Abstract
Objective
Physical activity, neighborhood walkability, and body mass index (BMI, kg/m2) associations were tested using quasi-experimental twin methods. We hypothesized that physical activity and walkability were independently associated with BMI within twin pairs, controlling for genetic and environmental background shared between them.
Methods
Data were from 6,376 (64% female; 58% identical) same-sex pairs, University of Washington Twin Registry, 2008–2013. Neighborhood walking, moderate-to-vigorous physical activity (MVPA), and BMI were self-reported. Residential address was used to calculate walkability. Phenotypic (non-genetically informed) and biometric (genetically informed) regression was employed, controlling for age, sex, and race.
Results
Walking and MVPA were associated with BMI in phenotypic analyses; associations were attenuated but significant in biometric analyses (Ps < 0.05). Walkability was not associated with BMI, however, was associated with walking (but not MVPA) in both phenotypic and biometric analyses (Ps < 0.05), with no attenuation accounting for shared genetic and environmental background.
Conclusions
The association between activity and BMI is largely due to shared genetic and environmental factors, but a significant causal relationship remains accounting for shared background. Although walkability is not associated with BMI, it is associated with neighborhood walking (but not MVPA) accounting for shared background, suggesting a causal relationship between them.
Keywords: Body Mass Index, Environment and Public Health, Heredity, Motor Activity, Twins
Introduction
Obesity is a major public health problem in the U.S.; prevalence rates have more than doubled over the past decades (Flegal et al., 2010; Flegal et al., 2002; Hedley et al., 2004; Ogden et al., 2006; Ogden et al., 2014). Obesity is associated with several adverse and costly health conditions (American Diabetes Association, 2008; Anderson et al., 2005; Clinical Guidelines, 1998; Flegal et al., 2005; Pronk et al., 1999; Wang et al., 2011). Effects of lifestyle behaviors such as diet and physical activity on health outcomes such as body mass index (BMI) are of primary interest because they are modifiable factors amenable to intervention.
A narrow focus on the effects of a behavior such as physical activity on BMI is shortsighted, however, because obesity is multifactorial and influenced by many interacting variables spanning biology to policy (National Heart Lung and Blood Institute, 2004). For example, most studies indicate moderate to strong genetic effects on BMI (Elks et al., 2012). The relative contributions of genetic and non-genetic influences on activity are controversial, however, with several European studies indicating moderate to strong genetic effects on physical activity participation (Carlsson et al., 2006; Eriksson et al., 2006; Joosen et al., 2005; Lauderdale et al., 1997; Maia et al., 2002; Perusse et al., 1989; Simonen et al., 2002; Stubbe et al., 2006). Environmental differences across the countries in which these studies were conducted could influence activity behaviors and limit the generalizability of findings, as demonstrated in a U.S. twin study in which unique environmental, and not genetic, factors primarily explained differences in physical activity participation (Duncan et al., 2008).
The role of the physical or built environment in supporting healthy lifestyles and subsequent effects on health outcomes has gained increased attention over the last decade. Consistent associations have been reported between built environmental features such as neighborhood “walkability” and activity outcomes, including more leisure-time and transportation-related walking (Ewing et al., 2003; Frank et al., 2004; Frank et al., 2007; Frank et al., 2006; Frank et al., 2005; Moudon et al., 2006; Moudon et al., 2007; Saelens et al., 2003). Neighborhood walkability itself has been associated with better health outcomes, including lower BMIs among residents (Black and Macinko, 2008; Diez Roux et al., 2002; Ewing et al., 2003; Frank et al., 2004; Frank et al., 2006; Papas et al., 2007). Neighborhood walkability is thought to exert its effects on health primarily by facilitating or inhibiting behaviors, such as physical activity and diet, which in turn strongly influence BMI (Papas et al., 2007). Others have suggested a broader view in which the physical and social environment affect the overall health and well-being of residents (Black and Macinko, 2008; CDC, 2007). For example, physical and/or social factors in the environment that contribute to stress, malnutrition, and inactivity could influence the risk of adverse health in the next generation through factors that act in utero and early life (Novak et al., 2006; Reynolds et al., 2013). Thus, neighborhood walkability could also influence genetic tendencies toward higher BMI. Regardless of the pathway linking neighborhood walkability to health, this ever-growing body of literature is dominated by observational studies with inherent methodological problems, chiefly an inability to adequately account for nonrandom selection of residents into neighborhoods (Oakes, 2004, 2006). Twins are an optimal way to address this self-selection problem–(Duncan et al., 2014; Turkheimer and Harden, 2014).
