Abstract
Purpose
The purpose of this study was to assess the prevalence of male infertility and treatment-related risk factors in childhood cancer survivors.
Methods
Within the Childhood Cancer Survivor Study, 1622 survivors and 274 siblings completed the Male Health Questionnaire. The analysis was restricted to survivors (938/1622; 57.8%) and siblings (174/274; 63.5%) who tried to become pregnant. Relative risks (RR) and 95% confidence intervals (CI) for the prevalence of self-reported infertility were calculated using generalized linear models for demographic variables and treatment-related factors to account for correlation among survivors and siblings of the same family. All statistical tests were two-sided.
Results
Among those who provided self-report data, the prevalence of infertility was 46.0% in survivors versus 17.5% in siblings (RR=2.64, 95% CI 1.88-3.70, p < 0.001). Of survivors who met the definition for infertility, 37% had reported at least one pregnancy with a female partner that resulted in a live birth. In a multivariable analysis, risk factors for infertility included an alkylating agent dose score (AAD) ≥ 3 (RR= 2.13, 95% CI 1.69-2.68 for AAD ≥ 3 versus AAD<3), surgical excision of any organ of the genital tract (RR=1.63, 95% CI 1.20-2.21), testicular radiation ≥ 4Gy (RR=1.99, 95% CI 1.52-2.61), and exposure to bleomycin (RR=1.55, 95% CI 1.20-2.01).
Conclusion
Many survivors who experience infertility father their own children suggesting episodes of both fertility and infertility. This and the novel association of infertility with bleomycin warrant further investigation.
Implications for Cancer Survivors
Though infertility is common, male survivors reporting infertility often father their own children. Bleomycin may pose some fertility risk.
Keywords: infertility, cancer, male, long-term survivors, pediatrics
Introduction
Survival from pediatric cancer has improved significantly over the last several decades with five-year survival rates increasing from 45 % in 1970 to over 80% in 2008[1]. However, survival may come at the cost of long-term side effects from cancer treatment such as infertility.
Many studies have reported on biologic aspects of fertility and reproductive outcomes in male survivors of childhood cancer [2-6]. A prior Childhood Cancer Survivor Study (CCSS) report compared fertility, defined as having fathered a pregnancy, in all male survivor and sibling participants [4]. Survivors were less likely than siblings (hazard ratio [HR] = 0.56; 95% confidence interval [CI] 0.49 to 0.63) to father a pregnancy. Exposure to testicular radiation of more than 7.5 Gy and a higher cumulative alkylating agent dose (AAD) score decreased the HR of fathering a pregnancy. To better understand the personal aspects of male reproductive health such as risk perception, sexual function, and individual choices about reproduction, the Male Health Questionnaire (MHQ) was administered to male CCSS survivors and siblings who agreed to participate and included detailed questions on these issues (Supplemental Figure 1). In this analysis of data collected with the MHQ we examine infertility in male survivors compared to their siblings. Infertility is different than fertility. The American Society of Reproductive Medicine (ASRM) defines “infertility” as “…the failure to achieve a successful pregnancy after 12 months or more of regular unprotected intercourse” (10). For this analysis we define infertility as the self-report of the inability of a female partner to conceive after 12 months of attempting to become pregnant by an adult male survivor of childhood cancer. The objective was to evaluate the prevalence of infertility and treatment-related risk factors which were hypothesized to be similar to exposures adversely impacting fertility in the prior analysis of males in the CCSS cohort. This study is unique in assessing infertility in males who may have fathered a pregnancy but had difficulty in doing so. Furthermore, the MHQ includes additional data on the desire and ability to have more children, the reasons for not having more children, and medical evaluation of infertility.
Materials and methods
The Childhood Cancer Survivor Study (CCSS)
The CCSS is a retrospective cohort consisting of 14,358 five-year survivors of childhood cancer ascertained from 26 participating institutions in the United States and Canada. Eligible participants were diagnosed between 1970 and 1986. The CCSS also includes 4,023 siblings of survivors who serve as the comparison group. Details of the study design and cohort have been previously published [7-9]. Data include self-report questionnaires and medical record abstraction of detailed treatment data from the treating institution as previously described [9]. The initial baseline questionnaire was administered beginning in 1994 and was updated periodically with follow-up questionnaires through 2009.
Male Heath Questionnaire (MHQ)
Survivor and sibling male participants 18 years of age or older at the time of the 2007 CCSS follow-up questionnaire, were asked if they would consider participating in a study “to better understand fertility and sexual function in males” by completing the MHQ. Of the 4,000 eligible male survivors and 1,097 male siblings who completed the 2007 follow-up questionnaire, 2,961 survivors and 723 siblings expressed potential interest in participating and the MHQ was mailed to them (Figure I). The MHQ was administered in 2008-2009 and included questions on pubertal development, sexual function/dysfunction, infertility, male health risk perceptions, testosterone therapy, erectile dysfunction, and use of fertility preservation techniques (questionnaires available at http://ccss.stjude.org/documents/original-cohort-questionnaires).
Figure 1. Participants in the Childhood Cancer Survivor Study Male Health Questionnaire and infertility analysis.
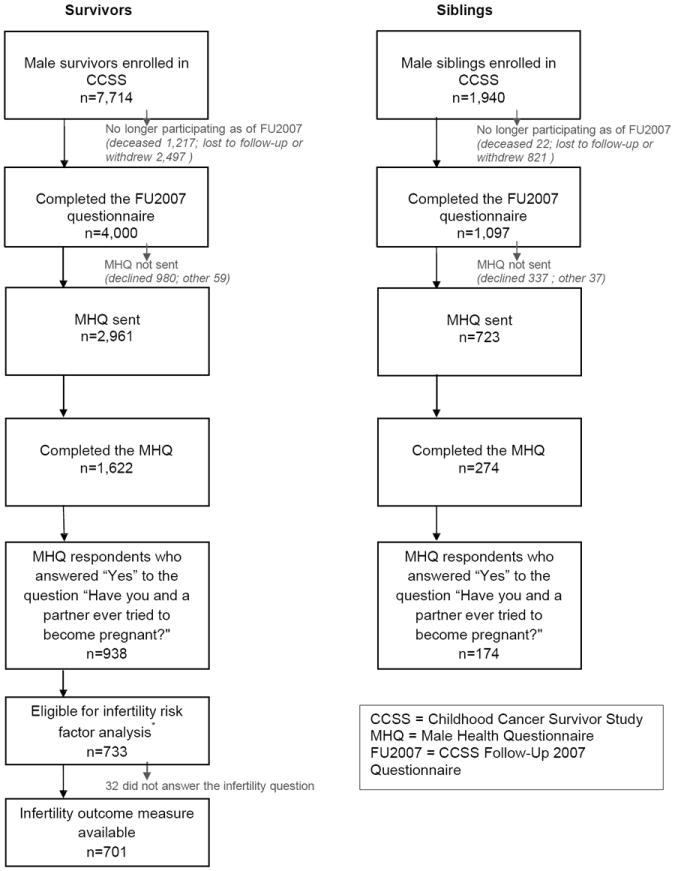
*Assessment of associations between infertility and cancer treatments received for primary cancer diagnosis was limited to subjects with no recurrence of the primary cancer and no secondary malignancy neoplasm.
