Background: IcaB is a poly-β-1,6-N-acetyl-d-glucosamine (PNAG) deacetylase required for polysaccharide intercellular adhesion-dependent biofilm formation by staphylococci.
Results: The structure of Ammonifex degensii IcaB has been determined and its catalytic mechanism and localization characterized.
Conclusion: IcaB is a membrane-associated PNAG deacetylase that uses an altered catalytic mechanism relative to other family 4 carbohydrate esterases.
Significance: First structural characterization of a Gram-positive PNAG deacetylase.
Keywords: Biofilm, Catalysis, Microbiology, Polysaccharide, Protein Structure, PNAG, Carbohydrate Esterase, Deacetylase, Exopolysaccharide Biosynthesis
Abstract
Exopolysaccharides are required for the development and integrity of biofilms produced by a wide variety of bacteria. In staphylococci, partial de-N-acetylation of the exopolysaccharide poly-β-1,6-N-acetyl-d-glucosamine (PNAG) by the extracellular protein IcaB is required for biofilm formation. To understand the molecular basis for PNAG de-N-acetylation, the structure of IcaB from Ammonifex degensii (IcaBAd) has been determined to 1.7 Å resolution. The structure of IcaBAd reveals a (β/α)7 barrel common to the family four carbohydrate esterases (CE4s) with the canonical motifs circularly permuted. The metal dependence of IcaBAd is similar to most CE4s showing the maximum rates of de-N-acetylation with Ni2+, Co2+, and Zn2+. From docking studies with β-1,6-GlcNAc oligomers and structural comparison to PgaB from Escherichia coli, the Gram-negative homologue of IcaB, we identify Arg-45, Tyr-67, and Trp-180 as key residues for PNAG binding during catalysis. The absence of these residues in PgaB provides a rationale for the requirement of a C-terminal domain for efficient deacetylation of PNAG in Gram-negative species. Mutational analysis of conserved active site residues suggests that IcaB uses an altered catalytic mechanism in comparison to other characterized CE4 members. Furthermore, we identified a conserved surface-exposed hydrophobic loop found only in Gram-positive homologues of IcaB. Our data suggest that this loop is required for membrane association and likely anchors IcaB to the membrane during polysaccharide biosynthesis. The work presented herein will help guide the design of IcaB inhibitors to combat biofilm formation by staphylococci.
Introduction
Staphylococcus epidermidis is a Gram-positive commensal bacteria that is the most prevalent organism found in indwelling medical-device-related bacterial infections (1). Nosocomial S. epidermidis infections are often caused by the bacteria forming a biofilm on biomaterials (2, 3). A biofilm is a cluster of microcolonies encapsulated within self-produced extracellular polymeric substances (4, 5). The components of the biofilms can be quite diverse but are generally composed of proteinaceous adhesins, nucleic acids, and exopolysaccharides (6–8). Exopolysaccharides are important for biofilm structure and architecture by allowing the diffusion of nutrients in and waste products out (4, 9). Exopolysaccharides have also been shown to function as adhesins, reduce the diffusion of antibiotics into the biofilm, and provide a barrier against phagocytosis (7, 10).
The conserved exopolysaccharide known as polysaccharide intercellular adhesin was originally identified in the biofilms of S. epidermidis (11) and Staphylococcus aureus (12) and has now been shown to be produced by various Gram-negative bacteria (13–19) and higher eukaryotes (20). Polysaccharide intercellular adhesin is synthesized as a β-1,6-linked poly-N-acetyl-d-glucosamine (PNAG)5 polymer and subsequently modified by partially de-N-acetylation and/or O-succinylation. In S. epidermidis, de-N-acetylated PNAG (dPNAG) production is dependent on the four-gene operon, icaADBC (21). IcaA, which is predicted to contain multiple transmembrane domains and a large cytosolic family 2 glycosyltransferase domain, is thought to be responsible for the production of PNAG and its translocation across the membrane (21, 22). IcaD is a small integral membrane protein that significantly increases PNAG biosynthesis when co-expressed with IcaA and potentially aids in PNAG translocation across the membrane (22). IcaC is an integral membrane protein that was originally predicted to be responsible for exporting mature long-chain PNAG (21). However, the proposed function of IcaC has recently been revisited (23). Bioinformatics analysis predicts that IcaC contains 10 transmembrane helices and is a member of a large acetyltransferase family, suggesting it plays a role in the O-modification of PNAG during biosynthesis (23). The presence of succinate groups on PNAG isolated from S. epidermidis (24) and S. aureus (25) supports the role of IcaC as an O-succinyltransferase. IcaB is an extracellular protein that is responsible for the partial de-N-acetylation of PNAG (26), a requirement for surface retention of the polymer. Furthermore, ΔicaB strains of S. epidermidis and S. aureus are unable to de-N-acetylate PNAG, do not form biofilms in vitro, and have highly attenuated virulence in a murine model for an indwelling medical device-related infection and bacteremia, respectively (26, 27). IcaB is a member of the family 4 carbohydrate esterases (CE4s) and has sequence homology (22% identity) to the N-terminal de-N-acetylase domain of Escherichia coli PgaB (PgaB22–309) (28). Recent characterization of PgaB has shown that its C-terminal domain (PgaB310–672) is required for binding and de-N-acetylation of PNAG, as the isolated PgaB22–309 de-N-acetylase domain is inactive (29).
Herein we present the first structure of IcaB from Ammonifex degensii (IcaBAd). The identification of key active site residues that are conserved within Gram-positive homologues provides a structural rationale for why IcaB, unlike its Gram-negative counterpart PgaB, does not require a C-terminal domain for enzymatic activity (29). Biochemical characterization of IcaBAd and S. epidermidis IcaB (IcaBSe) suggests the extracellular enzymes are membrane-associated and are anchored by a conserved hydrophobic loop. Furthermore, we provide the first mutational analysis of a PNAG deacetylase. The mutagenesis data suggests that the circular permutation of the CE4 motifs alters the enzymatic mechanism relative to other CE4s members (30, 31).
EXPERIMENTAL PROCEDURES
Cloning, Expression, and Purification of IcaBSe Constructs
The plasmid UT032 (32), which contains a codon-optimized version of the icaB gene from S. epidermidis (encoding residues 30–289) in pET16b, was used as a template to subclone icaB into the pET28a expression vector (Novagen). Inverse PCR was used with the forward and reverse primers GGGCATATGGCGAACGAAGAAAACAAAAAACTG and GGCTCGAGTCATTTTTCTTCGTCGAAACCGTCCC, which contain an NdeI and XhoI site, respectively, to yield plasmid pET28-IcaBSe30–289. The resulting plasmid encodes a thrombin-cleavable N-terminal hexahistidine tag fused to IcaBSe30–289. The D120N and H50A mutants of IcaBSe30–289 were generated using the QuikChange lightning site-directed mutagenesis kit (Agilent Technologies) as per the manufacturer's instructions with the forward and reverse primers GGATCAACTTCAACGACATGGACCAGACCATCTAC and GTAGATGGTCTGGTCCATGTCGTTGAAGTTGATCC and CTGGCGCTGAACTACGCCCGTGTTCG and CGAACACGGGCGTAGTTCAGCGCCAG, respectively. The hydrophobic loop deletion mutant of IcaBSe30–289 (IcaBSe30–289Δloop) was generated in three successive steps using the QuikChange lightening site-directed mutagenesis kit with the following modifications; (i) the denaturing and annealing duration steps were 30 s; (ii) the annealing temperature was 55 °C; (iii) the protocol was completed with 25 cycles. The first step deleted residues 54–72 using the forward and reverse primers CTACCACCGTGTTCGTAACTACTCTGTTACCG and CGGTAACAGAGTAGTTACGAACACGGTGGTAG. The second step inserted residues AAG after Arg-53 using the forward and reverse primers GAACTACCACCGTGTTCGTGCGGCGGGTGAAATCAAAAACTACTCTG and CAGAGTAGTTTTTGATTTCACCCGCCGCACGAACACGGTGGTAGTTC. The third step inserted residues EI to yield AAEIG after Arg-53 using the forward and reverse primers CCACCGTGTTCGTGCGGCGGAAATTGGTGAAATCAAAAAC and GTTTTTGATTTCACCAATTTCCGCCGCACGAACACGGTGG. The resulting construct IcaBSe30–289Δloop contains the mutations K54A, K55A, and K72G with residues 56–69 deleted.