Although significant associations among activity level, neighborhood walkability, and BMI are common, in the absence of random assignment to environmental conditions such relationships do not imply causality. There are two major unmeasured confounds in the observed relationship between BMI and activity level. The first is broadly characterized as genetic: if persons with certain genetic characteristics are more likely to engage in regular exercise, and the same genes contributing to activity also contribute to BMI, a correlation will exist regardless of any causal relationship. A second confound is environmental. If people raised in affluent families living in supportive environments are more likely to be both active and have a lower BMI, then any correlation between activity and BMI could be mediated by family income or residential location rather than result from a direct causal relationship.
The co-twin control design helps address the aforementioned limitations by providing a quasi-experimental method of accounting for genetic and environmental selection processes that might otherwise confound purported causal relations among uncontrolled variables (Turkheimer and Harden, 2014). Thus, despite evidence that neighborhood walkability may protect against high BMI between families, no study to date has investigated whether this effect remains significant within families. Therefore, the purpose of the current study was to use quasi-experimental twin methods to test associations between physical activity level and neighborhood walkability (an index of activity-supportive environments) (Frank et al., 2007; Frank et al., 2006; Frank et al., 2005; Frumkin et al., 2004; Moudon et al., 2006) with BMI in twins. We hypothesized that activity levels and walkability would each be independently associated with BMI within twin pairs, controlling for common genetic and environmental background shared between predictors and outcome.
Materials and Methods
Participants
The University of Washington Twin Registry (UWTR) is a community-based sample of twins reared together; construction methods appear elsewhere (Afari et al., 2006; Strachan et al., 2013). Briefly, twins completed a survey with items on socio-demographics, health, and lifestyle behaviors. Standard questions about childhood similarity that determine zygosity with greater than 90% accuracy when compared with DNA-based methods were used to classify twins as identical (monozygotic; MZ) or fraternal (dizygotic; DZ) (Eisen et al., 1989; Spitz et al., 1996; Torgersen, 1979). We used 2008–2013 survey data; residential addresses were not available prior to 2008. Written informed consent was obtained, approved by the University’s institutional review board. The final sample included 4,060 female (2,802 MZ, 1,258 DZ) and 2,316 male (1,585 MZ, 731 DZ) pairs. Overall, the sample was young (39.4 ± 17.6 years) and predominantly White (90.3%).
Measures
Outcome
The primary outcome was BMI (kg/m2) from self-reported height and weight. In a separate study, self-reported BMI and directly measured BMI were highly correlated (r = 0.98, p < 0.01) among 200 UWTR twin pairs, indicating strong construct validity in our sample. There was a tendency for higher BMI individuals to under-report to a greater extent than lower BMI individuals (r = −0.27, p < 0.01), but this discrepancy was not correlated with activity level (r = −0.01, p = 0.80) or walkability (r = −0.03, p = 0.59).
Predictors
Twins reported the number of days per week they engaged in vigorous physical activity for at least 20 minutes and in a separate question moderate physical activity for at least 30 minutes. A continuous activity measure was constructed by summing moderate and vigorous days weighted by their respective durations (MVPA). This measure provides an estimate directly corresponding to activity levels recommended for health (Garber et al., 2011; U.S. Department of Health and Human Services, 1996). In a sub-sample of 104 twins who wore accelerometers and GPS devices over a two-week period in an ongoing funded study, subjective MVPA correlated significantly with objectively measured MVPA (r = 0.46, p < 0.01). There was a tendency for more active individuals to under-report MVPA to a greater extent than less active individuals (r = −0.71, p < 0.01), but this discrepancy was not associated with BMI (r = 0.13, p = 0.18). Twins also reported how many days during a typical week they walked in their neighborhood and minutes per walking bout (Lee and Moudon, 2006; Moudon et al., 2006). Responses of less than 15 minutes were coded as 10 minutes, whereas responses of 90 or more were top coded as 90 minutes.
Neighborhood walkability was estimated using two methods: the commercially available Walk Score® (Walk Score, 2012) and an index commonly cited in the urban planning and health literature created using measures of urban form (Frank et al., 2005). Addresses were entered into the Walk Score® website, which uses data from business listings, road networks, schools, and public transit to map walking distance to amenities in nine different categories (e.g., schools, parks, restaurants) (Walk Score, 2011). The algorithm then uses distances, counts, and weights to create a continuous score normalized on a scale of 0–100, with 0 representing the least and 100 the most “walkable” neighborhoods (Walk Score, 2011). The utility of Walk Score® as a walkability index has been published (Carr et al., 2010, 2011). The “planner’s” walkability index uses measures of land-use mix, intersection density, and residential density within a 1-km network buffer around the residence. The z-score for each of these variables is calculated and entered into an equation (Frank et al., 2005), providing a continuous score ranging between −8.98 to 27.34 (higher is more walkable). In a sub-sample of 3,162 UWTR twins, the two indices correlated strongly with each other (r = 0.78, p = 0.01).
