Summary
During immune responses, B lymphocytes clonally expand and undergo secondary diversification of their immunoglobulin (Ig) genes in germinal centers (GCs)1–4. High affinity B cells are expanded through iterative interzonal cycles of division and hypermutation in the GC dark zone (DZ) followed by migration to the GC light zone (LZ) where they are selected based on affinity to return to the DZ5–10. Here we combine a transgenic strategy to measure cell division and a photoactivatable fluorescent reporter to examine whether the extent of clonal expansion and hypermutation are regulated during interzonal GC cycles. We find that both cell division and hypermutation are directly proportional to the amount of antigen captured and presented by GC B cells to T follicular helper (TFH) cells in the LZ. Our data explain how GC B cells with the highest affinity for antigen are selectively expanded and diversified.
Results
Clonal expansion is an essential feature of the immune response. B lymphocytes bearing antigen-specific Igs undergo this process in the GC, a specialized microanatomical compartment where B cells also diversify their Ig genes through somatic hypermutation (SHM)1–4. GC B cells expressing mutated surface Igs with the highest affinity are then positively selected by iterative cycles of cell division, SHM and selection5–10, endowing the host with high affinity humoral immunity4.
GC B cells divide and mutate in the DZ, and then migrate to the LZ where they capture antigen through surface Ig and present it as peptide bound to MHCII (pMHCII) to cognate TFH cells4,10–12. Migration between the two zones is mediated by the chemokine receptors CXCR4 and CXCR5, with 50% of DZ cells migrating to the LZ, and 10% returning to the DZ from the LZ within 6 hours5,10,13. Moreover, B cells in the two GC zones alternate between distinct genetic programs reflecting cell division in the DZ and selection in the LZ, but do so independently of local cues received in the two zones10,14. However, the precise mechanism by which the highest affinity cells are selected, and whether cell divisions and Ig mutations in the DZ are regulated, remains unknown14.
To determine whether the amount of antigen internalized by GC B cells governs the extent of clonal expansion, we titrated the amount of antigen delivered to GC B cells using antibodies that target DEC205, an endocytic receptor that carries antigen to intracellular MHCII-containing compartments10,15–18. GC responses were initiated by priming mice with ovalbumin (OVA) followed by boosting with OVA coupled to the hapten 4-hydroxy-3-nitrophenylacetyl (NP-OVA)9. Antigen-specific B cell responses were tracked by adoptive transfer of B1-8hi Ig heavy chain knock-in B cells, which are specific for NP when they express Ig lambda (Igλ) light chains19. To measure the relative expansion of B cells receiving graded amounts of antigen, GCs were induced in mice that received a mixture of B1-8hi DEC205+/+ and B1-8hi DEC205−/− B cells at a 5:95 ratio. Graded doses of antigen were delivered to DEC205+/+ GC B cells using chimeric DEC205 antibody fused to cognate antigen, OVA (αDEC-OVA, Fig. 1a)20. Whereas control injections with PBS had no effect, injection with 10 μg of αDEC-OVA resulted in selective expansion of the B1-8hi DEC205+/+ GC B cells (Fig. 1b, c and Extended Data Fig. 1). Decreasing the dose of antigen delivered, by mixing αDEC-OVA with a chimeric αDEC-205 antibody carrying the control irrelevant antigen Plasmodium Falciparum circumsporozoite protein (αDEC-CS), resulted in decreased expansion of B1-8hi DEC205+/+ GC B cells that was proportional to the dose of αDEC-OVA (Fig. 1b, c). Consistent with the idea that pMHCII-mediated selection occurs in the LZ and cell division in the DZ4,10, selective dose-dependent expansion of B1-8hi DEC205+/+ GC B cells was already evident at 48 hours in the DZ but only later in the LZ (Fig. 1d, e). In contrast, the B1-8hi DEC205−/− GC B cell population contracted in proportion to the amount of antigen delivered to the B1-8hi DEC205+/+ GC B cell population (Fig. 1b and Extended Data Fig. 1c). Thus, increasing the amount of cognate antigen presented by a subset of GC B cells to TFH cells leads to their proportional and selective expansion at the expense of GC B cells that present less antigen.
Figure 1. The amount of antigen captured and presented by GC B cells regulates their expansion.
a, Protocol for b–e. 1.5–5 × 106 B1-8hi DEC205+/+ and B1-8hi DEC205−/− B cells (≈ 1.5–5 × 105 Igλ+, NP-specific B cells) at a 5:95 ratio were transferred into OVA-primed WT mice, which were boosted with NP-OVA. After 6 days, mice were injected with PBS or αDEC-OVA mixed with αDEC-CS at ratios of 1:0, 1:3, 1:9, or 1:39. Lymph nodes were analyzed 2, 3, and 4 days after injection. b, Proportion of B1-8hi DEC205+/+ and B1-8hi DEC205−/− GC B cells 48 hours after treatment. c–e, Mean fraction of DEC205+/+ B cells among B1-8hi GC (c), DZ (CD86−−CXCR4+, d), and LZ (CD86+CXCR4−, e) cells. Error bars = SEM. Data represent 2–3 independent experiments at each time point with a total of 4–6 mice per condition for all time points.
