Abstract
Cre/loxP recombination is a powerful strategy widely used for in vivo conditional gene targeting. This technique has made possible many important discoveries of gene function in normal and disease biology. However, due to the transgenic nature of most Cre mouse strains undesired phenotypes occasionally occur in Cre mice. Here we report skeletal defects in Osterix-Cre (Osx-Cre) transgenic mice including delayed calvarial ossification and fracture calluses at multiple skeletal sites. These data suggest that Osx-Cre containing controls should be used for both in vivo and in vitro skeletal analyses of conditional knockout mice generated with this Osx-Cre mouse strain.
Keywords: Osterix-Cre, Skeletal defects, Mouse, Transgenic
Introduction
Application of the Cre/loxP site-specific recombination system has been widely used for chromosome modification and gene targeting studies in mice for almost two decades. When recombination is induced in vivo, not only can chromosomal deletions, duplications, inversions or translocations be generated, but conditional gene modification in a cell type, tissue or developmental stage specific manner can also be achieved (Yu and Bradley 2001, Kuhn and Torres 2002). Therefore, the Cre/loxP system has been a powerful tool in studies aimed at understanding basic biological processes as well as human diseases associated with genetic abnormalities.
Currently, one frequent application of the Cre/loxP strategy is to generate conditional knockout (CKO) mice that require generation both of a mouse strain harboring a loxP-flanked region of a target gene (floxed) and of a second mouse strain expressing the Cre recombinase constitutively. The latter is often achieved by making a Cre transgenic mouse line, where most of the time the insertion of a Cre-containing construct in the mouse genome is random and unknown. As a result, it can occasionally disrupt or enhance the function of an endogenous gene, causing undesired phenotypes in transgenic Cre mice themselves (Palmiter and Brinster 1986). An example of such a mouse line is the Osterix-Cre (Osx-Cre) transgenic mouse (Rodda and McMahon 2006).
The Osx-Cre transgenic mice were originally generated by the McMahon group (Rodda and McMahon 2006) and are now available at the Jackson Laboratory. Osterix (Osx) is a zinc finger-containing transcription factor required for osteoblast differentiation and bone formation (Nakashima et al. 2002). It is expressed in both endochondral and membranous osteoblasts and functions downstream of Runx2, another master transcription factor essential for osteogenesis (Komori et al. 1997). Although both Runx2 null and Osx null mutant mice exhibit similar bone defects characterized by a lack of bone formation and an absence of differentiated osteoblasts, Osx appears to strictly control osteoblast differentiation while Runx2 has a much earlier and broader effect on invasion of mesenchymal cells, osteoclasts and blood vessels into hypertrophic cartilage matrix as well (Komori et al. 1997, Nakashima et al. 2002). Clearly, expression of these transcription factors represents different stages in mesenchymal cell differentiation resulting in different cell behavior and fates. Therefore, mice expressing the Cre transgene under the control of the Runx2 or Osx promoter have become powerful genetic tools for studying skeletogenesis as well as specific stages in bone development (Rauch et al. 2010, Rodda and McMahon 2006). Here we report that Osx-Cre transgenic mice exhibit unexpected skeletal phenotypes that could confound bone studies using these mice. We suggest that proper controls must be used for in vivo and in vitro analyses involving this Osx-Cre mouse line.
Materials and Methods
Animals
Osx-Cre transgenic mice were kindly provided by Dr. Andy McMahon at Harvard University (Rodda and McMahon 2006) and also obtained from the Jackson Laboratory. Mice harboring floxed alleles of Pkd1 were described previously (Starremans et al. 2008). C57BL/6 mice were purchased from Charles River.
Skeletal staining
After euthanasia, mice were skinned and eviscerated. Skeletal specimens were then incubated in a staining solution containing 0.006% Alizarin red S, 0.005% Alcian blue, 0.13% glacial acetic acid and 62% ethanol. Stained skeletal samples were further cleared in 1.8% and 0.3% potassium hydroxide solutions and stored in 100% glycerol. All animal work was conducted according to protocols approved by the Institutional Animal Care and Use Committee at Harvard Medical School.
Micro-CT Scanning
After euthanasia, skulls of P6 pups were skinned, separated from the trunk and subjected to high-resolution micro-CT scanning (Xradia MicroXCT-200 system, Pleasanton CA, USA). Images were reconstructed and generated by the Xradia software.
