Abstract
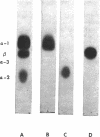
The major glycoprotein of the human erythrocyte membrane has been isolated by treatment with lithium di-iodosalicylate and found to be a single polypeptide chain with a molecular weight of about 50,000. This molecule, which is 60% carbohydrate and 40% protein, carries multiple blood-group antigens, the receptors for influenza viruses, and various plant agglutinins. Four unique carbohydrate-containing peptides (α-1, α-2, α-3, and β) are produced by tryptic digestion of the isolated glycoprotein; their order in the molecule has been determined by sequential tryptic digestion of intact erythrocyte membranes and partially digested glycoprotein fragments. Cleavage of the native protein with cyanogen bromide produces five fragments; two of these (C-5 and C-1) contain most of the carbohydrate in the molecule and are derived from the N-terminal half of the polypeptide chain. The nonpolar amino acids of this glycoprotein are located predominantly in the C-terminal fragment (C-2).
Phytohemagglutinin conjugated to ferritin has been used to map the distribution of glycoprotein receptors over the surfaces of intact erythrocytes by freeze-etching and electron microscopy. This label localizes to sites on the membrane that overlie the intramembranous particles. These findings suggest that the glycoprotein is oriented at the cell surface with its oligosaccharide-rich N-terminal end exposed to the exterior, while its C-terminal segment interacts with other components in the interior of the membrane to form intramembranous particles.
Keywords: tryptic peptides, cyanogen bromide peptides, intramembranous particles, receptors, blood-group antigens
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Branton D. Fracture faces of frozen membranes. Proc Natl Acad Sci U S A. 1966 May;55(5):1048–1056. doi: 10.1073/pnas.55.5.1048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bretscher M. S. Major human erythrocyte glycoprotein spans the cell membrane. Nat New Biol. 1971 Jun 23;231(25):229–232. doi: 10.1038/newbio231229a0. [DOI] [PubMed] [Google Scholar]
- COOK G. M., EYLAR E. H. SEPARATION OF THE M AND N BLOOD-GROUP ANTIGENS OF THE HUMAN ERYTHROCYTE. Biochim Biophys Acta. 1965 Mar 1;101:57–66. doi: 10.1016/0926-6534(65)90030-8. [DOI] [PubMed] [Google Scholar]
- Cook G. M. Glycoproteins in membranes. Biol Rev Camb Philos Soc. 1968 Aug;43(3):363–391. doi: 10.1111/j.1469-185x.1968.tb00964.x. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- DODGE J. T., MITCHELL C., HANAHAN D. J. The preparation and chemical characteristics of hemoglobin-free ghosts of human erythrocytes. Arch Biochem Biophys. 1963 Jan;100:119–130. doi: 10.1016/0003-9861(63)90042-0. [DOI] [PubMed] [Google Scholar]
- EYLAR E. H., MADOFF M. A., BRODY O. V., ONCLEY J. L. The contribution of sialic acid to the surface charge of the erythrocyte. J Biol Chem. 1962 Jun;237:1992–2000. [PubMed] [Google Scholar]
- Economidou J., Hughes-Jones N. C., Gardner B. Quantitative measurements concerning A and B antigen sites. Vox Sang. 1967 May;12(5):321–328. doi: 10.1111/j.1423-0410.1967.tb03362.x. [DOI] [PubMed] [Google Scholar]
- Hakomori S., Strycharz G. D. Investigations on cellular blood-group substances. I. Isolation and chemical composition of blood-group ABH and Le-b isoantigens of sphingoglycolipid nature. Biochemistry. 1968 Apr;7(4):1279–1286. doi: 10.1021/bi00844a005. [DOI] [PubMed] [Google Scholar]