To examine the mechanism by which increased T cell help leads to selective GC B cell expansion, we sought to measure cell division in the GC. Traditional dye based methods to monitor cell division are unsuitable in this context because B cells divide extensively and lose most of the dye before entering the GC. To circumvent this problem, we combined transgenes encoding the tetracycline transactivator (tTA) protein expressed under the Vav promoter and a histone H2B-mCherry fusion protein driven by a Doxycycline (DOX)-regulated promoter (tTA-H2B-mCh, Extended Data Fig. 2a)21,22. Under steady state conditions, tTA is expressed in hematopoietic cells and induces high levels of H2B-mCh expression (Extended Data Fig. 2b). Administration of DOX represses further H2B-mCh synthesis, and as a result the H2B-mCh dilutes in proportion to cell division (Extended Data Fig. 2c)23.
To determine whether tTA-H2B-mCh can be used to track antigen-specific GC B cell division in vivo, we repeated the prime-boost protocol described above using B1-8hi tTA-H2B-mCh B cells. As expected, non-proliferative follicular B cells did not dilute H2B-mCh after DOX treatment (Fig. 2a). In contrast, after 36 hours, a spectrum of discrete peaks of H2B-mCh expression corresponding to cell divisions became evident among GC B cells, and by 84 hours, GC B cells had completely diluted H2B-mCh (Fig. 2a). Thus, tTA-H2B-mCh can be used to monitor cell division in the GC.
Figure 2. T cell help regulates the number of GC B cell divisions.
a, H2B-mCh fluorescence among B1-8hi tTA-H2B-mCh B cells within the follicular (upper) or GC (lower) compartments of untreated mice (black) or after 36 (red) or 84 (blue) hours on DOX. Solid gray represents non-fluorescent cells. b, H2B-mCh fluorescence among B1-8hi tTA-H2B-mCh GC B cells in mice that received a 5:95 mixture of B1-8hi tTA-H2B-mCh and B1-8hi DEC205−/− B cells and were treated with PBS, αDEC-CS, or αDEC-OVA for 72 hours and administered DOX for 60 hours before analysis. c, Mean fraction of B1-8hi tTA-H2B-mCh GC B cells treated as in b. Error bars = SEM. d, Percent DZ cells among B1-8hi tTA-H2B-mCh GC B cells after treatment with PBS or αDEC-CS (control) or αDEC-OVA for 48 hours and DOX for 36 hours. Each symbol represents one mouse and lines represent mean values. Data represent 3–4 independent experiments for all time points with 2–3 mice for each condition per experiment. * p < 0.005; ** p < 0.001; *** p <0.0001, two-tailed Mann-Whitney test.
To determine whether the amount of antigen captured by GC B cells influences their degree of proliferation, we delivered additional antigen to GC B cells using αDEC-OVA. After 60 hours on DOX, B1-8hi DEC205+/+ tTA-H2B-mCh GC B cells from mice treated with control αDEC-CS or PBS were nearly evenly distributed among mChMed, Lo, and − groups, representing cells that underwent progressively more cell division. In contrast, after 60 hours on DOX, ~50% of GC B cells targeted with αDEC-OVA became mCh− and ~40% were mChLo (Fig. 2b, c). Therefore, increased antigen capture and presentation leads to increased rates of cell division by GC B cells.
Concomitant with the increased rate of cell division, there was a change in the zonal distribution of GC B cells. Whereas control cells equilibrated between DZ and LZ at a 2:1 ratio on average, αDEC-OVA targeted GC B cells were found nearly exclusively in the DZ (~90%, Fig 2d).
To examine cell cycle distribution during selection, we labeled GC B cells with an intravenous pulse of EdU followed 1 hour later by BrdU, allowing separation of GC B cells into the earliest (EdU−BrdU+), mid/late (EdU+BrdU+) and post- (EdU+BrdU−) S phase periods of the cell cycle (Fig. 3a and Extended Data Fig. 3). Control GC B cells in early and mid/late-S phases were found in both zones; post-S phase cells were primarily in the DZ (Fig. 3b). This supports the idea that GC B cells can initiate S phase in the LZ and then migrate to the DZ to divide10. Moreover, the fact that early S phase cells are distributed at a 2:1 DZ:LZ ratio suggests that, once in the DZ, GC B cells can initiate two cell divisions on average. In contrast, inducing selection by injecting αDEC-OVA shifted early S phase cells among selected GC B cells to the DZ (~86%), whereas slightly fewer early S phase cells were in the DZ (59%) among non-selected cells (Fig. 3b). We conclude that increasing the amount of antigen captured and presented to TFH cells increases the proportion of cells initiating S phase in the DZ. These findings suggest that GC B cells presenting the highest levels of antigen initiate additional cell divisions in the DZ before returning to the LZ.
