Abstract
Mammalian genomes can be edited by injecting pronuclear embryos with Cas9 cRNA and guide RNA (gRNA) but it is unknown whether editing can also occur during the onset of embryonic development, prior to pronuclear embryogenesis. We here report Cas9-mediated editing during sperm-induced meiotic exit and the initiation of development. Injection of unfertilized, mouse metaphase II (mII) oocytes with Cas9 cRNA, gRNA and sperm enabled efficient editing of transgenic and native alleles. Pre-loading oocytes with Cas9 increased sensitivity to gRNA ~100-fold. Paternal allelic editing occurred as an early event: single embryo genome analysis revealed editing within 3 h of sperm injection, coinciding with sperm chromatin decondensation during the gamete-to-embryo transition but prior to pronucleus formation. Maternal alleles underwent editing after the first round of DNA replication, resulting in mosaicism. Asymmetric editing of maternal and paternal alleles suggests a novel strategy for discriminatory targeting of parental genomes.
Gene-targeted mice facilitate functional genetic analysis in vivo but the manner in which they are typically produced via embryonic stem (ES) cells is laborious, time-consuming and expensive. Gene targeting in larger species, although increasingly relevant in biomedicine, is even more difficult.
Recently, two structurally-discrete classes of DNA binding domain, zinc finger nucleases (ZFNs) and transcription activator-like effector nucleases (TALENs), have been used to generate sequence-specific intracellular double-strand DNA breaks in metazoan genomes. ZFN- or TALEN-generated breaks may be repaired by error-prone nonhomologous end joining (NHEJ) to generate insertions or deletions (indels) that produce non-functional (null) alleles in cultured mammalian cells or single-cell embryos1,2. ZFN-generated double-strand breaks in cultured cells stimulate high-fidelity homology-dependent repair (HDR) by several orders of magnitude3,4,5 and enable HDR-mediated gene targeting in mouse and rat single-cell embryos (known as zygotes) at efficiencies of 2.4–25% (Ref. 1). However, the production of ZFNs and TALENs is complex, and pairs of each must be tailor-made for each targeted region. The efficiency of targeting is also variable and animals have not been produced by simultaneous ZFN- or TALEN-mediated targeting of multiple alleles.
These barriers have recently been negotiated by a powerful newcomer to mammalian genome engineering technology: the clustered, regularly interspaced, short palindromic repeat (CRISPR)/Cas9 system of adaptable bacterial immunity6,7. Cas9 is a DNA endonuclease whose site-specificity is determined by a single-stranded CRISPR RNA. CRISPR RNAs have been modified from their original bacterial source (eg Streptococcus pyogenes) and Cas9 codon-optimized to function effectively in mammalian cells6,8. The Cas9 system differs from ZFN and TALEN technologies because it utilizes a single protein - Cas9 - for all modifications, with target specificity provided by modified CRISPR guide RNA (gRNA). The gRNA contains a 20 nucleotide (nt) sequence that forms a heteroduplex with its complementary DNA target (frequently on the genome), which can be any sequence upstream of the proto-spacer adjacent motif (PAM), NGG6. Typical gRNAs are relatively short (~110 nt)6 and thus easy to synthesize. With these advantages, the Cas9 system has rapidly been adopted to introduce targeted mutations in yeast, plants, Drosophila, C. elegans, zebrafish, mice, rats, pigs and macaques (reviewed in Ref. 9). Cas9 has been used for multiplex targeting, with one-step NHEJ-mediated editing of 8 alleles at an efficiency of 10% in human cultured cells and biallelic editing with an 80% hit-rate in single-cell mouse embryos (pronuclear zygotes)10. Efficient early embryonic genome targeting is especially promising because it leads directly to mutant offspring11,12,13,14.
In fertilization, sperm combine with metaphase II (mII) oocytes and become denuded of nucleoprotein (protamine) in a process termed decondensation, before their genomes recondense with maternal histones and form a nuclear structure, the pronucleus15,16,17. (The maternal genome simultaneously forms a separate pronucleus.) The early phase in which sperm decondensation occurs coincides with the gamete-to-embryo transition and supports efficient transgene integration at quasi-random sites18,19; transgenes preferentially integrate into the sperm-derived genome (rather than the oocyte-derived genome), possibly because protein depletion during sperm decondensation makes it a better substrate for recombination17. This suggests that the paternal genome might also be a relatively good substrate for Cas9-mediated genome engineering20. We sought to test this possibility and report that Cas9 promotes high-efficiency genome editing in the decondensation phase that immediately follows intracytoplasmic sperm injection (ICSI) of unfertilized mII oocytes (Fig. 1A).
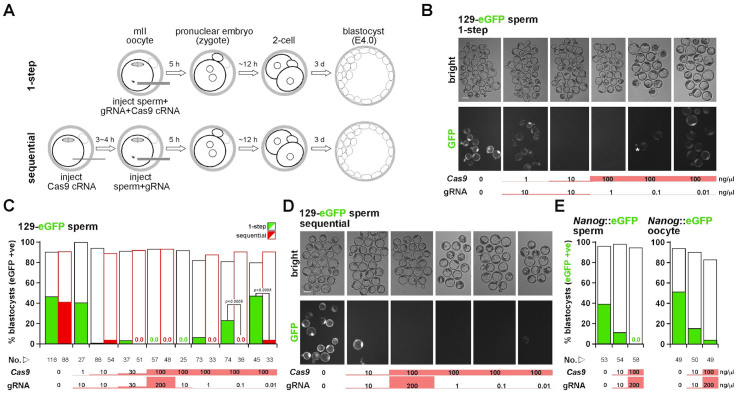
Figure 1. Cas9-mediated editing in mII exit following ICSI.
