Abstract
Background:
Neuromuscular Electrical Stimulation is a common intervention to address muscle weakness, however presents with many limitations such as fatigue, muscle damage, and patient discomfort that may influence its effectiveness. One novel form of electrical stimulation purported to improve neuromuscular re‐education is Patterned Electrical Neuromuscular Stimulation (PENS), which is proposed to mimic muscle‐firing patterns of healthy individuals. PENS provides patterned stimulating to the agonist muscle, antagonist muscle and then agonist muscle again in an effort to replicate firing patterns.
Purpose:
The purpose of this study was to determine the effect of a single PENS treatment on knee extension torque and quadriceps activation in individuals with quadriceps inhibition.
Methods:
18 subjects (10 males and 8 females: 24.2±3.4 years, 175.3±11.8cm, 81.8±12.4kg) with a history of knee injury/pain participated in this double‐blinded randomized controlled laboratory trial. Participants demonstrated quadriceps inhibition with a central activation ratio of ≤90%. Maximal voluntary isometric contraction of the quadriceps and central activation ratio were measured before and after treatment. The treatment intervention was a 15‐minute patterned electrical stimulation applied to the quadriceps and hamstring muscles with a strong motor contraction or a sham group, who received an identical set up as the PENS group, but received a 1mA subsensory stimulation. A 2×2 (group × time) ANCOVA was used to determine differences in maximal voluntary isometric contraction and central activation ratio between groups. The maximal voluntary isometric contraction was selected as a covariate due to baseline differences.
Results:
There were no differences in change scores between pre‐ and post‐intervention for maximal voluntary isometric contraction: (PENS: 0.09±0.32Nm/kg and Sham 0.15±0.18Nm/kg, p=0.713), or central activation ratio:(PENS: ‐1.22±6.06 and Sham: 1.48±3.7, p=0.270).
Conclusions:
A single Patterned Electrical Neuromuscular Stimulation treatment did not alter quadriceps central activation ratio or maximal voluntary isometric contraction. Unlike other types of muscle stimulation, PENS did not result in a reduction of quadriceps torque.
Level of Evidence:
Level III
Keywords: Electrical stimulation, muscle inhibition, quadriceps torque
INTRODUCTION
Quadriceps weakness and dysfunction are common repercussions following knee injury and surgery.1‐3 It is imperative for clinicians to restore quadriceps strength in order to retain normal knee joint function and encourage normal loading of the joint surfaces.4 Common practice aimed at improving quadriceps strength occurs during the rehabilitation process with use of isolated, functional, and sports specific exercises intended to hypertrophy the muscle. However, despite rehabilitation efforts, many individuals present with a reflexive response that results in prolonged inability to fully contract their quadriceps muscle, termed arthrogenic muscle inhibition (AMI).1,3 Quadriceps inhibition is theorized to contribute to ongoing muscle weakness, which ultimately can affect loading of the joint and researchers have identified consequences of altered gait kinematics,5 loss of dynamic knee stability,1 and increased propensity for developing tibiofemoral osteoarthritis (OA).6
Numerous interventions have been proposed to counter persistent muscle weakness following knee joint injury or surgery.2,7 An external electrical stimulus is applied focally over the joint or on the muscles with the goal of altering the motoneuron pool excitability. One common method to improve muscle strength is through the use of neuromuscular electrical stimulation (NMES).1,8 NMES is an electrically induced muscular contraction that is thought to bypass the reflexive inhibitory loop.9 The application of NMES can decrease atrophy,8 re‐educate the quadriceps muscle,10 and assist in regaining strength.9‐11 Several studies of both healthy subjects and subjects with pathology have supported NMES use in conjunction with volitional exercise over exercise alone.9‐11 NMES typically requires a stimulus that provides at least 60% of the maximal volitional isometric contraction (MVIC), in order to see strength improvements, and strength gains are linearly related to stimulus intensity.10 However, applying an electrically induced muscular contraction at such a high intensity can cause significant discomfort during the treatment and often results in fatigue.12 These limitations are not the only negative suggested implications of NMES, as other authors have proposed muscle damage,13 incomplete recruitment,12,14 and minimal functional activation.11
Patterned Electrical Neuromuscular Stimulation (PENS) is a novel application of electrical stimulation (ES) that can be used for neuromuscular re‐education and performance enhancement.15 PENS differs from NMES by attempting to replicate the typical firing sequence of muscle groups based on EMG patterns derived from healthy individuals during functional movement or activity.15,16 The stimulation pattern of a PENS treatment is delivered in a pattern that results in a contraction of the quadriceps muscle for 200ms, the hamstring muscle for 200ms and the quadriceps again for 120ms to mimic a voluntary movement pattern.16 This rhythmical pattern has been proposed to improve the neural drive by stimulating muscle stretch receptors and sensory neurons in both flexor and extensor motorneurons that have been found to replicate spinal alterations that are seen during locomotion.17 The PENS waveform is an asymmetrical biphasic square wave with a short phase duration (70μsec) and a 200‐millisecond stimulus train and a pulse rate of 50Hz. PENS also overcomes the limitation of functional use of stimulation seen with traditional NMES where a stimulus is delivered for 10 seconds with a 50 second rest period, which is not functionally applicable for activities besides early rehabilitation. PENS delivers the stimulus pattern continuously during the treatment session without requiring any off time.