The following protocol was used to express and purify all the IcaBSe constructs. E. coli BL21-CodonPlus cells transformed with the appropriate plasmid were grown in l liter of Luria-Bertani (LB) medium with 50 μg/ml kanamycin at 37 °C to an absorbance at 600 nm (A600) of ∼0.4–0.5 and moved to 25 °C. When the cultures reached an A600 of ∼0.6–0.7, protein expression was induced by the addition of isopropyl-d-1-thiogalactopyranoside to a final concentration of 1 mm. The cells were incubated for 3 h at 25 °C, harvested by centrifugation at 5000 × g for 20 min, and frozen on dry ice. Cell pellets were thawed and resuspended in 40 ml of lysis buffer (50 mm HEPES, pH 7.0, 1 m NaCl, 10 mm imidazole, 5% (v/v) glycerol, and one complete mini protease inhibitor mixture tablet (Roche Applied Science)). Resuspended cells were disrupted with three passes through an Emulsiflex-c3 (Avestin) at 15,000 p.s.i., and cell debris was removed by centrifugation at 31,000 × g for 30 min. The resulting supernatant was passed over a gravity column containing 3 ml of nickel-nitrilotriacetic acid resin (Qiagen) that was pre-equilibrated with buffer A (20 mm HEPES, pH 7.0, 1 m NaCl, 10 mm imidazole). Bound protein was washed with 10 column volumes of buffer A, 3 column volumes of buffer A with 20 mm imidazole, and eluted with 5 column volumes of buffer A with 250 mm imidazole. The eluted fraction was concentrated using a 10-kDa cut-off Amicon ultrafiltration device (EMD Millipore) and further purified and buffer-exchanged into buffer B (20 mm HEPES, pH 7.0, and 1 m NaCl) by gel filtration chromatography using a HiLoad 16/60 Superdex 200 prep-grade column (GE Healthcare). The purified IcaBSe constructs were >95% pure as judged by SDS-PAGE and stable for ∼1 week at 4 °C.
Cloning, Expression, and Purification of IcaBAd Constructs
A codon-optimized version of A. degensii icaB (GeneArt Invitrogen) was used as the template to subclone mature icaB into the pET28a expression vector (Novagen). Inverse PCR was used with the forward and reverse primers GGGCATATGGAAAGTCCGCGTACACCGGCAGGC and GGCTCGAGTTACGGGCTTGCCTGTGCCCATGCT, which contain an NdeI and XhoI site, respectively, to yield plasmid pET28-IcaBAd23–280. The resulting plasmid encodes a thrombin-cleavable N-terminal hexahistidine tag fused to IcaBAd residues 23–280. The hydrophobic loop deletion mutant of IcaBAd23–280 (IcaBAd23–280Δloop) was generated in one step using the same protocol as described above for IcaBSe30–289Δloop using the forward and reverse primers CATCGTGTTCTGCCGAGCAGCCGTTATGCAATTAGC and GCTAATTGCATAACGGCTGCTCGGCAGAACACGATG. The D114N and H44A mutants of IcaBAd23–280Δloop were generated in the same manner as described above for IcaBSe30–289 using the forward and reverse primers GCTTGTACTGGTTACCTTTAATGATGGTGATCTGAGCGand CGCTCAGATCACCATCATTAAAGGTAACCAGTACAAGC and GTTCTGTGTTATGCTCGTGTTCTGCCGAG and CTCGGCAGAACACGAGCATAACACAGAAC, respectively.
All the IcaBAd constructs were expressed and purified as described for IcaBSe with the following modifications; (i) cultures were moved prior to and induced at 18 °C for 16 h before being harvested; (ii) for the purification of IcaBAd23–280 the lysis buffer, buffer A, and buffer B contained 8, 4, and 1 mm CHAPS detergent, respectively; (iii) tris(2-carboxyethyl) phosphine was included in the lysis buffer and buffer A; (iv) 2 mm 2-mercaptoethanol was included in buffer B for fluorescamine assays; (v) buffer C (Tris, pH 7.0, 0.6 m NaCl, and 5 mm DTT) was used during gel-filtration chromatography purification of IcaBAd23–280Δloop for crystallization trials. The purified IcaBAd constructs were >95% pure as judged by SDS-PAGE and stable for ∼2 weeks at 4 °C.
Crystallization, Data Collection, and Structure Solution
Purified IcaBAd23–280Δloop was concentrated to ∼9–10 mg/ml and screened for crystallization conditions at 20 °C using hanging-drop vapor diffusion in 48-well VDX plates (Hampton Research) and the MCSG 1–4 sparse matrix suites (Microlytic). An initial crystallization hit was obtained in condition #72 from the MCSG-1 suite (30% (w/v) PEG 2000 monomethyl ether and 0.1 m potassium thiocyanate). Homemade grid-optimized screens failed to reproduce crystals, so a stock solution of MCSG-1 #72 was purchased from Microlytic. The entire 48-well plates were setup using the stock solution, and on average 3–5 drops per plate produced crystals in 1 month. One irregular shaped crystal (crystal 1) with dimensions of 50 × 50 × 20 μm was suitable for diffraction studies. After four months of incubation of the initial crystallization trials, bipyramidal-shaped crystals with dimensions of 60 × 30 × 30 μm also formed in condition #24 from the MCSG-1 suite (28% (w/v) PEG 2000 monomethyl ether, 0.1 m bis(2-hydroxyethyl)amino-tris(hydroxymethyl) methane (Bis-Tris), pH 6.5) and were suitable for diffraction studies (crystal 2). The crystals were cryoprotected for 5 s in reservoir solution supplemented with 20% (v/v) ethylene glycol before vitrification in liquid nitrogen. All data were collected on beam line X29 at the National Synchrotron Light Source (Table 1). A total of 360 images of 1° oscillation were collected for crystal 1 and crystal 2 on an ADSC Quantum-315r detector with a 300- or 260-mm crystal-to-detector distance, respectively, and an exposure time of 0.4 s per image. The data were indexed, integrated, and scaled using HKL2000 (33). The structure was determined for crystal 1 by molecular replacement with PHENIX AutoMR (34) using a truncated version of the N-terminal domain of PgaB42–655 (PDB ID 4F9D) as a search model with residues 43–48, 56–70, 146–160, 191–222, 276–288, and 310–646 removed from the model. The resulting electron density map enabled PHENIX AutoBuild (34) to build ∼40% of the protein. The remaining residues were built manually in COOT (35) and alternated with refinement using PHENIX.REFINE (34). The model from crystal 1 was then used to solve the structure of crystal 2 using molecular replacement, with manual model building and refinement as describe previously. Translation/Libration/Screw (TLS) groups were used during refinement and determined automatically using the TLSMD web server (36, 37). Structure figures were generated using the PyMOL Molecular Graphics System (DeLano Scientific), and quantitative electrostatics were calculated using PDB2PQR (38, 39) and APBS (40). Programs used for crystallographic data processing and analysis were accessed through SBGrid (41).
TABLE 1.
Summary of data collection and refinement statistics
Values in parentheses correspond to the highest resolution shell.
| IcaBAd23–280Δloop Crystal 1 | IcaBAd23–280Δloop Crystal 2 | |
|---|---|---|
| Data collection | ||
| Beamline | NSLS X29 | NSLS X29 |
| Wavelength (Å) | 1.075 | 1.075 |
| Space group | I4 | I4 |
| Unit-cell parameters (Å) | a = b = 84.6, c = 71.4 | a = b = 84.5, c = 71.9 |
| Resolution (Å) | 50.00-2.25 (2.35-2.25) | 50.00-1.70 (1.76-1.70) |
| Total no. of reflections | 173,478 | 411,607 |
| No. of unique reflections | 12,020 | 28,034 |
| Redundancy | 14.7 (10.6) | 14.7 (14.6) |
| Completeness (%) | 98.1 (84.7) | 100 (100) |
| Average I/σ(I) | 16.6 (2.8) | 60.5 (5.1) |
| Rmerge (%)a | 17.6 (59.5) | 8.8 (57.8) |
| Refinement | ||
| Rworkb/Rfreec | 20.3/23.1 | |
| No. of atoms | ||
| Protein | 1861 | |
| Zn2+ | 1 | |
| Chloride | 1 | |
| Water | 132 | |
| Average B-factors (Å2)d | ||
| Protein | 45.0 | |
| Zn2+ | 31.9 | |
| Chloride | 39.6 | |
| Water | 44.2 | |
| Root mean square deviation | ||
| Bond lengths (Å) | 0.006 | |
| Bond angles (°) | 1.09 | |
| Ramachandran plotd | ||
| Total favored (%) | 96.0 | |
| Total allowed (%) | 99.1 | |
| Disallowed (%) | 0.9 | |
| Coordinate error (Å)e | 0.22 | |
| PDB code | 4WCJ | |
a Rmerge = ΣΣ|I(k) − 〈I〉|/ΣI(k), where I(k) and 〈I〉 represent the diffraction intensity values of the individual measurements and the corresponding mean values. The summation is over all unique measurements.
b Rwork = Σ‖Fobs| − k|Fcalc‖/|Fobs|, where Fobs and Fcalc are the observed and calculated structure factors, respectively.
c Rfree is the sum extended over a subset of reflections (7%) excluded from all stages of the refinement.
d As calculated using MolProbity (62).
e Maximum-Likelihood Based Coordinate Error, as determined by PHENIX (34).
X-ray Absorption Spectroscopy Metal Analysis
X-ray absorption spectroscopy (XAS) was carried out on IcaBAd23–280Δloop crystal 1 on beam line X29, National Synchrotron Light Source. Two datasets were collected, at wavelengths of 1.2698 and 1.2964 Å, above and below the Zn2+ absorption edge, respectively. The data were collected, indexed, integrated, and scaled as described above. Anomalous electron density maps were generated for both datasets using sfall in the CCP4 suite (42).