This analysis focuses on infertility in those participants who answered affirmatively to the MHQ question, “Have you and a partner ever tried to become pregnant?”, thus excluding survivors and siblings who did not attempt to father a child. Infertility was defined as a positive response to the question, “Has a female partner ever had difficulty (it took more than a year) becoming pregnant by you?”
Exposure
Detailed data regarding chemotherapy and radiation therapy exposures, and all surgical procedures performed for the original cancer treatment within five years of diagnosis were obtained at baseline (abstraction form available at http://ccss.stjude.org/documents/original_cohort-questionnaire)[9]. Survivors with a history of a recurrence or secondary malignant neoplasm (SMN) were excluded from the assessment of cancer treatment variables as risk factors for infertility because, unlike the treatment data for the original cancer diagnosis obtained from the treating institution, treatment data for recurrences and SMNs were incomplete. Cumulative doses of specific chemotherapeutic agents, including alkylating agents, were abstracted from the medical records. The summed alkylating agent dose (AAD) score, which ranged from 0 (no alkylating agent exposure) to 7 in survivors responding to the MHQ, was used to evaluate risk associated with alkylating agent exposure [10, 11, 4]. To obtain this score, the distribution of cumulative doses for each of the alkylating agents was divided into tertiles and the AAD was calculated by adding the tertile score (1, 2, or 3) for each of the agents given to a particular patient. Other chemotherapeutic agents that were administered to at least 5% of the study population were evaluated as dichotomous variables. Radiation doses to the testes and hypothalamus/pituitary were estimated as previously described by Stovall et al. [12-14] Demographic characteristics, data on pregnancies and live births and current health status variables were obtained from CCSS baseline and follow-up questionnaires. Additional medical history variables relevant to men’s health were obtained from the MHQ.
Statistical analysis
Distributions of variables including demographics, cancer treatment, and infertility were summarized with frequencies and proportion estimates. Comparisons of proportions between independent groups used the chi-square test or, when appropriate due to low frequencies, Fisher’s exact test. Comparisons between survivors and siblings used generalized estimating equation modeling to account for potential within-family correlation [15].
In this study, infertility was a dichotomous (yes/no) outcome measure obtained via self-report on the MHQ. Risk factors for infertility were evaluated and relative risk (RR) estimates were calculated via general linear modeling. Since infertility was not rare in this study population, the odds ratio from a logistic model would not provide a good approximation of the relative risk parameter, so an alternative model with a log link function was used instead. [16]. All models accounted for age at the time of the MHQ by including age as an independent variable (categorical, 10 year increments). Individual factors relating to cancer treatment and demographic characteristics (see Table 3 for candidate variables) were first evaluated singly in age-adjusted models. Those factors that achieved a two-sided p-value of 0.20 or less were then considered candidates for further multivariable modeling. The final model was constructed by evaluating the candidate factors together in a multivariable context and then removing those factors that did not have an independent association with infertility. Factors were removed if they had a multivariable p-value above 0.20 and the omission of the factor from the model did not alter any other relative risk estimate by more than 10%. Cancer diagnosis was not used in the multivariable modeling because diagnosis is highly correlated with the cancer treatment variables. As mentioned above, since treatment data did not incorporate exposures received due to SMN or recurrence, models assessing treatment effects excluded patients who had an SMN or recurrence. All p-values are two-sided. P-values <0.05 were considered statistically significant.
Table 3. Infertility Prevalence by Demographic and Cancer Treatment Factors in Male Survivors of Childhood Cancer with No History of a Recurrence or Secondary Malignant Neoplasm.
| Characteristic | Number (percent) with infertility n=701 |
|---|---|
| Overall | 290 / 701 (41.4%) |
| DEMOGRAPHIC FACTORS | |
| Age at time of MHQ (Years) | |
| 20 to 29 | 12 / 50 (24.0%) |
| 30 to 39 | 142 / 337 (42.1%) |
| 40 to 49 | 121 / 282 (42.9%) |
| 50+ | 15 / 32 (46.9%) |
| Race/ethnicity group | |
| White (non-Hispanic) | 272 / 632 (43.0%) |
| Other | 10 / 31 (32.3%) |
| Unknown | 8 / 38 (21.1%) |
| Marital status | |
| Ever Married | 278 / 676 (41.1%) |
| Single (never married) | 11 / 23 (47.8%) |
| Unknown | 1 / 2 (50.0%) |
| Timing of Puberty Onset | |
| Early | 12 / 36 (33.3%) |
| Normal | 222 / 547 (40.6%) |
| Late | 52 / 113 (46.0%) |
| Unknown | 4 / 5 (80%) |
| CANCER CHARACTERISTICS AND CANCER TREATMENT FACTORS1 | |
| Age at diagnosis (Years) | |
| 0-4 | 69 / 191 (36.1%) |
| 5-9 | 63 / 163 (38.7%) |
| 10-14 | 91 / 186 (48.9%) |
| 15+ | 67 / 161 (41.6%) |
| Diagnosis group | |
| Acute lymphoblastic leukemia | 55 / 183 (30.1%) |
| AML and other leukemias | 3 / 10 (30.0%) |
| CNS tumors | 11 / 44 (25.0%) |
| Hodgkin lymphoma | 69 / 117 (59.0%) |
| Non-Hodgkin lymphoma | 36 / 102 (35.3%) |
| Kidney tumors | 15 / 49 (30.6%) |
| Neuroblastoma | 15 / 35 (42.9%) |
| Soft tissue sarcoma | 46 / 80 (57.5%) |
| Ewing sarcoma | 21 / 30 (70.0%) |
| Osteosarcoma | 17 / 48 (35.4%) |
| Other bone tumors | 2 / 3 (66.7%) |
| Testicular radiation dose | |
| No testicular radiation | 85 / 231 (36.8%) |
| <4 Gy | 166 / 397 (41.8%) |
| >=4 Gy | 19 / 23 (82.6%) |
| Unknown | 20 / 50 (40.0%) |
| Pituitary/hypothalamic radiation dose | |
| No pituitary/hypothalamic radiation | 85 / 231 (36.8%) |
| <20 Gy | 154 / 314 (49.0%) |
| 20-30 Gy | 21 / 81 (25.9%) |
| >=30 Gy | 10 / 23 (43.5%) |
| Unknown | 20 / 52 (38.5%) |
| TBI | |
| No | 264 / 640 (41.3%) |
| Yes | 2 / 2 (100%) |
| Unknown | 24 / 59 (40.7%) |
| Surgical excision of any organ of the genital tract22 | |
| No | 262 / 648 (40.4%) |
| Yes | 12 / 14 (85.7%) |
| Unknown | 16 / 39 (41.0%) |
| Surgical procedure on the spinal cord/canal or sympathetic nerves | |
| No | 269 / 648 (41.5%) |
| Yes | 5 / 14 (35.7%) |
| Unknown | 16 / 39 (41.0%) |
| AAD score | |
| 0 | 75 / 269 (27.9%) |
| 1 | 17 / 70 (24.3%) |
| 2 | 38 / 102 (37.3%) |
| 3 | 65 / 103 (63.1%) |
| 4 | 26 / 39 (66.7%) |
| 5+ | 27 / 37 (73.0%) |
| Unknown | 42 / 81 (51.9%) |
| Received bleomycin | |
| No | 241 / 611 (39.4%) |
| Yes | 33 / 51 (64.7%) |
| Unknown | 16 / 39 (41.0%) |
| Received methotrexate | |
| No | 163 / 339 (48.1%) |
| Yes | 111 / 323 (34.4%) |
| Unknown | 16 / 39 (41.0%) |
MHQ = Male Health Questionnaire; AML = acute myelogenous leukemia; CNS = central nervous system; TBI = total body irradiation; Gy = Gray; AAD = summed alkylating agent dose score
In addition to the cancer treatments summarized here, thirteen other chemotherapy agents received by >5% of patients in the analysis cohort were evaluated. Only those chemotherapy agents which appear in the final multivariable risk factor model are summarized in this table.
