Abstract
Obesity and type 2 diabetes mellitus (T2DM) often occur together and affect a growing number of individuals in both the developed and developing worlds. Both are associated with a number of other serious illnesses that lead to increased rates of mortality. There is likely a polygenic mode of inheritance underlying both disorders, but it has become increasingly clear that the pre- and postnatal environments play critical roles in pushing predisposed individuals over the edge into a disease state. This review focuses on the many genetic and environmental variables that interact to cause predisposed individuals to become obese and diabetic. The brain and its interactions with the external and internal environment are a major focus given the prominent role these interactions play in the regulation of energy and glucose homeostasis in health and disease.
I. INTRODUCTION
A. The Problem: Obesity, Diabetes, and Their Interactions
Obesity is defined as having an excess of body fat (267). However, for practical clinical and research purposes, obesity and overweight are most commonly defined by body mass index (BMI), the ratio of weight in kilograms divided by height in meters squared. Overweight is defined as a BMI of 25–29.9 kg/m2 and obesity as a BMI of >30 kg/m2. The category of obesity is further divided into subcategories of class I (BMI 30.0–34.9 kg/m2), class II (BMI 35.0–39.9 kg/m2), and class III (BMI ≥40 kg/m2) (355a). However, BMI does not provide a good measure of carcass adiposity, which is the true definition of obesity. For that reason, other measures such as waist circumference, waist-hip ratio, as well as percent body fat using DEXA, CT, and MRI have also been used (153). Such additional measures may be important because so-called normal weight obesity, the combination of normal BMI and high body fat content, is associated with a high prevalence of cardiometabolic dysregulation, metabolic syndrome, and cardiovascular risk and/or mortality factors (446). However, these other measures are not as widely used for a variety of reasons, including the expense of imaging equipment.
Diabetes is a chronic disease that is defined by hyperglycemia and is a heterogeneous condition that can be subdivided into a number of classes. Type 1 diabetes mellitus (T1DM) accounts for ∼5% of all cases of diabetes. It usually occurs in childhood or adolescence and is generally considered to be an autoimmune disease which attacks the pancreatic β-cells leading to profound hypoinsulinemia (222, 459). Type 2 diabetes mellitus (T2DM) is the most common form, accounting for almost 90% of cases. It usually occurs later in life, often, but not always, in association with obesity and almost always associated with early-onset peripheral insulin resistance and later β-cell dysfunction and insulin deficiency (110, 430). Its etiology is less well characterized and, along with T1DM, may well have an underlying genetic predisposition (22, 122, 126, 525). Specific diagnostic criteria have been defined by a number of organizations, including the World Health Organization and the American Diabetes Association. These are either a hemoglobin A1C of ≥6.5%, or fasting plasma glucose of ≥126 mg/dl, or a plasma glucose concentration of ≥200 mg/dl 2 h after a 75 g oral glucose tolerance test or a random plasma glucose measurement ≥200 mg/dl (www.diabetes.org). There are currently estimated to be around 382 million individuals worldwide that have diabetes. In the United States (US) alone, 25.8 million individuals (8.3% of the population) have diabetes. It was estimated that diabetes caused at least 548 billion dollars in health expenditure in 2013, and this figure is set to continue growing (International Diabetes Federation). Understanding the factors driving this increase is therefore of great economic and social importance.
B. Prevalence and Associated Morbidity and Mortality of Obesity
The prevalence of obesity and overweight in the United States is high. In 2007–2008, 32% of US men and 36% of US women were obese, and an additional 40% of men and 28% of women were overweight (149). In 2010, more than one-third of US children and adolescents were overweight or obese (368). About 5% of Americans have a class III obesity, i.e., a BMI of >40 kg/m2 (149). The prevalence of obesity and overweight has increased by 134 and 48%, respectively, since 1976–1980 (492). While overweight and obesity trends among women have remained stable, rates in men have continued to rise (149) with a 50 and 25% long-term risk of developing these conditions, respectively, in the Framingham study (531). These figures vary widely among sex, ethnic, and racial groups (149), as does the relationship between BMI and disease risk such that obesity prevalence is not a definite predictor of the degree of disease risk.
In general, obesity reduces life expectancy by 6–20 yr depending on age and race (152, 397), particularly among adults below the age of 65 (4, 114, 151, 152, 422). Cardiovascular disease, T2DM, cancer, and respiratory diseases are the leading causes of death in obese individuals (422). It is less clear whether being overweight carries the same increased mortality risk (4, 151, 286, 397, 422). The association between overweight/obesity and mortality risk, however, varies by sex, ethnicity, and age, which may be why data are mixed (71, 188, 229, 320, 497, 519). Being overweight or obese is associated with an increased risk of coronary heart disease (52, 91, 555). T2DM is strongly associated with obesity or overweight in both men and women (191), and a BMI of >25 kg/m2 was associated with a 2.2-fold greater risk of death from diabetes, a greater association than with any other cause of death (422). However, as with other diseases, the relationship between BMI and T2DM risk also varies by ethnicity (314, 499). Other diseases associated with obesity include various types of cancer (70, 112, 201, 433), ischemic stroke (358, 501, 579), heart failure (245), dementia (202), venous thrombosis (7), gallstones (489), gastroesophageal reflux disease (386), renal disease (145), sleep apnea (570), and osteoarthritis (83). Particularly pertinent to this review, maternal obesity is associated with gestational complications and adverse fetal and neonatal health outcomes (348, 513). However, there remains a controversy as to the higher rate of mortality among the overweight and obese, particularly using self-reported BMI (244). Some report the so-called obesity paradox whereby the overall mortality was lower among those with T2DM and cardiovascular comorbidity and weight loss but not weight gain was associated with increased mortality and morbidity (124, 125).
C. Genes × Environment Interactions: Imprinting (Epigenetics) as a Concept
Although a number of common genetic susceptibility loci for obesity and T2DM have been identified over the last decade, the rapid rise in prevalence of these conditions in the last two decades, a time frame which is not compatible with a change in our genetic make-up, suggests that the environment in which we live is an important determinant of obesity risk. Environmental factors that have been attributed to this rapidly increasing prevalence of obesity include increased consumption of highly processed foods that are high in saturated fat and refined carbohydrates as well as reduced physical activity (421). However, the wide variation in BMI among individuals living in the same “obesogenic” environment has led to the opinion that obesity risk is determined by a complex interaction between our genes and the environment in which we live. How these interactions could occur at the molecular level through epigenetic mechanisms and how there may be critical time periods during development when this is more likely to occur will be discussed in more detail below.
D. Historical Background
1. Early concepts of energy homeostasis regulation
In 1940, Hetherington and Ranson (209, 210) first demonstrated that lesions of the ventromedial hypothalamus caused rats to massively overeat and become obese. As later became apparent, to produce the massive obesity associated with the “classic” VMH lesion, damage usually extended to a quite large area including both the ventromedial (VMN) and arcuate (ARC) nuclei (127, 249, 462). However, it was not until several years after this fact became evident that the importance of the ARC and its resident proopiomelanocortin (POMC) and neuropeptide Y (NPY)/agouti-related peptide (AgRP) neurons in the regulation of energy and glucose homeostasis were recognized (38, 42, 43, 189, 467). Later, it was shown that large lesions of the lateral hypothalamic area (LHA) produce profound anorexia and weight loss (15), which led Stellar (493) to put forward the dual center hypothesis whereby the VMH was the “satiety center” and the LHA was the “feeding center.” This concept held sway for many years and led to the largely hypothalmocentric view of energy homeostasis control that still dominates the thinking and research of many investigators. However, we now recognize that such control resides within a distributed network of sites within the brain (183, 184) and that lesions in one part of this network can alter the defended level of body weight and adiposity (242). The observation that the level of defended body weight can be altered by lesions of areas such as the VMH and LHA led to the idea of a set-point whose level is set depending on the neural substrates as well as internal and external environments (242).
However, it was obvious that the brain required some means of monitoring the metabolic status of the periphery to enable it to control overall energy homeostasis. Kennedy (247) was among the first to suggest that body fat storage might be the source of such feedback. He suggested that adipose tissue produces a signal, in proportion to its mass, that is sensed by the brain to regulate changes in intake or expenditure, and this keeps body fat within a predefined set-point. This negative-feedback system has been termed the “lipostatic” hypothesis (247). In fact, the lipostatic factor postulated by Kennedy was eventually shown to be leptin, a hormone produced by adipose tissue in proportion to its overall mass (577). However, the basic concept of a set-point remains highly controversial, and extensive tomes have been written in defense (243) and rebuttal of this concept (396, 488, 558). What does seem clear is that in most humans, and some rodent strains that become obese, the defended body weight can be moved upward fairly easily while long-term attempts to move them below their higher body weight by caloric restriction is met with failure in upwards of 90% of individuals (288, 292, 302). The underlying reason for this observation remains unknown, but its existence serves as the main focus for most research which attempts to find treatments for obesity.
2. The discovery of leptin and how it changed things
In 1949, investigators at the Jackson laboratory in Bar Harbor reported a colony of mice showing severe obesity (223). These mice were first distinguishable from littermates at 4 wk of age but became four times heavier than wild-type littermates as adults. Offspring of heterozygous matings demonstrated the 3:1 ratio characteristic of a recessive gene, which was subsequently designated ob (now Lep) (223). In 1966, a second mouse strain with severe obesity syndrome was identified by Coleman and colleagues (220). Mice homozygous for the mutation were designated diabetes (db) and displayed early-onset obesity, hyperphagia, and diabetes. These fortuitous observations represented a major breakthrough in the field of the genetics of obesity, although the nature of the defective gene(s) remained to be discovered. Prior to the era of sophisticated transgenic approaches, Coleman and colleagues went on to perform heroic parabiosis experiments. They surgically connected the circulatory system of either wild-type or obese ob mice with diabetic db mice and found that it produced weight loss and hypophagia in wild-type and ob mice without affecting db mice. Based on these observations, Coleman and colleagues (220) proposed that ob mice lacked a circulating satiety factor and that db mice overproduced that circulating factor but could not respond to it. In 1994, Friedman and collaborators (577) cloned the defective gene of the ob mouse. Using positional cloning, they found that the ob gene encode a 4.5-kb RNA secreted by adipose tissue in proportion to its mass (577). As predicted, administration of the recombinant OB peptide reduced body weight and food intake of obese mice (73, 197, 399). Based on these physiological effects, Friedman named the peptide “leptin” from the Greek root leptos for “thin.” However, db mice were insensitive to the weight loss-inducing effect of leptin, suggesting that the db locus encodes the leptin receptor, which was subsequently cloned in 1996 (82, 283). Leptin appears to act primarily on the brain to mediate its effects on feeding and metabolism because central administration of leptin has a marked effect on feeding (73), and the strongest expression of leptin receptor occurs in the hypothalamus (283, 527). In fact, leptin fulfills all of the predicted “lipostatic” properties proposed by Kennedy in 1953 (247). Moreover, the observation that leptin is one of the first major metabolic hormones to appear during embryogenesis (215) suggests a role for leptin in perinatal development.
3. Early studies implicating the perinatal environment in the pathogenesis of obesity and diabetes
Some of the earliest evidence in support of the importance of the early life environment in determining long-term health came from studies in the United Kingdom and Sweden in the 1930s demonstrating that, within any one age group, death rates were most affected by the date of birth and not the year of death (248). Further support for the importance of the neonatal environment on long-term health emerged almost 50 years later in studies in Norway by Forsdahl (155) demonstrating that geographical variations in atherosclerotic disease were not associated with current mortality rates but correlated strongly with past infant mortality rates. The earliest evidence that nutrition during neonatal life could influence long-term metabolic health came from the study of individuals who were born during the Dutch Hunger Winter that occurred in the western part of the Netherlands at the end of World War II. These data suggested that low nutrient intake during early postnatal life actually reduced the risk of obesity at age 19 (428). These observations were supported by pioneering studies in rats by Kennedy (246) where he altered the plane of nutrition during the suckling period through manipulation of litter size. Rats reared in small litters where there is little competition for the mother's milk gain more weight during lactation and remain fatter and heavier throughout life even when fed a standard laboratory chow diet. In contrast, rats reared in large litters receive less milk and consequently gain less weight during suckling. These animals remain smaller and leaner throughout life. Importantly, it was demonstrated that if nutrient restriction was initiated for the same length of time post-weaning, rats rapidly caught up in weight (552). On the basis of these findings it was suggested that appetite was determined during the suckling period and that the hypothalamus played an important part in mediating these effects (553). These findings were supported in studies by others in subsequent decades (252, 377, 392, 413). More recent findings from animal models demonstrating the importance of the early postnatal period are discussed below.
Focus on the potential importance of the fetal environment arose from studies by Barker and colleagues (198) demonstrating a strong association between birth weight and subsequent risk of development of T2DM and other features of the metabolic syndrome. These studies demonstrated that individuals with the lowest birth weight were around six times more likely to have T2DM or impaired glucose tolerance at age 64 compared with those individuals with the highest birth weight. These findings have now been reproduced in over 50 studies worldwide. The relationship between birth weight and T2DM holds true in monozygotic (identical) twins (51, 417), suggesting that the fetal environment plays a critical role in mediating the relationship between birth weight and long-term metabolic health. While nutrient supply is one important determinant of fetal growth, assessing the importance of fetal nutrition in mediating these relationships is difficult in humans. However, evidence from studies of individuals who were in utero during periods of famine have provided direct evidence that alterations in maternal nutrition during pregnancy can influence long-term risk of T2DM. Prior to the “Dutch Hunger Winter,” the western part of the Netherlands was a well-nourished population. The abrupt onset of the famine and its short duration (5 mo) provided a unique opportunity to retrospectively study the effects of maternal nutrient restriction on offspring glucose tolerance. At age 50, those individuals who were in utero during the famine had worse glucose tolerance compared with those individuals born either the year before or the year after the famine (427). Those exposed during late gestation were most affected, suggesting that the third trimester represents a particularly vulnerable developmental period in terms of long-term regulation of glucose homeostasis. In contrast, risk of cardiovascular disease and obesity was more pronounced in those individuals exposed to famine during early gestation (428). This highlights the different critical periods of development for different organ systems. A subsequent, larger, study of a population exposed to the Chinese Famine (1959–1961) showed a similar association between exposure to suboptimal nutrition in utero and increased risk of T2DM in later life (309). In both studies, it was demonstrated that exposure to a nutritionally rich environment in later life exacerbated the detrimental effects of undernutrition in utero. The causative relationship between poor nutrition in utero and long-term health has been further substantiated by studies in animal models (see below).
II. CENTRAL REGULATION OF ENERGY AND GLUCOSE HOMEOSTASIS
A. The Central-Peripheral Conversation in the Control of Energy and Glucose Homeostasis
Energy homeostasis is defined as the balance between energy intake on the one hand and output as thermogenesis (heat production) on the other. When intake exceeds output, energy is stored primarily as fat in adipose depots. When food supplies are limited and intake is restricted, those adipose stores are called upon as the major energy source over long periods of time. While it is generally agreed that the brain is the controller of energy and glucose homeostasis, it is able to carry out this function only because it receives vital information about the metabolic and physiological status of the body from enteroceptive inputs from the various organs via metabolic signals and neural afferents. Afferents from the majority of viscera are carried primarily within the vagus (Xth) cranial nerve that has its cell bodies in the nodose ganglion. Their central axons terminate within the caudal part of the nucleus of the solitary tract (NTS) in the medulla (96, 442, 443, 466). Other small unmyelinated nerves from the viscera, which travel with somatic efferents, have their cell bodies in the dorsal root ganglia of the spinal cord. Their central processes also terminate in the caudal NTS. Thus the NTS represents the first important neural link between the viscera and the brain. These neural inputs carry sensations of stretch, pain in the viscera, as well as from chemical sensors within the portal vein, carotid body, and small intestines (96, 442, 443, 466). Importantly, the brain also monitors the metabolic status of the body by the transport of hormones such as leptin, insulin, and ghrelin and substrates such as glucose, free fatty acids, lactate, ketone bodies, and cytokines across the blood-brain barrier (BBB) (28, 29, 31, 362). The BBB excludes many toxins and molecules that do not have dedicated transporters from entering the brain by virtue of tight junctions between the vascular endothelial cells and apposition of astrocyte foot processes on cerebral microvessels. However, tight junctions in some vessels in areas such as the ARC may vary in permeability depending on the nutritional state of the individual (273). Finally, these neural, hormonal, and substrate signals from the body are integrated within a distributed network of brain sites that contain specialized metabolic sensing neurons (see below) which gather these signals from the body, together with indirect neural inputs from the primary senses of taste, smell, sight, hearing, and sensation, to alter their membrane potential, neural activity, neuro-transmitter and -peptide release, as well as gene transcription (303).
B. Metabolic Sensing Neurons: the Basic Integrators and Regulators of Glucose and Energy Homeostasis
In the 1950s Jean Mayer (322) first postulated that there were neurons in the hypothalamus that sensed changes in glucose oxidation as a means of regulating feeding. It was not until 1964 that Oomura et al. (372) and Anand et al. (16) identified such glucosensing neurons. The majority of neurons utilize glucose as their primary fuel to produce ATP when their activity increases. When neuronal activity increases, neuronal glucose transporters 3 (Glut3) increase the uptake of glucose proportionally (530). Most neurons can also utilize lactate, long-chain fatty acids, and ketone bodies as alternate fuels in some instances (47, 131, 312, 445). However, whereas metabolic sensing neurons also utilize glucose as a primary fuel, ambient extracellular levels of glucose and other metabolic substrates are “sensed” by these neurons using a variety of signaling and metabolic pathways as a means of regulating their activity. Thus, while most neurons utilize such substrates to fuel their ongoing activity, metabolic sensing neurons do as well, but also use these same substrates to regulate their activity (50, 280, 301, 303, 338).
These neurons either increase (glucose excited) or decrease (glucose inhibited) their activity as ambient glucose levels rise and are conversely inhibited and excited as glucose levels fall (16, 20, 304, 373). Thus, after a meal, glucose-excited neurons are generally activated, while glucose inhibited neurons are inactivated. During fasting or insulin-induced hypoglycemia, glucose inhibited neurons are powerfully activated (450, 452, 484). Within the ventromedial portion of the hypothalamus (VMH), which is composed of the ARC and VMN, ∼10–15% of neurons are either glucose excited or inhibited (305). Of those, 40–65% utilize the pancreatic form of glucokinase as a gatekeeper for the regulation of glucose-induced changes in their activity (236). Formation of ATP within glucose-excited neurons leads to inactivation of an ATP-sensitive K+ (KATP) channel leading to membrane depolarization, entry of calcium via a voltage-dependent calcium channel, increases in activity, propagation of an action potential, and release of neuro-transmitters and -peptides from their axon terminals (20, 305). Glucose-inhibited neurons form nitric oxide and, via activation of AMP-activated kinase and soluble guanylyl cyclase, increase neuronal firing when glucose levels fall by an action on the cystic fibrosis transmembrane receptor (148). Catabolic ARC POMC neurons are predominantly glucose excited (221), while anabolic ARC NPY/AgRP (351) and LHA orexin/hypocretin neurons (350) are mostly glucose inhibited in type. However, other glucosensing neurons have been identified which utilize several other ion channels and transporter mechanisms to regulate their activity (239, 365, 375, 390).
There remains a controversy as to whether physiological changes in blood and/or brain glucose are actually involved in the regulation of feeding as Mayer originally proposed (129, 172, 305). To summarize this controversy, studies using very high or low levels of glucose or glucose availability, especially in the brain, can inhibit or stimulate feeding, respectively (186, 474, 479, 529). Some investigators have shown a relationship between spontaneous, small dips in blood glucose preceding meals in rats and humans (72, 74, 313). However, others have failed to confirm such a relationship between blood or VMH glucose levels and meal onset (129). Also, manipulation of VMH neuronal glucosensing by altering glucokinase activity fails to affect either short- or long-term feeding (129), while it does markedly alter the counterregulatory responses to insulin-induced hypoglycemia (290). Such results suggest that hypothalamic glucosensing neurons are not critical regulators of normal feeding but are important for the defense against hypoglycemia.
Many of these same VMH glucosensing neurons are also fatty acid sensors which respond to long-chain fatty acids by altering their activity (230, 278, 280, 281, 337, 374). While early work suggested that this fatty acid sensing was mediated by intracellular metabolism of long-chain fatty acids (230), it now appears that much of this sensing is mediated by fatty acid translocator/CD36 (which appears to act as a receptor and may also be a transporter of fatty acids) in many VMH neurons and that this regulatory step is independent of neuronal fatty acid oxidation (278, 280, 281). Furthermore, although impairment of VMH glucosensing has no effect on energy homeostasis, altering fatty acid sensing by depletion of VMH neuronal CD36 inhibits linear growth as well as causes redistribution of fat stores from visceral to subcutaneous adipose depots and marked insulin resistance (278). Thus, while the glucosensing properties of VMH metabolic sensing neurons do not appear to be critical for the regulation of energy homeostasis, their ability to sense and respond to long-chain fatty acids is critical for some aspects of both energy and glucose homeostasis. Importantly for this review, the interaction among an obesity-prone genotype, diet, and the presence of maternal obesity has a major effect on both the glucose- and fatty acid-sensing properties of these VMH metabolic sensing neurons (281).
In addition to their responses to glucose and long-chain fatty acids, the activity of many of these same neurons is also altered by ambient levels of lactate (485) and ketone bodies (279, 510), both of which are produced locally by astrocytes (48, 49, 131). They also respond to hormones produced in the periphery such as leptin (225, 486), insulin (487, 541), and ghrelin (99) which are transported across the BBB. Thus the term metabolic (or nutrient) sensor is an apt term for these neurons. Importantly, while a great deal of the research on such neurons has focused on ARC and VMN neurons, glucosensing neurons have been identified in the lateral hypothalamus (16, 350), hypothalamic paraventricular nucleus (PVN) (128), amygdala (578), basal ganglia (285), NTS (343), and several other brain areas known to be involved in the regulation of both energy and glucose homeostasis (289, 305). Most of these neurons make critical connections with brain areas that provide efferent output to a variety of neuroendocrine, autonomic, and behavioral centers required for such homeostatic processes. The network of brain areas containing these metabolic sensors forms a distributed network that functions as an integrated system. Thus the early observations that destruction of the VMH or LHA leads to marked disturbances in energy and glucose homeostasis (209, 210, 240, 241, 341, 534) do not mean that these are satiety and feeding centers; it simply means that destroying one node of this distributed network can lead to dysfunction of its integrated function. While there is a great deal of redundancy in this distributed network, many of its component parts can undergo plasticity, particularly during early pre- and postnatal development through alterations in neural connections and expression of neuro-transmitters and -peptides (58, 59, 62, 98, 391–393, 490).
C. Homeostatic and Reward-Based Systems
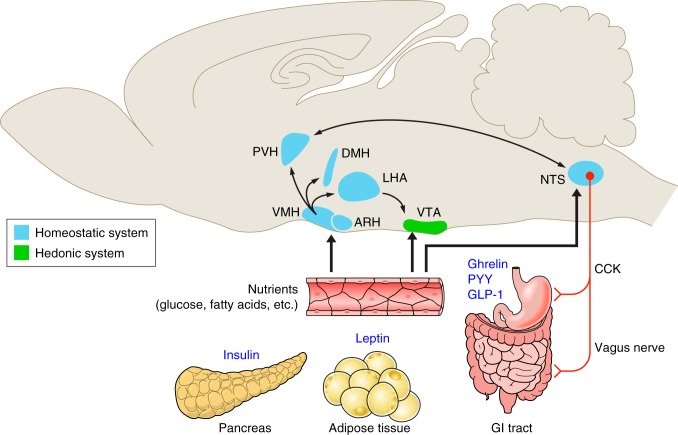
To ensure adequate nutrition, it is necessary for the brain to have intrinsic neural circuits that sense and regulate the levels of various nutrients in the blood and body stores. As mentioned above, a primary importance has been given to the hypothalamus, in part because this brain region can integrate hormonal, autonomic, and somatomotor control mechanisms and, in turn, induce a variety of neuroendocrine homeostatic responses (Figure 1). However, we now know that the central systems regulating energy homeostasis involve a distributed and interconnected neural network (181, 182, 301). For example, the ARC, that was originally thought to be exclusively “anorexigenic,” contains two chemically identified neuronal types that play opposite roles in energy balance regulation: the POMC neurons that are anorexigenic but also the NPY/AgRP neurons that are orexigenic (94, 483). Moreover, POMC neuronal activity can be modulated indirectly via transsynaptic GABAergic inputs arising from NPY neurons, showing the anatomical intricacy of these neural networks (17, 100, 516). Arcuate POMC and NPY neurons project to multiple hypothalamic and extrahypothalamic sites to regulate feeding (65, 94). Of particular importance are projections to the PVN because it is the most thoroughly characterized pathway involved in feeding and energy balance regulation, and the PVN is anatomically connected to endocrine, autonomic, and somatomotor systems (461, 506, 544). For example, the parvocellular part of the PVN contains corticotropin-releasing hormone and vasopressin neurons that regulate adrenocorticotropic hormone secretion and thyroid-stimulating hormone neurons that influence thyroid-stimulating hormone production in the pituitary. In addition to neuroendocrine neurons, the PVN also contains neurons that send direct projections to preautonomic sites, such as the brain stem and spinal cord (458, 506). In addition to forebrain structures, the caudal brain stem, and particularly the dorsal vagal complex, plays an essential role in the regulation of energy homeostasis. The dorsal vagal complex comprises the dorsal motor nucleus of the vagus nerve, NTS, and area postrema. Although the hypothalamus predominantly integrates long-term adiposity signals, dorsal vagal complex neurons appear to be more involved in the short-term control of feeding control in response to satiety signals (see Refs. 46, 182 for reviews).