Statistical analysis
All analyses were conducted using latent variable path analysis in Mplus (v. 7.0, Los Angeles, CA) (Muthen and Muthen, 2012) and maximum likelihood estimation. Analyses were conducted for same-sex pairs only and controlled for linear effects of age, sex, and race.
Univariate biometric decomposition
We used the classical twin model (ACE) to decompose variation in measures into three components: additive genetic (A), variance attributable to the additive effect of individual genes; shared environmental (C), variance attributable to environmental influences shared by twins raised in the same family; and non-shared environmental (E), variance attributable to environmental influences unique to the individual. This analysis lays the foundation for the more elaborate analyses that follow.
Biometric regression
The use of MZ and DZ twins in the regressions of BMI on walkability and activity level allows the separation of the phenotypic effect of the predictors from the genetic and shared environmental background that they may share. The regression of BMI on activity level, for example, has three components: the phenotypic regression of the outcome on the predictor, plus A and C regressions that provide alternative explanations of the predictor-outcome association. The A regression represents the tendency for pairs with high genetic loadings for activity to also have genetic loadings for BMI. Descriptively, an A regression of BMI on activity induces a covariance between the activity level of one member of a pair and the BMI of the other, more so for the MZ twins who share 100% of their genetic material than DZ twins who on average share half. The C regression represents the tendency for pairs to share a family environment conducive to both high activity and lower BMI. Descriptively, a C regression induces a covariance between the activity level of one twin and the BMI of the other, equally so for both MZ and DZ twins. With these pathways accounted for, the phenotypic regression represents the non-shared environmental effect of activity within pairs of twins. Descriptively, this means that a member of an identical twin pair with a higher activity level than the co-twin is also the twin with the lower BMI within the pair. The genetically informed phenotypic regression is the effect of the predictor on the outcome controlling for any common genetic or environmental background they share, and is therefore the best available approximation of any causal effect of the predictor (“quasi-causal” effect) (Turkheimer and Harden, 2014).
Results
Descriptive information for BMI and the predictors are shown in Table 1. Results were similar regardless of walkability index used; we present results using the Walk Score® derived index for simplicity. Walkability contained modest genetic and shared environmental variation, however, non-shared environment dominated (57%). Both MVPA and walking showed moderate heritability, almost no shared environmental variation, and large non-shared environmental variation (67% and 72%, respectively). In contrast, BMI consisted largely of additive genetic variation (76%), with no evidence of variation from the shared environment and modest contribution from the non-shared environment.
Table 1.
Descriptive statistics, twin intra-class correlations, and standardized variance components for walkability, physical activity, and body mass index, University of Washington Twin Registry, 2008–2013.
| Walkability | Moderate-to-Vigorous Physical Activity (min/week) | Walking (min/week) | Body Mass Index (kg/m2) | |
|---|---|---|---|---|
| Descriptive Statistics | ||||
| Mean (SD) | 40.6 (28.1) | 119.9 (92.2) | 88.4 (101.1) | 25.8 (5.5) |
| Sibling Correlations | ||||
| MZ | 0.43 (0.02) | 0.34 (0.01) | 0.29 (0.02) | 0.76 (0.01) |
| DZ | 0.34 (0.03) | 0.17 (0.02) | 0.13 (0.02) | 0.38 (0.02) |
| ACE Estimates | ||||
| h2 | 0.18 (0.06) | 0.31 (0.06) | 0.28 (0.02) | 0.76 (0.01) |
| c2 | 0.25 (0.06) | 0.02 (0.05) | 0.00 (0.01) | 0.00 (0.00) |
| e2 | 0.57 (0.02) | 0.67 (0.02) | 0.72 (0.02) | 0.24 (0.01) |
Abbreviations: dizygotic, DZ; monozygotic, MZ.
ACE estimates: additive genetic (h), variance attributable to the additive effect of individual genes; shared environmental (c), variance attributable to environmental influences shared by twins raised in the same family; and non-shared environmental (e), variance attributable to environmental influences unique to the individual.
Data presented as means and standard deviations in parentheses.
Next, we fit multivariate models in which activity level was regressed on walkability, and BMI was simultaneously regressed on walkability and activity. Unstandardized regression coefficients and standard errors for these non-genetically informed analyses are presented in the phenotypic regression columns of Table 2.
Table 2.
Trivariate models, body mass index predicted from environmental walkability and physical activity, University of Washington Twin Registry, 2008–2013.
| Regression | Moderate-to-Vigorous Physical Activity (min/week)
|
Walking (min/week)
|
||
|---|---|---|---|---|
| Phenotypic Regression (not genetically informed) | Quasi-Causal Regression (genetically informed) | Phenotypic Regression (not genetically informed) | Quasi-Causal Regression (genetically informed) | |
| Walkability → Activity | ||||
| bA | — | — | — | 0.09 (0.16) |
| bC | — | — | — | −0.06 (0.09) |
| bPhen | 0.01 (0.01) | 0.01 (0.01) | 0.04 (0.01)* | 0.04 (0.01)* |
| Walkability → BMI | ||||
| bA | — | — | — | — |
| bPhen | −0.02 (0.02) | −0.02 (0.02) | −0.01 (0.02) | −0.02 (0.02) |
| Activity → BMI | ||||
| bA | — | −0.92 (0.20)* | — | −0.91 (0.17)* |
| bPhen | −0.39 (0.03)* | −0.26 (0.03)* | −0.22 (0.03)* | −0.12 (0.03)* |
Abbreviation: body mass index, BMI (kg/m2).
