Abstract
Mesenchymal stem cells (MSCs) are multipotent cells that have the capability of differentiating into adipogenic, osteogenic, chondrogenic, and neural cells. With these multiple capabilities, MSCs have been highly regarded as effective transplantable cell source for regenerative medicine. A large bank of these cells can be found in several regions of the human umbilical cord (hUC) including the umbilical cord lining, the subendothelial layer, the perivascular zone, and most importantly in Wharton’s jelly (WJ). These cells, all umbilical cord-derived MSCs, are very durable, have large loading capacities, and are considered ethical to harvest because the umbilical cord is often considered a waste. These logistical advantages make WJ as appealing source of stem cells for transplant therapy. In particular, WJ is a predominantly good source of cells because MSCs in WJ (WJ-MSC) are maintained in a very early embryological phase and therefore have retained some of the primitive stemness properties. WJ-MSCs can easily differentiate into a plethora of cell types leading to a variety of applications. In addition, WJ-MSCs are slightly easier to harvest compared to other MSCs (such as bone marrow-derived MSCs). The fascinating stemness properties and therapeutic potential of WJ-MSCs provide great promise in many aspects of regenerative medicine and should be considered for further investigations as safe and effective donor cells for transplantation therapy in many debilitating disorders, which are discussed here. We previously reviewed WJ-MSCs therapeutic potential [1] and now provide an update on their recent preclinical and clinical applications.
Keywords: MSCs, multipotent cells, proliferation, differentiation, transplantation
Introduction
In recent years, medical research has focused on utilizing stem cell therapy to alleviate a number of debilitating disorders. In particular, recent efforts have turned to the human umbilical cord (hUC) for new sources of mesenchymal stem cells (MSCs). MSCs found in the hUC present several advantages over other stem cell tissue sources. First, hUC is seen as biological waste and typically discarded after birth. Its use therefore presents no ethical concerns [2]. Second, hUC cells exhibit reduced immunogenicity. Since these inactivated MSCs lack MHCII and other costimulatory molecules on their surface, they present no immune response in the host tissue. In laboratory studies, the allogenic transplantation of hUC cells into non-immune-suppressed animals did not produce rejection [3]. Third, hUC cells have an increased proliferative capacity, evidenced by a higher frequency of colony-forming-unit fibroblasts (CFU-F) and a shorter population doubling time than other cells [4].
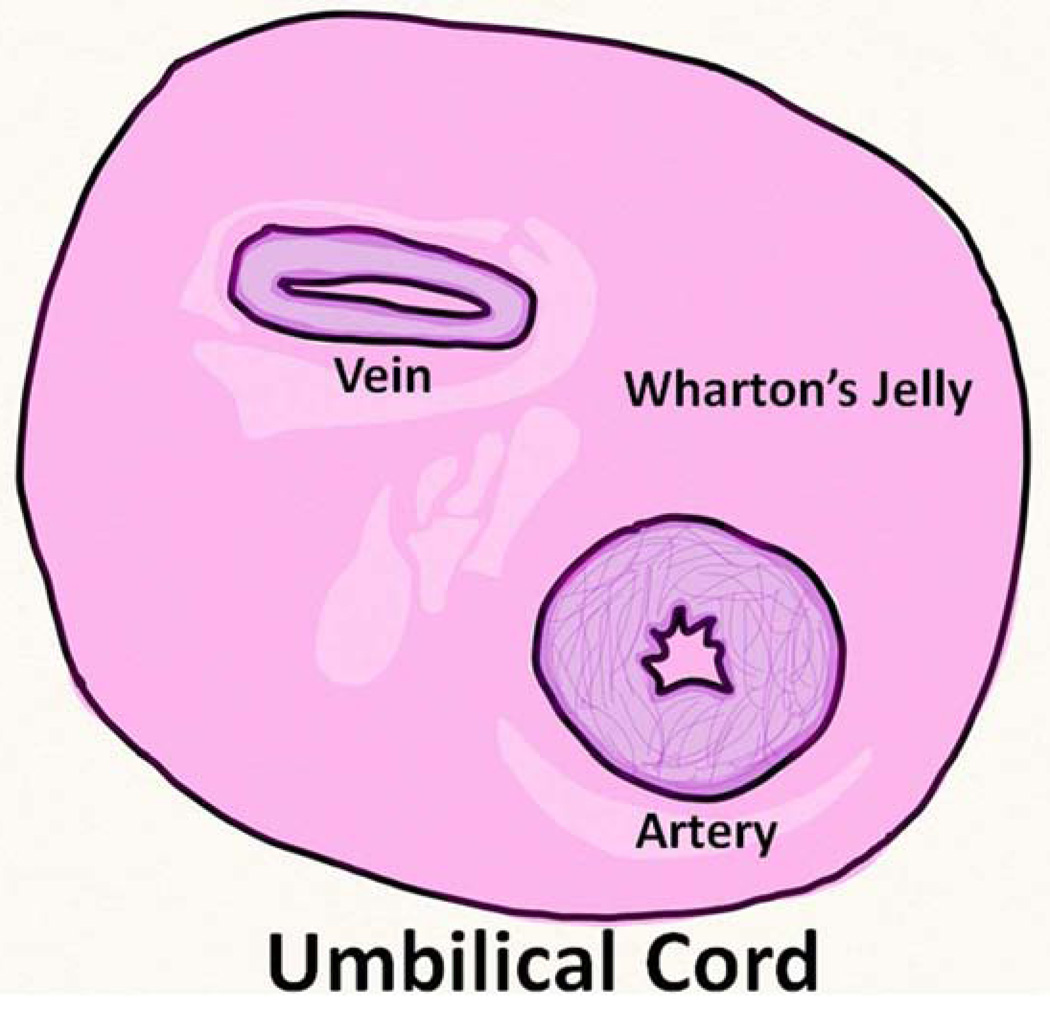
MSCs can be isolated from the umbilical cord (UC) lining, subendothelial layer, perivascular zone, and the Wharton’s Jelly (WJ) (the gelatinous matrix in the umbilical cord that provides insulation and protection of the vein and arteries of the umbilical cord) [5] (Figure 1). The MSCs found in these regions of the hUC are multipotent and can differentiate into adipogenic, osteogenic, chondrogenic, and neuronal cells [6].