Figure 3. Selective expansion involves increased S phase initiation in the DZ and longer DZ residence time.
a, EdU and BrdU incorporation among GC B cells. OVA-primed mice received 5 × 106 B1-8hi DEC205+/+ and B1-8hi DEC205−/− B cells at a 15:85 or 50:50 ratio. On day 6 after boosting with NP-OVA, mice receiving the 15:85 or the 50:50 B cell transfer were injected with αDEC-OVA or PBS, respectively. Two days later, mice received EdU and 1 hour later BrdU and were analyzed after 30 minutes (detailed in Extended Data Fig. 3a). b, Percent DZ cells among early (E), mid/late (M/L), and post- (P) S phase cells. Black squares denote αDEC-OVA treatment; white squares denote PBS control. Data represent two independent experiments with 6 or 7 mice per condition in total. Squares indicate mean values; error bars = SEM. c, d, OVA-primed mice received 5 × 106 B1-8hi PAGFP+ and B1-8hi DEC205−/− B cells at a 15:85 ratio or 5 × 106 B1-8hi PAGFP+ B cells alone and were boosted with NP-OVA. For mice receiving mixed B cell transfers, αDEC-OVA was injected on day 6. On day 8, DZ cells were photoactivated (c) and GC B cells were analyzed by flow cytometry (detailed in Extended Data Fig. 4a) (d). e, Percent LZ cells among photoactivated B1-8hi PAGFP+ GC B cells at 0 or 6 hours after DZ photoactivation. Each symbol represents one mouse. Data represent multiple independent experiments. ** p = 0.0022, two-tailed Mann-Whitney test.
We used photoactivation to test whether selected B cells presenting higher levels of antigen reside longer in the DZ10. Primed mice received a mixture of B1-8hi DEC205+/+ B cells expressing a photoactivatable green fluorescent protein (B1-8hi PAGFP+) and B1-8hi DEC205−/− B cells. On day 6 of the GC response, we induced selection by injecting αDEC-OVA. DZ cells were photoactivated and lymph nodes were processed for flow cytometry either 0 or 6 hours after photoactivation (Fig. 3c and Extended Data Fig. 4). Under control conditions, 57% of photoactivated GC B cells migrated to the LZ after 6 hours10. In contrast, B1-8hi PAGFP+ cells undergoing selective expansion migrated from the DZ to the LZ at half this rate (Fig. 3d, e). Thus, GC B cells undergoing selective expansion as a result of increased antigen presentation reside longer in the DZ, allowing them to undergo additional cell divisions before returning to the LZ. We conclude that TFH cells regulate the number of cell cycles a GC B cell initiates during each passage through the DZ, and that they do so in direct proportion to the amount of antigen captured and presented.
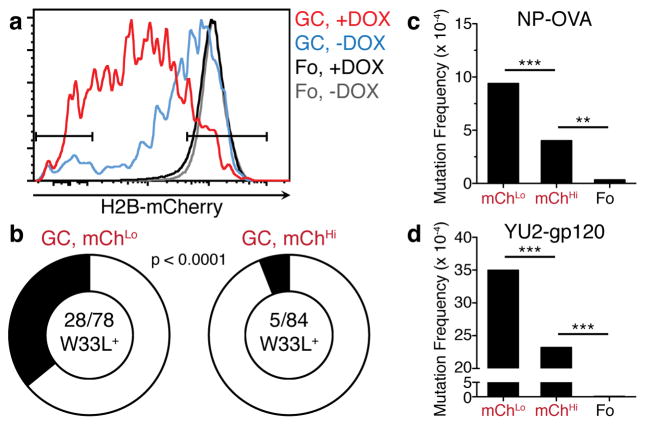
To determine whether variable numbers of cell divisions in the DZ are associated with selection during a polyclonal immune response, we immunized non-Ig transgenic, tTA-H2B-mCh mice with NP-OVA. Mice were given DOX at day 12.5 after immunization, and 36 hours later mChHi and mChLo GC B cells, representing lower and higher rates of proliferation respectively, were isolated by cell sorting (Fig. 4a, b). VH186.2 family genes were then analyzed for the characteristic high affinity anti-NP W33L mutation24. We found that 35.9%, of the highly divided GC B cells were W33L+; in contrast, only 6% of the cells that underwent the fewest divisions were W33L+ (p < 0.0001, Fig. 4b). Thus, selection of high affinity GC B cells in a polyclonal response is associated with increased rates of proliferation. Moreover, since each division can introduce mutations that increase or decrease affinity, even high affinity clones will rapidly partition into subclones that undergo different rates of proliferation, death or differentiation. This is likely reflected in the observation that mChLo cells are not all W33L+, and that W33L+ cells are not exclusively in the mChLo fraction.