FIGURE 1.
Major routes for regulation of feeding and energy balance. These simplified schematics illustrate the possible neural networks relaying metabolic signals from the periphery to the brain. The regulatory effects of peripheral hormones and nutrients involve a complex, distributed, and interconnected neuronal network involving neurons in the forebrain, midbrain, and hindbrain. The arcuate (ARC) and ventromedial (VMN) nuclei in the hypothalamus have high densities of neurons that directly respond to peripheral signals such as leptin, insulin, ghrelin, and glucose. In turn, neurons in the ARC and VMN send projections to other parts of the hypothalamus inculding the dorsomedial (DMN) and paraventicular (PVN) nuclei of the hypothalamus and the lateral hypothalamic area (LHA). Of particular importance are projections to PVN because it contains neurons that mediate autonomic and neuroendocrine responses associated with energy homeostasis. In addition to a direct action in the hypothalamus, peripheral metabolic signals also act on neurons located in the hindbrain, specifically on neurons located in the nucleus of the tractus solitarius (NTS). The NTS appears primarily involved in the short-term control of feeding control in response to satiety, such as peptide YY (PYY), cholecystokinin (CCK), and glucagon-like peptide-1 (GLP-1), and mechanical signals originating from the gastrointestinal (GI) tract. The central regulation of feeding also involves the neurons in the ventral tegmental area (VTA), which modulates behaviors and both our need to and desire to eat. This figure was created in part using illustrations from “Servier Medical Art” with permission.
If feeding were controlled solely by homeostatic systems, most individuals would likely maintain a stable, relatively lean body weight. However, virtually any mammal will eat beyond its homeostatic needs when exposed to highly palatable foods such as a high-fat/high-sucrose diet. Such observations support the contention that the hedonic (“reward”) system plays an important role in regulating feeding behavior (Figure 1). The hedonic system deals with the rewarding value of stimuli (e.g., food) and has neural circuits which encode wanting (incentive motivation) and liking (experienced pleasure) of those stimuli. A key neurobiological substrate involved in incentive motivation to eat is the mesolimbic dopaminergic pathway. This pathway is composed of dopamine neurons in the ventral tegmental area (VTA) of the midbrain that connects to limbic centers such as the nucleus accumbens, the amygdala, hippocampus, and medial prefrontal cortex (45). The observation that rodents with defective dopamine signaling in this mesolimbic system become aphagic and adipsic and can even die of starvation supports the idea that the mesolimbic dopaminergic system plays an incentive role in feeding regulation (507, 526). In addition to being activated by a variety of addictive substances, including cocaine and alcohol, VTA dopamine neurons are also directly modulated by metabolic hormones such as leptin and ghrelin. Leptin exerts a direct inhibitory influence on VTA dopamine neurons, and hyperphagia of leptin-deficient mice is blunted in the absence of dopamine (146, 163, 217, 507). In contrast, ghrelin increases the activity of VTA dopaminergic neurons and direct injection of ghrelin into the VTA promotes feeding (3, 354). These studies show that metabolic hormones are not only involved in the short- and long-term control of energy homeostasis, but also modulate motivated behaviors and both our need and desire to eat.
D. Central Roles for Leptin, Insulin, and Ghrelin
1. Leptin
The discovery of leptin reinforced the concepts originally proposed by Woods and Porte for insulin (561) that our subconscious motivation to eat can be powerfully and dynamically regulated by hormonal signals from the periphery. Although this 16-kDa protein is primarily produced by white adipose tissue, it rapidly became clear that leptin acts primarily in the brain to mediate its effects on feeding and metabolism. Leptin injection blunts food intake and decreases body weight, and this effect is more robust when the hormone is injected intracerebroventricularly compared with peripherally (73, 196, 197, 399). The long (signaling) form of the leptin receptor (LepRb) is expressed at high levels in the brain (76, 135, 470), and neuron-specific deletion of LepRb results in a phenotype that is a virtual carbon copy of whole-body leptin receptor-deficient db/db mouse. Reactivation of leptin receptors in the brain of db/db mice rescues their obese and diabetic phenotype, further demonstrating the importance of the brain in mediating leptin's effects (90, 117). Soon after the cloning of leptin and its receptors, considerable research focused on neurons located in the ARC, in part because of the proximity of this nucleus to the median eminence, a region outside of the BBB (87). Also the ARC contains the highest density of leptin receptors of any brain region (76, 135, 283, 334, 470) and lesions of VMH (that includes the ARC) prevent leptin's actions in the brain (263, 460). Within the ARC, leptin stimulates the activity of neurons that contain POMC-derived peptides and inhibits the activity of neurons that coproduce NPY and AgRP (333). Gain- and loss-of-function mutations of leptin receptors selectively in POMC neurons produce phenotypes that suggest a prominent role for POMC neurons in mediating leptin's effects on energy expenditure and glucose homeostasis with a more moderate effect on body weight regulation (27, 44).
Prior to 2005, a widely held view was that most, if not all, of leptin's effects are mediated by neurons located in the ARC. However, peripheral leptin administration also acts on neurons in other brains regions such as the VMN, LHA, VTA, and NTS (76, 134, 194, 195, 468). Such observations slowly moved the attention of the field away from the arcuate-centric notion of leptin action. Thus mice lacking LepRb in SF1-expressing neurons of the VMN develop mild obesity when fed a chow diet and are markedly sensitive to high-fat diet-induced obesity, supporting a role for VMN neurons in leptin's regulatory actions (121). In addition, targeted deletion of LepRb in LHA neurotensin neurons causes early-onset obesity due to hyperphagia and locomotor inactivity (284). Notably, neurotensin neurons appear anatomically well-poised to relay leptin's actions on the mesolimbic dopaminergic system, suggesting that neurotensin neurons may be a crucial point of convergence for homeostatic and hedonic interactions that regulate ingestive behavior. Supporting a role for leptin on brain reward circuits, leptin receptors are expressed and functional on dopaminergic neurons in the midbrain and direct manipulation of LepRb in VTA dopamine neurons influences feeding behavior (146, 163, 217). Another site of particular interest outside the hypothalamus is the NTS, a hindbrain nucleus involved in the processing of meal-related satiety signals where LepRb mRNA was shown to be expressed (335). But it was another 12 yr before the functional relevance of these NTS LepRbs was demonstrated. Downregulation of LepRb in the medial NTS led to increased body weight and adiposity and caused chronic hyperphagia, likely due to a reduction in leptin's potentiation of gastrointestinal satiation signaling such as cholecystokinin (CCK) (204). The NTS also receives neural inputs from the hypothalamus, and recent studies have demonstrated that leptin's modulation of energy expenditure and brown adipose thermogenesis is via a GABAergic ARC-PVN-hindbrain pathway (258). In summary, the effects of leptin on the central control of energy homeostasis are anatomically distributed and appear to involve a complex, distributed, and interconnected neuronal network involving neurons located in throughout the brain.
2. Insulin
Despite its sole production by the β-cells in the pancreas, plasma insulin, like leptin levels, generally parallel overall levels of carcass adiposity (23, 416). In addition, plasma insulin levels also vary over a wide range during ingestion and absorption of nutrients. While peripheral insulin's main actions are on glucose homeostasis, several lines of evidence suggest that insulin can act centrally to affect many brain functions. First, there are abundant levels of insulin receptors in several brain areas including the olfactory bulb, hippocampus, and hypothalamus (147, 226, 238, 573). There is still a debate about whether insulin is actually produced within the brain (376, 463), but it does appear that, despite its large size, it is transported across the BBB (30). During brain development, insulin acts on its brain receptors (sometimes in association with insulin-like growth factor I) as a trophic factor for facets of neural development (206, 423, 432) including neurite outgrowth (206, 464) and neuronal differentiation (355) and survival (359). However, when injected into the hypothalamus of rat neonates, insulin alters neuronal density in the VMN in association with increased body weight gain as adults (410). While controversial (159), some studies suggest that insulin might cross the placenta to enter the fetal circulation in humans (332). For example, in rats, insulin injections in third trimester dams predispose to adult obesity in offspring (232). However, maternal hyperinsulinemia might increase transplacental glucose transport to the fetus (378). Maternal hyperinsulinemia and hyperglycemia could thus cause fetal hyperglycemia with attendant hyperinsulinemia (235) and later increases in fetal weight in offspring of mothers with gestational diabetes (511). On the other hand, insulin clearly does cross the gut wall in the early postnatal development in rodents (213, 349) such that elevations in maternal milk insulin levels can be absorbed by the offspring as potential mediators of obesity development in later life (176).
In addition to these developmental effects, insulin has important glucose-dependent actions on the activity of hypothalamic metabolic sensing neurons (451, 487) as one way in which a signal relating to adiposity can be “sensed” by the brain. There is a large amount of literature on the effects of centrally injected insulin on food intake, energy, and glucose homeostasis. Both chronic and acute intracerebroventricular infusions of insulin reduce food intake (9, 560, 562) and reducing periventricular insulin receptors causes increased food intake, adiposity, and peripheral insulin resistance (367). However, reducing insulin receptors focally in the VMH causes glucose intolerance without altering body weight (388). In mice with selective neuronal knockout of insulin receptors, females have increased food intake, and both males and females develop diet-induced obesity, mild insulin resistance, and hypertriglyceridemia (68). However, such mice reportedly had no abnormalities of brain development or neuronal survival. Direct injections of insulin into the hypothalamus (415) or via the carotid arteries (426) alter hepatic glucose production (415), although the physiological significance of these studies has been questioned because of the large doses or nonphysiological conditions used to assess these central actions of insulin (306). Thus there is a great deal of conflicting information about the physiological role of insulin on brain development and the regulation of energy and glucose homeostasis. On balance, it seems likely that insulin is transported across the BBB and does have effects on all of these parameters.
3. Ghrelin
Ghrelin was originally discovered as an endogenous ligand for the growth hormone secretagogue receptor (GHSR) (254). In adults, ghrelin is mainly synthesized within oxyntic mucosa cells of the stomach, whereas the primary source of ghrelin production during neonatal life appears to be the pancreas (254, 454). In part because of its discovery from its linkage to GHSR, ghrelin was originally reported to stimulate growth hormone (GH) secretion (254). But it rapidly became evident that it also exerts an important role on feeding behavior. When injected peripherally or centrally, ghrelin promotes feeding, suppresses energy expenditure, and causes weight gain (276, 352, 563). Remarkably, ghrelin-induced hyperphagia occurs within 5 min and persists for 24 h after injection. The observations in both human and other animals of a preprandial rise and a postprandial decline in plasma ghrelin levels suggested that ghrelin plays a specific role in hunger and meal initiation (105, 106, 515). Based on these physiological effects, it is not surprising that GHSRs are abundantly expressed in various brain regions involved in somatic growth, food intake, and body weight regulation such as the hypothalamus, hindbrain, and midbrain (342, 580). Empirical studies employing direct intra-ARC injections of ghrelin and selective lesions of the ARC demonstrated the primary importance of ARC neurons, specifically in mediating ghrelin's action on feeding (509, 563). Within the ARC, the highest proportion of neurons activated by systemic ghrelin injection coexpress NPY and AgRP (100, 540, 554). Consistent with these findings, pharmacological blockade of NPY or its receptors blunts the effects of ghrelin on food intake (276, 352). Ghrelin can also regulate the activity of POMC neurons in the ARC, but this effect appears indirect and likely involves trans-synaptic GABAergic inputs arising from NPY neurons (17, 100, 516).
Leptin and ghrelin therefore appear as two complementary, yet antagonistic, regulators of energy balance. Notably, the distribution pattern of GHSR resembles that of LepRb (401), suggesting that leptin and ghrelin might reciprocally regulate many of the same neurons. However, whether there is a direct interaction between leptin and ghrelin signaling at the cellular level remains unclear. For example, although ARC neurons coexpress GHSR and LepRb, GHSR knockout mice display unaltered leptin sensitivity (401). Nevertheless, similar to leptin, the regulatory actions of ghrelin on feeding likely involve a complex and distributed neural network. In addition to its actions on hypothalamic neurons, ghrelin also regulates mesolimbic dopaminergic neurons in the midbrain to modulate more complex aspects of feeding such as food-reward behavior (3, 85, 354, 400, 478). More recent genetic evidence demonstrated that reactivation of GHSR signaling selectively in hindbrain neurons does not ameliorate ghrelin-induced food intake but rescues hypoglycemia of GHSR null mice, suggesting that hindbrain neurons relay ghrelin's effects on glucose homeostasis (471).
E. Neuronal Plasticity
The mammalian brain ensures adaptive behavior through its large capacity for cellular and circuit plasticity. One unique property of the hypothalamus, compared with other brain structures such as the cortex and hippocampus, is that its regulation is to a large degree activity-independent, but instead is controlled by physiological signals that reflect environmental conditions. The biological processes involved in neuronal plasticity fall into two major categories: the birth of new neurons (neurogenesis) and the reshaping of existing neural circuits (synaptic remodeling). Low rates of neurogenesis are observed in the mature hypothalamus under basal conditions (255, 256), and median eminence tanycytes appear to be a possible source of these newborn neurons (282). This constitutive hypothalamic neurogenesis can be enhanced by hormonal factors. For example, central injections of ciliary neurotrophic factor (CNTF) induced marked neurogenesis in the hypothalamus that appears to participate in the weight loss effects of CNTF in ob/ob and DIO mice (256). Moreover, microimplantation of neural progenitors that express leptin receptors into the hypothalamus of newborn db/db mice allows differentiation of the donor cells into neurons that integrate into functional neural circuits that lead to reduced hyperphagia and obesity (107). Nonneurotropic factors, such as aging and neurodegeneration, can also promote hypothalamic neurogenesis (405). Hypothalamic neurogenesis can also be downregulated. For example, high-fat feeding alters cellular remodeling as demonstrated by a reduction in the number of newly generated cells and the maintenance of old neurons in the mature hypothalamus (327). Together, these findings demonstrate that neurogenesis might represent an important adaptive cellular mechanisms in response to environmental insults.
Neuronal plasticity of hypothalamic feeding circuits also occurs through rearrangement of synapses. The excitatory and inhibitory synaptic inputs to the POMC and NPY neurons are markedly altered in adult ob/ob mice; leptin deficiency increases excitatory inputs on NPY/AgRP neurons while it decreases excitatory synaptic inputs to POMC neurons (406). Acute leptin injection in adult ob/ob mice rapidly (within hours) reverses these effects, both at the electrophysiological and ultrastructural levels. Other hormones, such as ghrelin and corticosterone, also have organizational effects on hypothalamic neural circuits by modulating the synaptic inputs of ARC POMC and NPY neurons in adult mice (193, 406). Moreover, a significant remodeling of synapses has been reported in obesity-prone (DIO) rats, with an increase in inhibitory inputs to POMC neurons in the ARC of DIO rats compared with diet-resistant (DR) rats (218). The capacity of nutritional challenges to cause structural changes also appears to differ between DIO and DR rats. High-fat feeding causes a loss of synapses onto POMC neurons in DIO rats, but a gain in synaptic coverage in obesity-resistant DR rats (218). Together, these observations indicate that remodeling of brain circuits involved in energy balance regulation occurs throughout the entire lifespan and is influenced by both metabolic and physiological cues and pathological insults. This neuronal plasticity allows the elaboration of adaptive behavioral and physiological responses that are essential for optimal regulation of energy balance.
F. Gut-Brain Interactions
1. Neurohumoral inputs
The brain receives a wide variety of signals from the gastrointestinal (GI) tract, via either sensory afferents or hormonal signals. The vagus nerve is indisputably the most important neural link between the gut and the brain. It is the longest of the cranial nerves and innervates the entire alimentary tract. It comprises fibers carrying afferent sensory information from the periphery to the brain, but also fibers carrying efferent motor information from the brain to the viscera (420). Afferent signals carried by the vagus nerve include information about gastric stretch, enteroendocrine signals from hormones released within the GI tract, and blood glucose and fatty acid levels. The caudal brain stem, and particularly the NTS via its vagal afferents and efferents, acts as a nodal point in the gut-brain axis. Vagal afferents from the GI tract synapse within subregions of the NTS, and the activation of these afferents regulates postprandial function by inhibiting food intake (465). In turn, the NTS sends reciprocal projections to other regions of the brain involved in feeding regulation such as the hypothalamus, amygdala, and nucleus accumbens. The NTS therefore represents a major portal through which visceral afferent information for homeostatic reflexes enters the brain.
Vagal afferent fibers are also sensitive to a variety of peripheral factors, including CCK, an endogenous peptide released by duodenal enteroendocrine cells (310). CCK is released after a meal and inhibits food intake [i.e., reduces meal size and induces meal termination (480)] in part by increasing the firing rate of vagal afferents projecting to the NTS (170, 347). The regulatory action of CCK on vagal-NTS projections appears to be mediated via the CCK-A receptor subtype (64, 259, 277, 395).
In addition to CCK, the gut secretes a number of other hormones that signal to the brain to regulate feeding. These hormonal effectors include ghrelin, peptide YY (PYY), and glucagon-like peptide-1 (GLP-1). Ghrelin is produced mainly by the gastric mucosa and is the only known peripheral hormone that promotes feeding. That secretion of ghrelin is increased in response to starvation, increased before a meal, and suppressed by meals, supports the hypothesis that ghrelin is primarily involved in meal initiation (105, 106, 515). The hypothalamus is a primary site of ghrelin's orexigenic effects. The highest density of ghrelin receptors and ghrelin-responsive neurons is found in the hypothalamus, particularly in the ARC, VMN, and PVN (211, 352, 342, 580). The observations that blockade of the gastric vagal afferent abolishes the feeding response to intravenous ghrelin and that GHSRs are expressed in vagal terminal suggest that ghrelin also induces some of its regulatory effects through the vagus nerve (115). For example, ghrelin does not stimulate feeding in human patients with surgical procedures involving vagotomy (115). However, data to the contrary exist regarding an essential role for the vagus in transmitting peripheral ghrelin's effects on feeding (19).
PYY is produced by L-type enteroendocrine cells, mainly in the ileum and colon, in response to the caloric content of the meal (5). The bioactive peptide, PYY3–36, is stimulated in proportion to the energy content of food and peaks 1–2 h postprandially. Peripheral administration of PYY3–36 inhibits food intake in rodents and humans (34, 35). PYY3–36 has a high affinity for the NPY Y2 receptors, which are widely distributed throughout the periphery and CNS, including in vagal endings (253). Consistent with these findings, gastric vagotomy blocks the anorectic effects of PYY3–36 (1, 253). In addition, PYY3–36 acts on hypothalamic neurons to reduce feeding and ARC injection of PYY3–36 inhibits food intake and inhibits the electrical activity of NPY nerve terminals causing a reduction of the inhibition of POMC neurons (35).
GLP-1, GLP-2, and oxyntomodulin are produced by the posttranslational processing of the preproglucagon gene in the gut and the brain stem (24). The GLPs are produced by intestinal L-cells in response to fatty acids or carbohydrates. GLP-1 is released into the circulation after a meal to inhibit gastric secretion and emptying and induce postprandial secretion of insulin (24, 268). Direct oxyntomodulin injection into the ARC causes a sustained reduction in refeeding after a fast, indicating the importance of the hypothalamus and particularly the ARC in mediating oxyntomodulin's anorectic action (113). However, intra-ARC administration of the GLP-1 receptor antagonist exendin9–39 does not block the anorectic action of GLP-1, indicating that oxyntomodulin and GLP-1 use different neural pathways to mediate their feeding effects (113). Sites of action of GLP-1 include neurons in autonomic control sites such as brain stem catecholamine neurons (565, 566).
2. Gut microbiota
Gut microflora and their interactions with obesity have become a subject of great interest in recent years. Leptin-deficient ob/ob mice have significant reductions in Bacteroides and increases in Firmicutes, two major gut bacterial phyla (307). Similarly, some obese humans demonstrate an increase in Firmicutes in their stools (308), and prolonged ingestion of a high-fat diet is associated with decreased bacterial abundance and increased Firmicutes content (520). Importantly, bacterial transplants from lean and obese mice into otherwise high-fat obesity-resistant, germ-free mice cause them to develop the weight gain phenotype of the donors, suggesting a causal role of gut microbiota in the development of obesity (521, 522). Also, increased body and fat mass in human twin pairs discordant for obesity could be transmitted to germ-free mice by transplantation of the fecal microbiota of those humans (438). The mechanism by which alterations in microbial gut flora might determine the propensity of an individual to become obese has not been established. However, one hypothesis is that these microflora might alter nutrient absorption by changing the absorptive surface of the gut in association with inflammatory changes induced by some diets (429, 520, 521). Such changes in gut permeability might become more important as the individual matures since large molecules such as antibodies, leptin, and insulin cross the neonatal intestinal barrier and enter the circulation (287, 349). Regardless of the specific mechanism, early postnatal nutrition and milk content might alter gut microbiota as an explanation for the increased obesity of diet-resistant pups cross-fostered to obese DIO dams (75, 176, 272, 315).
G. Peripheral Organs and Glucose Homeostasis
1. Pancreas
The pancreatic β-cells within the islets of Langerhans are the only cells that have the capability to secrete insulin. They are therefore central to the appropriate regulation of glucose homeostasis. The islets of Langerhans were first identified in 1869 by the German anatomist Paul Langerhans and, despite the fact that they constitute <5% of pancreatic mass, they are critical for maintenance of glucose homeostasis. They contain five major cell types: α-cells (that produce glucagon), δ-cells (that produce somatostatin), PP cells (that produce pancreatic polypeptide), ε-cells (that produce ghrelin), and β-cells (that produce insulin and amylin). Pancreatic β-cells produce insulin primarily in response to elevated levels of glucose. However, production can also be increased in response to other factors such as certain amino acids, free fatty acids, and the sulfonylurea class of antidiabetic drug. The stimulation of insulin secretion involves changes in β-cell electrical activity and ultimately exocytosis of insulin (reviewed in Rorsman and Braun, 447). T2DM is thought to arise in general when pancreatic β-cells malfunction such that they cannot further increase insulin secretion to compensate for progressive peripheral tissue insulin resistance. This may arise because of an inherent or progressive reduction in β-cells mass (reviewed in Weir and Bonner-Weir, 545), genetic defects that reduce β-cell function (reviewed in Bonnefond et al., 54), programming events that occurred in early life resulting in a permanent reduction in β-cell mass and/or function (reviewed in Reusens et al., 435), or postnatal triggers that could involve epigenetic mechanisms (171).
2. Liver
The liver is the major site of glucose production under fasting conditions, and thus resistance to the action of insulin to inhibit hepatic glucose production can contribute to hyperglycemia (66). There are a number of mechanisms by which hepatic insulin resistance can occur. Nonalcoholic fatty liver disease (NAFLD), which is thought to affect up to 30% of the population in the Western world, is thought to be a major contributing factor (571). Under physiological conditions fatty acids enter hepatocytes and are either oxidized by mitochondria or stored in the form of triglycerides. However, under conditions where there is an imbalance between influx and oxidation excessive storage occurs. This can occur, for example, when lipid storage capacity of adipose tissue becomes exceeded, leading to increased flux of fatty acids into the liver and consequently increased deposition of triglycerides and other lipid intermediates such as phosphatidic acid and diacylglycerol (21). These can result in activation of various kinases (e.g., inhibitor of kappa B kinase and Jun NH2-terminal kinase) that inhibit insulin signaling through serine phosphorylation of IRS-1 and consequently cause hepatic insulin resistance. In addition, there is evidence to suggest that under conditions of hyperinsulinemia, as a consequence of resistance to the action of insulin in relation to inhibition of hepatic glucose production, insulin's ability to promote de novo lipogenesis can remain intact. This will further promote hepatic triglyceride accumulation (66). There is good evidence to suggest that fatty liver and hepatic insulin resistance can develop as a result of both early environmental (86) and genetic factors (168).
3. Skeletal muscle
Skeletal muscle is the major site of glucose disposal postprandially and thus insulin resistance at this site is a substantial contributor to the development of T2DM. Skeletal muscle takes up glucose in an insulin-dependent manner as a result of the stimulation of translocation of the glucose transporter GLUT4 to the plasma membrane via stimulation of the phosphoinositol 3-kinase-protein kinase B (Akt) pathway. In addition to this insulin-stimulated pathway, there is an alternative pathway that potentiates glucose uptake into skeletal muscle that is activated by exercise and caloric restriction (453). This is mediated by AMP kinase, which has therefore become a focus of potential therapeutic strategies for insulin resistance and associated syndromes. As with liver, skeletal muscle is a major site of triglyceride accumulation in situations where the adipocyte lipid storage capacity has been exceeded. There is a strong positive correlation between muscle triglyceride content and insulin resistance (385). The mechanism(s) by which increased lipid accumulation induces insulin resistance in skeletal muscle remains a subject of debate (reviewed in 55). However, it has been suggested that such lipotoxicity results in increased levels of bioactive lipid metabolites such as ceramides that are known to inhibit activation of protein kinase B. Paradoxically intramyocellular triglycerides are also increased in highly insulin-sensitive trained athletes (reviewed in 89). This suggests that it is not the presence of the triglycerides per se that is causing the insulin resistance and that perhaps if their turnover is increased, for example, by regular exercise, generation of lipotoxic intermediates is reduced.