Figure 1.
Anatomy of the human umbilical cord showing Wharton’s Jelly.
However, limitations still remain for the isolation of UC-MSCs for clinical use. For cord lining MSCs, the isolation methods are incredibly time-consuming. In addition, the current procedure for isolation of WJ-MSCs involves fetal bone serum (FBS) as its nutrient enhancement. The problem that results from this supplement is that viral and prion diseases become a major concern. Thus, standard isolation still needs to be modified [7]. An additional problem with their ability for clinical uses is the UC-MSC’s property to proliferate at high rates and, after tissue repair, differentiate into two daughter cells with asymmetric portions of the parent cell`s cytoplasm. Due to this, the MSCs present an enigmatic problem of not being capable to be tracked through MRIs after a short period of time elapses. Therefore determining whether the MSCs fully developed into the cell type it was primarily intended to and whether it is functioning correctly will be an arduous and inefficient process. Currently the stains of SPIO and Mn2+ are being used for the identification and tracking of MSCs. However these both present problems. The Mn stain has a high cytotoxicity and, although there is a solution to minimize the cytotoxicity, the impending risk is too high [8]. While SPIO stain does not possess this property it cannot track the MSC cells accurately and descends in clarity as more time elapses [9]. An experiment conducted with Prussian blue staining demonstrates this lack of consistency [10]. Therefore, future research efforts must focus on methods to more efficiently isolate MSCs from the umbilical cord [11] and an appropriate method of staining to enable MRI imaging of the MSCs.
Despite the setbacks in isolation, WJ-MSCs still present perhaps the best opportunity for cell therapy in the future. With a greater proliferative capacity and tri-lineage differentiation potential, these cells can be induced to form a diverse array of cell types than MSCs found in both the bone marrow and from different regions of the umbilical cord. This high differentiation potential can be envisioned to target a variety of disorders (Table 1). For example, UC stem cells exhibit an interesting potency to heal cutaneous burn wounds [12]. Both UC-epithelial cells and UC-MSCs can be grown on scaffolds and grafted to treat partial thickness and full thickness burns [12].
Table 1.
Milestone Discoveries for WJ Transplantation
| Disease Indication | WJ Cell Therapy Outcomes |
|---|---|
| Cancer | |
| Liver Disease | |
| Peripheral Nerve Damage: | |
| Cardiovascular Repair: | |
| Connective Tissue Repair: |
|
| Obesity and Diabetes: |
|
Cell Therapy for Cancer
In comparison to other sources of MSCs derived from the bone marrow, WJ-MSCs exhibit reduced immunogenicity. WJ-MSCs lack costimulatory ligands which activate an immune response from both B and T cells [13]. At the same time, WJ-MSCs express HLA-G, a protein that induces the expansion of regulatory T cells and suppresses cytotoxic T cells and Natural Killer cells, at a high level [14].
Interestingly enough, WJ-MSCs (referred to as UCMSCs in [14]) also possess properties that make them potential tools for cancer therapy. Cancer cells secrete cytokines and growth factors, and WJ-MSCs have receptors for these molecules in the cell membrane [14]. This interaction between cytokines and growth factors and their receptors results in WJ-MSCs exhibiting a tropism towards the inflammatory cancer and tumor tissues [14].
Moreover, WJ-MSCs also present tumorcidal abilities. Although bone marrow-derived MSCs have been shown to stimulate tumor growth, WJ-MSCs have been demonstrated to attenuate the growth of tumors [14]. While most of these properties are not fully understood, there are two mechanisms for cancer suppression that are known. The first is that WJ-MSCs produce several secretory proteins which in turn promote the cell death of cancer cells and stop the cell cycle [14]. In addition, WJ-MSCs also caused more tumor suppressing genes to be expressed thus aiding in the cancer treatment. The second mechanism by which cancer can be suppressed is through the enhancement of the immune system reaction to the cancer cells [14]. Studies have shown that rats treated with rat WJ-MSC displayed great improvement to tumors that they have [15]. Those rats possessed a highly reduced tumor, and an abundant amount of lymphocytes in the area, which infiltrate the tissue of the tumor and assist in therapy. In addition to their anti-cancer benefits, WJ-MSCs are also considered safe therapeutic cells because they do not pose a risk of spreading tumor tissue into other parts of the body or other adverse effects [16]. If research efforts can develop tumor suppressor genes or anticancer drugs in the future, WJ-MSCs can potentially be utilized as vehicles for targeted cancer therapy because of their durability, large loading capacity, ability to be harvested in large numbers with no risk to the donor, and tumor tropism [14].
