Abstract
In patients managed with opioids for chronic pain, opioid-induced bowel dysfunction—specifically, opioid-induced constipation (OIC)—is a common side effect, which has a significant impact on quality of life (QoL). The most recent developments for management of OIC are opioid antagonists, including naloxone, a competitive antagonist of peripheral opioid receptors that reverses opioid-induced peripheral gastrointestinal (GI) effects. A prolonged-release formulation of naloxone is available in combination with oxycodone (OXN PR). To review the specific role of OXN PR in the management of chronic pain and OIC and its impact on QoL and healthcare costs, a review of available relevant literature was conducted. Healthcare costs can be up to ten times higher for patients with GI events than for those without. Assessment of QoL in patients with OIC is essential, and multiple tools for its evaluation are available. The Bowel Function Index (BFI), a tool that was specifically developed and validated to measure bowel function in patients with OIC, can be an indication of QoL. In patients with moderate-to-severe chronic pain, randomized trials have demonstrated that OXN PR has equal analgesic efficacy and safety, but results in improved bowel function, compared with prolonged-release oxycodone (Oxy PR) alone. In conclusion, randomized studies using the BFI, as well as real-world clinical practice observations, have demonstrated improved QoL for patients taking OXN PR. This combination should allow more patients to benefit from the analgesic efficacy of opioid therapy and should minimize the side effects of constipation that correspond to improvements in QoL and healthcare offsets.
Key Points
| Opioid induced constipation is a medical condition that causes a substantial burden to the patient and the healthcare system. |
| In clinical studies, oxycodone/naloxone has been shown to improve bowel function and is estimated to be cost-effective according to health economic models. |
Introduction
Chronic pain is a common and disabling condition, which can significantly affect quality of life (QoL) [1–5]. A variety of definitions may be used to define chronic pain; the International Association for the Study of Pain defines it as pain without apparent biological value that has persisted beyond the normal tissue healing time (usually taken to be 3 months) [6]. In a large-scale survey of chronic pain (defined as pain lasting >6 months with an intensity of ≥5 on a 1–10 scale) across 15 European countries plus Israel, prevalence rates of 12–30 % were reported [2]. Prevalence rates, however, vary widely between studies because of differences in population characteristics, sampling methods and the criteria used to define chronic pain. Pain is a particular concern for cancer patients and has a significant impact on their QoL [7]. Up to 70 % of patients with advanced cancer have been reported to experience chronic pain [8, 9].
Chronic pain can stem from a variety of underlying conditions, which may be musculoskeletal, neuropathic, ischaemic or cancer related [2, 10], and may be seen as a biopsychosocial phenomenon in which pain interacts with psychosocial factors [11]. Chronic pain is also strongly associated with development of comorbidities, including depression and anxiety, impaired sleep and alterations in immune function [5, 12–15].
Management strategies for chronic pain are multimodal, with the aim of reducing pain and maximizing QoL [11, 16]. Non-pharmacotherapeutic management may include physical and psychological interventions [16]. The major pharmacological agents currently used to treat chronic pain include non-opioid analgesics (e.g. paracetamol and non-steroidal anti-inflammatory drugs, including cyclooxygenase-2 inhibitors) and opioids. In addition, atypical analgesics, including antidepressants and anticonvulsants, may be used. Pain intensity generally guides the process of analgesic choice and, in many cases, high-potency drugs are the first choice of treatment [16, 17]. For cancer pain, a stepwise approach to pharmacotherapy is generally advised on the World Health Organization (WHO) sequential three-step analgesic ladder from non-opioids to weak opioids, followed by strong opioids [17]. Opioids, however, are the mainstay and sometimes the first-line option for pain relief in cancer pain, as they offer the most effective analgesic therapy [7, 18, 19]. Recent clinical practice guidelines from the European Society for Medical Oncology and the European Association for Palliative Care (EAPC) state that low doses of morphine, oxycodone or a oxycodone–naloxone combination can be used as step II on the WHO ladder [7, 20]. Several medical societies have endorsed the use of opioids for non-cancer pain as a legitimate medical practice and have published guidelines for its safe use [21–23]. The value of opioid therapy, however, must be confirmed for each patient [24]. Morphine is generally the opioid of choice for the treatment of moderate-to-severe chronic pain [7, 20]. Nonetheless, in a systematic review for an update of the EAPC guidelines, oxycodone was shown to be as effective as morphine and hydromorphone for cancer pain in terms of analgesia and is one of the most commonly used strong opioids in Europe [7, 25].
Maximizing QoL is particularly important in individuals with chronic pain, who may already suffer reduced QoL because of their pain [26]. Several variables affect QoL, one of which is pharmacotherapy-related side effects. Despite the effectiveness of opioids, side effects compromise their therapeutic potential. Side effects may include nausea, central nervous system events such as confusion and hallucinations, pruritus and, particularly, opioid-induced bowel dysfunction (OIBD). OIBD includes a spectrum of symptoms, the primary one being opioid-induced constipation (OIC) [27–29]. With long-term use, many side effects associated with opioid therapy may subside because of tolerance; however, OIC often lingers and can significantly affect QoL [2, 28]. Optimal management of chronic pain with opioids requires a careful balance between efficacy and tolerability to ensure that QoL is maintained. One approach to overcome the gastrointestinal (GI) side effects of opioids is the use of opioid antagonists, such as naloxone, naloxegol, methylnaltrexone and alvimopan [27, 30]. Co-administration of oxycodone (a semi-synthetic opioid analgesic) with the opioid antagonist naloxone (OXN) has been demonstrated to counteract the GI side effects of the analgesic [31–33]. When administered orally, naloxone has low systemic bioavailability (<2 %) and antagonizes peripheral opioid receptors in the GI tract with little impact on centrally acting opioid analgesia [34]. In this review, we focus on the specific role of OXN in the management of chronic pain and OIC, and its impact on QoL and healthcare costs.
Opioid-Induced Bowel Dysfunction and Opioid-Induced Constipation
The GI tract plays a key role in the development of opioid-related adverse effects, as the main opioid receptors are widely distributed in the gut. Activation of GI opioid receptors by exogenous opioids disrupts GI motility and secretion, thereby inhibiting normal bowel function [35]. The primary adverse effects of opioids frequently lead to secondary complications, including a constellation of signs and symptoms known as OIBD. OIBD comprises constipation, anorexia, nausea and vomiting, gastro-oesophageal reflux, delayed digestion, abdominal pain, flatulence, bloating, hard stools, straining during bowel movement and incomplete evacuation. In some cases, it may lead to more serious complications such as bowel faecal impaction with overflow diarrhoea and faecal incontinence, pseudo-obstruction (which may cause anorexia, nausea and vomiting), disturbance of drug absorption, urine retention and urine incontinence. These symptoms further the impact on patients’ health [35–37] (Fig. 1).
Fig. 1.