Docking Studies
The binding of β-1,6-(GlcNAc)3 and β-1,6-(GlcNAc)5 were investigated using AutoDock Vina 1.1.2 (43). The structure files for β-1,6-(GlcNAc)3 and β-1,6-(GlcNAc)5 were constructed using the web-based Glycam Biomolecule Builder (44). Receptor and ligand Protein Data Bank files were prepared for docking using scripts provided from MGLTools 1.5.6 (45). Docking was conducted with a grid spacing of 0.37 and xyz of 23 × 19 × 18 using a rigid receptor with 10 poses computed. The top productive pose (N-Ac group bound to the zinc ion) for β-1,6-(GlcNAc)3 and β-1,6-(GlcNAc)5 had scores of −7.2 kcal/mol and −6.0 kcal/mol, respectively.
Preparation of PNAG Oligomers
β-1,6-GlcNAc oligomers were synthesized and purified, and their identities were confirmed as outlined previously (46). The β-1,6-GlcNAc oligomers were stored as lyophilized powders at room temperature and dissolved with deionized water for use in assays. Accurate oligomer concentrations were determined by 1H NMR using dimethylformamide as an internal standard.
Fluorescamine Enzyme Activity Assays
The de-N-acetylation activity of various IcaB constructs and mutants on PNAG oligomers was determined by labeling the free amine groups produced during catalysis using a fluorescamine-based assay (47). The assay was performed as described previously (32) with the following modification; BRAND black 96-well plates were used for fluorescence measurements using a SpectraMax M2 plate reader from Molecular Devices (Sunnyvale, CA). For the metal-preference assay, IcaBAd23–280Δloop (30 μm) in buffer B was preincubated in the presence of various divalent metals as chloride salt solutions (30 μm) or metal chelators (1 mm) at room temperature for 30 min. The mixture was then incubated at 37 °C for 24 h with 50 mm PNAG tetramer and pentamer oligomer mixture, and the amount of de-N-acetylation was quantified using the fluorescamine assay. For the pH dependence of the enzymes, reactions were measured between pH 5.0 and 9.0 using a three-component MES-HEPES-borate buffer system (100 mm) and NaCl (500 mm). Reactions were conducted at 37 °C using 30 μm enzyme, 30 μm CoCl2 or NiCl2, and 50 mm PNAG tetramer and pentamer oligomer mixture. A glucosamine standard curve was used to quantify the observed de-N-acetylation activity.
Membrane Binding Assays
Staphylococcus carnosus was grown in 1 liter of LB media at 37 °C for 16 h, and cells were harvested at 5000 × g for 20 min. The cell pellet was resuspended and digested with 20 mg/ml lysozyme in 25 ml of buffer D (50 mm HEPES, pH 8.0, 300 mm NaCl, and 2 mm tris(2-carboxyethyl) phosphine) for 90 min at 25 °C. Digested cells were then further disrupted with five passes through an Emulsiflex-c3 at 25,000 p.s.i., and cell debris was removed by centrifugation at 31,000 × g for 30 min. Membranes were then isolated by centrifugation at 200,000 × g for 1 h. Total membranes were weighed and resuspended using a hand press in buffer D with the addition of a protease tablet to a concentration of 75–80 mg/ml. For each reaction a 500 μl volume was used with 10 μm concentrations of protein incubated with ∼35 mg of membranes in buffer D. The reactions were incubated for 2 h at 4 °C, and membrane fractions were isolated by centrifugation at 100,000 × g for 45 min. Supernatants were removed, and the pellets were washed 3 times and then resuspended with 500 μl buffer D. The supernatant and resuspended pellet fractions were analyzed by Western blot using α-His6 conjugated to alkaline phosphatase (Sigma) and developed with nitro blue tetrazolium chloride/5-bromo-4-chloro-3′-indolyphosphate p-toluidine salt reagent (Thermo Scientific Pierce).
RESULTS
Generation of Stable IcaB Constructs
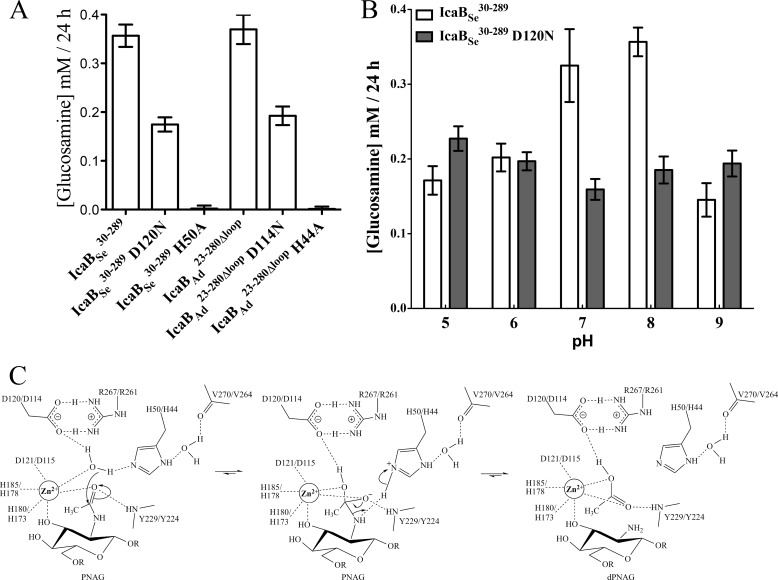
The production and purification of IcaB from Gram-positive bacteria for functional characterization purposes has proven to be challenging. Our group recently reported the generation of the first stable soluble IcaB construct from S. epidermidis (32) using a codon-optimized gene, induction and expression at 10 °C, and with high ionic strength buffers present during the purification process. Despite this success, excessive degradation of the protein occurred during the long induction period, and the protein could not be concentrated, making it unsuitable for structural studies. By reducing the induction period to 2–3 h at 25 °C and using 1 m NaCl in the purification buffers we have been able to reduce protein degradation and concentrate IcaBSe30–289 to >5 mg/ml. Unfortunately this protein was recalcitrant to crystallization. A survey of IcaB homologues from Staphylococcus, Lactobacillus, and Bacillus spp. revealed that they all contain high lysine content (8–15%), which is unfavorable for crystallization. To find an IcaB homologue more suitable for crystallization, an in-depth search using the STRING database (48) for bacterial species with the icaABCD operon was performed. This search identified IcaB from A. degensii, which has low lysine (4%) and high arginine (8%) content. An IcaBAd23–280 construct lacking the predicted signal sequence was, therefore, constructed and found to fractionate with E. coli membranes during purification. Although the protein could be purified when detergent was added to the buffers, it was also recalcitrant to crystallization. Examination of multiple sequence alignments revealed a predicted hydrophobic loop that was only conserved within Gram-positive homologues of IcaB (i.e. not within PgaB) (Fig. 1A). We predicted that the removal of this loop might improve the stability and solubility of the enzymes and in turn make the protein more amenable to crystallization. Deletion of the predicted hydrophobic loop, residues 56–69 and 50–64 in IcaBSe and IcaBAd, respectively, did not improve the solubility of IcaBSe30–289Δloop but greatly improved solubility characteristics of IcaBAd23–280Δloop. To determine whether the proposed hydrophobic rich loop was involved in de-N-acetylation, fluorescamine assays using PNAG oligomers were completed on all four enzyme constructs. These assays showed no significant differences in activity between IcaBSe30–289, IcaBSe30–289Δloop, IcaBAd23–280, and IcaBAd23–280Δloop (Fig. 1B), suggesting that the loop does not play a role in catalysis.
FIGURE 1.
A hydrophobic loop in IcaB is not required for PNAG de-N-acetylation. A, sequence alignment of IcaB from A. degensii KC4 (IcaBAd), S. epidermidis RP62A (IcaBSe), S. aureus MRSA252 (IcaBSa), Lactococcus lactis Il1403 (IcaBLl), Bacillus megaterium WSH-002 (IcaBBm), and E. coli K-12 PgaB (PgaBEc) reveals the presence of a conserved hydrophobic loop in Gram-positive homologues (gray box). Residue numbers are listed before and after the respective sequence. The ★ and ● represent the catalytic histidine and active site pocket residues conserved only within Gram-positive homologues, respectively. B, fluorescamine assays comparing the de-N-acetylation activity of IcaBSe30–289, IcaBSe30–289Δloop, IcaBAd23–280, and IcaBAd23–280Δloop suggest that the hydrophobic loop is not required for PNAG de-N-acetylation. Bars represent triplicate experiments with S.D.
IcaBAd23–280Δloop Is a (β/α)7 Barrel with Unique Structural Features
To gain insight into the structure of IcaB, IcaBAd23–280Δloop was subjected to crystallization trials. Crystals were obtained from two different crystallization conditions, and diffraction data were collected to 2.25 and 1.7 Å (crystal 1 and 2, respectively, Table 1). The structure of crystal 1 was solved using molecular replacement with E. coli PgaB as the model template (PDB ID 4F9D). Crystal 1 of IcaBAd23–280Δloop crystallized in the tetragonal space group I4 with one molecule in the asymmetric unit. Crystal 2 originally indexed in the space group P42 and could be solved by molecular replacement using the model from crystal 1. In this space group two IcaB molecules are present in the asymmetric unit. However, density for the second molecule was poor, and the R factors could not be refined below 30%. Patterson analysis revealed significant pseudo-translational symmetry with an off-origin peak 96% of the origin peak located at (x,y,z) = (0.5,0.5,0.5) suggesting the true space group was I4. Re-indexing the dataset in space group I4 produced excellent density maps with one molecule per asymmetric unit and a drop in R factors by ∼10%. Refinement of crystal 2 produced a final model with good geometry and R factors of 20.3% (Rwork) and 23.1% (Rfree) (Table 1). Residues 23–30, 278–280, and the hexahistidine tag were not included in the final model as there was no interpretable electron density present.