Figure 4. Increased cell division in polyclonal GCs is associated with higher Ig affinity and somatic hypermutation.
a–d, tTA-H2B-mCh mice were immunized with NP-OVA (a–c) or YU2-gp120 (d) and administered DOX for 36 hours before purification. a, Representative histogram displaying mChHi and mChLo gates. b, Frequency of W33L mutation among VH186.2 clones in mChHi and mChLo GC B cells on day 14 after immunization. The number of W33L+ clones among VH186.2 sequences is shown in the center of the pie chart and was compared using Fisher’s Exact test. Data are pooled from 2 independent experiments with 2 mice each. c–d, SHM in the JH4 intron among mChHi and mChLo GC B cells on day 7 after NP-OVA (c) and day 9 after YU2-gp120 immunization (d). Data in (c) and (d) are pooled from 2 independent experiments with 2–3 mice each. Over 160 clones in (c) and 140 clones in (d) were analyzed for mChHi and mChLo GC B cells and 51 clones in (c) and 94 clones in (d) were analyzed for follicular B cells. Fo, follicular B cells. ** p = 0.0033; *** p < 0.0001, χ2 test with Yates correction.
Each round of cell division in the GC is predicted to produce 1 somatic mutation per 103 base pairs in Ig genes8. The finding that TFH cells control the number of B cell divisions per GC cycle suggests that they may also regulate hypermutation. To determine whether there is a correlation between selection, proliferation and SHM in the GC, we immunized tTA-H2B-mCh mice with NP-OVA, purified mChHi and mChLo GC B cells by cell sorting and analyzed the intron downstream of JH4 for somatic mutations, since this region is targeted for SHM but is not subject to selection25. GC B cells that had diluted H2B-mCh by undergoing a greater number of divisions were significantly more mutated than less divided cells (p < 0.0001, Fig. 4c).
High levels of mutation are required to produce broadly neutralizing antibodies to HIV-1 and are believed to be an impediment to vaccine development26. To determine whether mutations accumulate differentially among GC B cells responding to HIV-1 gp120, we immunized tTA-H2B-mCh mice with HIV-1YU2 gp120 and compared GC B cells that had undergone different levels of division. Similar to NP-OVA, the more divided mChLo B cells in the anti-gp120 GCs were significantly more mutated than either mChHi GC or follicular B cells (Fig. 4d). We conclude that B cells undergo a variable number of divisions in the DZ before returning to the LZ, and that increased cell division is associated with higher Ig affinity and increased SHM.
The GC is a site of intense Ig diversification and selection, from which high affinity B cells emerge that seed the memory and plasma cell compartments3,4,27. Affinity-based Ig selection is accomplished iteratively. B cell clones expanding and mutating in the DZ travel to the LZ to compete for help from TFH cells, with only a fraction of LZ cells selected to return to the DZ to continue cycling10. Our experiments indicate that the number of GC B cell divisions per DZ cycle is variable, ranging from 1 to 6, and is regulated by the amount of antigen captured in the LZ. By capturing more antigen and dividing a greater number of times in the DZ during each interzonal cycle, high affinity GC B cells outcompete lower affinity cells that capture less antigen and divide fewer times. The magnitude of T cell help provided in the LZ therefore regulates the behavior of a DZ cell – inducing selected cells to re-enter the cell cycle a variable number of times before migrating back to the LZ. This finding is consistent with and may explain the observation that the switch from DZ to LZ phenotype is independent of cues received in the DZ14. Moreover, the least proliferative GC B cells have the fewest affinity-enhancing mutations and are disproportionately found in the LZ (Fig. 2d and Fig. 4b). The precise contributions of death and differentiation to the eventual disappearance of these cells from the GC remain to be determined.
An important feature of differential cell division in the DZ is that each round of division is associated with increased accumulation of SHM. Consistent with this idea, highly proliferative GC B cells have both higher Ig affinity and a greater number of somatic mutations. Our experiments reveal a feed-forward loop in the GC, whereby SHM is greatest among GC B cells whose antibodies provide the highest affinity template for further diversification. This finding may be particularly relevant for designing immunization strategies that elicit broadly neutralizing antibodies to pathogens, like HIV-1, that require exceptionally high levels of mutation.
