Abstract
Starting with assignments of proton nuclear magnetic resonance previously made for oxytocin in deuterated dimethylsulfoxide at 220 MHz, we have assigned resonances for the mammalian antidiuretic hormone, lysine vasopressin. The results demonstrate that spectral assignments of neurohypophyseal hormones and their congeners can, within certain limits, be derived from each other. Comparison of the spectra of lysine vasopressin and oxytocin suggests that the gross backbone conformations of their 20-membered ring components are for the most part similar in deuterated dimethylsulfoxide, whereas the C-terminal acyclic amino-acid sequence of lysine vasopressin is more flexible than that of oxytocin.
Keywords: proton magnetic resonance, neurohypophyseal hormones, three-dimensional structure, pseudo-frequency sweep decoupling, spectral assignments
Full text
PDF

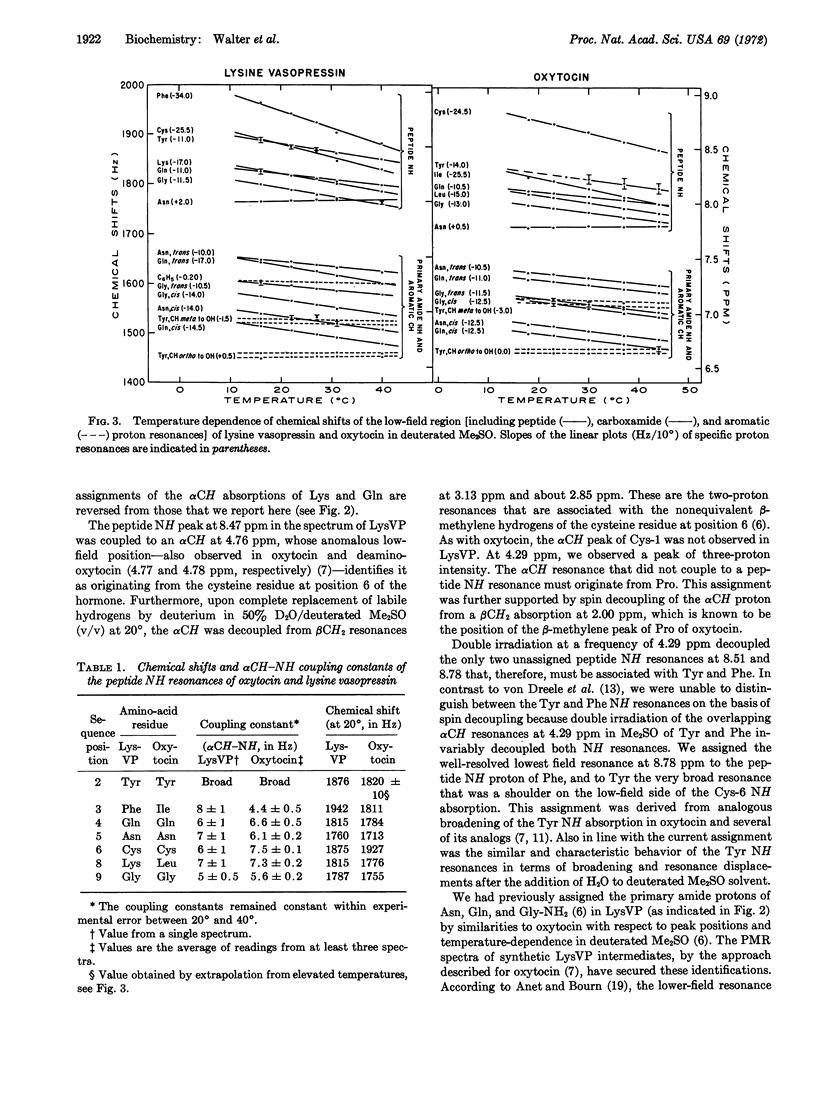


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bystrov V. F., Portnova S. L., Tsetlin V. I., Ivanov V. T., Ovchinnikov Y. A. Conformational studies of peptide systems. The rotational states of the NH--CH fragment of alanine dipeptides by nuclear magnetic resonance. Tetrahedron. 1969 Feb;25(3):493–515. doi: 10.1016/s0040-4020(01)83261-0. [DOI] [PubMed] [Google Scholar]
- Deber C. M., Bovey F. A., Carver J. P., Blout E. R. Nuclear magnetic resonance evidence for cis-peptide bonds in proline oligomers. J Am Chem Soc. 1970 Oct 21;92(21):6191–6198. doi: 10.1021/ja00724a016. [DOI] [PubMed] [Google Scholar]
- Deslauriers R., Smith I. C. Evidence from proton magnetic resonance data for the stacking of aromatic amino acids in lysine-vasopressin: comparison with oxytocin derivatives and related dipeptides. Biochem Biophys Res Commun. 1970 Jul 13;40(1):179–185. doi: 10.1016/0006-291x(70)91063-6. [DOI] [PubMed] [Google Scholar]
- Ferguson D. R., Heller H. Distribution of neurohypophysial hormones in mammals. J Physiol. 1965 Oct;180(4):846–863. doi: 10.1113/jphysiol.1965.sp007735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geddes A. J., Parker K. D., Atkins E. D., Beighton E. "Cross-beta" conformation in proteins. J Mol Biol. 1968 Mar 14;32(2):343–358. doi: 10.1016/0022-2836(68)90014-4. [DOI] [PubMed] [Google Scholar]
- Gibbons W. A., Némethy G., Stern A., Craig L. C. An approach to conformational analysis of peptides and proteins in solution based on a combination of nuclear magnetic resonance spectroscopy and conformational energy calculations. Proc Natl Acad Sci U S A. 1970 Sep;67(1):239–246. doi: 10.1073/pnas.67.1.239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HODGKIN D. C., OUGHTON B. M. Possible molecular models for gramicidin S and their relationship to present ideas of protein structure. Biochem J. 1957 Apr;65(4):752–756. doi: 10.1042/bj0650752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson L. F., Schwartz I. L., Walter R. Oxytocin and neurohypophyseal peptides: spectral assignment and conformational analysis by 220 MHz nuclear magnetic resonance. Proc Natl Acad Sci U S A. 1969 Dec;64(4):1269–1275. doi: 10.1073/pnas.64.4.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kopple K. D., Ohnishi M., Go A. Conformations of cyclic peptides. IV. Nuclear magnetic resonance studies of cyclo-pentaglycyl-L-leucyl and cyclo-diglycyl-L-histidyldiglycyl-L-tyrosyl. Biochemistry. 1969 Oct;8(10):4087–4095. doi: 10.1021/bi00838a028. [DOI] [PubMed] [Google Scholar]