This surgery variable equals “Yes” if the subject’s cancer treatment records included one or more of the following procedures: orchiectomy, radical prostatectomy, total cystectomy or retroperitoneal dissection.
Ethical Approval
The study was reviewed and approved by the institutional review board at each institution. Written informed consent was received from all participants 18 years of age or older and from a parent or guardian of participants younger than 18 years.
Results
Participants and non-participants
Participants and non-participants in the MHQ were compared to determine the representativeness of the population (Table 1). The questionnaire was completed and returned by 1,622 survivors (54.8% of those who indicated they would be willing to complete the supplemental MHQ) and 274 siblings (37.9% of those siblings who indicated they would complete the MHQ) (Supplemental Figure I). Survivors who did complete the MHQ were slightly older at each time point than non-participants in the MHQ. Participants were more likely to be white, have married or lived as married, and have reported at least one partner pregnancy. Treatment characteristics were similar; however participants were more likely to have been survivors of Hodgkin lymphoma and less likely to have survived a central nervous system (CNS) neoplasm.
Table 1. Comparison of Participants and Non-Participants in the MHQ among Surviving Male CCSS Participants.
| Survivors | Siblings | |||
|---|---|---|---|---|
| Subject characteristic: | MHQ participants (n=1622) | MHQ non participants* (n=4875) | MHQ participants (n=274) | MHQ non participants† (n=1644) |
| Race/ethnicity | ||||
| White (non-Hispanic) | 1441 (93.4%) | 3967 (84.8%) | 256 (97.0%) | 1429 (90.8%) |
| Black (non-Hispanic) | 30 (1.9%) | 254 (5.4%) | 0 (0.0%) | 45 (2.9%) |
| Hispanic | 44 (2.9%) | 280 (6.0%) | 5 (1.9%) | 57 (3.6%) |
| Other | 27 (1.8%) | 177 (3.8%) | 3 (1.1%) | 42 (2.7%) |
| Ever married or lived as married (status as of FU2007) | ||||
| Yes | 1225 (75.9%) | 1411§ (60.5%) | 227 (82.8%) | 625 § (77.1%) |
| No | 389 (24.1%) | 923 § (39.5%) | 47 (17.2%) | 186 § (22.9%) |
| Partner pregnancies and live births reported on CCSS questionnaires (status as of FU2007)† | ||||
| 0 pregnancies reported | 882 (54.4%) | 1461§ (61.4%) | 96 (35.0%) | 292§ (35.5%) |
| ≥ 1 pregnancy reported, no live births reported‡ | 97 (6.0%) | 111§ (4.7%) | 15 (5.5%) | 49§ (6.0%) |
| ≥ 1 live birth reported | 641 (39.6%) | 808§ (33.9%) | 163 (59.5%) | 482§ (58.6%) |
| Age at start of MHQ distribution (Years) | ||||
| 20-29 | 307 (18.9%) | 1370 (28.1%) | 56 (20.5%) | 507 (30.9%) |
| 30-39 | 712 (43.9%) | 2206 (45.3%) | 104 (38.1%) | 589 (35.9%) |
| 40-49 | 525 (32.4%) | 1150 (23.6%) | 83 (30.4%) | 450 (27.4%) |
| 50+ | 78 (4.8%) | 149 (3.1%) | 30 (11.0%) | 96 (5.8%) |
| Mean at start of MHQ distribution | 37.2 | 35.3 | 38.3 | 35.6 |
| Mean age at MHQ completion | 37.9 | n.a. | 39.4 | n.a. |
| Age at initial cancer diagnosis (Years) | ||||
| 0-4 | 553 (34.1%) | 2128 (43.6%) | n.a. | n.a. |
| 5-9 | 360 (22.2%) | 1165 (23.9%) | n.a. | n.a. |
| 10-14 | 377 (23.2%) | 897 (18.4%) | n.a. | n.a. |
| 15+ | 332 (20.5%) | 685 (14.1%) | n.a. | n.a. |
| Mean age at initial cancer diagnosis | 9.0 | 7.6 | n.a. | n.a. |
| Diagnosis | ||||
| Acute lymphoblastic leukemia | 500 (30.8%) | 1497 (30.7%) | n.a. | n.a. |
| AML and other leukemias | 35 (2.2%) | 158 (3.2%) | n.a. | n.a. |
| CNS tumors | 138 (8.5%) | 649 (13.3%) | n.a. | n.a. |
| Hodgkin lymphoma | 259 (16.0%) | 555 (11.4%) | n.a. | n.a. |
| Non-Hodgkin lymphoma | 179 (11.0%) | 505 (10.4%) | n.a. | n.a. |
| Kidney tumors | 132 (8.1%) | 417 (8.6%) | n.a. | n.a. |
| Neuroblastoma | 81 (5.0%) | 341 (7.0%) | n.a. | n.a. |
| Soft tissue sarcoma | 145 (8.9%) | 408 (8.4%) | n.a. | n.a. |
| Ewing sarcoma | 48 (3.0%) | 109 (2.2%) | n.a. | n.a. |
| Osteosarcoma | 100 (6.2%) | 220 (4.5%) | n.a. | n.a. |
| Other bone tumors | 5 (0.3%) | 16 (0.3%) | n.a. | n.a. |
| AAD score in first 5 years following diagnosis | ||||
| 0 | 678 (47.7%) | 1950 (51.8%) | n.a. | n.a. |
| 1 | 160 (11.3%) | 440 (11.7%) | n.a. | n.a. |
| 2 | 203 (14.3%) | 454 (12.1%) | n.a. | n.a. |
| 3 | 222 (15.6%) | 561 (14.9%) | n.a. | n.a. |
| 4 | 81 (5.7%) | 170 (4.5%) | n.a. | n.a. |
| ≥5 | 77 (5.4%) | 188 (5.0%) | n.a. | n.a. |
| Treatment with testicular irradiation in first 5 years following diagnosis | ||||
| No testicular irradiation | 506 (33.8%) | 1408 (35.3%) | n.a. | n.a. |
| < 4 Gy | 875 (58.5%) | 2316 (58.1%) | n.a. | n.a. |
| ≥4 Gy | 114 (7.6%) | 261 (6.6%) | n.a. | n.a. |
| Total body irradiation in first 5 years following diagnosis | ||||
| Yes | 24 (1.6%) | 56 (1.4%) | n.a. | n.a. |
| No | 1460 (98.4%) | 3859 (98.6%) | n.a. | n.a. |
Male Health Questionnaire = MHQ; Childhood Cancer Survivor Study = CCSS; FU2007 = CCSS Follow-Up 2007 Questionnaire; AML = acute myelogenous leukemia; CNS = central nervous system; AAD = summed alkylating agent dose score; Gy = gray