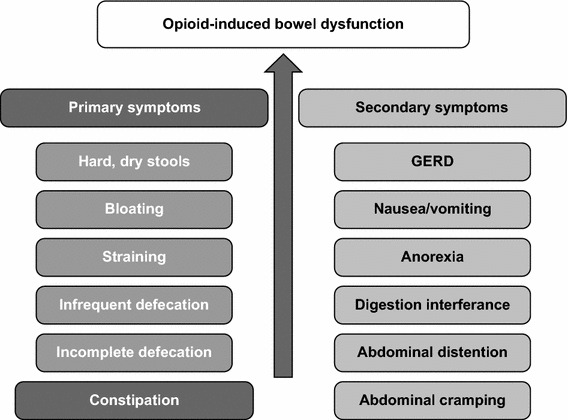
Primary and secondary symptoms and complications of opioid-induced bowel dysfunction. Adapted from Holzer [27]. GERD gastroesophageal reflux disease
OIC can have a particularly significant impact on patients’ lives [37, 38]. Estimates of the prevalence of constipation adverse effects in patients taking opioids varies because of variations in patient populations, differences in administration route, dose and type of the various opioids available [29, 39, 40]. In patients with chronic non-cancer pain, prevalence rates of up to 90 % have been reported [29, 41]. In patients with cancer, a large series of US hospice studies reported prevalence rates of 40–63 % [42]. In patients taking opioids, constipation has a serious impact on QoL and may be even more distressing for the patient than the pain itself [26, 43–45]. In a large international survey of patients taking opioids for >6 months, those suffering constipation were more likely to visit physicians, miss work and feel that their performance at work and their ability to undertake daily activities was impaired, compared with patients without constipation [46]. Some patients may even discontinue opioid therapy because of symptoms of constipation [47]. For these reasons, effective pharmacological therapy for OIC is considered to be an unmet need [48].
Bowel dysfunction in patients with OIC, however, is not caused exclusively by the opioid medication. Factors such as pain, medications other than opioids, diet and underlying disease factors play an important role in the complex pathophysiology of constipation [26].
Quality of Life in Patients with Chronic Pain
QoL is one of the most important patient-centred outcomes of medical care [49, 50] and is a multidimensional parameter for which coverage may be categorized within five dimensions: physical wellbeing, material wellbeing, social wellbeing, emotional wellbeing, and development and activity [51]. In some individuals, such as older patients with long-term illness and those with no possible cure, QoL can be the only achievable outcome [50]. In a comprehensive international study covering eight countries, it was demonstrated that suffering from a chronic condition substantially reduces QoL [52].
Chronic pain is a condition that has a significant impact on the social and working lives of sufferers and may even lead to social isolation. A large pan-European study involving >45,000 individuals suffering chronic pain reported that 21 % were diagnosed with depression due to their pain, 61 % were less able or unable to work outside the home, 19 % had lost their job and 13 % had changed jobs because of their pain [2]. A total of 60 % of these individuals had visited their doctor for their pain 2–9 times in the previous 6 months [2].
In addition to the reduced QoL due to pain, sufferers of moderate-to-severe pain treated with opioid medication often bear the additional burden of adverse effects of their pain medication, particularly OIC [28, 35]. Assessment of QoL in patients with OIC can assist therapeutic choices, and numerous assessment tools are available to assist in evaluation.
Tools to Measure Quality of Life in Patients with Chronic Pain and Constipation
Numerous tools are available to measure QoL in patients who have chronic pain and constipation. These tools can be general QoL measures that are designed to evaluate the effects of interventions or can be utility measures designed to evaluate judgements of health outcomes. A selection of QoL tools are listed in Table 1 [53].
Table 1.
Example instruments and tools available for measurement of quality of life in patients with pain and constipation
| General health-related measures |
| Medical Outcomes Study 36-Item Short-Form (SF-36) [93] |
| Medical Outcomes Study 12-Item Short-Form (SF-12) [94] |
| Nottingham Health Profile [95] |
| World Health Organization Quality of Life (WHOQOL) pain subscale [96] |
| Utility measuresa |
| EuroQoL [97] |
| Health Utilities Index (HUI) [98] |
| Cancer pain-specific tools |
| Functional Living Index-Cancer (FLIC) [99] |
| Functional Assessment of Cancer Therapy-General (FACT-G) [100] |
| European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC-QLQ) [101]b |
| Gastrointestinal-specific tools for constipation |
| Gastrointestinal Symptom Rating Scale (GSRS) [54] |
| Elderly Bowel Symptom Questionnaire (EBSQ) [55] |
| Patient Assessment of Constipation Symptoms (PAC-SYM) [56]c |
| Specific quality of life for constipation |
| Patient Assessment of Constipation Quality of Life (PAC-QoL) [57]d |
| Opioid-induced constipation-specific tools |
| Bowel Function Index (BFI) [58]e |
aSF-36 may be used as a utility measure
bDeveloped for use in international clinical trials
cA 12-item self-report instrument divided into abdominal, rectal and stool domains designed to assess symptom frequency and severity, and validated for use in patients with opioid-induced constipation
dIncludes four subscales: worries and concerns, physical discomfort, psychosocial discomfort and satisfaction
eThe BFI is owned by Mundipharma Laboratories GmbH, Switzerland (2002); the BFI is the subject of a European Patent Application (Publication No. EP 1,860,988) and corresponding patents and patent applications in other countries
Gastrointestinal-Specific Tools to Measure Quality of Life in Patients with Constipation
Several tools are available to measure GI symptoms in patients with constipation (Table 1). These include GI-specific tools for measuring constipation, including the Gastrointestinal Symptom Rating Scale [54], the Elderly Bowel Symptom Questionnaire [55] and the Patient Assessment of Constipation Symptoms (PAC-SYM) Questionnaire [56]. The Patient Assessment of Constipation Quality of Life (PAC-QoL) was designed to include specific QoL outcomes for constipation; was developed to address the need for a standardized, patient-reported outcomes measure to evaluate constipation over time; and includes specific QoL-related measures [57].
The Bowel Function Index: a Specific Validated Tool to Measure Bowel Function in Patients with Opioid-Induced Constipation
The Bowel Function Index (BFI1) is a validated, clinician-administered, patient-reported, three-item questionnaire (ease of defecation, feeling of incomplete evacuation and patient personal judgment of constipation) designed to evaluate OIC in cancer and non-cancer chronic pain patients [58–62]. Although not a direct measure of QoL, the BFI has been shown to be statistically correlated (r = 0.571; p < 0.0001) with the Knowles–Eccersley–Scott symptom score and matches up with PAC-QoL and, to a lesser extent, with the Medical Outcomes Study 12-Item Short-Form (SF-12) generic QoL questionnaire [63–65].