The models represent the phenotypic regression of the outcome on the predictor (bPhen), plus A (bA) and C (bC) regressions that provide alternative explanations of the association between the predictor and the outcome.
Data presented as regression coefficients with standard errors in parentheses.
Statistically significant (p < 0.05) regression coefficient.
Walkability was not associated with MVPA (bPhen = 0.01, p = 0.12) or BMI (bPhen = −0.02, p = 0.32). Higher MVPA, however, predicted lower BMI such that each additional hour per week was associated with a 0.39 kg/m2 decrease in BMI. Walkability and MVPA jointly predicted 1.2% of the variance in BMI. Walkability and walking were positively associated; each additional unit in walkability was associated with a 2.4-minute increase in walking. As we observed in models using MVPA as the indicator for physical activity, walkability did not predict BMI (bPhen = −0.01, p = 0.55). More walking predicted lower BMI; each additional hour of walking was associated with a 0.22 kg/m2 decrease in BMI. Walkability and walking jointly predicted 0.5% of the variance in BMI.
We then decomposed nonzero phenotypic regression coefficients into their ACE components. The model is illustrated in Figure 1; unstandardized regression coefficients and standard errors for these genetically informed analyses are presented in the quasi-causal regression columns in Table 2. Controlling for genetic-based selection, the inverse association between MVPA on BMI remained statistically significant, although the association was reduced in magnitude (−0.26 vs. −0.39 for quasi-causal and phenotypic regressions, respectively). The A regression of BMI on MVPA was significant and substantially larger than the genetically informed bPhen (bA = −0.92 vs. −0.26).
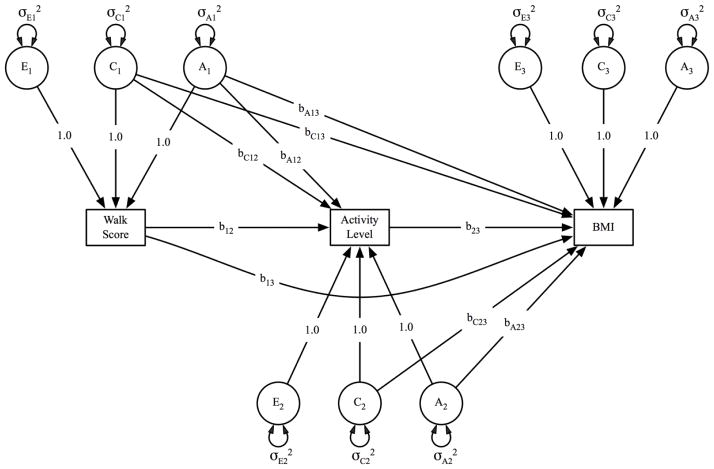
Figure 1.
Path diagram of the multivariate twin model.
Activity level is regressed on walkability (Walk Score®) via a phenotypic pathway (b12) in addition to genetic and shared environmental pathways (bA12 and bC12). Body mass index (BMI) is regressed on Walk Score and activity level via phenotypic pathways (b13 and b23, respectively), in addition to genetic (bA13 and bA23) and shared environmental (bC13 and bC23).
The regression of walking on walkability remained significant after accounting for genetic and shared environment and was not attenuated compared with the phenotypic regression. The walking-BMI association also remained significant while controlling for common genetic background and showed evidence of genetic-based selection (bA = −0.91, p < 0.01). The quasi-causal effect was substantially attenuated (bPhen = −0.22 and −0.12 in phenotypic and quasi-causal analyses, respectively).
Discussion
As expected, consistent with the literature, activity levels and BMI were significantly associated in the non-genetically informed analyses; each additional hour of MVPA was associated with a 0.39 unit reduction while each additional hour of neighbrohood walking was associated with a 0.22 unit reduction in BMI. However, a novel finding from this study relates to the genetically informed twin analysis, which suggests that much of these phenotypic relationships were the result of a common genetic background, as evidenced by reductions in the magnitude of the phenotypic relationship after controlling for A confounds. Nonetheless, a significant and potentially causal relationship of both MVPA and neighborhood walking with BMI remained after the common background was accounted for.
