Abstract
Recombination signal binding protein for Ig-κJ region (RBP-J), the major downstream effector of Notch signaling, is necessary to maintain the number of renin-positive juxtaglomerular cells and the plasticity of arteriolar smooth muscle cells to re-express renin when homeostasis is threatened. We hypothesized that RBP-J controls a repertoire of genes that defines the phenotype of the renin cell. Mice bearing a bacterial artificial chromosome reporter with a mutated RBP-J binding site in the renin promoter had markedly reduced reporter expression at the basal state and in response to a homeostatic challenge. Mice with conditional deletion of RBP-J in renin cells had decreased expression of endocrine (renin and Akr1b7) and smooth muscle (Acta2, Myh11, Cnn1, and Smtn) genes and regulators of smooth muscle expression (miR-145, SRF, Nfatc4, and Crip1). To determine whether RBP-J deletion decreased the endowment of renin cells, we traced the fate of these cells in RBP-J conditional deletion mice. Notably, the lineage staining patterns in mutant and control kidneys were identical, although mutant kidneys had fewer or no renin-expressing cells in the juxtaglomerular apparatus. Microarray analysis of mutant arterioles revealed upregulation of genes usually expressed in hematopoietic cells. Thus, these results suggest that RBP-J maintains the identity of the renin cell by not only activating genes characteristic of the myo-endocrine phenotype but also, preventing ectopic gene expression and adoption of an aberrant phenotype, which could have severe consequences for the control of homeostasis.
Keywords: renin, cell fate, juxtaglomerular cell, smooth muscle, hematopoiesis
Juxtaglomerular (JG) cells are crucial for the maintenance of BP and fluid/electrolyte homeostasis. These cells act as sensors that constantly monitor the physiologic status of the animal and convey information to adjacent cells within the JG apparatus and along the renal arterioles. Under normal circumstances, renin secretion by JG cells suffices to maintain homeostasis. However, when there is a more profound physiologic challenge, to restore homeostasis, preexistent smooth muscle (SM) cells along the arterioles, mesangial cells, and interstitial pericytes1 gradually dedifferentiate into renin-producing cells in a pattern resembling the one found in fetal life.2–6
JG cells express a unique set of genes characteristic of both endocrine and SM cells.7 This dual endocrine–contractile phenotype is crucial for the rapid regulation of BP and renal hemodynamics. However, the factors controlling this dual phenotype and the mechanisms that regulate the ability of SM cells along the afferent arterioles to reacquire the renin phenotype have not been fully defined.
JG cells and adjacent cells along the arterioles express all the components of the Notch signaling pathway,7 an ancestral cell–cell communication system involved in cell fate decisions. We hypothesized that the Notch pathway through its major transcriptional effector, recombination signal binding protein for Ig-κJ region (RBP-J), maintains the identity of the JG cell and the ability of cells along the arterioles to regain the renin phenotype when homeostasis is threatened. In fact, mice with conditional deletion of RBP-J (conditional knockout [cKO]) in cells from the renin lineage have very few JG cells, and SM cells along the afferent arterioles cannot reacquire the renin phenotype on a physiologic challenge.8 However, it was unclear whether in vivo Notch/RBP-J regulated the renin promoter directly and/or the expression of genes known to be characteristic of or responsible for the dual endocrine–contractile phenotype of the renin cell. Therefore, we designed a series of experiments to test the hypothesis that RBP-J regulates a gene network that controls the dual endocrine–contractile identity of the JG cell and the ability of cells upstream from the glomerulus to reacquire the renin phenotype.
Results
RBP-J Activates the Renin Promoter
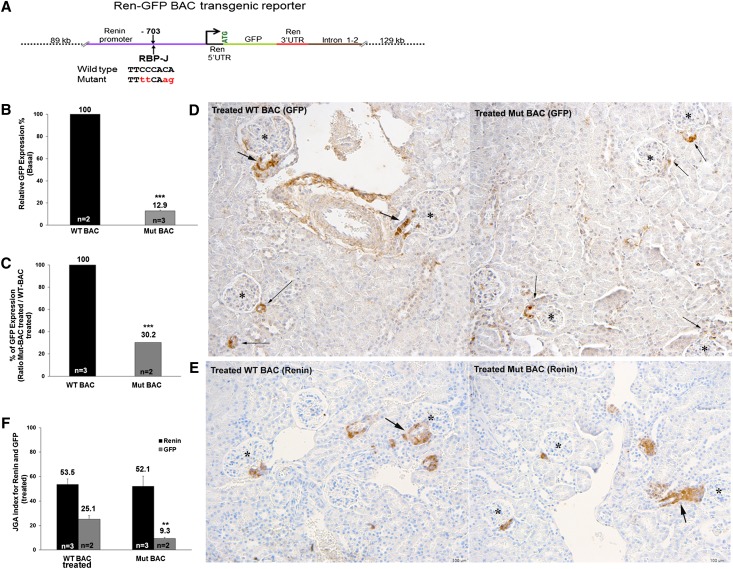
To determine whether RBP-J directly affects renin expression, we used a bacterial artificial chromosome (BAC) system to generate control wild-type BAC (WT-BAC) transgenic mice, in which the first exon of the Ren1c gene was replaced with an enhanced green fluorescent protein (GFP), and mutant BAC (Mut-BAC) mice, in which the four nucleotides in the RBP-J consensus sequence critical for its binding9 were substituted in the BAC construct (Figure 1A).
Figure 1.
RBP-J regulates the renin promoter in vivo. Mutation of the RBP-J site in the renin promoter diminishes GFP expression, a surrogate of renin expression. (A) Diagram of the BAC reporter construct used to generate BAC transgenic mice. The dual kanamycin/streptomycin selection strategy was used to replace the first exon of renin with enhanced GFP, insert the renin gene 3′ untranslated region (UTR), and modify the RBP-J binding site (nucleotides are in red). (B) Quantitative RT-PCR for GFP mRNA expression in kidneys from mice harboring the mutated RBP-J reporter (Mut-BAC) is significantly lower than in the control (WT-BAC) mice at basal state. (C–F) Mice were treated with low sodium diet+captopril for 5 days to induce re-expression of renin along arterioles. (C) Mut-BAC mice do not increase GFP mRNA to the same level as WT-BAC mice. (D and E) Immunohistochemistry for GFP and renin expression in response to homeostatic challenge. (D) In WT-BAC mice, GFP (brown) is in JG cells, small arterioles, and along the arterioles, recapitulating the renin pattern. Mut-BAC mice have no GFP along the arterioles and few GFP-positive JGAs (arrows). (E) Renin staining in WT-BAC and Mut-BAC mice shows that, as expected, the endogenous, nonmutated renin gene responds by increasing the number of renin-positive cells along the arterioles properly and equally in both groups. *Glomeruli with GFP-positive or renin-positive JGA. (F) The JGA index for renin in both transgenic mice shows that harboring the BAC transgene does not affect expression of the endogenous renin gene. Values are means±SEMs. **P<0.01; ***P<0.001.
We studied two independent transgenic lines each containing two copies of the WT-BAC or Mut-BAC transgene. In the basal state, Mut-BAC mice had 87% lower GFP mRNA expression than WT-BAC mice (Figure 1B). To investigate whether Mut-BAC mice can increase the expression of GFP under a physiologic threat, we treated Mut-BAC and WT-BAC mice with captopril and sodium depletion for 5 days, a manipulation known to induce arteriolar SM cells upstream from the JG area (JGA) to reacquire the renin phenotype. Treated Mut-BAC mice had diminished capacity to increase GFP expression (only 30% of that in treated WT-BAC mice) (Figure 1C). In correspondence with the smaller increase in GFP expression in the treated Mut-BAC mice, immunohistochemistry for GFP showed that, in contrast to treated WT-BAC mice, treated Mut-BAC mice did not have GFP-positive cells upstream of the arterioles distant from the glomeruli (Figure 1D). Quantification of the immunostaining results showed that the JGA index for GFP was significantly lower in treated Mut-BAC than treated WT-BAC mice (Figure 1F). As expected, renin staining and JGA index for renin were similar in both groups, indicating that introduction of the BAC transgene did not disrupt the expression of the endogenous renin gene (Figure 1, E and F). Together, these data indicate that RBP-J regulates the renin promoter directly (Figure 7A) and is involved in the ability of SM cells along the arteriole to reacquire the renin phenotype.