Generic Quality-of-Life Measures
Generic QoL measures are becoming increasingly popular with health policy makers. One advantage of generic measures is that they are non-disease specific, so they allow the policy maker to compare the impact of therapies relative to therapies in another disease area. These measures are commonly applied in resource allocation decisions as policy makers are allocating budgets across multiple and diverse disease areas. The Medical Outcomes Study 36-Item Short-Form Health Survey (SF-36) is one of the most widely used generic QoL measures and has been used in articles describing more than 200 diseases and conditions [66, 67]. One special type of QoL measure is the utility value, which is implied in health economic models based on a cost-utility analysis [68, 69]. Utility score health is usually measured between 0 and 1, where perfect health is given a score of 1 (although, in extreme cases, the score can go below 0, which is classified as ‘worse than death’). There have been a number of utility studies showing the reduction in QoL due to constipation. One study in patients with a severe non-curable disease and relatively short life expectancy treated with opioids found that QoL, as measured by the EuroQoL five-dimension questionnaire (EQ-5D), for those without advanced illness who were not constipated, was much higher (score 0.65) than the score for those who were constipated (score 0.31) [44]. In another study in patients with chronic functional constipation, a cost-utility model used to define health status in terms of the number of quality-adjusted life years (QALYs) demonstrated a 1 % improvement in health gain for one laxative over another [70].
Economic Burden of Constipation
The economic burden of constipation is vast in terms of both direct and indirect costs [26, 71]. Constipation-associated direct costs include physician visits, hospitalization, procedures and medications. Indirect costs include self-medication, lost earnings, restricted activity and costs of caregivers [26]. In studies conducted in the USA in patients receiving opioid treatment, direct costs have been demonstrated to be significantly higher in patients reporting GI events and constipation than in those not reporting such events [72, 73]. In an analysis of the costs of medical and pharmacy claims in patients receiving a new prescription for oral, short-acting opioid treatment, adjusted mean healthcare costs per patient during the 90 days after the opioid therapy were more than ten times higher for patients experiencing GI events than for those without GI events (US$40,133 versus US$3,981) [72]. A study conducted in Sweden in patients treated with strong opioids demonstrated that total (direct and indirect) costs are particularly high in patients with severe constipation, reaching €1,525 per patient per month, compared with €1,034 for patients with no constipation (Fig. 2) [74]. In cancer patients on opioid therapy, costs associated with constipation were found to be especially significant, with increases in total costs of 109 % for patients who had constipation, compared with those who did not [71]. Patients with constipation had significantly higher rates of concurrent use of ≥2 opioids, opioid discontinuation, opioid switching, nausea with vomiting, and respiratory depression than those without constipation. Patients with constipation also received more in-patient, hospice, home health, laboratory, other outpatient, emergency, office visit and nursing home care [71]. Resource utilization associated with the diagnosis and management of constipation is a significant cost driver, and constipation prevention programmes can lead to substantial cost savings [26].
Fig. 2.
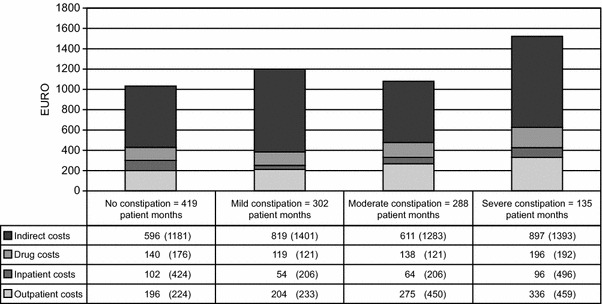
Mean cost per patient per month for opioid-treated patients with and without constipation [74]
Strategies for the Management of Opioid-Induced Constipation
Strategies for managing OIC include rotation or reduction in the dose of the opioid and management of OIC symptoms [75]. There are currently two main strategies for the management of OIC symptoms: the use of laxatives and treatment with opioid antagonists.
Laxatives
Laxatives are the most common therapeutic strategy for OIC [76]. Conventional laxatives either act by affecting osmotic condition, stimulate bowel action or have emollient properties. Inhibition of small-intestinal motility is a prominent feature of opioids and is probably one of the reasons why laxatives (which act predominantly on the colon) do not have a satisfactory effect on the majority of patients with OIC [40]. In addition, laxatives have the potential for over-use and dependency, and are associated with adverse effects such as dehydration, bloating and dermatitis [29, 37]. Despite the wide availability of different laxative types, OIC persists in many patients. In a multinational survey (PROBE 1), 45 % of patients receiving opioids and laxatives reported fewer than three bowel movements per week, 81 % of patients reported constipation and 58 % reported straining to pass a bowel movement [40]. In another study, 54 % of patients did not achieve a desired result with laxatives even half of the time. Despite the fact that OIC is usually treated with laxatives, there is insufficient clinical evidence that laxatives are efficacious in this indication [77]. In addition, it appears that laxatives are often not prescribed, even when they are indicated. In an audit of clinical practice in Scotland, 98 % of opioid prescriptions for patients needing an opioid were for the opioid alone [78].
Opioid Antagonists
Opioid antagonists aim to counteract the constipation effects of opioids by antagonizing the peripheral opioid receptors in the GI tract, while avoiding an impact on centrally acting opioid analgesia [79, 80]. The main challenge with opioid antagonists is to inhibit peripheral actions without affecting their central action, which may lead to opiate withdrawal symptoms or reversal of the analgesic effect [79]. Several single-entity opioid antagonists, including peripherally acting mu-opioid receptor antagonists (PAMORAs), are available and in development. These include methylnaltrexone, naloxegol and alvimopan [81]. Methylnaltrexone, which is administered as an emergency-relief subcutaneous injection when traditional oral laxatives fail, provides a bowel movement in approximately 50–60 % of patients but is approved only for palliative care in patients with advanced illness [75, 82]. Alvimopan, which is approved by the US Food and Drug Administration (FDA) and is indicated to accelerate the time to upper and lower GI recovery following surgeries that include partial bowel resection with primary anastomosis, has also been shown to increase spontaneous bowel movements, but cardiovascular safety concerns have hindered development of this agent [81]. Naloxegol has been recently approved by the FDA as an add-on to existing pain therapy and has shown efficacy for increasing bowel movements [81]. Oral formulations such as naloxegol and alvimopan provide obvious advantages over methylnaltrexone in terms of patient acceptance and QoL. However, these single-entity agents need to be combined with analgesic agents and therefore carry an intrinsic risk of lower patient compliance. Unfortunately, as yet, there are no directly comparative published data for these PAMORAs, but individual efficacy and safety analyses have been reviewed in several publications elsewhere [30, 81, 83, 84].
Naloxone is a competitive antagonist of peripheral opioid receptors with low systemic bioavailability and therefore has little impact on centrally acting opioid analgesia [34]. Naloxone, as part of a prolonged-release (PR) combination, reverses opioid-induced peripheral GI effects [85]. In patients with moderate-to-severe chronic pain, phase II and phase III randomized trials have demonstrated that combination PR oxycodone/naloxone (OXN PR) has equal analgesic efficacy and safety, but with improved bowel function, compared with oxycodone alone [31–33, 86, 87]. The beneficial effects of OXN PR, compared with oxycodone, have also been demonstrated over the long term (<52 weeks) [88].