Neighborhood walkability was not associated with BMI in phenotypic analysis. Although neighborhood walkability had little effect on mean level of MVPA, it was associated with more neighborhood walking. Extending the literature, we found that this association remained significant even after accounting for genetic and shared environmental confounds, suggesting that neighborhood walkability is in fact causally related to more neighborhood walking. Together, our findings provide evidence supporting the ecological framework whereby a measure of the built environment – neighborhood walkability – is causally associated with physical activity behavior, which is subsequently causally associated with BMI.
Twin models not only account for genetic effects, twins reared together are also matched for shared environmental background, including numerous childhood and adolescent exposures. The ability to control for these confounds is another strength of the twin model, and has implications for overcoming the bias resulting from neighborhood self-selection. Studies using a categorical description of urbanization in non-U.S. samples have documented the influence of shared environmental factors on residence (Whitfield et al., 2005; Willemsen et al., 2005), other studies have revealed subtleties in the data. For example, a more recent report (Duncan et al., 2012) using a continuous walkability index employing specific information about neighborhood urban form (Frank et al., 2005) observed that shared environmental factors were the largest contributor to residential selection among younger twins (ages 18–24.9) whereas unique environment was largest in older twins (ages 25+). Although differences among these studies underline the complexity of residential selection, it is clear that choice of residence should not be considered a random environmental exposure. Both genetic and shared environmental selection processes are determinants of residential selection and must be taken into account in any non-experimental study of environmental risk factors. Although some artifacts of self-selection will exist in any study of the relationship between environment and behavior, using twins in this type of investigation controls for as many potential confounds as possible.
Strengths and limitations
The sample, although representative of Washington State, was largely White and middle class; thus, results are not necessarily generalizable to other geographic or socio-demographic groups. Both activity and BMI were self-reported and may suffer from measurement bias. Additionally, the physical activity measure did not capture details on activity type (e.g., running, swimming, etc.) or domains of activity (e.g., household, occupational, transportation, etc.), nor durations greater than 20 minutes for vigorous and 30 minutes for moderate activity. However, any potential underestimation of activity would lead to an underestimation of the “true” association between physical activity and BMI; thus, the magnitude of association found between MVPA/walking and BMI in the present study is a conservative estimate. In addition, the survey did not assess usual dietary intake, an unaddressed potential confounder. The use of the commercially-available Walk Score® algorithm for estimating walkability is also a limitation because the measure is primarily an index of utilitarian destinations accessible by walking, and to a lesser extent neighborhood density, and not other specific measures of urban form that have been correlated with physical activity in the literature. Given this potential limitation, we conducted analyses using two measures of walkability and results were consistent regardless of exposure used.
Finally, although we have used the twin method to control for genetic and environmental confounds of possible causal predictors of BMI, even these methods have residual confounding and are not as robust as random assignment in establishing causal relations. Because the data utilized here were strictly cross-sectional, we are unable to rule out the possibility of reverse causality – i.e., that differences in BMI cause differences in activity or residential selection. Data collection in the UWTR is ongoing, and longitudinal designs are planned.
Conclusions
Using a multivariate twin model, we demonstrated how twins can be used to compute quasi-experimental estimates of causal relations between uncontrolled factors, such as neighborhood walkability and physical activity, and health outcomes, such as BMI. The results suggest that much of the phenotypic relationship between physical activity (both MVPA and neighborhood walking) and BMI is the result of a common genetic background, but a significant and potentially causal relationship remains after this common background is accounted for. We also observed a tendency for walkability to promote walking (but not MVPA) above and beyond any genetic or shared environmental confounds.
Highlights.
Twin models can be used to compute quasi-experimental associations.
Moderate-to-vigorous activity is causally associated with body mass index.
Neighborhood walkability is causally association with neighborhood walking.
Neighborhood walking is causally associated with body mass index.
Acknowledgments
Funding: This work was supported by a grant from the National Institutes of Health (R01 AG042176 to G.E.D.). The National Institutes of Health played no role in the study design; the collection, analysis, or interpretation of data; the drafting of this manuscript; or the decision to submit it for publication.
The authors thank Ally Avery, Scientific Operations Manager, and the entire Registry staff for their diligent work in data collection, and the twins for taking part in the Registry.
Footnotes
Conflict of Interest: The authors have no competing interests to declare in relation to this manuscript.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Afari N, Noonan C, Goldberg J, Edwards K, Gadepalli K, Osterman B, Evanoff C, Buchwald D. University of Washington Twin Registry: construction and characteristics of a community-based twin registry. Twin Res Human Genet. 2006;9:1023–9. doi: 10.1375/183242706779462543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Diabetes Association. Economic costs of diabetes in the U.S. in 2007. Diabetes Care. 2008;31:596–615. doi: 10.2337/dc08-9017. [DOI] [PubMed] [Google Scholar]
- Anderson LH, Martinson BC, Crain AL, Pronk NP, Whitebird RR, O’Connor PJ, Fine LJ. Health care charges associated with physical inactivity, overweight, and obesity. Prev Chronic Dis. 2005;2:A09. [PMC free article] [PubMed] [Google Scholar]
- Black JL, Macinko J. Neighborhoods and obesity. Nutrition reviews. 2008;66:2–20. doi: 10.1111/j.1753-4887.2007.00001.x. [DOI] [PubMed] [Google Scholar]
- Carlsson S, Andersson T, Lichtenstein P, Michaelsson K, Ahlbom A. Genetic effects on physical activity: results from the Swedish Twin Registry. Med Sci Sports Exerc. 2006;38:1396–401. doi: 10.1249/01.mss.0000228941.17034.c1. [DOI] [PubMed] [Google Scholar]
- Carr LJ, Dunsiger SI, Marcus BH. Walk Score (TM) As a Global Estimate of Neighborhood Walkability. Am J Prev Med. 2010;39:460–63. doi: 10.1016/j.amepre.2010.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr LJ, Dunsiger SI, Marcus BH. Validation of Walk Score for estimating access to walkable amenities. Br J Sports Med. 2011;45:1144–8. doi: 10.1136/bjsm.2009.069609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. About Healthy Places. Centers for Disease Control and Prevention, National Center for Environmental Health; Atlanta, GA: 2007. [Google Scholar]
- Clinical Guidelines on the Identification, Evaluation and Treatment of Overweight and Obesity in Adults--The Evidence Report. National Institutes of Health. Obesity Res. 1998;6(Suppl 2):51S–209S. [PubMed] [Google Scholar]
- Diez Roux AV, Jacobs DR, Kiefe CI. Neighborhood characteristics and components of the insulin resistance syndrome in young adults: the coronary artery risk development in young adults (CARDIA) study. Diabetes Care. 2002;25:1976–82. doi: 10.2337/diacare.25.11.1976. [DOI] [PubMed] [Google Scholar]
- Duncan GE, Dansie EJ, Strachan E, Munsell M, Huang R, Vernez Moudon A, Goldberg J, Buchwald D. Genetic and environmental influences on residential location in the US. Health Place. 2012;18:515–9. doi: 10.1016/j.healthplace.2012.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan GE, Goldberg J, Noonan C, Moudon AV, Hurvitz P, Buchwald D. Unique environmental effects on physical activity participation: a twin study. PLoS One. 2008;3:e2019. doi: 10.1371/journal.pone.0002019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan GE, Mills B, Strachan E, Hurvitz P, Huang R, Moudon AV, Turkheimer E. Stepping towards causation in studies of neighborhood and environmental effects: How twin research can overcome problems of selection and reverse causation. Health Place. 2014;27C:106–11. doi: 10.1016/j.healthplace.2014.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisen S, Neuman R, Goldberg J, Rice J, True W. Determining zygosity in the Vietnam Era Twin Registry: an approach using questionnaires. Clin Genet. 1989;35:423–32. doi: 10.1111/j.1399-0004.1989.tb02967.x. [DOI] [PubMed] [Google Scholar]
- Elks CE, den Hoed M, Zhao JH, Sharp SJ, Wareham NJ, Loos RJ, Ong KK. Variability in the heritability of body mass index: a systematic review and meta-regression. Front Endocrinol. 2012;3:29. doi: 10.3389/fendo.2012.00029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eriksson M, Rasmussen F, Tynelius P. Genetic factors in physical activity and the equal environment assumption-- the Swedish young male twins study. Behav Genet. 2006;36:238–47. doi: 10.1007/s10519-005-9018-7. [DOI] [PubMed] [Google Scholar]
- Ewing R, Schmid T, Killingsworth R, Zlot A, Raudenbush S. Relationship between urban sprawl and physical activity, obesity, and morbidity. Am J Health Promot. 2003;18:47–57. doi: 10.4278/0890-1171-18.1.47. [DOI] [PubMed] [Google Scholar]
- Flegal KM, Carroll MD, Ogden CL, Curtin LR. Prevalence and trends in obesity among US adults, 1999–2008. JAMA. 2010;303:235–41. doi: 10.1001/jama.2009.2014. [DOI] [PubMed] [Google Scholar]