Results
Osx-Cre transgenic mice exhibit multiple bone defects
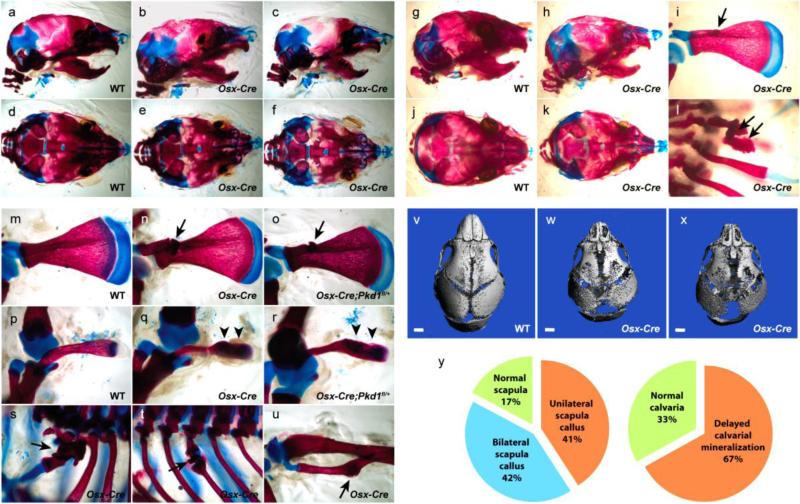
The Osx-Cre mice provided by Dr. Andy McMahon were crossed to C57BL/6 mice in our laboratory for three generations. Although these mice carry the tetracycline-controlled Tet-Off regulation of Osx-Cre, we did not administer any doxycycline to the mice throughout our examination in order to observe the effect of actively expressed Osx-Cre. To examine whether the Osx-Cre transgene alone affects normal bone development, we analyzed skeletons from newborn to one-week-old pups. Newborn Osx-Cre pups were indistinguishable from their wild type (WT) littermates. However, body weight comparison at P6 showed that Osx-Cre pups were significantly lighter than their WT littermates, weighing 3.60g and 4.13g on average, respectively (n=8 for WT, n=9 for Osx-Cre, P<0.01). Alcian blue and Alizarin red staining of skeletons revealed that Osx-Cre pups exhibited multiple skeletal defects including delayed calvarial and clavicular mineralization and frequent fracture callus spots on the scapula, ribs and fibula (Figure 1a-f, mu). To confirm that the delayed calvarial mineralization in Osx-Cre pups is not an artifact from the skeletal staining method, which can occasionally occur, micro-CT analysis was carried out on the calvaria of P6 mice. As shown in Figure 1v-x, Osx-Cre mice exhibited delayed and reduced calvarial mineralization at P6. To determine whether the observed skeletal defects in Osx-Cre mice were associated with the Osx-Cre transgene expression or due to normal variation among newborn littermates, we examined the frequency of skeletal defects in both Osx-Cre pups and their WT littermates. Over 83% (10 out of 12) P6 Osx-Cre pups had a scapula callus defect with 42% having bilateral calluses. Delayed calvarial mineralization was seen in 67% of Osx-Cre pups (8/12) (Figure 1y). In contrast, none of the 13 WT pups exhibited scapula calluses or obviously delayed calvarial ossification (Figure 1y). These results suggest that the observed bone defects are highly associated with the expression of the Osx-Cre transgene, although the phenotype is not fully penetrant.
Figure 1. Skeletal defects in Osx-Cre transgenic mice.
Alcian blue and Alizarin red skeletal staining shows delayed calvarial (b, c, e, f, h, k) and clavicular (q, r) mineralization in Osx-Cre pups compared to WT littermate controls (a, d, g, j, p). Fracture callus spots are frequently observed on the scapula (i, n, o), ribs (l, s, t) and fibula (u) in Osx-Cre but not in control pups (m, p)Micro-CT analysis of the skull further confirms delayed calvarial mineralization in Osx-Cre pups (v-x). The occurrence of scapula calluses and delayed calvarial mineralization in Osx-Cre transgenic mice has a high frequency of 83% and 67%, respectively (y). These skeletal defects are observed in Osx-Cre pups both on a mixed (a-f, m-u) and C57BL/6 background (g-l). Arrow: fracture callus; arrowhead: delayed clavicular mineralization.