Quality-of-Life Benefits of Oxycodone/Naloxone
In the trials evaluating OXN PR, bowel function and QoL were investigated using the BFI and other tools (Table 2). In the three phase III trials, conducted mainly in patients with moderate-to-severe non-malignant pain, significant improvements in the BFI were seen in patients taking OXN PR versus those taking oxycodone PR (Oxy PR) [31–33] (Table 2). These improvements were generally seen after just 1 week of treatment and continued throughout the trials [31–33]. Significant and clinically relevant improvements in symptoms, using PAC-SYM and the Patient Assessment of Opioid-Induced Constipation, were also demonstrated [31–33]. The improvements in bowel function were confirmed in a pooled analysis of the phase III trials, in which significant improvements were demonstrated in the BFI for patients receiving OXN PR, compared with those receiving Oxy PR [87]. Statistically significant and clinically relevant improvements in the BFI were observed at week 1 and at every subsequent time point during the study (−15.1; 95 % CI −17.3 to −13.0; p < 0.0001 versus baseline) (Fig. 3) [87]. Similar significant improvements in QoL in terms of the BFI were demonstrated in a randomized phase II study in patients with severe chronic pain [86]. A 52-week extension study, in which patients were maintained on OXN PR or switched from Oxy PR to OXN PR, demonstrated that these improvements in the BFI are maintained over the long term [88]. In an evaluation of OXN PR, using the generic QoL tool SF-36, significant improvements were seen in social functioning (p = 0.012), vitality (p = 0.010) and the general health subscale (p = 0.039) at week 12 [89].
Table 2.
Overview of quality-of-life-based outcomes using the Bowel Function Index (BFI) in prolonged-release (PR) oxycodone/naloxone (OXN) clinical trials
| Trial design | Population | Measure | Results | References | |
|---|---|---|---|---|---|
| 1. |
12-week, phase III RCT Combined OXN PR vs Oxy PR Primary objective: constipation as measured by BFI |
Moderate-to-severe non-malignant pain |
BFI PAC-SYM |
Significant improvement for OXN PR vs Oxy PR in BFI at 4 weeks (treatment difference: −14.9; p < 0.0001) Clinically relevant improvement in PAC-SYM Most common AEs were GI (24 % for OXN PR vs 16 % for Oxy PR) |
Lowenstein et al. [31] |
| 2. |
12-week, phase III RCT Combined OXN PR vs Oxy PR Primary objective: constipation as measured by BFI |
Moderate-to-severe non-cancer pain |
BFI PACOI |
Significant improvement for OXN PR vs Oxy PR in BFI at 4 weeks (treatment difference: −15.2; p < 0.0001) Significant improvement in PACOI at 4 weeks (p < 0.0001) Most common AEs were GI (19 % for OXN PR vs 30 % for Oxy PR) |
Simpson et al. [33] |
| 3. |
12-week, phase III RCT Combined OXN PR vs Oxy PR vs PBO Primary objective: analgesic efficacy Constipation, as measured by BFI, was an exploratory analysis |
Moderate-to-severe non-malignant lower back pain |
BFI BPI-SF (modified)a |
Clinically relevant improvement in BFI Mean BFI reduction vs baseline at 12 weeks: −23 for OXN PR vs −11 for Oxy PRb Significant improvement in BPI-SF for OXN PR (p = 0.0057) and Oxy PR (p = 0.0030) vs PBO Most common AEs were GI (23 % for OXN PR vs 27 % for Oxy PR) |
Vondrackova et al. [32] |
| 4. |
12-week, phase II RCT Oxy PR + naloxone PR vs Oxy PR Primary objectives: constipation, as measured by BFI, and analgesic efficacy, including optimal dose identification (oxycodone to naloxone ratio) |
Severe chronic non-cancer pain and cancer pain (2.5 % of patients) | BFI |
Significant improvement in BFI at 4 weeks (treatment difference: −16.2; p < 0.05 for 2:1 ratio group) Similar overall AEs between groups (62–70 %) |
Meissner et al. [86] |
| 5. |
Pooled analysis of trial 1 (Lowenstein et al. [31]) and trial 2 (Simpson et al. [33]) Primary objective: analgesic efficacy Constipation, as measured by BFI, was an secondary end point |
Moderate-to-severe non-malignant pain | BFI |
Significant improvement in BFI at 12 weeks (−15.1; p < 0.0001) Most common AEs were GI (21 %) |
Lowenstein et al. [87] |
| 6. |
Long-term (≤52 weeks) open-label extension of trial 3 (Vondrackova et al. [32]) and trial 2 (Simpson et al. [33]) OXN PR No primary end point was set. Analgesic efficacy and constipation, as measured by BFI, were assessed as two separate analysesc |
Moderate-to-severe non-cancer pain | BFI |
BFI reduction vs baseline at 12 months (−15) Most common AEs were infections and infestations (40 %) |
Sander-Kiesling et al. [88] |
AE adverse event, BFI Bowel Function Index, BPI Brief Pain Inventory, GI gastrointestinal, Oxy oxycodone, PACOI Patient Assessment of Opioid-Induced Constipation (sum score of the rectal and stools subscale score of PAC-SYM), PAC-SYM Patient Assessment of Constipation Symptoms, PBO placebo, RCT randomized controlled trial, SF Short Form
aBPI interference of pain with sleep item
bSubgroup of patients with BFI ≥50 at baseline; no statistical analysis was conducted on this patient population
cResults for BFI analysis are reported here
Fig. 3.
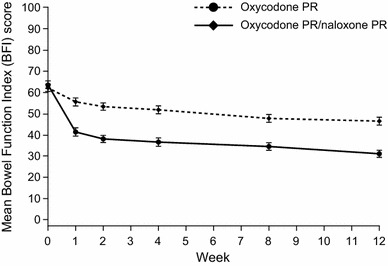
Mean Bowel Function Index score over time in patients treated with prolonged-release (PR) oxycodone/naloxone and oxycodone PR (pooled analysis of two randomized phase III trials) [87]
In real-world clinical practice, a large non-interventional study reported improvements in QoL for patients taking OXN PR [90]. In this German study involving 7,836 individuals with severe chronic pain of various aetiologies treated with OXN PR, significant reductions in the BFI were seen both in opioid-naïve and opioid pre-treated patients [90]. QoL, as measured by the Short-Form Brief Pain Inventory (BPI-SF), demonstrated an improvement of 43 % in mean overall QoL (in 2,023 individuals) after just 4 weeks of treatment [90].
Health Economic Benefits of Oxycodone/Naloxone
A UK comparison of the cost effectiveness of OXN PR versus Oxy PR, using a cost-utility model combining the costs of pain therapy, laxatives and other resources to manage constipated patients with moderate-to-severe non-malignant pain, demonstrated a QoL gain with OXN PR versus Oxy PR [89]. The incremental gain in QALYs was based on the SF-36 results mapped to EQ-5D and implied that patients treated with OXN PR in real-world clinical practice will experience a QoL gain. Treatment costs were slightly higher for OXN PR, but this was offset by increased QALY gains. The ratio of incremental cost to incremental QALY gain was substantially below commonly applied decision thresholds in the UK, suggesting that OXN PR is cost effective and should be adopted in the health system [89]. In some scenarios, total cost savings to the health system have been demonstrated. In a German study in which patients received OXN PR or other strong (WHO step III) opioids at 6 months, direct treatment costs were lower in the OXN PR cohort, and there was a QALY gain, compared with the cohort taking other strong opioids [91]. Similar results were seen in a Spanish study in patients with chronic severe pain and OIC, in which an incremental cost-effectiveness ratio far below the efficiency threshold commonly cited in Spain was demonstrated [92].