(A) Schematic of 1-step (upper) and sequential methods of Cas9-mediated mII editing. mII, metaphase II. (B) Paired Hoffman modulation (upper) and eGFP expression (eGFP) images of E4.0 blastocysts produced by 1-step injection of wt mII oocytes with 129-eGFP sperm from hemizygotes, with concentrations of injected Cas9 cRNA and eGFP gRNA indicated below. An asterisk indicates a presumptively phenotypic mosaic. Bar, 100 μm. (C) Numerical representation of embryo development and green fluorescence following injection of sperm from 129-eGFP hemizygous males. Percentages are of blastocyst development on embryonic day 4 (E4.0) (open) and of blastocysts that fluoresced green (filled) indicating 129-eGFP transgene expression in 1-step (green) or sequential (red) methods. Starting embryo numbers and injected concentrations of Cas9 cRNA and eGFP gRNA (gRNA) are shown beneath. (D) Paired Hoffman modulation (upper) and eGFP expression (eGFP) images as for (B) except that the sequential method of injection (Fig. 1A) was used. (E) Histograms as for (C) except that one of either the injected sperm (left) or oocyte carried a Nanog-eGFP knock-in allele. Red highlights provide at-a-glance indication of RNA concentrations in (B) to (E).
Results and discussion
To evaluate genome editing during meiotic exit, we first generated a test-bed transgenic line (129-eGFP) by introducing a single-copy ubiquitously-expressed pCAG-eGFP transgene (eGFP) onto the 129/Sv background. When sperm heads from 129-eGFP hemizygotes (ie eGFP+/− males) were injected into wild-type (wt) B6D2F1 mII oocytes and the resulting embryos cultured, over-all, 90.5% (n = 116) developed to the blastocyst stage by embryonic day 4 (E4.0), of which 46.6% fluoresced green, indicating that they expressed a functional eGFP gene (Fig. 1B,C). When sperm from 129-eGFP hemizygous males were coinjected with 100 ng/μl cRNA encoding Cas9 and 200 ng/μl eGFP gRNA (the 1-step method of Fig. 1A), 93.3% developed to the blastocyst stage (n = 57) but none (0%) fluoresced green (Fig. 1B,C). This is the expected result if the eGFP alleles had been efficiently edited to become non-functional. When we titrated Cas9 cRNA and eGFP gRNA under these conditions, we found that they were effective at concentrations at or above 1–10 ng/μl (Fig. 1C). Paternal mosaicism was occasionally observed at near-threshold concentrations of active gRNA (~1 ng/μl; asterisk in Fig. 1B).
Our previous work has shown that cRNA injected into mII oocytes takes 3~4 h before it is discernibly expressed21,22. We therefore reasoned that loading mII oocytes with Cas9 cRNA prior to sperm/gRNA injection might enhance editing by lengthening the time for Cas9 expression. To investigate this, we sequentially injected oocytes first with Cas9 cRNA and after 3~4 h, eGFP gRNA plus sperm from a 129-eGFP hemizygote (Fig. 1A). As in the 1-step method, the Cas9 system efficiently eliminated green fluorescence compared to controls (Fig. 1B–D). These results were confirmed by analogous experiments with a Nanog-eGFP knock-in line (Fig. 1E and Supplementary Fig. S1A). This shows that genome targeting by injecting mII oocytes with sperm, Cas9 cRNA and gRNA can be achieved high efficiencies and is not locus-specific.
To evaluate whether the sequential injection enhanced editing, we performed a comparative titration of gRNA (1-step vs sequential methods), holding the concentration of injected Cas9 cRNA constant and non-limiting at 100 ng/μl (Fig. 1B–D). Editing in the 1-step method became inefficient when ~1 ng/μl eGFP gRNA was injected (Fig. 1C). However, a similar editing efficiency in the sequential method was obtained with 0.01 ng/μl eGFP gRNA, corresponding to ~7 × 108 gRNA molecules per injection (Fig. 1C). Therefore, in these experiments, the sequential method of Cas9-mediated paternal genomic editing was ~100-fold more sensitive than the 1-step method. One interpretation of this finding is that the potential for editing in the 1-step method declines before sufficient Cas9 has been synthesized, implying that paternal genome editing is generally an early event.
To assess Cas9 activity towards the maternal genome, wt B6D2F1 sperm were injected into oocytes from females carrying an eGFP transgene. Interpretation of these experiments in the 129-eGFP line was confounded by carry-over of eGFP protein expressed during oocyte maturation, but the problem was mitigated with oocytes from Nanog-eGFP hemizygotes, in which there is little or no carry-over (Fig. 1E and Supplementary Fig. S1B). Following injection with 200 ng/μl gRNA and 100 ng/μl Cas9 cRNA, which targeted the paternal genome with ~100% efficiency, 4.0% of E4.0 blastocysts were GFP-positive (49<n<58, p<0.0005) (Fig. 1E and Supplementary Fig. S1B).