Mouse genetic backgrounds can often affect the manifestation of a skeletal defect. To determine whether the bone phenotype in our Osx-Cre mice is affected by the mixed mouse strain background, we purchased the same line of Osx-Cre mice from the Jackson Laboratory where they had been backcrossed to C57BL/6 mice for at least 10 generations. At age P6, these Osx-Cre mice showed similar skeletal defects as described above (Figure 1g-l), including reduced body weight (WT: 3.43g n=21, Osx-Cre: 2.60g n=12, P<0.01), indicating that the skeletal defects observed in the Osx-Cre mice are not related to their mixed strain background.
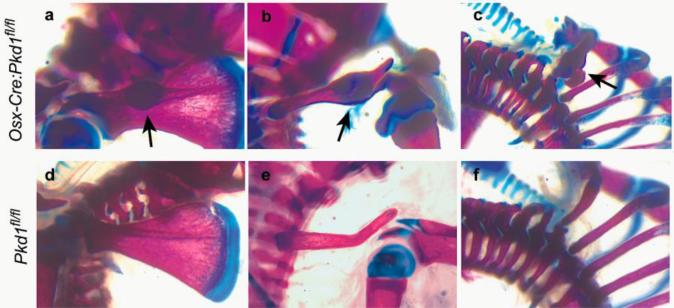
In studies using the Cre/loxP CKO model, mice with homozygously floxed target gene (fl/fl) are often chosen as controls for the CKO mice that would in addition express a Cre transgene (Ogata et al. 2011, Kamiya et al. 2008). To assess to what extent the bone defects in Osx-Cre mice could affect analysis of bone phenotypes in other CKO mice generated with this deleter strain, we examined the skeleton of polycystin 1 (Pkd1) CKO mice using Osx-Cre. Not surprisingly, homozygous Pkd1 CKO mice (Osx-Cre:Pkd1fl/fl) also had fracture calluses often on the rib, clavicle and scapula (Figure 2a-c) as well as delayed calvarial mineralization (data not shown) at early ages. As this phenotype was not seen in Pkd1fl/fl mice (Figure 2d-f), Pkd1fl/fl mice would not be the correct controls to use in these experiments. Instead, Osx-Cre or Osx-Cre:Pkd1fl/+ mice need to be used.
Figure 2. Skeletal defects in Osx-Cre:Pkd1fl/fl conditional knockout mice.
Alcian blue and Alizarin red skeletal staining shows frequent fracture calluses on the scapula (a), clavicle (b) and ribs (c) in P6 Osx-Cre:Pkd1fl/fl conditional knockout mice, but none in Pkd1fl/fl pups (d-f).
Discussion
Here we report that the Osx-Cre transgenic mice available from the Jackson Laboratory exhibit skeletal defects including delayed calvarial ossification and fracture healing calluses at multiple skeletal sites. Decreased body weight and delayed cortical bone development have been reported in young adults of these Osx-Cre mice (Davey et al. 2012). Modest increase in trabecular number and separation has also been shown after body weight correction in these mice (Davey et al. 2012). Taken together, the skeletal abnormalities in Osx-Cre mice appear to occur both in membranous and long bones.
Although the phenotype is mild, it raises the issue of proper controls when this Cre line is used for skeletal analyses. Several skeletal studies have been published using this Osx-Cre transgenic mouse strain (Greenblatt et al. 2010, Razidlo et al. 2010, Zhu et al. 2011). However, some of the studies describing skeletal abnormalities or defective cellular functions did not include Osx-Cre in their controls. Our data suggest that heterozygous knockouts (expressing both Osx-Cre and heterozygous floxed target gene) or Osx-Cre mice are necessary controls for Osx-Cre mediated CKO mice. Only when such controls are used, can the possibility that some of the observed skeletal abnormalities are due to expression of the Cre transgene rather than deletion of the target gene be excluded. Moreover, primary osteoblast isolation and culture are often carried out from calvariae in P0-P3 pups for in vitro analyses. As Osx-Cre pups clearly had delayed calvarial ossification in vivo, it is highly likely that primary osteoblasts isolated from Osx-Cre mice have impaired differentiation, proliferation or osteogenic function. Therefore, in vitro primary osteoblast analyses should also include cells from Osx-Cre mice to control for the effect of the Osx-Cre.
