Abstract
Allergen-specific IgE serology tests became commercially available in the 1980s. Since then these tests have been widely used to diagnose and treat allergic skin diseases. However, the relationship between a positive reaction and disease occurrence has been controversial. The purpose of this study was to evaluate allergens using a serologic allergy test in dogs with atopic dermatitis (AD). Dogs clinically diagnosed with AD (n=101) were tested using an allergen-specific IgE immunoassay. Among the total 92 environmental and food allergens, house dust and house dust mites were the most common. Several allergens including airborne pollens and molds produced positive reactions, and which was considered increasing allergens relating to the climate changes. The presence of antibodies against staphylococci and Malassezia in cases of canine AD was warranted in this study. Additionally, strong (chicken, turkey, brown rice, brewer's yeast, and soybean) and weakly (rabbit, vension, duck, and tuna) positive reactions to food allergens could be used for avoidance and limited-allergen trials.
Keywords: atopic dermatitis, dogs, IgE, serologic allergy test
Introduction
Atopic dermatitis (AD) is a genetically predisposed pruritic inflammatory allergic skin disease that is common among humans. The incidence of AD has increased rapidly worldwide during the past 30 years [19]. As previously described [15], this condition is also common in dogs and recognized as a multifactorial disease involving IgE-mediated allergic reactions to various environmental allergens, alterations of skin barrier function, microbial colonization, and abnormal cutaneous immunity. In general, canine AD is diagnosed based on medical history, physical examination, exclusion of other diseases that cause similar clinical signs, and allergy test results [11]. Among these, the intradermal skin test (IDST) is considered the gold standard. However, it seems that IDST for dogs and cats is not practical for general practitioners. Allergen-specific IgE serology is now widely used in clinical practice [4]. In general, allergen-specific IgE serology appears to produce false positive results (low specificity) and cannot be used to confirm a diagnosis, but offers useful information about avoidance allergens and the inclusion of allergens for allergen-specific immunotherapy [4,15]. Causative allergens vary according to geographical region, climate, environmental pollution/hygiene, and residential environments [7,19]. Allergen-specific IgE serology is also required for large-scale investigations of canine AD. The purpose of the current study was to measure the sensitization rates of environmental allergens in dogs with AD using an allergen-specific IgE serology test and identify the causative allergens.
Materials and Methods
Criteria for animal selection
A total of 101 dogs admitted to the Konkuk University Veterinary Medical Teaching Hospital (Korea) between 2003 and 2010 were included in this study. All animals were privately owned patients that had been diagnosed with AD based on clinical history, signs, and diagnostic criteria. Each dogs fulfilled three major plus three minor criteria described by Willemse [27], three criteria noted by Prélaud et al. [21], and at least five criteria described by Farvot [18]. Administration of anti-allergy drugs such as antihistamines, glucocorticoids, and cyclosporine had to be discontinued at least 4 weeks prior to enrolment.
Exclusion criteria
Exclusion of animals with other pruritic skin disease including bacterial/fungal skin infection, yeast overgrowth, and ectoparasite skin disease was performed using appropriate diagnostic tests (multiple skin scrapings, impression cytology, acetate tape preparations, otic examination, and fungal culture). A strict hypoallergenic hydrolyzed commercial diet was administered for at least 8 weeks, and responses to external parasite control were also used as methods to rule out food allergy dermatitis and ectoparasite skin disease. Furthermore, AD dogs with concurrent conditions that required treatment (oral and/or topical anti-allergy drugs) were excluded from the study.
Serologic allergy test (SAT)
Serological allergen-specific IgE levels were measured with an ELISA using a spot test (Spectrum Laboratories, USA). Blood was drawn by jugular and cephalic venipuncture, allowed to clot at room temperature, and centrifuged at 2,000 × g for 15 min at 4℃. Serum samples were collected, frozen at -20℃, and shipped to Spectrum Laboratory within 2 days after obtaining the blood. A complete IgE panel assay using a polyclonal anti-IgE that minimized non-specific IgG and IgE binding was performed. Test results ranged from 0 to 10,000 units. Results over 200 units were considered positive based on the manufacturer's recommendations. Ninety-two allergens belonging to a total of 12 groups were tested. The groups include grasses, trees, weeds, mold, epidermals, house dust, foods, indoor allergens, insects, fleas, Staphylococcus, and Malassezia.
