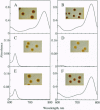
Abstract
Protochlorophyllide reductase (NADPH:protochlorophyllide oxidoreductase; EC 1.6.99.1) catalyzes the light-dependent reduction of protochlorophyllide to chlorophyllide, a key regulatory step in the chlorophyll biosynthetic pathway. We have developed an expression system in which the protochlorophyllide reductase from pea (Pisum sativum L.) is used to complement protochlorophyllide reduction mutants in the photosynthetic bacterium Rhodobacter capsulatus, allowing analysis of wild-type and mutant forms of the enzyme. By protein sequence comparisons, we have identified the plant protochlorophyllide reductases as belonging to the family of short-chain alcohol dehydrogenases. Based on our protein sequence alignments, we have identified and mutated two conserved residues (Tyr-275 and Lys-279) within the proposed active site of the enzyme and shown that they are critical for activity. A model of the enzyme reaction mechanism for light-dependent protochlorophyllide reduction is proposed.
Full text
PDF


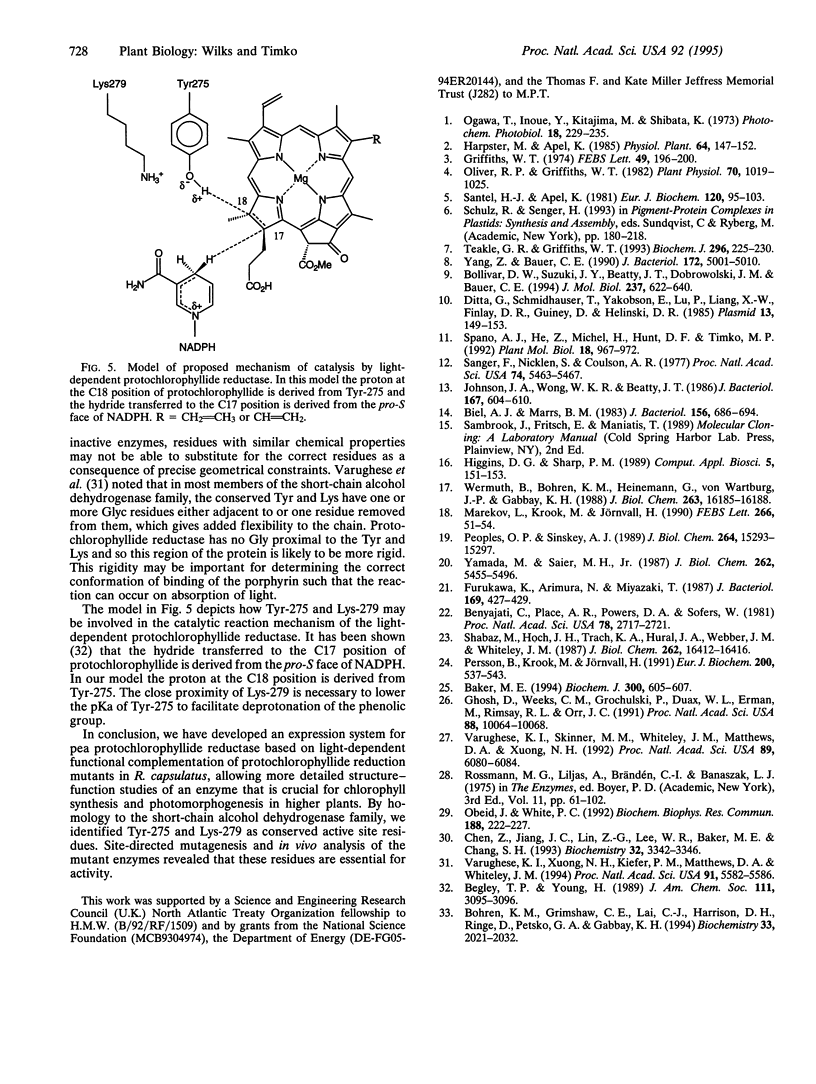
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baker M. E. Protochlorophyllide reductase is homologous to human carbonyl reductase and pig 20 beta-hydroxysteroid dehydrogenase. Biochem J. 1994 Jun 1;300(Pt 2):605–607. doi: 10.1042/bj3000605b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benyajati C., Place A. R., Powers D. A., Sofer W. Alcohol dehydrogenase gene of Drosophila melanogaster: relationship of intervening sequences to functional domains in the protein. Proc Natl Acad Sci U S A. 1981 May;78(5):2717–2721. doi: 10.1073/pnas.78.5.2717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biel A. J., Marrs B. L. Transcriptional regulation of several genes for bacteriochlorophyll biosynthesis in Rhodopseudomonas capsulata in response to oxygen. J Bacteriol. 1983 Nov;156(2):686–694. doi: 10.1128/jb.156.2.686-694.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohren K. M., Grimshaw C. E., Lai C. J., Harrison D. H., Ringe D., Petsko G. A., Gabbay K. H. Tyrosine-48 is the proton donor and histidine-110 directs substrate stereochemical selectivity in the reduction reaction of human aldose reductase: enzyme kinetics and crystal structure of the Y48H mutant enzyme. Biochemistry. 1994 Mar 1;33(8):2021–2032. doi: 10.1021/bi00174a007. [DOI] [PubMed] [Google Scholar]
- Bollivar D. W., Suzuki J. Y., Beatty J. T., Dobrowolski J. M., Bauer C. E. Directed mutational analysis of bacteriochlorophyll a biosynthesis in Rhodobacter capsulatus. J Mol Biol. 1994 Apr 15;237(5):622–640. doi: 10.1006/jmbi.1994.1260. [DOI] [PubMed] [Google Scholar]