4. White adipose tissue
In recent years, the contribution of adipose tissue to whole body glucose homeostasis and regulation of energy balance has been increasingly recognized, and it is therefore no longer considered merely a site of lipid storage. It can both directly and indirectly influence glucose homeostasis. Adipose tissue takes up glucose in an insulin-dependent manner. Although it was initially considered to account for only ∼5% of postprandial glucose uptake, studies with transgenic animals have suggested that loss of insulin-dependent glucose uptake to adipose tissue leads to substantial loss of glucose tolerance (2). In addition to directly taking up glucose, adipose tissue can indirectly affect whole body glucose homeostasis through release of factors including free fatty acids, adipokines (e.g., resistin and adiponectin), and inflammatory mediators (e.g., TNF-α) that influence glucose uptake and/or insulin action in other tissues, especially skeletal muscle (reviewed in 165). It is well established that obesity-associated insulin resistance is associated with inflammation of adipose tissue and consequently increased production of inflammatory markers and cytokines (including TNF-α, IL-6, and IL-1β) that inhibit insulin signaling (reviewed in 144). Adipose tissue is also the major site of leptin production, a major regulator of energy balance across the life course (discussed in detail elsewhere in this review).
III. PERINATAL BRAIN DEVELOPMENT
The hypothalamus develops from the rostral diencephalon after induction by the underlying prechordal plate. Classical birth dating studies using [3H]thymidine or the thymidine analog BrdU revealed that the majority of neurons composing the hypothalamus are born between embryonic day (E) 11 and E14 in mice and E12 and E17 in rats (14, 101, 227, 317, 383). Hypothalamic neurons acquire their terminal peptidergic phenotype soon after they are generated. For example, melanin concentrating hormone neurons in the LHA are born between E12 and E13 in rats, and its mRNA is detected in the LHA as early as E13 (63). More recent genetic cell lineage experiments also indicated that hypothalamic progenitor cells can give rise to neurons that express antagonistic neuropeptides in adult life. For example, embryonic Pomc-expressing precursors can subsequently adopt either a POMC or an NPY phenotype (383).
Although hypothalamic neuronal proliferation and differentiation occurs primarily during the second half of gestation in rodents, the rodent hypothalamus remains relatively immature at birth and continues to grow during the first 2–3 wk of postnatal life. Axonal tract tracing experiments in mice showed that hypothalamic axonal connections are not formed at birth. For example, ARC axons reach their target nuclei between postnatal day (P) 6 and P16 (60). Axon terminals containing NPY/AgRP are found in a pattern that coincides with the innervation of axons from the ARC (25, 187, 361). Efferent projections from the VMN and dorsomedial nucleus (DMN) appear to develop prior to those from the ARC and are fully established by P6 and P10, respectively (60). Synapses are another key component of neuronal connectivity. We still know relatively little about the exact time point (if any) at which synapse assembly is fully established in the hypothalamus, but a few reports indicate that synapses mature gradually in the hypothalamus from birth to adulthood (319, 328).
Brain stem projections develop relatively early in rodents. Brain stem catecholaminergic inputs to the PVN are present as early as P1 in rats (440). However, different neurotransmitter systems show different developmental patterns. For example, the density of noradrenergic projections to the PVN is relatively low at birth and gradually increases to reach adult levels at weaning. In contrast, adrenergic projections are relatively high in the PVN of newborn rats but gradually decrease until weaning (440). Reciprocal descending projections from the hypothalamus to the caudal brain stem also develop early in life. Retrograde tracing experiments showed that hypothalamic neurons, such as those in the DMN, PVN, and LHA, send axonal projection to dorsal vagal complex neurons at birth and continue to develop to achieve adult-like patterns at weaning (439, 441). In summary, projections to and from the hypothalamus and brain stem develop primarily after birth and appear chemically and structurally immature until weaning.
The considerable importance of postnatal hypothalamic development in rodents differs from that in humans and non-human primates where the hypothalamus develops almost entirely during fetal life. For example, in Japanese macaques NPY/AgRP fibers innervate the PVN as early as gestational day 100 (i.e., late second trimester) and a mature pattern of NPY/AgRP projections is not apparent until gestational day 170 (180). These findings emphasize the importance of recognizing species differences in terms of timeline of developmental events. Although the regional development of the rodent hypothalamus proceeds on a timeline of days, the same developmental process takes weeks to months in human and non-human primates. Similar to non-human primates, the human hypothalamus also develops primarily prenatally with NPY-containing axons detected in the ARC and PVN as early as at 21 weeks of gestation (262).
IV. GENETIC BASIS OF OBESITY
A. Single Gene Mutations
Although single gene mutations that cause obesity are rare, their identification has helped greatly in our understanding of energy homeostasis regulation. One very successful approach to identify monogenic forms of obesity has been to focus on children who were extremely obese from an early age and to use a combination of biochemical and genetic approaches to identify the affected locus (reviewed in 366). O'Rahilly and colleagues (345) used this approach to identify a pair of cousins who were severely obese as a result of having undetectable levels of leptin. They were established as having a homozygous frame shift mutation in the leptin gene (345). Treatment of these and other leptin-deficient individuals with daily injections of recombinant leptin normalized their body weight, thus proving causality between the single gene mutation and the obese phenotype (143). To date, there are still only 24 confirmed instances of individuals with this mutation (S. Farooqi, personal communication). Furthermore, these studies demonstrated that human food intake regulation, as in the leptin-deficient ob/ob mouse, was dependent on a functional leptin-signaling pathway. Since these initial studies, it has been demonstrated that human obesity can result from defects in various components of the leptin signaling pathway including the leptin receptor (88), POMC (270), and the melanocortin-4 receptor (MC4R) (569). The latter is now thought to be the most common monogenic form of obesity, with some studies demonstrating that ∼1 in 200 obese people have disease-causing mutations in the MC4R (12, 274). There are now over 20 single gene disorders that have been shown to cause severe obesity. In addition to direct components of the leptin signaling pathway, they include genes such as prohormone convertase 1 (which is required for the processing of pro-peptides into active peptides such as POMC) (228), SIM 1 (a transcription factor required for hypothalamic development) (425) and SH2B1 (an adaptor protein that modulates signaling through tyrosine kinase and JAK-associated cytokine receptors) (123). It is notable that these single gene mutations generally influence central sensing and control of energy homeostasis rather than through peripheral systems. Further analyses of these individuals demonstrate that the defects influence appetite and satiety resulting in increased food intake. In contrast, little or no effect is observed on energy expenditure, with MC4R mutation patients being the exception and showing a small but significant reduction in metabolic rate (264).
B. Obesity as a Polygenic Disorder
As above, although there are several single gene mutations that have been identified which cause obesity and diabetes in humans (142), approximately two-thirds of obesity is inherited in what is probably a polygenic fashion (57, 502). Genome-wide association studies (GWAS) were greatly facilitated by the International HapMap (www.hapmap.org) defining common single-nucleotide polymorphisms (SNPs) and existing linkage disequilibrium that provided near-genomic coverage of common genetic variations. We are now in the fourth wave of GWAS studies of obesity that has used a variety of variables such as BMI as a continuous trait or extremes of obesity in large populations of children or adults. FTO was one of the first genes identified, originally as having a high association with T2DM but later showing that this was through its association with obesity (158). Similarly, although homozygous inheritance of mutations of the MC4R leads to severe obesity (142), variants near the MC4R gene have a relatively strong association with obesity (269, 581). Other variants with obesity associations are BDNF, TMEM18, SH2B1, NEGR1, MTCH2, FAIM2, and GNPDA2 (36, 203, 216, 219, 434). It is important to point out that, as opposed to being causal for obesity, the way that direct mutations of the MC4R gene are (142), these GWAS genes are merely associations. Many are in noncoding areas of the genome and might be markers rather than playing any contributory role in obesity causation (456). However, several of the genes such as BDNF, MC4R, SH2B1, NRXN3, TMEM18, and NEGR1 are known to be involved in the regulation of energy homeostasis, reward, and/or neural development (142, 158, 179, 205, 321). Importantly, FTO has been shown to play a critical role in leptin receptor trafficking (500). There are also likely to be many other genes that singly or in combination contribute to the genetic propensity to become obese which have yet to be identified by such studies. In addition, epigenetic modifications of some of these known or as yet to be identified genes are likely to play a critical role in determining their expression under conditions of varying environmental conditions.
V. PERINATAL ENVIRONMENT AND THE DEVELOPMENT OF OBESITY AND T2DM
A. Prenatal Influences
1. Parental undernutrition
Addressing the consequences of parental undernutrition is technically challenging in a human context. The best evidence for a direct effect of undernutrition during pregnancy on long-term metabolic health of the offspring has come from the study of individuals who were in utero during periods of famine such as the Dutch Hunger Winter (427) and the Chinese Famine (309) (see above). These have demonstrated effects of severe under nutrition during pregnancy and risk of T2DM in the offspring. Few human studies have established an effect of more physiological differences in diet during pregnancy on long-term health of the offspring. Studies of a large Danish cohort revealed that dairy protein consumption during pregnancy was positively associated with birth weight (370). However, data are not yet available regarding the possibility that increased dairy protein consumption is also associated with reduced risk of T2DM. The importance of appropriate micronutrient intake during pregnancy for the health of the offspring at age six has been suggested by a study of an Indian cohort (564). This revealed that low maternal vitamin B12 at 18 wk of pregnancy and high maternal erythrocyte folate concentrations at 28 wk of pregnancy were associated with increased insulin resistance in the offspring. Similar observations were made in a Nepalese cohort with maternal vitamin B12 deficiency being associated with insulin resistance in the offspring at age 6–8 yr (498).
In contrast to the paucity of evidence from humans, there is extensive evidence from animal models to suggest that maternal undernutrition during pregnancy is associated with increased risk of glucose intolerance, insulin resistance, and obesity in the offspring (Figure 2). This includes detrimental consequences of total caloric restriction, macronutrient, as well as micronutrient deficiency. Varying degrees of total caloric restriction have been demonstrated to result in metabolic dysfunction in the offspring. Reducing caloric intake by 50% during the last week of pregnancy and throughout lactation in rats led to loss of glucose tolerance in the offspring as they aged (169). Similar effects were observed in a sheep model where nutrient restriction was initiated in late pregnancy (167), perhaps suggesting that the third trimester is a critical time period of exposure for risk of T2DM (as also indicated from studies of the Dutch Hunger Winter Cohort). More severe caloric restriction (to 30% ad libitum) in the rat has also been shown to be associated with insulin resistance and obesity in the offspring (532).
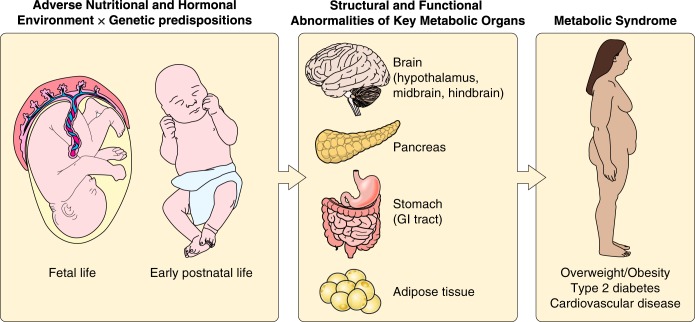
FIGURE 2.
Developmental origins of metabolic disease. The developmental programming of key regulatory systems by the perinatal environment and/or genetic background represents a possible mechanism by which alterations in maternal and/or early postnatal nutrition predispose the offspring to obesity and type 2 diabetes. This figure was created in part using illustrations from “Servier Medical Art” with permission.
The most extensively studied rodent model of macronutrient deficiency is that of isocaloric protein restriction. Offspring of dams fed diets containing 5–8% protein demonstrate impaired insulin secretion in adulthood (336) (that is exaggerated if the offspring are fed a high fat diet, 556) and insulin resistance (382). These defects in insulin action and secretion are associated with an age-dependent loss of glucose tolerance and development of a T2DM phenotype in later life (402). If followed by rapid postnatal catch-up growth, maternal protein restriction during pregnancy is associated with increased adiposity in the offspring (381). The propensity to the development of obesity is further exaggerated if the offspring are weaned onto a highly palatable diet (381). Although most studies of parental macronutrient deficiency have focused on nutrient restriction in the mother, there are now emerging studies to suggest that there can be detrimental consequences of paternal nutrient deficiency; offspring of males fed a low-protein diet displayed increased expression of genes involved in lipid and cholesterol biosynthesis in the liver (77).
A number of studies in animal models have also investigated the effects of micronutrient deficiency in the offspring. Maternal zinc deficiency in rats has been associated with increased leptin levels, insulin resistance, and impaired glucose tolerance in the offspring (233). Similar observations were observed in a rat model of maternal dietary chromium restriction (384). Maternal anemia in the rat leads to increased blood pressure in the offspring but as yet has not been associated with changes in offspring adiposity or glucose intolerance (166).
2. Parental obesity
Initial programming studies focused primarily on the detrimental consequences of parental undernutrition on the long-term metabolic health of the offspring (see above). However, following the growing epidemic of obesity during the last decade, a rapidly increasing number of studies have focused on the consequences of parental overnutrition and/or obesity during the periconceptual period on the risk of obesity and T2DM in the offspring. The potential detrimental consequences of maternal obesity during pregnancy on offspring risk of obesity in humans was first inferred from a number of observational studies which demonstrated that children born to obese mothers were more at risk of obesity/increased adiposity than those born to obese fathers (79, 214, 275, 369, 424, 550). This was further supported by evidence that maternal BMI is independently associated with offspring BMI (261, 551), adiposity (164), and insulin resistance (53, 339) and that there are strong associations between maternal weight gain during pregnancy and offspring adiposity (103, 436). Some evidence for these associations being causal has come from studies of siblings born before and after the mother had bariatric surgery to reduce her weight. These studies demonstrated that offspring born after surgery had a reduced risk of obesity and insulin resistance compared with those born prior to surgery (265, 481). It has recently been shown that these differences in risk were also associated with changes in DNA methylation (i.e., an epigenetic change) of a large number of loci, including genes involved in glucose homeostasis (190). These studies show a disproportionate risk of disease in offspring born to the same mother under different in utero conditions thereby providing evidence that an obesogenic environment experienced during this critical period of development directly influences long-term risk of obesity.
Studies in animal models support the findings from the human studies described above, supporting a causal relationship between maternal overnutrition/obesity during pregnancy and offspring adiposity and insulin resistance (reviewed in Alfaradhi and Ozanne, 10) (Figure 2). The earliest studies addressed the detrimental consequences of maternal high-fat feeding where rat dams were fed a saturated-fat-rich diet during pregnancy and lactation leading to obesity, insulin resistance, and dysregulated glucose homeostasis in the offspring (192, 512). These effects were not only a consequence of maternal diets rich in saturated fats. Maternal diets rich in n-6 polyunsaturated fatty acids have also been demonstrated to have detrimental effects on insulin sensitivity and adiposity in rat offspring (69). Similar detrimental effects of maternal high-fat feeding have been observed in non-human primate models. These include increased adiposity, as well as increased hepatic triglyceride deposition in the offspring at 6 mo of age (325). Such abnormalities in mouse models have been shown to persist into adulthood (67). Encouragingly, it has been demonstrated that dietary intervention prior to pregnancy in rats that had been fed a high-fat diet for 90 days, at least in part ameliorated the detrimental effects of the high fat diet on offspring adiposity and insulin resistance (574).
Although the studies above show proof of principle that maternal high-fat feeding has detrimental consequences for the metabolic health of the offspring, the experimental diets utilized are not representative of the obesogenic diets currently consumed by women (and men) living in the western world. In addition, in general, the rodent models of maternal high fat feeding are not associated with maternal obesity, unless they are fed for long periods of time, as rodents are good at regulating their caloric intake in response to high-fat foods which they do not find that palatable. A more recent approach has therefore been to employ highly palatable/cafeteria style diets rich in simple sugars that override the natural satiety signals of rodents and are more representative of a typical human Western diet. Feeding of such diets to pregnant rodents leads to the development of insulin resistance, increased adiposity, and impaired glucose tolerance in the offspring (84, 455, 469). These changes are associated with altered structure and function of central appetite regulatory circuits (84, 251, 535). The effects of maternal diet-induced obesity on offspring body weight and adiposity are most apparent when the offspring themselves are fed an obesogenic diet highlighting an interaction between fetal and early postnatal nutrition (37, 473). This provides one potential explanation for the variation in obesity observed in individuals all living in the same obesogenic environment. Detrimental effects of a maternal obesogenic diet have also been observed in larger animal models such as sheep. Feeding pregnant ewes an obesogenic diet that was 150% of their energy requirement led to increased intramuscular triglyceride and skeletal muscle insulin resistance (567). There is also evidence to suggest that the effect of maternal high-fat feeding/obesity can be transmitted through at least two generations, through both the male and female lineage (130).
Finally, in animal models there is now also emerging evidence to suggest that paternal obesity at the time of conception is associated with metabolic dysfunction in the offspring. Chronic high-fat feeding in male rats led to increased adiposity, glucose intolerance, and insulin resistance in female offspring (357). Most recently it has been shown that these effects of paternal diet-induced obesity on offspring obesity and insulin resistance persist to at least two generations, at least in mice (162). This is accompanied by changes in gene expression in testes and sperm and global DNA methylation in the sperm.
3. Prenatal stress and offspring obesity and diabetes
Psychosocial stress is a common factor in human existence. Such stress increases the likelihood of becoming obese and diabetic in humans (316, 449) and rodents (140, 431, 503). Not surprisingly, severe stress during pregnancy can have major adverse effects on offspring. Many of these effects are likely due to the fact that cortisol, which is released in stressful situations, can cross the placenta and alter the development of the brain and other organs (137, 237, 364). In addition to a range of abnormalities in behavior and cognitive function (364), there is evidence that severe maternal psychosocial stress is associated with higher BMI, percent body fat, insulin resistance, and abnormal lipid profiles (137, 139) and hypothalamo-pituitary-adrenal dysregulation in young adult offspring (138). Much of our knowledge of the mechanisms underlying these abnormalities comes from rodent studies in which dams are subjected to different types of stress and/or corticosteroids during various stages of gestation. As a broad generalization, depending on the stage of pregnancy, prenatal stress or exogenous glucocorticoids can have a major adverse impact on the development of the brain, including neurotransmitter systems and brain areas involved in the regulation of energy and glucose homeostasis (161, 237, 437) and pathways regulating motivated and reward behaviors (208, 323). Depending on the timing of stress and the sex of the offspring, adverse offspring outcomes of prenatal stress include permanent dysfunction of the neuroendocrine axis (237) and stress responsiveness (160, 208), delayed learning (160) and abnormal glucose tolerance, hyperphagia, as well as increased body weight and adiposity (111, 363, 387, 508, 546). Importantly, prenatal stress results in less maternal grooming and attention in offspring (81, 418), which can have important effects on offspring behavior and metabolic phenotype (80, 81, 95). In keeping with these fetal/neonate-maternal interactions, at least some of the abnormalities in offspring stress responsivity can be reversed by blocking the mother's stress-induced corticosterone response (32), by fostering their offspring to nonstressed dams (32) or by postnatal handling (482). Intriguingly, in addition to an effect of maternal stress on developing offspring, paternal stress prior to mating significantly reduced the stress responsivity of resultant offspring with global changes in transcriptional regulation suggestive of epigenetic programming (444). Unfortunately, no data were presented with regard to either alteration in adiposity or glucose tolerance in this study. Nevertheless, such studies, if they can be translated to the human condition, suggest that much of the damage done by prenatal stress can be undone by either ameliorating the mother's stress response or by postnatal manipulations that control the offspring-mother interactions.
4. Gestational diabetes
Initial epidemiological studies highlighted the association between low birth weight and increased metabolic disease risk in later life, observations that have been reproduced in over 40 populations worldwide (356). However, in some of these studies, such as those of native North American population, increased risk of T2DM and metabolic syndrome was also observed at the high birth weight end of the spectrum (324). These populations have a high prevalence of T2DM, obesity, and consequently gestational diabetes (>10% of all pregnancies) (157). Therefore, the increased risk of metabolic disease in individuals with high birth weight was proposed to reflect an increased risk of diabetes in the macrosomic offspring of women with gestational diabetes (108, 318, 403, 536). This hypothesis is supported by sib pair studies that have demonstrated a greater prevalence of T2DM and high BMI in siblings born after the mother was diagnosed with T2DM compared with those born prior to the development of T2DM (109). Further evidence for the association between maternal gestational diabetes and increased offspring weight being causative has come from a retrospective study that demonstrated that intensive treatment by diet and/or insulin of gestational diabetic mothers attenuated this association (212).
Studies in animal models have also provided strong evidence that gestational diabetes can cause increased risk of diabetes in the offspring (Figure 2). In most rodent studies, the effects of maternal diabetes have generally been assessed using models where diabetes is induced in the mother by chemical destruction of the maternal β-cells using streptozotocin (reviewed by Van Assche et al., 528). The phenotype of the offspring is determined by the severity of the glucose intolerance induced in the mother. The offspring of mildly diabetic mothers are large at birth and in neonatal life demonstrate an apparent enhanced development of their endocrine pancreas. However, in adulthood they have a deficit in their insulin secreting capacity (199) and develop impaired glucose tolerance (6, 472). The offspring are also hyperphagic, leptin resistant, and obese (491). This is associated with hypothalamic defects (409) including a reduction in neuronal connections between the ARC and the PVN (491). If the maternal diabetes is severe, the offspring are born small for gestational age. As a result of overstimulation by the high glucose levels, the offspring β-cells are almost completely degranulated with lower insulin content and the offspring become insulin resistant as adults (6). In light of the growing epidemic of obesity, a growing number of animal models of maternal diet-induced obesity are being established (see above and below). In some of these it has been demonstrated (unsurprisingly) that the dams develop impaired glucose tolerance during pregnancy. Although gestational diabetes is not the only altered metabolic parameter in these models, it is conceivable that at least some of the detrimental consequences of maternal obesity in the offspring are caused by accompanying gestational diabetes.
B. Postnatal Influences on Offspring Metabolic Outcomes
1. Maternal-infant interactions
Early infancy exposure to a variety of experiences and metabolic milieus can have an important impact on the ways in which the infant learns to cope with their environment. The content of breast milk is influenced by the physiological and metabolic state of the mother and can have important effects on the metabolic state and feeding preferences of their infants. Hormones such as leptin and insulin are secreted into the milk and, during early infancy, can be absorbed directly into the bloodstream of suckling infants (78, 176, 213, 234, 349). In addition, the milk content of nutrients such as essential fatty acids which are required for neural development (524) are heavily influenced by the genetic and metabolic status of the mother (176). While many studies support a protective effect of breast versus formula feeding during infancy against later obesity and glucose intolerance (104, 154, 266, 379), some suggest that factors such as maternal diabetes might have an adverse effect on the metabolic development of their offspring (408). In rodents, cross-fostering of genetically obesity-resistant (DR) pups to obese dams with a genetic propensity to become obese on high-fat diets (DIO) causes them to become obese and insulin resistant when subsequently exposed to a high-fat diet as adults (176). Much of this effect may be attributed to abnormalities in milk content of nutrients such as poly- and monounsaturated fatty acids and hormones such as insulin and leptin which are essential for normal brain development (176). Similarly, dietary choices of the breast-feeding mother or early exposure to specific tastes and orders in infant formulas can have marked effects on dietary and taste preferences of the developing infant (154, 329–331, 518). In both humans and experimental animals, the major issue left unanswered is what basic mechanisms underlie these persistent changes in behavior as well as metabolic and physiological function. Some are associated with changes in the anatomical development of pathways critical to these functions (62), while others may be due to epigenetic changes in gene expression, or both.
2. Catch-up growth in intrauterine growth retardation and accelerated postnatal growth
Accelerated early neonatal growth and/or obesity has been shown to amplify the detrimental consequences of being born small for gestational age on metabolic health outcomes. The original Hertfordshire studies by Hales et al. (198) demonstrated that the men with the worst glucose tolerance at age 64 were those that were in the lowest quartile of birth weight but who were obese as adults. Likewise, in the Dutch Hunger Winter studies, the worst glucose tolerance was observed in individuals who were exposed to famine in utero but became obese as adults (427). The particular detrimental effects of rapid growth during childhood following fetal growth restriction emerged from a study of primary school children in South Africa. Those with a low birth weight who gained weight rapidly during early childhood had the worst glucose tolerance at age 7 (102). Studies in Finland also demonstrated that men and women who develop T2DM are those born small for gestational age and then cross BMI centiles between the ages of 2 and 11 (141). These detrimental effects of catch-up growth may be related to the observation that during periods of such accelerated growth there is preferential accumulation of fat mass rather than lean tissue (344). Studies in animal models reinforce this concept that rapid postnatal growth following in utero growth restriction is detrimental to long-term metabolic health, including increased risk of obesity. Rodent models of maternal protein restriction, caloric restriction, and intrauterine artery ligation, which all demonstrate low birth weight, develop increased adiposity when suckled by normally fed dams during the lactation period and therefore undergo postnatal catch up growth (381, 475, 532).