While it is clear that transgenic Osx-Cre mice display unexpected skeletal defects, the reason why they occur remains unknown. Recently, several studies have shown that in addition to osteoblasts, osteocytes and hypertrophic chondrocytes, Osx-Cre also targets many other cell types including stromal cells, adipocytes and perivascular cells in the bone marrow (Chen et al. 2014), brain cells (Park et al. 2011), and lung cells (our unpublished data). Whether any of these cell types, particularly bone marrow stromal cells and brain cells, contribute to the observed skeletal defects in Osx-Cre mice awaits further investigation. In addition to the non-skeletal tissue expression of Osx-Cre, it is also possible that these undesired phenotypes are caused by impaired osteoblast function due to insertional effects of the transgene. It has been estimated that 5% of established transgenic lines result in insertional mutation effects (Meisler 1992). Several possibilities may explain such effects. First, the site at which the Cre transgene inserts into the mouse genome could possibly disrupt the normal function of an endogenous gene. Second, deletion or duplication of large chromosomal DNA regions could occur during transgene integration (Palmiter and Brinster 1986). Third, prokaryotic vector sequences have been shown to be inhibitory for some mammalian genes and could have a disruptive effect (Townes et al. 1985). Fourth, the Osx promoter, GFP and Cre containing bacterial artificial chromosome used to generate this Osx-Cre line (Rodda and McMahon 2006) is a relatively large piece of foreign DNA that potentially could exert an interruptive effect.
In our study, Osx-Cre mice were backcrossed to C57BL/6 mice for either three (our laboratory) or more than ten generations (Jackson Laboratory). Although mouse genetic backgrounds play important roles in the formation of certain skeletal defects (Lee et al. 2010), it does not appear to affect the manifestation of skeletal defects in these Osx-Cre mice. Due to the various genetic backgrounds in which mice carrying floxed alleles of interest are often developed, CKO mice using Cre are also frequently generated with mixed backgrounds. Therefore, it is of great importance to be aware of undesired defects in a Cre deleter mouse line and properly control for such defects in the associated analyses.
Acknowledgements
We thank Nicholas Brady for micro CT analyses. We thank Naomi Fukai for help with technique-related work. We also thank Sofiya Plotkina for help with mouse genotyping. These studies were supported by grant AR36819 (to BRO) from the National Institutes of Health.
References
- Chen J, Shi Y, Regan J, Karuppaiah K, Ornitz DM, Long F. Osx-Cre targets multiple cell types besides osteoblast lineage in postnatal mice. PLoS One. 2014;9(1):e85161. doi: 10.1371/journal.pone.0085161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davey RA, Clarke MV, Sastra S, Skinner JP, Chiang C, Anderson PH, Zajac JD. Decreased body weight in young Osterix-Cre transgenic mice results in delayed cortical bone expansion and accrual. Transgenic Res. 2012;21(4):885–893. doi: 10.1007/s11248-011-9581-z. [DOI] [PubMed] [Google Scholar]
- Greenblatt MB, Shim JH, Zou W, Sitara D, Schweitzer M, Hu D, Lotinun S, Sano Y, Baron R, Park JM, Arthur S, Xie M, Schneider MD, Zhai B, Gygi S, Davis R, Glimcher LH. The p38 MAPK pathway is essential for skeletogenesis and bone homeostasis in mice. J Clin Invest. 2010;120(7):2457–2473. doi: 10.1172/JCI42285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamiya N, Ye L, Kobayashi T, Mochida Y, Yamauchi M, Kronenberg HM, Feng JQ, Mishina Y. BMP signaling negatively regulates bone mass through sclerostin by inhibiting the canonical Wnt pathway. Development. 2008;135(22):3801–3811. doi: 10.1242/dev.025825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komori T, Yagi H, Nomura S, Yamaguchi A, Sasaki K, Deguchi K, Shimizu Y, Bronson RT, Gao YH, Inada M, Sato M, Okamoto R, Kitamura Y, Yoshiki S, Kishimoto T. Targeted disruption of Cbfa1 results in a complete lack of bone formation owing to maturational arrest of osteoblasts. Cell. 1997;89(5):755–764. doi: 10.1016/s0092-8674(00)80258-5. [DOI] [PubMed] [Google Scholar]