Methods
Mice
C57/BL6, B6.SJL, and Col1A1-tetO-H2B-mCherry mice were purchased from Jackson Laboratories. Vav-tTA transgenic mice that had been backcrossed to C57/BL6 were obtained from S.W. Lowe and crossed to Col1A1-tetO-H2B-mCherry mice to produce tTA-H2B-mCh mice21,22. B1-8hi, DEC205−/−, and PAGFP transgenic mice were described previously10,19,28. All experiments were performed with authorization from the Institutional Review Board and the IACUC at The Rockefeller University.
B cell transfer and culture
Resting B cells were purified by forcing spleen tissue through a 40 μm mesh into complete RPMI media (Gibco) with 6% serum. Single cell suspensions were purified by magnetic cells sorting (MACS) using CD43 beads, according to the manufacturer’s instructions (Miltenyi Biotec). Indicated cell numbers were transferred intravenously into recipient mice. To assess proliferation in vitro, resting B cells purified from tTA-H2B-mCh mice were labeled at 37 °C in 5 μM carboxyfluorescein succinimidyl ester. Labeled B cells were then stimulated with LPS and IL-4 for 72 hours as previously described30. DOX was added to the cultures at 500 ng/mL.
Immunizations and treatments
C57BL/6 or B6.SJL male recipient mice (6–8 weeks of age) were primed by intraperitoneal immunization with 100 μl containing 50 μg of OVA (Grade V, Sigma) precipitated in alum at a 2:1 ratio in PBS. Two to six weeks after priming, mice received adoptive cell transfers of indicated B cells and were boosted the following day with 25 μg of NP14-OVA (Biosearch Technologies) in hind footpads. Popliteal lymph nodes were harvested for flow cytometric analysis. To generate polyclonal GCs, mice were immunized with 50 μg intraperitoneally and 12.5 μg in the hind footpads of NP14-OVA in alum. HIV-1YU2 gp120-specific GCs were induced by immunizing mice intraperitoneally and in hind footpads with 12.5 μg and 6.25 μg of gp120 protein, respectively, in alum. αDEC-OVA and αDEC-CS chimeric antibodies were produced by transient transfection in 293T cells, as described20. 10 μg of chimeric antibody in PBS was injected into footpads of mice at indicated time points, except in Fig. 3b in which 5 μg of chimeric antibody was injected into footpads. Mice were administered DOX by intraperitoneal injection of 1.6 mg DOX (Sigma) in PBS and hind footpad injection of 0.2 mg DOX in PBS. Mice were maintained on DOX by adding DOX (2 mg/ml) and sucrose (10 mg/ml) to the drinking water for the indicated periods of time.
Flow cytometry
Lymph nodes were harvested by forcing tissue through 40 μm mesh into complete RPMI media (Gibco) with 6% serum. Single cell suspensions were treated at 4 °C for 10 min with 1 μg/ml anti-CD16/32 (2.4G2, Bio-X-Cell) and then stained for 25 min at 4 °C. B220, CD38, CD86, CD45.1 and CD45.2 antibodies were from eBioscience. FAS, CXCR4, CD45.1, CD45.2, Igλ1-3, GL7, streptavidin-PE and streptavidin-APC were from BD Biosciences. Streptavidin-Alexa Fluor 488 was from Invitrogen. For cell cycle and S phase analysis, mice were injected intravenously with 2 mg BrdU (Sigma-Aldrich) and 1 mg EdU (Life Technologies) in PBS. Cells were then stained for surface antigens as described above and processed using an anti-BrdU-FITC kit (BD Biosciences) and Click-iT EdU-Pacific Blue kit (Life Technologies) according to manufacturers’ protocols. All samples were analyzed on a BD Fortessa. GC B cells were gated as live/single, B220+, CD38− and FAS+. DZ and LZ GC B cells were further gated as CXCR4+CD86− and CXCR4−CD86+, respectively. CD45.1 and CD45.2 allotypic markers were used to trace adoptively transferred B cells of genotypes B1-8hi DEC205+/+ (CD45.1+), B1-8hi DEC205−/− (CD45.1+CD45.2+), and B1-8hi DEC205+/+ tTA-H2B-mCh (CD45.2+) within either C57/BL6 (CD45.2+) or B6.SJL (CD45.1+) recipient mice.