IcaBAd23–280Δloop adopts a (β/α)7 barrel fold common to CE4s (49) (Fig. 2A). The core of the structure is composed of seven parallel β-strands arranged in a barrel that is surrounded by seven α-helices with one of the helices capping the bottom of the barrel (Fig. 2A). Six loops are present on the top face of the (β/α)7 barrel (the C termini of the β-strands) and are important for active site architecture. In addition to the (β/α)7 core, three β-hairpins are located in loops L1, L3, and L4, which contain residues important for the formation of the active site groove (Fig. 2A). The deleted hydrophobic loop in IcaBAd23–280Δloop is located in loop L1 between Ser-49 and Ser-65 (Fig. 2) and would result in a long structural extension not required for folding the (β/α)7 barrel. Electrostatic potential analysis of IcaBAd23–280Δloop reveals two distinct features; first, is an electronegative active site pocket that is along the top of the (β/α)7 barrel (Fig. 2B). Second, is an electropositive patch located on one side of the (β/α)7 barrel near loops L1 and L2, in close proximity to where the deleted hydrophobic loop would extend (Fig. 2B).
FIGURE 2.
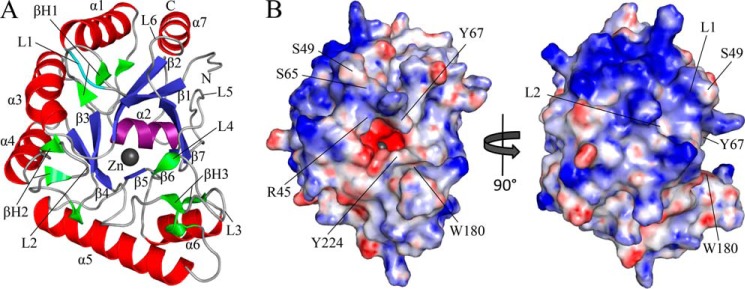
Structure and electrostatic surface representation of IcaBAd23–280Δloop. A, IcaBAd23–280Δloop shown in the schematic with the canonical (β/α)7 barrel fold labeled β1–7 and α1–7 colored blue (β-strands) and red (α-helices), respectively, except for the “capping” helix colored purple located on the bottom of the barrel. Additional secondary structure elements are colored green with β-hairpins labeled βH1–3, loops labeled L1–6 and colored light gray, and the Zn2+ ion colored dark gray and shown as a sphere. N and C termini are labeled accordingly, and the site where the hydrophobic loop was deleted is colored cyan. B, electrostatic surface representation of IcaBAd23–280Δloop shown in the same orientation as panel A and rotated 90° to the right show an electronegative active site pocket and a conserved electropositive patch adjacent to L1 and L2. Quantitative electrostatics are colored from red (−10 kT/e) to blue (+10 kT/e).
IcaBAd23–280Δloop Active Site Metal Analysis Suggests an Ordered Zn2+
Examination of IcaBAd23–280Δloop difference electron density maps revealed a large 28σ peak at the top of the (β/α)7 barrel, suggesting the presence of a metal ion. The metal ion is coordinated in an octahedral manner by the side chains of Asp-115, His-173, His-178, and three water molecules (Fig. 3A). The arrangement of the Asp-His-His residues is characteristic of the metal coordination found in most CE4s. To determine the identity of the metal, anomalous diffraction data were collected on IcaBAd23–280Δloop crystal 1 above and below the absorption edge for the transition metal Zn2+, as it is the most common metal cofactor found in CE4 structures. A significant decrease in anomalous signal was observed for the 28σ peak for the data collected below the Zn2+ absorption edge (1.2964 Å) (Fig. 3B), confirming the identity of Zn2+ as the metal ion.
FIGURE 3.
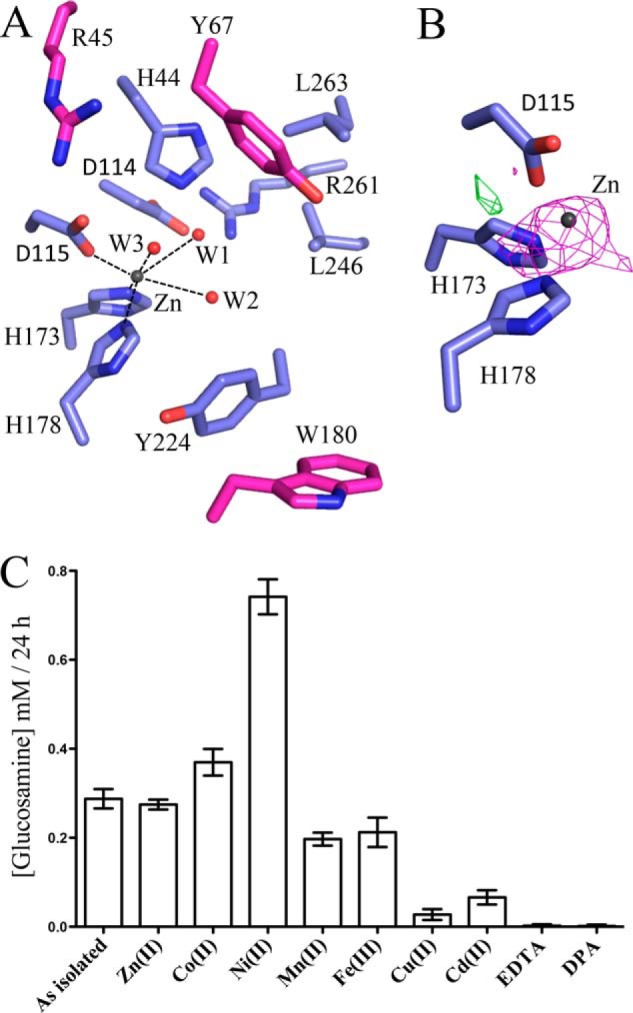
IcaBAd23–280Δloop active site and metal-dependent activity. A, the active site of IcaBAd23–280Δloop is shown in stick representation with the canonical CE4 motif residues, binding groove residues, and the Zn2+ ion colored blue, magenta, and dark gray, respectively. B, metal coordinating triad of IcaBAd23–280Δloop shown in stick representation (blue) with the anomalous difference maps above (magenta) and below (green) the Zn2+ absorption edge shown as a mesh and contoured at 2.0σ. C, fluorescamine de-N-acetylation activity assay comparison of IcaBAd23–280Δloop (as isolated) and incubated with various divalent metal chlorides or metal chelators. DPA, dipicolinic acid. Bars represent triplicate experiments with S.D.
Previous metal-dependent activity studies on IcaBSe30–289 had shown a preference for Zn2+ and Co2+ in PNAG de-N-acetylation. To determine whether IcaBAd23–280Δloop shows the same metal preference as IcaBSe30–289, metal-dependent fluorescamine assays were conducted (Fig. 3C). IcaBAd23–280Δloop showed similar levels of activity compared with the as-isolated enzyme in the presence of Zn2+, Mn2+, and Fe3+ and increases of ∼1.3- and 2.3-fold in the presence of Co2+ and Ni2+, respectively. Enzyme activity was abolished when incubated with 1 mm EDTA or dipicolinic acid.
Structural Comparison of IcaBAd and PgaB Reveals the Basis for PNAG Binding
Structural alignment of IcaBAd23–280Δloop with the N-terminal domain of PgaB, residues 43–309 (PgaB43–309, PDB ID 4F9D), shows strong conservation of the CE4 domain with a root mean square deviation of 1.7 Å over 219 equivalent Cα atoms (Fig. 4A). Two topological differences were found between IcaBAd23–280Δloop and PgaB43–309. First, α7 of IcaBAd23–280Δloop is tilted 105° relative to the equivalent α-helix in PgaB43–309 (Fig. 4A). Second, IcaBAd23–280Δloop only has three β-hairpin motifs (βH1–3), and the loops they reside in (L1, L3, and L4) are much shorter than the equivalent loops in PgaB43–309 (Fig. 4A). The difference in loop architecture alters the architecture of the active site binding pocket (Fig. 4, B and C). Residues Tyr-67 and Trp-180 narrow the active site pocket to 10 Å in IcaBAd23–280Δloop as compared with 16 Å in PgaB43–309 (Met-67 and Gly-191) (Fig. 4, B and C). Furthermore, Arg-45 in IcaBAd23–280Δloop is located at the beginning of the cleft at the −1 subsite and would be available for hydrogen bonding to PNAG. The equivalent residue in PgaB43–309 is an asparagine (Fig. 1A) that is buried in the structure by 3 aromatic residues Phe-66, Tyr-117, and Phe-154.
FIGURE 4.