Results
Forty-six out of 101 dogs (45.5%) were males, seven (15.2%) of which were castrated. Fifty-five of the 101 dogs (54.5%) were females, four (7.3%) of which were spayed. The ratio of male : female animals was 1 : 1.2. There were 16 different breeds included in the study: mixed (n = 13), cocker spaniel (n = 15), Yorkshire terrier (n = 13), Shih Tzu (n = 13), Maltese (n = 12), Pekingese (n = 4), miniature pinscher (n = 4), beagle (n = 4), Dachshund (n = 3), miniature Schnauzer (n = 3), pug, Cavalier King Charles spaniel, Jindo dog, Chihuahua, and Bichon Firse (one each). Animal age at presentation ranged from 5 months to 13 years (median, 3 years) and most dogs were less than 5 years old (77.9%). The dogs developed clinical signs and skin lesions between 6 months and 2 years, and duration of the clinical signs prior to diagnosis ranged from 2 weeks to 8 years.
Environmental allergens
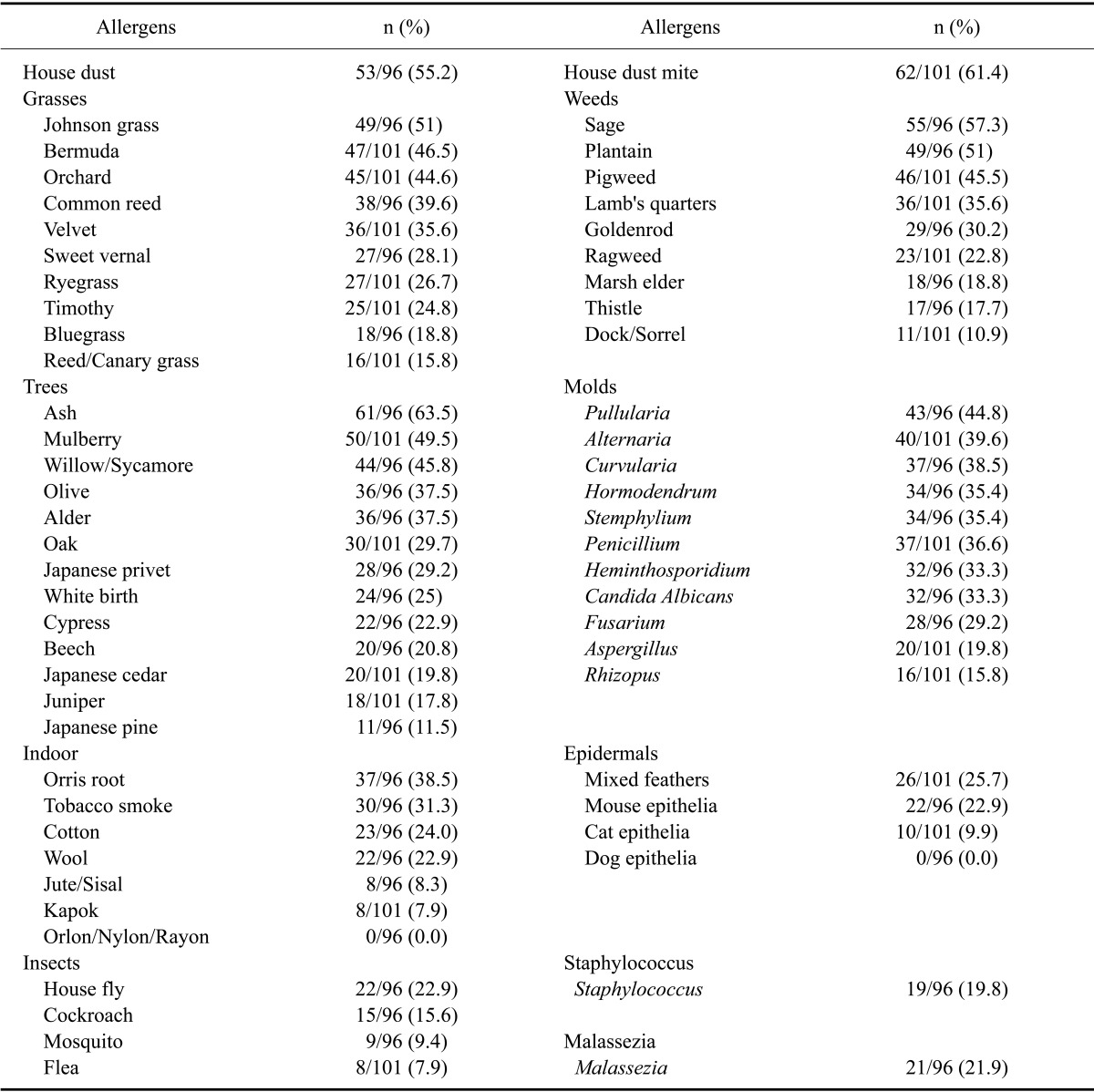
The overall sensitization rates of the tested allergens are shown in Table 1.
Table 1.
Sensitization rates for environmental allergens of each group of atopic dermatitis (AD) dogs
n: number of positive reactions/number of examinations.
House dust and house dust mites
The sensitization rates for house dust and house dust mites were 55.2% and 61.4%. These rates were higher compared to those of other allergens in this study.
Airborne pollen (grass, weeds, and trees)