Direct sequencing of reverse-transcriptase PCR amplimers corresponding to paternal eGFP transcripts (ie derived from a sperm-borne eGFP transgene) in completely non-green-fluorescent E4.0 blastocysts produced unambiguous reads (Fig. 2A,B). The unmixed sequences revealed that single targeting events had occurred to produce indels within or immediately 5' of the gRNA-matching eGFP genomic segment (n = 26) for both sequential and 1-step editing, whether pCAG-eGFP or Nanog-eGFP transgenes were targeted (Fig. 2A,B). However, targeting of maternal pCAG-eGFP or Nanog-eGFP transgenes derived from oocytes typically (in 14/15 cases) produced sequence mosaicism (Fig. 2C). In some cases, the sequence ambiguity suggested that cells contained a non-mutated allele as well as a mutated one (Fig. 2C) and Nanog-eGFP mutations were detected in two green fluorescent embryos, presumably either reflecting mosaicism or functionality retained by mutated eGFP alleles.
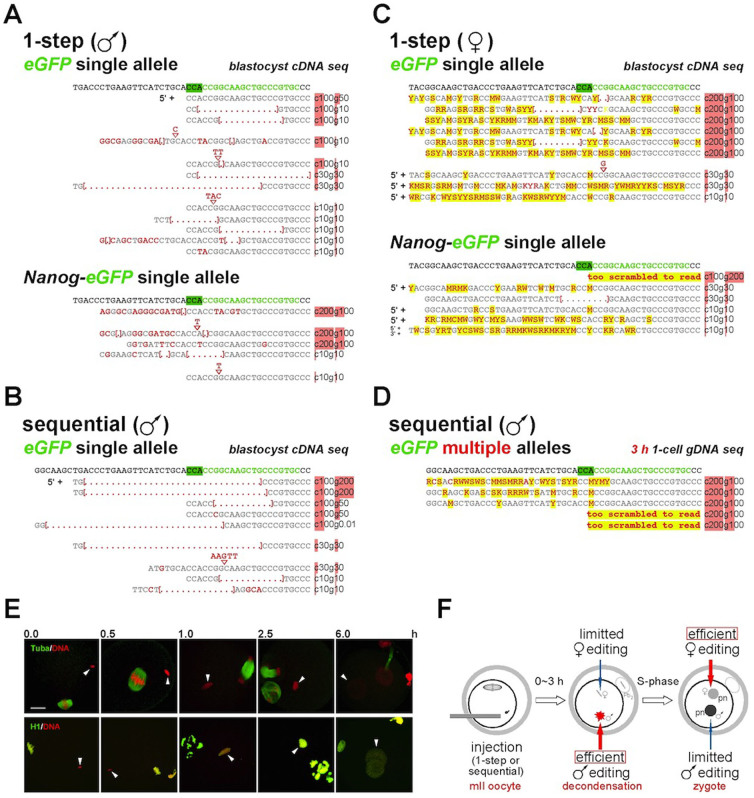
Figure 2. Sequence analysis reveals allelic asymmetry in parental genome mII editing.
(A) Sequences of reverse-transcriptase PCR products from embryonic day 4 (E4.0) blastocysts developing after the 1-step method of editing (Fig. 1A) in which Cas9 cRNA and eGFP gRNA were co-injected into mII oocytes with sperm from 129-eGFP single-copy hemizygotes (eGFP single). The gRNA-target sequence (green typeface) plus adjacent 5′ sequence is displayed on the top row and mutants beneath (grey typeface), with the corresponding Cas9 cRNA (c) and eGFP gRNA (g) concentrations (ng/μl) that were injected to produced them. The proto-spacer adjacent motif (PAM) is highlighted in green. Mutations are indicated in red typeface. 5′ +, mutations detected 5′ (but not 3′) of the displayed sequence. (B) Sequences of editing mutants produced as per (A), by injecting sperm from 129-eGFP (upper) or Nanog-eGFP knock-in hemizygotes with wt oocytes, except by the sequential method (Fig. 1A). (C) Sequences of editing mutants as for (A), except that the transgenic alleles were maternal; wt 129 sperm were injected into mII oocytes obtained from 129-eGFP single copy (upper) or Nanog-eGFP knock-in hemizygotes. 3′ +, mutations were detected 3′ of the displayed sequence. Yellow highlighting indicates ambiguous calls presumptively produced by multiple targeting events. (D) Whole genome amplification of individual embryos collected 3 h after injection with sperm from a hemizygous transgenic line (eGFP multi) carrying two copies of the eGFP transgene. The annotation used is as for (C). Red highlights provide at-a-glance indication of RNA concentrations in (A) to (D). (E) Merged confocal immunofluorescence images of single embryos at the times indicated (h) after ICSI, showing DNA labelled with propidium iodide (red) and antibody labeling (green) of tubulin-α (Tuba, upper panels) or histone H1 (H1). Both sperm and oocytes were wt. White arrowheads indicate paternal chromatin. Bar, 100 μm. (F) Schematic depicting a model for Cas9-mediated editing following injection of mII oocytes (mII). Limitted editing of maternal alleles during the gamete-to-embryo transition is inherent to the system, whereas limitted editing of paternal alleles in zygotes is because available targets have already been removed. Pb2, second polar body; pn, pronucleus.