Excludes the1, 217 male subjects known to have died as of FU2007. The n=4,875 for this column is derived by subtracting the1,217 deceased males and the 1,622 living male survivors who participated in the MHQ from the total of 7,714 male CCSS survivors.
Excludes the 22 male siblings known to have died as of FU2007. The n=1,644 for this column is derived by subtracting the 22 deceased male siblings and the 274 living male siblings who participated in the MHQ from the total of 1,940 male CCSS siblings.
The marriage and pregnancy variables in this column are summarized only for MHQ non-participants who completed the FU2007 questionnaire. This was done to avoid ascertainment bias and ensure that the marriage and pregnancy proportion estimates shown for the MHQ participants, all of whom completed the FU2007 questionnaire, and for the MHQ non participant column are both based on similar duration of follow-up.
Includes 12 survivors and 3 siblings who reported that their partners were currently pregnant at the time of the FU2007 questionnaire completion.
Infertility in survivors compared to siblings
Of those who responded to the MHQ, 938 (57.8%) survivors, and 174 (63.5%) sibling controls reported having tried to become pregnant with a partner and were thus included in the infertility analysis (Figure I). Prevalence of infertility and infertility evaluations was compared in survivors and siblings (Table II). Infertility was reported by 46.0% of survivors and 17.5% of siblings (p<0.001), while 37.5% of survivors and 16.3% of siblings (p<0.001) reported they were not able to have all the children they wanted. Survivors who were not able to have all the children they wanted reported that male infertility was the most common reason (63.5%) compared to 14.8% of siblings (p<0.001).
Table 2. Summary of Infertility Prevalence and Infertility Evaluations among Male Survivors and Siblings Who Have Tried to Become Pregnant with a Partner.
| Survivors | Siblings | |
|---|---|---|
| Questionnaire Item 1 | Number (%) | Number (%) |
| Infertility Prevalence | ||
| Has a female partner ever had difficulty (it took >1 year) becoming pregnant by you? | n=938 | n=174 |
| --- Yes | 412 (46.0%)* | 29 (17.5%) |
| --- No | 483 (54.0%) | 137 (82.5%) |
| --- Unknown | 43 -- | 8 -- |
| Were you able to have all the children you wanted to have? | n=938 | n=174 |
| --- Yes | 568 (62.5%) | 139 (83.7%) |
| --- No | 341 (37.5%)* | 27 (16.3%) |
| --- Unknown | 29 -- | 8 -- |
| If no, which of you wanted more children? | n=341 | n=27 |
| --- I wanted more children but my partner did not | 19 (6.1%) | 3 (12.5%) |
| --- My partner wanted more children but I did not | 11 (3.5%) | 4 (16.7%) |
| --- We both wanted more children but we could not have more | 282 (90.4%)† | 17 (70.8%) |
| --- Unknown | 29 -- | 3 -- |
| If more children were wanted, what were the reasons for not having more children?2 | n=340 | n=27 |
| --- male infertility (% Yes) | 216 (63.5%)* | 4 (14.8%) |
| --- other health issues related to cancer treatment (% Yes) | 9 (2.6%) | 0 (0.0%) |
| --- other health issues not related to cancer treatment (% Yes) | 2 (0.6%) | 1 (3.7%) |
| --- female infertility (% Yes) | 35 (10.3%) | 6 (22.2%) |
| --- partner health issues (% Yes) | 22 (6.5%) | 4 (14.8%) |
| --- tried, but partner could not become pregnant, reason unknown (% Yes) | 61 (17.9%) | 5 (18.5%) |
| --- issues other than health (% Yes) | 51 (15.0%)† | 11 (40.7%) |
| Medical Evaluation for Infertility | ||
| Have you or a female partner ever been evaluated for infertility? | n=938 | n=174 |
| --- Yes | 490 (53.6%)* | 36 (21.4%) |
| --- No | 420 (45.9%) | 131 (78.0%) |
| --- I don’t know | 5 (0.5%) | 1 (0.6%) |
| --- No response | 23 -- | 6 -- |
| If you or your partner were evaluated for decreased fertility, was a problem identified? | n=490 | n=36 |
| --- Yes. A fertility problem was found in my partner | 5 (1.0%) | 2 (5.6%) |
| --- Yes. A fertility problem was found in me | 421 (85.9%) | 30 (83.3%) |
| --- Both of the above | 49 (10.0%) | 2 (5.6%) |
| --- No | 12 (2.5%) | 2 (5.6%) |
| --- I don’t know | 3 (0.6%) | 0 (0.0%) |
| Self-report of semen analysis results (among those who reported that a male fertility problem was identified in medical evaluations) | n=470 (421+49) | n=32 (30+2) |
| --- Normal sperm count and normal motility | 122 (26.0%) | 19 (59.4%) |
| --- Low sperm count and/or low motility | 306 (65.1%)* | 8 (25.0%) |
| --- Subject did not know or declined to report semen analysis results | 33 (7.0%) | 3 (9.4%) |
| --- Subject indicated that he had never had a semen analysis | 9 (1.9%) | 2 (6.2%) |
Subjects who responded “Yes” to the question “Have you ever tried with a partner to become pregnant?” were included in this table. The infertility section of the MHQ contained built-in skip patterns to ensure that each respondent only answered items that were relevant to his particular situation. The total size of the relevant subject pool for each question is given at the top of each section. Subjects who did not supply an answer were excluded from the denominator in the percentage calculations.