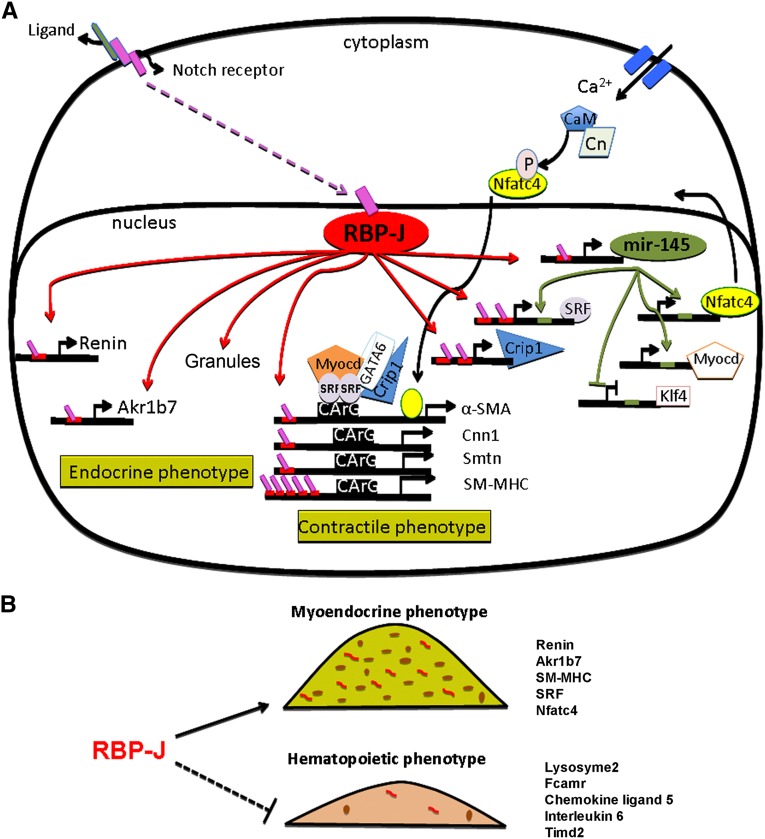
Figure 7.
RBP-J acts as a master regulator that maintains the identity of the JG cell. (A) RBP-J regulates a network of genes that confers the endocrine–contractile phenotype of the JG cell. Red bars located in the promoters of target genes represent RBP-J binding sites. Endocrine genes, such as renin and Akr1b7, and generation of renin granules are indicated on the left side of the diagram. Genes necessary to maintain the contractile phenotype are indicated on the right side. RBP-J regulates the SM genes directly through their promoter regions and indirectly by upregulating the expression of miR-145–5p and SRF. miR-145–5p positively regulates SRF, myocardin, and Nfatc4, and they act together to activate the expression of SM genes.15 SRF binds to CArG sites located in the SM genes. Given that SRF is an miR-145–5p target gene and has two RBP-J sites in its promoter, it is likely that both miR-145–5p and RBP-J regulate the transcriptional activity of SRF. In addition, miR-145–5p also promotes the expression of SM genes by repressing Klf4, a transcription factor that forms a complex with SRF to prevent transcriptional activity of SM genes.46 Crip1, another RBP-J predicted target gene, is an SM marker known to form a complex with GATA 6 and SRF to promote the contractile phenotype. We hypothesize that the canonical Notch signaling pathway is involved in maintaining the myo-endocrine phenotype of the JG cell. The ligand–Notch receptor interaction (yet to be identified) results in the release of the Notch intracellular domain (pink boxes), allowing its translocation to the nucleus, where it binds RBP-J to activate transcription. CaM, calmodulin; Cn, calcineurin; P, phosphate; Smtn, smoothelin. (B) RBP-J maintains the identity of the JG cells by not only activating genes characteristic of their myo-endocrine phenotype but also, preventing the undesirable ectopic expression of genes from other lineages.
RBP-J Deletion Does Not Affect the Endowment of Cells from the Renin Lineage
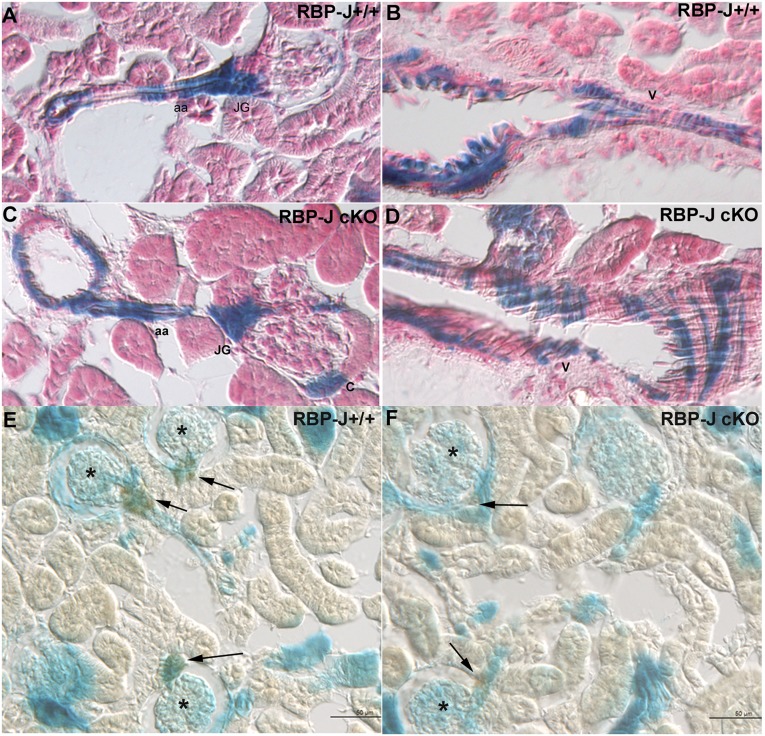
To determine whether the marked diminution in the number of JG cells resulted from a decreased population or a change in the distribution of cells from the renin lineage, we performed lineage studies in cKO and control mice harboring the R26R10 reporter. After cre-mediated recombination in the control and cKO mice, cells of the renin lineage express β-galactosidase (β-gal), effectively labeling renin cells and their descendants. We found that, as expected, these RBP-J cKO;R26R mice had reduced renin expression (Supplemental Table 1) as previously described in cKO mice lacking the reporter.8 Interestingly, the distribution of β-gal–positive cells was identical in the control and cKO kidneys (Figure 2), and the β-gal JGA indices were the same (Supplemental Figure 1). In contrast to controls, costaining for renin verified that cKO mice had few or no renin-expressing cells in the JGAs, but they were still β-gal–positive (Figure 2F, Supplemental Table 1). Furthermore, assays for apoptosis (Supplemental Table 2) and cell proliferation (not shown) showed no difference between control and cKO mice. These data indicate that the decrease in the number of renin-expressing cells was not caused by an increase in the percentage of dead cells or a decrease in the number and/or location of the renin precursors and subsequent progeny of renin-derived cells. Therefore, former renin-expressing cells and their descendants are still present in the appropriate locations in cKO mice, although they are no longer capable of expressing renin, suggesting the possibility that they have adopted a different phenotype.
Figure 2.
RBP-J deletion does not affect the endowment of cells from the renin lineage. Kidneys from control and RBP-J cKO;R26R adult mice were subjected to the X-gal reaction to detect β-gal, which marks cells that expressed renin earlier in development (blue). (A and B) In RBP-J+/+ control kidneys, blue staining is seen in the JGA (JG), along the afferent arteriole (aa), and in a large vessel (V). (C and D) RBP-J cKO kidneys have the same blue staining pattern as controls. (E and F) Double staining for β-gal (blue) and renin (brown). Whereas β-gal labels the developmental history of renin expression (thus, blue staining is observed along arterioles and glomeruli), the actual expression of renin (brown) in adult animals is confined to JG cells. In cKO mice, the distribution of blue cells in the JGA and arterioles is not different from controls; however, cKO mice have fewer renin+ JGAs (arrows) than in control mice. *Glomeruli with renin+ JGAs.
RBP-J Deletion Affects the Myo-Endocrine Phenotype of Cells of the Renin Lineage
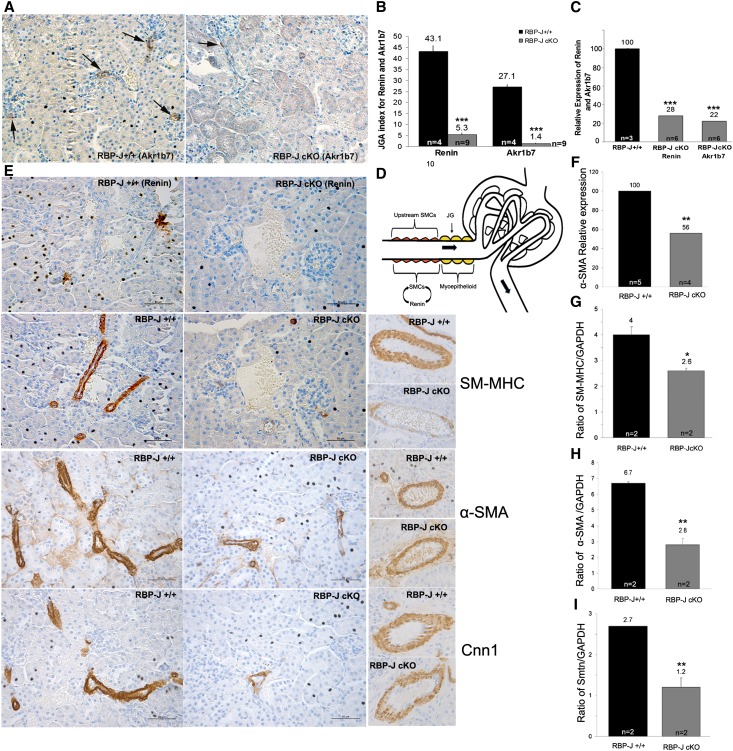
Given that cells of the renin lineage were still present in the appropriate locations in RBP-J cKO mice, suggesting that they may have switched their phenotype, led us to investigate whether repression of renin was accompanied by alterations in the expression of other genes characteristic of renin cells.7 Aldo-keto reductase 1b7 (Akr1b7), an independent endocrine marker of JG cells, is coexpressed at almost the same level as renin in JG cells throughout development and in response to physiologic manipulations.7 Akr1b7 immunostaining in cKO mice was markedly diminished with respect to controls (Figure 3A), and quantitation showed a significantly lower Akr1b7 JGA index (Figure 3B). The decrease in the number of JGAs expressing Akr1b7 in the cKO mice was accompanied by a reduction in Akr1b7 mRNA expression to the same level as renin mRNA (Figure 3C). The effects of RBP-J on the endocrine phenotype of the JG cell are illustrated in Figure 7A.