There is now also growing evidence to suggest that accelerated postnatal growth not only exaggerates the effects of suboptimal growth in utero but can also have detrimental effects on later health regardless of an individual's birth weight. This is particularly prominent in relation to risk of increased adiposity and obesity. At least three systematic reviews demonstrate in humans that accelerated postnatal growth increases risk of subsequent obesity (26, 346, 371). These studies show associations, but do not provide information regarding the causes of the accelerated growth. However, in humans, both observational and randomized feeding trials suggest that nutritionally induced rapid weight gain in the first half of infancy predicts later obesity and cardiovascular risk factors such as higher blood pressure (173, 523, 547). Studies comparing breast-fed infants to formula-fed infants revealed that the former were at reduced risk of obesity (18, 200). These observational studies do not provide causal evidence that nutrition per se mediates these relationships. However, it is well known that formula-fed infants gain more weight over the first year of life than breast-fed infants (120). Causal relationships between nutrition during infancy and subsequent metabolic health have emerged from randomized intervention studies and control trials. In these studies low levels of nutrient intake during the neonatal period are protective against risk of obesity and cardiovascular disease (257, 476, 477). The precise duration of this early neonatal critical time window for determination of obesity risk is not clear. However, it has been suggested that it could be as little as the first postnatal week of life (495). Animal models have again confirmed these studies in humans. Use of a range of animal models has repeatedly confirmed the fact that early overnutrition in the neonatal period predisposes to later obesity (Figure 2). Raising rodent pups in small litters increases their intake and markedly increases their propensity to become obese as adults (231, 246). Similarly, overfeeding neonatal rats for the first 18 days of life by intragastric tubes markedly increases their body weight gain (549). On the other hand, raising rodent pups in large litters restricts their access to food and can protect even genetically obesity-prone animals from becoming obese (231, 392).
VI. GENE-ENVIRONMENT INTERACTIONS
A. Epigenetics
The term epigenetics (literally meaning “above the genetics”) was first defined by the developmental biologist Conrad Waddington as the “interactions of genes with their environment which bring the phenotype into being” (539). The epigenetic changes that mediate this interaction include alterations in DNA methylation, covalent modifications of histone tails (e.g., acetylation, methylation, phosphorylation, and ubiquitination), and expression of noncoding RNAs (e.g., miRNAs). The phenomenon of epigenetics therefore explains how one genotype can give rise to multiple different phenotypes through alterations in the epigenotype. It also provides a molecular framework through which the environment can interact with the genome to alter gene expression and thereby influence phenotype. As gene-environment interactions are key to the concept of developmental programming, much attention has been directed towards the potential role of epigenetic mechanisms in mediating the effects of a suboptimal exposure of a fetus in utero to permanent changes in its long-term metabolic health including risk of T2DM and obesity. Epigenetics provides an attractive mechanism to underlie the cellular memory by which a suboptimally exposed cell during a critical period of development stably affects gene expression following multiple rounds of cell division.
The potential for diet during pregnancy to permanently alter the epigenotype and therefore adult phenotype and disease susceptibility was first demonstrated 15 years ago using the Agouti viable yellow (Avy) mouse (559). The Avy allele is epigenetically sensitive as a result of a retrotransposon insertion upstream of the Agouti gene. When the retrotransposon is hypermethylated and thus silenced, the agouti gene is expressed only in skin and produces a lean mouse with an agouti-colored coat (termed pseudo-agouti). In contrast, hypomethylation of the retrotransposon generates a ubiquitously expressed transcript that causes yellow coat color and obesity. It has been demonstrated that when Avy pregnant dams are fed a diet supplemented in methyl donors and cofactors (e.g., choline, folic acid, vitamin B12) they tend to have offspring that are pseudo-agouti and lean rather than being yellow and obese as seen when the Agouti gene is ubiquitously active (543, 559). This effect of maternal diet on offspring coat color occurred by increasing the level of methylation at the Avy allele.
Rodent models have also demonstrated that physiological changes in maternal diet in wild-type animals can alter the epigenotype of the offspring (reviewed in 380). Transcription factors have emerged as common targets of epigenetic programming by changes in the early environment. They are also conceptually attractive targets of programming. Through epigenetic programming of such factors, a coordinated network of genes can be modified since transcription factors themselves regulate expression of gene networks. Examples of transcription factors that are epigenetically programmed through changes in DNA methylation and histone modifications include the pancreatic β-cell developmental transcription factors and MODY genes PDX-1 (as a result of placental insufficiency) (389) and HNF-α (as a result of maternal protein restriction) (457). Both of these studies demonstrated that the programmed changes were dynamic and changed as the animals aged, perhaps explaining the importance of the ageing process in development of the diabetic phenotype in both of these animal models of programming. Peroxisome proliferator-activated receptor-α in the liver is another example of a transcription factor that is epigenetically programmed by maternal protein restriction in rats (311). Studies in ovine models have also demonstrated that maternal undernutrition can lead to epigenetic alterations in the offspring. For example, periconceptual undernutrition in sheep led to changes in the methylation of the POMC locus in the fetus later on in gestation (496). Altered DNA methylation of a gene involved in gluconeogenesis (PEPCK1) has also been reported in a non-human primate model of maternal undernutrition during pregnancy (360).
In addition to programmed epigenetic changes in response to fetal/maternal undernutrition, animal models have also highlighted the potential for maternal overnutrition to influence epigenetic modifications in the offspring. Using a mouse model of maternal high-fat feeding, Vucetic et al. (537) demonstrated that components of central reward pathways such as the μ-opioid receptor were susceptible to epigenetic programming by maternal overnutrition. Maternal diet-induced obesity in rats has also been shown to lead to programmed changes in DNA methylation of pro-adipogenic genes (including C/EBP-β) in adipose tissue in the offspring (56). Growing evidence suggesting that epigenetic programming by overnutrition not only occurs through the maternal line but can also be transmitted through the paternal line is emerging. Chronic high-fat feeding of male rats led to pancreatic β-cell dysfunction in female offspring that was associated with changes in methylation of the Il13ra2 locus (357). Furthermore, consistent with epidemiological evidence, overnutrition during the neonatal period leads to permanent changes in epigenetic modifications. Using a model of neonatal overfeeding through litter size manipulation, Plagemann and colleagues (407, 414) demonstrated programmed changes in the POMC and insulin receptor loci in the hypothalamus.
Data from humans in relation to evidence for epigenetic modifications contributing to the developmental origins of T2DM and obesity are much more limited and are often hindered by the lack of availability of metabolically relevant tissues from living humans. The majority of studies have therefore focused on clinically accessible tissues such as white blood cells or umbilical cord. However, a major goal has been to identify epigenetic changes in these tissues that are reflective of epigenetic changes in tissues such as adipose tissue, the brain, and the endocrine pancreas. Genome-wide methylation analysis of cord blood cells demonstrated that intrauterine growth restriction in humans was associated with altered methylation of the HNF-α locus, again highlighting the potential importance of programming of transcription factors (132). Human studies have also demonstrated association between patterns of early postnatal growth and epigenetic modifications. Groom et al. (185) reported a link between rapid postnatal growth and differential methylation of the TACSTD2 locus, a gene associated with childhood adiposity. Evidence for the effects of diet during pregnancy and epigenetic changes in the offspring in humans is sparse, and most has come from studies of individuals who were in utero during the Dutch Hunger Winter. Initial studies of this cohort identified differential methylation of the Igf2 locus six decades after exposure to the famine in utero (207), and a further five vulnerable loci were identified in a subsequent study (514). Other human studies have demonstrated the potential use of epigenetic modifications as markers of future risk of metabolic disease. In two separate cohorts, Godfrey et al. (175) demonstrated that methylation of the retinoid X receptor in umbilical cord tissue correlated strongly with percent fat mass later on in childhood and explained ∼25% of the variation in adiposity.
In addition to studies showing associations between changes in early patterns of growth and nutrition, there are also a limited number of studies showing epigenetic variation in candidate genes associated with T2DM and obesity. Small but significant differences in methylation of FTO (39), insulin (568), and KCNQ1 (517) loci have all been shown to correlate with disease risk. Furthermore, there is evidence that lifestyle factors associated with changes in obesity risk can alter promoter methylation of key genes in skeletal muscle including PGC-1α, PDK4, and PPAR-δ (33).
B. Hormonal Influences
As discussed above, a plethora of data from rodent and human studies have suggested that changes in nutrition during perinatal life have a significant impact on the development of obesity and related diseases in later life. Hormones, such as leptin, insulin, and ghrelin, are dynamically regulated by nutritional and metabolic status and are therefore major signals to the developing fetus and neonate of nutrient availability (Figure 2). In addition, hormones produce a multitude of effects on functions in the developing fetus and neonate that are well outside the functions they serve in later life. Thus the biological actions of several metabolic hormones are different during neonatal versus adult epochs. For example, in sharp contrast to the potent effects of leptin and ghrelin on feeding in adults, peripheral leptin or ghrelin injections have no significant effects on milk intake or body weight during the first 2–3 wk of postnatal life in rats and mice (340, 404, 490). These observations suggest that leptin and ghrelin might exert different functions during neonatal life such as altering neural development. Early observations by Bereiter and Jeanrenaud (40, 41) reported structural defects in the obese ob/ob mice, including a reduction in soma size of cells in the VMN and dorsal motor vagal nucleus neurons, as well as alterations in the dendritic orientation of VMN and LHA neurons. Twenty years later, Ahima and Flier (8) showed that the same mutant mice display an immature pattern of expression of synaptic and glial proteins. This pioneer work paved the way for subsequent research on leptin in brain development and plasticity.
The availability of ob/ob mice and more modern neuroanatomical tools to study neural circuits allowed more detailed studies on the role of leptin on hypothalamic development. Axonal tracing of ARC neurons demonstrated that the leptin deficiency permanently disrupts the development of projections from the ARC to each of its major targets, including the PVN (61). Remarkably, peripheral leptin injection in ob/ob neonates restores the density of ARC axons to a density that was comparable to that of wild-type littermates, but the treatment of adult ob/ob mice with leptin is largely ineffective (61). Also, leptin restores normal brain weight in ob/ob mice but only when the hormone is injected during early life (494). These observations suggest that leptin acts primarily during a restricted critical neonatal period to exert its neurotrophic effects. Notably, obesogenic environments, such as maternal obesity, diabetes, and postnatal overnutrition, can cause hyperleptinemia throughout postnatal life and impair central leptin sensitivity during critical periods of hypothalamic development (62, 174, 250, 491). Notably, this early leptin resistance is associated with a disrupted development of ARC neural projections to the PVN (62, 174, 250, 491). In contrast, maternal undernutrition during pregnancy and lactation or the postnatal period blunts the naturally occurring postnatal leptin surge and also causes abnormal development of ARC projections (97, 118, 572), and daily leptin treatment during early postnatal life in pups born to undernourished dams normalizes their metabolic abnormalities (533). These findings show the importance of neonatal leptin in life-long metabolic regulation and raise the importance of early endocrine intervention in metabolic (mal)programming.
More recent studies have also implicated ghrelin in the development of metabolic systems. Ghrelin is one of the first major metabolic hormones to appear during development. It is expressed in embryos as early as the morula stage and continues to be expressed in the developing fetus and neonate. During perinatal development, ghrelin is transiently expressed in the pancreatic α-cells where it colocalizes with glucagon (116). But ghrelin is also produced by the pancreatic β-cells (419). This transient expression of ghrelin appears to play a role in pancreas development. Newborn rats exposed to ghrelin for 7 or 14 days had reduced pancreatic weights, attenuated pancreatic DNA synthesis, and reduced DNA content (119). The morphological effects of neonatal ghrelin appear widespread because chronic neonatal ghrelin injections also reduce growth of the stomach, as evidenced by a decrease in gastric weight, DNA synthesis, and DNA content. On the other hand, ghrelin injections in adult animals increase pancreatic and gastric weight, DNA synthesis, and DNA content (119, 542), indicating that ghrelin can induce biphasic effects on gastric growth depending on the age of exposure.
Ghrelin also exerts developmental effects on the brain. In vitro incubation of hypothalamic and brain stem cells with ghrelin induces proliferation with many of the resultant newborn cells acquiring a neuronal and/or glial phenotype (224, 575, 576). Insulin has also long been associated with brain development. Consistent with a trophic role of insulin in the developing hypothalamus, offspring of insulin-deficient mothers display a reduced number of ARC neurons, and this reduction of neuronal cell number is preventable by the normalization of glycemia using pancreatic islet transplantation (156). Moreover, hypoinsulinemic pups born to protein-restricted dams display a reduction in the number of astrocytes (411), while the offspring of gestationally diabetic mothers, which have increased insulin levels, have increased numbers of astrocytes (409, 412). In addition to influencing hypothalamic cell numbers, insulin can also influence hypothalamic neuronal connectivity. Pups born to insulin-deficient dams display abnormally organized POMC and NPY/AgRP neural projections that could result from the attenuated responsiveness of hypothalamic neurons to the neurotrophic actions of leptin during neonatal development (491). Notably, intrahypothalamic insulin injections during early postnatal life cause life-long metabolic dysregulation, raising the importance of neonatal insulin in the developing brain on life-long metabolic regulation (410, 412).
C. Rodent Models of Gene-Environment Interactions
1. Mouse models
Although transgenic and knockout experiments are typically conducted in mice, a significant variability in adiposity, DIO, and obesity-related diabetes exists among the mouse strains commonly used in laboratory research (see 548 for a review). The inbred C57BL/6J (B6) strain is probably the most widely used strain to conduct transgenic and knockout experiments, in part because of its susceptibility to develop obesity on high-fat diets. C57BL/6J mice are not obese on a standard chow, but when fed a high-fat diet they develop hyperglycemia, hyperinsulinemia, and hyperleptinemia (133, 505, 548). In contrast, some strains, such as 129/Sv and A/J mice, are almost totally resistant to obesity and diabetes when fed a high-fat diet (503). Remarkably, both 129/Sv and C57BL/6J mice eat an equal number of calories when fed a high-fat diet (13), suggesting that C57BL/6J have a higher feeding efficiency and gain greater weight per calorie consumed. Even within the C57 mouse strain there are significant differences among sub-strains in response to the high-fat diet. Thus C57BL/6J mice fed a high-fat diet exhibit a marked metabolic phenotype, whereas C57BL/6KsJ mice only display a weak phenotype (93). Furthermore, in some laboratories it has been noted that C57BL/6J mice within the same colony exhibit a bimodal response to high-fat diet; half develop DIO, and half are obesity-resistant (136). Given the fact that they all share the identical genotype, this marked difference in metabolic phenotypes when offered a high-fat diet suggests the presence of an as yet to be determined epigenetic influence. Background genes also appear to play an important role in determining the metabolic phenotype of mice with naturally occurring mutations or mice that have been genetically altered by introduction of transgenes. For example, ob/ob and db/db mice on the C57BL/Ks background are obese and develop severe diabetes and a marked hyperglycemia, whereas ob/ob mice on the C57BL/6J background are obese but only exhibit mild diabetes and hyperglycemia (92). Similarly, mice with a double-heterozygous deletion of the insulin receptor and insulin receptor substrate-1 become insulin resistant and severely hyperinsulinemic on the C57BL/6J background, but on the 129/Sv background these double mutant mice only exhibit a mild hyperinsulinemia (271). Together, these observations indicate that background genes in mice greatly influence the development of obesity and obesity-related diseases, such as T2DM, in response to either an obesogenic environment or genetic defects.
2. Rat models
The selectively bred DIO and DR strains of rats have proven to be a valuable model for studying the interactions of genes with environment. These strains were derived from the outbred Charles River Sprague-Dawley rat. Sprague-Dawley rats from this breeder have the fairly unique characteristic of showing a wide variation in body weight and adipose gain when placed on a relatively high-fat (31%), high-sucrose (25%) diet, designated as a “high energy” (HE) diet (296). Approximately half the rats placed on such a diet overeat for 4–6 wk and become obese (296). The remaining rats overeat for only a few days and gain no more weight than controls fed a low-fat chow diet (299). Importantly, these outbred rats have been selectively bred to produce DIO and DR strains which have maintained their distinctive phenotypes for more than 50 generations. The obesity of the DIO rat appears to have a genetic origin since breeding DIO males with another obesity-resistant strain of rats passes on this phenotype to the offspring of these crosses in an apparently polygenic manner of transmission similar to most human obesity (57, 298, 502). This model is an excellent one for the study of human obesity since, like most obese humans, it maintains its higher body weight and adipose set-points even when switched to a low-fat diet or after being calorically restricted for many weeks (291, 302). This defense of a higher body weight set-point is what occurs in obese humans and is likely the reason for the high recidivism rate in the medical treatment of obesity and the extreme measures many previously obese individuals must undertake to keep off lost weight (326, 448, 538, 557).
The DIO/DR model is extremely useful for the study of gene-environment interactions associated with maternal obesity and insulin resistance since dams can be fed the same high-fat diet but only the DIO dams become obese and insulin resistant during gestation and lactation (176, 177, 294, 300). This obesity of DIO dams is not accompanied by an increase in offspring body weight unless such offspring are also fed HE diet from weaning. As opposed to DIO offspring, offspring of DR dams, whether the dams were made obese with a highly palatable diet or stayed lean on HE diet during gestation and lactation, gained no more weight or adiposity than controls regardless of their postweaning diets. However, maternal obesity, regardless of genotype, was associated with enlargement of the VMN and DMN and differentially affected the density of norepinephrine and serotonin transporters in the PVN (294). On the other hand, offspring of DIO dams, regardless of whether their dams were lean or obese during gestation and lactation, showed defective development of the α-melanocyte stimulating hormone (α-MSH, a catabolic peptide derived from POMC) and AgRP pathways projections from the ARC POMC and NPY/AgRP neurons to the PVN. These defective projections appeared to be due to the inherent leptin resistance of the DIO rat (176, 178, 295, 297, 299, 392), since leptin is required for normal development of this pathway (62).
Although it is uncertain whether DIO pups are born with inherent leptin resistance, it does appear in the first few days of life (62), making this early postnatal period an important focus of potential interventions that might alter later life development of obesity. In fact, cross-fostering DR pups from lean DR dams to obese, but not lean, DIO dams fed HE diet causes them to become obese and insulin resistant when they are fed HE diet as adults (176). This is associated with an “anabolic” shift in the expression of hypothalamic genes including increased ARC AgRP and decreased VMN leptin, insulin, and MC3R expression (176). As mentioned above, the major difference between obese DIO dams and all others was the high insulin and leptin and low mono- and polyunsaturated fatty acid contents of their milk, suggesting an early influence on both brain development and obesity-proneness in this otherwise highly obesity-resistant strain (176). However, cross-fostering DIO pups from either lean or obese DIO dams to lean DR dams had no effect on their propensity to become obese or insulin resistant when fed HE diet as adults. While fostering DIO pups with lean DR dams failed to alter their obesity-prone phenotype, raising them in large litters, which severely restricts intake (246), prevents them from becoming obese as adults. This obesity resistance was associated with normalization of their ARC leptin receptor binding and leptin sensitivity with a presumptively resultant normalization of their ARC-PVN α-MSH and AgRP pathway outgrowth (392). Thus early dietary interventions can have a major effect on later life development of obesity that is highly dependent on the interventions and the genetic makeup of the individual.
While it is clear that interventions during gestation and early postnatal development can have major impacts on the long-term development of systems involved in the regulation of energy and glucose homeostasis, it appears that the critical period for altering some of these systems may extend well into adolescence and early adulthood in both humans and other mammals. An excellent example of this is the effect of early-onset exercise on altering the development of obesity in selectively bred DIO rats. These rats are intrinsically less active than DR rats both in their home cages (260) and in running wheels. Yet, when running wheels are made available, only DIO rats lose weight despite running only half as much as DR rats (293). When early postweaning DIO rats are fed a high-fat diet and simultaneously provided with a running wheel for 3 wk or more, they have a persistent protection from becoming obese which is associated with increased leptin signaling in the ARC and VMN (394). Because ARC-PVN α-MSH and NPY/AgRP pathways have already completed their development by the time the wheels are introduced (60), the increased leptin signaling and obesity protection are not associated with changes in the density of these fiber pathways (391). However, this does not exclude the possibility that other brain areas that have not fully developed might be altered by such early-onset exercise. In addition, it is possible that the increased leptin signaling might be associated with epigenetic changes, although this possibility has not been explored.
VII. HOW CAN WE USE THIS INFORMATION TO PREVENT AND TREAT OBESITY AND DIABETES?
This review presents data that clearly demonstrate the major importance of the perinatal environment and genetic predisposition in determining the development of neural pathways and organs involved in the regulation of energy and glucose homeostasis. While we do understand many of the predisposing factors, it remains challenging to identify those individuals who are most at risk and the predisposing factors that push them into a vicious cycle of obesity and insulin resistance from which few can recover. Because organs, particularly the brain, undergo the majority of their development during the perinatal period, there is a premium on identifying at risk individuals and risk factors during this critical period. Importantly, while most organs undergo continuing change of structure and function throughout life, the brain is much less plastic with regard to changing the connections of critical neuronal pathways established during critical periods of early development. The problem is that, even if we could reliably identify such individuals and risk factors, we are a long way from knowing how to alter the perinatal environment to prevent offspring from being set on the path to near-permanent predisposition to obesity and diabetes.
Also, we understand even less about the factors that make obesity, once it develops, a near-permanent condition in so many individuals. Given our current state of knowledge, there are some possible guidelines, although some of these are based on animal research that might not apply to humans. First, several factors increase the probability of offspring obesity and/or diabetes. These include obesity in one or both parents, gestational diabetes, intake of a high-fat, calorically dense diet during pregnancy and lactation, gestational undernutrition with postnatal overfeeding (“catch up growth”), genetic mutations known to cause obesity in affected individuals, and possibly some gene variants which have a high association with obesity such as FTO. However, it is important to recognize that these latter gene variants are only associations, and we are a long way from understanding the combinations of genes and the epigenetic modifications of these and other genes that promote obesity. Similarly, while research in animal models has identified several factors that appear to adversely alter the development of neural pathways involved in the regulation of energy and glucose homeostasis, it is unclear if these same factors apply to humans and, if they do, the stage of gestational and postnatal development which is most at risk. Finally, even if we could identify at risk individuals and obesogenic factors, changing the perinatal environment is a socioeconomic and cultural challenge for which we have so far failed to find a practical solution in the vast majority of at risk individuals. The hope would be that continued research into the factors that predispose individuals to become obese might identify those that lend themselves to relatively simple, straightforward interventions.
GRANTS
This work was supported by National Institute of Diabetes and Digestive and Kidney Diseases Grants R01 30066 and 53181 and the Research Service of the Veterans Administration (to B. E. Levin). S. E. Ozanne is supported by the MRC Metabolic Diseases Unit (MRC_MC_UU_12012/4). S. G. Bouret is supported by the National Institutes of Health Grants R01DK84142 and P01ES022845, United States Environment Protection Agency Grant RD83544101, the Foundation for Prader-Willi Research, and the EU FP7 integrated project (grant agreement no. 266408, “Full4Health”).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
Acknowledgments
Present addresses: S. Bouret, The Saban Research Institute, Neuroscience Program, Childrens Hospital Los Angeles, Univ. of Southern California, Los Angeles, CA 90027 (e-mail: sbouret@chla.usc.edu); and S. E. Ozanne, Univ. of Cambridge Institute of Metabolic Science and MRC Metabolic Diseases Unit, Cambridge CB2 2QR, UK (e-mail: seo10@mole.bio.cam.ac.uk).
Address for reprint requests and other correspondence: B. E. Levin, Neurology Service (127C), VA Medical Center, 385 Tremont Ave., East Orange, NJ 07018 (e-mail: levin@njms.rutgers.edu).