These findings suggest that parent-of-origin-specific chromatin behaviour during the gamete-to-embryo transition leads to parental asymmetries in Cas9-mediated genome editing. The generation in most cases of mixed maternal alleles (mosaicism) where only one allele was available for editing indicates that maternal genome editing usually occurred after DNA replication (S-phase). Although maternal editing typically occurred after S-phase, paternal genome editing at non-limiting Cas9 and gRNA concentrations was usually complete pre-S-phase, giving rise to a single event that removed the gRNA target and prevented subsequent editing. Since pCAG-eGFP or Nanog-eGFP transgenic lines are present in different backgrounds, this phenomenon is allele- and strain-independent.
We next sought to delineate the time-frame for paternal genome editing. To do this, we performed single-cell whole genome amplification and target locus genomic PCR after wt oocytes had been coinjected with eGFP transgene-carrying sperm plus Cas9 and gRNA (Fig. 2D). To increase the likelihood of detecting editing in single cells, we used males from a different line carrying 2 eGFP transgene copies. Sampling was 3 h after injection. All five (100%) of the transgenic 1-cell embryos analyzed had undergone editing of at least one genomic copy of eGFP (Fig. 2D). The 3 h editing time-frame corresponds to the period in which sperm decondensation occurs (Fig. 2E,F), well before pronuclear formation or the onset of mitotic S-phase after ~7 h23. This distinguishes Cas9-mediated editing following mII oocyte injection from that following injection of pronuclear zygotes which have entered the mitotic cell cycle and accordingly contain mitotic chromatin17.
We next evaluated editing of native alleles in the gamete-to-embryo transition and monitored transmission of mutations to offspring. Tyr was chosen because its null phenotypes are readily detectable: Tyr mutations in C57BL/6 (black) mice result in a white coat colour and/or changes in eye morphology and pigmentation24,25. Cas9 cRNA and Tyr gRNA were coinjected with wt C57BL/6 sperm into wt C57BL/6 oocytes and resultant 2-cell embryos transferred to pseudo-pregnant recipient surrogate mothers. Some experiments included an inert gRNA directed against Foxn1. Between 14.8 and 24.0% of founders exhibited complete or mosaic phenotypes expected for loss of Tyr function for 1-step and sequential methods (Table 1; Supplementary Fig. S2). These comprised loss of pigmentation in fur and both eyes, sometimes predisposing to ophthalmic irregularities or anophthalmia26.
Table 1. Development of embryos and phenotypes of offspring in experiments to edit Tyr alleles.
| in vitro | in vivo | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| method | [gRNA] | [Cas9 cRNA] | survived | pn | 2-cell | blast | born/tf | preg/tf | white | mosaic | % phen |
| 1-step | Tyr30 | 30 ng/μl | 148/160 | 136 | 132 | 19/26 | 60/106 | 7/7 | 0 | 9 | 15.8 |
| 1-step | Tyr30 + Fox30 | 30 ng/μl | 129/148 | 128 | 127 | 17/25 | 11/60 | 1/3 | 0 | 2 | 20.0 |
| 1-step | Tyr 200 | 100 ng/μl | 59/67 | 57 | 56 | 7/8 | 28/48 | 3/3 | 2 | 4 | 24.0 |
| 1-step | Tyr200 + Fox200 | 100 ng/μl | 54/67 | 51 | 48 | nd | 31/48 | 3/3 | 0 | 5 | 17.9 |
| seq | Tyr30 + Fox30 | 30 ng/μl | 84/93 | 78 | 78 | 8/18 | 30/60 | 3/3 | 2 | 2 | 14.8 |
Experiments were initiated on ≥2 experimental days for each treatment. seq, sequential method (Fig. 1A); Tyr30, Tyr gRNA at 30 ng/μl; Fox30, Foxn1 gRNA at 30 ng/μl; pn, pronclear embryos 6~8 h after sperm injection; blast, blastocyst; nd, not determined; tf, embryo transfer at the 2-cell stage; preg, number of recipients (recipients falling pregnant). Mosaic phenotypes group coat colour and eye changes. Some pups were rejected by foster mothers prior to weaning. Percentages with phenotype changes (% phen) exclude pups that died perinatally.
Most (23/27, 85.2%) founder phenotype changes only affected part of the mouse (Supplementary Fig. S2A), reflecting somatic mosaicism and showing that editing had occurred after the first S-phase. We investigated this by sequencing DNA from offspring with and without white coat or eye phenotypes (Supplementary Fig. S3A, B). Most of the founders with evident phenotypic alterations contained one or more Tyr mutations (Supplementary Fig. S3A). In addition, many (4/6) randomly-selected founders with no discernable phenotypic changes also possessed Tyr mutations. Mutations were typically (10/12) of the mixed type (Supplementary Fig. S3A). Since a similar mixed pattern of mutations was observed for maternal alleles of eGFP (Fig. 2C) the phenomenon is not locus- or sequence-specific. Consistent with our findings, multiple edited alleles have also been observed following injection of Cas9/gRNA components into pronuclear zygotes25.