Figure 3.
Deletion of RBP-J affects expression of genes marking the dual endocrine and SM phenotype of renin cells. (A–C) Expression of Akr1b7, an endocrine phenotype marker. (A) Immunostaining for Akr1b7 shows that RBP-J+/+ mice express Akr1b7 in the JGAs (arrows in left panel). RBP-J cKO mice have few or no Akr1b7-positive JGAs (arrow in right panel). (B) In RBP-J cKO mice, the JGA indices for both Akr1b7 and renin are significantly reduced. (C) Akr1b7 and renin mRNA levels in RBP-J cKO mice decreased significantly to the same extent compared with controls. (D–I) Expression of SM phenotype markers assessed by (D and E) immunostaining and (F–I) mRNA expression. (D) The sketch illustrates the location of JG cells (yellow) in the adult animal and the location of VSMCs upstream in the afferent arterioles, where expression of SM genes is reduced in RBP-J cKO mice. The diagram also indicates that the SM cells (SMCs) have the plasticity to re-express the renin phenotype when confronted with a homeostatic threat. (E) Consecutive sections stained with renin and SM-MHC show that RBP-J cKO mice had few renin-positive cells, and the expression of SM-MHC was markedly reduced, particularly in the interlobular arteries and upstream portion of the afferent arterioles. Consecutive sections stained for α-SMA and Cnn1 show that α-SMA staining upstream in the afferent arterioles is thinner with decreased intensity and extent of expression. Cnn1 staining shows an overall decrease in the number of Cnn1-positive cells. In large arteries (right panels), cKO mice had fewer SM-MHC–positive cells around the artery, but localization of α-SMA and Cnn1 was not different from controls. Notice that SM-MHC in mutant mice had a scant and dot-like distribution of SM-MHC–positive cells around the artery. (F) α-SMA mRNA expression was reduced to 56% of control in kidneys from cKO mice. (G–I) Semiquantitative PCR in isolated kidney arterioles showed that α-SMA, SM-MHC, and smoothelin expression decreased 2.4-, 1.5-, and 2.3-fold, respectively, in the RBP-J cKO mice. Values are means±SEMs. Scale bars, 50 µm. *P<0.05; **P<0.01; ***P<0.001.
The expression of SM genes is an important defining characteristic of the JG cells, which must have contractile function to properly regulate renal hemodynamics.7 We evaluated the effect of RBP-J deletion on expression of specific SM genes in the JG and the upstream segments of the afferent arterioles and along larger intrarenal arteries (diagram in Figure 3D). Immunohistochemistry for SM myosin heavy chain11 (SM-MHC), α-SM actin (α-SMA), and calponin1 (Cnn1) showed decreased expression in cKO mice: cells were thinner with lesser amounts of staining (Figure 3E), particularly in the interlobular arteries and upstream of the afferent arterioles. Interestingly, the larger intrarenal arteries had fewer SM-MHC–positive cells, whereas staining for α-SMA and Cnn1 was similar to controls (Figure 3E). Although immunostaining for α-SMA and Cnn1 in large arteries appeared to not be different from controls, their expression was diminished in arterioles (Figure 3E). Thus, deletion of RBP-J has a differential effect on the expression of SM proteins depending on the blood vessel size, with greater effect on the expression of SM-MHC in large arteries than α-SMA and Cnn1.
The decreased expression of SM markers in the upstream portion of arterioles was accompanied by lower mRNA levels. Measurement of α-SMA, SM-MHC, and smoothelin (a marker for terminally differentiated SM cells) using quantitative RT-PCR and semiquantitative PCR in total kidney and/or isolated arterioles showed that, in cKO mice, total kidney α-SMA mRNA was 44% lower than in controls (Figure 3F), and in isolated arterioles, expression of α-SMA, SM-MHC, and smoothelin was 2.4-, 1.5-, and 2.3-fold lower, respectively, compared with controls. (Figure 3, G–I).
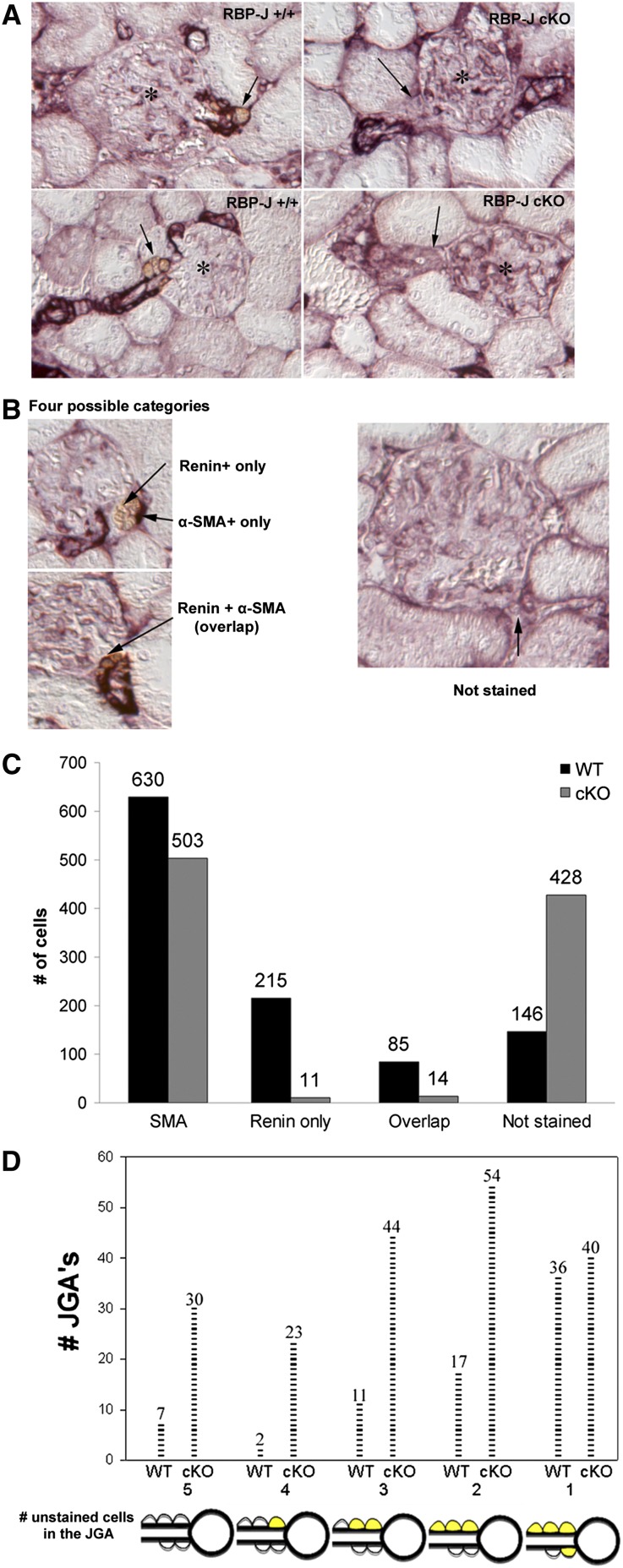
The observation that the expression of the SM genes was reduced led us to further investigate in topological detail whether the expression of SM genes was specifically affected in the cells of the JGA. Using double immunostaining for renin and α-SMA (Figure 4), we found that a significant proportion of JGAs in the cKO mice was not stained with either antibody. We quantified the costaining for renin and α-SMA as follows. Five JG cells/JGA were scored and divided into four categories based on the expression of one or both markers or no expression: (1) renin only, (2) α-SMA only, (3) overlapping staining (renin and α-SMA), and (4) unstained cells (Figure 4, B–D). As expected, the cKO mice had a significant decrease in the number of renin-positive cells (Figure 4C). Interestingly, cKO mice had a 2.9-fold increase in the total number of unstained (with either antibody) cells compared with controls (Figure 4C). We then took a closer look at the unstained category in which five cells/JGA were evaluated and found that cKO animals had a significant number of JGAs with two to five cells that have lost the expression of renin and SM proteins (Figure 4D). Furthermore, double staining for Cnn1 and renin showed a similar result: a 2.4-fold increase in the number of unstained JGA cells in cKO mice (Supplemental Figure 2). Altogether, these data indicated that deletion of RBP-J in JG cells affects not only expression of renin and Akr1b7 but also, the expression of genes characteristic of the SM phenotype, suggesting that RBP-J also maintains the contractile machinery of the JG cells. This point is illustrated in Figure 7A.
Figure 4.
Deletion of RBP-J increases the number of JG cells that express neither renin nor SM markers. (A) Double staining for renin (brown) and α-SMA (purple) shows that cKO mice have few or no renin-positive JG cells (arrows) and diminished or absent α-SMA expression in JGAs. *Glomeruli. (B) Five cells in each JGA were counted and categorized as (1) renin+ only, (2) α-SMA+ only, (3) both renin+ and α-SMA+, and (4) not stained. (C) A summary of the number of cells in each category shows that RBP-J cKO mice had an increased number of unstained cells. (D) The number of JGAs with one to five cells unstained with either antibody. RBP-J cKO mice had more JGAs with two to five unstained cells, whereas the number of JGAs with one unstained cell is similar in RBP-J cKO and control animals. In total, 219 JGA cells were counted in the controls (n=2), and 216 JGA cells were counted in the mutants (n=2). Each tick indicates a JGA. Cells in yellow are cells stained with either antibody.
RBP-J Regulates the SM Program of the JG Cell
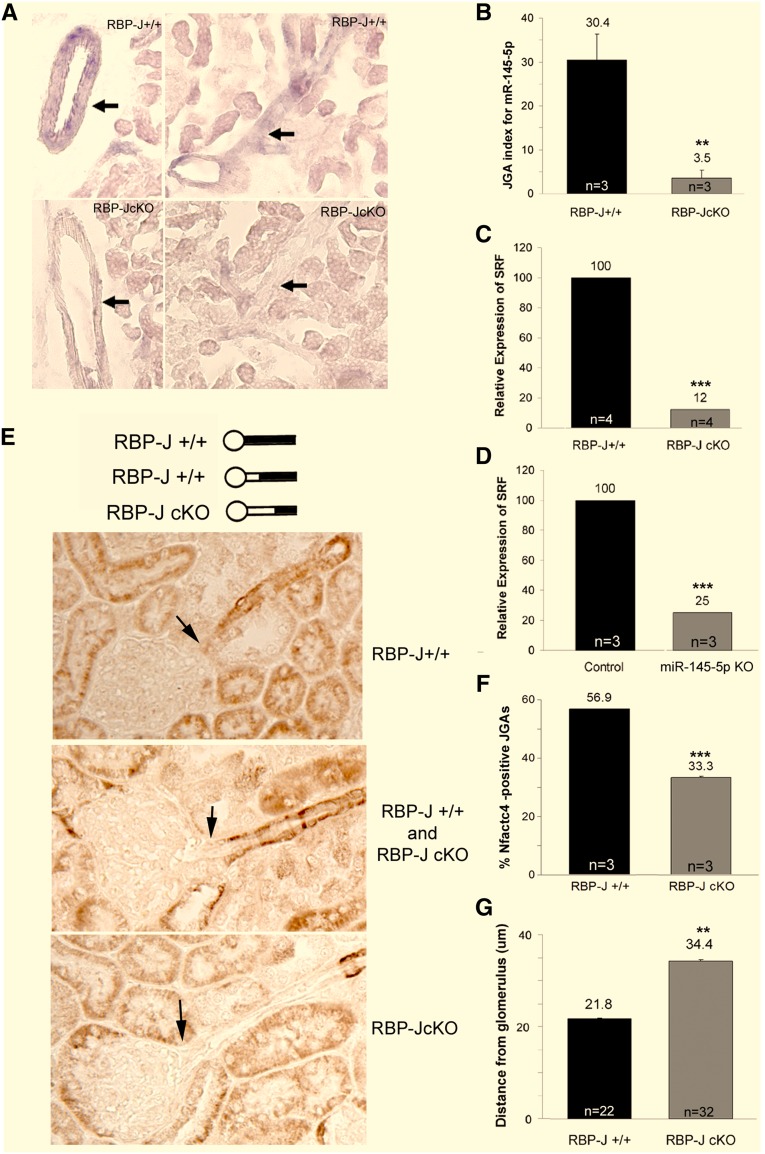
We previously reported that microRNAs (miRs), a group of small noncoding RNAs,11 are necessary to maintain the phenotype of renin-expressing cells and the integrity of the kidney vasculature.12 Given that miR-145–5p is prominently expressed in the kidney arterioles and may be important to sustain the SM phenotype of JG cells, we evaluated whether expression of miR-145–5p was altered by deletion of RBP-J. In situ hybridization for miR-145–5p in RBP-J cKO mice revealed a significant decrease in the number of miR-145–5p-positive cells in the JGA and renal arterioles (Figure 5A) and a significantly lower miR-145–5p JGA index (Figure 5B).
Figure 5.
Deletion of RBP-J reduces expression of miR-145–5p and its downstream SM gene targets. (A) In situ hybridization for miR-145–5p. In control mice, miR-145–5p is expressed in SM cells along the arterioles and in major arteries (arrows), whereas the RBP-J cKO mice exhibit a significant decrease in the overall expression of miR-145–5p. (B) In addition, the number of JGAs positive for miR-145–5p was decreased. (C) SRF mRNA expression is reduced significantly in both RBP-J cKO and miR-145–5p KO mice. (D–G) Nfatc4 expression in RBP-J cKO mice. (E) Staining for Nfatc4 showed that some JGAs (arrows) were Nfatc4-positive and that some were negative; however, the upstream portion of the arterioles was positive. The diagram illustrates the distribution of Nfatc4-positive cells. (F) JGAs with a visible arteriole were examined in control and cKO mice, and it was seen that the percent of Nfatc4-positive JGAs is reduced in RBP-J cKO mice. (G) To define whether RBP-J deletion altered the distribution (distance) of Nfatc4-positive cells from the glomerulus, we measured the distance from the JGA to the first Nfatc4-positive cells along the arteriole upstream from the glomerulus. In RBP-J cKO mice, the majority of the Nfatc4-positive cells were farther upstream of the JGA compared with controls. Values are means±SEMs. **P<0.01; ***P<0.001.
To examine whether deletion of RBP-J affects the expression of additional key SM regulators, we studied the expression of SRF, Crip1, and NF of activated T cells 4 (Nfatc4). SRF binds the CArG box in the promoters of SM genes to activate transcription. SRF forms a complex with myocardin,13,14 which is positively regulated by miR-145–5p and also modulates miR-145–5p in a positive feedback loop (Figure 7A).15,16 SRF mRNA expression in cKO mice was significantly lower than in controls (Figure 5C). To further investigate whether expression of SRF is altered by deletion of miR-145–5p, we performed quantitative RT-PCR for SRF in miR-145–5p KO mice. Results show that kidney cortex from miR-145–5p KO mice had significantly reduced SRF expression (Figure 5D), suggesting that both RBP-J and miR-145–5p are necessary to maintain SRF expression in the kidney vasculature. In addition to the SRF–myocardin complex, SRF associates with the SM marker Crip1, a double zinc finger-like molecule that mediates protein–protein interactions to promote the contractile phenotype15,17 (Figure 7A). Crip1 distribution in cKO mice was diminished in both JGA and arteriolar SM cells, indicating that RBP-J controls the expression of another key regulator of the SM phenotype (Supplemental Figure 3).
Nfactc4, another gene known to be important in regulating the expression of SM genes, is a transcription factor that modulates SM gene transcription in a Ca2+- dependent manner18 (Figure 7A). We found that cKO mice had a significant decrease in the number of JGAs positive for Nfatc4 compared with controls (Figure 5F). Interestingly, in Nfatc4-negative JGAs (controls as well as cKO), their corresponding arterioles had Nfatc4-positive cells farther upstream from the glomeruli (Figure 5E). Although expression of Nfatc4 was heterogeneous, the distance from the junction where the arteriole enters the glomerulus to the beginning of the Nfatc4-positive cells along the afferent arteriole (diagram in Figure 5E) was 1.6-fold greater in cKO mice (Figure 5G). Thus, control and cKO mice exhibit heterogeneous Nfatc4 distribution within the afferent arteriole, but deletion of RBP-J results in a decrease in the number of Nfatc4-positive JGAs and fewer Nfatc4-positive cells along the renal arterioles.