REFERENCES
- 1.Abbott CR, Monteiro M, Small CJ, Sajedi A, Smith KL, Parkinson JR, Ghatei MA, Bloom SR. The inhibitory effects of peripheral administration of peptide YY(3–36) and glucagon-like peptide-1 on food intake are attenuated by ablation of the vagal-brainstem-hypothalamic pathway. Brain Res 1044: 127–131, 2005. [DOI] [PubMed] [Google Scholar]
- 2.Abel ED, Peroni O, Kim JK, Kim YB, Boss O, Hadro E, Minnemann T, Shulman GI, Kahn BB. Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver. Nature 409: 729–733, 2001. [DOI] [PubMed] [Google Scholar]
- 3.Abizaid A, Liu ZW, Andrews ZB, Shanabrough M, Borok E, Elsworth JD, Roth RH, Sleeman MW, Picciotto MR, Tschop MH, Gao XB, Horvath TL. Ghrelin modulates the activity and synaptic input organization of midbrain dopamine neurons while promoting appetite. J Clin Invest 116: 3229–3239, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Adams KF, Schatzkin A, Harris TB, Kipnis V, Mouw T, Ballard-Barbash R, Hollenbeck A, Leitzmann MF. Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N Engl J Med 355: 763–778, 2006. [DOI] [PubMed] [Google Scholar]
- 5.Adrian TE, Ferri GL, Bacarese-Hamilton AJ, Fuessl HS, Polak JM, Bloom SR. Human distribution and release of a putative new gut hormone, peptide YY. Gastroenterology 89: 1070–1077, 1985. [DOI] [PubMed] [Google Scholar]
- 6.Aerts L, Sodoyez-Goffaux F, Sodoyez JC, Malaisse WJ, Van Assche FA. The diabetic intrauterine milieu has a long-lasting effect on insulin secretion by b cells and on insulin uptake by target tissues. Am J Obstet Gynecol 159: 1287–1292, 1988. [DOI] [PubMed] [Google Scholar]
- 7.Ageno W, Becattini C, Brighton T, Selby R, Kamphuisen PW. Cardiovascular risk factors and venous thromboembolism: a meta-analysis. Circulation 117: 93–102, 2008. [DOI] [PubMed] [Google Scholar]
- 8.Ahima RS, Bjorbaek C, Osei S, Flier JS. Regulation of neuronal and glial proteins by leptin: implications for brain development. Endocrinology 140: 2755–2762, 1999. [DOI] [PubMed] [Google Scholar]
- 9.Air EL, Benoit SC, Blake Smith KA, Clegg DJ, Woods SC. Acute third ventricular administration of insulin decreases food intake in two paradigms. Pharmacol Biochem Behav 72: 423–429, 2002. [DOI] [PubMed] [Google Scholar]
- 10.Alfaradhi MZ, Ozanne SE. Developmental programming in response to maternal overnutrition. Front Genet 2: 27, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alharbi KK, Spanakis E, Tan K, Smith MJ, Aldahmesh MA, O'Dell SD, Sayer AA, Lawlor DA, Ebrahim S, Davey Smith G, O'Rahilly S, Farooqi S, Cooper C, Phillips DI, Day IN. Prevalence and functionality of paucimorphic and private MC4R mutations in a large, unselected European British population, scanned by meltMADGE. Hum Mutat 28: 294–302, 2007. [DOI] [PubMed] [Google Scholar]
- 13.Almind K, Kahn CR. Genetic determinants of energy expenditure and insulin resistance in diet-induced obesity in mice. Diabetes 53: 3274–3285, 2004. [DOI] [PubMed] [Google Scholar]
- 14.Altman J, Bayer SA. The development of the rat hypothalamus. Adv Anat Embryol Cell Biol 100: 1–178, 1986. [PubMed] [Google Scholar]
- 15.Anand BK, Brobeck JR. Hypothalamic control of food intake in rats and cats. Yale J Biol Med 24: 123–146, 1951. [PMC free article] [PubMed] [Google Scholar]
- 16.Anand BK, China GS, Sharma KN, Dua S, Singh B. Activity of single neurons in the hypothalamus feeding centers: effect of glucose. Am J Physiol 207: 1146–1154, 1964. [DOI] [PubMed] [Google Scholar]
- 17.Andrews ZB, Liu ZW, Walllingford N, Erion DM, Borok E, Friedman JM, Tschop MH, Shanabrough M, Cline G, Shulman GI, Coppola A, Gao XB, Horvath TL, Diano S. UCP2 mediates ghrelin's action on NPY/AgRP neurons by lowering free radicals. Nature 454: 846–851, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Arenz S, Ruckerl R, Koletzko B, von Kries R. Breast-feeding and childhood obesity–a systematic review. Int J Obesity Relat Metab Disorders 28: 1247–1256, 2004. [DOI] [PubMed] [Google Scholar]
- 19.Arnold M, Mura A, Langhans W, Geary N. Gut vagal afferents are not necessary for the eating-stimulatory effect of intraperitoneally injected ghrelin in the rat. J Neurosci 26: 11052–11060, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ashford MLJ, Boden PR, Treherne JM. Glucose-induced excitation of hypothalamic neurones is mediated by ATP-sensitive K+ channels. Pflügers Arch 415: 479–483, 1990. [DOI] [PubMed] [Google Scholar]
- 21.Asrih M, Jornayvaz FR. Inflammation as a potential link between nonalcoholic fatty liver disease and insulin resistance. J Endocrinol 218: 25–36, 2013. [DOI] [PubMed] [Google Scholar]
- 22.Awdeh ZL, Yunis EJ, Audeh MJ, Fici D, Pugliese A, Larsen CE, Alper CA. A genetic explanation for the rising incidence of type 1 diabetes, a polygenic disease. J Autoimmun 27: 174–181, 2006. [DOI] [PubMed] [Google Scholar]
- 23.Bagdade JD, Bierman EL, Porte D Jr. The significance of basal insulin levels in the evaluation of the insulin response to glucose in diabetic and nondiabetic subjects. J Clin Invest 46: 1549–1557, 1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology 132: 2131–2157, 2007. [DOI] [PubMed] [Google Scholar]
- 25.Bai FL, Yamano M, Shiotani Y, Emson PC, Smith AD, Powell JF, Tohyama M. An arcuato-paraventricular and dorsomedial hypothalamic neuropeptide Y containing system which lacks norepinephrine in the rat. Brain Res 331: 172–175, 1985. [DOI] [PubMed] [Google Scholar]
- 26.Baird J, Fisher D, Lucas P, Kleijnen J, Roberts H, Law C. Being big or growing fast: systematic review of size and growth in infancy and later obesity. Bone Miner J 331: 929, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Balthasar N, Coppari R, McMinn J, Liu SM, Lee CE, Tang V, Kenny CD, McGovern RA, Chua SC Jr, Elmquist JK, Lowell BB. Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron 42: 983–991, 2004. [DOI] [PubMed] [Google Scholar]
- 28.Banks WA, Jaspan JB, Huang W, Kastin AJ. Transport of insulin across the blood-brain barrier: saturability at euglycemic doses of insulin. Peptides 18: 1423–1429, 1997. [DOI] [PubMed] [Google Scholar]
- 29.Banks WA, Kastin AJ. Blood to brain transport of interleukin links the immune and central nervous systems. Life Sci 48: 117–121, 1991. [DOI] [PubMed] [Google Scholar]
- 30.Banks WA, Kastin AJ. Differential permeability of the blood-brain barrier to two pancreatic peptides: insulin and amylin. Peptides 19: 883–889, 1998. [DOI] [PubMed] [Google Scholar]
- 31.Banks WA, Kastin AJ, Huang W, Jaspan JB, Maness LM. Leptin enters the brain by a saturable system independent of insulin. Peptides 17: 305–311, 1996. [DOI] [PubMed] [Google Scholar]
- 32.Barbazanges A, Piazza PV, Le Moal M, Maccari S. Maternal glucocorticoid secretion mediates long-term effects of prenatal stress. J Neurosci 16: 3943–3949, 1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Barres R, Yan J, Egan B, Treebak JT, Rasmussen M, Fritz T, Caidahl K, Krook A, O'Gorman DJ, Zierath JR. Acute exercise remodels promoter methylation in human skeletal muscle. Cell Metab 15: 405–411, 2012. [DOI] [PubMed] [Google Scholar]
- 34.Batterham RL, Cohen MA, Ellis SM, Le Roux CW, Withers DJ, Frost GS, Ghatei MA, Bloom SR. Inhibition of food intake in obese subjects by peptide YY3–36. N Engl J Med 349: 941–948, 2003. [DOI] [PubMed] [Google Scholar]
- 35.Batterham RL, Cowley MA, Small CJ, Herzog H, Cohen MA, Dakin CL, Wren AM, Brynes AE, Low MJ, Ghatei MA, Cone RD, Bloom SR. Gut hormone PYY(3–36) physiologically inhibits food intake. Nature 418: 650–654, 2002. [DOI] [PubMed] [Google Scholar]
- 36.Bauer F, Elbers CC, Adan RA, Loos RJ, Onland-Moret NC, Grobbee DE, van Vliet-Ostaptchouk JV, Wijmenga C, van der Schouw YT. Obesity genes identified in genome-wide association studies are associated with adiposity measures and potentially with nutrient-specific food preference. Am J Clin Nutr 90: 951–959, 2009. [DOI] [PubMed] [Google Scholar]
- 37.Bayol SA, Farrington SJ, Stickland NC. A maternal “junk food” diet in pregnancy and lactation promotes an exacerbated taste for “junk food” and a greater propensity for obesity in rat offspring. Br J Nutr 98: 843–851, 2007. [DOI] [PubMed] [Google Scholar]
- 38.Beck B, Burlet A, Nicolas JP, Burlet C. Hyperphagia in obesity is associated with a central peptidergic dysregulation in rats. J Nutr 120: 806–811, 1990. [DOI] [PubMed] [Google Scholar]
- 39.Bell CG, Finer S, Lindgren CM, Wilson GA, Rakyan VK, Teschendorff AE, Akan P, Stupka E, Down TA, Prokopenko I, Morison IM, Mill J, Pidsley R, International Type 2 Diabetes 1q C Deloukas P, Frayling TM, Hattersley AT, McCarthy MI, Beck S, Hitman GA. Integrated genetic and epigenetic analysis identifies haplotype-specific methylation in the FTO type 2 diabetes and obesity susceptibility locus. PloS One 5: e14040, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bereiter DA, Jeanrenaud B. Altered dendritic orientation of hypothalamic neurons from genetically obese (ob/ob) mice. Brain Res 202: 201–206, 1980. [PubMed] [Google Scholar]
- 41.Bereiter DA, Jeanrenaud B. Altered neuroanatomical organization in the central nervous system of the genetically obese (ob/ob) mouse. Brain Res 165: 249–260, 1979. [DOI] [PubMed] [Google Scholar]
- 42.Bergen HT, Mizuno T, Taylor J, Mobbs CV. Resistance to diet-induced obesity is associated with increased proopiomelanocortin mRNA and decreased neuropeptide Y mRNA in the hypothalamus. Brain Res 851: 198–203, 1999. [DOI] [PubMed] [Google Scholar]
- 43.Bergen HT, Mizuno TM, Taylor J, Mobbs CV. Hyperphagia and weight gain after gold-thioglucose: relation to hypothalamic neuropeptide Y and proopiomelanocortin. Endocrinology 139: 4483–4488, 1998. [DOI] [PubMed] [Google Scholar]
- 44.Berglund ED, Vianna CR, Donato J Jr, Kim MH, Chuang JC, Lee CE, Lauzon DA, Lin P, Brule LJ, Scott MM, Coppari R, Elmquist JK. Direct leptin action on POMC neurons regulates glucose homeostasis and hepatic insulin sensitivity in mice. J Clin Invest 122: 1000–1009, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Berthoud HR, Lenard NR, Shin AC. Food reward, hyperphagia, obesity. Am J Physiol Regul Integr Comp Physiol 300: R1266–R1277, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Berthoud HR. Multiple neural systems controlling food intake and body weight. Neurosci Biobehav Rev 26: 393–428, 2002. [DOI] [PubMed] [Google Scholar]
- 47.Bittar PG, Charnay Y, Pellerin L, Bouras C, Magistretti PJ. Selective distribution of lactate dehydrogenase isoenzymes in neurons and astrocytes of human brain. J Cereb Blood Flow Metab 16: 1079–1089, 1996. [DOI] [PubMed] [Google Scholar]
- 48.Blazquez C, Sanchez C, Velasco G, Guzman M. Role of carnitine palmitoyltransferase I in the control of ketogenesis in primary cultures of rat astrocytes. J Neurochem 71: 1597–1606, 1998. [DOI] [PubMed] [Google Scholar]
- 49.Blazquez C, Woods A, de Ceballos ML, Carling D, Guzman M. The AMP-activated protein kinase is involved in the regulation of ketone body production by astrocytes. J Neurochem 73: 1674–1682, 1999. [DOI] [PubMed] [Google Scholar]
- 50.Blouet C, Schwartz GJ. Hypothalamic nutrient sensing in the control of energy homeostasis. Behav Brain Res 209: 1–12, 2010. [DOI] [PubMed] [Google Scholar]
- 51.Bo S, Cavallo-Perin P, Gentile L, Repetti E, Pagano G. Influence of a familial history of diabetes on the clinical characteristics of patients with Type 2 diabetes mellitus. Diabetic Med 17: 538–542, 2000. [DOI] [PubMed] [Google Scholar]
- 52.Bogers RP, Bemelmans WJ, Hoogenveen RT, Boshuizen HC, Woodward M, Knekt P, van Dam RM, Hu FB, Visscher TL, Menotti A, Thorpe RJ Jr, Jamrozik K, Calling S, Strand BH, Shipley MJ, Investigators B-CC. Association of overweight with increased risk of coronary heart disease partly independent of blood pressure and cholesterol levels: a meta-analysis of 21 cohort studies including more than 300,000 persons. Arch Internal Med 167: 1720–1728, 2007. [DOI] [PubMed] [Google Scholar]
- 53.Boney CM, Verma A, Tucker R, Vohr BR. Metabolic syndrome in childhood: association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 115: e290–296, 2005. [DOI] [PubMed] [Google Scholar]
- 54.Bonnefond A, Froguel P, Vaxillaire M. The emerging genetics of type 2 diabetes. Trends Mol Med 16: 407–416, 2010. [DOI] [PubMed] [Google Scholar]
- 55.Boren J, Taskinen MR, Olofsson SO, Levin M. Ectopic lipid storage and insulin resistance: a harmful relationship. J Internal Med 274: 25–40, 2013. [DOI] [PubMed] [Google Scholar]
- 56.Borengasser SJ, Zhong Y, Kang P, Lindsey F, Ronis MJ, Badger TM, Gomez-Acevedo H, Shankar K. Maternal obesity enhances white adipose tissue differentiation and alters genome-scale DNA methylation in male rat offspring. Endocrinology 154: 4113–4125, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bouchard C, Perusse L. Genetics of obesity. Annu Rev Nutr 13: 337–354, 1993. [DOI] [PubMed] [Google Scholar]
- 58.Bouret SG. Neurodevelopmental actions of leptin. Brain Res 1350: 2–9, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bouret SG. Role of early hormonal and nutritional experiences in shaping feeding behavior and hypothalamic development. J Nutr 140: 653–657, 2010. [DOI] [PubMed] [Google Scholar]
- 60.Bouret SG, Draper SJ, Simerly RB. Formation of projection pathways from the arcuate nucleus of the hypothalamus to hypothalamic regions implicated in the neural control of feeding behavior in mice. J Neurosci 24: 2797–2805, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bouret SG, Draper SJ, Simerly RB. Trophic action of leptin on hypothalamic neurons that regulate feeding. Science 304: 108–110, 2004. [DOI] [PubMed] [Google Scholar]
- 62.Bouret SG, Gorski JN, Patterson CM, Chen S, Levin BE, Simerly RB. Hypothalamic neural projections are permanently disrupted in diet-induced obese rats. Cell Metab 7: 179–185, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Brischoux F, Fellmann D, Risold PY. Ontogenetic development of the diencephalic MCH neurons: a hypothalamic “MCH area” hypothesis. Eur J Neurosci 13: 1733–1744, 2001. [DOI] [PubMed] [Google Scholar]
- 64.Broberger C, Holmberg K, Shi TJ, Dockray G, Hokfelt T. Expression and regulation of cholecystokinin and cholecystokinin receptors in rat nodose and dorsal root ganglia. Brain Res 903: 128–140, 2001. [DOI] [PubMed] [Google Scholar]
- 65.Broberger C, Johansen J, Johansson C, Schalling M, Hokfelt T. The neuropeptide Y/agouti gene-related protein (AGRP) brain circuitry in normal, anorectic, and monosodium glutamate-treated mice. Proc Natl Acad Sci USA 95: 15043–15048, 1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Brown MS, Goldstein JL. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab 7: 95–96, 2008. [DOI] [PubMed] [Google Scholar]
- 67.Bruce KD, Cagampang FR, Argenton M, Zhang J, Ethirajan PL, Burdge GC, Bateman AC, Clough GF, Poston L, Hanson MA, McConnell JM, Byrne CD. Maternal high-fat feeding primes steatohepatitis in adult mice offspring, involving mitochondrial dysfunction and altered lipogenesis gene expression. Hepatology 50: 1796–1808, 2009. [DOI] [PubMed] [Google Scholar]
- 68.Bruning JC, Gautam D, Burks DJ, Gillette J, Schubert M, Orban PC, Klein R, Krone W, Muller-Wieland D, Kahn CR. Role of brain insulin receptor in control of body weight and reproduction. Science 289: 2122–2125, 2000. [DOI] [PubMed] [Google Scholar]
- 69.Buckley AJ, Keseru B, Briody J, Thompson M, Ozanne SE, Thompson CH. Altered body composition and metabolism in the male offspring of high fat-fed rats. Metab Clin Exp 54: 500–507, 2005. [DOI] [PubMed] [Google Scholar]
- 70.Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of US adults. N Engl J Med 348: 1625–1638, 2003. [DOI] [PubMed] [Google Scholar]
- 71.Calle EE, Thun MJ, Petrelli JM, Rodriguez C, Heath CW Jr. Body-mass index and mortality in a prospective cohort of US adults. N Engl J Med 341: 1097–1105, 1999. [DOI] [PubMed] [Google Scholar]
- 72.Campfield LA, Smith FJ. Functional coupling between transient declines in blood glucose and feeding behavior: temporal relationships. Brain Res Bull 17: 427–433, 1986. [DOI] [PubMed] [Google Scholar]
- 73.Campfield LA, Smith FJ, Guisez Y, Devos R, Burn P. Recombinant mouse OB protein: evidence for a peripheral signal linking adiposity and central neural networks. Science 269: 546–549, 1995. [DOI] [PubMed] [Google Scholar]
- 74.Campfield LA, Smith FJ, Rosenbaum M. Human hunger: is there a role for blood glucose dynamics? Appetite 18: 244, 1992. [DOI] [PubMed] [Google Scholar]
- 75.Canani RB, Costanzo MD, Leone L, Bedogni G, Brambilla P, Cianfarani S, Nobili V, Pietrobelli A, Agostoni C. Epigenetic mechanisms elicited by nutrition in early life. Nutr Res Rev 24: 198–205, 2011. [DOI] [PubMed] [Google Scholar]
- 76.Caron E, Sachot C, Prevot V, Bouret SG. Distribution of leptin-sensitive cells in the postnatal and adult mouse brain. J Comp Neurol 518: 459–476, 2010. [DOI] [PubMed] [Google Scholar]
- 77.Carone BR, Fauquier L, Habib N, Shea JM, Hart CE, Li R, Bock C, Li C, Gu H, Zamore PD, Meissner A, Weng Z, Hofmann HA, Friedman N, Rando OJ. Paternally induced transgenerational environmental reprogramming of metabolic gene expression in mammals. Cell 143: 1084–1096, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Casabiell X, Pineiro V, Tome MA, Peino R, Dieguez C, Casanueva FF. Presence of leptin in colostrum and/or breast milk from lactating mothers: a potential role in the regulation of neonatal food intake. J Clin Endocrinol Metab 82: 4270–4273, 1997. [DOI] [PubMed] [Google Scholar]
- 79.Catalano PM, Farrell K, Thomas A, Huston-Presley L, Mencin P, de Mouzon SH, Amini SB. Perinatal risk factors for childhood obesity and metabolic dysregulation. Am J Clin Nutr 90: 1303–1313, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Champagne DL, Bagot RC, van Hasselt F, Ramakers G, Meaney MJ, de Kloet ER, Joels M, Krugers H. Maternal care and hippocampal plasticity: evidence for experience-dependent structural plasticity, altered synaptic functioning, and differential responsiveness to glucocorticoids and stress. J Neurosci 28: 6037–6045, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Champagne FA, Meaney MJ. Stress during gestation alters postpartum maternal care and the development of the offspring in a rodent model. Biol Psychiatry 59: 1227–1235, 2006. [DOI] [PubMed] [Google Scholar]
- 82.Chen H, Charlat O, Tartaglia LA, Woolf EA, Weng X, Ellis SJ, Lakey ND, Culpepper J, Moore KJ, Breitbart RE, Duyk GM, Tepper RI, Morgenstern JP. Evidence that the diabetes gene encodes the leptin receptor: identification of a mutation in the leptin receptor gene in db/db mice. Cell 84: 491–495, 1996. [DOI] [PubMed] [Google Scholar]
- 83.Chen H, Guo X. Obesity and functional disability in elderly Americans. J Am Geriatr Soc 56: 689–694, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chen H, Simar D, Lambert K, Mercier J, Morris MJ. Maternal and postnatal overnutrition differentially impact appetite regulators and fuel metabolism. Endocrinology 149: 5348–5356, 2008. [DOI] [PubMed] [Google Scholar]
- 85.Chuang JC, Perello M, Sakata I, Osborne-Lawrence S, Savitt JM, Lutter M, Zigman JM. Ghrelin mediates stress-induced food-reward behavior in mice. J Clin Invest 121: 2684–2692, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Cianfarani S, Agostoni C, Bedogni G, Berni Canani R, Brambilla P, Nobili V, Pietrobelli A. Effect of intrauterine growth retardation on liver and long-term metabolic risk. Int J Obes 36: 1270–1277, 2012. [DOI] [PubMed] [Google Scholar]
- 87.Ciofi P. The arcuate nucleus as a circumventricular organ in the mouse. Neurosci Lett 487: 187–190, 2011. [DOI] [PubMed] [Google Scholar]
- 88.Clement K, Vaisse C, Lahlou N, Cabrol S, Pelloux V, Cassuto D, Gourmelen M, Dina C, Chambaz J, Lacorte JM, Basdevant A, Bougneres P, Lebouc Y, Froguel P, Guy-Grand B. A mutation in the human leptin receptor gene causes obesity and pituitary dysfunction. Nature 392: 398–401, 1998. [DOI] [PubMed] [Google Scholar]
- 89.Coen PM, Goodpaster BH. Role of intramyocelluar lipids in human health. Trends Endocrinol Metab 23: 391–398, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Cohen P, Zhao C, Cai X, Montez JM, Rohani SC, Feinstein P, Mombaerts P, Friedman JM. Selective deletion of leptin receptor in neurons leads to obesity. J Clin Invest 108: 1113–1121, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Colditz GA, Willett WC, Rotnitzky A, Manson JE. Weight gain as a risk factor for clinical diabetes mellitus in women. Ann Intern Med 122: 481–486, 1995. [DOI] [PubMed] [Google Scholar]
- 92.Coleman DL, Hummel KP. The influence of genetic background on the expression of the obese (Ob) gene in the mouse. Diabetologia 9: 287–293, 1973. [DOI] [PubMed] [Google Scholar]
- 93.Collins S, Martin TL, Surwit RS, Robidoux J. Genetic vulnerability to diet-induced obesity in the C57BL/6J mouse: physiological and molecular characteristics. Physiol Behav 81: 243–248, 2004. [DOI] [PubMed] [Google Scholar]
- 94.Cone RD. Anatomy and regulation of the central melanocortin system. Nature Neurosci 8: 571–578, 2005. [DOI] [PubMed] [Google Scholar]
- 95.Connor KL, Vickers MH, Beltrand J, Meaney MJ, Sloboda DM. Nature, nurture or nutrition? Impact of maternal nutrition on maternal care, offspring development and reproductive function. J Physiol 590: 2167–2180, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Contreras RJ, Beckstead RM, Norgren R. The central projections of the trigeminal, facial, glossopharyngeal and vagus nerves: an autoradiographic study in the rat. J Auton Nerv Syst 6: 303–322, 1982. [DOI] [PubMed] [Google Scholar]
- 97.Coupe B, Amarger V, Grit I, Benani A, Parnet P. Nutritional programming affects hypothalamic organization and early response to leptin. Endocrinology 151: 702–713, 2010. [DOI] [PubMed] [Google Scholar]
- 98.Coupe B, Bouret SG. Development of the hypothalamic melanocortin system. Front Endocrinol 4: 38, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Cowley MA, Cone RD, Enriori P, Louiselle I, Williams SM, Evans AE. Electrophysiological actions of peripheral hormones on melanocortin neurons. Ann NY Acad Sci 994: 175–186, 2003. [DOI] [PubMed] [Google Scholar]
- 100.Cowley MA, Smith RG, Diano S, Tschop M, Pronchuk N, Grove KL, Strasburger CJ, Bidlingmaier M, Esterman M, Heiman ML, Garcia-Segura LM, Nillni EA, Mendez P, Low MJ, Sotonyi P, Friedman JM, Liu H, Pinto S, Colmers WF, Cone RD, Horvath TL. The distribution and mechanism of action of ghrelin in the CNS demonstrates a novel hypothalamic circuit regulating energy homeostasis. Neuron 37: 649–661, 2003. [DOI] [PubMed] [Google Scholar]