Mutations were transmitted to offspring, including inheritance of the non-pigmented eye phenotype in F1 progeny of coat colour founders (with overtly normal eyes), indicating that coat colour and eye phenotypes have a shared aetiology and further revealing founder mosaicism (Supplementary Fig. S2 and Supplementary Fig. S3B).
These experiments demonstrate that injecting mII oocytes with Cas9 cRNA, gRNA and sperm efficiently produces embryos and offspring with edited genomes. Single cell analysis revealed that editing was detectable by 3 h. This period (the first 3 h) overlaps with the gamete-to-embryo transition involving meiotic exit, sperm decondensation (Fig. 2E,F) and polar body cytokinesis28,27 and precedes by several hours the appearance of pronuclear structures that define the zygote. Recombination within 3 h of sperm delivery would account for the distinctive patterns of paternal and maternal editing we observed (Fig. 2A–C) if sperm decondensation provided a unique genomic opportunity for recombination. Editing of the paternal allele removes the gRNA target, precluding subsequent editing. We did not detect mixed-type paternal allelic editing under optimal, non-limiting conditions (Fig. 1B,C), suggesting that editing occurred during developmental onset, prior to S-phase. By contrast, edited maternal alleles were typically mixed (Fig. 2C). Editing when the Cas9 system is injected into pronuclear zygotes also causes mosaicism25, indicating that the same mechanism operated. This implies that, consistent with observations in transgenesis18, meiotic exit and the gamete-to-embryo transition do not efficiently support maternal genome editing, plausibly because the structure of maternal chromatin during this phase is refractory to the editing machinery. Although the maternal genome is inherently refractory to editing in the gamete-to-embryo transition, the limitted evidence here for paternal genome editing in zygotes presumably reflects the removal of available targets by efficient editing at the earlier stage (Fig. 2F).
Genome editing during meiotic progression has several practical implications. The possibility that the paternal genome is preferentially targeted during decondensation implies that it might be possible to devise strategies to alter only one parental allele prescriptively, even where both have identical DNA sequences. Selective editing of the paternal allele, for example, may have utility in the study of imprinting or to alter subtle deleterious (epi)mutations. The relative recombinogenicity of decondensing paternal chromatin also opens the possibility that it might support a broader repertoire of targeting strategies, including the use of ‘nickases' (Ref. 28), nuclear transfer, different delivery platforms29, and the efficient deletion12 or integration30 of large DNA fragments; a comprehensive evaluation of large fragment targeting would benefit from a more complete description of HDR in mammalian mII oocytes20.
In addition, editing during the gamete-to-embryo transition may have utility in larger species. Although obtaining pronuclear zygotes is relatively straightforward in the mouse, this is not always the case for large commercial breeds and the efficiency of integration and transmission of exogenous DNA remains low31. The use of mII oocytes, which in some species (eg pigs) can be derived by in vitro maturation, may address this limitation, with delivery of the editing system by ICSI32 or other methods29. Finally, ICSI in human assisted reproduction is widely used33 and this or analogous approaches may one day enable human genome targeting or editing during very early development. This formal possibility will require exhaustive evaluation, but if successful, could enable genomic surgery for gene repair during the initiation of embryogenesis.
Methods
Animals
Animal procedures complied with the statutes of the Animals (Scientific Procedures) Act, 1986, approved by the University of Bath Animal Welfare and Ethical Review Body and the Biosciences Services Unit. Wild-type mouse strains were bred from stocks in-house or otherwise supplied by Charles River (L'Arbresle, France). A pCAG-eGFP transgene was introduced into the 129Sv background to produce multi- and single-copy, high-expressing lines18, and additional transgenic lines were generated in-house or were kind gifts and contained pCAG-eGFP or pNanog-eGFP transgenes on mixed background hybrid lines containing a contribution from C57BL/6.
Collection and culture of oocytes
Oocytes were collected from 8~12-week-old females following standard super-ovulation by serial intraperitoneal injection of 5 IU pregnant mare serum gonadotropin (PMSG) and 5 IU human chorionic gonadotropin (hCG). Oviductal metaphase II (mII) oocytes were collected in M2 medium (Specialty Media, USA) ~15 h post-hCG injection essentially as described34. After repeated washing in M2, denuded oocytes were incubated in kalium simplex optimized medium (KSOM; Specialty Media, USA)35 under mineral oil in humidified 5% CO2 (v/v air) at 37°C, until required.