Deletion of RBP-J Alters the Fate of Cells from the Renin Lineage
In an attempt to define the identity of the JG cells that have lost their endocrine–contractile phenotype, we screened for the expression of markers characteristic of stem cells (Oct-4 and Nanog) and mesenchymal (vimentin), endothelial (PECAM), and epithelial (cytokeratin) cells but found no changes in staining in the JGA of cKO mice (data not shown). Given those negative results and with the goal of defining the repertoire of genes enriched in renin lineage cells lacking RBP-J, we performed mRNA microarray analysis in purified arterioles from control and cKO mice. We first confirmed by semiquantitative PCR that, as expected, renin and RBP-J expressions were significantly decreased in arterioles from cKO mice (Supplemental Figure 4). Results from the microarray analysis were striking: arterioles from cKO mice had increased representation of genes that belong to the immune response system (Figure 6A). A closer look at the top significantly upregulated genes in the cKO arterioles shows that many of these genes are normally expressed in hematopoietic cells (B cells: Ccl9, Il6, Fcamr, Bcl2a1c, Igk-C, Rgs1, H2Aa, and Cd72; T cells: Timd2, Ccl9, Il6, and Ccl5) (Figure 6B, Supplemental Table 3). We performed RT-PCR analysis and confirmed the increased expression of Timd2, Ccl2, Ccl5, Fcamr, and Abp1 in kidneys from cKO mice (Figure 6C). These genes and numerous other upregulated genes are involved in B cell differentiation, directing migration of monocytes, macrophages, or T cells, and immune and inflammatory responses. Thus, these data suggest that cells of the renin lineage lacking RBP-J now are expressing genes normally confined to cells of the hematopoietic/immune response system.
Figure 6.
Genes associated with the hematopoietic immune response are upregulated in arterioles from RBP-J cKO mice. (A) Gene Ontology analysis of the mRNA microarray data in isolated arterioles from RBP-J cKO mice shows increased representation of biologic processes related to the immune response. (B) Immune response-related genes upregulated in RBP-J cKO arterioles. (C) Validation of upregulated genes by PCR of RNA from control and RBP-J cKO kidneys. GAPDH was used for normalization.
Discussion
This series of studies shows that RBP-J, the main effector of the Notch pathway, (1) controls the genetic program that confers the dual endocrine–contractile phenotype of the JG cell, (2) sustains the memory of SM cells upstream in the arterioles to reacquire the renin phenotype, and (3) suppresses the extemporaneous ectopic expression of genes from other lineages. These findings are illustrated in Figure 7.
The expression of two crucial genes characteristic of the JG cell endocrine phenotype, renin and Akr1b7, is profoundly diminished by RBP-J deletion in both the basal unstressed state and response to the homeostatic challenge of sodium depletion and captopril treatment. Using BAC-GFP reporter mice, we show that RBP-J directly regulates the renin promoter in vivo. This effect is manifested in the unstressed animal as well as in response to the physiologic challenge mentioned above, indicating that RBP-J controls the phenotype of the JG cell as well as the response of the SM cells upstream from the glomerulus. These results agree with in vitro studies showing that RBP-J activates the renin promoter.19 Using chromatin immunoprecipitation, we showed that RBP-J is expressed in JG cells and binds the renin promoter,7 and now, it provides a functional correlate in vivo. In other systems, RBP-J is a repressor or activator depending on the tissue context.20 Our results show, for the first time, that, in vivo, RBP-J acts as a direct transcriptional activator of the renin gene.
In addition to renin, RBP-J regulates the expression of Akr1b7, a newly discovered marker for renin cells.7 Akr1b7, which also possesses RBP-J binding sites in its promoter, is expressed in kidney cells programmed to the renin phenotype, even when the renin gene has been deleted.7 Akr1b7 belongs to the Akr superfamily of enzymes that catalyzes the reduction to alcohol of harmful aldehydes and ketones generated by hormone synthesizing cells.21 This detoxifying function of Akr1b7 seems crucial to protect renin cells from those harmful compounds and promote cell survival. In the cKO mice, Akr1b7 mRNA and protein expression are reduced in parallel with renin. This finding is in agreement with our previous observation that Akr1b7 is expressed together with renin in JG cells. When there is a challenge to BP/fluid electrolyte homeostasis, there is a recruitment of cells coexpressing both proteins (Akr1b7+/renin+ cells) along the kidney vasculature, indicating that SM cells along the arterioles, upstream from the glomerulus, are again transformed to the endocrine phenotype, a situation resembling the fetal pattern of expression for both genes. Interestingly, we have found that, when the renin gene is deleted, there is still an unsuccessful attempt to increase the number of renin cells. The expression of Akr1b7 in those cells is simultaneously upregulated along the arterioles, indicating that Akr1b7 is part of a genetic program that coactivates both genes as well as the machinery that controls renin granule formation when the endocrine phenotype is turned on.7,8 Until recently, the molecules that governed the expression of the endocrine phenotype of the JG cell were unknown. Our findings indicate that RBP-J is at least one of such molecules and that RBP-J is fundamental to sustain the endocrine status of the renin cell (Figure 7A).
The actions of RBP-J are not limited to the endocrine functions of the renin cell. In addition to renin, JG cells express numerous SM genes7 crucial to maintain the contractile phenotype required to regulate glomerular blood flow and response to physiologic cues. Here, we show that RBP-J deletion affects the expression of SM genes and their master regulators. Expression of SM genes was markedly reduced in afferent arterioles and interlobular arteries. Interestingly, SM-MHC expression was severely decreased in large arteries, indicating a heterogeneous response of the SM genes along the kidney microvasculature. Of note, SM-MHC has five predicted RBP-J binding sites in its promoter, whereas both α-SMA and Cnn1 have only one site (Figure 7A), suggesting that the number of RBP-J binding sites may influence the level of expression of individual SM genes. Additional studies are needed to answer this question. In agreement with the present results, other studies show that Notch signaling by RBP-J directly and/or indirectly affects adult vascular SM cell (VSMC) differentiation in a cell type-specific manner and that RBP-J has a role in activating the SM-MHC and α-SMA genes in VSMCs.22,23 In silico analyses showed that multiple SM genes expressed in the JG cells have RBP-J binding sites (Figure 7A). Although RBP-J can directly control SM gene transcription,22,23 it is likely that RBP-J also regulates a network that reinforces the acquisition and maintenance of the JG cell SM program. We, therefore, examined whether lack of RBP-J affected the expression of key upstream regulators of the SM program. One such regulator is miR-145–5p, which is important for muscle development in other systems24 and specifically expressed in JG cells and SM cells along the afferent arterioles.12 We found reduced miR-145–5p expression in cKO renal arterioles and JG cells, suggesting that RBP-J regulates miR-145–5p directly through its promoter. In fact, the human miR-145–5p promoter has seven predicted RBP-J binding sites,25 and studies in human aortic VSMCs showed that activation of the canonical Notch signaling pathway is required to maintain miR-145–5p transcription.25 These findings, together with our observations, suggest that miR-145–5p functions to maintain the SM phenotype of JG cells and arteriolar VSMCs. miR-145–5p may promote the contractile phenotype by upregulating myocardin,25 which forms a complex with the transcription factor SRF to activate SM genes13 (Figure 7A). Increasing miR-145–5p expression in fibroblasts in vitro reprograms them into VSMCs, in part by the positive effect of miR-145–5p on myocardin in a feedback loop in which miR-145–5p upregulates myocardin, allowing the formation of the SRF–myocardin complex, and subsequent SRF expression reinforces miR-145–5p to maintain myocardin expression.15 In fact, lack of RBP-J resulted in a decrease in SRF mRNA, suggesting the possibility that RBP-J deletion decreased miR-145–5p levels, and therefore, miR-145–5p could not reinforce expression of SRF. In support of this conclusion, we found that miR-145–5p KO mice had a marked decrease in SRF expression, thus supporting the hypothesis that miR-145–5p serves as a positive regulator of SRF and the SM phenotype in JG cells (Figure 7A). Furthermore, our data showing that SRF mRNA levels are decreased significantly in both RBP-J cKO and miR-145–5p KO mice suggest that RBP-J and miR-145–5p jointly regulate SRF expression and therefore, SM expression in JG cells.
Our laboratory has shown that Nfatc4 is expressed in JG cells as well as the kidney arterioles, suggesting that Nfatc4 contributes to maintenance of the contractile phenotype.7 Nfat proteins are transcription factors important for the development of the cardiovascular system26 and the migration and proliferation of SMCs.27 Nfatc4 also positively regulates α-SMA expression through its promoter.28 We found that the number of Nfatc4-positive cells in cKO mice is diminished significantly in the JGA and along the afferent arterioles. Our observation that Nfatc4 decreased in cKO mice provides an additional link between miR-145–5p and Nfatc4 activity, because Nfatc4 is a target of miR-145–5p (Figure 7A).29 It is likely that decreased Nfatc4 in the RBP-J cKO is caused by diminished miR-145–5p expression, although additional experiments will be required to confirm whether miR-145–5p directly regulates Nfatc4 in JG cells.