- Chen Z., Jiang J. C., Lin Z. G., Lee W. R., Baker M. E., Chang S. H. Site-specific mutagenesis of Drosophila alcohol dehydrogenase: evidence for involvement of tyrosine-152 and lysine-156 in catalysis. Biochemistry. 1993 Apr 6;32(13):3342–3346. doi: 10.1021/bi00064a017. [DOI] [PubMed] [Google Scholar]
- Ditta G., Schmidhauser T., Yakobson E., Lu P., Liang X. W., Finlay D. R., Guiney D., Helinski D. R. Plasmids related to the broad host range vector, pRK290, useful for gene cloning and for monitoring gene expression. Plasmid. 1985 Mar;13(2):149–153. doi: 10.1016/0147-619x(85)90068-x. [DOI] [PubMed] [Google Scholar]
- Furukawa K., Arimura N., Miyazaki T. Nucleotide sequence of the 2,3-dihydroxybiphenyl dioxygenase gene of Pseudomonas pseudoalcaligenes. J Bacteriol. 1987 Jan;169(1):427–429. doi: 10.1128/jb.169.1.427-429.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh D., Weeks C. M., Grochulski P., Duax W. L., Erman M., Rimsay R. L., Orr J. C. Three-dimensional structure of holo 3 alpha,20 beta-hydroxysteroid dehydrogenase: a member of a short-chain dehydrogenase family. Proc Natl Acad Sci U S A. 1991 Nov 15;88(22):10064–10068. doi: 10.1073/pnas.88.22.10064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths W. T. Protochlorophyll and protochlorophyllide as precursors for chlorophyll synthesis in vitro. FEBS Lett. 1974 Dec 15;49(2):196–200. doi: 10.1016/0014-5793(74)80510-7. [DOI] [PubMed] [Google Scholar]
- Higgins D. G., Sharp P. M. Fast and sensitive multiple sequence alignments on a microcomputer. Comput Appl Biosci. 1989 Apr;5(2):151–153. doi: 10.1093/bioinformatics/5.2.151. [DOI] [PubMed] [Google Scholar]
- Johnson J. A., Wong W. K., Beatty J. T. Expression of cellulase genes in Rhodobacter capsulatus by use of plasmid expression vectors. J Bacteriol. 1986 Aug;167(2):604–610. doi: 10.1128/jb.167.2.604-610.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marekov L., Krook M., Jörnvall H. Prokaryotic 20 beta-hydroxysteroid dehydrogenase is an enzyme of the 'short-chain, non-metalloenzyme' alcohol dehydrogenase type. FEBS Lett. 1990 Jun 18;266(1-2):51–54. doi: 10.1016/0014-5793(90)81504-h. [DOI] [PubMed] [Google Scholar]
- Obeid J., White P. C. Tyr-179 and Lys-183 are essential for enzymatic activity of 11 beta-hydroxysteroid dehydrogenase. Biochem Biophys Res Commun. 1992 Oct 15;188(1):222–227. doi: 10.1016/0006-291x(92)92373-6. [DOI] [PubMed] [Google Scholar]
- Oliver R. P., Griffiths W. T. Pigment-protein complexes of illuminated etiolated leaves. Plant Physiol. 1982 Oct;70(4):1019–1025. doi: 10.1104/pp.70.4.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peoples O. P., Sinskey A. J. Poly-beta-hydroxybutyrate biosynthesis in Alcaligenes eutrophus H16. Characterization of the genes encoding beta-ketothiolase and acetoacetyl-CoA reductase. J Biol Chem. 1989 Sep 15;264(26):15293–15297. [PubMed] [Google Scholar]