Conclusions
Management of chronic pain is challenging, and unmet needs remain. Traditional opioids are often the treatment of choice in moderate and severe pain, but their use is often compromised by side effects; in particular, OIBD can substantially reduce QoL. Most treatments for OIBD are not very effective and are not mechanism based. OXN is one of the first therapies to act directly on the mechanism of OIBD and has been shown to be an effective treatment for this (to date, unmet) need.
The BFI is a specific validated measurement tool that can assist in the evaluation of bowel function in patients with OIBD, and specifically with OIC. Treatment of patients with moderate-to-severe pain with OXN PR has been demonstrated to significantly improve QoL. Use of OXN PR should allow more patients to benefit from the analgesic efficacy of opioid therapy and should minimize the side effects of constipation, with a corresponding improvement in QoL. Health economic analysis has shown OXN PR to be below commonly applied thresholds, with some cost offsets because of fewer resources being required to treat the constipation.
Disclosures and Acknowledgments
Bart Morlion has received consulting fees or honoraria from Johnson & Johnson and Grünenthal, and lecture fees from Pfizer, Mundipharma, Grünenthal and Lilly. Katri Elina Clemens has received lecture fees and honoraria from Mundipharma GmbH & Co. KG. Bart Morlion and Katri Elina Clemens have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. Will Dunlop is an employee of Mundipharma International.
Medical writing assistance was provided by Joan Thomas, Ph.D., of Discovery London and was funded by Mundipharma Research GmbH & Co. KG.
Footnotes
The BFI is owned by Mundipharma Laboratories GmbH, Switzerland (2002); the BFI is the subject of a European Patent Application (Publication No. EP 1,860,988) and corresponding patents and patent applications in other countries.
References
- 1.Blyth FM, et al. Chronic pain in Australia: a prevalence study. Pain. 2001;89(2–3):127–134. doi: 10.1016/S0304-3959(00)00355-9. [DOI] [PubMed] [Google Scholar]
- 2.Breivik H, et al. Survey of chronic pain in Europe: prevalence, impact on daily life, and treatment. Eur J Pain. 2006;10(4):287–333. doi: 10.1016/j.ejpain.2005.06.009. [DOI] [PubMed] [Google Scholar]
- 3.Johannes CB, et al. The prevalence of chronic pain in United States adults: results of an Internet-based survey. J Pain. 2010;11(11):1230–1239. doi: 10.1016/j.jpain.2010.07.002. [DOI] [PubMed] [Google Scholar]
- 4.Toblin RL, et al. A population-based survey of chronic pain and its treatment with prescription drugs. Pain. 2011;152(6):1249–1255. doi: 10.1016/j.pain.2010.12.036. [DOI] [PubMed] [Google Scholar]
- 5.Vetter TR. A primer on health-related quality of life in chronic pain medicine. Anesth Analg. 2007;104(3):703–718. doi: 10.1213/01.ane.0000255290.64837.61. [DOI] [PubMed] [Google Scholar]
- 6.Pain., I.A.f.t.S.o., Pain, 1986;Suppl 3:S1–S225.
- 7.Ripamonti CI, et al. Management of cancer pain: ESMO Clinical Practice Guidelines. Ann Oncol. 2012;23(Suppl 7):7139–7154. doi: 10.1093/annonc/mds233. [DOI] [PubMed] [Google Scholar]
- 8.Colvin L, Forbes K, Fallon M. Difficult pain. BMJ. 2006;332(7549):1081–1083. doi: 10.1136/bmj.332.7549.1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Breivik H, et al. Cancer-related pain: a pan-European survey of prevalence, treatment, and patient attitudes. Ann Oncol. 2009;20(8):1420–1433. doi: 10.1093/annonc/mdp001. [DOI] [PubMed] [Google Scholar]
- 10.Apkarian AV, Baliki MN, Geha PY. Towards a theory of chronic pain. Prog Neurobiol. 2009;87(2):81–97. doi: 10.1016/j.pneurobio.2008.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gatchel RJ, et al. The biopsychosocial approach to chronic pain: scientific advances and future directions. Psychol Bull. 2007;133(4):581–624. doi: 10.1037/0033-2909.133.4.581. [DOI] [PubMed] [Google Scholar]
- 12.Ohayon MM, Schatzberg AF. Using chronic pain to predict depressive morbidity in the general population. Arch Gen Psychiatry. 2003;60(1):39–47. doi: 10.1001/archpsyc.60.1.39. [DOI] [PubMed] [Google Scholar]
- 13.Bair MJ, et al. Depression and pain comorbidity: a literature review. Arch Intern Med. 2003;163(20):2433–2445. doi: 10.1001/archinte.163.20.2433. [DOI] [PubMed] [Google Scholar]
- 14.Nicholson B, Verma S. Comorbidities in chronic neuropathic pain. Pain Med. 2004;5(Suppl 1):S9–S27. doi: 10.1111/j.1526-4637.2004.04019.x. [DOI] [PubMed] [Google Scholar]
- 15.Marchand F, Perretti M, McMahon SB. Role of the immune system in chronic pain. Nat Rev Neurosci. 2005;6(7):521–532. doi: 10.1038/nrn1700. [DOI] [PubMed] [Google Scholar]
- 16.Morlion B. Chronic low back pain: pharmacological, interventional and surgical strategies. Nat Rev Neurol. 2013;9(8):462–473. doi: 10.1038/nrneurol.2013.130. [DOI] [PubMed] [Google Scholar]
- 17.Kroenke K, Krebs EE, Bair MJ. Pharmacotherapy of chronic pain: a synthesis of recommendations from systematic reviews. Gen Hosp Psychiatry. 2009;31(3):206–219. doi: 10.1016/j.genhosppsych.2008.12.006. [DOI] [PubMed] [Google Scholar]
- 18.Colson J, et al. A systematic review of observational studies on the effectiveness of opioid therapy for cancer pain. Pain Physician. 2011;14(2):E85–E102. [PubMed] [Google Scholar]
- 19.Manchikanti L, et al. Effectiveness of long-term opioid therapy for chronic non-cancer pain. Pain Physician. 2011;14(2):E133–E156. [PubMed] [Google Scholar]
- 20.Caraceni A, et al. Use of opioid analgesics in the treatment of cancer pain: evidence-based recommendations from the EAPC. Lancet Oncol. 2012;13(2):e58–e68. doi: 10.1016/S1470-2045(12)70040-2. [DOI] [PubMed] [Google Scholar]
- 21.de Leon-Casasola OA. Opioids for chronic pain: new evidence, new strategies, safe prescribing. Am J Med. 2013;126(3 Suppl 1):S3–S11. doi: 10.1016/j.amjmed.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 22.Practice guidelines for chronic pain management: an updated report by the American Society of Anesthesiologists Task Force on Chronic Pain Management and the American Society of Regional Anesthesia and Pain Medicine. Anesthesiology, 2010;112(4):810–33. [DOI] [PubMed]