PENS application has been used for performance enhancement with healthy individuals and has had positive results in an OA population.15,18 It has been hypothesized that motor patterned ES can improve contractility and muscle strength.19 Furthermore, because sub‐maximal contractions are elicited, less fatigue has been proposed with the PENS treatment, however these have not been examined following an immediate ES treatment. The purpose of this study was to determine the effect of a single PENS treatment on knee extension torque and quadriceps activation in individuals with quadriceps inhibition.
METHODS
Design
This study was a double‐blinded, randomized, sham controlled laboratory study. The dependent variables were quadriceps activation assessed by the central activation ratio (CAR) and peak knee extension torque measured by MVIC and normalized to body mass. The independent variables included treatment groups (PENS and Sham) and time (Pre‐ and post‐intervention). A blinded investigator measured outcomes during this study. An outside researcher conducted simple randomization from a random number generator before participant enrollment to the study.
Subjects
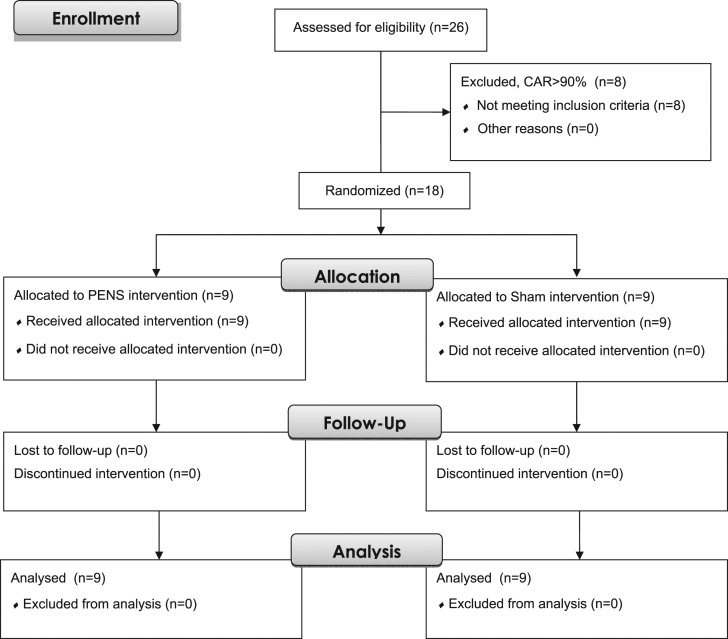
Eighteen participants (10 males and 8 females: age 24.2±3.4 years, height 175.3±11.8cm, mass 81.8±12.4kg) with a variety of knee pathologies were enrolled in the study (Table 1). Subjects were included if they self‐reported a previous diagnosed knee injury or surgery, were between the ages of 18 and 40 and presented with quadriceps inhibition measured by a CAR of less than 90% during the initial screening. Subjects were excluded if they presented with a knee surgery or injury within the prior six months, lower extremity neuropathy, or the presence of a tendinopathy. Additionally, participants with cardiac history, muscular abnormality, active infection over the site of the electrode placements (thigh), or those who were hypersensitive to electrical stimulation or pregnant were excluded. (Figure 1) All participants were not currently participating in supervised rehabilitation. The study was approved by the University of Virginia Institutional Review Board and all participants provided informed written consent prior to enrollment.
Table 1.