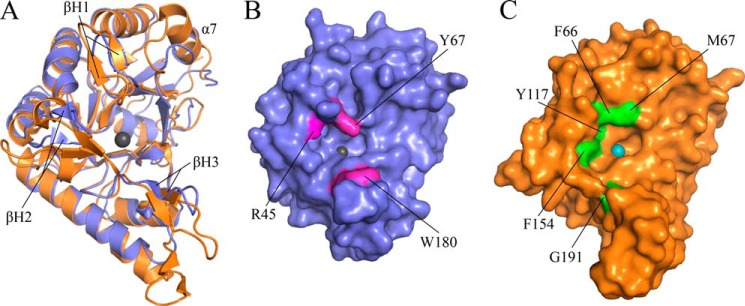
IcaBAd has a narrow active site pocket compared with PgaB43–309. A, superposition of IcaBAd23–280Δloop (blue) and PgaB43–309 (orange) shows a strong conservation of the CE4 fold with a root mean square deviation of 1.7 Å over 219 equivalent Cα atoms. β-Hairpin loops for IcaBAd are labeled βH1–3, and the Zn2+ ion is shown as a sphere colored dark gray. Surface representation of IcaBAd23–280Δloop (B) and PgaB43–309 (C) is shown in the same representation as panel A with residues contributing to the difference in active site architecture colored magenta and green for IcaBAd23–280Δloop and PgaB43–309, respectively. The Ni2+ ion in PgaB is shown as a teal-colored sphere.
Docking Studies with PNAG Oligomers Suggest Three GlcNAc Binding Sites
Docking studies with β-1,6-(GlcNAc)3 and β-1,6-(GlcNAc)5 were conducted using IcaBAd23–280Δloop to gain insight into the determinants for PNAG binding. The predicted affinity for β-1,6-(GlcNAc)5 (−6.0 kcal/mol) was lower than β-1,6-(GlcNAc)3 (−7.2 kcal/mol) in the docking studies, and inspection of the poses computed showed that the first and fifth residues of β-1,6-(GlcNAc)5 (subsites −2 and +2) do not make contacts with IcaBAd23–280Δloop. Furthermore, only the top β-1,6-(GlcNAc)3 pose showed hydrogen-bonding contacts with all three N-Ac groups to IcaBAd23–280Δloop and thus was used for further analysis. The docked β-1,6-(GlcNAc)3 binds IcaBAd23–280Δloop along the active site groove occupying subsites −1, 0, and +1 (Fig. 5A). GlcNAc in the −1 subsite has hydrogen bonds between Arg-45 and Glu-149 to the C5 oxygen (3.6 Å) and N-Ac nitrogen (2.9 Å), respectively (Fig. 5B). GlcNAc in the 0 subsite makes contacts with the Zn2+ ion via the hydroxyl at C3 (OH3) (2.2 Å) and the N-Ac carbonyl (2.3 Å), Asp-114 and OH3 (2.7 Å), Arg-45 and OH4 (3.6 Å), and stacking interactions with Tyr-67 (Fig. 5B). GlcNAc in the +1 subsite has hydrogen bonds between the backbone carbonyl of Tyr-224 and both the OH3 (3.6 Å) and the N-Ac carbonyl (3.5 Å) and T-shaped stacking interactions with Trp-180 (Fig. 5B). Interestingly, the N-Ac carbonyl and OH3 of the 0 subsite GlcNAc are in close proximity to the crystallographic waters, W2 and W3, with distances of 0.6 and 0.2 Å, respectively (Fig. 5B), This observation reinforces the confidence of the docking studies and the positioning of the 0 subsite GlcNAc during catalysis.
FIGURE 5.
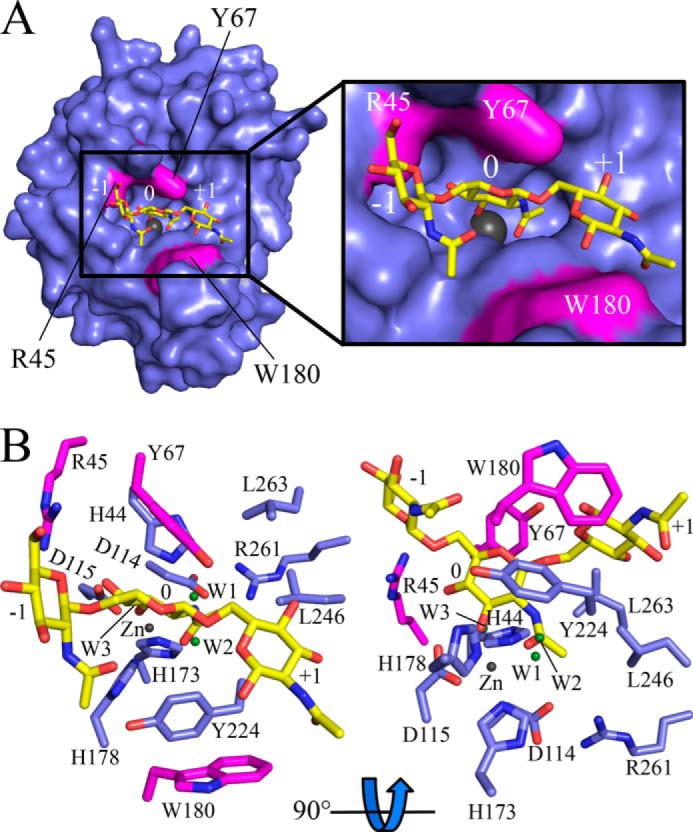
Docking of PNAG oligomers. A, IcaBAd23–280Δloop (blue) with a β-1,6-(GlcNAc)3 (yellow) docked in the active site shown in surface and stick representations, respectively. The model predicts that Arg-45, Tyr-67, and Trp-180 (magenta) are important for the binding of β-1,6-(GlcNAc)3. B, active site close-up of the docked β-1,6-(GlcNAc)3 shows the central sugar binds to the Zn2+ ion and the crystallographic waters (W2 and W3, green spheres) overlap with the N-Ac carbonyl and OH-3 oxygens. The Zn2+ ion is shown as a dark gray colored sphere, and the GlcNAc subsites are labeled from the non-reducing to reducing end as −1, 0, and +1 in both panels.
Mutational Analysis of IcaB Suggests an Altered CE4 Catalytic Mechanism
CE4s have been proposed to catalyze the deacetylation of their substrates using a metal-assisted general acid and base mechanism (30). The first conserved aspartate found in CE4 motif 1, TFDD, is considered the catalytic base. The conserved histidine in CE4 motif 5, LXH, is proposed to be the catalytic acid. From structural and mutational studies on the Streptococcus pneumoniae peptidoglycan deacetylase PgdA (30) and the Vibrio cholerae chitin deacetylase CDA (31), we hypothesized that His-50/His-44 and Asp-120/Asp-114 are the catalytic acid and base in IcaBSe/IcaBAd, respectively. To assess the role of these residues in the catalytic mechanism H50A/H44A and D120N/D114N variants were assayed for de-N-acetylation activity. The histidine variants were completely inactive (Fig. 6A), suggesting this conserved residue plays a key role in catalysis. However, the IcaBSe and IcaBAd aspartate variants displayed 53 and 56% of wild type activity, respectively (Fig. 6A), suggesting these residues do not serve essential roles in the catalytic mechanism. Asp-120/Asp-114 may actually be involved in tuning the electrostatics of the active site as the asparagine variants lose their pH activation profile (Fig. 6B). To identify a residue in the active site pocket that could potentially serve as the base in the reaction, we used the pKa prediction server PROPKA3 (50). The predicted pKa of His-50/His-44 is ∼3, suggesting it is basic under the assay conditions and could potentially have a bifunctional role in catalysis (Fig. 6C).
FIGURE 6.
Mutational analysis and proposed catalytic mechanism. A, fluorescamine de-N-acetylation activity comparison of wild type IcaBSe and IcaBAd, and their D120N/D114N and H50A/H44A variants. B, pH dependence of the fluorescamine de-N-acetylation activity of wild type IcaBSe30–289 and its D120N variant. C, proposed catalytic mechanism for IcaB-dependent de-N-acetylation of PNAG. Bars represent triplicate experiments with S.D.
The Conserved Hydrophobic Loop of IcaB Is Involved in Membrane Association
The presence of a solvent-accessible hydrophobic loop in Gram-positive homologues of IcaB suggests it may have a functional importance. Given that these proteins are found on the extracellular surface of the bacteria, we hypothesized that the loop may play a role in membrane localization. IcaBSe30–289, IcaBSe30–289Δloop, IcaBAd23–280, and IcaBAd23–280Δloop were, therefore, assayed for their ability to bind and associate with staphylococcal membranes. Using a membrane pulldown assay, IcaBSe30–289 and IcaBAd23–280 co-fractioned into the pellet when incubated with staphylococcal membranes (Fig. 7A). IcaBAd23–280Δloop was only present in the soluble fraction, and IcaBSe30–289Δloop had significantly reduced quantities found in the insoluble fraction (Fig. 7A). These data suggest that the hydrophobic loop plays a role in binding and associating with the membrane. Structural modeling of the hydrophobic loop in IcaBSe30–289 using Phyre2 (51) predicts an α-helix spanning residues 59–66 that is amphipathic, with Phe-61, Ile-62, Leu-64, and Leu-65 lying on the solvent exposed face (Fig. 7B). A similar result was also seen for Trp-54, Gly-55, Leu-57, and Phe-58 in IcaBAd23–280.