The reasons listed in this section are not mutually exclusive. Each row in this section was summarized and tested as a separate “Yes/No” item. One of the 341 survivors eligible to report on reasons skipped this section entirely; hence the denominator for the % Yes calculation among survivors is 340.
The test for differences in proportions between survivors and siblings was statistically significant (* indicates p<0.001, † indicates p<0.05).
Of the 412 survivors who met the definition for infertility, 152 (37%) had reported at least one pregnancy with a female partner that resulted in a live birth. Another 34 of the 412 survivors had reported at least one pregnancy with a female partner, but no live births, so a total of 186 (45%) reported ever fathering a pregnancy. Of the 29 siblings who met the definition for infertility, 20 (69%) had reported at least one pregnancy with a female partner that resulted in a live birth. The other 9 siblings with infertility reported no partner pregnancies at all.
Over half of survivors (53.6%) reported that they and/or their female partner had been medically evaluated for infertility in comparison to 21.4% of siblings (p<0.001). Among both siblings and survivors, the majority reported a male contribution to an identified fertility problem (95.9% for survivors; 88.9% for siblings). Among males who reported a male fertility problem was identified in medical evaluations, survivors were more likely than siblings to report low sperm count and low sperm motility (65.1% for survivors; 25.0% for siblings).
Assessment of risk factors for infertility
The prevalence of infertility by demographic characteristics and cancer treatment factors is provided in Table III for survivors with no history of a recurrence or SMN. Infertility was 24% among survivors younger than 30 years of age, and increased to greater than 40% among those older than 30 years of age. Differences based on race/ethnicity were difficult to assess due to the small numbers of minority participants. The prevalence of infertility among survivors was not significantly associated with marital status, college education, full time employment, income and health insurance status.
Survivors of bone tumors other than osteosarcoma (23/33; 69.7%), Hodgkin lymphoma (69/117; 59.0%), and soft tissue sarcomas (46/80; 57.5%) had a high prevalence of infertility (Table III). The prevalence of infertility increased with higher AAD. Similar rates of infertility were seen among those who received no radiation to the testes (85/231; 36.8%) or less than 4 Gy (166/397; 41.8%). However, among those receiving greater than or equal to 4 Gy (mean dose 10.3; range 4 – 29.1), 82.6% (19/23) were infertile. The prevalence of infertility did not consistently increase with increasing radiation dose to the hypothalamus/pituitary. Operations involving the spinal cord/canal or sympathetic nerves were not associated with higher rates of infertility. Among the small number of patients whose cancer treatment included surgical excision of any of the organs of the genital tract (orchiectomy, radical prostatectomy or total cystectomy), there was a very high prevalence of infertility (12/14, 85.7%), though most of the affected survivors had multiple treatment-related risk factors and two patients with bilateral orchiectomies were included. Eight out of ten survivors with unilateral orchiectomy reported infertility; however seven also had an AAD ≥ 3.
In the multivariable analysis, an AAD ≥3, surgical excision of any organ of the genital tract, and testicular radiation dose ≥ 4Gy were all statistically significant independent risk factors for infertility (Table IV). An AAD ≥ 3 (RR= 2.13, 95% CI 1.69-2.68) was associated with a high risk for infertility versus an AAD <3. Among the non-alkylating chemotherapy agents that were evaluated, bleomycin was the only one independently associated with an increased relative risk (RR) of infertility (RR 1.55; 95% CI 1.2 – 2.01, p=0.0008). Exposure to methotrexate was not a statistically significant risk factor after adjusting for the other variables, but it was retained in the model because it did not meet the pre-defined criteria (p>0.20 and <10% change in all other relative risk parameters) necessary to allow its removal.
Table 4. Multivariable Model Assessing Risk Factors for Infertility in Male Cancer Survivors with No History of a Recurrence or Secondary Malignant Neoplasm.
| Factors1 | Factor Level | Multivariable Model Estimates | |||
|---|---|---|---|---|---|
| Relative Risk for Infertility | 95% CI | p-value | |||
| Lower Limit | Upper Limit | ||||
| Age at time of MHQ questionnaire (Years) | 20 to 29 | 1.00 | -- | -- | -- |
| 30 to 39 | 1.46 | 0.82 | 2.60 | 0.20 | |
| 40 to 49 | 1.39 | 0.78 | 2.48 | 0.27 | |
| 50+ | 1.91 | 0.98 | 3.74 | 0.06 | |
| Surgical excision of any organ of the genital tract2 | No | 1.00 | -- | -- | -- |
| Yes | 1.63 | 1.20 | 2.21 | 0.002 | |
| Testicular radiation dose | No testicular radiation | 1.00 | -- | -- | -- |
| <4Gy | 1.04 | 0.84 | 1.29 | 0.70 | |
| >=4Gy | 1.99 | 1.52 | 2.61 | <0.0001 | |
| AAD (non-platinum) score | 0 | 1.00 | -- | -- | -- |
| 1 | 1.06 | 0.66 | 1.69 | 0.82 | |
| 2 | 1.31 | 0.94 | 1.84 | 0.11 | |
| 3 | 2.04 | 1.58 | 2.64 | <0.0001 | |
| 4 | 2.07 | 1.52 | 2.82 | <0.0001 | |
| 5+ | 2.39 | 1.79 | 3.20 | <0.0001 | |
| Received bleomycin | No | 1.00 | -- | -- | -- |
| Yes | 1.55 | 1.20 | 2.01 | <0.001 | |
| Received methotrexate | No | 1.00 | -- | -- | -- |
| Yes | 0.84 | 0.67 | 1.04 | 0.11 | |
CI = Confidence interval; MHQ = Male Health Questionnaire; Gy = Gray; AAD = summed alkylating agent dose score
The factors included in this multivariable model are those that showed independent contributions to model fit (see methods section for details). Other factors that were evaluated in preliminary modeling, but not included in the final multivariable model due to lack of independent association with infertility were: race, marital status, age at diagnosis, timing of puberty onset, operations on the spinal canal or sympathetic nerves, hypothalamic/pituitary radiation dose and the indicator variables for exposure to anthracyclines, platinum-containing agents, vincristine, vinblastine, Ara-C, mercaptopurine, thioguanine, L-asparaginase, hydroxyurea, prednisone, dexamethasone and actinomycin D.