- KATHAN R. H., WINZLER R. J., JOHNSOM C. A. Preparation of an inhibitor of viral hemagglutination from human erythrocytes. J Exp Med. 1961 Jan 1;113:37–45. doi: 10.1084/jem.113.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornfeld S., Kornfeld R. Solubilization and partial characterization of a phytohemagglutinin receptor site from human erythrocytes. Proc Natl Acad Sci U S A. 1969 Aug;63(4):1439–1446. doi: 10.1073/pnas.63.4.1439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laico M. T., Ruoslahti E. I., Papermaster D. S., Dreyer W. J. Isolation of the fundamental polypeptide subunits of biological membranes. Proc Natl Acad Sci U S A. 1970 Sep;67(1):120–127. doi: 10.1073/pnas.67.1.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenard J. Protein and glycolipid components of human erythrocyte membranes. Biochemistry. 1970 Mar 3;9(5):1129–1132. doi: 10.1021/bi00807a012. [DOI] [PubMed] [Google Scholar]
- MORAWIECKI A. DISSOCIATION OF M- AND N-GROUP MUCOPROTEINS INTO SUBUNITS IN DETERGENT SOLUTIONS. Biochim Biophys Acta. 1964 Nov 1;83:339–347. doi: 10.1016/0926-6526(64)90012-6. [DOI] [PubMed] [Google Scholar]
- Maddy A. H. The properties of the protein of the plasma membrane of ox erythrocytes. Biochim Biophys Acta. 1966 Mar 28;117(1):193–200. doi: 10.1016/0304-4165(66)90166-8. [DOI] [PubMed] [Google Scholar]
- Maizel J. V., Jr Acrylamide-gel electrophorograms by mechanical fractionation: radioactive adenovirus proteins. Science. 1966 Feb 25;151(3713):988–990. doi: 10.1126/science.151.3713.988. [DOI] [PubMed] [Google Scholar]
- Marchesi V. T., Andrews E. P. Glycoproteins: isolation from cellmembranes with lithium diiodosalicylate. Science. 1971 Dec 17;174(4015):1247–1248. doi: 10.1126/science.174.4015.1247. [DOI] [PubMed] [Google Scholar]
- Nicolson G. L., Singer S. J. Ferritin-conjugated plant agglutinins as specific saccharide stains for electron microscopy: application to saccharides bound to cell membranes. Proc Natl Acad Sci U S A. 1971 May;68(5):942–945. doi: 10.1073/pnas.68.5.942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phillips D. R., Morrison M. Exposed protein on the intact human erythrocyte. Biochemistry. 1971 May 11;10(10):1766–1771. doi: 10.1021/bi00786a006. [DOI] [PubMed] [Google Scholar]
- Pinto da Silva P., Douglas S. D., Branton D. Localization of A antigen sites on human erythrocyte ghosts. Nature. 1971 Jul 16;232(5307):194–196. doi: 10.1038/232194a0. [DOI] [PubMed] [Google Scholar]
- Poulik M. D., Lauf P. K. Heterogeneity of water-soluble structural components of human red cell membrane. Nature. 1965 Nov 27;208(5013):874–876. doi: 10.1038/208874a0. [DOI] [PubMed] [Google Scholar]
- Segrest J. P., Jackson R. L., Andrews E. P., Marchesi V. T. Human erythrocyte membrane glycoprotein: a re-evaluation of the molecular weight as determined by SDS polyacrylamide gel electrophoresis. Biochem Biophys Res Commun. 1971 Jul 16;44(2):390–395. doi: 10.1016/0006-291x(71)90612-7. [DOI] [PubMed] [Google Scholar]
- Springer G. F., Nagai Y., Tegtmeyer H. Isolation and properties of human blood-group NN and meconium-Vg antigens. Biochemistry. 1966 Oct;5(10):3254–3272. doi: 10.1021/bi00874a028. [DOI] [PubMed] [Google Scholar]
- Tillack T. W., Marchesi V. T. Demonstration of the outer surface of freeze-etched red blood cell membranes. J Cell Biol. 1970 Jun;45(3):649–653. doi: 10.1083/jcb.45.3.649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WARREN L. The thiobarbituric acid assay of sialic acids. J Biol Chem. 1959 Aug;234(8):1971–1975. [PubMed] [Google Scholar]
- Weber T., Nordman C. T., Gräsbeck R. Separation of lymphocyte-stimulating and agglutinating activities in phytohaemagglutinin (PHA) from Phaseolus vulgaris. Scand J Haematol. 1967;4(1):77–80. doi: 10.1111/j.1600-0609.1967.tb01601.x. [DOI] [PubMed] [Google Scholar]
- Whittemore N. B., Trabold N. C., Reed C. F., Weed R. I. Solubilized glycoprotein from human erythrocyte membranes possessing blood group A, B and H activity. Vox Sang. 1969 Oct;17(4):289–299. doi: 10.1111/j.1423-0410.1969.tb00398.x. [DOI] [PubMed] [Google Scholar]
- Winzler R. J. Carbohydrates in cell surfaces. Int Rev Cytol. 1970;29:77–125. doi: 10.1016/s0074-7696(08)60033-9. [DOI] [PubMed] [Google Scholar]