Photoactivation
Intravital imaging and photoactivation were performed as described previously10,18,29. Anasthesia was induced with 100 mg ketamine, 15 mg xylazine, and 2.5 mg acepromazine per kg of body weight and maintained with 1.25% isofluorane in 100% oxygen. A double-edged razor blade was used to shave hind legs. Mice were restrained on a 37°C stage warmer (BioTherm Micro S37; Biogenics) and an incision was made behind the knee to allow exposure of the popliteal lymph node. Once exposed, the lymph node was restrained with a metal strap and visualized with a microscope objective and a 40°C objective heater. To label LZ-resident follicular dendritic cells, 1 μg of the red fluorescent protein tdTomato conjugated to NP was injected in hind footpads one day before imaging10,18. To photoactivate DZ B cells, B1-8hi PAGFP+ GC B cells were photoactivated external to NP-tdTomato-labeled FDCs in the LZ by scanning with a femtosecond-pulsed multiphoton laser tuned to 820 nm wavelength and imaged at 940 nm wavelength, as previously described10,18. Cell motility was monitored immediately following photoactivation to ensure cell viability. Incisions were sutured and mice were allowed to recover for 6 hours before flow cytometric analysis. To determine the accuracy of this strategy, B1-8hi PAGFP+ GC B cells were photoactivated in the DZ of explanted lymph nodes and analyzed immediately for surface phenotype. Imaging experiments were performed with an Olympus BX61 upright microscope (Olympus 25X 1.05 NA Plan objective), fitted with a Coherent Chameleon Vision II laser (Rockefeller University Bio-Imaging Resource Center).
Ig sequence analysis
B cells were purified from spleen and lymph node tissues of immunized mice using CD43 magnetic beads (MACS). Single cell suspensions were stained as described above and sorted using a FACS Aria II (Becton Dickinson). GC B cells were gated as live/single, B220+CD38−FAS+ (Fig. 4c) or B220+CD38−FAS+GL7+ (Fig. 4d) and mChHi or mChLo. Genomic DNA was extracted from sorted cell populations and PCR was performed from DNA corresponding to 1,000–5,000 cells using Phusion HF (New England Biolabs). PCR products were gel extracted and cloned into Zero Blunt TOPO vectors (Invitrogen). For VH186.2 sequence analysis, GC B cells were further gated as Igλ+ and clones were amplified as described previously10,15. Ig sequences were analyzed using the IMGT/V-QUEST system to identify W33L mutations. JH4 intronic sequences were amplified using 5′-TCCTAGGAACCAACTTAAGAGT-3′ and 5′-TGGAGTTTTCTGAGCATTGCAG-3′ primers and 35 cycles with an annealing temperature of 57°C and an extension time at 72°C for 1 minute. High quality traces were analyzed using MacVector 12.7 for base pair mismatches and deletions as compared to the germline sequence. Both mismatches and deletions were counted as mutations. To calculate mutation frequency, the total number of mutations from all clones was summed and divided by the total number of base pairs analyzed for each group. Mutation frequencies among different groups were compared and analyzed for statistical significance by the χ2 test with Yates correction using Prism software v. 5.0 (Graphpad).
Extended Data
Extended Data Figure 1. Flow cytometric analysis of germinal center B cells, related to Figure 1.
a, Representative flow cytometry plots display gating strategy used to analyze experiments in Fig. 1. Live B220+ singlets were gated on GC cells (CD38-FAS+) and divided into LZ (CD86+CXCR4−) and DZ (CD86−CXCR4+) cells. B1-8hi DEC205+/+ cells within these compartments were identified as CD45.1+ and B1-8hi DEC205−/− cells were identified as CD45.1+CD45.2+. b, c, Total number of B1-8hi DEC205+/+ (b) and B1-8hi DEC205−/− (c) GC B cells per 106 lymph node cells from the experiments reported in Fig. 1. Bars represent mean values; error bars = SEM.
Extended Data Figure 2. tTA-H2B-mCh system, related to Figure 2.
a, Diagrammatic representation of the Vav-tTA and Tet-Op-H2B-mCh transgenes that were combined (tTA-H2B-mCh) to label B cells with H2B-mCh in order to inducibly measure cell division in the GC with DOX. b, Histogram displaying H2B-mCh expression among B220+ lymphocytes from the peripheral blood of WT (gray), Vav-tTA+ (red), Tet-Op-H2B-mCh+ (blue), and tTA-H2B-mCh mice (black). c, Purified B cells from tTA-H2B-mCh mice were labeled with CFSE and stimulated with LPS and IL-4. Left, H2B-mCh levels after 0 or 72 hours in culture (gray and black, respectively). Middle, H2B-mCh gates for B cells activated for 72 hours are color-coded. Right, histogram displaying the CFSE levels for the color-coded H2B-mCh gates. Data are representative of two independent experiments.