Cell Therapy for Liver Disease
WJ-MSCs have also been explored as a method to cure liver diseases. Because of WJ-MSCs excessive proliferation and ability to differentiate into different cells they are perfect candidates for this type of treatment. These specific stem cells have been known to differentiate into adipocytes, osteoblasts, and also neurons [17, 18]. Because of the need for donors of livers and the harmful side effects of the liver transplantations, stem cell therapies are becoming recognized to be a much better method of curing liver disease. Morphological analysis demonstrates that WJ-MSCs express markers that correlate with the phenotype of hepatoblasts (the precursor to hepatocytes, the cell of the primary liver tissue), suggesting the ability for WJ-MSCs to differentiate into liver cells [19]. Hepatic commitment of these cells can potentially be induced in the presence of specific growth factors or the host liver cell environment. Moreover, in a model of fibrosis (the body’s response to chronic liver damage), the introduction of WJ-MSCs into the injured livers of mice was able to relieve fibrosis and reduce hepatic inflammation [20], possibly by abrogating extracellular matrix accumulation (caused by fibrosis). However, these results are controversial because other studies found that MSCs produce no benefit to hepatic function in the long term [21].
Transplantation of WJ-MSCs has also been tested in liver fibrosis. Using carbon tetrachloride (CCl4), rats were experimentally induced display liver fibrosis and 4 weeks later received WJ-MSCs injections [22]. After an additional 4 weeks, there was a remarkable decrease in the liver fibrosis in the rats treated with WJ-MSCs as compared to the rats that were not treated with the WJ-MSCs. Some WJ-MSCs exhibited phenotypes of the liver, and those WJ-MSCs that did not differentiate had the capability to secrete cytokines that have the potential to restore liver function [23]. These observations indicate a multi-pronged reparative mechanism of WJ-MSCs involving specific lineage differentiation and therapeutic molecules that are key pathways towards tissue repair.
Cell Therapy for Peripheral Nerve Damage
WJ-MSCs have also been proposed as a potential cure for peripheral nervous system (PNS) injuries. When a peripheral neuron is damaged, the Schwann cells lose contact to the next axon and therefore self-degrade their own myelin sheaths. The body responds with a proliferation of Schwann cells that support axonal regrowth and regeneration of myelin [24]. Therefore, research in cell therapy has explored the efficacy of transplanting Schwann cells to heal the injury. This is difficult, however, because isolation of Schwann cells can cause damage to other peripheral nerves, while the amount of Schwann cells able to be isolated is typically very low. MSCs offer a novel alternative stem cell source because they are easily accessible and highly proliferative. Because of the trans-differentiation potential of WJ-MSCs into ectoderm-derived cells, MSCs can potentially differentiate into functional Schwann cells and be applied to heal injured peripheral neurons [25]. To this end, the transplantation of WJ-MSCs into a rat with peripheral nerve damage revealed that the injected cells labeled with alentivirus green fluorescent protein to allow visualization of myelin of the regenerated axons, were able to differentiate and become functioning Schwann cells with an efficiency of 97%. These findings indicate that WJ-MSCs appear as effective donor cells for cell therapy in the future [25].
Cell Therapy for Cardiovascular and Connective Tissue Repair
Another potential use for WJ-MSCs is in cardiovascular tissue engineering. The cardiovascular system has low regenerative potential, which makes the use of WJ-MSCs an ideal alternative in cardiovascular tissue repair with their immunomodulatory properties and self-regenerating capacity [26]. Interestingly, biologically active heart valve leaflets could be engineered using only cells from the human umbilical cord [27]. These leaflets showed complex tissues that closely resembled native tissues. With such robust results, the use of WJ-MSCs became a large step in overcoming limiting factors in repairing congenital malformations. These efficacy readouts have also been confirmed in large animal studies, where engineered heart tissues were successfully transplanted into sheep, even showing good functional performance after 20 weeks [28]. However, uncertainty still remains over whether these engineered tissues can survive successfully in the long-term [29]. Additional research studies have found that culturing UC-MSCs in the presence of certain growth factors and hormones more efficiently induces UC-MSCs to differentiate into cardiomyogenic lineages. In particular, the presence of the hormone oxytocin gave rise to the most efficient differentiation [30].
UC-MSCs may also be beneficial in the treatment of cartilage injuries. Cartilage injuries are the result of a metabolic imbalance of an organism’s chondrocytes (the primary cell of cartilage tissue), and the natural growth and repair of cartilage tissue is slow [31]. Transplantation of UC-MSCs presents a potentially effective mechanism for cell therapy [32]. UC-MSCs, when cultured in a medium containing ascorbic acid, transferrin, dexamethasone, and other molecules, can differentiate into chondrocyte-like cells [33]. Therefore, stem cell transplantation stands as a strategy to substantially increase the number of chondrocytes, enabling a quicker recovery of cartilage diseases [34].