- Flegal KM, Carroll MD, Ogden CL, Johnson CL. Prevalence and trends in obesity among US adults, 1999–2000. JAMA. 2002;288:1723–7. doi: 10.1001/jama.288.14.1723. [DOI] [PubMed] [Google Scholar]
- Flegal KM, Graubard BI, Williamson DF, Gail MH. Excess deaths associated with underweight, overweight, and obesity. JAMA. 2005;293:1861–7. doi: 10.1001/jama.293.15.1861. [DOI] [PubMed] [Google Scholar]
- Frank LD, Andresen MA, Schmid TL. Obesity relationships with community design, physical activity, and time spent in cars. Am J Prev Med. 2004;27:87–96. doi: 10.1016/j.amepre.2004.04.011. [DOI] [PubMed] [Google Scholar]
- Frank LD, Saelens BE, Powell KE, Chapman JE. Stepping towards causation: do built environments or neighborhood and travel preferences explain physical activity, driving, and obesity? Soc Sci Med. 2007;65:1898–914. doi: 10.1016/j.socscimed.2007.05.053. [DOI] [PubMed] [Google Scholar]
- Frank LD, Sallis JF, Conway TL, Chapman JE, Saelens BE, Bachman W. Many pathways from land use to health - Associations between neighborhood walkability and active transportation, body mass index, and air quality. J Am Plann Assoc. 2006;72:75–87. [Google Scholar]
- Frank LD, Schmid TL, Sallis JF, Chapman J, Saelens BE. Linking objectively measured physical activity with objectively measured urban form: findings from SMARTRAQ. Am J Prev Med. 2005;28:117–25. doi: 10.1016/j.amepre.2004.11.001. [DOI] [PubMed] [Google Scholar]
- Frumkin H, Frank L, Jackson R. Urban Sprawl and Public Health. Designing, Planning, and Building for Healthy Communities. Island Press; Washington, D.C: 2004. [Google Scholar]
- Garber CE, Blissmer B, Deschenes MR, Franklin BA, Lamonte MJ, Lee IM, Nieman DC, Swain DP. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43:1334–59. doi: 10.1249/MSS.0b013e318213fefb. [DOI] [PubMed] [Google Scholar]
- Hedley AA, Ogden CL, Johnson CL, Carroll MD, Curtin LR, Flegal KM. Prevalence of overweight and obesity among US children, adolescents, and adults, 1999–2002. JAMA. 2004;291:2847–50. doi: 10.1001/jama.291.23.2847. [DOI] [PubMed] [Google Scholar]
- Joosen AM, Gielen M, Vlietinck R, Westerterp KR. Genetic analysis of physical activity in twins. Am J Clin Nutr. 2005;82:1253–9. doi: 10.1093/ajcn/82.6.1253. [DOI] [PubMed] [Google Scholar]
- Lauderdale DS, Fabsitz R, Meyer JM, Sholinsky P, Ramakrishnan V, Goldberg J. Familial determinants of moderate and intense physical activity: a twin study. Med Sci Sports Exerc. 1997;29:1062–8. doi: 10.1097/00005768-199708000-00012. [DOI] [PubMed] [Google Scholar]
- Lee C, Moudon AV. Correlates of Walking for Transportation or Recreation Purposes. J Physical Act Health. 2006;3:S77–S98. doi: 10.1123/jpah.3.s1.s77. [DOI] [PubMed] [Google Scholar]
- Maia JA, Thomis M, Beunen G. Genetic factors in physical activity levels: a twin study. Am J Prev Med. 2002;23:87–91. doi: 10.1016/s0749-3797(02)00478-6. [DOI] [PubMed] [Google Scholar]
- Moudon AV, Lee C, Cheadle AD, Garvin C, Johnson D, Schmid TL, Weathers RD, Lin L. Operational definitions of walkable neighborhood: theoretical and empirical insights. J Physical Act Health. 2006;3:S99–S117. doi: 10.1123/jpah.3.s1.s99. [DOI] [PubMed] [Google Scholar]
- Moudon AV, Lee C, Cheadle AD, Garvin C, Rd DB, Schmid TL, Weathers RD. Attributes of environments supporting walking. Am J Health Promot. 2007;21:448–59. doi: 10.4278/0890-1171-21.5.448. [DOI] [PubMed] [Google Scholar]
- Muthen LK, Muthen BO. Mplus. Statistical analysis with latent variables. User’s Guide. 7. Muthen & Muthen; Los Angeles, CA: 2012. [Google Scholar]
- National Heart Lung and Blood Institute. Predictors of obesity, weight gain, diet, and physical activity workshop. National Institutes of Health; 2004. [Accessed Sep 15, 2014]. Available at: http://www.nhlbi.nih.gov/research/reports/2004-obesity/index.htm. [Google Scholar]
- Novak DA, Desai M, Ross MG. Gestational programming of offspring obesity/hypertension. J Matern Fetal Neonatal Med. 2006;19:591–9. doi: 10.1080/14767050600708233. [DOI] [PubMed] [Google Scholar]
- Oakes JM. The (mis)estimation of neighborhood effects: causal inference for a practicable social epidemiology. Soc Sci Med. 2004;58:1929–52. doi: 10.1016/j.socscimed.2003.08.004. [DOI] [PubMed] [Google Scholar]