Means and Standard Deviations of Selected Demographic and Physical Characteristics of Sample by Musculoskeletal Injury Status.
| Demographics | PENS (n = 9) | Sham (n = 9) | p‐value |
|---|---|---|---|
| Sex | 5 M; 4 F | 5 M; 4 F | 1.0 |
| Age, yrs | 22.9 ± 2.2 | 25.5 ± 4.0 | 0.11 |
| Height, cm | 175.3 ± 12.6 | 175.3 ± 11.7 | 1.0 |
| Mass. kg | 80.9 ± 16.2 | 82.6 ± 7.9 | 0.79 |
| IKDC | 92.1 ± 6.9 | 83.9 ± 9.7 | 0.06 |
| KOOS | 93.8 ± 4.8 | 89.3 ± 10.8 | 0.27 |
| ACLr | 4 | 6 | |
| Anterior Knee Pain | 4 | 2 | |
| Meniscus Tear | 1 | 1 |
IKDC= International Knee Documentation Committee Score; KOOS= Knee Injury and Osteoarthritis Outcome Score; ACLr= Anterior Cruciate Ligament Reconstruction
Figure 1.
CONSORT Flow Diagram
Procedures
Participants reported to the laboratory where subjective demographics were collected using general health history questionnaires. Participants reported pain levels using the Visual Analog Scale (VAS), and completed two functional scales, the International Knee Documentation Committee (IKDC) and the Knee Injury and Osteoarthritis Outcome Score (KOOS). Upon completion of the questionnaires, participants were screened for CAR of the involved leg. The involved leg was the limb that participants self‐reported a previous history of knee injury or surgery. This limb was assessed throughout the entire testing session. If participants had a history of bilateral injuries or surgery, the limb they identified as more problematic during functional activities was utilized for data collection.
Knee extension torque was measured using the Biodex Multimodal Dynamometer (System 3, Biodex Medical Systems, Inc, Shirley, NY). A remote access port digitized at 125 Hz served as the analog to digital converter, 16 bits, was used to export data (MP150, Biopac Systems, Inc, Santa Barbara, CA) and record knee extension torque output.
Two 3” × 5” self‐adhesive electrodes were placed over the area of greatest bulk of the vastus medialis located superomedial to the patella and over the proximal‐lateral quadriceps in the area of the vastus lateralis muscle.7,20 Participants were seated in the dynamometer chair with their involved hip and knee flexed to 85°and 90°, respectively. The axis of rotation was aligned with the lateral femoral condyle and the participants were asked to maintain this position with their arms folded across their chest during the procedure. Participants were secured to the chair with a lap belt and Velcro straps proximal to the ankle joint to decrease accessory movement.21
MVIC Assessment: Gradual submaximal isometric contractions were performed to allow participants to become accustomed to the dynamometer and to perform a graded warm up. Participants were instructed to extend their knee until they were producing approximately 25% of their full effort. Submaximal isometric contractions were increasing by increments of 25% until reaching full isometric contraction (25%, 50%, 75%, and 100%).21 Three maximal trials were collected while providing continuous verbal encouragement, each separated by 60 seconds of rest.
CAR Assessment: Once the participant was comfortable with the MVIC task, the individual performed an MVIC with submaximal stimuli, which was also progressed in 25% increments until a maximal 125V stimulus was administered to the quadriceps muscle. Quadriceps CAR was measured through the Superimposed Burst (SIB) technique using a Grass S88 Stimulator and Stimulus Isolation Unit (Grass‐Telefactor, West Warwick, RI). The Isolation unit delivered a 100‐millisecond train of 10 square‐wave pulses at an intensity of 125V with a pulse duration of 600μs at a frequency of 100 pulses per second. During each MVIC trial, the researcher manually administered the 125V electrical stimulus when a steady plateau was visible during real‐time quadriceps torque output from a near computer. Those participants who had an average CAR under 90% (established a priori) were randomized to the intervention groups.7 (Table 1)
PENS was administered with an Omnistim® FX2 Pro Electrotherapy system (Accelerated Care Plus, Reno, NV) to the treatment group's involved limb. According to the manufacturer's guidelines, the treatment was delivered in a stimulation pattern to the agonist (quadriceps), the antagonist (hamstrings) and then the agonist (quadriceps) muscle groups. 15 (Figure 2) The intensity of the stimulation was delivered at a strong, comfortable contraction where a motor response can be seen in an alternating pattern between the quadriceps and hamstring muscle groups for 15‐minutes while the participant was seated on a plinth.
Figure 2.
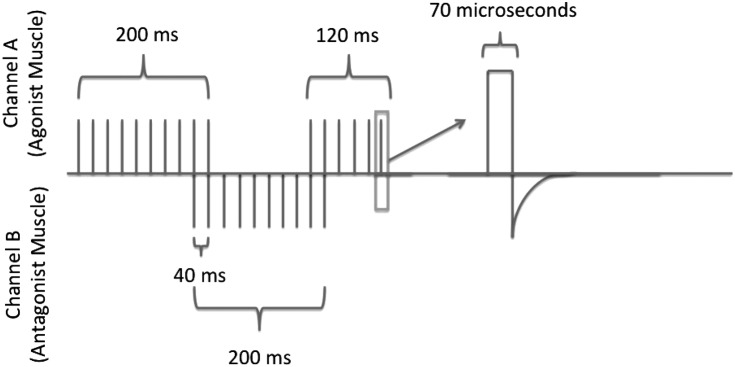
Patterned Electrical Neuromuscular Stimulation Pattern
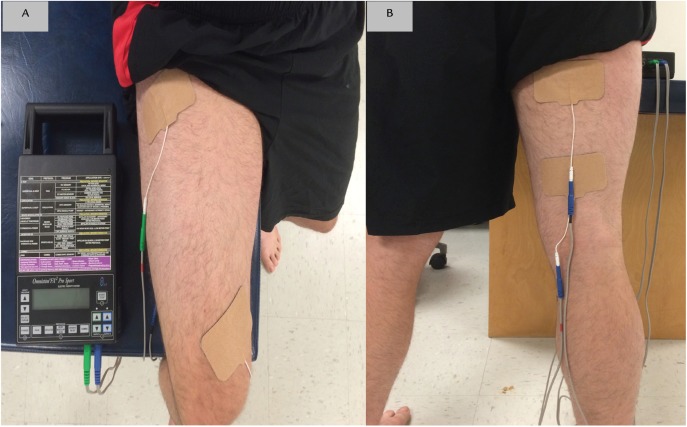
The individuals enrolled in the study were randomly assigned via concealed numbered envelope allocation to either the PENS treatment, or a sham treatment. The blinded clinician provided treatment that was enclosed in the envelops in the numerical order that was established during the randomization process. Two channels were used to deliver an alternating stimulus pattern between the agonist and antagonist muscles of the involved limb. Channel A consisted of two 2”×4” pre‐gelled self‐ adhesive stimulating electrodes placed on the anterior thigh, one over the proximal vastus laterals and one placed over the distal vastus medialis obliquus. Channel B also consisted of two 2”x4” pre‐gelled self‐ adhesive stimulating electrodes placed on the posterior thigh, one was positioned on the distal hamstring and the second was placed over the proximal hamstring. (Figure 3) Participants who received the PENS group received a 15‐minute lower extremity treatment that involved increasing the amplitude until a strong motor contraction in the participant's leg could be seen. Channel A and B were increased separately until strong motor contractions could be seen in both the quadriceps and hamstring muscles. Participants were told they would be receiving sensory treatment that might produce a muscle contraction.
Figure 3.
PENS electrode placement
The Sham group was set up in the same manner as the PENS group, however participants were told they were receiving a stimulus that was a “sub‐sensory” treatment. The sham treatment was applied by increasing the amplitude to 1mA, the minimal stimulus the system could deliver, yet not great enough to elicit a sensory or motor effect. This minimal stimulus was needed in order to light the unit display and to activate the timer. The treatment duration was also 15‐minutes and participants were informed to notify the researcher if anything was felt during the treatment.
Following the intervention, PENS electrodes were removed from the participant's limb and two 3” × 5” electrodes to assess the quadriceps CAR were replaced on the quadriceps muscle of the involved limb. Identical methods were performed from the screening process for three additional trials for both MVIC and CAR for all participants. A post intervention VAS was re‐administered immediately following the post‐intervention testing, which concluded the study.
Data Analysis
The MVIC was normalized to the participant's body mass and was reported in Nm/Kg. The CAR was calculated by using both the MVIC and the increased torque produced due to the superimposed burst (SIB) from the electrical stimulus. The MVIC was divided by the MVIC torque plus the SIB to calculate the activation ratio of what the participant is able to generate maximally compared to the potential of the quadriceps muscle torque production 22 using the following equation:
Statistical Analysis
A 2×2 (Group × Time) ANCOVA was used to evaluate the differences between MVIC and CAR pre‐ and post‐ intervention. The MVIC was selected as a covariate due to differences that existed between groups at baseline. Dependent t‐tests were used for VAS scores pre‐ and post‐intervention. The alpha level was set a priori at 0.05. Statistical analyses were performed using SPSS (version 20; SPSS Inc., Chicago, IL). Standardized effect sizes were estimated with Cohen's d using a pooled standard deviation with 95% confidence intervals (CI).23
Sample size was calculated a priori, based off the research of Pietrosimone et al. which looked at improving CAR measures in individuals with tibiofemoral OA.7 The study had a variability of 0.13 at baseline and expected mean change of 0.15 in CAR following intervention.7 Those expected mean change and variability values along with an alpha level of 0.05 and 1‐beta level of 0.8 indicated the need for a sample size of 9 per group for a total of 18 participants.
RESULTS
There were no statistical differences between groups for any pre‐intervention measure. There were no differences between pre‐ to post‐intervention change scores for MVIC: (PENS: 0.09±0.32Nm/kg, sham 0.15±0.18Nm/kg, p=0.713) or CAR:(PENS: ‐1.22±6.06, sham: 1.48±3.7, p=0.270). No between‐subject effect was seen in the MVIC (p=0.295) or CAR (p=0.138). There were no significant differences in VAS in either the PENS (Pre:0.22±0.66 Post:0.18±0.53, p=0.886) or the sham groups (Pre:0.47±0.88, Post:0.60±1.02, p=0.709).
Strong effect sizes were calculated in the MVIC (Cohen d = 0.72, 95% CI = ‐0.44, 1.89) and CAR (Cohen d= 0.93, 95% CI= ‐0.27, 2.12) when comparing the percentage change scores between the PENS group and the sham group. Post hoc power analysis was calculated for both MVIC (.174) and CAR (.311). With the effect sized values produced within this study, a sample size needed to see differences after treatment in both MVIC and CAR would be 32 and 20 per group.
DISCUSSION
The primary goal of this study was to examine the influence of a single 15‐minute PENS treatment on knee extension torque and quadriceps activation in individuals with quadriceps activation failure following a knee injury or surgery. The authors' hypothesized that a patterned stimulation would increase the knee extension torque and reduce the amount of activation failure. The results of this study suggest that a single PENS treatment did not alter knee extension torque or quadriceps activation when immediately assessed by MVIC or CAR.
Similar changes in the MVIC torque production were found in both PENS and sham groups, 3% and 2.8% respectfully. These results differ from other previous NMES studies that examined MVIC torque production following a single electrical stimulation treatment. The previous ES parameters utilized a frequency of 75Hz and duty cycle of 6.25‐seconds on, 20‐seconds off. 14,24 Knee extension torque was found to decrease by 20% following 30 electrically induced contractions.14 This is similar to results found when assessing plantar flexor torque production after stimulation, which was reduced by 9.4% following a single session of 30 electrically induced contractions.24 These are substantially greater changes in torque output as compared to those seen in the groups within the current study. Those authors hypothesized this decrease was due to peripheral fatigue, which occurs between the neuromuscular junction and actin‐myosin cross bridges which are responsible for force production.14,24
The NMES frequencies used in the cited NMES studies are greater than the 50Hz pulse rate in the PENS parameters, which may be a reason for the greater reduction in torque when compared to the current study. There is evidence that NMES parameters such as frequency and stimulus intensity are directly correlated to both torque output and fatigue rate.25 Lower frequency protocols have been found to allow participants to maintain a more constant torque output over time when compared to greater frequency protocols.26 However, the authors' are unsure of the extent of a difference that would occur in knee extensor torque production between 75 and 50Hz treatments.
It is of interest that 15‐minutes of consistent electrical stimulation did not create deficits in knee extension torque in the PENS group. Traditional NMES parameters have suggested to use greater on‐off cycles to prevent common limitations such as spatial recruitment of muscle fibers, muscle fatigue, and muscle damage, which have all been found to decrease maximal voluntary contraction.11 Duty cycle was one of the first NMES parameters to be examined during initial research on Russian stimulation experiments and supports current evidence that a decrease in torque production and fatigue occurs as the duty cycle increases from a 1:1 ratio to a 1:5 ratio.27,28 One explanation for this may be due to the patterned delivery of stimulus in the PENS compared to the tetanic contraction traditionally produced using NMES protocols. While the PENS treatment lasted for a 15‐minute continuous treatment, the stimulus train targeting the quadriceps muscle was alternating at 200 and 120 milliseconds repetitively during the treatment, yet the amplitude was not a tetanic contraction. Without a strong tetanus contraction, there may be less metabolic demands placed on the muscle fibers, which may influence the muscle's ability to maintain a constant torque production.
The stimulus pattern targeting both the agonist and antagonist muscle groups may also provide insight into the lack of a decrease in torque, as some research supports electrically activated co‐activation to improve strength gains.9 Researchers have begun to explore stimulation being applied to opposing muscle in order to reduce muscle damage, patient discomfort and improve strength gains.29,30 While there is limited research on PENS, previous research using NMES on both quadriceps and hamstring muscles has demonstrated a decrease in torque production by 4.4%.30 The concept of administering electrical stimulation to agonist and antagonist muscle groups arose from co‐contraction of the quadriceps and hamstring simultaneously following anterior cruciate ligament reconstruction in order to minimize anterior translation of the tibia and undue forces on the graft.31 While advancements in surgical technique have minimized this concern, use of electrical stimulation using opposing muscle groups has continued to gain interest due to a more functional knee movement.32,33 NMES can be administered to opposing muscle groups, however it is either during a simultaneous co‐contraction or the muscles are stimulated reciprocally.30,31 The proposed benefit to utilizing PENS is that the agonist and antagonist are stimulated in a precise pattern based on EMG patterns suggested to help re‐train impaired lower extremity function caused by neural components. Those neural components have been proposed to improve with PENS due to an efferent contraction of lower extremity muscles, which can improve voluntary movements.16,34 Electrical stimulation in a pattern to the agonist, antagonist, agonist muscles have produced promising results at reproducing quick movements in other extremities, but has yet to be studied
While the results of this study did not demonstrate a change in quadriceps MVIC or CAR in a subjects with knee pathology following a single PENS treatment there are several limitations. First while the population utilized in this study were all those with self‐reported pathology, there was no delimitation to the specific pathology for inclusion. Inclusion of multiple injuries may be responsible for variation between the two groups with regard to both MVIC and self reported deficits when assessed by the IKDC. Subjects with multiple pathologies were included with the premise that multiple pathologies may present with quadriceps inhibition following injury, however, studying a single pathology may have provided more insight on the potential benefits of this novel form of electrical stimulation. Second, the authors' only assessed knee extension torque and quadriceps activation failure, however examining fatigue with electromyography would add more insight into using a novel ES device. Third, only a single treatment of PENS was administered, and multiple treatments may produce a cumulative effect on the measures of interest. Previous researchers studying traditional NMES have indicated that in order to see improvements in strength, electrically produced muscular contractions should be able to reach 60% of the individuals MVIC. The current study did not assess the percentage of quadriceps force produced by PENS during the treatment. Finally, the applicability of the findings of this study may be decreased due to the relatively small sample size. A larger sample size (as indicated by the post‐experiment power calculations) would allow a greater chance to detect group differences.
Future research should compare PENS to traditional NMES for both effectiveness at improving strength gains in individuals with muscle weakness, as well as comparing differences in commonly reported side effects of NMES including fatigue, discomfort and muscle damage.
CONCLUSION
The use of a single treatment of PENS did not change quadriceps extension MVIC or central activation in subjects with self‐reported knee pathologies as compared to controls. While strengthening parameters were not seen within this study, typical decreases in quadriceps MVIC following electrical stimulation were also not present following a PENS treatment. Future research should examine the use of this form of electrical stimulation at greater amplitudes and within multiple treatments of PENS in order to examine long‐term improvement on both strength and activation.
Acknowledgments:
The authors would like to thank Aaron Bottinick, Luke Donovan, and Grant Norte for their assistance with administering the blinded interventions during this project.
REFERENCES
- 1.Rice DA McNair PJ Quadriceps arthrogenic muscle inhibition: Neural mechanisms and treatment perspectives. Semin Arthritis Rheum. 2010;40(3):250‐266. [DOI] [PubMed] [Google Scholar]
- 2.Hart JM Kuenze CM Pietrosimone BG Ingersoll CD Quadriceps function in anterior cruciate ligament‐deficient knees exercising with transcutaneous electrical nerve stimulation and cryotherapy: A randomized controlled study. Clin Rehabil. 2012;26(11):974‐981. [DOI] [PubMed] [Google Scholar]
- 3.Hopkins J Ingersoll C Arthrogenic muscle inhibition: A limiting factor in joint rehabilitation. Journal of Sport Rehabilitation. 2000;9:135‐159. [Google Scholar]
- 4.Lewek M Rudolph K Axe M Snyder‐Mackler L The effect of insufficient quadriceps strength on gait after anterior cruciate ligament reconstruction. Clin Biomech (Bristol, Avon). 2002;17(1):56‐63. [DOI] [PubMed] [Google Scholar]
- 5.Torry MR Decker MJ Viola RW O'Connor DD Steadman JR Intra‐articular knee joint effusion induces quadriceps avoidance gait patterns. Clin Biomech (Bristol, Avon). 2000;15(3):147‐159. [DOI] [PubMed] [Google Scholar]
- 6.Palmieri‐Smith RM Thomas AC Wojtys EM Maximizing quadriceps strength after ACL reconstruction. Clin Sports Med. 2008;27(3):405‐24, vii‐ix. [DOI] [PubMed] [Google Scholar]
- 7.Pietrosimone BG Hart JM Saliba SA Hertel J Ingersoll CD Immediate effects of transcutaneous electrical nerve stimulation and focal knee joint cooling on quadriceps activation. Med Sci Sports Exerc. 2009;41(6):1175‐1181. [DOI] [PubMed] [Google Scholar]
- 8.Bax L Staes F Verhagen A Does neuromuscular electrical stimulation strengthen the quadriceps femoris? A systematic review of randomised controlled trials. Sports Med. 2005;35(3):191‐212. [DOI] [PubMed] [Google Scholar]
- 9.Snyder‐Mackler L Ladin Z Schepsis AA Young JC Electrical stimulation of the thigh muscles after reconstruction of the anterior cruciate ligament. effects of electrically elicited contraction of the quadriceps femoris and hamstring muscles on gait and on strength of the thigh muscles. J Bone Joint Surg Am. 1991;73(7):1025‐1036. [PubMed] [Google Scholar]
- 10.Snyder‐Mackler L Delitto A Bailey SL Stralka SW Strength of the quadriceps femoris muscle and functional recovery after reconstruction of the anterior cruciate ligament. A prospective, randomized clinical trial of electrical stimulation. J Bone Joint Surg Am. 1995;77(8):1166‐1173. [DOI] [PubMed] [Google Scholar]
- 11.Maffiuletti NA Minetto MA Farina D Bottinelli R Electrical stimulation for neuromuscular testing and training: State‐of‐the art and unresolved issues. Eur J Appl Physiol. 2011;111(10):2391‐2397. [DOI] [PubMed] [Google Scholar]
- 12.Jubeau M Sartorio A Marinone PG et al. Comparison between voluntary and stimulated contractions of the quadriceps femoris for growth hormone response and muscle damage. J Appl Physiol. 2008;104(1):75‐81. [DOI] [PubMed] [Google Scholar]
- 13.Nosaka K Aldayel A Jubeau M Chen TC Muscle damage induced by electrical stimulation. Eur J Appl Physiol. 2011;111(10):2427‐2437. [DOI] [PubMed] [Google Scholar]
- 14.Zory R Boerio D Jubeau M Maffiuletti NA Central and peripheral fatigue of the knee extensor muscles induced by electromyostimulation. Int J Sports Med. 2005;26(10):847‐853. [DOI] [PubMed] [Google Scholar]
- 15.Gulick DT Castel JC Palermo FX Draper DO Effect of patterned electrical neuromuscular stimulation on vertical jump in collegiate athletes. Sports Health. 2011;3(2):152‐157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cooke JD Brown SH Movement‐related phasic muscle activation. II. generation and functional role of the triphasic pattern. J Neurophysiol. 1990;63(3):465‐472. [DOI] [PubMed] [Google Scholar]
- 17.McCrea DA Rybak IA Organization of mammalian locomotor rhythm and pattern generation. Brain Res Rev. 2008;57(1):134‐146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Burch F Tarro J Greenberg J Carroll W Evaluating the benefits of patterned stimulation in the treatment of osteoarthritis of the knee. Osteoarthritis Cartilage. 2008;16(8):865‐872. [DOI] [PubMed] [Google Scholar]
- 19.Sheffler LR Chae J Neuromuscular electrical stimulation in neurorehabilitation. Muscle Nerve. 2007;35(5):562‐590. [DOI] [PubMed] [Google Scholar]
- 20.Pietrosimone BG Selkow NM Ingersoll CD Hart JM Saliba SA Electrode type and placement configuration for quadriceps activation evaluation. J Athl Train. 2011;46(6):621‐628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Roberts D Kuenze C Saliba S Hart JM Accessory muscle activation during the superimposed burst technique. J Electromyogr Kinesiol. 2012;22(4):540‐545. [DOI] [PubMed] [Google Scholar]
- 22.Kent‐Braun JA Le Blanc R Quantitation of central activation failure during maximal voluntary contractions in humans. Muscle Nerve. 1996;19(7):861‐869. 2‐7. [DOI] [PubMed] [Google Scholar]
- 23.McGough JJ Faraone SV Estimating the size of treatment effects: Moving beyond p values. Psychiatry (Edgmont). 2009;6(10):21‐29. [PMC free article] [PubMed] [Google Scholar]
- 24.Boerio D Jubeau M Zory R Maffiuletti NA Central and peripheral fatigue after electrostimulation‐induced resistance exercise. Med Sci Sports Exerc. 2005;37(6):973‐978. [PubMed] [Google Scholar]
- 25.Binder‐Macleod SA McDermond LR Changes in the force‐frequency relationship of the human quadriceps femoris muscle following electrically and voluntarily induced fatigue. Phys Ther. 1992;72(2):95‐104. [DOI] [PubMed] [Google Scholar]
- 26.Gorgey AS Mahoney E Kendall T Dudley GA Effects of neuromuscular electrical stimulation parameters on specific tension. Eur J Appl Physiol. 2006;97(6):737‐744. [DOI] [PubMed] [Google Scholar]
- 27.Babkin D 1977. Notes from Y. M. Kots', PhD, (USSR) lectures and laboratory periods. Canadian‐Soviet exchange symposium on electrostimulation of skeletal muscles. [Google Scholar]
- 28.Ward AR Shkuratova N Russian electrical stimulation: The early experiments. Phys Ther. 2002;82(10):1019‐1030. [PubMed] [Google Scholar]
- 29.Yanagi T Shiba N Maeda T et al. Agonist contractions against electrically stimulated antagonists. Arch Phys Med Rehabil. 2003;84(6):843‐848. [DOI] [PubMed] [Google Scholar]
- 30.Vanderthommen M Triffaux M Demoulin C Crielaard JM Croisier JL Alteration of muscle function after electrical stimulation bout of knee extensors and flexors. J Sports Sci. 2012;11:592‐599. [PMC free article] [PubMed] [Google Scholar]
- 31.Delitto A Rose SJ McKowen JM Lehman RC Thomas JA Shively RA Electrical stimulation versus voluntary exercise in strengthening thigh musculature after anterior cruciate ligament surgery. Phys Ther. 1988;68(5):660‐663. [DOI] [PubMed] [Google Scholar]
- 32.Lynch CL Popovic MR A stochastic model of knee angle in response to electrical stimulation of the quadriceps and hamstrings muscles. Artif Organs. 2011;35(12):1169‐1174. [DOI] [PubMed] [Google Scholar]
- 33.del‐Ama AJ Gil‐Agudo A Pons JL Moreno JC Hybrid FES‐robot cooperative control of ambulatory gait rehabilitation exoskeleton. J Neuroeng Rehabil. 2014;11:27‐0003‐11‐27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gartner A Staiger V Neurotrophin secretion from hippocampal neurons evoked by long‐term‐potentiation‐inducing electrical stimulation patterns. Proc Natl Acad Sci U S A. 2002;99(9):6386‐6391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wierzbicka MM Wiegner AW Shahani BT Role of agonist and antagonist muscles in fast arm movements in man. Exp Brain Res. 1986;63(2):331‐340. [DOI] [PubMed] [Google Scholar]