Sperm preparation and microinjection
Sperm preparation was essentially as previously described21,22. Briefly, cauda epididymidal sperm from 8~12-week-old males were triturated for 45 sec in nuclear isolation medium (NIM; 125 mM KCl, 2.6 mM NaCl, 7.8 mM Na2HPO4, 1.4 mM KH2PO4, 3.0 mM EDTA; pH 7.0) containing 1.0% (w/v) 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonate (CHAPS) at room temperature (25°C). Sperm were washed twice in NIM and pelleted (1,890 g) at ambient temperature; head-tail detachment was enhanced by trituration during pellet resuspension. Sperm were resuspended in ice-cold NIM (~0.5 ml per epididymis) and stored at 4°C for up to 3 h until injection. Typically, ~50 μl of each suspension was mixed with 20 μl of polyvinylpyrrolidone (PVP, average Mr ≈ 360,000; Sigma, UK) solution (15% [w/v]) and sperm injected (ICSI) using a piezo-actuated pipette (Prime Tech, Japan) into oocytes in a droplet of M2 within ~60 min, essentially as described34. Assuming an inner pipette tip diameter of 6 μm, we estimate that 2~2.5 pl were introduced per injection. After a brief (5~15 min) recovery period, injected oocytes were transferred to KSOM under mineral oil equilibrated in humidified 5% CO2 (v/v air) at 37°C.
Immunocytochemistry and imaging
Immunocytochemistry, differential interference contrast microscopy (DIC) and fluorescence imaging were essentially as described previously21,22. In brief, mII oocyte and 1-cell embryo samples for the images in Figure 2E were incubated overnight at 4°C with mouse anti-α-tubulin (1:2000 [v/v]; Sigma) or -H1 (1:1000; Santa Cruz, USA) antibodies, followed by a 1 h incubation at 37°C with secondary antibody (1:250; Life Technologies Ltd., UK) conjugated to Alexa 488. DNA was stained by incubating samples at 37°C for 20 min in propidium iodide (1:200; Sigma, USA). Fluorescence was visualized on an Eclipse E600 (Nikon, Japan) microscope equipped with a Radiance 2100 laser scanning confocal system (BioRad, USA). Images were processed with ImageJ (http://imagej.nih.gov/ij/) analysis software. Embryos were imaged on an Olympus IX71 equipped with an Andro Zyla sCMOS camera and OptoLED illumination system (Cairn Research Ltd., UK) and processed using Metamorph software (Molecular Devices, LLC, USA). Excitation at 484 nm with an ET-EYFP filter system was used to detect eGFP epifluorescence. Imaging of blastocysts for Figure 1 and Supplementary Figure S1 was following culture in drops of KSOM (Specialty Media, USA) under mineral oil in 6 cm dishes in a humidified 5% CO2 (v/v air) incubator at 37°C21,22.
Embryo transfer
E1.5 (2-cell) embryos (the day following activation) were transferred to the oviductal ampullae of pseudo-pregnant CD-1 females at day 0.5 (ie plugged females that had been mated with vasectomized males the previous night). Pups were delivered by natural birth and where appropriate, fetuses, pups and placentae collected by Caesarian section at the desired time point. Newborn pups were fostered by CD-1 females as appropriate.
Vector construction
For target gRNA synthesis, we employed the pT7-gRNA backbone vector system36. Target gRNA sequences were selected using the CRISPR gRNA design tool (DNA 2.0) and informed the design of complementary oligonucleotides of the general sequences: TAGGN20 (forward) and AAACN20 (reverse)(Eurofins MWG Operon). Forward and reverse oligonucleotides (10 μM each) were annealed by incubating in 20 μl 1× NEB buffer solution at 95°C for 5 min, ramping down to 50°C at 0.1°C/sec followed by 50°C for 10 min then ramping to 4°C at 1°C/sec. 1 μl of annealed oligonucleotides were mixed with 400 ng of pT7-gRNA vector, 0.5 μl BsmBI, 0.3 μl BglII, 0.3 μl SalGI, 0.5 μl T4 DNA ligase, 1 μl 10× NEB buffer 3, 1 μl T4 ligase buffer and 4 μl nuclease-free water. One-step digestion and ligation were performed in a PCR machine using the parameters: 3 cycles of 37°C for 20 min, 16°C for 15 min, followed by incubation at 37°C for 10 min and 55°C for 15 min. Products (2 μl) were used to transform DH5α. Positives (>90%) were verified by diagnostic BglII digestion and sequencing and purified pT7-gRNA constructs were linealized by BamH1 digestion and used to program in vitro gRNA synthesis using a MEGAscript T7 Transcription Kit (Life Technologies).
Synthesis of cRNA and gRNA
5′-capped and polyadenylated Cas9 cRNA was synthesized in a T7 mScript™ Standard mRNA Production System (Cellscript, USA) as previously described21,22 from the T7 P3s-Cas9HC vector. Guide RNA (gRNA) was synthesized using a MEGAshortscript T7 Transcription Kit (Invitrogen, USA) according to the instructions of the manufacturer. RNAs were dissolved in nuclease-free water, quantified on a nanophotometer (Implen, Germany) and stored in aliquots at −80°C until required. Immediately prior to injection, RNA solutions were diluted as appropriate with nuclease-free water. In the sequential injection method, oocytes were first injected with Cas9 cRNA solution and following culture for 3~4 h the same oocyte was injected with a single sperm in the appropriate gRNA dilution. For 1-step injection, gRNA and Cas9 cRNA were mixed with a sperm suspension to give the appropriate final injection concentrations and a single sperm injected into the oocyte. Following injection, embryos were cultured in vitro either to the blastocyst stage to allow evaluation of genome editing in preimplantation embryos, or transferred at the 2-cell stage to pseudopregnant recipients so that mutations could be characterised in offspring.