The same is true for tendon injuries. Human umbilical cord perivascular cells (HUCPVCs) have been shown to produce collagen that repairs tendon injuries in rats [35]. Additionally, the presence of HUCPVCs facilitated a change in structure and organization of the collagen fibers [35]. Instead of being disorganized, these collagen fibers were arranged in linear parallel bundles, thereby increasing the tendon’s tensile strength in comparison to the control group [36].
Cell Therapy for Obesity and Diabetes
The prenatal differentiation of WJ-MSCs may play a factor in determining an individual’s susceptibility to obesity and related disorders later in life. Obesity can be affected by increased adipogenesis, the early determination of MSCs to adipocytes before birth. This phenomenon is influenced heavily by the prenatal environment [37]. The changes in the prenatal environment have been largely ascribed to the mother’s health condition. When healthy mothers were compared to diabetic mothers a major difference was observed in the prenatal environments were the protein levels [37]. The changes in the concentration of CD90 is related to the change in plasticity and the up-regulation of CD44, CD29, CD73, CD166, SSEA4 while TERT is related to the increase of the proliferative ability of the cells. Studies show that WJ-MSCs from mothers with hyperglycemia or gestational diabetes mellitus have a higher affinity towards adipocyte differentiation and increased adipocyte differentiation efficiency than those from lean mothers [37]. The findings suggest that the changes in the prenatal environment in obese mothers pre-dispose the child to becoming obese or having Type II Diabetes later in life [37]. Also additional research suggests that WJ-MSC may have the potential to benefit in the direct treatment of diabetes mellitus [38]. By using markers that indicate when certain genes are expressed, models have shown that WJ-MSCs have the capability to differentiate into all sorts of pancreatic cells including the insulin-producing β cells [39]. Using immunohistochemistry and ELISA assays, a significantly greater amount of insulin and C-peptide protein was released from the differentiated cells than from the undifferentiated cells. These results suggest that the WJ-MSC did in fact turn insulin-producing cells. In just a week, WJ-MSCs can turn into the exact cells that are attacked by this aggressive disease. With more research and study, WJ-MSCs may provide a means to alleviate diabetes in the future [40].
Moreover, new cell isolation methods have emerged to more efficiently culture and expand the population of WJ-MSCs in a laboratory setting. A lower oxygen concentration (5%) than room air (21%), as well as a low plate density (10 cells/cm2), has powerful effects on WJ cell expansion, shortening population doubling time and substantially increasing the colony forming efficiency of the cells [41]. These new methods are exciting and hold promise as researchers search for even more efficient methods to harvest stem cells for developing cell therapies [41].
As mentioned previously, WJ-MSCs are potentially viable resources as donor cells for clinical transplantation use, due primarily to their high proliferative capacity and ability to differentiate between three different tissue lineages (ectoderm, mesoderm, and endoderm). WJ-MSCs are likely more beneficial than some other sources of MSCs (Table 2). For example, for several years, bone marrow MSCs have been demonstrated as the future of hematopoietic stem cell transplantation, primarily due to their intrinsic micro-environmental support for hematopoietic stem cells and ability to differentiate into various mesodermal lineages, much like the WJ-MSCs, with limited difficulty in isolation. However these stem cells are inferior due to the invasive procedures which are required for its aspiration [9].
Table 2.
Comparison of MSC Sources
| MSC Source | Potential Benefits | Potential Drawbacks |
|---|---|---|
| WJ | ||
| Bone Marrow | ||
| UC Blood |
|
|
| Amniotic Fluid |
|
|
| UC Matrix |
|
Another potent source of MSCs is the umbilical cord blood. Much like WJ-MSCs these are multi-faceted cells with the ability to differentiate into lineages whilst in either in vitro or in vivo condition and possess higher proliferative capacity than those MSCs of bone marrow [4]. However, studies suggest that MSCs derived from the umbilical cord blood are limited in their utility because technical challenges in extracting sufficient amounts of these cells.
Amniotic Fluid MSCs (AF-MSCs) are similar to those of Wharton’s Jelly in that they have a good differentiation capability and can be efficiently obtained from the placenta [11]. These multipotent stem cells may have a future in cell therapy and clinical use. However, since these cells have only recently emerged in the field, most researchers believe that further studies must be conducted to develop a proficient method to culture and isolate these cells [11].
Lastly, similarly to WJ-MSCs, Umbilical Cord Matrix MSCs (UCM-MSCs) are useful in tissue engineering and possibly cell therapy. They also have differentiation abilities, immunodulatory properties, and trophic activity [4]. However, the number of UCM-MSCs extracted is limited and unique ex-vivo expansion method is necessary to obtain sufficient numbers of UCM-MSCs [4].
Although all the sources of MSCs possess comparable stemness properties (differentiation, proliferation capacity), and present with potential for clinical cell-based therapeutic use, and all sources display nearly analogous post-transplantational effects, the ease in isolating and propagating ample supply of WJ-MSCs, combined with their high proliferative capacity and ability to differentiate into the three germ lineages make WJ-MSCs appealing donor cells for transplantation therapy.
Conclusions
Recent evidence demonstrates that WJ-MSCs are potential transplantable cells for treatment of devastating diseases, such as cancer and diabetes. Their use in cell therapy will be an integral addition to the field of regeneration. WJ-MSCs have a multitude of benefits such as their high proliferation rate [42], lower doubling time, and ability to function with non-immune-suppressed animals [43]. However, there remains paucity in the translation of WJ-MSCs for clinical use, largely due to the cells’ heterogeneity, which results from the current isolation methods and inefficient staining methods [44]. There are two primary explanations for heterogeneity. First, the hUC has multiple distinct anatomical zones, and previous attempts at isolating WJ-MSCs have inadvertently harvested cells from different anatomical structures of the hUC in addition to the Wharton’s Jelly. Second, there is currently wide variation in procedures to harvest WJ-MSCs, and this variation can produce inconsistent results between studies [11]. Future research and refinement of isolation procedures can potentially overcome these obstacles [11]. Regardless of these drawbacks WJ-MCSs are still the ideal future for cell therapy; their properties of high proliferation capability and versatility to differentiate between three lineages allow them to lower immunogenicity and have the potential to treat an array of diseases and disorders [45].
In addition, WJ-MSCs stimulate immune responses from B and T cells [13] and suppress cytotoxic and natural killer cells [14]. WJ-MSCs possess cytokines and growth factor receptors, which allow them to be vital tools for cancer therapy. In such therapy, WJ-MCSs drastically weaken the cancerous tumors by secreting therapeutic proteins which promote the cancerous cells to undergo cell death and to stop the cell cycle [46]; moreover, WJ-MSCs enhance the immune response to cancer cells. With the minimum risk of spreading the cancer cells throughout the body or to the MSC donor, WJ-MSCs have the potential to serve as vehicles for delivery of tumor suppressive genes and anticancer drugs occur.
Apart from cancer treatment WJ-MCSs also can facilitate cell-based therapies for liver diseases and diabetes mellitus due to their high proliferation and differentiation ability [47], e.g., WJ-MSCs can express hepatoblastic phenotypes and can become liver cells or pancreatic cells [48].
As we recognized the many versatile capabilities of WJ-MSCs, their documented efficacy in animal models and limited clinical trials as therapeutic cells advances the field of regenerative medicine. With more research, WJ-MSCs may someday become recognized as routine donor cells for cell-based therapies [49, 50, 51].
Acknowledgements
CVB is funded by National Institutes of Health 1R01NS071956-01A1, Department of Defense W81XWH-11-1-0634, and VA Merit Review.
Abbreviations
- hUC
Human Umbilical Cord
- MSC
Mesenchymal Stem Cells
- CFU-F
Colony-Forming-Unit Fibroblasts
- FBS
Fetal Bone Serum
- UC-MSC
Umbilical Cord Mesenchymal Stem Cells
- WJ-MSC
Wharton’s Jelly Mesenchymal Stem Cells
- WJ
Wharton's Jelly
- HUCPVC
Human Umbilical Cord Perivascular Cells
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosure of Interest: CVB holds patents and has pending patents in stem cell biology and applications.
References
- 1.Kim DW, Staples M, Shinozuka K, Pantcheva P, Kang SD, Borlongan CV. Wharton's Jelly-Derived Mesenchymal Stem Cells: Phenotypic Characterization and Optimizing Their Therapeutic Potential for Clinical Applications. International Journal of Molecular Sciences. 2013;14(6):11692–11712. doi: 10.3390/ijms140611692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Batsali AK, Kastrinaki MC, Papadaki HA, Pontikoglou C. Mesenchymal Stem Cells Derived from Wharton's Jelly of the Umbilical Cord: Biological Properties and Emerging Clinical Applications. Current Stem Cell Research and Therapy. 2013;8(2):144–155. doi: 10.2174/1574888x11308020005. [DOI] [PubMed] [Google Scholar]
- 3.Cho PS, Messina DJ, Hirsh EL, Chi N, Goldman SN, Lo DP, Harris IR, Popma SH, Sachs DH, Huang CA. Immunogenicity of umbilical cord tissue–derived cells. Blood Journal. 2008;111(1):430–438. doi: 10.1182/blood-2007-03-078774. [DOI] [PubMed] [Google Scholar]
- 4.Conconi MT, Di Liddo R, Tommasini M, Calore C, Parnigotto PP. Phenotype and Differentiation Potential of Stromal Populations Obtained from Various Zones of Human Umbilical Cord: An Overview. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:6–20. [Google Scholar]