Among the three types of airborne pollens, the most common offending allergen was pollen from ash (63.5%) followed that produced by sage (57.3%), Johnson grass (51%), plaintain (51%), mulberry (49.5%), Bermuda grass (46.5%), willow (45.8%), and pigweed (45.5%). The sensitization rates for Japanese pine allergens and dock/sorrel (Rumex) were lower (11.5% and 10.9%, respectively) compared to those of the other allergens.
Molds
The sensitization rate for pullularia and alternaria were 44.8% and 39.6%, respectively; these rates were higher than those for other mold allergens. Additional important molds were curvularia, penicillium, hormodendrum, stemphylium, heinthosporidium, and candida albicans. The lowest sensitization rate was that for rhizopus (15.8%).
Household inhalants (epithelials and indoor allergens)
Epithelials had a lower sensitization rate compared to other groups of allergens. Positive reactions to both mixed feather and mouse epithelials were 25.7% and 22.9%, respectively. The most common indoor allergens were orris root (38.5%) followed by tobacco smoke (31.3%), wool (22.9%), and cotton (24%). No allergic reaction to dog epithelials or synthetic fibers (orlon, nylon, or rayon) was observed.
Insects
The highest sensitization ratio among insects was for houseflies (22.9%). The positive rate for flea allergens was only 7.9%.
Microorganisms
For Staphyloccus and Malassezia allergens, 19.8% and 21.9%, respectively, of the dogs produced positive responses.