Genomic DNA analysis
For standard genotyping, mouse ear-punch tissue samples were digested at 55°C for 3 h in 25–100 μl of a lysis buffer containing 10% (w/v) sodium dodecyl sulphate and with 2 mg/ml proteinase K (Sigma). 1 μl of a 1:10 dilution of each sample was used for genotyping by PCR in a 20 μl reaction volume.
For genomic qPCR, crude lysates were diluted 50-fold and 5 μl mixed with 20 μl SYBR® Green PCR Master Mix (Life Technologies). qPCR was performed on an ABI7500 Real Time PCR machine (Applied Biosystems) with the following parameters: 95°C, 10 sec (once); 95°C, 5 sec; 62°C, 31 sec (40 cycles); 72°C, 35 sec (once). DNA from the Nanog-eGFP line served as a single eGFP copy reference. Relative expression of EGFP was calculated using the ΔCt between eGFP and H3f3a. Fold change was calculated as E = 2-ΔΔCt. Sequences of PCR primers used here are given in Supplementary Table S1.
Whole genome amplification of single oocytes 3 h post-ICSI was performed with a GenomePlex® Single Cell Whole Genome Amplification kit (Sigma) in accordance with the supplied instructions. In brief, mII oocytes from superovulated B6D2F1 females were injected with sperm from either 129-GFP homozygous or hemizygous 8~12-week-old males. Injected cells were singly transferred to a 0.2 ml PCR tube in a minimal volume 3 h after ICSI and flash-frozen until processing shortly after. Frozen samples were adjusted to 9 μl with nuclease-free water and single-cell lysis and DNA fragmentation were performed by heating to 50°C for 1 h followed by 99°C for 4 min in the presence of 1 μl Proteinase K (0.5 mg/ml) in the Single Cell Lysis & Fragmentation Buffer provided. Library preparation was performed on fragmented DNA samples using the solution provided and incubated in a thermal cycler with the following parameters (one cycle): 16°C, 20 min; 24°C, 20 min; 37°C, 20 min; 75°C, 5 min; hold at 4°C. Genome amplification was performed with the addition of 7.5 μl Amplification Master Mix and 5.0 μl WGA DNA polymerase in a thermal cycler with the following parameters immediately after a single denaturing step (95°C, 3 min): 94°C, 30 sec; 65°C, 5 min (25 cycles); hold at 4°C. Using WGA samples as input DNA, eGFP sequences were amplified by PCR (Supplementary Table S1) and amplimers purified from 1.4% (w/v) agarose gels using the Wizard® SV Gel and PCR Clean-Up System (Promega) and supplied for sequencing with mixed read sequences (Source BioScience).
Statistical analysis
Statistical differences between pairs of data sets were analyzed by a chi-squared test.
Author Contributions
A.C.F.P. conceived the experiments with additional imput from T.S. and M.A. Micromanipulation was performed by T.S. and molecular analyses by M.A. Data analysis was by T.S., M.A. and A.C.F.P. A.C.F.P. wrote the manuscript with suggestions from T.S. and M.A.
Supplementary Material
Supplementary information
Acknowledgments
The authors thank Animal Facility support staff for ensuring the welfare of animals used in this work. We are grateful to Drs M. Leeb, M. VerMilyea, M. Furutani-Seiki, J. Correia and Mr D. Sharma and acknowledge Project Grant support from the Medical Research Council, UK (G1000839) and an EU Reintegration Grant (PIRG06-GA-2009-256408) to A.C.F.P.
References
- Cui X. et al. Targeted integration in rat and mouse embryos with zinc-finger nucleases. Nat. Biotechnol. 29, 64–67 (2011). [DOI] [PubMed] [Google Scholar]
- Sung Y. et al. Knockout mice created by TALEN-mediated gene targeting. Nat. Biotechnol. 31, 23–24 (2013). [DOI] [PubMed] [Google Scholar]
- Porteus M. H. & Baltimore D. Chimeric nucleases stimulate gene targeting in human cells. Science 300, 763 (2003). [DOI] [PubMed] [Google Scholar]
- Urnov F. D. et al. Highly efficient endogenous human gene correction using designed zinc-finger nucleases. Nature 435, 646-651 (2005). [DOI] [PubMed] [Google Scholar]
- Meyer M., de Angelis M. H., Wurst W. & Kühn R. (2010).Gene targeting by homologous recombination in mouse zygotes mediated by zinc-finger nucleases. Proc. Natl. Acad. Sci. USA 107, 15022–15026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mali P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu P. D., Lander E. S. & Zhang F. Development and applications of CRISPR-Cas9 for genome engineering. Cell 157, 1262–1278 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cong L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sander J. D. & Joung J. K. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat. Biotechnol. 32, 347–355 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H. et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell 153, 910–918 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen B. et al. Generation of gene-modified mice via Cas9/RNA-mediated gene targeting. Cell Res. 23, 720–723 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii W., Kawasaki K., Sugiura K. & Naito K. Efficient generation of large-scale genome-modified mice using gRNA and CAS9 endonuclease. Nucleic Acids Res. 41, e187 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H. et al. One-step generation of mice carrying reporter and conditional alleles by CRISPR/Cas-mediated genome engineering. Cell 154, 1370–1379 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou J. et al. One-step generation of different immunodeficient mice with multiple gene modifications by CRISPR/Cas9 mediated genome engineering. Int. J. Biochem. Cell Biol. 46, 49–55 (2014). [DOI] [PubMed] [Google Scholar]