- 101.Croizier S, Amiot C, Chen X, Presse F, Nahon JL, Wu JY, Fellmann D, Risold PY. Development of posterior hypothalamic neurons enlightens a switch in the prosencephalic basic plan. PloS One 6: e28574, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Crowther NJ, Cameron N, Trusler J, Gray IP. Association between poor glucose tolerance and rapid post natal weight gain in seven-year-old children. Diabetologia 41: 1163–1167, 1998. [DOI] [PubMed] [Google Scholar]
- 103.Crozier SR, Inskip HM, Godfrey KM, Cooper C, Harvey NC, Cole ZA, Robinson SM, Southampton Women's Survey Study G. Weight gain in pregnancy and childhood body composition: findings from the Southampton Women's Survey. Am J Clin Nutr 91: 1745–1751, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Crume TL, Ogden LG, Mayer-Davis EJ, Hamman RF, Norris JM, Bischoff KJ, McDuffie R, Dabelea D. The impact of neonatal breast-feeding on growth trajectories of youth exposed and unexposed to diabetes in utero: the EPOCH Study. Int J Obes 36: 529–534, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Cummings DE, Purnell JQ, Frayo RS, Schmidova K, Wisse BE, Weigle DS. A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 50: 1714–1719, 2001. [DOI] [PubMed] [Google Scholar]
- 106.Cummings DE, Weigle DS, Frayo RS, Breen PA, Ma MK, Dellinger EP, Purnell JQ. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med 346: 1623–1630, 2002. [DOI] [PubMed] [Google Scholar]
- 107.Czupryn A, Zhou YD, Chen X, McNay D, Anderson MP, Flier JS, Macklis JD. Transplanted hypothalamic neurons restore leptin signaling and ameliorate obesity in db/db mice. Science 334: 1133–1137, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Dabelea D. The predisposition to obesity and diabetes in offspring of diabetic mothers. Diabetes Care 30Suppl 2: S169–174, 2007. [DOI] [PubMed] [Google Scholar]
- 109.Dabelea D, Knowler WC, Pettitt DJ. Effect of diabetes in pregnancy on offspring: follow-up research in the Pima Indians. J Matern Fetal Med 9: 83–88, 2000. [DOI] [PubMed] [Google Scholar]
- 110.Dagogo-Jack S, Santiago JV. Pathophysiology of type 2 diabetes and modes of action of therapeutic interventions. Arch Intern Med 157: 1802–1817, 1997. [PubMed] [Google Scholar]
- 111.Dahlgren J, Nilsson C, Jennische E, Ho HP, Eriksson E, Niklasson A, Bjorntorp P, Albertsson WK, Holmang A. Prenatal cytokine exposure results in obesity and gender-specific programming. Am J Physiol Endocrinol Metab 281: E326–E334, 2001. [DOI] [PubMed] [Google Scholar]
- 112.Dai Z, Xu YC, Niu L. Obesity and colorectal cancer risk: a meta-analysis of cohort studies. World J Gastroenterol 13: 4199–4206, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Dakin CL, Small CJ, Batterham RL, Neary NM, Cohen MA, Patterson M, Ghatei MA, Bloom SR. Peripheral oxyntomodulin reduces food intake and body weight gain in rats. Endocrinology 145: 2687–2695, 2004. [DOI] [PubMed] [Google Scholar]
- 114.Danaei G, Ding EL, Mozaffarian D, Taylor B, Rehm J, Murray CJ, Ezzati M. The preventable causes of death in the United States: comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med 6: e1000058, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Date Y, Murakami N, Toshinai K, Matsukura S, Niijima A, Matsuo H, Kangawa K, Nakazato M. The role of the gastric afferent vagal nerve in ghrelin-induced feeding and growth hormone secretion in rats. Gastroenterology 123: 1120–1128, 2002. [DOI] [PubMed] [Google Scholar]
- 116.Date Y, Nakazato M, Hashiguchi S, Dezaki K, Mondal MS, Hosoda H, Kojima M, Kangawa K, Arima T, Matsuo H, Yada T, Matsukura S. Ghrelin is present in pancreatic alpha-cells of humans and rats and stimulates insulin secretion. Diabetes 51: 124–129, 2002. [DOI] [PubMed] [Google Scholar]
- 117.De Luca C, Kowalski TJ, Zhang Y, Elmquist JK, Lee C, Kilimann MW, Ludwig T, Liu SM, Chua SC Jr. Complete rescue of obesity, diabetes, and infertility in db/db mice by neuron-specific LEPR-B transgenes. J Clin Invest JCI24059, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Delahaye F, Breton C, Risold PY, Enache M, Dutriez-Casteloot I, Laborie C, Lesage J, Vieau D. Maternal perinatal undernutrition drastically reduces postnatal leptin surge and affects the development of arcuate nucleus POMC neurons in neonatal male rat pups. Endocrinology 149: 470–475, 2008. [DOI] [PubMed] [Google Scholar]
- 119.Dembinski A, Warzecha Z, Ceranowicz P, Bielanski W, Cieszkowski J, Dembinski M, Pawlik WW, Kuwahara A, Kato I, Konturek PC. Variable effect of ghrelin administration on pancreatic development in young rats. Role of insulin-like growth factor-1. J Physiol Pharmacol 56: 555–570, 2005. [PubMed] [Google Scholar]
- 120.Dewey KG. Growth characteristics of breast-fed compared to formula-fed infants. Biol Neonate 74: 94–105, 1998. [DOI] [PubMed] [Google Scholar]
- 121.Dhillon H, Zigman JM, Ye C, Lee CE, McGovern RA, Tang V, Kenny CD, Christiansen LM, White RD, Edelstein EA, Coppari R, Balthasar N, Cowley MA, Chua S Jr, Elmquist JK, Lowell BB. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 49: 191–203, 2006. [DOI] [PubMed] [Google Scholar]
- 122.Do R, Bailey SD, Desbiens K, Belisle A, Montpetit A, Bouchard C, Perusse L, Vohl MC, Engert JC. Genetic variants of FTO influence adiposity, insulin sensitivity, leptin levels, and resting metabolic rate in the Quebec Family Study. Diabetes 57: 1147–1150, 2008. [DOI] [PubMed] [Google Scholar]
- 123.Doche ME, Bochukova EG, Su HW, Pearce LR, Keogh JM, Henning E, Cline JM, Saeed S, Dale A, Cheetham T, Barroso I, Argetsinger LS, O'Rahilly S, Rui L, Carter-Su C, Farooqi IS. Human SH2B1 mutations are associated with maladaptive behaviors and obesity. J Clin Invest 122: 4732–4736, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Doehner W, Clark A, Anker SD. The obesity paradox: weighing the benefit. Eur Heart J 31: 146–148, 2010. [DOI] [PubMed] [Google Scholar]
- 125.Doehner W, Erdmann E, Cairns R, Clark AL, Dormandy JA, Ferrannini E, Anker SD. Inverse relation of body weight and weight change with mortality and morbidity in patients with type 2 diabetes and cardiovascular co-morbidity: an analysis of the PROactive study population. Int J Cardiol 162: 20–26, 2012. [DOI] [PubMed] [Google Scholar]
- 126.Dotta F, Fondelli C, Di Mario U. Type 1 diabetes mellitus as a polygenic multifactorial disease: immunopathogenic mechanisms of beta-cell destruction. Acta Biomed 76Suppl 3: 14–18, 2005. [PubMed] [Google Scholar]
- 127.Dube MG, Xu B, Kalra PS, Sninsky CA, Kalra SP. Disruption in neuropeptide Y and leptin signaling in obese ventromedial hypothalamic-lesioned rats. Brain Res 816: 38–46, 1999. [DOI] [PubMed] [Google Scholar]
- 128.Dunn-Meynell AA, Govek E, Levin BE. Intracarotid glucose infusions selectively increase Fos-like immunoreactivity in paraventricular, ventromedial and dorsomedial nuclei neurons. Brain Res 748: 100–106, 1997. [DOI] [PubMed] [Google Scholar]
- 129.Dunn-Meynell AA, Sanders NM, Compton D, Becker TC, Eiki J, Zhang BB, Levin BE. Relationship among brain and blood glucose levels and spontaneous and glucoprivic feeding. J Neurosci 29: 7015–7022, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Dunn GA, Bale TL. Maternal high-fat diet promotes body length increases and insulin insensitivity in second-generation mice. Endocrinology 150: 4999–5009, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Edmond J, Robbins RA, Bergstrom JD, Cole RA, de Vellis J. Capacity for substrate utilization in oxidative metabolism by neurons, astrocytes, and oligodendrocytes from developing brain in primary culture. J Neurosci Res 18: 551–561, 1987. [DOI] [PubMed] [Google Scholar]
- 132.Einstein F, Thompson RF, Bhagat TD, Fazzari MJ, Verma A, Barzilai N, Greally JM. Cytosine methylation dysregulation in neonates following intrauterine growth restriction. PloS One 5: e8887, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Eldar-Finkelman H, Schreyer SA, Shinohara MM, LeBoeuf RC, Krebs EG. Increased glycogen synthase kinase-3 activity in diabetes- and obesity-prone C57BL/6J mice. Diabetes 48: 1662–1666, 1999. [DOI] [PubMed] [Google Scholar]
- 134.Elias CF, Kelly JF, Lee CE, Ahima RS, Drucker DJ, Saper CB, Elmquist JK. Chemical characterization of leptin-activated neurons in the rat brain. J Comp Neurol 423: 261–281, 2000. [PubMed] [Google Scholar]
- 135.Elmquist JK, Bjorbaek C, Ahima RS, Flier JS, Saper CB. Distributions of leptin receptor mRNA isoforms in the rat brain. J Comp Neurol 395: 535–547, 1998. [PubMed] [Google Scholar]
- 136.Enriori PJ, Evans AE, Sinnayah P, Jobst EE, Tonelli-Lemos L, Billes SK, Glavas MM, Grayson BE, Perello M, Nillni EA, Grove KL, Cowley MA. Diet-induced obesity causes severe but reversible leptin resistance in arcuate melanocortin neurons. Cell Metab 5: 181–194, 2007. [DOI] [PubMed] [Google Scholar]
- 137.Entringer S, Buss C, Wadhwa PD. Prenatal stress and developmental programming of human health and disease risk: concepts and integration of empirical findings. Curr Opin Endocrinol Diabetes Obesity 17: 507–516, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Entringer S, Kumsta R, Hellhammer DH, Wadhwa PD, Wust S. Prenatal exposure to maternal psychosocial stress and HPA axis regulation in young adults. Horm Behav 55: 292–298, 2009. [DOI] [PubMed] [Google Scholar]
- 139.Entringer S, Wust S, Kumsta R, Layes IM, Nelson EL, Hellhammer DH, Wadhwa PD. Prenatal psychosocial stress exposure is associated with insulin resistance in young adults. Am J Obstet Gynecol 199: 491–497, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Epel EE, Moyer AE, Martin CD, Macary S, Cummings N, Rodin J, Rebuffe-Scrive M. Stress-induced cortisol, mood, and fat distribution in men. Obes Res 7: 9–15, 1999. [DOI] [PubMed] [Google Scholar]
- 141.Eriksson JG, Osmond C, Kajantie E, Forsen TJ, Barker DJ. Patterns of growth among children who later develop type 2 diabetes or its risk factors. Diabetologia 49: 2853–2858, 2006. [DOI] [PubMed] [Google Scholar]
- 142.Farooqi IS. Monogenic human obesity. Front Horm Res 36: 1–11, 2008. [DOI] [PubMed] [Google Scholar]
- 143.Farooqi IS, Jebb SA, Langmack G, Lawrence E, Cheetham CH, Prentice AM, Hughes IA, McCamish MA, O'Rahilly S. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N Engl J Med 341: 879–884, 1999. [DOI] [PubMed] [Google Scholar]
- 144.Feng B, Zhang T, Xu H. Human adipose dynamics and metabolic health. Ann NY Acad Sci 1281: 160–177, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Ferris M, Hogan SL, Chin H, Shoham DA, Gipson DS, Gibson K, Yilmaz S, Falk RJ, Jennette JC. Obesity, albuminuria, and urinalysis findings in US young adults from the Add Health Wave III study. Clin J Am Soc Nephrol 2: 1207–1214, 2007. [DOI] [PubMed] [Google Scholar]
- 146.Figlewicz DP, Benoit SC. Insulin, leptin, and food reward: update 2008. Am J Physiol Regul Integr Comp Physiol 296: R9–R19, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Figlewicz DP, Dorsa DM, Stein LJ, Baskin DG, Paquette T, Greenwood MRC, Woods SC, Porte D Jr. Brain and liver insulin binding is decreased in Zucker rats carrying the “fa” gene. Endocrinology 117: 1537–1543, 1985. [DOI] [PubMed] [Google Scholar]
- 148.Fioramonti X, Song Z, Vazirani RP, Beuve A, Routh VH. Hypothalamic nitric oxide in hypoglycemia detection and counterregulation: a two-edged sword. Antioxidants Redox Signaling 14: 505–517, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Flegal KM, Carroll MD, Ogden CL, Curtin LR. Prevalence and trends in obesity among US adults, 1999–2008. JAMA 303: 235–241, 2010. [DOI] [PubMed] [Google Scholar]
- 151.Flegal KM, Graubard BI, Williamson DF, Gail MH. Excess deaths associated with underweight, overweight, and obesity. JAMA 293: 1861–1867, 2005. [DOI] [PubMed] [Google Scholar]
- 152.Fontaine KR, Redden DT, Wang C, Westfall AO, Allison DB. Years of life lost due to obesity. JAMA 289: 187–193, 2003. [DOI] [PubMed] [Google Scholar]
- 153.Force USPST. Screening for obesity in adults: recommendatino and rationale. Ann Internal Med 139: 930–932, 2003. [DOI] [PubMed] [Google Scholar]
- 154.Forestell CA, Mennella JA. Early determinants of fruit and vegetable acceptance. Pediatrics 120: 1247–1254, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Forsdahl A. Are poor living conditions in childhood and adolescence and important risk factor for arteriosclerotic heart disease? Int J Rehab Res 2: 238–239, 1979. [DOI] [PubMed] [Google Scholar]
- 156.Franke K, Harder T, Aerts L, Melchior K, Fahrenkrog S, Rodekamp E, Ziska T, Van Assche FA, Dudenhausen JW, Plagemann A. Programming' of orexigenic and anorexigenic hypothalamic neurons in offspring of treated and untreated diabetic mother rats. Brain Res 1031: 276–283, 2005. [DOI] [PubMed] [Google Scholar]
- 157.Franks PW, Looker HC, Kobes S, Touger L, Tataranni PA, Hanson RL, Knowler WC. Gestational glucose tolerance and risk of type 2 diabetes in young Pima Indian offspring. Diabetes 55: 460–465, 2006. [DOI] [PubMed] [Google Scholar]
- 158.Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, Lindgren CM, Perry JR, Elliott KS, Lango H, Rayner NW, Shields B, Harries LW, Barrett JC, Ellard S, Groves CJ, Knight B, Patch AM, Ness AR, Ebrahim S, Lawlor DA, Ring SM, Ben-Shlomo Y, Jarvelin MR, Sovio U, Bennett AJ, Melzer D, Ferrucci L, Loos RJ, Barroso I, Wareham NJ, Karpe F, Owen KR, Cardon LR, Walker M, Hitman GA, Palmer CN, Doney AS, Morris AD, Smith GD, Hattersley AT, McCarthy MI. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 316: 889–894, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Freinkel N. Banting Lecture 1980: of pregnancy and progeny. Diabetes 29: 1023–1035, 1980. [DOI] [PubMed] [Google Scholar]
- 160.Fride E, Dan Y, Feldon J, Halevy G, Weinstock M. Effects of prenatal stress on vulnerability to stress in prepubertal and adult rats. Physiol Behav 37: 681–687, 1986. [DOI] [PubMed] [Google Scholar]
- 161.Fujioka T, Sakata Y, Yamaguchi K, Shibasaki T, Kato H, Nakamura S. The effects of prenatal stress on the development of hypothalamic paraventricular neurons in fetal rats. Neuroscience 92: 1079–1088, 1999. [DOI] [PubMed] [Google Scholar]
- 162.Fullston T, Ohlsson Teague EM, Palmer NO, Deblasio MJ, Mitchell M, Corbett M, Print CG, Owens JA, Lane M. Paternal obesity initiates metabolic disturbances in two generations of mice with incomplete penetrance to the F2 generation and alters the transcriptional profile of testis and sperm microRNA content. FASEB J 27: 4226–4243, 2013. [DOI] [PubMed] [Google Scholar]
- 163.Fulton S, Pissios P, Manchon RP, Stiles L, Frank L, Pothos EN, Maratos-Flier E, Flier JS. Leptin regulation of the mesoaccumbens dopamine pathway. Neuron 51: 811–822, 2006. [DOI] [PubMed] [Google Scholar]
- 164.Gale CR, Javaid MK, Robinson SM, Law CM, Godfrey KM, Cooper C. Maternal size in pregnancy and body composition in children. J Clin Endocrinol Metab 92: 3904–3911, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Galic S, Oakhill JS, Steinberg GR. Adipose tissue as an endocrine organ. Mol Cell Endocrinol 316: 129–139, 2010. [DOI] [PubMed] [Google Scholar]
- 166.Gambling L, Dunford S, Wallace DI, Zuur G, Solanky N, Srai SK, McArdle HJ. Iron deficiency during pregnancy affects postnatal blood pressure in the rat. J Physiol 552: 603–610, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Gardner DS, Tingey K, Van Bon BWM, Ozanne SE, Wilson V, Dandrea J, Keisler DH, Stephenson T, Symonds ME. Programming of glucose-insulin metabolism in adult sheep after maternal undernutrition. Am J Physiol Regul Integr Comp Physiol 289: R947–R954, 2005. [DOI] [PubMed] [Google Scholar]
- 168.Gariani K, Philippe J, Jornayvaz FR. Non-alcoholic fatty liver disease and insulin resistance: from bench to bedside. Diabetes Metab 39: 16–26, 2013. [DOI] [PubMed] [Google Scholar]
- 169.Garofano A, Czernichow P, Breant B. Effect of ageing on beta-cell mass and function in rats malnourished during the perinatal period. Diabetologia 42: 711–718, 1999. [DOI] [PubMed] [Google Scholar]
- 170.Gibbs J, Falasco JD, McHugh PR. Cholecystokinin-decreased food intake in rhesus monkeys. Am J Physiol 230: 15–18, 1976. [DOI] [PubMed] [Google Scholar]
- 171.Gilbert ER, Liu D. Epigenetics: the missing link to understanding beta-cell dysfunction in the pathogenesis of type 2 diabetes. Epigenetics 7: 841–852, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Gilbert M, Magnan C, Turban S, Andre J, Guerre-Millo M. Leptin receptor-deficient obese Zucker rats reduce their food intake in response to a systemic supply of calories from glucose. Diabetes 52: 277–282, 2003. [DOI] [PubMed] [Google Scholar]
- 173.Gillman MW. Early infancy as a critical period for development of obesity and related conditions. Nestle Nutrition workshop series. Paediatric Programme 65: 13–20, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Glavas M, Kirigiti M, Xiao X, Enriori P, Fisher S, Evans A, Grayson B, Cowley M, Smith M, Grove K. Early overnutrition results in early-onset arcuate leptin resistance and increased sensitivity to high-fat diet. Endocrinology 151: 1598–1610, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Godfrey KM, Sheppard A, Gluckman PD, Lillycrop KA, Burdge GC, McLean C, Rodford J, Slater-Jefferies JL, Garratt E, Crozier SR, Emerald BS, Gale CR, Inskip HM, Cooper C, Hanson MA. Epigenetic gene promoter methylation at birth is associated with child's later adiposity. Diabetes 60: 1528–1534, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Gorski J, Dunn-Meynell AA, Hartman TG, Levin BE. Postnatal environment overrides genetic and prenatal factors influencing offspring obesity and insulin resistance. Am J Physiol Regul Integr Comp Physiol 291: R768–R778, 2006. [DOI] [PubMed] [Google Scholar]
- 177.Gorski J, Levin BE. Effects of cross fostering on body weight, adiposity and insulin sensitivity in selectively bred obesity-prone and resistant rats (Abstract). Obesity Res 12: A103, 2004. [Google Scholar]
- 178.Gorski JN, Dunn-Meynell AA, Levin BE. Maternal obesity increases hypothalamic leptin receptor expression and sensitivity in juvenile obesity-prone rats. Am J Physiol Regul Integr Comp Physiol 292: R1782–R1791, 2007. [DOI] [PubMed] [Google Scholar]
- 179.Gratacos M, Gonzalez JR, Mercader JM, de Cid R, Urretavizcaya M, Estivill X. Brain-derived neurotrophic factor Val66Met and psychiatric disorders: meta-analysis of case-control studies confirm association to substance-related disorders, eating disorders, and schizophrenia. Biol Psychiatry 61: 911–922, 2007. [DOI] [PubMed] [Google Scholar]
- 180.Grayson BE, Allen SE, Billes SK, Williams SM, Smith MS, Grove KL. Prenatal development of hypothalamic neuropeptide systems in the nonhuman primate. Neuroscience 143: 975–986, 2006. [DOI] [PubMed] [Google Scholar]
- 181.Grill HJ. Distributed neural control of energy balance: contributions from hindbrain and hypothalamus. Obesity 14Suppl 5: 216S–221S, 2006. [DOI] [PubMed] [Google Scholar]
- 182.Grill HJ, Hayes MR. Hindbrain neurons as an essential hub in the neuroanatomically distributed control of energy balance. Cell Metab 16: 296–309, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Grill HJ, Kaplan JM. Interoceptive and integrative contributions of forebrain and brainstem to energy balance control. Int J Obes Relat Metab Disorders 25Suppl 5: S73–77, 2001. [DOI] [PubMed] [Google Scholar]
- 184.Grill HJ, Kaplan JM. The neuroanatomical axis for control of energy balance. Front Neuroendocrinol 23: 2–40, 2002. [DOI] [PubMed] [Google Scholar]
- 185.Groom A, Potter C, Swan DC, Fatemifar G, Evans DM, Ring SM, Turcot V, Pearce MS, Embleton ND, Smith GD, Mathers JC, Relton CL. Postnatal growth and DNA methylation are associated with differential gene expression of the TACSTD2 gene and childhood fat mass. Diabetes 61: 391–400, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Grossman SP. The role of glucose, insulin and glucagon in the regulation of food intake and body weight. Neurosci Biobehav Rev 10: 295–315, 1986. [DOI] [PubMed] [Google Scholar]
- 187.Grove KL, Allen S, Grayson BE, Smith MS. Postnatal development of the hypothalamic neuropeptide Y system. Neuroscience 116: 393–406, 2003. [DOI] [PubMed] [Google Scholar]
- 188.Gu D, He J, Duan X, Reynolds K, Wu X, Chen J, Huang G, Chen CS, Whelton PK. Body weight and mortality among men and women in China. JAMA 295: 776–783, 2006. [DOI] [PubMed] [Google Scholar]
- 189.Guan XM, Yu H, Van der Ploeg LH. Evidence of altered hypothalamic pro-opiomelanocortin/neuropeptide Y mRNA expression in tubby mice. Brain Res 59: 273–279, 1998. [DOI] [PubMed] [Google Scholar]
- 190.Guenard F, Deshaies Y, Cianflone K, Kral JG, Marceau P, Vohl MC. Differential methylation in glucoregulatory genes of offspring born before vs. after maternal gastrointestinal bypass surgery. Proc Natl Acad Sci USA 110: 11439–11444, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Guh DP, Zhang W, Bansback N, Amarsi Z, Birmingham CL, Anis AH. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health 9: 88, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Guo F, Jen KL. High-fat feeding during pregnancy and lactation affects offspring metabolism in rats. Physiol Behav 57: 681–686, 1995. [DOI] [PubMed] [Google Scholar]
- 193.Gyengesi E, Liu Z, D'Agostino G, Gan G, Horvath T, Gao X, Diano S. Corticosterone regulates synaptic input organization of POMC and NPY/AgRP neurons in adult mice. Endocrinology 151: 5395–5402, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Hakansson ML, Brown H, Ghilardi N, Skoda RC, Meister B. Leptin receptor immunoreactivity in chemically defined target neurons of the hypothalamus. J Neurosci 18: 559–572, 1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Hakansson ML, Meister B. Transcription factor STAT3 in leptin target neurons of the rat hypothalamus. Neuroendocrinology 68: 420–427, 1998. [DOI] [PubMed] [Google Scholar]
- 196.Halaas JL, Boozer C, Blair-West J, Fidahusein N, Denton DA, Friedman JM. Physiological response to long-term peripheral and central leptin infusion in lean and obese mice. Proc Natl Acad Sci USA 94: 8878–8883, 1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Halaas JL, Gajiwala KS, Maffei M, Cohen SL, Rabinowitz D, Lallone RL, Burley SK, Friedman JM. Weight-reducing effects of plasma protein encoded by the obese gene. Science 269: 543–546, 1995. [DOI] [PubMed] [Google Scholar]