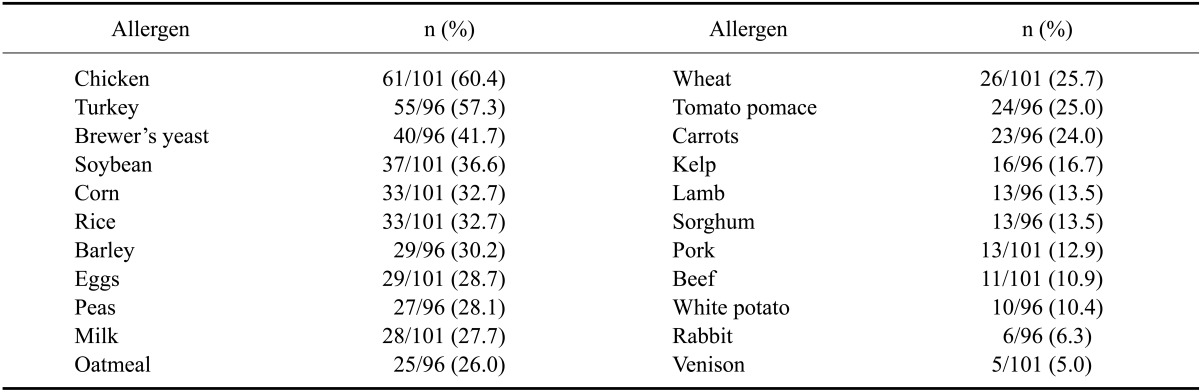
Food allergens
Among the food allergens, the most common was chicken (60.4%) followed by turkey (57.3%), brown rice (42.7%), brewer's yeast (41.7%), and soybeans (36.6%). The sensitization rates for rabbit, vension, duck, and tuna were lower compared to those of the other allergens (less than 10%; Table 2).
Table 2.
Sensitization rates for food allergens among AD dogs
Discussion
Identify causative allergens associated with AD has been a very important issue in veterinary dermatology because this condition is the most common cause of pruritus in dogs [25]. Many studies have been conducted to assess the value of allergen-specific IgE serology in cases of canine AD and canine food allergic dermatitis [4,5,12,23]. Interpretation of the test results should be performed cautiously and only used once a clinical diagnosis of AD has been made because positive test results are not specific for canine AD [11]. Traditionally, house dust mites and house dust have been considered the most important allergens in humans and dogs [7,19]. In canines, human dander, feathers, kapok, molds, weeds, grasses, and trees are also important allergens [25].
In the present study, 101 dogs with AD were tested with 92 allergens (70 environmental allergens and 22 food allergens). Among the environmental allergens, the sensitization rates for house dust and house dust mites were high; these are considered important allergens in veterinary and human medicine. The high sensitization rate (61.4%) for house dust mites was similar to ones previously reported in studies performed in the same geographic location [7,26,28]. However, the sensitization rate for house dust in the present study (55.2%) was higher than those observed in previous investigations. The sensitization rate for house dust was just 5.3% among 35 dogs in 2002 [28], and 17.5% among 22 dogs in 2005 [26].
In addition, reactions to airborne pollen (grasses, weeds, and trees) and molds were relatively common in this study although pollen is not usually considered an important allergen. Grasses produced negative results in three previous studies [7,26,28]. Sensitization to weeds was not observed and the rate for trees was just 5.3% among 22 dogs in 2005 [26]; few or no allergens in these groups caused positive reactions in 2002 [28]. The most recent study results obtained in 2010 [7] showed that Rhizopus had a higher positive rate (49.1%) compared to the one observed in the current study (15.8%), which was the lowest sensitization rate among molds. According to other previous reports, sensitivity of the allergen-specific IgE serology test to molds was lower than the IDST, and serology test results are less reliable for detecting molds hypersensitivities [5,14].
Flea and insect groups produced relatively weak positive reactions in this study. Flea populations tend to grow in warm temperatures and high humidity, and these insects are very common allergens of canines in the world [25]. However, flea allergies were rare in the present study because most of the evaluated dogs lived primarily indoors. The results of our study also suggested that the sensitized rate was low. On the other hand, mosquitos are very common in Korea, and it was recently observed that these insects have an increasingly close relationship with AD in humans and dogs [7]. However, the sensitization rate for mosquitos was only 9.4% in our investigation.