- 23.Franklin GM. Opioids for chronic noncancer pain: a position paper of the American Academy of Neurology. Neurology. 2014;83(14):1277–1284. doi: 10.1212/WNL.0000000000000839. [DOI] [PubMed] [Google Scholar]
- 24.Baldini A, Von Korff M, Lin EH. A review of potential adverse effects of long-term opioid therapy: a practitioner’s guide. Prim Care Companion CNS Disord. 2012. 14(3). [DOI] [PMC free article] [PubMed]
- 25.Caraceni A, Pigni A, Brunelli C. Is oral morphine still the first choice opioid for moderate to severe cancer pain? A systematic review within the European Palliative Care Research Collaborative guidelines project. Palliat Med. 2011;25(5):402–409. doi: 10.1177/0269216310392102. [DOI] [PubMed] [Google Scholar]
- 26.Dennison C, et al. The health-related quality of life and economic burden of constipation. Pharmacoeconomics. 2005;23(5):461–476. doi: 10.2165/00019053-200523050-00006. [DOI] [PubMed] [Google Scholar]
- 27.Holzer P. Opioid antagonists for prevention and treatment of opioid-induced gastrointestinal effects. Curr Opin Anaesthesiol. 2010;23(5):616–622. doi: 10.1097/ACO.0b013e32833c3473. [DOI] [PubMed] [Google Scholar]
- 28.Holzer P, et al. Opioid-induced bowel dysfunction in cancer-related pain: causes, consequences, and a novel approach for its management. J Opioid Manag. 2009;5(3):145–151. doi: 10.5055/jom.2009.0015. [DOI] [PubMed] [Google Scholar]
- 29.Panchal SJ, Muller-Schwefe P, Wurzelmann JI. Opioid-induced bowel dysfunction: prevalence, pathophysiology and burden. Int J Clin Pract. 2007;61(7):1181–1187. doi: 10.1111/j.1742-1241.2007.01415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chey WD, et al. Naloxegol for opioid-induced constipation in patients with noncancer pain. N Engl J Med. 2014;370(25):2387–2396. doi: 10.1056/NEJMoa1310246. [DOI] [PubMed] [Google Scholar]
- 31.Lowenstein O, et al. Combined prolonged-release oxycodone and naloxone improves bowel function in patients receiving opioids for moderate-to-severe non-malignant chronic pain: a randomised controlled trial. Expert Opin Pharmacother. 2009;10(4):531–543. doi: 10.1517/14656560902796798. [DOI] [PubMed] [Google Scholar]
- 32.Vondrackova D, et al. Analgesic efficacy and safety of oxycodone in combination with naloxone as prolonged release tablets in patients with moderate to severe chronic pain. J Pain. 2008;9(12):1144–1154. doi: 10.1016/j.jpain.2008.06.014. [DOI] [PubMed] [Google Scholar]
- 33.Simpson K, et al. Fixed-ratio combination oxycodone/naloxone compared with oxycodone alone for the relief of opioid-induced constipation in moderate-to-severe noncancer pain. Curr Med Res Opin. 2008;24(12):3503–3512. doi: 10.1185/03007990802584454. [DOI] [PubMed] [Google Scholar]
- 34.Smith K, et al. Low absolute bioavailability of oral naloxone in healthy subjects. Int J Clin Pharmacol Ther. 2012;50(5):360–367. doi: 10.5414/CP201646. [DOI] [PubMed] [Google Scholar]
- 35.Mehendale SR, Yuan CS. Opioid-induced gastrointestinal dysfunction. Dig Dis. 2006;24(1–2):105–112. doi: 10.1159/000090314. [DOI] [PubMed] [Google Scholar]
- 36.Leppert W. Are we able to manage effectively opioid-induced bowel dysfunction? J Autacoids. 2012(1):1000e113.
- 37.Pappagallo M. Incidence, prevalence, and management of opioid bowel dysfunction. Am J Surg. 2001;182(5A Suppl):11S–18S. doi: 10.1016/S0002-9610(01)00782-6. [DOI] [PubMed] [Google Scholar]
- 38.Wald A, et al. The burden of constipation on quality of life: results of a multinational survey. Aliment Pharmacol Ther. 2007;26(2):227–236. doi: 10.1111/j.1365-2036.2007.03376.x. [DOI] [PubMed] [Google Scholar]
- 39.Rosti G, et al. Opioid-related bowel dysfunction: prevalence and identification of predictive factors in a large sample of Italian patients on chronic treatment. Eur Rev Med Pharmacol Sci. 2010;14(12):1045–1050. [PubMed] [Google Scholar]
- 40.Bell TJ, et al. The prevalence, severity, and impact of opioid-induced bowel dysfunction: results of a US and European Patient Survey (PROBE 1) Pain Med. 2009;10(1):35–42. doi: 10.1111/j.1526-4637.2008.00495.x. [DOI] [PubMed] [Google Scholar]
- 41.Allan L, et al. Randomised crossover trial of transdermal fentanyl and sustained release oral morphine for treating chronic non-cancer pain. BMJ. 2001;322(7295):1154–1158. doi: 10.1136/bmj.322.7295.1154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.McMillan SC. Assessing and managing opiate-induced constipation in adults with cancer. Cancer Control. 2004;11(3 Suppl):3–9. doi: 10.1177/10732748040110S302. [DOI] [PubMed] [Google Scholar]
- 43.Fallon MT, Hanks GW. Morphine, constipation and performance status in advanced cancer patients. Palliat Med. 1999;13(2):159–160. doi: 10.1191/026921699677653615. [DOI] [PubMed] [Google Scholar]
- 44.Penning-van Beest FJ, et al. Quality of life in relation to constipation among opioid users. J Med Econ. 2010;13(1):129–135. doi: 10.3111/13696990903584436. [DOI] [PubMed] [Google Scholar]
- 45.McMillan SC, Small BJ. Symptom distress and quality of life in patients with cancer newly admitted to hospice home care. Oncol Nurs Forum. 2002;29(10):1421–1428. doi: 10.1188/02.ONF.1421-1428. [DOI] [PubMed] [Google Scholar]
- 46.Bell T, Annunziata K, Leslie JB. Opioid-induced constipation negatively impacts pain management, productivity, and health-related quality of life: findings from the National Health and Wellness Survey. J Opioid Manag. 2009;5(3):137–144. doi: 10.5055/jom.2009.0014. [DOI] [PubMed] [Google Scholar]
- 47.Kurz A, Sessler DI. Opioid-induced bowel dysfunction: pathophysiology and potential new therapies. Drugs. 2003;63(7):649–671. doi: 10.2165/00003495-200363070-00003. [DOI] [PubMed] [Google Scholar]
- 48.Ketwaroo GA, Cheng V, Lembo A. Opioid-induced bowel dysfunction. Curr Gastroenterol Rep. 2013;15(9):344. doi: 10.1007/s11894-013-0344-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Leplege A, Hunt S. The problem of quality of life in medicine. JAMA. 1997;278(1):47–50. doi: 10.1001/jama.1997.03550010061041. [DOI] [PubMed] [Google Scholar]