- McDonald C. C., Phillips W. D. Proton magnetic resonance spectra of proteins in random-coil configurations. J Am Chem Soc. 1969 Mar 12;91(6):1513–1521. doi: 10.1021/ja01034a039. [DOI] [PubMed] [Google Scholar]
- Meienhofer J., Sano Y. A solid-phase synthesis of [lysine]-vasopressin through a crystalline protected nonapeptide intermediate. J Am Chem Soc. 1968 May 22;90(11):2996–2997. doi: 10.1021/ja01013a067. [DOI] [PubMed] [Google Scholar]
- Ohnishi M., Urry D. W. Temperature dependence of amide proton chemical shifts: the secondary structures of gramicidin S and valinomycin. Biochem Biophys Res Commun. 1969 Jul 23;36(2):194–202. doi: 10.1016/0006-291x(69)90314-3. [DOI] [PubMed] [Google Scholar]
- Pinkerton M., Steinrauf L. K., Dawkins P. The molecular structure and some transport properties of valinomycin. Biochem Biophys Res Commun. 1969 May 22;35(4):512–518. doi: 10.1016/0006-291x(69)90376-3. [DOI] [PubMed] [Google Scholar]
- Ramachandran G. N., Chandrasekaran R., Kopple K. D. Variation of the NH-C alpha-H coupling constant with dihedral angle in the NMR spectra of peptides. Biopolymers. 1971 Nov;10(11):2113–2131. doi: 10.1002/bip.360101108. [DOI] [PubMed] [Google Scholar]
- Rudko A. D., Lovell F. M., Low B. W. Prolactin synthesis in primates. Nat New Biol. 1971 Jul 7;232(27):18–19. [PubMed] [Google Scholar]
- SCHWARTZ I. L., LIVINGSTON L. M. CELLULAR AND MOLECULAR ASPECTS OF THE ANTIDIURETIC ACTION OF VASOPRESSINS AND RELATED PEPTIDES. Vitam Horm. 1964;22:261–358. doi: 10.1016/s0083-6729(08)60341-6. [DOI] [PubMed] [Google Scholar]
- Stern A., Gibbons W. A., Craig L. C. A conformational analysis of gramicidin S-A by nuclear magnetic resonance. Proc Natl Acad Sci U S A. 1968 Oct;61(2):734–741. doi: 10.1073/pnas.61.2.734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urry D. W., Ohnishi M., Walter R. Secondary structure of the cyclic moiety of the peptide hormone oxytocin and its deamino analog. Proc Natl Acad Sci U S A. 1970 May;66(1):111–116. doi: 10.1073/pnas.66.1.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urry D. W., Quadrifoglio F., Walter R., Schwartz I. L. Conformational studies on neurohypophyseal hormones: the disulfide bridge of oxytocin. Proc Natl Acad Sci U S A. 1968 Jul;60(3):967–974. doi: 10.1073/pnas.60.3.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urry D. W., Walter R. Proposed conformation of oxytocin in solution. Proc Natl Acad Sci U S A. 1971 May;68(5):956–958. doi: 10.1073/pnas.68.5.956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venkatachalam C. M. Stereochemical criteria for polypeptides and proteins. V. Conformation of a system of three linked peptide units. Biopolymers. 1968 Oct;6(10):1425–1436. doi: 10.1002/bip.1968.360061006. [DOI] [PubMed] [Google Scholar]
- Von Dreele P. H., Brewster A. I., Bovey F. A., Scheraga H. A., Ferger M. F., Du Vigneaud V. Nuclear magnetic resonance studies of lysine-vasopressin: structural constraints. Proc Natl Acad Sci U S A. 1971 Dec;68(12):3088–3091. doi: 10.1073/pnas.68.12.3088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Von Dreele P. H., Brewster A. I., Scheraga H. A., Ferger M. F., Du Vigneaud V. Nuclear magnetic resonance spectrum of lysine-vasopressin and its structural implications. Proc Natl Acad Sci U S A. 1971 May;68(5):1028–1031. doi: 10.1073/pnas.68.5.1028. [DOI] [PMC free article] [PubMed] [Google Scholar]