This surgery variable equals “Yes” if the subject’s cancer treatment records included one or more of the following procedures: orchiectomy, radical prostatectomy or total cystectomy.
Discussion
The CCSS is a large cohort of childhood cancer survivors with detailed exposure data and a sibling control group. In this study we evaluated infertility, defined as the inability of a female partner to conceive after 12 months of trying to become pregnant [17], among adult male CCSS participants compared to male sibling controls. It is widely accepted that treatment in the form of certain chemotherapy, radiation therapy, and surgeries, can have adverse and long-term effects on male fertility. The MHQ provided additional data on male-factor infertility including the desire and ability to have more children, the reasons for not having more children, and medical evaluation of infertility. This analysis is a unique evaluation of fertility problems in male survivors of childhood cancer in that it includes males who may have fathered a pregnancy but had difficulty in doing so.
Survivors demonstrated an increased prevalence of infertility (46.0%), and high utilization of fertility services with more than one half of these survivors undergoing an infertility evaluation. However, survivors were also more likely to not have all the children they wanted and attribute male-factor infertility as the reason for not having more children (p < 0.001 for all) compared to their siblings. Encouragingly, 37% of survivors who met the definition for infertility had reported at least one pregnancy with a female partner that resulted in a live birth compared to 69% of siblings.
While research on the psychosocial implications of male infertility and its treatment lags behind studies in women there is growing evidence to suggest that regardless of gender differences in coping styles, men are emotionally affected by infertility [18]. Recent studies suggest the psychologic effect of not having a child may in fact be greater for men versus women and that men undergoing treatment for infertility suffer similar levels of distress compared to women[18, 19].
In the United States, the National Survey of Family Growth (NSFG) estimated 7.4% of couples to be infertile [20], though analyses in Canada (11.5-15.7%) and France (24%), give higher estimates [21, 22]. The NSFG also estimates a rate of 7.4% of sexually active men who personally sought help in regard to having a child and 18.1% of males who sought an evaluation were diagnosed to have a male-related infertility condition. [23] In this analysis, the rate of infertility, infertility evaluations pursued by survivors, and male contribution to infertility was also higher than population estimates.
In survivors infertility was most prevalent among those treated for sarcomas other than osteosarcoma (Ewing sarcoma, soft tissue sarcoma, and other bone tumors) and Hodgkin lymphoma, which are commonly treated with high-dose alkylating agent therapy. Infertility was least prevalent among survivors of CNS tumors which may be due to a high prevalence of surgery alone as treatment (16/41; 39.0%). However, the percentage of survivors reporting infertility across all cancer diagnoses was greater compared to siblings. This is consistent with results of the prior CCSS analysis in which the HRs of fathering a pregnancy were reduced in all male survivors except those with a primary diagnosis of Wilms tumor or neuroblastoma when compared to siblings [4]. In the treatment model, high-dose alkylating agent exposure (AAD ≥ 3), surgical excision of any male genital tract organ, and testicular radiation dose ≥ 4 Gy were associated with infertility, as found in previous studies [24-30].
Bleomycin, commonly used in regimens for Hodgkins and germ cell tumors, has not previously been associated with testicular germ cell damage and infertility and the current association with infertility is a surprising and novel finding of this study in need of further investigation [31, 32, 25]. While this may be a false positive finding, bleomycin may contribute to infertility but to a lesser extent than alkylators. In a large study of male infertility in Hodgkin lymphoma survivors, an elevated follicular stimulating hormone (FSH) was found in 8% (8/101) of males who had received non-alkylator regimens with either ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) or EBVP (epirubicin, bleomycin, vinblastine, prednisone) and in 3% of males treated with radiation only (which did not include direct gonadal exposure), however persistent FSH elevation was uncommon [33]. Also, many of the survivors with bleomycin exposure had a primary diagnosis of Hodgkin lymphoma (24/51; 47.1%) in which there is known to be testicular dysfunction at baseline prior to cancer treatment.[34]. Survivors of germ cell tumors are not included in the CCSS cohort.
Therapies that affect the hypothalamic/pituitary axis have been associated with impaired fertility in some studies [2, 35-37, 4, 38]. In our analysis the prevalence of infertility was similar among those who received no hypothalamic/pituitary radiation (36.8%), low-dose hypothalamic/pituitary radiation (< 30 Gy) (44.3%), or high-dose hypothalamic/pituitary radiation (≥ 30 Gy) (43.5%). CNS tumor survivors were underrepresented in the MHQ. Many of these received no radiation in their treatment (44%), which may explain the inability to demonstrate increasing central radiation as a risk factor.
The present study has some limitations. First, we used a medical definition of infertility. Therefore, those who did not “try” to father a pregnancy but were able to do so and those who decided not to try to father a pregnancy for either medical or psychosocial reasons are not included. Second, the CCSS is a retrospective cohort and relies on self-report questionnaires. This is not the most accurate or direct measure of infertility and also limited our ability to look at variables such as body mass index. This analysis was based on the MHQ and thus was limited by the questions asked in this ancillary study of the CCSS. As a cross-sectional study data was not available on the timing of the bout of infertility or infertility evaluation in relation to pregnancies, lives births or previous CCSS questionnaires. This limited our ability to address primary versus secondary infertility and analyze associations with other time dependent variables. The treatment analysis did not include survivors who had experienced a recurrence or SMN, likely underestimating reported infertility in this population of male cancer survivors as those likely to be most heavily treated were excluded. Lastly, data also suggests that males may underreport pregnancies, partly because they may not be informed of all pregnancies by partners [4, 41].
The sensitive nature of questions in addition to demographic and treatment factors may have affected those who participated in this survey and accuracy of the reporting. In this analysis, 17.5% of male siblings who reported infertility is in broad agreement with worldwide estimates from Canada and France of 11.5 - 24%, though higher than those reported in the United States (7.4%) [22, 20, 21]. The rate of infertility evaluations pursued by siblings (21%) is also higher than the baseline rate from the NSFG in the general population of 7.4% [23]. Male contribution to infertility was identified in the majority of siblings who underwent a fertility evaluation (95.9% for survivors; 88.9% for siblings) which is substantially higher than population estimates from the NSFG of 18.1% suggesting some potential selection bias [23]. Survivor and sibling participants were more likely to be white, married, and older at the time of survey administration which are all factors associated with a higher likelihood of seeking an infertility evaluation. Older age is also a risk factor for infertility. However, survivors still reported relatively greater rates of infertility, not having all the children they wanted, pursuing infertility evaluations and male-factor infertility. Survivor participants were more likely to have Hodgkin lymphoma, associated with more fertility problems, and less likely to have CNS tumors, associated with less fertility problems, thus potentially overestimating the general prevalence. Survivor participants were also more likely to have fathered a pregnancy versus non-participants which may have caused bias in either direction.