FIGURE 7.
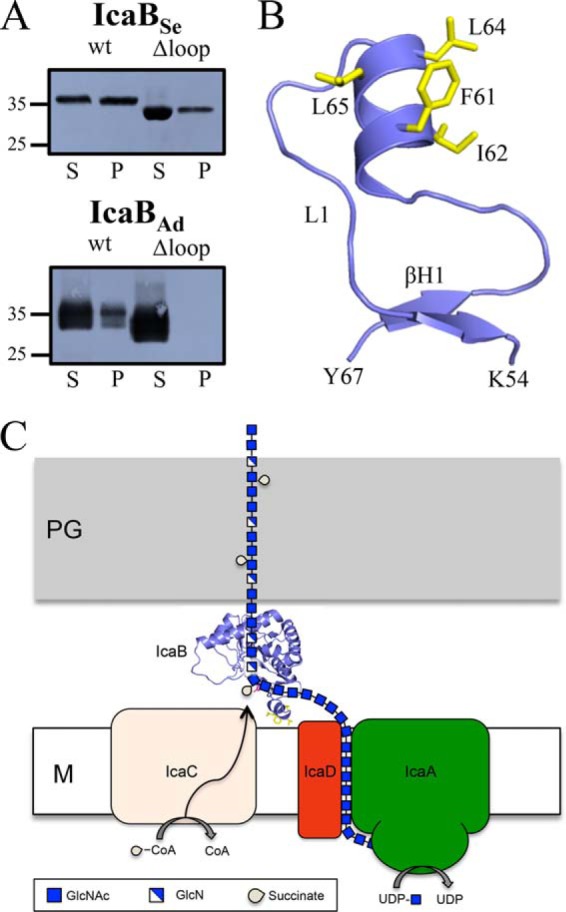
Membrane localization of IcaB. A, staphylococcal membrane pulldown assays of purified IcaB proteins show the hydrophobic loop is important for membrane association. S, supernatant; P, pellet; wt, wild type. Molecular mass markers are indicated on the left side of the blots in kDa. B, predicted structure for the IcaBSe hydrophobic loop reveals an amphipathic α-helix. C, proposed model for PNAG biosynthesis and modification. IcaB associates with the membrane via its hydrophobic loop and electropositive surface and de-N-acetylates PNAG after export. M, membrane; PG, peptidoglycan; CoA, coenzyme A.
DISCUSSION
IcaB has been identified as an attractive target for the design of inhibitors to combat biofilm-related infections by S. epidermidis and S. aureus for two reasons. First, it is an extracellular protein (26), which eliminates the need for membrane permeable drugs. Second, it is responsible for PNAG de-N-acetylation, which is required for biofilm formation and virulence in animal models (26, 27). Our recent study of IcaBSe30–289 was the first functional characterization of its metal- and length-dependent de-N-acetylation activity (32). IcaBSe and its Gram-negative homologue PgaB have the same enzymatic function but displayed different metal- and length-dependent activity on PNAG oligomers (28, 32). Additionally, the domain architecture and localization of the two proteins are drastically different. IcaBSe is a single-domain extracellular protein, whereas PgaB is a two-domain periplasmic lipoprotein (52, 53).
IcaB contains a hydrophobic loop that is conserved only in Gram-positive homologues (Fig. 1A). This hydrophobic loop is not required for de-N-acetylation of PNAG oligomers (Fig. 1B) but likely contains an amphipathic α-helix that can bind to staphylococcal membranes (Fig. 7). This type of amphipathic α-helix has been found in other integral or associated membrane proteins (54, 55) and is likely, at least in part, responsible for the retention of IcaB to the cell surface. Furthermore, the electropositive patch found on the structure of IcaBAd23–280Δloop (Fig. 2B) would be well suited to interact with the negatively charged phosphate groups on the phospholipids and lipoteichoic acids. This correlates well with previous in vivo data that shows IcaBSe as a membrane-associated extracellular protein (26).
IcaBAd23–280Δloop contains an Asp-His-His triad that coordinates an ordered Zn2+ ion in the structure (Fig. 3B). IcaBAd23–280Δloop displays metal-dependent de-N-acetylation of PNAG oligomers with a preference for Ni2+, Co2+, and Zn2+ (Fig. 3C). PgaB displayed the highest activity with Co2+ and Ni2+ cofactors (28), whereas IcaBSe30–289 had the highest activity with Co2+ and Zn2+ (32). The metal ion promiscuity in IcaB appears to be a continuing trend for the CE4 family. IcaBAd23–280Δloop shows structural similarity to PgaB43–309 and contains the same circularly permuted arrangement of the CE4 motifs, a feature unique to these CE4 members (28, 56). Despite the high structural similarity and metal ion preference between IcaB and PgaB43–309, only IcaB is enzymatically active on PNAG oligomers; PgaB requires both N- and C-terminal domains for de-N-acetylation activity (29). Structural studies on CDA and comparison to known CE4 structures reveals that loop architecture plays a major role in substrate binding and the enzymatic mechanism (31). The loop architecture of IcaBAd23–280Δloop is similar to other CE4s, like PgdA (30), which has a narrow binding pocket. Additionally, the conserved residues Arg-45, Tyr-67, and Trp-180 make important interactions with the three GlcNAc moieties at −1, 0, and +1 subsites, respectively, in the docking studies (Fig. 5). PgaB43–309 does not contain any of these conserved elements (Fig. 4, A and C), likely leading to loss of PNAG binding and thus rendering the isolated domain inactive. Previous docking and simulation studies have suggested that conserved elements, including a conserved arginine and tryptophan, on PgaB310–672 are required for PNAG binding and catalysis in the E. coli enzyme (29). The cleft formed between the two domains of PgaB also creates a long extended active site (29). This correlates with the enzymatic data that shows length-dependent de-N-acetylation of the oligosaccharides up to β-1,6-(GlcNAc)5 with preference for the central GlcNAc (28). The structure of IcaBAd23–280Δloop suggests only three GlcNAc subsites within the active site pocket (Fig. 5). This is supported by previous studies with IcaBSe30–289 that showed similar de-N-acetylation of β-1,6-(GlcNAc)3 to β-1,6-(GlcNAc)6 and displayed deacetylation preference at the second GlcNAc from the reducing end (32). The involvement of PgaB310–672 during de-N-acetylation appears to be part of a mechanism for PgaB to continuously associate with the dPNAG polymer (29). This type of processive mechanism would produce an energetically favorable extended state of dPNAG to allow for efficient periplasmic translocation and hand-off to the export machinery (29). Because IcaB is an extracellular protein it does not need an auxiliary domain for continual processing and export of dPNAG. It is probable that IcaB will interact with dPNAG multiple times as they both are present in the extracellular matrix. This is consistent with the higher levels of deacetylation observed for S. epidermidis (∼15–20%) over E. coli (∼3–5%) in dPNAG isolated from source even though IcaB displays lower levels of activity than PgaB in vitro. The circular permutation of the CE4 motifs in IcaB and PgaB must play a role other than substrate binding during catalysis. PgaB has been proposed to operate through a similar enzymatic mechanism to other CE4 members (57) but is lacking a key catalytic residue that has been proposed to attenuate its catalytic efficiency (28). Our preliminary mutational analysis suggests Asp-120/Asp-114 is not the catalytic base as the equivalent D275N variant in PgdA inactivates the enzyme. In the IcaBAd23–280Δloop structure Asp-114 is in a bi-dentate salt bridge with Arg-261 (Asp-120 and Arg-267 in IcaBSe), which would prevent a free carboxylate group that could serve as the base. The equivalent salt bridge in both PgdA and CDA are not bi-dentate as the conserved arginine spans the active site in a different orientation. This would free the aspartate carboxylate group to abstract a proton from the nucleophilic water. Thus, we propose His-50/His-44 to be a bifunctional acid base catalyst in the enzymatic mechanism (Fig. 6C). In our proposed scheme, hydrolysis of the 0 subsite GlcNAc N-Ac group would be initiated by the Zn2+-coordinated water molecule. Deprotonation of the water by His-50/His-44 would lead to nucleophilic attack of the N-Ac carbonyl group to produce a tetrahedral oxyanion intermediate that would be stabilized by the Zn2+ ion and the backbone amide of Tyr-229/Tyr-224. The intermediate would then be protonated on the N-Ac nitrogen by the imidazolium of His-50/His-44. This would generate a free amine leaving group (dPNAG) and the acetic acid byproduct. Diffusion of acetic acid out and water into the active site would regenerate the enzyme and lead to another round of de-N-acetylation on the dPNAG polymer. Similar bifunctional roles have been proposed for a glutamic acid in the zinc metalloamidases LpxC (58, 59) and carboxypeptidase A (60, 61). Together, these results suggest that the circular permutation of the CE4 motifs in IcaB and its Gram-negative homologue PgaB may result in an altered CE4 enzymatic mechanism.