Differences among the causative allergens when comparing studies might be due to several reasons. Direct comparison between IDST and allergen?specific IgE serology test results are impossible because the two techniques detect IgE from different sources such as skin and serum [5,25]. Discrepancies between IDST and serology data may be explained by variations in testing methodologies, allergen extracts, and sensitivity [3,5,10,22]. In previous studies of causative allergens of AD dogs in Korea, an IDST was performed. Thus, complete correlations between results from those publications with ones from the current study cannot be identified.
As previously described [19], geographic and climate conditions are closely associated with environmental allergens. In particular, pollens and molds are affected by temperature and humidity [1]. Not surprisingly, these factors represent recently emerging allergens that cause allergenic disease in humans [17]. Positive responses to environmental allergens identified in the present study can be related to increased average temperatures and CO2 concentrations in Korea due to global warming and rapid urbanization. Previous studies conducted in our country were performed during different years (2002, 2005, and 2010), and the general sensitization rates of allergens might be increased overall 10 years. In addition, previous investigations included a relatively small number of dogs and were conducted in different regions of Korea [7,26,28].
Secondary infection associated with Malassezia or staphylococci is quite frequent, and recent studies have focused on the relationship of the associated antigens with allergies in dogs [15,16]. Antibodies to microorganisms in AD dogs had never been reported in Korea. In the present investigation, the sensitization rates for Staphyloccus and Malassezia were 19.8% and 21.9%, respectively. Results from previous studies revealed that AD dogs produce higher levels of IgE to these microorgainisms. [2,16]. These data indicate an immunopathogenic role of the microorganisms; however, the exact pathogenesis and clinical significance are uncertain. In humans, Staphylococcus aureus plays an important role in AD development and staphylococcal superantigens have been implicated in AD pathogenesis [9]. Thus, secondary infection by Malassezia or Staphyloccus should be monitored and promptly identified in AD dogs.
Animals with food allergic dermatitis were excluded from our using an elimination diet. However, AD dogs are believed to be sensitive to various allergens, and cross-reactivity of airborne and food allergens has been detected in humans [8]. Among food allergens, chicken, turkey, brown rice, brewer's yeast, and soybean are considered common. These allergens are frequently used ingredients for commercial dog food that most dogs in this study were mainly fed. The meaningful of food-specific IgE test is still controversial [13], but which also can be used for limited-antigen dietary trials [6]. Based on the results from the present study, chicken, turkey, brown rice, brewer's yeast, and soybeans can be included in avoidance diets. Rabbit, venison, duck, and tuna can also be used for dietary trials of canine AD.
The clinical history, characteristic signs, and several diagnostic criteria [18,21,27] were used for diagnosing canine AD in the current investigation. The sensitization rates for house dust and house dust mites were high, while allergies against airborne pollen and molds were relatively common in our study. The sensitization rates for Malassezia or staphylococci were approximately 20%. Reducing the allergen load and exposure as well as promptly identifying and treating secondary infections may be helpful for controlling the symptoms of AD dogs. There are several reports indicating that the SAT has high specificity and relatively low sensitivity [4,15]. Thus, some false positive reactions might have been obtained in our study. However, both the IDST and SAT are mostly used for allergen-specific immunotherapy to treat AD [11], and have been found to have equal efficacy for identifying allergens as a part of immunotherapy regimens for AD dogs [20,24].
In summary, we measured the sensitization rates for selected allergens in AD dogs using allergen-specific IgE serology. This SAT is easy to perform, less stressful to the patients, has a low cost compared to the IDST, and produces fewer false negative results. When accounting for the patient's history, clinical signs, and exposure to allergens and eliminating other causes of pruritus, allergen-specific IgE serology might be valuable for controlling canine AD.
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation Korea (NRF) funded by the Ministry of Education (no. 2013R1A1A2006152).
Footnotes
There is no conflict of interest.