- 198.Hales CN, Barker DJ, Clark PM, Cox LJ, Fall C, Osmond C, Winter PD. Fetal and infant growth and impaired glucose tolerance at age 64. BMJ 303: 1019–1022, 1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Han J, Xu J, Long YS, Epstein PN, Liu YQ. Rat maternal diabetes impairs pancreatic beta-cell function in the offspring. Am J Physiol Endocrinol Metab 293: E228–E236, 2007. [DOI] [PubMed] [Google Scholar]
- 200.Harder T, Bergmann R, Kallischnigg G, Plagemann A. Duration of breastfeeding and risk of overweight: a meta-analysis. Am J Epidemiol 162: 397–403, 2005. [DOI] [PubMed] [Google Scholar]
- 201.Harriss DJ, Atkinson G, George K, Cable NT, Reilly T, Haboubi N, Zwahlen M, Egger M, Renehan AG, Group CC. Lifestyle factors and colorectal cancer risk (1): systematic review and meta-analysis of associations with body mass index. Colorectal Dis 11: 547–563, 2009. [DOI] [PubMed] [Google Scholar]
- 202.Hassing LB, Dahl AK, Thorvaldsson V, Berg S, Gatz M, Pedersen NL, Johansson B. Overweight in midlife and risk of dementia: a 40-year follow-up study. Int J Obes 33: 893–898, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Haupt A, Thamer C, Heni M, Machicao F, Machann J, Schick F, Stefan N, Fritsche A, Haring HU, Staiger H. Novel obesity risk loci do not determine distribution of body fat depots: a whole-body MRI/MRS study. Obesity 18: 1212–1217, 2010. [DOI] [PubMed] [Google Scholar]
- 204.Hayes MR, Skibicka KP, Leichner TM, Guarnieri DJ, DiLeone RJ, Bence KK, Grill HJ. Endogenous leptin signaling in the caudal nucleus tractus solitarius and area postrema is required for energy balance regulation. Cell Metab 11: 77–83, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Heard-Costa NL, Zillikens MC, Monda KL, Johansson A, Harris TB, Fu M, Haritunians T, Feitosa MF, Aspelund T, Eiriksdottir G, Garcia M, Launer LJ, Smith AV, Mitchell BD, McArdle PF, Shuldiner AR, Bielinski SJ, Boerwinkle E, Brancati F, Demerath EW, Pankow JS, Arnold AM, Chen YD, Glazer NL, McKnight B, Psaty BM, Rotter JI, Amin N, Campbell H, Gyllensten U, Pattaro C, Pramstaller PP, Rudan I, Struchalin M, Vitart V, Gao X, Kraja A, Province MA, Zhang Q, Atwood LD, Dupuis J, Hirschhorn JN, Jaquish CE, O'Donnell CJ, Vasan RS, White CC, Aulchenko YS, Estrada K, Hofman A, Rivadeneira F, Uitterlinden AG, Witteman JC, Oostra BA, Kaplan RC, Gudnason V, O'Connell JR, Borecki IB, van Duijn CM, Cupples LA, Fox CS, North KE. NRXN3 is a novel locus for waist circumference: a genome-wide association study from the CHARGE Consortium. PLoS Genet 5: e1000539, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Heidenreich KA, Toledo SP. Insulin receptors mediate growth effects in cultured fetal neurons. II. Activation of a protein kinase that phosphorylates ribosomal protein S6. Endocrinology 125: 1458–1463, 1989. [DOI] [PubMed] [Google Scholar]
- 207.Heijmans BT, Tobi EW, Stein AD, Putter H, Blauw GJ, Susser ES, Slagboom PE, Lumey LH. Persistent epigenetic differences associated with prenatal exposure to famine in humans. Proc Natl Acad Sci USA 105: 17046–17049, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Henry C, Kabbaj M, Simon H, Le Moal M, Maccari S. Prenatal stress increases the hypothalamo-pituitary-adrenal axis response in young and adult rats. J Neuroendocrinol 6: 341–345, 1994. [DOI] [PubMed] [Google Scholar]
- 209.Hetherington AW. Obesity in the rat following the injection of chronic acid into the hypophysis. Endocrinology 26: 264–268, 1940. [Google Scholar]
- 210.Hetherington AW, Ranson SW. Hypothalamic lesions and adiposity in the rat. Anat Rec 78: 149–172, 1940. [Google Scholar]
- 211.Hewson AK, Dickson SL. Systemic administration of ghrelin induces Fos and Egr-1 proteins in the hypothalamic arcuate nucleus of fasted and fed rats. J Neuroendocrinol 12: 1047–1049, 2000. [DOI] [PubMed] [Google Scholar]
- 212.Hillier TA, Pedula KL, Schmidt MM, Mullen JA, Charles MA, Pettitt DJ. Childhood obesity and metabolic imprinting: the ongoing effects of maternal hyperglycemia. Diabetes Care 30: 2287–2292, 2007. [DOI] [PubMed] [Google Scholar]
- 213.Hirsova D, Koldovsky O. On the question of the absorption of insulin from the gastrointestinal tract during postnatal development. Physiol Bohem 18: 281–284, 1969. [PubMed] [Google Scholar]
- 214.Hochner H, Friedlander Y, Calderon-Margalit R, Meiner V, Sagy Y, Avgil-Tsadok M, Burger A, Savitsky B, Siscovick DS, Manor O. Associations of maternal prepregnancy body mass index and gestational weight gain with adult offspring cardiometabolic risk factors: the Jerusalem Perinatal Family Follow-up Study. Circulation 125: 1381–1389, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Hoggard N, Hunter L, Duncan JS, Williams LM, Trayhurn P, Mercer JG. Leptin and leptin receptor mRNA and protein expression in the murine fetus and placenta. Proc Natl Acad Sci USA 94: 11073–11078, 1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Holzapfel C, Grallert H, Huth C, Wahl S, Fischer B, Doring A, Ruckert IM, Hinney A, Hebebrand J, Wichmann HE, Hauner H, Illig T, Heid IM. Genes and lifestyle factors in obesity: results from 12,462 subjects from MONICA/KORA. Int J Obes 34: 1538–1545, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Hommel JD, Trinko R, Sears RM, Georgescu D, Liu ZW, Gao XB, Thurmon JJ, Marinelli M, DiLeone RJ. Leptin receptor signaling in midbrain dopamine neurons regulates feeding. Neuron 51: 801–810, 2006. [DOI] [PubMed] [Google Scholar]
- 218.Horvath TL, Sarman B, Garcia-Caceres C, Enriori PJ, Sotonyi P, Shanabrough M, Borok E, Argente J, Chowen JA, Perez-Tilve D, Pfluger PT, Bronneke HS, Levin BE, Diano S, Cowley MA, Tschop MH. Synaptic input organization of the melanocortin system predicts diet-induced hypothalamic reactive gliosis and obesity. Proc Natl Acad Sci USA 107: 14875–14880, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Hotta K, Nakamura M, Nakamura T, Matsuo T, Nakata Y, Kamohara S, Miyatake N, Kotani K, Komatsu R, Itoh N, Mineo I, Wada J, Masuzaki H, Yoneda M, Nakajima A, Funahashi T, Miyazaki S, Tokunaga K, Kawamoto M, Ueno T, Hamaguchi K, Tanaka K, Yamada K, Hanafusa T, Oikawa S, Yoshimatsu H, Nakao K, Sakata T, Matsuzawa Y, Kamatani N, Nakamura Y. Association between obesity and polymorphisms in SEC16B, TMEM18, GNPDA2, BDNF, FAIM2 and MC4R in a Japanese population. J Hum Genet 54: 727–731, 2009. [DOI] [PubMed] [Google Scholar]
- 220.Hummel KP, Dickie MM, Coleman DL. Diabetes, a new mutation in the mouse. Science 153: 1127–1128, 1966. [DOI] [PubMed] [Google Scholar]
- 221.Ibrahim N, Smart JL, Rubenstein M, Low MJ, Kelly MJ. Mouse hypothalamic POMC neurons are modulated by KATP channel activity. Abstr Soc Neurosci 31: 733–711, 2001. [Google Scholar]
- 222.Imagawa A, Hanafusa T, Tamura S, Moriwaki M, Itoh N, Yamamoto K, Iwahashi H, Yamagata K, Waguri M, Nanmo T, Uno S, Nakajima H, Namba M, Kawata S, Miyagawa JI, Matsuzawa Y. Pancreatic biopsy as a procedure for detecting in situ autoimmune phenomena in type 1 diabetes: close correlation between serological markers and histological evidence of cellular autoimmunity. Diabetes 50: 1269–1273, 2001. [DOI] [PubMed] [Google Scholar]
- 223.Ingalls AM, Dickie MM, Snell GD. Obese, a new mutation in the house mouse. J Hered 41: 317–318, 1950. [DOI] [PubMed] [Google Scholar]
- 224.Inoue Y, Nakahara K, Kangawa K, Murakami N. Transitional change in rat fetal cell proliferation in response to ghrelin and des-acyl ghrelin during the last stage of pregnancy. Biochem Biophys Res Commun 393: 455–460, 2010. [DOI] [PubMed] [Google Scholar]
- 225.Irani B, Le Foll C, Dunn-Meynell AA, Levin BE. Effects of leptin on rat ventromedial hypothalamic neurons. Endocrinology 149: 5145–5154, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Irani BG, Dunn-Meynell AA, Levin BE. Altered hypothalamic leptin, insulin and melanocortin binding associated with moderate fat diet and predisposition to obesity. Endocrinology 148: 310–316, 2007. [DOI] [PubMed] [Google Scholar]
- 227.Ishii Y, Bouret SG. Embryonic birthdate of hypothalamic leptin-activated neurons in mice. Endocrinology 153: 3657–3667, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Jackson RS, Creemers JW, Ohagi S, Raffin-Sanson ML, Sanders L, Montague CT, Hutton JC, O'Rahilly S. Obesity and impaired prohormone processing associated with mutations in the human prohormone convertase 1 gene. Nature Genet 16: 303–306, 1997. [DOI] [PubMed] [Google Scholar]
- 229.Jee SH, Sull JW, Park J, Lee SY, Ohrr H, Guallar E, Samet JM. Body-mass index and mortality in Korean men and women. N Engl J Med 355: 779–787, 2006. [DOI] [PubMed] [Google Scholar]
- 230.Jo YH, Su Y, Gutierrez-Juarez R, Chua S Jr. Oleic acid directly regulates POMC neuron excitability in the hypothalamus. J Neurophysiol 101: 2305–2316, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Johnson PR, Stern JS, Greenwood MR, Zucker LM, Hirsch J. Effect of early nutrition on adipose cellularity and pancreatic insulin release in the Zucker rat. J Nutr 103: 738–743, 1973. [DOI] [PubMed] [Google Scholar]
- 232.Jones AP, Olster DH, States B. Maternal insulin manipulations in rats organize body weight and noradrenergic innervation of the hypothalamus in gonadally intact male offspring. Dev Brain Res 97: 16–21, 1996. [DOI] [PubMed] [Google Scholar]
- 233.Jou MY, Philipps AF, Lonnerdal B. Maternal zinc deficiency in rats affects growth and glucose metabolism in the offspring by inducing insulin resistance postnatally. J Nutr 140: 1621–1627, 2010. [DOI] [PubMed] [Google Scholar]
- 234.Jovanovic-Peterson L, Fuhrmann K, Hedden K, Walker L, Peterson CM. Maternal milk and plasma glucose and insulin levels: studies in normal and diabetic subjects. J Am Coll Nutr 8: 125–131, 1989. [DOI] [PubMed] [Google Scholar]
- 235.Kainer F, Weiss PA, Huttner U, Haas J, Reles M. Levels of amniotic fluid insulin and profiles of maternal blood glucose in pregnant women with diabetes type-I. Early Hum Dev 49: 97–105, 1997. [DOI] [PubMed] [Google Scholar]
- 236.Kang L, Routh VH, Kuzhikandathil EV, Gaspers L, Levin BE. Physiological and molecular characteristics of rat hypothalamic ventromedial nucleus glucosensing neurons. Diabetes 53: 549–559, 2004. [DOI] [PubMed] [Google Scholar]
- 237.Kapoor A, Dunn E, Kostaki A, Andrews MH, Matthews SG. Fetal programming of hypothalamo-pituitary-adrenal function: prenatal stress and glucocorticoids. J Physiol 572: 31–44, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Kar S, Chabot JG, Quirion R. Quantitative autoradiographic localization of [125I]insulin-like growth factor I, [125I]insulin-like growth factor II, and [125I]insulin receptor binding sites in developing and adult rat brain. J Comp Neurol 333: 375–397, 1993. [DOI] [PubMed] [Google Scholar]
- 239.Karnani MM, Venner A, Jensen LT, Fugger L, Burdakov D. Direct and indirect control of orexin/hypocretin neurons by glycine receptors. J Physiol 589: 639–651, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Keesey RE, Boyle PC, Storlien LH. Food intake and utilization in lateral hypothalamically lesioned rats. Physiol Behav 21: 265–268, 1978. [DOI] [PubMed] [Google Scholar]
- 241.Keesey RE, Corbett SW. Metabolic defense of the body weight set-point. In: Eating and Its Disorders, edited by Stunkard AJ, Stellar E. New York: Raven, 1984, p. 87–96. [PubMed] [Google Scholar]
- 242.Keesey RE, Mitchell JS, Kemnitz JW. Body weight and body composition of male rats following hypothalamic lesions. Am J Physiol Regul Integr Comp Physiol 237: R68–R73, 1979. [DOI] [PubMed] [Google Scholar]
- 243.Keesey RE, Powley TL. Body energy homeostasis. Appetite 51: 442–445, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Keith SW, Fontaine KR, Pajewski NM, Mehta T, Allison DB. Use of self-reported height and weight biases the body mass index-mortality association. Int J Obes 35: 401–408, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Kenchaiah S, Evans JC, Levy D, Wilson PW, Benjamin EJ, Larson MG, Kannel WB, Vasan RS. Obesity and the risk of heart failure. N Engl J Med 347: 305–313, 2002. [DOI] [PubMed] [Google Scholar]
- 246.Kennedy GC. The development with age of hypothalamic restraint upon the appetite of the rat. J Endocrinol 16: 9–17, 1957. [DOI] [PubMed] [Google Scholar]
- 247.Kennedy GC. The role of depot fat in the hypothalamic control of food intake in the rat. Proc R Soc Lond B Biol Sci 140: 578–592, 1953. [DOI] [PubMed] [Google Scholar]
- 248.Kermack WO, McKendrick AG, McKinlay PL. Death-rates in Great Britain and Sweden: expression of specific mortality rates as products of two factors, and some consequences thereof. J Hygiene 34: 433–457, 1934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.King BM. The rise, fall, and resurrection of the ventromedial hypothalamus in the regulation of feeding behavior and body weight. Physiol Behav 87: 221–244, 2006. [DOI] [PubMed] [Google Scholar]
- 250.Kirk S, Samuelsson A, Argenton M, Dhonye H, Kalamatianos T, Poston L, Taylor P, Coen C. Maternal obesity induced by diet in rats permanently influences central processes regulating food intake in offspring. PloS One 4: e5870, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Kirk SL, Samuelsson AM, Argenton M, Dhonye H, Kalamatianos T, Poston L, Taylor PD, Coen CW. Maternal obesity induced by diet in rats permanently influences central processes regulating food intake in offspring. PloS One 4: e5870, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Knittle JL, Hirsch J. Effect of early nutrition on the development of rat epididymal fat pads: cellularity and metabolism. J Clin Invest 47: 2091–2098, 1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Koda S, Date Y, Murakami N, Shimbara T, Hanada T, Toshinai K, Niijima A, Furuya M, Inomata N, Osuye K, Nakazato M. The role of the vagal nerve in peripheral PYY3–36-induced feeding reduction in rats. Endocrinology 146: 2369–2375, 2005. [DOI] [PubMed] [Google Scholar]
- 254.Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402: 656–660, 1999. [DOI] [PubMed] [Google Scholar]
- 255.Kokoeva MV, Yin H, Flier JS. Evidence for constitutive neural cell proliferation in the adult murine hypothalamus. J Comp Neurol 505: 209–220, 2007. [DOI] [PubMed] [Google Scholar]
- 256.Kokoeva MV, Yin H, Flier JS. Neurogenesis in the hypothalamus of adult mice: potential role in energy balance. Science 310: 679–683, 2005. [DOI] [PubMed] [Google Scholar]
- 257.Koletzko B, von Kries R, Closa R, Escribano J, Scaglioni S, Giovannini M, Beyer J, Demmelmair H, Gruszfeld D, Dobrzanska A, Sengier A, Langhendries JP, Rolland Cachera MF, Grote V, European Childhood Obesity Trial Study. Lower protein in infant formula is associated with lower weight up to age 2 y: a randomized clinical trial. Am J Clin Nutr 89: 1836–1845, 2009. [DOI] [PubMed] [Google Scholar]
- 258.Kong D, Tong Q, Ye C, Koda S, Fuller PM, Krashes MJ, Vong L, Ray RS, Olson DP, Lowell BB. GABAergic RIP-Cre neurons in the arcuate nucleus selectively regulate energy expenditure. Cell 151: 645–657, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Kopin AS, Mathes WF, McBride EW, Nguyen M, Al-Haider W, Schmitz F, Bonner-Weir S, Kanarek R, Beinborn M. The cholecystokinin-A receptor mediates inhibition of food intake yet is not essential for the maintenance of body weight. J Clin Invest 103: 383–391, 1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Kotz CM, Teske JA, Billington CJ. Neuroregulation of nonexercise activity thermogenesis and obesity resistance. Am J Physiol Regul Integr Comp Physiol 294: R699–R710, 2008. [DOI] [PubMed] [Google Scholar]
- 261.Koupil I, Toivanen P. Social and early-life determinants of overweight and obesity in 18-year-old Swedish men. Int J Obes 32: 73–81, 2008. [DOI] [PubMed] [Google Scholar]
- 262.Koutcherov Y, Mai JK, Ashwell KW, Paxinos G. Organization of human hypothalamus in fetal development. J Comp Neurol 446: 301–324, 2002. [DOI] [PubMed] [Google Scholar]
- 263.Koyama K, Shimabukuro M, Chen G, Wang MY, Lee Y, Kalra PS, Dube MG, Kalra SP, Newgard CB, Unger RH. Resistance to adenovirally induced hyperleptinemia in rats. Comparison of ventromedial hypothalamic lesions and mutated leptin receptors. J Clin Invest 102: 728–733, 1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Krakoff J, Ma L, Kobes S, Knowler WC, Hanson RL, Bogardus C, Baier LJ. Lower metabolic rate in individuals heterozygous for either a frameshift or a functional missense MC4R variant. Diabetes 57: 3267–3272, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Kral JG, Biron S, Simard S, Hould FS, Lebel S, Marceau S, Marceau P. Large maternal weight loss from obesity surgery prevents transmission of obesity to children who were followed for 2 to 18 years. Pediatrics 118: e1644–1649, 2006. [DOI] [PubMed] [Google Scholar]
- 266.Kramer MS. Do breast-feeding and delayed introduction of solid foods protect against subsequent obesity? J Pediatr 98: 883–887, 1981. [DOI] [PubMed] [Google Scholar]
- 267.Krebs NF, Himes JH, Jacobson D, Nicklas TA, Guilday P, Styne D. Assessment of child and adolescent overweight and obesity. Pediatrics 120Suppl 4: S193–228, 2007. [DOI] [PubMed] [Google Scholar]
- 268.Kreymann B, Williams G, Ghatei MA, Bloom SR. Glucagon-like peptide-1 7–36: a physiological incretin in man. Lancet 2: 1300–1304, 1987. [DOI] [PubMed] [Google Scholar]
- 269.Kring SI, Holst C, Toubro S, Astrup A, Hansen T, Pedersen O, Sorensen TI. Common variants near MC4R in relation to body fat, body fat distribution, metabolic traits and energy expenditure. Int J Obes 34: 182–189, 2010. [DOI] [PubMed] [Google Scholar]
- 270.Krude H, Biebermann H, Luck W, Horn R, Brabant G, Gruters A. Severe early-onset obesity, adrenal insufficiency and red hair pigmentation caused by POMC mutations in humans. Nature Genet 19: 155–157, 1998. [DOI] [PubMed] [Google Scholar]
- 271.Kulkarni RN, Almind K, Goren HJ, Winnay JN, Ueki K, Okada T, Kahn CR. Impact of genetic background on development of hyperinsulinemia and diabetes in insulin receptor/insulin receptor substrate-1 double heterozygous mice. Diabetes 52: 1528–1534, 2003. [DOI] [PubMed] [Google Scholar]
- 272.Laitinen K, Collado MC, Isolauri E. Early nutritional environment: focus on health effects of microbiota and probiotics. Beneficial Microbes 1: 383–390, 2010. [DOI] [PubMed] [Google Scholar]
- 273.Langlet F, Levin BE, Luquet S, Mazzone M, Messina A, Dunn-meynell AA, Balland E, Lacombe A, Mazur D, Carmeliet P, Bouret SG, Prevot V, Dehouck B. Tanycyte VEGF-A boosts blood-hypothalamic barrier plasticity and acces of metabolic signals to the arcuate nucleus in response to fasting. Cell Metab 17: 607–617, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Larsen LH, Echwald SM, Sorensen TI, Andersen T, Wulff BS, Pedersen O. Prevalence of mutations and functional analyses of melanocortin 4 receptor variants identified among 750 men with juvenile-onset obesity. J Clin Endocrinol Metab 90: 219–224, 2005. [DOI] [PubMed] [Google Scholar]
- 275.Lawlor DA, Smith GD, O'Callaghan M, Alati R, Mamun AA, Williams GM, Najman JM. Epidemiologic evidence for the fetal overnutrition hypothesis: findings from the mater-university study of pregnancy and its outcomes. Am J Epidemiol 165: 418–424, 2007. [DOI] [PubMed] [Google Scholar]
- 276.Lawrence CB, Snape AC, Baudoin FM, Luckman SM. Acute central ghrelin and GH secretogogues induce feeding and activate brain appetite centers. Endocrinology 143: 155–162, 2002. [DOI] [PubMed] [Google Scholar]
- 277.Laye MJ, Rector RS, Warner SO, Naples SP, Perretta AL, Uptergrove GM, Laughlin MH, Thyfault JP, Booth FW, Ibdah JA. Changes in visceral adipose tissue mitochondrial content with type 2 diabetes and daily voluntary wheel running in OLETF rats. J Physiol 587: 3729–3739, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Le Foll C, Dunn-Meynell A, Musatov S, Magnan C, Levin BE. FAT/CD36: A major regulator of neuronal fatty acid sensing and energy homeostasis in rats and mice. Diabetes 62: 2709–2716, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Le Foll C, Dunn-Meynell AA, Miziorko HM, Levin BE. Regulation of hypothalamic neuronal sensing and food intake by ketone bodies and fatty acids. Diabetes. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Le Foll C, Irani BG, Magnan C, Dunn-Meynell AA, Levin BE. Characteristics and mechanisms of hypothalamic neuronal fatty acid sensing. Am J Physiol Regul Integr Comp Physiol 297: R655–R664, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Le Foll C, Irani BG, Magnan C, Dunn-Meynell AA, Levin BE. Effects of maternal genotype and diet on offspring glucose and fatty acid sensing ventromedial hypothalamic nucleus neurons. Am J Physiol Regul Integr Comp Physiol 297: R1351–R1357, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Lee DA, Bedont JL, Pak T, Wang H, Song J, Miranda-Angulo A, Takiar V, Charubhumi V, Balordi F, Takebayashi H, Aja S, Ford E, Fishell G, Blackshaw S. Tanycytes of the hypothalamic median eminence form a diet-responsive neurogenic niche. Nature Neurosci 15: 700–702, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Lee GH, Proenca R, Montez JM, Carroll KM, Darvishzadeh JG, Lee JI, Friedman JM. Abnormal splicing of the leptin receptor in diabetic mice. Nature 379: 632–635, 1996. [DOI] [PubMed] [Google Scholar]
- 284.Leinninger GM, Opland DM, Jo YH, Faouzi M, Christensen L, Cappellucci LA, Rhodes CJ, Gnegy ME, Becker JB, Pothos EN, Seasholtz AF, Thompson RC, Myers MG Jr. Leptin action via neurotensin neurons controls orexin, the mesolimbic dopamine system and energy balance. Cell Metab 14: 313–323, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Lenard L, Karadi Z, Faludi B, Czurko A, Niedetzky C, Vida I, Nishino H. Glucose-sensitive neurons of the globus pallidus. I. Neurochemical characteristics. Br Res Bull 37: 149–155, 1995. [DOI] [PubMed] [Google Scholar]
- 286.Lenz M, Richter T, Muhlhauser I. The morbidity and mortality associated with overweight and obesity in adulthood: a systematic review. Deutsches Arzteblatt Int 106: 641–648, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Levin BE. Interaction of perinatal and pre-pubertal factors with genetic predisposition in the development of neural pathways involved in the regulation of energy homeostasis. Brain Res 1350: 10–17, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Levin BE. Metabolic imprinting on genetically predisposed neural circuits perpetuates obesity. Nutrition 16: 909–915, 2000. [DOI] [PubMed] [Google Scholar]