Remarkably, the decreased number of renin-expressing cells found in RBP-J cKO mice was not accompanied by apoptosis, necrosis, and/or changes in proliferation. More important, lineage tracing clearly showed that, in cKO mice, cells of the renin lineage were present in all of their normal locations. Altogether, those findings indicate that cells from the renin lineage lacking RBP-J did not die but switched their phenotype to another cell type. Because Notch/RBP-J has been proposed to control binary cell fate decisions,30,31 we expected that suppression of renin would simply cause a switch to the SM phenotype. However, this result was not the case. In fact, expression of SM genes (and the expression of their main regulators) was also markedly suppressed. How can these findings be better understood? Although the phylogenetically conserved role of Notch is to link the fate choice of one cell to that of neighboring cells, this outcome is contextual, depending on the proliferative state, the degree of differentiation of interacting cells, and the physiologic and maturational status of the surrounding tissue where those cells exist. Recent information from several laboratories clearly illustrates this point and helps in understanding our findings: we have recently shown that deletion of RBP-J in renal stromal Foxd1+ cells, the earliest progenitors for all the mural cells of the kidney vasculature,32 leads to decreased endowment of renin cells, mesangial cells, and SM cells, resulting in aberrant morphogenesis of arterioles and the formation of glomerular aneurysms.33 In this study, however, deletion of RBP-J in cells already committed to a renin cell fate (and therefore, located downstream in the Foxd1 lineage hierarchy) does not result in overt morphogenetic/developmental defects, but it does lead to a remarkable switch in the fate/identity of the adult JG cells. Thus, mutation of RBP-J in the earliest progenitor leads to a decrease in the pool of descendants, whereas deletion in the more differentiated renin precursors results in a significant change in the choice of cell identity. In line with our own observations and contrary to the situation in the embryo, Notch signaling in the adult gut influences cell fate without altering morphogenesis.34 In those studies, ectopic Notch signaling in the adult mouse intestinal progenitors directed cells away from a secretory fate, whereas in the embryo, Notch overexpression resulted in defects in gut morphogenesis. Altogether, the aforementioned experiments indicate that the actions of Notch/RBP-J are influenced by the degree of and potential for differentiation of the target cells.
As mentioned above, our findings support the notion that RBP-J sustains the memory of SM cells upstream in the arterioles to reacquire the renin phenotype. Under normal circumstances, renin secretion from JG cells is sufficient to respond to the minute-to-minute physiologic demands of daily life. However, when there is a more profound challenge to homeostasis (such as sodium depletion and diminished angiotensin actions as in these experiments), preexisting SM cells along the renal arterioles are gradually called on to produce renin and restore homeostasis. This process, historically mislabeled as recruitment, is not because of proliferation and/or migration of new cells, but it is achieved instead by the reacquisition of the capability of SM cells to synthesize renin as they did in fetal life.4,35,36 We show here that SM cells harboring the deletion are incapable of such transformation, suggesting the distinct possibility that RBP-J controls the molecular machinery responsible for the switch in cell type. The intricate mechanism has yet to be delineated, but our previous observations in vitro and in vivo indicate that histone acetyl transferases such as CBP/p300, which partner with RBP-J, may be involved.37 Because Notch receptors are activated in SM cells and RBP-J moves to the nucleus, it interacts with several partners, including CBP/p300. We have proposed that histone H4 acetylation with the involvement of the Creb/CBP/p300 pathway leads to the opening of chromatin, allowing transcription of the renin gene to proceed.37–39 Furthermore, our observations using histone deacetylase inhibitors in JG cells in culture and deletion of CBP/p300 in vivo support this possibility.37–39 Additional work will be needed, however, to delineate the chain of molecular events responsible for the SM cells’ plasticity. Lastly, the findings raise an intriguing hypothesis. It has been suggested that modulation of the Notch pathway in cells that are not terminally differentiated provides them with the plasticity to change fate.40 Because SM cells along the arterioles are capable of transiently alternating between the SM and the renin phenotype, their chromatin must be constantly poised to allow such changes to occur, suggesting the possibility that SM cells of the renal arterioles are not terminally differentiated (being, in fact, a pool of cells that can be called into action when more renin is needed to control homeostasis).6 This hypothesis remains to be tested.
Our findings also point to a repressor role of Notch/RBP-J. A close look at the microanatomy of the afferent arterioles where they abut the glomeruli showed that cKO mice have a significant number of cells in the JG segment that expresses neither SM nor renin. Those dually negative cells did not express endothelial, epithelial, or stem cell markers. However, arteriolar cells express transcripts characteristic of hematopoietic cells, indicating that, in renin cells, RBP-J normally suppresses the ectopic expression of genes characteristic of other lineages. In retrospect, perhaps these findings should not be as surprising: canonical Notch/RBP-J signaling is essential for the development and function of lymphocytes,41 and in fact, JG cells share several transcription factors crucial for the commitment and differentiation of hematopoietic cells, including E2A, EBF1, Ikaros, and RBP-J.7 Altogether, this information suggests a remarkable conservation of the transcriptional machinery and a possible lineage relationship between these cell types. How RBP-J suppresses the expression of hematopoietic genes in renin cells remains to be determined. However, it should be noted that a number of the upregulated genes in our cKO mice (e.g., Abp1, Ccl2, Ccl5, Ccl9, Fcamr, and Timd2) have in silico predicted RBP-J binding sites in their promoters and are likely to be regulated directly by RBP-J. Overall, our findings uncovered an unforeseen and powerful function of RBP-J in renin cells: the control of their identity not only by stimulating the expression of myo-epithelioid genes as discussed above, but also by its function as a repressor. This view is portrayed schematically in Figure 7B.
Finally, given that Notch signaling is a cell-to-cell communication system, a crucial question that remains to be explored is the identity of the ligand(s) and receptor(s) involved in initiating and sustaining the response in both the basal state and response to major acute and chronic physiologic challenges, such as dehydration, sodium depletion, or inhibition of angiotensin actions. It is very likely that the same ligand(s) and receptor(s) involved in the maintenance of the JG cell are involved in a graded manner to acute and chronic challenges to homeostasis. This hypothesis, however, remains to be tested. Our own assessment so far indicates that Jagged1 and Jagged 2 are both expressed in renin cells, and both may contribute to the response. We are currently exploring this possibility and delineating the nature of the receptor(s) involved.
In summary, in the normal animal, RBP-J maintains the identity of the JG cells by not only activating genes characteristic of their myo-endocrine phenotype but also, preventing the undesirable ectopic expression of genes from other lineages and the adoption of an aberrant phenotype with potentially devastating consequences for the control of homeostasis. Furthermore, RBP-J controls the memory of cells to reacquire the renin phenotype when homeostasis is challenged.
Clearly, regulation of renin cell identity by the Notch pathway is a complex process, whereby RBP-J, a master transcriptional regulator, governs a broad network of genes, which acting in concert, culminates with the precise physiologically able phenotype of the renin cell.
Concise Methods
Mice
The RBP-Jfl/fl mice42 were the gift of Tasuku Honjo, and miR-145–5-p KO mice43 were obtained from Eric N. Olson. The R26R reporter mouse, which expresses β-gal in a cre-inducible manner,10 was used to trace the cells of the renin lineage in the RBP-J cKO mice. To generate the Ren1dcre/+;RBP-Jfl/fl;R26R+/− study animals, RBP-Jfl/fl;R26R+/+ mice were crossed with Ren1dcre/cre;RBP-Jfl/fl mice. The control mice, Ren1dcre/+;RBP-J+/+;R26R+/−, are WT for RBP-J and heterozygous for both Ren1dcre and R26R. The BAC transgenic mice were generated using the services of the University of Virginia Gene Targeting and Transgenic Facility. Founder mice carrying the BAC transgene were identified using primer sets targeting the area of GFP insertion, yielding an 860-bp product (5′-GAAGGAGAGCAAAAGGTAAGAG-3′ and 5′-GCCGATGGGGGTGTTCTG-3′). The presence of the intended mutation in the RBP-J binding site in the Mut-BAC mice was confirmed by sequencing (Supplemental Material, Supplemental Figure 5). The control WT-BAC and Mut-BAC transgenic founder mice were bred to C57BL/6 mice, and GFP expression was quantified in 3.5-month-old animals from the F1 generation.
All procedures followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Animal Care and Use Committee of the University of Virginia.