- 50.Netuveli G, et al. Functional limitation in long standing illness and quality of life: evidence from a national survey. BMJ. 2005;331(7529):1382–1383. doi: 10.1136/bmj.331.7529.1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Felce D, Perry J. Quality of life: its definition and measurement. Res Dev Disabil. 1995;16(1):51–74. doi: 10.1016/0891-4222(94)00028-8. [DOI] [PubMed] [Google Scholar]
- 52.Alonso J, et al. Health-related quality of life associated with chronic conditions in eight countries: results from the International Quality of Life Assessment (IQOLA) Project. Qual Life Res. 2004;13(2):283–298. doi: 10.1023/B:QURE.0000018472.46236.05. [DOI] [PubMed] [Google Scholar]
- 53.Bonomi AE, Shikiar R, Legro MW. Quality-of-life assessment in acute, chronic, and cancer pain: a pharmacist’s guide. J Am Pharm Assoc (Wash) 2000;40(3):402–416. doi: 10.1016/s1086-5802(16)31089-0. [DOI] [PubMed] [Google Scholar]
- 54.Svedlund J, Sjodin I, Dotevall G. GSRS—a clinical rating scale for gastrointestinal symptoms in patients with irritable bowel syndrome and peptic ulcer disease. Dig Dis Sci. 1988;33(2):129–134. doi: 10.1007/BF01535722. [DOI] [PubMed] [Google Scholar]
- 55.O’Keefe EA, et al. A bowel symptom questionnaire for the elderly. J Gerontol. 1992;47(4):M116–M121. doi: 10.1093/geronj/47.4.M116. [DOI] [PubMed] [Google Scholar]
- 56.Slappendel R, et al. Validation of the PAC-SYM questionnaire for opioid-induced constipation in patients with chronic low back pain. Eur J Pain. 2006;10(3):209–217. doi: 10.1016/j.ejpain.2005.03.008. [DOI] [PubMed] [Google Scholar]
- 57.Marquis P, et al. Development and validation of the Patient Assessment of Constipation Quality of Life questionnaire. Scand J Gastroenterol. 2005;40(5):540–551. doi: 10.1080/00365520510012208. [DOI] [PubMed] [Google Scholar]
- 58.Rentz AM, et al. Validation of the Bowel Function Index to detect clinically meaningful changes in opioid-induced constipation. J Med Econ. 2009;12(4):371–383. doi: 10.3111/13696990903430481. [DOI] [PubMed] [Google Scholar]
- 59.Rentz AM, et al. Observational, nonintervention, multicenter study for validation of the Bowel Function Index for constipation in European countries. Curr Med Res Opin. 2011;27(1):35–44. doi: 10.1185/03007995.2010.535270. [DOI] [PubMed] [Google Scholar]
- 60.Coffin B, Causse C. Constipation assessment scales in adults: a literature review including the new Bowel Function Index. Expert Rev Gastroenterol Hepatol. 2011;5(5):601–613. doi: 10.1586/egh.11.53. [DOI] [PubMed] [Google Scholar]
- 61.Ueberall MA, et al. The Bowel Function Index for evaluating constipation in pain patients: definition of a reference range for a non-constipated population of pain patients. J Int Med Res. 2011;39(1):41–50. doi: 10.1177/147323001103900106. [DOI] [PubMed] [Google Scholar]
- 62.Ducrotte P, Causse C. The Bowel Function Index: a new validated scale for assessing opioid-induced constipation. Curr Med Res Opin. 2012;28(3):457–466. doi: 10.1185/03007995.2012.657301. [DOI] [PubMed] [Google Scholar]
- 63.Knowles CH, et al. Linear discriminant analysis of symptoms in patients with chronic constipation: validation of a new scoring system (KESS) Dis Colon Rectum. 2000;43(10):1419–1426. doi: 10.1007/BF02236639. [DOI] [PubMed] [Google Scholar]
- 64.Abramowitz L, et al. Further validation of the psychometric properties of the Bowel Function Index for evaluating opioid-induced constipation (OIC) J Med Econ. 2013;16(12):1434–1441. doi: 10.3111/13696998.2013.851083. [DOI] [PubMed] [Google Scholar]
- 65.Abramowitz L, et al. Prevalence and impact of constipation and bowel dysfunction induced by strong opioids: a cross-sectional survey of 520 patients with cancer pain: DYONISOS study. J Med Econ. 2013;16(12):1423–1433. doi: 10.3111/13696998.2013.851082. [DOI] [PubMed] [Google Scholar]
- 66.Garratt A, et al. Quality of life measurement: bibliographic study of patient assessed health outcome measures. BMJ. 2002;324(7351):1417. doi: 10.1136/bmj.324.7351.1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ware JE, Jr, Sherbourne SD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473–483. doi: 10.1097/00005650-199206000-00002. [DOI] [PubMed] [Google Scholar]
- 68.Feeny D. A utility approach to the assessment of health-related quality of life. Med Care. 2000;38(9 Suppl):2151–2154. doi: 10.1097/00005650-200009002-00022. [DOI] [PubMed] [Google Scholar]
- 69.Torrance GW. Utility approach to measuring health-related quality of life. J Chronic Dis. 1987;40(6):593–603. doi: 10.1016/0021-9681(87)90019-1. [DOI] [PubMed] [Google Scholar]
- 70.Guest JF, Clegg JP, Helter MT. Cost-effectiveness of macrogol 4000 compared to lactulose in the treatment of chronic functional constipation in the UK. Curr Med Res Opin. 2008;24(7):1841–1852. doi: 10.1185/03007990802102349. [DOI] [PubMed] [Google Scholar]
- 71.Candrilli SD, Davis KL, Iyer S. Impact of constipation on opioid use patterns, health care resource utilization, and costs in cancer patients on opioid therapy. J Pain Palliat Care Pharmacother. 2009;23(3):231–241. doi: 10.1080/15360280903098440. [DOI] [PubMed] [Google Scholar]
- 72.Kwong WJ, Diels J, Kavanagh S. Costs of gastrointestinal events after outpatient opioid treatment for non-cancer pain. Ann Pharmacother. 2010;44(4):630–640. doi: 10.1345/aph.1M520. [DOI] [PubMed] [Google Scholar]
- 73.Iyer S, Davis KL, Candrilli S. Opioid use patterns and health care resource utilization in patients prescribed opioid therapy with and without constipation. Manag Care. 2010;19(3):44–51. [PubMed] [Google Scholar]
- 74.Hjalte F, et al. The direct and indirect costs of opioid-induced constipation. J Pain Symptom Manage. 2010;40(5):696–703. doi: 10.1016/j.jpainsymman.2010.02.019. [DOI] [PubMed] [Google Scholar]
- 75.Swegle JM, Logemann C. Management of common opioid-induced adverse effects. Am Fam Physician. 2006;74(8):1347–1354. [PubMed] [Google Scholar]
- 76.Tamayo AC, Diaz-Zuluaga PA. Management of opioid-induced bowel dysfunction in cancer patients. Support Care Cancer. 2004;12(9):613–618. doi: 10.1007/s00520-004-0649-7. [DOI] [PubMed] [Google Scholar]