- 5.Sibov TT, Pavon LF, Miyaki LA, Mamani JB, Nucci LP, Alvarim LT, Silveira PH, Marti LC, Gamarra L. Umbilical cord mesenchymal stem cells labeled with multimodal iron oxide nanoparticles with fluorescent and magnetic properties: application for in vivo cell tracking. International Journal of Nanomedicine. 2014;9:337–350. doi: 10.2147/IJN.S53299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang HS, Hung SC, Peng ST, Huang CC, Wei HM, Guo YJ, Fu YS, Lai MC, Chen CC. Mesenchymal stem cells in the Wharton's jelly of the human umbilical cord. Stem Cells. 2004;22(7):1330–1337. doi: 10.1634/stemcells.2004-0013. [DOI] [PubMed] [Google Scholar]
- 7.Venugopal P, Balasubramanian S, Majumdar AS, Ta M. Isolation, characterization, and gene expression analysis of Wharton's jelly-derived mesenchymal stem cells under xeno-free culture conditions. Stem Cells Cloning. 2011;4:39–50. doi: 10.2147/SCCAA.S17548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kim DW, Staples M, Shinozuka K, Pantcheva P, Kang SD, Borlongan CV. Wharton's Jelly-Derived Mesenchymal Stem Cells: Phenotypic Characterization and Optimizing Their Therapeutic Potential for Clinical Applications. Int J Mol Sci. 2013;14(6):11692–11712. doi: 10.3390/ijms140611692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Prasanna SJ, Jahnavi VS. Wharton's Jelly Mesenchymal Stem Cells as Off-The-Shelf Cellular Therapeutics: A Closer Look into their Regenerative and Immunomodulatory Properties. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:28–38. [Google Scholar]
- 10.Chung J, Yamada M, Yan PC. Magnetic resonance imaging of human embryonic stem cells. Current Protocols in Stem Cell Biology. 2009;Chapter 5(Unit 5A) doi: 10.1002/9780470151808.sc05a03s10. [DOI] [PubMed] [Google Scholar]
- 11.Jeschke MG, Gauglitz GG, Phan TT, Herndon DN, Kita K. Umbilical Cord Lining Membrane and Wharton’s Jelly-Derived Mesenchymal Stem Cells: the Similarities and Differences. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:21–27. [Google Scholar]
- 12.Branski LK, Gauglitz GG, Herndon DN, Jeschke MG. A review of gene and stem cell therapy in cutaneous wound healing. Burns: journal of the International Society for Burn Injuries. 2009;35:171–180. doi: 10.1016/j.burns.2008.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Racz GZ, Kadar K, Foldes A, Kallo K, Perczel-Kovach K, Keremi B, Nagy A, Varga G. Immunomodulatory and potential therapeutic role of mesenchymal stem cells in periodontitis. J Physiol Pharmacol. 2014;65(3):327–339. [PubMed] [Google Scholar]
- 14.Tamura M, Kawabata A, Ohta N, Uppalapati L, Becker KG, Troyer D. Wharton’s Jelly Stem Cells as Agents for Cancer Therapy. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:39–47. [Google Scholar]
- 15.Chanran Ganta, Doi Chiyo, Rie Ayuzawa, Rajashekar Rachakatla, Marla Pyle, Gordon Andrews, Mark Weiss, Masaaki Tamura, Deryl Troyer. Rat Umbilical Cord Stem Cells Completely Abolish Rat Mammary Carcinomas with No Evidence of Metastasis or Recurrence 100 Days Post-tumor Cell Inoculation. Cancer Res. 2009;69(5):1815–1820. doi: 10.1158/0008-5472.CAN-08-2750. [DOI] [PubMed] [Google Scholar]
- 16.Matsuzuka T, Rachakatla RS, Doi C, Maurya DK, Ohta N, Kawabata A, Pyle MM, Pickel L, Reischman J, Marini F, Troyer D, Tamura M. Human umbilical cord matrix-derived stem cells expressing interferon-beta gene significantly attenuate bronchioloalveolar carcinoma xenografts in SCID mice. Lung Cancer. 2010;70(1):28–36. doi: 10.1016/j.lungcan.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Scheers I, Lombard C, Paganelli M, Campard D, Najimi M, Gala JL, Decottignies A, Sokal E. Human umbilical cord matrix stem cells maintain multilineage differentiation abilities and do not transform during long-term culture. PLoS One. 2013;8(8) doi: 10.1371/journal.pone.0071374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kim T, Momin E, Choi J, Yuan K, Zaidi H, Kim J, Park M, Lee N, McMahon MT, Quinones-Hinojosa A, Bulte JW, Hyeon T, Gilad AA. Mesoporous Silica-Coated Hollow Manganese Oxide Nanoparticles as Positive T Contrast Agents for Labeling and MRI Tracking of Adipose-Derived Mesenchymal Stem Cells. J Am Chem Soc. 2011 Mar 9;133(9):2955–2961. doi: 10.1021/ja1084095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Scheers I, Lombard C, Najimi M, Sokal EM. Cell Therapy for the Treatment of Metabolic Liver Disease: An Update on the Umbilical Cord Derived Stem Cells Candidates. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:48–53. [Google Scholar]
- 20.Lin SZ, Chang YJ, Liu JW, Chang LF, Sun LY, Li YS, Luo GH, Liao CH, Chen PH, Chen TM, Lee RP, Yang KL, Harn HJ, Chiou TW. Transplantation of Human Wharton's Jelly-Derived Stem Cells Alleviates Chemically Induced Liver Fibrosis in Rats. Cell Transplantation. 2010;19(11):1451–1463. doi: 10.3727/096368910X514198. [DOI] [PubMed] [Google Scholar]