- Yanagimachi R. Mammalian fertilization. In: The Physiology of Reproduction (E. Knobil & J.D.Neill, eds), 2nd edition, pp. 189–317. Raven Press, New York (1994). [Google Scholar]
- McLay D. W. & Clarke H. J. Remodelling the paternal chromatin at fertilization in mammals. Reproduction 125, 625–633 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida N., Brahmajosyula M., Shoji S., Amanai M. & Perry A. C. F. Epigenetic discrimination by mouse metaphase II oocytes mediates asymmetric chromatin remodeling independently of meiotic exit. Dev. Biol. 301, 464–477 (2007). [DOI] [PubMed] [Google Scholar]
- Perry A. C. F. et al. Mammalian transgenesis by intracytoplasmic sperm injection. Science 284, 1180–1183 (1999). [DOI] [PubMed] [Google Scholar]
- Perry A. C. F. et al. Efficient metaphase II transgenesis with different transgene archetypes. Nature Biotechnol. 19, 1071–1073 (2001). [DOI] [PubMed] [Google Scholar]
- Perry A. C. F. Hijacking oocyte DNA repair machinery in transgenesis? Mol. Reprod. Dev. 56, 319–324 (2000). [DOI] [PubMed] [Google Scholar]
- Suzuki T., Yoshida N., Suzuki E., Okuda E. & Perry A. C. F. Full-term mouse development by abolishing Zn2+-dependent metaphase II arrest without Ca2+ release. Development 137, 2659–2669 (2010). [DOI] [PubMed] [Google Scholar]
- Suzuki T. et al. Mouse Emi2 as a distinctive regulatory hub in second meiotic metaphase. Development 137, 3281–3291 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aoki F. & Schultz R. M. DNA replication in the 1-cell mouse embryo: stimulatory effect of histone acetylation. Zygote 7, 165–172 (1999). [DOI] [PubMed] [Google Scholar]
- Mizuno S. et al. Simple generation of albino C57BL/6J mice with G291T mutation in the tyrosinase gene by the CRISPR/Cas9 system. Mamm. Genome 25, 327–334 (2014). [DOI] [PubMed] [Google Scholar]
- Yen S. T. et al. Somatic mosaicism and allele complexity induced by CRISPR/Cas9 RNA injections in mouse zygotes. Dev. Biol. 393, 3–9 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cronin C. A., Ryan A. B., Talley E. M. & Scrable H. Tyrosinase expression during neuroblast divisions affects later pathfinding by retinal ganglion cells. J. Neurosci. 23, 11692–11697 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perry A. C. F. & Verlhac M.-H. Second meiotic arrest and exit in frogs and mice. EMBO Rep. 9, 246–251 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mali P. et al. CAS9 transcriptional activators for target specificity screening and paired nickases for cooperative genome engineering. Nat. Biotechnol. 31, 833–838 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lois C., Hong E. J., Pease S., Brown E. J. & Baltimore D. Germline transmission and tissue-specific expression of transgenes delivered by lentiviral vectors. Science 295, 868–872 (2002). [DOI] [PubMed] [Google Scholar]
- Gennequin B., Otte D. M. & Zimmer A. CRISPR/Cas-induced double-strand breaks boost the frequency of gene replacements for humanizing the mouse Cnr2 gene. Biochem. Biophys. Res. Commun. 441, 815–819 (2013). [DOI] [PubMed] [Google Scholar]
- Robl J. M., Wang Z., Kasinathan P. & Kuroiwa Y. Transgenic animal production and animal biotechnology. Theriogenology 67, 127–133 (2007). [DOI] [PubMed] [Google Scholar]
- Onishi A. & Perry A. C. F. “Livestock Production via Micromanipulation” pp 371-375 in ‘Practical Manual of In Vitro Fertilization'. Nagy, Z.P., Varghese, A.C. and Agarwal, A. (Eds.) Springer, US (2012).
- Mansour R. et al. International Committee for Monitoring Assisted Reproductive Technologies world report: Assisted Reproductive Technology 2006. Hum. Reprod. 29, 1536–1551 (2014). [DOI] [PubMed] [Google Scholar]
- Yoshida N. & Perry A. C. F. Piezo-actuated mouse intracytoplasmic sperm injection (ICSI). Nature Protoc. 2, 296–304 (2007). [DOI] [PubMed] [Google Scholar]
- Erbach G. T., Lawitts J. A., Papaioannou V. E. & Biggers J. D. Differential growth of the mouse preimplantation embryo in chemically defined media. Biol. Reprod. 50, 1027–1033 (1994). [DOI] [PubMed] [Google Scholar]
- Jao L. E., Wente S. R. & Chen W. Efficient multiplex biallelic zebrafish genome editing using a CRISPR nuclease system. Proc. Natl. Acad. Sci. USA 110, 13904–13909 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary information