA synthesis using SP6 and T7 RNA polymerases. Anal Biochem. 1991 Jun;195(2):207–213. doi: 10.1016/0003-2697(91)90318-n. [DOI] [PubMed] [Google Scholar]
- Jaenicke R. Folding and association of proteins. Prog Biophys Mol Biol. 1987;49(2-3):117–237. doi: 10.1016/0079-6107(87)90011-3. [DOI] [PubMed] [Google Scholar]
- KIHO Y., RICH A. INDUCED ENZYME FORMED ON BACTERIAL POLYRIBOSOMES. Proc Natl Acad Sci U S A. 1964 Jan;51:111–118. doi: 10.1073/pnas.51.1.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim P. S., Baldwin R. L. Intermediates in the folding reactions of small proteins. Annu Rev Biochem. 1990;59:631–660. doi: 10.1146/annurev.bi.59.070190.003215. [DOI] [PubMed] [Google Scholar]
- Kunkel T. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Proc Natl Acad Sci U S A. 1985 Jan;82(2):488–492. doi: 10.1073/pnas.82.2.488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Sinclair J. F., Waddle J. J., Waddill E. F., Baldwin T. O. Purified native subunits of bacterial luciferase are active in the bioluminescence reaction but fail to assemble into the alpha beta structure. Biochemistry. 1993 May 18;32(19):5036–5044. doi: 10.1021/bi00070a010. [DOI] [PubMed] [Google Scholar]
- Sugihara J., Baldwin T. O. Effects of 3' end deletions from the Vibrio harveyi luxB gene on luciferase subunit folding and enzyme assembly: generation of temperature-sensitive polypeptide folding mutants. Biochemistry. 1988 Apr 19;27(8):2872–2880. doi: 10.1021/bi00408a031. [DOI] [PubMed] [Google Scholar]
- Wolin S. L., Walter P. Ribosome pausing and stacking during translation of a eukaryotic mRNA. EMBO J. 1988 Nov;7(11):3559–3569. doi: 10.1002/j.1460-2075.1988.tb03233.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziegler M. M., Goldberg M. E., Chaffotte A. F., Baldwin T. O. Refolding of luciferase subunits from urea and assembly of the active heterodimer. Evidence for folding intermediates that precede and follow the dimerization step on the pathway to the active form of the enzyme. J Biol Chem. 1993 May 25;268(15):10760–10765. [PubMed] [Google Scholar]