- 21.Wang H, Zhao T, Xu F, Li Y, Wu M, Zhu D, Cong X, Liu Y. How important is differentiation in the therapeutic effect of mesenchymal stromal cells in liver disease? Cytotherapy. 2014;16(3):309–316. doi: 10.1016/j.jcyt.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 22.Atasever A, Yaman D. The effects of grape seed and colchicine on carbon tetrachloride induced hepatic damage in rats. Exp Toxicol Pathol. 2014 doi: 10.1016/j.etp.2014.04.008. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 23.Tsai PC, Fu TW, Chen YM, Ko TL, Chen TH, Shih YH, Hung SC, Fu YS. The therapeutic potential of human umbilical mesenchymal stem cells from Wharton's jelly in the treatment of rat liver fibrosis. Liver Transplantation. 2009;15(5):484–495. doi: 10.1002/lt.21715. [DOI] [PubMed] [Google Scholar]
- 24.Matsuse D1, Kitada M, Kohama M, Nishikawa K, Makinoshima H, Wakao S, Fujiyoshi Y, Heike T, Nakahata T, Akutsu H, Umezawa A, Harigae H, Kira J, Dezawa M. Human umbilical cord-derived mesenchymal stromal cells differentiate into functional Schwann cells that sustain peripheral nerve regeneration. J Neuropathol Exp Neurol. 2010;69(9):973–985. doi: 10.1097/NEN.0b013e3181eff6dc. [DOI] [PubMed] [Google Scholar]
- 25.Kuroda Y, Kitada M, Wakao S, Dezawa M. Mesenchymal Stem Cells and Umbilical Cord as Sources for Schwann Cell Differentiation: their Potential in Peripheral Nerve Repair. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:54–63. [Google Scholar]
- 26.Corotchi MC, Popa MA, Remes A, Sima LE, Gussi I, LupuPlesu M. Isolation method and xeno-free culture conditions influence multipotent differentiation capacity of human Wharton's jelly-derived mesenchymal stem cells. Stem Cell Research and Therapy. 2013 doi: 10.1186/scrt232. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schmidt D1, Mol A, Breymann C, Achermann J, Odermatt B, Gössi M, Neuenschwander S, Prêtre R, Genoni M, Zund G, Hoerstrup SP. Living Autologous Heart Valves Engineered From Human Prenatally Harvested Progenitors. Circulation. 2006;114:125–131. doi: 10.1161/CIRCULATIONAHA.105.001040. [DOI] [PubMed] [Google Scholar]
- 28.Alexandre N, Ribeiro J, Gärtner A, Pereira T, Amorim I, Fragoso J, Lopes A, Fernandes J, Costa E, Santos-Silva A, Rodrigues M, Santos JD, Maurício AC, Luís AL. Biocompatibility and hemocompatibility of polyvinyl alcohol hydrogel used for vascular grafting—In vitro and in vivo studies. Journal of Biomedical materials Research. 2014 doi: 10.1002/jbm.a.35098. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 29.Semenov OV, Breymann C. Mesenchymal Stem Cells Derived from Wharton’s Jelly and their Potential for Cardio-Vascular Tissue Engineering. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:64–71. [Google Scholar]
- 30.Hollweck T, Hartmann I, Eblenkamp M, Wintermantel E, Reichart B, Überfuhr P, Eissner G. Cardiac Differentiation of Human Wharton`s Jelly Stem Cells–Experimental Comparison of Protocols. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:95–102. [Google Scholar]
- 31.Chung JY, Song M, Ha CW, Kim JA, Lee CH, Park YB. Comparison of articular cartilage repair with different hydrogel-human umbilical cord blood-derived mesenchymal stem cell composites in a rat model. Stem cell Res Ther. 2014 doi: 10.1186/scrt427. Epud ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dahlin RL, Kinard LA, Lam J, Needham CJ, Lu S, Kasper FK, Mikos AG. Articular chondrocytes and mesenchymal stem cells seeded on biodegradable scaffolds for the repair of cartilage in a rat osteochondral defect model. Biomaterials. 2014 doi: 10.1016/j.biomaterials.2014.05.055. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fensky F, Reichert JC, Traube A, Rackwitz L, Siebenlist S, Nöth U. Chondrogenicpredifferentiation of human mesenchymal stem cells in collagen type I hydrogels. Biomed Tech (Berl) 2014 doi: 10.1515/bmt-2013-0076. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 34.Anzalone R, Lo Iacono M, Corrao S, Magno F, Loria T, Cappello F, Zummo G, Farina F, La Rocca G. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:72–81. [Google Scholar]
- 35.Emrani H, Davies JE. Umbilical Cord Perivascular Cells: A Mesenchymal Cell Source for Treatment of Tendon Injuries. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:112–119. [Google Scholar]
- 36.Hernigou P, FlouzatLachaniette CH, Delambre J, Zilber S, Duffiet P, Chevallier N, Rouard H. Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. International Orthopaedics. 2014 doi: 10.1007/s00264-014-2391-1. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 37.Pierdomenico L, Lanuti P, Lachmann R, Grifone G, Cianci E, Gialò L, Pacella S, Romano M, Vitacolonna E, Miscia S. Diabetes Mellitus During Pregnancy Interferes with the Biological Characteristics of Wharton's Jelly Mesenchymal Stem Cells. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:103–111. [Google Scholar]