The structure, functional characterization, and membrane association of IcaB reported here will help facilitate additional studies that probe our proposed model for the biosynthesis and modification of PNAG in Gram-positive bacteria (Fig. 7C) and will help guide the development of novel inhibitors to prevent biofilm-related infection caused by S. epidermidis and S. aureus.
Acknowledgments
We thank Patrick Yip for technical assistance and the Trimble laboratory at The Hospital for Sick Children for the use of their micro-ultracentrifuge. The National Synchrotron Light Source beam line X29A is supported by the United States Department of Energy Office of Biological and Environmental Research and the National Institutes of Health National Centre for Research Resources.
This work was supported by Canadian Institutes of Health Research Grants 43998 (to P. L. H.) and 89708 (to M. N.).
The atomic coordinates and structure factors (code 4WCJ) have been deposited in the Protein Data Bank (http://wwpdb.org/).
- PNAG
- poly-β-1,6-N-acetyl-d-glucosamine
- dPNAG
- de-N-acetylated PNAG
- CE4
- family 4 carbohydrate esterase
- Bis-Tris
- 2-[bis(2-hydroxyethyl)amino]-2-(hydroxymethyl)propane-1,3-diol.
REFERENCES
- 1. O'Gara J. P., Humphreys H. (2001) Staphylococcus epidermidis biofilms: importance and implications. J. Med. Microbiol. 50, 582–587 [DOI] [PubMed] [Google Scholar]
- 2. von Eiff C., Peters G., Heilmann C. (2002) Pathogenesis of infections due to coagulase-negative staphylococci. Lancet Infect. Dis. 2, 677–685 [DOI] [PubMed] [Google Scholar]
- 3. Rohde H., Frankenberger S., Zähringer U., Mack D. (2010) Structure, function, and contribution of polysaccharide intercellular adhesin (PIA) to Staphylococcus epidermidis biofilm formation and pathogenesis of biomaterial-associated infections. Eur. J. Cell Biol. 89, 103–111 [DOI] [PubMed] [Google Scholar]
- 4. Hall-Stoodley L., Costerton J. W., Stoodley P. (2004) Bacterial biofilms: from the natural environment to infectious diseases. Nat. Rev. Microbiol. 2, 95–108 [DOI] [PubMed] [Google Scholar]
- 5. López D., Vlamakis H., Kolter R. (2010) Biofilms. Cold Spring Harb. Perspect. Biol. 2, a000398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sutherland I. (2001) Biofilm exopolysaccharides: a strong and sticky framework. Microbiology 147, 3–9 [DOI] [PubMed] [Google Scholar]
- 7. Vu B., Chen M., Crawford R. J., Ivanova E. P. (2009) Bacterial extracellular polysaccharides involved in biofilm formation. Molecules 14, 2535–2554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Branda S. S., Vik S., Friedman L., Kolter R. (2005) Biofilms: the matrix revisited. Trends Microbiol. 13, 20–26 [DOI] [PubMed] [Google Scholar]
- 9. Sutherland I. W. (2001) The biofilm matrix: an immobilized but dynamic microbial environment. Trends Microbiol. 9, 222–227 [DOI] [PubMed] [Google Scholar]
- 10. Zhao K., Tseng B. S., Beckerman B., Jin F., Gibiansky M. L., Harrison J. J., Luijten E., Parsek M. R., Wong G. C. (2013) Psl trails guide exploration and microcolony formation in Pseudomonas aeruginosa biofilms. Nature 497, 388–391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Mack D., Fischer W., Krokotsch A., Leopold K., Hartmann R., Egge H., Laufs R. (1996) The intercellular adhesin involved in biofilm accumulation of Staphylococcus epidermidis is a linear β-1,6-linked glucosaminoglycan: purification and structural analysis. J. Bacteriol. 178, 175–183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. McKenney D., Pouliot K. L., Wang Y., Murthy V., Ulrich M., Döring G., Lee J. C., Goldmann D. A., Pier G. B. (1999) Broadly protective vaccine for Staphylococcus aureus based on an in vivo-expressed antigen. Science 284, 1523–1527 [DOI] [PubMed] [Google Scholar]
- 13. Wang X., Preston J. F., 3rd, Romeo T. (2004) The pgaABCD locus of Escherichia coli promotes the synthesis of a polysaccharide adhesin required for biofilm formation. J. Bacteriol. 186, 2724–2734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Choi A. H., Slamti L., Avci F. Y., Pier G. B., Maira-Litrán T. (2009) The pgaABCD locus of Acinetobacter baumannii encodes the production of poly-β-1–6-N-acetylglucosamine, which is critical for biofilm formation. J. Bacteriol. 191, 5953–5963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Sloan G. P., Love C. F., Sukumar N., Mishra M., Deora R. (2007) The Bordetella Bps polysaccharide is critical for biofilm development in the mouse respiratory tract. J. Bacteriol. 189, 8270–8276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Conover M. S., Sloan G. P., Love C. F., Sukumar N., Deora R. (2010) The Bps polysaccharide of Bordetella pertussis promotes colonization and biofilm formation in the nose by functioning as an adhesin. Mol. Microbiol. 77, 1439–1455 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Izano E. A., Sadovskaya I., Vinogradov E., Mulks M. H., Velliyagounder K., Ragunath C., Kher W. B., Ramasubbu N., Jabbouri S., Perry M. B., Kaplan J. B. (2007) Poly-N-acetylglucosamine mediates biofilm formation and antibiotic resistance in Actinobacillus pleuropneumoniae. Microb. Pathog. 43, 1–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Jarrett C. O., Deak E., Isherwood K. E., Oyston P. C., Fischer E. R., Whitney A. R., Kobayashi S. D., DeLeo F. R., Hinnebusch B. J. (2004) Transmission of Yersinia pestis from an infectious biofilm in the flea vector. J. Infect. Dis. 190, 783–792 [DOI] [PubMed] [Google Scholar]
- 19. Yakandawala N., Gawande P. V., LoVetri K., Cardona S. T., Romeo T., Nitz M., Madhyastha S. (2011) Characterization of the poly-β-1,6-N-acetylglucosamine polysaccharide component of Burkholderia biofilms. Appl. Environ. Microbiol. 77, 8303–8309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Cywes-Bentley C., Skurnik D., Zaidi T., Roux D., Deoliveira R. B., Garrett W. S., Lu X., O'Malley J., Kinzel K., Zaidi T., Rey A., Perrin C., Fichorova R. N., Kayatani A. K., Maira-Litràn T., Gening M. L., Tsvetkov Y. E., Nifantiev N. E., Bakaletz L. O., Pelton S. I., Golenbock D. T., Pier G. B. (2013) Antibody to a conserved antigenic target is protective against diverse prokaryotic and eukaryotic pathogens. Proc. Natl. Acad. Sci. U.S.A. 110, E2209–E2218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Heilmann C., Schweitzer O., Gerke C., Vanittanakom N., Mack D., Götz F. (1996) Molecular basis of intercellular adhesion in the biofilm-forming Staphylococcus epidermidis. Mol. Microbiol. 20, 1083–1091 [DOI] [PubMed] [Google Scholar]
- 22. Gerke C., Kraft A., Süssmuth R., Schweitzer O., Götz F. (1998) Characterization of the N-acetylglucosaminyltransferase activity involved in the biosynthesis of the Staphylococcus epidermidis polysaccharide intercellular adhesin. J. Biol. Chem. 273, 18586–18593 [DOI] [PubMed] [Google Scholar]
- 23. Atkin K. E., MacDonald S. J., Brentnall A. S., Potts J. R., Thomas G. H. (2014) A different path: revealing the function of staphylococcal proteins in biofilm formation. FEBS Lett. 588, 1869–1872 [DOI] [PubMed] [Google Scholar]
- 24. Sadovskaya I., Vinogradov E., Flahaut S., Kogan G., Jabbouri S. (2005) Extracellular carbohydrate-containing polymers of a model biofilm-producing strain, Staphylococcus epidermidis RP62A. Infect. Immun. 73, 3007–3017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Joyce J. G., Abeygunawardana C., Xu Q., Cook J. C., Hepler R., Przysiecki C. T., Grimm K. M., Roper K., Ip C. C., Cope L., Montgomery D., Chang M., Campie S., Brown M., McNeely T. B., Zorman J., Maira-Litrán T., Pier G. B., Keller P. M., Jansen K. U., Mark G. E. (2003) Isolation, structural characterization, and immunological evaluation of a high-molecular-weight exopolysaccharide from Staphylococcus aureus. Carbohydr. Res. 338, 903–922 [DOI] [PubMed] [Google Scholar]
- 26. Vuong C., Kocianova S., Voyich J. M., Yao Y., Fischer E. R., DeLeo F. R., Otto M. (2004) A crucial role for exopolysaccharide modification in bacterial biofilm formation, immune evasion, and virulence. J. Biol. Chem. 279, 54881–54886 [DOI] [PubMed] [Google Scholar]