Animals were anesthetized with tribromoethanol, and the kidneys were removed, weighed, and preserved for different purposes. For RNA extraction, kidneys were stored in RNA later (Ambion, Austin, TX). For immunohistochemistry in paraffin sections, they were fixed in Bouin solution (overnight) or 10% formalin for 30 minutes, and for frozen sections, they were fixed in 4% paraformaldehyde in PBS for 1 hour, cryoprotected in 30% sucrose in PBS with 2 mM MgCl2 at 4°C overnight, and then embedded in OCT compound embedding medium (Miles, Elkhart, IL).
Immunohistochemistry and In Situ Hybridization
Immunohistochemistry was performed as previously described6 using antibodies against α-SMA (isotype IgG2a, clone 1A4, dilution 1:10,000; Sigma-Aldrich, St. Louis, MO), Cnn1 (1806–1, dilution 1:500; Epitomics Inc., Burlingame, CA), SM-MHC11 (5121–1, dilution 1:1000; Epitomics Inc.), Nfatc4 (ab62613, dilution 1:500; Abcam, Inc., Cambridge, MA), rabbit anti-mouse renin antibody37 (dilution 1:500), and Akr1b7 antibody (dilution 1:200, with citrate antigen retrieval; Santa Cruz Biotechnology, Santa Cruz, CA). For GFP detection, we used an anti-GFP antibody (A6455, dilution 1:1000; Invitrogen, Grand Island, NY) and the TSA Biotin System protocol (NEL700A001KT; PerkinElmer, Waltham, MA) to amplify the GFP signal.
β-Gal expression was evaluated in cryosections (7 μm) as previously described.6
In situ hybridization to detect miR-145–5p expression in kidney sections was performed as previously described.12
Kidney Microvessel Isolation
Preglomerular microvessels were isolated using iron oxide perfusion and magnet separation of the vessels as previously described.38 The isolated microvessels were passed through 21- and 23-gauge needles to remove the glomeruli from the vessels, and the arterioles were collected on a 100-µm filter.
Generation of the BAC Construct with Mutated RBP-J Binding Site
The BAC clone RP2388K7 containing the renin gene was purchased from CHORI (Oakland, CA). We used the dual kanamycin and streptomycin selection method44 to replace the first exon of the renin gene with enhanced green fluorescent protein (EGFP) (Clontech Laboratories, Mountain View, CA) and the renin 3′ untranslated region for the WT reporter (WT-BAC) and then introduced the mutation in the RBP-J binding site in the renin promoter for the Mut-BAC. Supplemental Material has details of the BAC constructs and primers used.
DNA and RNA Extraction
To determine the transgene copy number in the transgenic mice, we extracted DNA from mouse tails using a proteinase K digestion plus phenol/chloroform extraction protocol (The Jackson Laboratory, Bar Harbor, ME). The phenol/chloroform extraction step was repeated two times to obtain optimal 260/230 ratios.
RNA was extracted from kidneys and isolated arterioles using Trizol, and cDNA was prepared as previously described.8 For GFP quantification, cDNA was prepared from 1 μg total kidney RNA using the High Capacity cDNA Reverse Transcription Kit (part no. 4368814; Applied Biosystems, Carlsbad, CA) according to the manufacturer’s instructions.
RT-PCR and Quantitative RT-PCR Analyses
Semiquantitative RT-PCR was assessed using the integrated OD value (defined as a pixel value) by drawing equal sized boxes (for each sample) around bands, with background values (water) taken for each sample. Quantitative real-time PCR using SYBR Green (Invitrogen) to measure GAPDH and renin levels was performed as previously described.8 EGFP and GAPDH in the transgenic mice were measured by quantitative RT-PCR using the TaqMan Universal Master MixII and TaqMan expression assay primers for glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (4331182) and EGFP (4400291; Applied Biosystems).
The copy number of the BAC transgene in the transgenic mice was determined using the services of the University of Virginia Biomolecular Research Facility using TaqMan copy number assays (Applied Biosystems) for EGFP (4400291) and Tfrc (4458366; transferrin receptor) as the single-copy genomic standard.
RBP-J Binding Site Prediction
To identify potential RBP-J binding sites in the promoter region of genes, we performed in silico analysis using the Transcription Element Search System program (http://www.cbil.upenn.edu/cgi-bin/tess/tess?RQ=WELCOME) or MatInspector.45
Microarray Analyses
To identify genes altered by deletion of RBP-J, we performed mRNA microarray analysis in isolated arterioles from control and RBP-J cKO mice. Total RNA was extracted from microvessels isolated from adult mice (as described above), and hybridization to the Mouse Gene 1.0 ST array (Affymetrix, Santa Clara, CA) was performed by the Biomolecular Research Facility at the University of Virginia. Two groups were used (control versus mutant). Each group was a pool of RNA samples (controls: n=3; mutants: n=4). Microarray data were analyzed through the use of Ingenuity Pathway Analysis (Ingenuity Systems) with a significance cutoff of P<0.05.
Statistical Analyses
Data are expressed as means±SEMs. Significance was determined by t test. P<0.05 was considered significant.
Disclosures
None.
Supplementary Material
Acknowledgments
The authors thank Takele Yazew and Kimberly Hilsen-Durette for technical assistance with the mouse work.
This work was supported by National Institutes of Health Grants DK075481 (to M.L.S.S.-L.), R37-HL066242 (Diversity Supplement Graduate Research Assistant; to R.A.G), and R01-HL096735 (to R.A.G.).
Part of this work was presented at the High Blood Pressure Research Conference (September 19–22, 2012) in Washington, DC and published in abstract form (Ruth M. Castellanos Rivera; Ellen S. Pentz; Kenneth W. Gross; Silvia Medrano; Jing Yu; Maria Luisa S. Sequeira-Lopez; R. Ariel Gomez, Hypertension, 60: A38, 2012).
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2013101045/-/DCSupplemental.
References
- 1.Berg AC, Chernavvsky-Sequeira C, Lindsey J, Gomez RA, Sequeira-Lopez ML: Pericytes synthesize renin. World J Nephrol 2: 11–16, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gomez RA, Chevalier RL, Sturgill BC, Johns DW, Peach MJ, Carey RM: Maturation of the intrarenal renin distribution in Wistar-Kyoto rats. J Hypertens 4: S31–S33, 1986 [Google Scholar]
- 3.Gomez RA, Lynch KR, Chevalier RL, Wilfong N, Everett A, Carey RM, Peach MJ: Renin and angiotensinogen gene expression in maturing rat kidney. Am J Physiol 254: F582–F587, 1988 [DOI] [PubMed] [Google Scholar]
- 4.Gomez RA, Chevalier RL, Everett AD, Elwood JP, Peach MJ, Lynch KR, Carey RM: Recruitment of renin gene-expressing cells in adult rat kidneys. Am J Physiol 259: F660–F665, 1990 [DOI] [PubMed] [Google Scholar]
- 5.Sequeira Lopez ML, Pentz ES, Robert B, Abrahamson DR, Gomez RA: Embryonic origin and lineage of juxtaglomerular cells. Am J Physiol Renal Physiol 281: F345–F356, 2001 [DOI] [PubMed] [Google Scholar]
- 6.Sequeira López ML, Pentz ES, Nomasa T, Smithies O, Gomez RA: Renin cells are precursors for multiple cell types that switch to the renin phenotype when homeostasis is threatened. Dev Cell 6: 719–728, 2004 [DOI] [PubMed] [Google Scholar]
- 7.Brunskill EW, Sequeira-Lopez ML, Pentz ES, Lin E, Yu J, Aronow BJ, Potter SS, Gomez RA: Genes that confer the identity of the renin cell. J Am Soc Nephrol 22: 2213–2225, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Castellanos Rivera RM, Monteagudo MC, Pentz ES, Glenn ST, Gross KW, Carretero O, Sequeira-Lopez ML, Gomez RA: Transcriptional regulator RBP-J regulates the number and plasticity of renin cells. Physiol Genomics 43: 1021–1028, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tun T, Hamaguchi Y, Matsunami N, Furukawa T, Honjo T, Kawaichi M: Recognition sequence of a highly conserved DNA binding protein RBP-J kappa. Nucleic Acids Res 22: 965–971, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Soriano P: Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet 21: 70–71, 1999 [DOI] [PubMed] [Google Scholar]
- 11.Bartel DP: MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 116: 281–297, 2004 [DOI] [PubMed] [Google Scholar]