This study highlights risk factors for fertility problems among male survivors of childhood cancer and the important reality that many male survivors of childhood cancer and their partners are unable to have all the children they want due to male-factor infertility. Risk factors for infertility include a testicular radiation dose of 4 Gy or more, surgical excision of any organ of the genital tract, exposure to higher doses of alkylating agents, and bleomycin exposure. The novel association of infertility and bleomycin will require future investigation. This information is important in counseling patients prior to cancer treatment as they make decisions about cryopreservation of semen. It is also helpful at the completion of cancer treatment as they make life decisions related to reproduction that include ongoing maintenance of cryopreserved sperm and pursuit of fertility evaluations. Future studies should examine infertility in male survivors of childhood cancer longitudinally in order to further investigate primary versus secondary infertility as well as fertility recovery over time. The psychological and social aspects of infertility for male survivors of childhood cancer also warrant further study.
Supplementary Material
Acknowledgments
Funding:
This work was supported by the Lance Armstrong Foundation (L.R.M.); and the National Cancer Institute of the National Institutes of Health, Department of Health and Human Services (U24- CA 55727 to L.L.R.).
Footnotes
Conflict of Interest: There are no conflicts of interest to declare
References
- 1.Howlader N, Noone AM, Krapcho M, Neyman N, Aminou R, Waldron W, et al. SEER Cancer Statistics Review, 1975-2008. National Cancer Institute; Bethesda, MD: 2011. http://seer.cancer.gov/csr/1975_2008. [Google Scholar]
- 2.Byrne J, Fears TR, Mills JL, Zeltzer LK, Sklar C, Meadows AT, et al. Fertility of long-term male survivors of acute lymphoblastic leukemia diagnosed during childhood. Pediatric blood & cancer. 2004;42(4):364–72. doi: 10.1002/pbc.10449. [DOI] [PubMed] [Google Scholar]
- 3.Cvancarova M, Samuelsen SO, Magelssen H, Fossa SD. Reproduction rates after cancer treatment: experience from the Norwegian radium hospital. J Clin Oncol. 2009;27(3):334–43. doi: 10.1200/JCO.2007.15.3130. [DOI] [PubMed] [Google Scholar]
- 4.Green DM, Kawashima T, Stovall M, Leisenring W, Sklar CA, Mertens AC, et al. Fertility of male survivors of childhood cancer: a report from the Childhood Cancer Survivor Study. J Clin Oncol. 2010;28(2):332–9. doi: 10.1200/JCO.2009.24.9037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Magelssen H, Melve KK, Skjaerven R, Fossa SD. Parenthood probability and pregnancy outcome in patients with a cancer diagnosis during adolescence and young adulthood. Hum Reprod. 2008;23(1):178–86. doi: 10.1093/humrep/dem362. [DOI] [PubMed] [Google Scholar]
- 6.Pivetta E, Maule MM, Pisani P, Zugna D, Haupt R, Jankovic M, et al. Marriage and parenthood among childhood cancer survivors: a report from the Italian AIEOP Off-Therapy Registry. Haematologica. 2011;96(5):744–51. doi: 10.3324/haematol.2010.036129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Robison LL, Armstrong GT, Boice JD, Chow EJ, Davies SM, Donaldson SS, et al. The Childhood Cancer Survivor Study: a National Cancer Institute-supported resource for outcome and intervention research. J Clin Oncol. 2009;27(14):2308–18. doi: 10.1200/JCO.2009.22.3339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Leisenring WM, Mertens AC, Armstrong GT, Stovall MA, Neglia JP, Lanctot JQ, et al. Pediatric cancer survivorship research: experience of the Childhood Cancer Survivor Study. J Clin Oncol. 2009;27(14):2319–27. doi: 10.1200/JCO.2008.21.1813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Robison LL, Mertens AC, Boice JD, Breslow NE, Donaldson SS, Green DM, et al. Study design and cohort characteristics of the Childhood Cancer Survivor Study: a multi-institutional collaborative project. Med Pediatr Oncol. 2002;38(4):229–39. doi: 10.1002/mpo.1316. [DOI] [PubMed] [Google Scholar]
- 10.Tucker MA, Meadows AT, Boice JD, Jr, Stovall M, Oberlin O, Stone BJ, et al. Leukemia after therapy with alkylating agents for childhood cancer. J Natl Cancer Inst. 1987;78(3):459–64. [PubMed] [Google Scholar]
- 11.Green DM, Kawashima T, Stovall M, Leisenring W, Sklar CA, Mertens AC, et al. Fertility of female survivors of childhood cancer: a report from the childhood cancer survivor study. J Clin Oncol. 2009;27(16):2677–85. doi: 10.1200/JCO.2008.20.1541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stovall M, Donaldson SS, Weathers RE, Robison LL, Mertens AC, Winther JF, et al. Genetic effects of radiotherapy for childhood cancer: gonadal dose reconstruction. International journal of radiation oncology, biology, physics. 2004;60(2):542–52. doi: 10.1016/j.ijrobp.2004.03.017. [DOI] [PubMed] [Google Scholar]
- 13.Stovall M, Weathers R, Kasper C, Smith SA, Travis L, Ron E, et al. Dose reconstruction for therapeutic and diagnostic radiation exposures: use in epidemiological studies. Radiat Res. 2006;166(1 Pt 2):141–57. doi: 10.1667/RR3525.1. [DOI] [PubMed] [Google Scholar]
- 14.Packer RJ, Gurney JG, Punyko JA, Donaldson SS, Inskip PD, Stovall M, et al. Long-term neurologic and neurosensory sequelae in adult survivors of a childhood brain tumor: childhood cancer survivor study. J Clin Oncol. 2003;21(17):3255–61. doi: 10.1200/JCO.2003.01.202. [DOI] [PubMed] [Google Scholar]
- 15.Zeger SL, Liang KY. Longitudinal data analysis using generalized linear models. Biometrika. 1986;73:13–22. [Google Scholar]
- 16.Zou G. A modified poisson regression approach to prospective studies with binary data. American journal of epidemiology. 2004;159(7):702–6. doi: 10.1093/aje/kwh090. [DOI] [PubMed] [Google Scholar]