Extended Data Figure 3. Edu/BrdU labeling strategy to analyze the progression of S phase, related to Figure 3a, b.
a, Mice with ongoing GCs were administered intravenous EdU followed by intravenous BrdU 1 hour later. A half-hour after BrdU administration, mice were analyzed by flow cytometry. Cells in early S phase at the time of analysis incorporate only the second nucleotide analog and can therefore be identified as EdU−BrdU+. Cells in mid/late S phase replicate DNA during both the EdU and BrdU injections and are therefore EdU+BrdU+. Cells that completed S phase in the hour between EdU and BrdU administration are post-S phase cells at the time of analysis. These cells incorporate the first label, but not the second, making them EdU+BrdU−. b, Gating strategy used in Fig. 3a, b. B1-8hi DEC205+/+ and B1-8hi DEC205−/− GC B cells were identified among CD19+CD38−FAS+ cells using CD45 allotypic markers and were further subdivided based on EdU and BrdU incorporation and DZ/LZ surface phenotype. c, Flow cytometry plots displaying EdU and BrdU incorporation in GC and follicular B cells of mice receiving EdU and/or BrdU. d, Percent of B1-8hi DEC205+/+ (black) and B1-8hi DEC205−/− (gray) GC B cells in early (EdU−BrdU+, left), mid/late (EdU+BrdU+, middle), and post- (EdU+BrdU−, right) S phase periods in control (PBS) or αDEC-OVA treated mice. Data represent values from the same experiments reported in Fig. 3a, b. Error bars = SEM; ** p = 0.0022, two-tailed Mann-Whitney test.
Extended Data Figure 4. DZ photoactivation protocol and flow cytomeric analysis, related to Figure 3c–e.
a, Diagrammatic representation of the protocol used in Figure 3c–e. b, Flow cytometric gating strategy used to analyze GC B cells photoactivated in the DZ. Live singlets were gated as B220+PAGFP+CD38−FAS+Active PAGFP+.
Acknowledgments
S.W. Lowe and D.R. Fooksman for mice; D. Bosque and T. Eisenreich for help with mouse colony management; A. Abadir for protein production; K. Yao for technical help; K. Velinzon for help with cell sorting; D. Mucida, and all members of the Nussenzweig lab for discussion. Support for the Rockefeller University multiphoton microscope was granted by the Empire State Stem Cell Fund through New York State Department of Health contract C023046. Supported by NIH Medical Scientist Training Program grant T32GM07739 to the Weill Cornell/Rockefeller/Sloan-Kettering Tri-Institutional MD-PhD Program (A.D.G.); NIH grants AI037526-19 and AI072529-06 (M.C.N.); and the NIH Center for HIV/AIDS Vaccine Immunology and Immunogen Discovery (CHAVI-ID) 1UM1 AI100663-01 (M.C.N). Z.S. is a Human Frontiers of Science Fellow. M.C.N. is an HHMI investigator.
Footnotes
Author contributions
A.D.G. planned and performed experiments and wrote the manuscript. A.D.G. and Z.S. planned and performed photoactivation experiments. M.C.N. planned experiments and wrote the manuscript.
The authors declare no competing financial interest.
References
- 1.Berek C, Berger A, Apel M. Maturation of the immune response in germinal centers. Cell. 1991;67:1121–1129. doi: 10.1016/0092-8674(91)90289-b. [DOI] [PubMed] [Google Scholar]
- 2.Jacob J, Kelsoe G, Rajewsky K, Weiss U. Intraclonal generation of antibody mutants in germinal centres. Nature. 1991;354:389–392. doi: 10.1038/354389a0. [DOI] [PubMed] [Google Scholar]
- 3.Rajewsky K. Clonal selection and learning in the antibody system. Nature. 1996;381:751–758. doi: 10.1038/381751a0. [DOI] [PubMed] [Google Scholar]
- 4.Victora GD, Nussenzweig MC. Germinal centers. Annual review of immunology. 2012;30:429–457. doi: 10.1146/annurev-immunol-020711-075032. [DOI] [PubMed] [Google Scholar]
- 5.Allen CD, Okada T, Tang HL, Cyster JG. Imaging of germinal center selection events during affinity maturation. Science. 2007;315:528–531. doi: 10.1126/science.1136736. [DOI] [PubMed] [Google Scholar]
- 6.Hauser AE, et al. Definition of germinal-center B cell migration in vivo reveals predominant intrazonal circulation patterns. Immunity. 2007;26:655–667. doi: 10.1016/j.immuni.2007.04.008. [DOI] [PubMed] [Google Scholar]
- 7.Kocks C, Rajewsky K. Stepwise intraclonal maturation of antibody affinity through somatic hypermutation. Proceedings of the National Academy of Sciences of the United States of America. 1988;85:8206–8210. doi: 10.1073/pnas.85.21.8206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McKean D, et al. Generation of antibody diversity in the immune response of BALB/c mice to influenza virus hemagglutinin. Proceedings of the National Academy of Sciences of the United States of America. 1984;81:3180–3184. doi: 10.1073/pnas.81.10.3180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schwickert TA, et al. In vivo imaging of germinal centres reveals a dynamic open structure. Nature. 2007;446:83–87. doi: 10.1038/nature05573. [DOI] [PubMed] [Google Scholar]