References
- 1.Beggs PJ. Impacts of climate changes on aeroallergens: past and future. Clin Exp Allergy. 2004;34:1507–1513. doi: 10.1111/j.1365-2222.2004.02061.x. [DOI] [PubMed] [Google Scholar]
- 2.Bexley J, Nuttall TJ, Hammerberg B, Fitzgerald JR, Halliwell RE. Serum anti-Staphylococcus pseudintermedius IgE and IgG antibodies in dogs with atopic dermatitis and nonatopic dogs. Vet Dermatol. 2013;24:19–24.e6. doi: 10.1111/j.1365-3164.2012.01109.x. [DOI] [PubMed] [Google Scholar]
- 3.Codner EC, Lessard P. Comparision of intradermal allergy test and enzyme-linked immunosorbent assay in dogs with allergic skin disease. J Am Vet Med Assoc. 1993;202:739–743. [PubMed] [Google Scholar]
- 4.DeBoer DJ, Hillier A. The ACVD task force on canine atopic dermatitis (XVI): laboratory evaluation of dogs with atopic dermatitis with serum-based "allergy" tests. Vet Immunol Immunopathol. 2001;81:277–287. doi: 10.1016/s0165-2427(01)00304-x. [DOI] [PubMed] [Google Scholar]
- 5.Foster AP, Littlewood JD, Webb P, Wood JLN, Rogers K, Shaw SE. Comparison of intradermal and serum testing for allergen-specific IgE using a FcεRIα-based assay in atopic dogs in the UK. Vet Immunol Immunopathol. 2003;93:51–60. doi: 10.1016/s0165-2427(03)00052-7. [DOI] [PubMed] [Google Scholar]
- 6.Halliwell REW, Gordon C, Horvath C, Wagner R. IgE and IgG antibodies to food antigens in sera from normal dogs, atopic dogs and dogs with adverse food reactions. Vet Dermatol. 2004;15(Suppl 1):2–3. [Google Scholar]
- 7.Kim HJ, Kang MH, Park HM. Common allergens of atopic dermatitis in dogs: comparative finings based on intradermal tests. J Vet Sci. 2011;12:287–290. doi: 10.4142/jvs.2011.12.3.287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Koo SW, Kim YC, Kim SC, Kim DK. The study of prick test results and serum IgE levels in patients with atopic dermatitis. Korean J Dermatol. 1992;30:43–50. [Google Scholar]
- 9.Leung DY, Hauk P, Strickland I, Travers JB, Norris DA. The role of superantigens in human diseases: therapeutic implications for the treatment of skin diseases. Br J Dermatol. 1998;139(Suppl 53):17–29. doi: 10.1046/j.1365-2133.1998.1390s3017.x. [DOI] [PubMed] [Google Scholar]
- 10.Lian TM, Halliwell RE. Allergen-specific IgE and IgGd antibodies in atopic and normal dogs. Vet Immunol Immunopathol. 1998;66:203–223. doi: 10.1016/s0165-2427(98)00199-8. [DOI] [PubMed] [Google Scholar]
- 11.Marsella R. Canine atopic dermatitis: what's new? Compend Contin Educ Vet. 2010;32:E1–E4. [PubMed] [Google Scholar]
- 12.Martín Á, Sierra MP, González JL, Arévalo MÁ. Identification of allergens responsible for canine cutaneous adverse food reactions to lamb, beef and cow's milk. Vet Dermatol. 2004;15:349–356. doi: 10.1111/j.1365-3164.2004.00404.x. [DOI] [PubMed] [Google Scholar]
- 13.Mueller R, Tsohalis J. Evaluation of serum allergenspecific IgE for the diagnosis of food adverse reactions in the dog. Vet Dermatol. 1998;9:167–171. doi: 10.1046/j.1365-3164.1998.00107.x. [DOI] [PubMed] [Google Scholar]
- 14.Mueller RS, Burrows A, Tsohalis J. Comparison of intradermal testing and serum testing for allergen-specific IgE using monoclonal IgE antibodies in 84 atopic dogs. Aust Vet J. 1999;77:290–294. doi: 10.1111/j.1751-0813.1999.tb10263.x. [DOI] [PubMed] [Google Scholar]