- 17.Practice Committee of tAmerican Society for Reproductive M. Definitions of infertility and recurrent pregnancy loss. Fertility and sterility. 2008;90(5 Suppl):S60. doi: 10.1016/j.fertnstert.2008.08.065. [DOI] [PubMed] [Google Scholar]
- 18.Culley L, Hudson N, Lohan M. Where are all the men? The marginalization of men in social scientific research on infertility. Reproductive biomedicine online. 2013 doi: 10.1016/j.rbmo.2013.06.009. [DOI] [PubMed] [Google Scholar]
- 19.Peronace LA, Boivin J, Schmidt L. Patterns of suffering and social interactions in infertile men: 12 months after unsuccessful treatment. Journal of psychosomatic obstetrics and gynaecology. 2007;28(2):105–14. doi: 10.1080/01674820701410049. [DOI] [PubMed] [Google Scholar]
- 20.Chandra A, Martinez GM, Mosher WD, Abma JC, Jones J. Fertility, family planning, and reproductive health of U.S. women: data from the 2002 National Survey of Family Growth. Vital Health Stat. 2005;23(25):1–160. [PubMed] [Google Scholar]
- 21.Slama R, Hansen OK, Ducot B, Bohet A, Sorensen D, Giorgis Allemand L, et al. Estimation of the frequency of involuntary infertility on a nation-wide basis. Hum Reprod. 2012;27(5):1489– 98. doi: 10.1093/humrep/des070. [DOI] [PubMed] [Google Scholar]
- 22.Bushnik T, Cook JL, Yuzpe AA, Tough S, Collins J. Estimating the prevalence of infertility in Canada. Human Reproduction. 2012;27(3):738–46. doi: 10.1093/humrep/der465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Anderson JE, Farr SL, Jamieson DJ, Warner L, Macaluso M. Infertility services reported by men in the United States: national survey data. Fertility and sterility. 2009;91(6):2466–70. doi: 10.1016/j.fertnstert.2008.03.022. [DOI] [PubMed] [Google Scholar]
- 24.Rowley MJ, Leach DR, Warner GA, Heller CG. Effect of graded doses of ionizing radiation on the human testis. Radiat Res. 1974;59(3):665–78. [PubMed] [Google Scholar]
- 25.Dohle GR. Male infertility in cancer patients: Review of the literature. Int J Urol. 2010;17(4):327–31. doi: 10.1111/j.1442-2042.2010.02484.x. [DOI] [PubMed] [Google Scholar]
- 26.Howell S, Shalet S. Gonadal damage from chemotherapy and radiotherapy. Endocrinology and metabolism clinics of North America. 1998;27(4):927–43. doi: 10.1016/s0889-8529(05)70048-7. [DOI] [PubMed] [Google Scholar]
- 27.Jaffe N, Sullivan MP, Ried H, Boren H, Marshall R, Meistrich M, et al. Male reproductive function in long-term survivors of childhood cancer. Med Pediatr Oncol. 1988;16(4):241–7. doi: 10.1002/mpo.2950160404. [DOI] [PubMed] [Google Scholar]
- 28.Kenney LB, Laufer MR, Grant FD, Grier H, Diller L. High risk of infertility and long term gonadal damage in males treated with high dose cyclophosphamide for sarcoma during childhood. Cancer. 2001;91(3):613–21. doi: 10.1002/1097-0142(20010201)91:3<613∷AID-CNCR1042>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 29.Relander T, Cavallin-Stahl E, Garwicz S, Olsson AM, Willen M. Gonadal and sexual function in men treated for childhood cancer. Med Pediatr Oncol. 2000;35(1):52–63. doi: 10.1002/1096-911X(200007)35:1<52∷AID-MPO9>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 30.Romerius P, Stahl O, Moell C, Relander T, Cavallin-Stahl E, Wiebe T, et al. High risk of azoospermia in men treated for childhood cancer. Int J Androl. 2011;34(1):69–76. doi: 10.1111/j.1365-2605.2010.01058.x. [DOI] [PubMed] [Google Scholar]
- 31.Viviani S, Santoro A, Ragni G, Bonfante V, Bestetti O, Bonadonna G. Gonadal toxicity after combination chemotherapy for Hodgkin’s disease. Comparative results of MOPP vs ABVD. Eur J Cancer Clin Oncol. 1985;21(5):601–5. doi: 10.1016/0277-5379(85)90088-4. [DOI] [PubMed] [Google Scholar]
- 32.Brougham MF, Wallace WH. Subfertility in children and young people treated for solid and haematological malignancies. Br J Haematol. 2005;131(2):143–55. doi: 10.1111/j.1365-2141.2005.05740.x. [DOI] [PubMed] [Google Scholar]
- 33.van der Kaaij MA, Heutte N, Le Stang N, Raemaekers JM, Simons AH, Carde P, et al. Gonadal function in males after chemotherapy for early-stage Hodgkin’s lymphoma treated in four subsequent trials by the European Organisation for Research and Treatment of Cancer: EORTC Lymphoma Group and the Groupe d’Etude des Lymphomes de l’Adulte. J Clin Oncol. 2007;25(19):2825–32. doi: 10.1200/JCO.2006.10.2020. [DOI] [PubMed] [Google Scholar]
- 34.Rueffer U, Breuer K, Josting A, Lathan B, Sieber M, Manzke O, et al. Male gonadal dysfunction in patients with Hodgkin’s disease prior to treatment. Ann Oncol. 2001;12(9):1307– 11. doi: 10.1023/a:1012464703805. [DOI] [PubMed] [Google Scholar]
- 35.Byrne J, Fears TR, Mills JL, Zeltzer LK, Sklar C, Nicholson HS, et al. Fertility in women treated with cranial radiotherapy for childhood acute lymphoblastic leukemia. Pediatric blood & cancer. 2004;42(7):589–97. doi: 10.1002/pbc.20033. [DOI] [PubMed] [Google Scholar]
- 36.Darzy KH. Radiation-induced hypopituitarism after cancer therapy: who, how and when to test. Nat Clin Pract Endocrinol Metab. 2009;5(2):88–99. doi: 10.1038/ncpendmet1051. [DOI] [PubMed] [Google Scholar]
- 37.Schmiegelow M, Lassen S, Poulsen HS, Schmiegelow K, Hertz H, Andersson AM, et al. Gonadal status in male survivors following childhood brain tumors. The Journal of clinical endocrinology and metabolism. 2001;86(6):2446–52. doi: 10.1210/jcem.86.6.7544. [DOI] [PubMed] [Google Scholar]
- 38.Green DM, Nolan VG, Kawashima T, Stovall M, Donaldson SS, Srivastava D, et al. Decreased fertility among female childhood cancer survivors who received 22-27 Gy hypothalamic/pituitary irradiation: a report from the Childhood Cancer Survivor Study. Fertility and sterility. 2011;95(6):1922-7–7 e1. doi: 10.1016/j.fertnstert.2011.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