- Kuhn R, Torres RM. Cre/loxP recombination system and gene targeting. Methods Mol Biol. 2002;180:175–204. doi: 10.1385/1-59259-178-7:175. [DOI] [PubMed] [Google Scholar]
- Lee GS, Liao X, Shimizu H, Collins MD. Genetic and pathologic aspects of retinoic acid-induced limb malformations in the mouse. Birth Defects Res A Clin Mol Teratol. 2010;88(10):863–882. doi: 10.1002/bdra.20712. [DOI] [PubMed] [Google Scholar]
- Meisler MH. Insertional mutation of 'classical' and novel genes in transgenic mice. Trends Genet. 1992;8(10):341–344. doi: 10.1016/0168-9525(92)90278-c. [DOI] [PubMed] [Google Scholar]
- Nakashima K, Zhou X, Kunkel G, Zhang Z, Deng JM, Behringer RR, de Crombrugghe B. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell. 2002;108(1):17–29. doi: 10.1016/s0092-8674(01)00622-5. [DOI] [PubMed] [Google Scholar]
- Ogata N, Shinoda Y, Wettschureck N, Offermanns S, Takeda S, Nakamura K, Segre GV, Chung UI, Kawaguchi H. G alpha(q) signal in osteoblasts is inhibitory to the osteoanabolic action of parathyroid hormone. J Biol Chem. 2011;286(15):13733–13740. doi: 10.1074/jbc.M110.200196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmiter RD, Brinster RL. Germ-line transformation of mice. Annu Rev Genet. 1986;20:465–499. doi: 10.1146/annurev.ge.20.120186.002341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JS, Baek WY, Kim YH, Kim JE. In vivo expression of Osterix in mature granule cells of adult mouse olfactory bulb. Biochem Biophys Res Commun. 2011;407(4):842–847. doi: 10.1016/j.bbrc.2011.03.129. [DOI] [PubMed] [Google Scholar]
- Rauch A, Seitz S, Baschant U, Schilling AF, Illing A, Stride B, Kirilov M, Mandic V, Takacz A, Schmidt-Ullrich R, Ostermay S, Schinke T, Spanbroek R, Zaiss MM, Angel PE, Lerner UH, David JP, Reichardt HM, Amling M, Schutz G, Tuckermann JP. Glucocorticoids suppress bone formation by attenuating osteoblast differentiation via the monomeric glucocorticoid receptor. Cell Metab. 2010;11(6):517–531. doi: 10.1016/j.cmet.2010.05.005. [DOI] [PubMed] [Google Scholar]
- Razidlo DF, Whitney TJ, Casper ME, McGee-Lawrence ME, Stensgard BA, Li X, Secreto FJ, Knutson SK, Hiebert SW, Westendorf JJ. Histone deacetylase 3 depletion in osteo/chondroprogenitor cells decreases bone density and increases marrow fat. PLoS One. 2010;5(7):e11492. doi: 10.1371/journal.pone.0011492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodda SJ, McMahon AP. Distinct roles for Hedgehog and canonical Wnt signaling in specification, differentiation and maintenance of osteoblast progenitors. Development. 2006;133(16):3231–3244. doi: 10.1242/dev.02480. [DOI] [PubMed] [Google Scholar]
- Starremans PG, Li X, Finnerty PE, Guo L, Takakura A, Neilson EG, Zhou J. A mouse model for polycystic kidney disease through a somatic in-frame deletion in the 5' end of Pkd1. Kidney Int. 2008;73(12):1394–1405. doi: 10.1038/ki.2008.111. [DOI] [PubMed] [Google Scholar]
- Townes TM, Lingrel JB, Chen HY, Brinster RL, Palmiter RD. Erythroid-specific expression of human beta-globin genes in transgenic mice. EMBO J. 1985;4(7):1715–1723. doi: 10.1002/j.1460-2075.1985.tb03841.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Y, Bradley A. Engineering chromosomal rearrangements in mice. Nat Rev Genet. 2001;2(10):780–790. doi: 10.1038/35093564. [DOI] [PubMed] [Google Scholar]
- Zhu W, Liang G, Huang Z, Doty SB, Boskey AL. Conditional inactivation of the CXCR4 receptor in osteoprecursors reduces postnatal bone formation due to impaired osteoblast development. J Biol Chem. 2011;286(30):26794–26805. doi: 10.1074/jbc.M111.250985. [DOI] [PMC free article] [PubMed] [Google Scholar]