- Oakes JM. Commentary: advancing neighbourhood-effects research--selection, inferential support, and structural confounding. Int J Epidemiol. 2006;35:643–7. doi: 10.1093/ije/dyl054. [DOI] [PubMed] [Google Scholar]
- Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA. 2006;295:1549–55. doi: 10.1001/jama.295.13.1549. [DOI] [PubMed] [Google Scholar]
- Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011–2012. JAMA. 2014;311:806–14. doi: 10.1001/jama.2014.732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papas MA, Alberg AJ, Ewing R, Helzlsouer KJ, Gary TL, Klassen AC. The built environment and obesity. Epidemiologic Reviews. 2007;29:129–43. doi: 10.1093/epirev/mxm009. [DOI] [PubMed] [Google Scholar]
- Perusse L, Tremblay A, Leblanc C, Bouchard C. Genetic and environmental influences on level of habitual physical activity and exercise participation. Am J Epidemiol. 1989;129:1012–22. doi: 10.1093/oxfordjournals.aje.a115205. [DOI] [PubMed] [Google Scholar]
- Pronk NP, Goodman MJ, O’Connor PJ, Martinson BC. Relationship between modifiable health risks and short-term health care charges. JAMA. 1999;282:2235–9. doi: 10.1001/jama.282.23.2235. [DOI] [PubMed] [Google Scholar]
- Reynolds RM, Labad J, Buss C, Ghaemmaghami P, Raikkonen K. Transmitting biological effects of stress in utero: Implications for mother and offspring. Psychoneuroendocrinology. 2013;38:1843–9. doi: 10.1016/j.psyneuen.2013.05.018. [DOI] [PubMed] [Google Scholar]
- Saelens BE, Sallis JF, Black JB, Chen D. Neighborhood-based differences in physical activity: an environment scale evaluation. Am J Public Health. 2003;93:1552–8. doi: 10.2105/ajph.93.9.1552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simonen RL, Perusse L, Rankinen T, Rice T, Rao DC, Bouchard C. Familial aggregation of physical activity levels in the Quebec Family Study. Med Sci Sports Exerc. 2002;34:1137–42. doi: 10.1097/00005768-200207000-00014. [DOI] [PubMed] [Google Scholar]
- Spitz E, Moutier R, Reed T, Busnel MC, Marchaland C, Roubertoux PL, Carlier M. Comparative diagnoses of twin zygosity by SSLP variant analysis, questionnaire, and dermatoglyphic analysis. Behav Genet. 1996;26:55–63. doi: 10.1007/BF02361159. [DOI] [PubMed] [Google Scholar]
- Strachan E, Hunt C, Afari N, Duncan G, Noonan C, Schur E, Watson N, Goldberg J, Buchwald D. University of washington twin registry: poised for the next generation of twin research. Twin Res Human Genet. 2013;16:455–62. doi: 10.1017/thg.2012.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stubbe JH, Boomsma DI, Vink JM, Cornes BK, Martin NG, Skytthe A, Kyvik KO, Rose RJ, Kujala UM, et al. Genetic influences on exercise participation in 37.051 twin pairs from seven countries. PLoS ONE. 2006;1:e22. doi: 10.1371/journal.pone.0000022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torgersen S. The determination of twin zygosity by means of a mailed questionnaire. Acta Genet Med Gemellol. 1979;28:225–36. doi: 10.1017/s0001566000009077. [DOI] [PubMed] [Google Scholar]
- Turkheimer E, Harden KP. Behavior genetic research methods: Testing quasi-causal hypotheses using multivariate twin data. In: Reis HT, Judd CM, editors. Handbook of research methods in personality and social psychology. 2. Cambridge University Press; Cambridge, U.K: 2014. pp. 159–87. [Google Scholar]
- U.S. Department of Health and Human Services. Physical activity and health: a report of the Surgeon General. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion; Atlanta, GA: 1996. [Google Scholar]
- Walk Socre. [Accessed Sep 15, 2014];Walk Score Methodology. 2011 Available at: http://www.walkscore.com/methodology.shtml.
- Walk Score. [Accessed Sep 15, 2014];Get Your Walk Score - A Walkability Score For Any Address. 2012 Available at: www.walkscore.com.
- Wang YC, McPherson K, Marsh T, Gortmaker SL, Brown M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet. 2011;378:815–25. doi: 10.1016/S0140-6736(11)60814-3. [DOI] [PubMed] [Google Scholar]
- Whitfield JB, Zhu G, Heath AC, Martin NG. Choice of residential location: Chance, family influences, or genes? Twin Res Human Genet. 2005;8:22–26. doi: 10.1375/1832427053435391. [DOI] [PubMed] [Google Scholar]
- Willemsen G, Posthuma D, Boomsma DI. Environmental factors determine where the Dutch live: Results from the Netherlands Twin Register. Twin Res Human Genet. 2005;8:312–17. doi: 10.1375/1832427054936655. [DOI] [PubMed] [Google Scholar]