- 10.Victora GD, et al. Germinal center dynamics revealed by multiphoton microscopy with a photoactivatable fluorescent reporter. Cell. 2010;143:592–605. doi: 10.1016/j.cell.2010.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Allen CD, Okada T, Cyster JG. Germinal-center organization and cellular dynamics. Immunity. 2007;27:190–202. doi: 10.1016/j.immuni.2007.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Oprea M, Perelson AS. Somatic mutation leads to efficient affinity maturation when centrocytes recycle back to centroblasts. J Immunol. 1997;158:5155–5162. [PubMed] [Google Scholar]
- 13.Allen CD, et al. Germinal center dark and light zone organization is mediated by CXCR4 and CXCR5. Nature immunology. 2004;5:943–952. doi: 10.1038/ni1100. [DOI] [PubMed] [Google Scholar]
- 14.Bannard O, et al. Germinal center centroblasts transition to a centrocyte phenotype according to a timed program and depend on the dark zone for effective selection. Immunity. 2013;39:912–924. doi: 10.1016/j.immuni.2013.08.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dominguez-Sola D, et al. The proto-oncogene MYC is required for selection in the germinal center and cyclic reentry. Nature immunology. 2012;13:1083–1091. doi: 10.1038/ni.2428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jiang W, et al. The receptor DEC-205 expressed by dendritic cells and thymic epithelial cells is involved in antigen processing. Nature. 1995;375:151–155. doi: 10.1038/375151a0. [DOI] [PubMed] [Google Scholar]
- 17.Kamphorst AO, Guermonprez P, Dudziak D, Nussenzweig MC. Route of antigen uptake differentially impacts presentation by dendritic cells and activated monocytes. J Immunol. 2010;185:3426–3435. doi: 10.4049/jimmunol.1001205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shulman Z, et al. T follicular helper cell dynamics in germinal centers. Science. 2013;341:673–677. doi: 10.1126/science.1241680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shih TA, Roederer M, Nussenzweig MC. Role of antigen receptor affinity in T cell-independent antibody responses in vivo. Nature immunology. 2002;3:399–406. doi: 10.1038/ni776. [DOI] [PubMed] [Google Scholar]
- 20.Boscardin SB, et al. Antigen targeting to dendritic cells elicits long-lived T cell help for antibody responses. The Journal of experimental medicine. 2006;203:599–606. doi: 10.1084/jem.20051639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Egli D, Rosains J, Birkhoff G, Eggan K. Developmental reprogramming after chromosome transfer into mitotic mouse zygotes. Nature. 2007;447:679–685. doi: 10.1038/nature05879. [DOI] [PubMed] [Google Scholar]
- 22.Wiesner SM, Jones JM, Hasz DE, Largaespada DA. Repressible transgenic model of NRAS oncogene-driven mast cell disease in the mouse. Blood. 2005;106:1054–1062. doi: 10.1182/blood-2004-08-3306. [DOI] [PubMed] [Google Scholar]
- 23.Tumbar T, et al. Defining the epithelial stem cell niche in skin. Science. 2004;303:359–363. doi: 10.1126/science.1092436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Allen D, Simon T, Sablitzky F, Rajewsky K, Cumano A. Antibody engineering for the analysis of affinity maturation of an anti-hapten response. The EMBO journal. 1988;7:1995–2001. doi: 10.1002/j.1460-2075.1988.tb03038.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jolly CJ, Klix N, Neuberger MS. Rapid methods for the analysis of immunoglobulin gene hypermutation: application to transgenic and gene targeted mice. Nucleic acids research. 1997;25:1913–1919. doi: 10.1093/nar/25.10.1913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.West AP, Jr, et al. Structural Insights on the Role of Antibodies in HIV-1 Vaccine and Therapy. Cell. 2014;156:633–648. doi: 10.1016/j.cell.2014.01.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tarlinton D, Good-Jacobson K. Diversity among memory B cells: origin, consequences, and utility. Science. 2013;341:1205–1211. doi: 10.1126/science.1241146. [DOI] [PubMed] [Google Scholar]
- 28.Guo M, et al. A monoclonal antibody to the DEC-205 endocytosis receptor on human dendritic cells. Human immunology. 2000;61:729–738. doi: 10.1016/s0198-8859(00)00144-0. [DOI] [PubMed] [Google Scholar]
- 29.Schwickert TA, et al. A dynamic T cell-limited checkpoint regulates affinity-dependent B cell entry into the germinal center. The Journal of experimental medicine. 2011;208:1243–1252. doi: 10.1084/jem.20102477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Robbiani DF, et al. AID is required for the chromosomal breaks in c-myc that lead to c-myc/IgH translocations. Cell. 2008;135:1028–1038. doi: 10.1016/j.cell.2008.09.062. [DOI] [PMC free article] [PubMed] [Google Scholar]