- Persson B., Krook M., Jörnvall H. Characteristics of short-chain alcohol dehydrogenases and related enzymes. Eur J Biochem. 1991 Sep 1;200(2):537–543. doi: 10.1111/j.1432-1033.1991.tb16215.x. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santel H. J., Apel K. The protochlorophyllide holochrome of barley (Hordeum vulgare L.). The effect of light on the NADPH:protochlorophyllide oxidoreductase. Eur J Biochem. 1981 Nov;120(1):95–103. doi: 10.1111/j.1432-1033.1981.tb05674.x. [DOI] [PubMed] [Google Scholar]
- Shahbaz M., Hoch J. A., Trach K. A., Hural J. A., Webber S., Whiteley J. M. Structural studies and isolation of cDNA clones providing the complete sequence of rat liver dihydropteridine reductase. J Biol Chem. 1987 Dec 5;262(34):16412–16416. [PubMed] [Google Scholar]
- Spano A. J., He Z., Michel H., Hunt D. F., Timko M. P. Molecular cloning, nuclear gene structure, and developmental expression of NADPH: protochlorophyllide oxidoreductase in pea (Pisum sativum L.). Plant Mol Biol. 1992 Mar;18(5):967–972. doi: 10.1007/BF00019210. [DOI] [PubMed] [Google Scholar]
- Teakle G. R., Griffiths W. T. Cloning, characterization and import studies on protochlorophyllide reductase from wheat (Triticum aestivum). Biochem J. 1993 Nov 15;296(Pt 1):225–230. doi: 10.1042/bj2960225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varughese K. I., Skinner M. M., Whiteley J. M., Matthews D. A., Xuong N. H. Crystal structure of rat liver dihydropteridine reductase. Proc Natl Acad Sci U S A. 1992 Jul 1;89(13):6080–6084. doi: 10.1073/pnas.89.13.6080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varughese K. I., Xuong N. H., Kiefer P. M., Matthews D. A., Whiteley J. M. Structural and mechanistic characteristics of dihydropteridine reductase: a member of the Tyr-(Xaa)3-Lys-containing family of reductases and dehydrogenases. Proc Natl Acad Sci U S A. 1994 Jun 7;91(12):5582–5586. doi: 10.1073/pnas.91.12.5582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wermuth B., Bohren K. M., Heinemann G., von Wartburg J. P., Gabbay K. H. Human carbonyl reductase. Nucleotide sequence analysis of a cDNA and amino acid sequence of the encoded protein. J Biol Chem. 1988 Nov 5;263(31):16185–16188. [PubMed] [Google Scholar]
- Yamada M., Saier M. H., Jr Glucitol-specific enzymes of the phosphotransferase system in Escherichia coli. Nucleotide sequence of the gut operon. J Biol Chem. 1987 Apr 25;262(12):5455–5463. [PubMed] [Google Scholar]
- Yang Z. M., Bauer C. E. Rhodobacter capsulatus genes involved in early steps of the bacteriochlorophyll biosynthetic pathway. J Bacteriol. 1990 Sep;172(9):5001–5010. doi: 10.1128/jb.172.9.5001-5010.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]