- 38.Cao X, Han ZB, Zhao H, Liu Q. Transplantation of mesenchymal stem cells recruits trophic macrophages to induce pancreatic beta cell regeneration in diabetic mice. The International Journal of Biochemistry and Cell Biology. 2014 doi: 10.1016/j.biocel.2014.06.003. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 39.Bhandari DR, Seo KW, Sun B, Seo MS, Kim HS, Seo YJ, Marcin J, Forraz N, Roy HL, Larry D, Colin M, Kang KS. The simplest method for in vitro β-cell production from human adult stem cells. Differentiation. 2011;82:144–152. doi: 10.1016/j.diff.2011.06.003. [DOI] [PubMed] [Google Scholar]
- 40.D'Addio F, Trevisani A, Ben Nasr M, Bassi R, El Essawy B, Abdi R, Secchi A, Fiorina P. Harnessing the immunological properties of stem cells as a therapeutic option for diabetic nephropathy. Acta Diabetologica. 2014 doi: 10.1007/s00592-014-0603-1. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 41.López Y, Seshareddy K, Trevino E, Cox J, Weiss ML. Evaluating the Impact of Oxygen Concentration and Plating Density on Human Wharton’s Jelly-Derived Mesenchymal Stromal Cells. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:82–94. [Google Scholar]
- 42.Usha Nekanti, Sumitava Dastidar, Parvathy Venugopal, Satish Totey, Malancha Ta. Increased Proliferation and Analysis of Differential Gene Expression in Human Wharton’s Jelly-derived Mesenchymal Stromal Cells under Hypoxia. Int J Biol Sci. 2010;6(5):499–512. doi: 10.7150/ijbs.6.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nagamura-Inoue T, He H. Umbilical cord-derived mesenchymal stem cells: Their advantages and potential clinical utility. World J Stem Cells. 2014;6(2):195–202. doi: 10.4252/wjsc.v6.i2.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Christodoulou I, Kolisis FN, Papaevangeliou D, Zoumpourlis V. Comparative Evaluation of Human Mesenchymal Stem Cells of Fetal (Wharton's Jelly) and Adult (Adipose Tissue) Origin during Prolonged In Vitro Expansion: Considerations for Cytotherapy. Stem Cells Int. 2013;2013:246134. doi: 10.1155/2013/246134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wu S, Ju G-Q, Du T, Zhu Y-J, Liu G-H. Microvesicles Derived from Human Umbilical Cord Wharton’s Jelly Mesenchymal Stem Cells Attenuate Bladder Tumor Cell Growth In Vitro and In Vivo. PLoS ONE. 2013;8(4):e61366. doi: 10.1371/journal.pone.0061366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Han Ihn, Yun Miyong, Eun-Ok Kim, Bonglee Kim, Min-Hyung Jung, Sung-Hoon Kim. Umbilical cord tissue-derived mesenchymal stem cells induce apoptosis in PC-3 prostate cancer cells through activation of JNK and downregulation of PI3K/AKT signaling. Stem Cell Res Ther. 2014;5(2):54. doi: 10.1186/scrt443. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 47.Hongwu Wang, Xiaoyan Qiu, Ping Ni, Xuerong Qiu, Xiaobo Lin, Weizhao Wu, Lichun Xie, Limin Lin, Juan Min, Xiulan Lai, Yunbin Chen, Guyu Ho, Lian Ma. Immunological characteristics of human umbilical cord mesenchymal stem cells and the therapeutic effects of their transplantion on hyperglycemia in diabetic rats. Int J Mol Med. 2014 Feb;33(2):263–270. doi: 10.3892/ijmm.2013.1572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Patcharee Prasajak, Wilairat Leeanansaksiri. Developing a New Two-Step Protocol to Generate Functional Hepatocytes from Wharton's Jelly-Derived Mesenchymal Stem Cells under Hypoxic Condition. Stem Cells Int. 2013;2013:762196. doi: 10.1155/2013/762196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lange-Consiglio A, Corradetti B, Rutigliano L, Cremonesi F, Bizzaro D. In Vitro Studies of Horse Umbilical Cord Matrix-Derived Cells: From Characterization to Labeling for Magnetic Resonance Imaging. The Open Tissue Engineering and Regenerative Medicine Journal. 2011;4:120–133. [Google Scholar]
- 50.De la Fuente A, Mateos J, Lesende-Rodríguez I, Calamia V, Fuentes-Boquete I, de Toro FJ, Arufe MC, Blanco FJ. Proteome analysis during chondrocyte differentiation in a new chondrogenesis model using human umbilical cord stromamesenchymal stem cells. Mol Cell Proteomics. 2012;11 doi: 10.1074/mcp.M111.010496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu KH1, Zhou B, Lu SH, Feng B, Yang SG, Du WT, Gu DS, Han ZC, Liu YL. In vitro and in vivo differentiation of human umbilical cord derived stem cells into endothelial cells. J Cell Biochem. 2007;100(3):608–616. doi: 10.1002/jcb.21078. [DOI] [PubMed] [Google Scholar]