- 27. Cerca N., Jefferson K. K., Maira-Litrán T., Pier D. B., Kelly-Quintos C., Goldmann D. A., Azeredo J., Pier G. B. (2007) Molecular basis for preferential protective efficacy of antibodies directed to the poorly acetylated form of staphylococcal poly-N-acetyl-β-(1–6)-glucosaminev. Infect. Immun. 75, 3406–3413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Little D. J., Poloczek J., Whitney J. C., Robinson H., Nitz M., Howell P. L. (2012) The structure- and metal-dependent activity of Escherichia coli PgaB provides insight into the partial de-N-acetylation of poly-β-1,6-N-acetyl-d-glucosamine. J. Biol. Chem. 287, 31126–31137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Little D. J., Li G., Ing C., DiFrancesco B. R., Bamford N. C., Robinson H., Nitz M., Pomès R., Howell P. L. (2014) Modification and periplasmic translocation of the biofilm exopolysaccharide poly-β-1,6-N-acetyl-d-glucosamine. Proc. Natl. Acad. Sci. U.S.A. 111, 11013–11018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Blair D. E., Schüttelkopf A. W., MacRae J. I., van Aalten D. M. (2005) Structure- and metal-dependent mechanism of peptidoglycan deacetylase, a streptococcal virulence factor. Proc. Natl. Acad. Sci. U.S.A. 102, 15429–15434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Andrés E., Albesa-Jové D., Biarnés X., Moerschbacher B. M., Guerin M. E., Planas A. (2014) Structural basis of chitin oligosaccharide deacetylation. Angew. Chem. Int. Ed. Engl. 53, 6882–6887 [DOI] [PubMed] [Google Scholar]
- 32. Pokrovskaya V., Poloczek J., Little D. J., Griffiths H., Howell P. L., Nitz M. (2013) Functional characterization of Staphylococcus epidermidis IcaB, a de-N-acetylase important for biofilm formation. Biochemistry 52, 5463–5471 [DOI] [PubMed] [Google Scholar]
- 33. Otwinowski Z., Minor W. (1997) Processing of x-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 [DOI] [PubMed] [Google Scholar]
- 34. Adams P. D., Afonine P. V., Bunkóczi G., Chen V. B., Davis I. W., Echols N., Headd J. J., Hung L. W., Kapral G. J., Grosse-Kunstleve R. W., McCoy A. J., Moriarty N. W., Oeffner R., Read R. J., Richardson D. C., Richardson J. S., Terwilliger T. C., Zwart P. H. (2010) PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Emsley P., Cowtan K. (2004) Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 [DOI] [PubMed] [Google Scholar]
- 36. Painter J., Merritt E. A. (2006) Optimal description of a protein structure in terms of multiple groups undergoing TLS motion. Acta Crystallogr. D Biol. Crystallogr. 62, 439–450 [DOI] [PubMed] [Google Scholar]
- 37. Painter J., Merritt E. A. (2006) TLSMD web server for the generation of multi-group TLS models. J. Appl. Crystallogr. 39, 109–111 [Google Scholar]
- 38. Dolinsky T. J., Czodrowski P., Li H., Nielsen J. E., Jensen J. H., Klebe G., Baker N. A. (2007) PDB ID 2PQR: expanding and upgrading automated preparation of biomolecular structures for molecular simulations. Nucleic Acids Res. 35, W522–W525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Dolinsky T. J., Nielsen J. E., McCammon J. A., Baker N. A. (2004) PDB ID 2PQR: an automated pipeline for the setup of Poisson-Boltzmann electrostatics calculations. Nucleic Acids Res. 32, W665–W667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Baker N. A., Sept D., Joseph S., Holst M. J., McCammon J. A. (2001) Electrostatics of nanosystems: application to microtubules and the ribosome. Proc. Natl. Acad. Sci. U.S.A. 98, 10037–10041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Morin A., Eisenbraun B., Key J., Sanschagrin P. C., Timony M. A., Ottaviano M., Sliz P. (2013) Collaboration gets the most out of software. eLife 2, e01456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Winn M. D., Ballard C. C., Cowtan K. D., Dodson E. J., Emsley P., Evans P. R., Keegan R. M., Krissinel E. B., Leslie A. G., McCoy A., McNicholas S. J., Murshudov G. N., Pannu N. S., Potterton E. A., Powell H. R., Read R. J., Vagin A., Wilson K. S. (2011) Overview of the CCP4 suite and current developments. Acta Crystallogr. D Biol. Crystallogr. 67, 235–242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Trott O., Olson A. J. (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31, 455–461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Woods Group (2005–2014) GLYCAM Web. Complex Carbohydrate Research Center, University of Georgia, Athens, GA [Google Scholar]
- 45. Sanner M. F. (1999) Python: a programming language for software integration and development. J. Mol. Graph. Model. 17, 57–61 [PubMed] [Google Scholar]
- 46. Leung C., Chibba A., Gómez-Biagi R. F., Nitz M. (2009) Efficient synthesis and protein conjugation of β-(1→6)-d-N-acetylglucosamine oligosaccharides from the polysaccharide intercellular adhesin. Carbohydr. Res. 344, 570–575 [DOI] [PubMed] [Google Scholar]
- 47. Udenfriend S., Stein S., Böhlen P., Dairman W., Leimgruber W., Weigele M. (1972) Fluorescamine: a reagent for assay of amino acids, peptides, proteins, and primary amines in the picomole range. Science 178, 871–872 [DOI] [PubMed] [Google Scholar]
- 48. Snel B., Lehmann G., Bork P., Huynen M. A. (2000) STRING: a web-server to retrieve and display the repeatedly occurring neighbourhood of a gene. Nucleic Acids Res. 28, 3442–3444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Blair D. E., van Aalten D. M. (2004) Structures of Bacillus subtilis PdaA, a family 4 carbohydrate esterase, and a complex with N-acetylglucosamine. FEBS Lett. 570, 13–19 [DOI] [PubMed] [Google Scholar]
- 50. Olsson M., Søndergaard C. R. (2011) PROPKA3: consistent treatment of internal and surface residues in empirical pKa predictions. J. Chem. Theory Comput. 7, 525–537 [DOI] [PubMed] [Google Scholar]
- 51. Kelley L. A., Sternberg M. J. (2009) Protein structure prediction on the Web: a case study using the Phyre server. Nat Protoc 4, 363–371 [DOI] [PubMed] [Google Scholar]
- 52. Itoh Y., Rice J. D., Goller C., Pannuri A., Taylor J., Meisner J., Beveridge T. J., Preston J. F., 3rd, Romeo T. (2008) Roles of pgaABCD genes in synthesis, modification, and export of the Escherichia coli biofilm adhesin, poly-β-1,6-N-acetyl-d-glucosamine (PGA). J. Bacteriol. 190, 3670–3680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Little D. J., Whitney J. C., Robinson H., Yip P., Nitz M., Howell P. L. (2012) Combining in situ proteolysis and mass spectrometry to crystallize Escherichia coli PgaB. Acta Crystallogr Sect. F. Struct. Biol. Cryst. Commun. 68, 842–845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Lovering A. L., Lin L. Y., Sewell E. W., Spreter T., Brown E. D., Strynadka N. C. (2010) Structure of the bacterial teichoic acid polymerase TagF provides insights into membrane association and catalysis. Nat. Struct. Mol. Biol. 17, 582–589 [DOI] [PubMed] [Google Scholar]
- 55. Bracey M. H., Hanson M. A., Masuda K. R., Stevens R. C., Cravatt B. F. (2002) Structural adaptations in a membrane enzyme that terminates endocannabinoid signaling. Science 298, 1793–1796 [DOI] [PubMed] [Google Scholar]
- 56. Nishiyama T., Noguchi H., Yoshida H., Park S. Y., Tame J. R. (2013) The structure of the deacetylase domain of Escherichia coli PgaB, an enzyme required for biofilm formation: a circularly permuted member of the carbohydrate esterase 4 family. Acta Crystallogr. D Biol. Crystallogr. 69, 44–51 [DOI] [PubMed] [Google Scholar]
- 57. Little D. J., Bamford N. C., Nitz M., Howell P. L. (2014) Metal-dependent polysaccharide deacetylase PgaB. Encyclopedia of Inorganic and Bioinorganic Chemistry (Scott R.A., ed.) pp. 1–11, John Wiley & Sons, Inc., Chichester, UK [Google Scholar]
- 58. Whittington D. A., Rusche K. M., Shin H., Fierke C. A., Christianson D. W. (2003) Crystal structure of LpxC, a zinc-dependent deacetylase essential for endotoxin biosynthesis. Proc. Natl. Acad. Sci. U.S.A. 100, 8146–8150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Hernick M., Fierke C. A. (2005) Zinc hydrolases: the mechanisms of zinc-dependent deacetylases. Arch. Biochem. Biophys. 433, 71–84 [DOI] [PubMed] [Google Scholar]
- 60. Christianson D. W., David P. R., Lipscomb W. N. (1987) Mechanism of carboxypeptidase A: hydration of a ketonic substrate analogue. Proc. Natl. Acad. Sci. U.S.A. 84, 1512–1515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Christianson D. W., Lipscomb W. N. (1989) Carboxypeptidase A. Acc. Chem. Res. 22, 62–69 [Google Scholar]
- 62. Chen V. B., Arendall W. B., 3rd, Headd J. J., Keedy D. A., Immormino R. M., Kapral G. J., Murray L. W., Richardson J. S., Richardson D. C. (2010) MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 [DOI] [PMC free article] [PubMed] [Google Scholar]