- 77.Holzer P. Non-analgesic effects of opioids: management of opioid-induced constipation by peripheral opioid receptor antagonists: prevention or withdrawal? Curr Pharm Des. 2012;18(37):6010–6020. doi: 10.2174/138161212803582388. [DOI] [PubMed] [Google Scholar]
- 78.Lanza P, Carey M. The impact of opioid and laxative prescribing habits on constipation in the primary care setting before and after the introduction of SIGN 44: control of pain in patients with cancer. Primary Health Care Res Dev. 2006;7:3–9. doi: 10.1191/1463423606pc267oa. [DOI] [Google Scholar]
- 79.Camilleri M. Opioid-induced constipation: challenges and therapeutic opportunities. Am J Gastroenterol. 2011;106(5):835–842. doi: 10.1038/ajg.2011.30. [DOI] [PubMed] [Google Scholar]
- 80.Choi YS, Billings JA. Opioid antagonists: a review of their role in palliative care, focusing on use in opioid-related constipation. J Pain Symptom Manage. 2002;24(1):71–90. doi: 10.1016/S0885-3924(02)00424-4. [DOI] [PubMed] [Google Scholar]
- 81.Poulsen JL, et al. Clinical potential of naloxegol in the management of opioid-induced bowel dysfunction. Clin Exp Gastroenterol. 2014;7:345–358. doi: 10.2147/CEG.S52097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bader S, Durk T, Becker G. Methylnaltrexone for the treatment of opioid-induced constipation. Expert Rev Gastroenterol Hepatol. 2013;7(1):13–26. doi: 10.1586/egh.12.63. [DOI] [PubMed] [Google Scholar]
- 83.Diego L, et al. Novel opioid antagonists for opioid-induced bowel dysfunction. Expert Opin Investig Drugs. 2011;20(8):1047–1056. doi: 10.1517/13543784.2011.592830. [DOI] [PubMed] [Google Scholar]
- 84.Ford AC, Brenner DM, Schoenfeld PS. Efficacy of pharmacological therapies for the treatment of opioid-induced constipation: systematic review and meta-analysis. Am J Gastroenterol. 2013;108(10):1566–1574. doi: 10.1038/ajg.2013.169. [DOI] [PubMed] [Google Scholar]
- 85.Smith K, et al. Naloxone as part of a prolonged release oxycodone/naloxone combination reduces oxycodone-induced slowing of gastrointestinal transit in healthy volunteers. Expert Opin Investig Drugs. 2011;20(4):427–439. doi: 10.1517/13543784.2011.563236. [DOI] [PubMed] [Google Scholar]
- 86.Meissner W, et al. A randomised controlled trial with prolonged-release oral oxycodone and naloxone to prevent and reverse opioid-induced constipation. Eur J Pain. 2009;13(1):56–64. doi: 10.1016/j.ejpain.2008.06.012. [DOI] [PubMed] [Google Scholar]
- 87.Lowenstein O, et al. Efficacy and safety of combined prolonged-release oxycodone and naloxone in the management of moderate/severe chronic non-malignant pain: results of a prospectively designed pooled analysis of two randomised, double-blind clinical trials. BMC Clin Pharmacol. 2010;10:12. doi: 10.1186/1472-6904-10-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sandner-Kiesling A, et al. Long-term efficacy and safety of combined prolonged-release oxycodone and naloxone in the management of non-cancer chronic pain. Int J Clin Pract. 2010;64(6):763–774. doi: 10.1111/j.1742-1241.2010.02360.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Dunlop W, et al. Quality of life benefits and cost impact of prolonged release oxycodone/naloxone versus prolonged release oxycodone in patients with moderate-to-severe non-malignant pain and opioid-induced constipation: a UK cost-utility analysis. J Med Econ. 2012;15(3):564–575. doi: 10.3111/13696998.2012.665279. [DOI] [PubMed] [Google Scholar]
- 90.Schutter U, et al. Innovative pain therapy with a fixed combination of prolonged-release oxycodone/naloxone: a large observational study under conditions of daily practice. Curr Med Res Opin. 2010;26(6):1377–1387. doi: 10.1185/03007991003787318. [DOI] [PubMed] [Google Scholar]
- 91.Rychlik R, Kiencke P, Kresimon J. Healthcare research study into quality of life and pharmacoeconomic aspects of patients with chronic back pain being treated with oxycodone/naloxone or other WHO step opiods. Interim analysis. Article in German. Gesundh Okon Qual Manag. 2011;16:10–19. doi: 10.1055/s-0029-1246102. [DOI] [Google Scholar]
- 92.Galvez R, Provencio M, Sanz-Ortiz J. Análisis económico de oxicodona LP/naloxona LP en el manejo del dolor intenso y el estreñimiento asociado al tratamiento con opioides en España. Pharmacoecon Span Res Arti. 2012;9(1):21–32. [Google Scholar]
- 93.McHorney CA, Ware JE, Jr, Raczek AE. The MOS 36-Item Short-Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med Care. 1993;31(3):247–263. doi: 10.1097/00005650-199303000-00006. [DOI] [PubMed] [Google Scholar]
- 94.Ware J, Jr, Kosinski M, Keller SD. A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care. 1996;34(3):220–233. doi: 10.1097/00005650-199603000-00003. [DOI] [PubMed] [Google Scholar]
- 95.Hunt SM, et al. The Nottingham Health Profile: subjective health status and medical consultations. Soc Sci Med A. 1981;15(3 Pt 1):221–229. doi: 10.1016/0271-7123(81)90005-5. [DOI] [PubMed] [Google Scholar]
- 96.The World Health Organization Quality of Life Assessment (WHOQOL): development and general psychometric properties. Soc Sci Med. 1998;46(12):1569–85. [DOI] [PubMed]
- 97.EuroQol—a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199–208. [DOI] [PubMed]
- 98.Feeny D, et al. Multi-attribute health status classification systems. Health Utilities Index. Pharmacoeconomics. 1995;7(6):490–502. doi: 10.2165/00019053-199507060-00004. [DOI] [PubMed] [Google Scholar]
- 99.Schipper H, et al. Measuring the quality of life of cancer patients: the Functional Living Index-Cancer: development and validation. J Clin Oncol. 1984;2(5):472–483. doi: 10.1200/JCO.1984.2.5.472. [DOI] [PubMed] [Google Scholar]
- 100.Cella DF, et al. The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J Clin Oncol. 1993;11(3):570–579. doi: 10.1200/JCO.1993.11.3.570. [DOI] [PubMed] [Google Scholar]
- 101.Aaronson NK, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365–376. doi: 10.1093/jnci/85.5.365. [DOI] [PubMed] [Google Scholar]