- 12.Sequeira-Lopez ML, Weatherford ET, Borges GR, Monteagudo MC, Pentz ES, Harfe BD, Carretero O, Sigmund CD, Gomez RA: The microRNA-processing enzyme dicer maintains juxtaglomerular cells. J Am Soc Nephrol 21: 460–467, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sun Q, Taurin S, Sethakorn N, Long X, Imamura M, Wang DZ, Zimmer WE, Dulin NO, Miano JM: Myocardin-dependent activation of the CArG box-rich smooth muscle gamma-actin gene: Preferential utilization of a single CArG element through functional association with the NKX3.1 homeodomain protein. J Biol Chem 284: 32582–32590, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang Z, Wang DZ, Pipes GC, Olson EN: Myocardin is a master regulator of smooth muscle gene expression. Proc Natl Acad Sci U S A 100: 7129–7134, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cordes KR, Sheehy NT, White MP, Berry EC, Morton SU, Muth AN, Lee TH, Miano JM, Ivey KN, Srivastava D: miR-145 and miR-143 regulate smooth muscle cell fate and plasticity. Nature 460: 705–710, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang Z, Wang DZ, Hockemeyer D, McAnally J, Nordheim A, Olson EN: Myocardin and ternary complex factors compete for SRF to control smooth muscle gene expression. Nature 428: 185–189, 2004 [DOI] [PubMed] [Google Scholar]
- 17.Henderson JR, Macalma T, Brown D, Richardson JA, Olson EN, Beckerle MC: The LIM protein, CRP1, is a smooth muscle marker. Dev Dyn 214: 229–238, 1999 [DOI] [PubMed] [Google Scholar]
- 18.Graef IA, Chen F, Chen L, Kuo A, Crabtree GR: Signals transduced by Ca(2+)/calcineurin and NFATc3/c4 pattern the developing vasculature. Cell 105: 863–875, 2001 [DOI] [PubMed] [Google Scholar]
- 19.Pan L, Glenn ST, Jones CA, Gross KW: Activation of the rat renin promoter by HOXD10.PBX1b.PREP1, Ets-1, and the intracellular domain of notch. J Biol Chem 280: 20860–20866, 2005 [DOI] [PubMed] [Google Scholar]
- 20.Furriols M, Bray S: A model Notch response element detects Suppressor of Hairless-dependent molecular switch. Curr Biol 11: 60–64, 2001 [DOI] [PubMed] [Google Scholar]
- 21.Bohren KM, Bullock B, Wermuth B, Gabbay KH: The aldo-keto reductase superfamily. cDNAs and deduced amino acid sequences of human aldehyde and aldose reductases. J Biol Chem 264: 9547–9551, 1989 [PubMed] [Google Scholar]
- 22.Doi H, Iso T, Sato H, Yamazaki M, Matsui H, Tanaka T, Manabe I, Arai M, Nagai R, Kurabayashi M: Jagged1-selective notch signaling induces smooth muscle differentiation via a RBP-Jkappa-dependent pathway. J Biol Chem 281: 28555–28564, 2006 [DOI] [PubMed] [Google Scholar]
- 23.Noseda M, Fu Y, Niessen K, Wong F, Chang L, McLean G, Karsan A: Smooth Muscle alpha-actin is a direct target of Notch/CSL. Circ Res 98: 1468–1470, 2006 [DOI] [PubMed] [Google Scholar]
- 24.Boettger T, Beetz N, Kostin S, Schneider J, Krüger M, Hein L, Braun T: Acquisition of the contractile phenotype by murine arterial smooth muscle cells depends on the Mir143/145 gene cluster. J Clin Invest 119: 2634–2647, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Boucher JM, Peterson SM, Urs S, Zhang C, Liaw L: The miR-143/145 cluster is a novel transcriptional target of Jagged-1/Notch signaling in vascular smooth muscle cells. J Biol Chem 286: 28312–28321, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.de la Pompa JL, Timmerman LA, Takimoto H, Yoshida H, Elia AJ, Samper E, Potter J, Wakeham A, Marengere L, Langille BL, Crabtree GR, Mak TW: Role of the NF-ATc transcription factor in morphogenesis of cardiac valves and septum. Nature 392: 182–186, 1998 [DOI] [PubMed] [Google Scholar]
- 27.Yellaturu CR, Ghosh SK, Rao RK, Jennings LK, Hassid A, Rao GN: A potential role for nuclear factor of activated T-cells in receptor tyrosine kinase and G-protein-coupled receptor agonist-induced cell proliferation. Biochem J 368: 183–190, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gonzalez Bosc LV, Layne JJ, Nelson MT, Hill-Eubanks DC: Nuclear factor of activated T cells and serum response factor cooperatively regulate the activity of an alpha-actin intronic enhancer. J Biol Chem 280: 26113–26120, 2005 [DOI] [PubMed] [Google Scholar]
- 29.Dweep H, Sticht C, Pandey P, Gretz N: miRWalk—database: Prediction of possible miRNA binding sites by “walking” the genes of three genomes. J Biomed Inform 44: 839–847, 2011 [DOI] [PubMed] [Google Scholar]
- 30.Artavanis-Tsakonas S, Matsuno K, Fortini ME: Notch signaling. Science 268: 225–232, 1995 [DOI] [PubMed] [Google Scholar]
- 31.Ohlstein B, Spradling A: Multipotent Drosophila intestinal stem cells specify daughter cell fates by differential notch signaling. Science 315: 988–992, 2007 [DOI] [PubMed] [Google Scholar]
- 32.Sequeira Lopez ML, Gomez RA: Development of the renal arterioles. J Am Soc Nephrol 22: 2156–2165, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lin EE, Sequeira-Lopez ML, Gomez RA: RBP-J in FOXD1+ renal stromal progenitors is crucial for the proper development and assembly of the kidney vasculature and glomerular mesangial cells. Am J Physiol Renal Physiol 306: F249–F258, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Stanger BZ, Datar R, Murtaugh LC, Melton DA: Direct regulation of intestinal fate by Notch. Proc Natl Acad Sci U S A 102: 12443–12448, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cantin M, Araujo-Nascimento MD, Benchimol S, Desormeaux Y: Metaplasia of smooth muscle cells into juxtaglomerular cells in the juxtaglomerular apparatus, arteries, and arterioles of the ischemic (endocrine) kidney. An ultrastructural-cytochemical and autoradiographic study. Am J Pathol 87: 581–602, 1977 [PMC free article] [PubMed] [Google Scholar]
- 36.Gomez RA, Lynch KR, Chevalier RL, Everett AD, Johns DW, Wilfong N, Peach MJ, Carey RM: Renin and angiotensinogen gene expression and intrarenal renin distribution during ACE inhibition. Am J Physiol 254: F900–F906, 1988 [DOI] [PubMed] [Google Scholar]
- 37.Gomez RA, Pentz ES, Jin X, Cordaillat M, Sequeira Lopez ML: CBP and p300 are essential for renin cell identity and morphological integrity of the kidney. Am J Physiol Heart Circ Physiol 296: H1255–H1262, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pentz ES, Lopez ML, Cordaillat M, Gomez RA: Identity of the renin cell is mediated by cAMP and chromatin remodeling: An in vitro model for studying cell recruitment and plasticity. Am J Physiol Heart Circ Physiol 294: H699–H707, 2008 [DOI] [PubMed] [Google Scholar]
- 39.Pentz ES, Cordaillat M, Carretero OA, Tucker AE, Sequeira Lopez ML, Gomez RA: Histone acetyl transferases CBP and p300 are necessary for maintenance of renin cell identity and transformation of smooth muscle cells to the renin phenotype. Am J Physiol Heart Circ Physiol 302: H2545–H2552, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fre S, Bardin A, Robine S, Louvard D: Notch signaling in intestinal homeostasis across species: The cases of Drosophila, Zebrafish and the mouse. Exp Cell Res 317: 2740–2747, 2011 [DOI] [PubMed] [Google Scholar]
- 41.Yuan JS, Kousis PC, Suliman S, Visan I, Guidos CJ: Functions of notch signaling in the immune system: Consensus and controversies. Annu Rev Immunol 28: 343–365, 2010 [DOI] [PubMed] [Google Scholar]
- 42.Han H, Tanigaki K, Yamamoto N, Kuroda K, Yoshimoto M, Nakahata T, Ikuta K, Honjo T: Inducible gene knockout of transcription factor recombination signal binding protein-J reveals its essential role in T versus B lineage decision. Int Immunol 14: 637–645, 2002 [DOI] [PubMed] [Google Scholar]
- 43.Xin M, Small EM, Sutherland LB, Qi X, McAnally J, Plato CF, Richardson JA, Bassel-Duby R, Olson EN: MicroRNAs miR-143 and miR-145 modulate cytoskeletal dynamics and responsiveness of smooth muscle cells to injury. Genes Dev 23: 2166–2178, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang S, Zhao Y, Leiby M, Zhu J: A new positive/negative selection scheme for precise BAC recombineering. Mol Biotechnol 42: 110–116, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cartharius K, Frech K, Grote K, Klocke B, Haltmeier M, Klingenhoff A, Frisch M, Bayerlein M, Werner T: MatInspector and beyond: Promoter analysis based on transcription factor binding sites. Bioinformatics 21: 2933–2942, 2005 [DOI] [PubMed] [Google Scholar]
- 46.Xu N, Papagiannakopoulos T, Pan G, Thomson JA, Kosik KS: MicroRNA-145 regulates OCT4, SOX2, and KLF4 and represses pluripotency in human embryonic stem cells. Cell 137: 647–658, 2009 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.