- 15.Nuttall T, Uri M, Halliwell R. Canine atopic dermatitis -what have we learned? Vet Rec. 2013;172:201–207. doi: 10.1136/vr.f1134. [DOI] [PubMed] [Google Scholar]
- 16.Nuttall TJ, Halliwell RE. Serum antibodies to Malassezia yeasts in canine atopic dermatitis. Vet Dermatol. 2001;12:327–332. doi: 10.1046/j.0959-4493.2001.00261.x. [DOI] [PubMed] [Google Scholar]
- 17.Oh JW, Kang IJ, Kim SW, Kook MH, Kim BS, Shin KS, Hahn YS, Lee HB, Shon MH, Cheong JT, Lee HR, Kim KE. The correlation between increased sensitization rate to weeds in children and the annual increase in weed pollen in Korea. Pediatr Allergy Respir Dis. 2006;16:114–121. [Google Scholar]
- 18.Olivry T. New diagnostic criteria for canine atopic dermatitis. Vet Dermatol. 2010;21:123–126. doi: 10.1111/j.1365-3164.2009.00776.x. [DOI] [PubMed] [Google Scholar]
- 19.Park SH, Lim DH, Son BK, Kim JH, Song YE, Oh IB, Kim YH, Lee KH, Kim SY, Hong SC. Sensitization rates of airborne pollen and mold in children. Korean J Pediatr. 2012;55:322–329. doi: 10.3345/kjp.2012.55.9.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Plant JD, Neradelik MB, Polissar NL, Fadok VA, Scott BA. Agreement between allergen-specific IgE assays and ensuing immunotherapy recommendations from four commercial laboratories in the USA. Vet Dermatol. 2014;25:15–e6. doi: 10.1111/vde.12104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Prélaud P, Guagère E, Alhaidari Z, Faivre N, Héripret D, Gayerie A. Reevaluation of diagnostic criteria of canine atopic dermatitis. Rev Med Vet (Toulouse) 1998;149:1057–1064. [Google Scholar]
- 22.Roque JB, O'Leary CA, Kyaw-Tanner M, Latter M, Mason K, Shipstone M, Vogelnest L, Duffy D. High allergen-specific serum immunoglobulin E levels in nonatopic West Highland white terriers. Vet Dermatol. 2011;22:257–266. doi: 10.1111/j.1365-3164.2010.00939.x. [DOI] [PubMed] [Google Scholar]
- 23.Saevik BK, Thoresen SI, Kristensen F. A retrospective study of hyposensitization in canine atopy based on a polyclonal ELISA test. Vet Res Commun. 2002;26:613–624. doi: 10.1023/a:1020968703929. [DOI] [PubMed] [Google Scholar]
- 24.Schnabl B, Bettenay SV, Dow K, Mueller RS. Results of allergen-specific immunotherapy in 117 dogs with atopic dermatitis. Vet Rec. 2006;158:81–85. doi: 10.1136/vr.158.3.81. [DOI] [PubMed] [Google Scholar]
- 25.Scott DW, Miller WH, Jr, Griffin CE. Canine Atopic Disease. In: Scott DW, Miller WH Jr, Griffin CE, editors. Muller and Kirk's Small Animal Dermatology. 6th ed. Philadelphia: WB Saunders; 2001. pp. 574–601. [Google Scholar]
- 26.Song KH, Lee JY, Liu J, Lee SE, Park SJ, Kim DH. Prevalence of causative allergens on canine atopic dermatitis in Daejeon area. J Vet Clin. 2005;22:26–30. [Google Scholar]
- 27.Willemse T. Atopic skin disease: a review and a reconsideration of diagnostic criteria. J Small Anim Pract. 1986;27:771–778. [Google Scholar]
- 28.Youn HY, Kang HS, Bhang DH, Kim MK, Hwang CH, Han HR. Allergens causing atopic diseases in canine. J Vet Sci. 2002;3:335–341. [PubMed] [Google Scholar]