- 289.Levin BE. Metabolic sensing neurons and the control of energy homeostasis. Physiol Behav 89: 486–489, 2006. [DOI] [PubMed] [Google Scholar]
- 290.Levin BE, Becker TC, Eiki J, Zhang BB, Dunn-Meynell AA. Ventromedial hypothalamic glucokinase is an important mediator of the counterregulatory response to insulin-induced hypoglycemia. Diabetes 57: 1371–1379, 2008. [DOI] [PubMed] [Google Scholar]
- 291.Levin BE, Dunn-Meynell AA. Defense of body weight against chronic caloric restriction in obesity-prone and -resistant rats. Am J Physiol Regul Integr Comp Physiol 278: R231–R237, 2000. [DOI] [PubMed] [Google Scholar]
- 292.Levin BE, Dunn-Meynell AA. Defense of body weight depends on dietary composition and palatability in rats with diet-induced obesity. Am J Physiol Regul Integr Comp Physiol 282: R46–R54, 2002. [DOI] [PubMed] [Google Scholar]
- 293.Levin BE, Dunn-Meynell AA. Differential effects of exercise on body weight gain and adiposity in obesity-prone and -resistant rats. Int J Obes 30: 722–727, 2006. [DOI] [PubMed] [Google Scholar]
- 294.Levin BE, Dunn-Meynell AA. Maternal obesity alters adiposity and monoamine function in genetically predisposed offspring. Am J Physiol Regul Integr Comp Physiol 283: R1087–R1093, 2002. [DOI] [PubMed] [Google Scholar]
- 295.Levin BE, Dunn-Meynell AA. Reduced central leptin sensitivity in rats with diet-induced obesity. Am J Physiol Regul Integr Comp Physiol 283: R941–R948, 2002. [DOI] [PubMed] [Google Scholar]
- 296.Levin BE, Dunn-Meynell AA, Balkan B, Keesey RE. Selective breeding for diet-induced obesity and resistance in Sprague-Dawley rats. Am J Physiol Regul Integr Comp Physiol 273: R725–R730, 1997. [DOI] [PubMed] [Google Scholar]
- 297.Levin BE, Dunn-Meynell AA, Banks WA. Obesity-prone rats have normal blood-brain barrier transport but defective central leptin signaling prior to obesity onset. Am J Physiol Regul Integr Comp Physiol 286: R143–R150, 2004. [DOI] [PubMed] [Google Scholar]
- 298.Levin BE, Dunn-Meynell AA, McMinn JE, Alperovich M, Cunningham-Bussel A, Chua SC Jr. A new obesity-prone, glucose-intolerant rat strain (FDIO). Am J Physiol Regul Integr Comp Physiol 285: R1184–R1191, 2003. [DOI] [PubMed] [Google Scholar]
- 299.Levin BE, Dunn-Meynell AA, Ricci MR, Cummings DE. Abnormalities of leptin and ghrelin regulation in obesity-prone juvenile rats. Am J Physiol Endocrinol Metab 285: E949–E957, 2003. [DOI] [PubMed] [Google Scholar]
- 300.Levin BE, Govek E. Gestational obesity accentuates obesity in obesity-prone progeny. Am J Physiol Regul Integr Comp Physiol 275: R1374–R1379, 1998. [DOI] [PubMed] [Google Scholar]
- 301.Levin BE, Kang L, Sanders NM, Dunn-Meynell AA. Role of neuronal glucosensing in the regulation of energy homeostasis. Diabetes 55Suppl 2: S122–S130, 2006. [Google Scholar]
- 302.Levin BE, Keesey RE. Defense of differing body weight set-points in diet-induced obese and resistant rats. Am J Physiol Regul Integr Comp Physiol 274: R412–R419, 1998. [DOI] [PubMed] [Google Scholar]
- 303.Levin BE, Magnan C, Dunn-Meynell A, Le Foll C. Metabolic sensing and the brain: who, what, where, and how? Endocrinology 152: 2552–2557, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Levin BE, Routh VH, Dunn-Meynell AA. Glucosensing neurons in the central nervous system. In: Neural and Metabolic Control of Macronutrient Intake, edited by Berthoud H-R, Seeley RJ. New York: CRC, 1999, p. 325–337. [Google Scholar]
- 305.Levin BE, Routh VH, Kang L, Sanders NM, Dunn-Meynell AA. Neuronal glucosensing: what do we know after 50 years? Diabetes 53: 2521–2528, 2004. [DOI] [PubMed] [Google Scholar]
- 306.Levin BE, Sherwin RS. Peripheral glucose homeostasis: does brain insulin matter? J Clin Invest 121: 3392–3395, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Ley RE, Backhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI. Obesity alters gut microbial ecology. Proc Natl Acad Sci USA 102: 11070–11075, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature 444: 1022–1023, 2006. [DOI] [PubMed] [Google Scholar]
- 309.Li Y, He Y, Qi L, Jaddoe VW, Feskens EJ, Yang X, Ma G, Hu FB. Exposure to the Chinese famine in early life and the risk of hyperglycemia and type 2 diabetes in adulthood. Diabetes 59: 2400–2406, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Liddle RA. Regulation of cholecystokinin secretion by intraluminal releasing factors. Am J Physiol Gastrointest Liver Physiol 269: G319–G327, 1995. [DOI] [PubMed] [Google Scholar]
- 311.Lillycrop KA, Phillips ES, Jackson AA, Hanson MA, Burdge GC. Dietary protein restriction of pregnant rats induces and folic acid supplementation prevents epigenetic modification of hepatic gene expression in the offspring. J Nutr 135: 1382–1386, 2005. [DOI] [PubMed] [Google Scholar]
- 312.Lopes-Cardozo M, Larsson OM, Schousboe A. Acetoacetate and glucose as lipid precursors and energy substrates in primary cultures of astrocytes and neurons from mouse cerebral cortex. J Neurochem 46: 773–778, 1986. [DOI] [PubMed] [Google Scholar]
- 313.Louis-Sylvestre J, Le Magnen J. Fall in blood glucose level precedes meal onset in free-feeding rats. Neurosci Biobehav Rev 4: 13–15, 1980. [DOI] [PubMed] [Google Scholar]
- 314.Low S, Chin MC, Ma S, Heng D, Deurenberg-Yap M. Rationale for redefining obesity in Asians Ann Acad Med Singapore 38: 66–69, 2009. [PubMed] [Google Scholar]
- 315.Luoto R, Kalliomaki M, Laitinen K, Isolauri E. The impact of perinatal probiotic intervention on the development of overweight and obesity: follow-up study from birth to 10 years. Int J Obes 34: 1531–1537, 2010. [DOI] [PubMed] [Google Scholar]
- 316.Marin P, Darin N, Amemiya T, Andersson B, Jern S, Bjorntorp P. Cortisol secretion in relation to body fat distribution in obese premenopausal women. Metab Clin Exp 41: 882–886, 1992. [DOI] [PubMed] [Google Scholar]
- 317.Markakis EA. Development of the neuroendocrine hypothalamus. Front Neuroendocrinol 23: 257–291, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Martin FI, Heath P, Mountain KR. Pregnancy in women with diabetes mellitus. Fifteen years' experience: 1970–1985. Medical J Australia 146: 187–190, 1987. [PubMed] [Google Scholar]
- 319.Matsumoto A, Arai Y. Developmental changes in synaptic formation in the hypothalamic arcuate nucleus of female rats. Cell Tissue Res 169: 143–156, 1976. [DOI] [PubMed] [Google Scholar]
- 320.Matsuo T, Sairenchi T, Iso H, Irie F, Tanaka K, Fukasawa N, Ota H, Muto T. Age- and gender-specific BMI in terms of the lowest mortality in Japanese general population. Obesity 16: 2348–2355, 2008. [DOI] [PubMed] [Google Scholar]
- 321.Maures TJ, Kurzer JH, Carter-Su C. SH2B1 (SH2-B) and JAK2: a multifunctional adaptor protein and kinase made for each other. Trends Endocrinol Metab 18: 38–45, 2007. [DOI] [PubMed] [Google Scholar]
- 322.Mayer J. Glucostatic mechanism of regulation of food intake. N Engl J Med 249: 13–16, 1953. [DOI] [PubMed] [Google Scholar]
- 323.McArthur S, McHale E, Dalley JW, Buckingham JC, Gillies GE. Altered mesencephalic dopaminergic populations in adulthood as a consequence of brief perinatal glucocorticoid exposure. J Neuroendocrinol 17: 475–482, 2005. [DOI] [PubMed] [Google Scholar]
- 324.McCance DR, Pettitt DJ, Hanson RL, Jacobsson LT, Knowler WC, Bennett PH. Birth weight and non-insulin dependent diabetes: thrifty genotype, thrifty phenotype, or surviving small baby genotype? BMJ 308: 942–945, 1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.McCurdy CE, Bishop JM, Williams SM, Grayson BE, Smith MS, Friedman JE, Grove KL. Maternal high-fat diet triggers lipotoxicity in the fetal livers of nonhuman primates. J Clin Invest 119: 323–335, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.McGuire MT, Wing RR, Klem ML, Seagle HM, Hill JO. Long-term maintenance of weight loss: do people who lose weight through various weight loss methods use different behaviors to maintain their weight? Int J Obes Relat Metab Disorders 22: 572–577, 1998. [DOI] [PubMed] [Google Scholar]
- 327.McNay DE, Briancon N, Kokoeva MV, Maratos-Flier E, Flier JS. Remodeling of the arcuate nucleus energy-balance circuit is inhibited in obese mice. J Clin Invest 122: 142–152, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Melnick I, Pronchuk N, Cowley MA, Grove KL, Colmers WF. Developmental switch in neuropeptide Y and melanocortin effects in the paraventricular nucleus of the hypothalamus. Neuron 56: 1103–1115, 2007. [DOI] [PubMed] [Google Scholar]
- 329.Mennella JA, Beauchamp GK. The effects of repeated exposure to garlic-flavored milk on the nursling's behavior. Pediatr Res 34: 805–808, 1993. [DOI] [PubMed] [Google Scholar]
- 330.Mennella JA, Castor SM. Sensitive period in flavor learning: effects of duration of exposure to formula flavors on food likes during infancy. Clin Nutr 31: 1022–1025, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Mennella JA, Jagnow CP, Beauchamp GK. Prenatal and postnatal flavor learning by human infants. Pediatrics 107: E88, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Menon RK, Cohen RM, Sperling MA, Cutfield WS, Mimouni F, Khoury JC. Transplacental passage of insulin in pregnant women with insulin-dependent diabetes mellitus. Its role in fetal macrosomia. N Engl J Med 323: 309–315, 1990. [DOI] [PubMed] [Google Scholar]
- 333.Mercer JG, Hoggard N, Williams LM, Lawrence CB, Hannah LT, Morgan PJ, Trayhurn P. Coexpression of leptin receptor and preproneuropeptide Y mRNA in arcuate nucleus of mouse hypothalamus. J Neuroendocrinol 8: 733–735, 1996. [DOI] [PubMed] [Google Scholar]
- 334.Mercer JG, Hoggard N, Williams LM, Lawrence CB, Hannah LT, Trayhurn P. Localization of leptin receptor mRNA and the long form splice variant (Ob-Rb) in mouse hypothalamus and adjacent brain regions by in situ hybridization. FEBS Lett 387: 113–116, 1996. [DOI] [PubMed] [Google Scholar]
- 335.Mercer JG, Moar KM, Hoggard N. Localization of leptin receptor (Ob-R) messenger ribonucleic acid in the rodent hindbrain. Endocrinology 139: 29–34, 1998. [DOI] [PubMed] [Google Scholar]
- 336.Merezak S, Reusens B, Renard A, Goosse K, Kalbe L, Ahn MT, Tamarit-Rodriguez J, Remacle C. Effect of maternal low-protein diet and taurine on the vulnerability of adult Wistar rat islets to cytokines. Diabetologia 47: 669–675, 2004. [DOI] [PubMed] [Google Scholar]
- 337.Migrenne S, Cruciani-Guglielmacci C, Kang L, Wang R, Rouch C, Lefevre AL, Ktorza A, Routh VH, Levin BE, Magnan C. Fatty acid signaling in the hypothalamus and the neural control of insulin secretion. Diabetes 55Suppl 2: S139–144, 2006. [Google Scholar]
- 338.Migrenne S, Le Foll C, Levin BE, Magnan C. Brain lipid sensing and nervous control of energy balance. Diabetes Metab 37: 83–88, 2011. [DOI] [PubMed] [Google Scholar]
- 339.Mingrone G, Manco M, Mora ME, Guidone C, Iaconelli A, Gniuli D, Leccesi L, Chiellini C, Ghirlanda G. Influence of maternal obesity on insulin sensitivity and secretion in offspring. Diabetes Care 31: 1872–1876, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Mistry AM, Swick A, Romsos DR. Leptin alters metabolic rates before acquisition of its anorectic effect in developing neonatal mice. Am J Physiol Regul Integr Comp Physiol 277: R742–R747, 1999. [DOI] [PubMed] [Google Scholar]
- 341.Mitchel JS, Keesey RE. Defense of a lowered weight maintenance level by lateral hypothalamically lesioned rats: Evidence from a restriction-refeeding regimen. Physiol Behav 18: 1121–1125, 1977. [DOI] [PubMed] [Google Scholar]
- 342.Mitchell V, Bouret S, Beauvillain JC, Schilling A, Perret M, Kordon C, Epelbaum J. Comparative distribution of mRNA encoding the growth hormone secretagogue-receptor (GHS-R) in Microcebus murinus (primate, lemurian) and rat forebrain and pituitary. J Comp Neurol 429: 469–489, 2001. [DOI] [PubMed] [Google Scholar]
- 343.Mizuno Y, Oomura Y. Glucose responding neurons in the nucleus tractus solitarius of the rat, in vitro study. Brain Res 307: 109–116, 1984. [DOI] [PubMed] [Google Scholar]
- 344.Modi N, Thomas EL, Harrington TA, Uthaya S, Dore CJ, Bell JD. Determinants of adiposity during preweaning postnatal growth in appropriately grown and growth-restricted term infants. Pediatr Res 60: 345–348, 2006. [DOI] [PubMed] [Google Scholar]
- 345.Montague CT, Farooqi IS, Whitehead JP, Soos MA, Rau H, Wareham NJ, Sewter CP, Digby JE, Mohammed SN, Hurst JA, Cheetham CH, Earley AR, Barnett AH, Prins JB, O'Rahilly S. Congenital leptin deficiency is associated with severe early-onset obesity in humans. Nature 387: 903–908, 1997. [DOI] [PubMed] [Google Scholar]
- 346.Monteiro PO, Victoria CG. Rapid growth in infancy and childhood and obesity in later life- a systematic review. Obesity Rev 6: 143–154, 2005. [DOI] [PubMed] [Google Scholar]
- 347.Moran TH, Sawyer TK, Seeb DH, Ameglio PJ, Lombard MA, McHugh PR. Potent and sustained satiety actions of a cholecystokinin octapeptide analogue. Am J Clin Nutr 55: 286S–290S, 1992. [DOI] [PubMed] [Google Scholar]
- 348.Morin KH. Perinatal outcomes of obese women: a review of the literature. J Obstet Gynecol Neonatal Nursing 27: 431–440, 1998. [DOI] [PubMed] [Google Scholar]
- 349.Mosinger B, Placer Z, Koldovsky O. Passage of insulin through the wall of the gastro-intestinal tract of the infant rat. Nature 184Suppl 16: 1245–1246, 1959. [DOI] [PubMed] [Google Scholar]
- 350.Muroya S, Uramura K, Sakurai T, Takigawa M, Yada T. Lowering glucose concentrations increases cytosolic Ca2+ in orexin neurons of the rat lateral hypothalamus. Neurosci Lett 309: 165–168, 2001. [DOI] [PubMed] [Google Scholar]
- 351.Muroya S, Yada T, Shioda S, Takigawa M. Glucose-sensitive neurons in the rat arcuate nucleus contain neuropeptide Y. Neurosci Lett 264: 113–116, 1999. [DOI] [PubMed] [Google Scholar]
- 352.Nakazato M, Murakami N, Date Y, Kojima M, Matsuo H, Kangawa K, Matsukura S. A role for ghrelin in the central regulation of feeding. Nature 409: 194–198, 2001. [DOI] [PubMed] [Google Scholar]
- 354.Naleid A, Grace M, Cummings D, Levine A. Ghrelin induces feeding in the mesolimbic reward pathway between the ventral tegmental area and the nucleus accumbens. Peptides 26: 2274–2279, 2005. [DOI] [PubMed] [Google Scholar]
- 355.Nataf V, Monier S. Effect of insulin and insulin-like growth factor I on the expression of the catecholaminergic phenotype by neural crest cells. Brain Res Dev Brain Res 69: 59–66, 1992. [DOI] [PubMed] [Google Scholar]
- 355a.National Heart, Lung, and Blood Institute. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults: the Evidence Report. Bethesda, MD: NIH, 1998. [Google Scholar]
- 356.Newsome CA, Shiell AW, Fall CH, Phillips DI, Shier R, Law CM. Is birth weight related to later glucose and insulin metabolism? A systematic review. Diabetic Med 20: 339–348, 2003. [DOI] [PubMed] [Google Scholar]
- 357.Ng SF, Lin RC, Laybutt DR, Barres R, Owens JA, Morris MJ. Chronic high-fat diet in fathers programs beta-cell dysfunction in female rat offspring. Nature 467: 963–966, 2010. [DOI] [PubMed] [Google Scholar]
- 358.Ni Mhurchu C, Rodgers A, Pan WH, Gu DF, Woodward M, Asia Pacific Cohort Studies. Body mass index and cardiovascular disease in the Asia-Pacific Region: an overview of 33 cohorts involving 310,000 participants. Int J Epidemiol 33: 751–758, 2004. [DOI] [PubMed] [Google Scholar]
- 359.Niblock MM, Brunso-Bechtold JK, Riddle DR. Laminar variation in neuronal viability and trophic dependence in neocortical slices. J Neurosci Res 65: 455–462, 2001. [DOI] [PubMed] [Google Scholar]
- 360.Nijland MJ, Mitsuya K, Li C, Ford S, McDonald TJ, Nathanielsz PW, Cox LA. Epigenetic modification of fetal baboon hepatic phosphoenolpyruvate carboxykinase following exposure to moderately reduced nutrient availability. J Physiol 88: 1349–1359, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 361.Nilsson I, Johansen JE, Schalling M, Hokfelt T, Fetissov SO. Maturation of the hypothalamic arcuate agouti-related protein system during postnatal development in the mouse. Brain Res 155: 147–154, 2005. [DOI] [PubMed] [Google Scholar]
- 362.Nonaka N, Shioda S, Niehoff ML, Banks WA. Characterization of blood-brain barrier permeability to PYY3–36 in the mouse. J Pharmacol Exp Ther 306: 948–953, 2003. [DOI] [PubMed] [Google Scholar]
- 363.Nyirenda MJ, Welberg LA, Seckl JR. Programming hyperglycaemia in the rat through prenatal exposure to glucocorticoids-fetal effect or maternal influence? J Endocrinol 170: 653–660, 2001. [DOI] [PubMed] [Google Scholar]
- 364.O'Donnell K, O'Conner TG, Glover V. Prenatal stress and neurodevelopment of the child: focus on the HPA axis and role of the placenta. Dev Neurosci 31: 285–292, 2009. [DOI] [PubMed] [Google Scholar]
- 365.O'Malley D, Reimann F, Simpson AK, Gribble FM. Sodium-coupled glucose cotransporters contribute to hypothalamic glucose sensing. Diabetes 55: 3381–3386, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.O'Rahilly S. Human genetics illuminates the paths to metabolic disease. Nature 462: 307–314, 2009. [DOI] [PubMed] [Google Scholar]
- 367.Obici S, Feng Z, Karkanias G, Baskin DG, Rossetti L. Decreasing hypothalamic insulin receptors causes hyperphagia and insulin resistance in rats. Nature Neurosci 5: 566–572, 2002. [DOI] [PubMed] [Google Scholar]
- 368.Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity and trends in body mass index among US children and adolescents, 1999–2010. JAMA 307: 483–490, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Oken E. Maternal and child obesity: the causal link. Obstet Gynecol Clin N Am 36: 361–377, 2009. [DOI] [PubMed] [Google Scholar]
- 370.Olsen SF, Halldorsson TI, Willett WC, Knudsen VK, Gillman MW, Mikkelsen TB, Olsen J, Consortium N. Milk consumption during pregnancy is associated with increased infant size at birth: prospective cohort study. Am J Clin Nutr 86: 1104–1110, 2007. [DOI] [PubMed] [Google Scholar]
- 371.Ong KK, Loos RJ. Rapid infancy weight gain and subsequent obesity: systematic reviews and hopeful suggestions. Acta Paediatr 95: 904–908, 2006. [DOI] [PubMed] [Google Scholar]
- 372.Oomura Y, Kimura K, Ooyama H, Maeno T, Iki M, Kuniyoshi M. Reciprocal activities of the ventromedial and lateral hypothalamic areas of cats. Science 143: 484–485, 1964. [DOI] [PubMed] [Google Scholar]
- 373.Oomura Y, Kimura K, Ooyama H, Maeo T, Iki M, Kuniyoshi N. Reciprocal activities of the ventromedial and lateral hypothalamic area of cats. Science 143: 484–485, 1964. [DOI] [PubMed] [Google Scholar]
- 374.Oomura Y, Nakamura T, Sugimori M, Yamada Y. Effect of free fatty acid on the rat lateral hypothalamic neurons. Physiol Behav 14: 483–486, 1975. [DOI] [PubMed] [Google Scholar]
- 375.Oomura Y, Ooyama H, Sugimori M, Nakamura T, Yamada Y. Glucose inhibition of the glucose-sensitive neurone in the rat lateral hypothalamus. Nature 247: 284–286, 1974. [DOI] [PubMed] [Google Scholar]
- 376.Orosco M, Nicolaidis S. Insulin and glucose-induced changes in feeding and medial hypothalamic monoamines revealed by microdialysis in rats. Brain Research Bull 33: 289–297, 1994. [DOI] [PubMed] [Google Scholar]
- 377.Oscai LB, McGarr JA. Evidence that the amount of food consumed in early life fixes appetite in the rat. Am J Physiol Regul Integr Comp Physiol 235: R141–R144, 1978. [DOI] [PubMed] [Google Scholar]
- 378.Osmond DT, King RG, Brennecke SP, Gude NM. Placental glucose transport and utilisation is altered at term in insulin-treated, gestational-diabetic patients. Diabetologia 44: 1133–1139, 2001. [DOI] [PubMed] [Google Scholar]
- 379.Owen C, Martin R, Whincup P, Smith G, Cook D. Effect of infant feeding on the risk of obesity across the life course: a quantitative review of published evidence. Pediatrics 115: 1367–1377, 2005. [DOI] [PubMed] [Google Scholar]
- 380.Ozanne SE, Constancia M. Mechanisms of disease: the developmental origins of disease and the role of the epigenotype. Nature Clin Pract Endocrinol Metab 3: 539–546, 2007. [DOI] [PubMed] [Google Scholar]
- 381.Ozanne SE, Lewis R, Jennings BJ, Hales CN. Early programming of weight gain in mice prevents the induction of obesity by a highly palatable diet. Clin Sci 106: 141–145, 2004. [DOI] [PubMed] [Google Scholar]
- 382.Ozanne SE, Olsen GS, Hansen LL, Tingey KJ, Nave BT, Wang CL, Hartil K, Petry CJ, Buckley AJ, Mosthaf-Seedorf L. Early growth restriction leads to down regulation of protein kinase C zeta and insulin resistance in skeletal muscle. J Endocrinol 177: 235–241, 2003. [DOI] [PubMed] [Google Scholar]
- 383.Padilla SL, Carmody JS, Zeltser LM. POMC-expressing progenitors give rise to antagonistic neuronal populations in hypothalamic feeding circuits. Nature Med 16: 403–405, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Padmavathi IJ, Rao KR, Venu L, Ganeshan M, Kumar KA, Rao Ch N, Harishankar N, Ismail A, Raghunath M. Chronic maternal dietary chromium restriction modulates visceral adiposity: probable underlying mechanisms. Diabetes 59: 98–104, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Pan DA, Lillioja S, Kriketos AD, Milner MR, Baur LA, Bogardus C, Jenkins AB, Storlien LH. Skeletal muscle triglyceride levels are inversely related to insulin action. Diabetes 46: 983–988, 1997. [DOI] [PubMed] [Google Scholar]
- 386.Pandolfino JE, El-Serag HB, Zhang Q, Shah N, Ghosh SK, Kahrilas PJ. Obesity: a challenge to esophagogastric junction integrity. Gastroenterology 130: 639–649, 2006. [DOI] [PubMed] [Google Scholar]
- 387.Pankevich DE, Mueller BR, Brockel B, Bale TL. Prenatal stress programming of offspring feeding behavior and energy balance begins early in pregnancy. Physiol Behav 98: 94–102, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Paranjape SA, Chan O, Zhu W, Horblitt AM, Grillo CA, Wilson S, Reagan L, Sherwin RS. Chronic reduction of insulin receptors in the ventromedial hypothalamus produces glucose intolerance and islet dysfunction in the absence of weight gain. Am J Physiol Endocrinol Metab 301: E978–E983, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 389.Park JH, Stoffers DA, Nicholls RD, Simmons RA. Development of type 2 diabetes following intrauterine growth retardation in rats is associated with progressive epigenetic silencing of Pdx1. J Clin Invest 118: 2316–2324, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Parsons MP, Hirasawa M. ATP-sensitive potassium channel-mediated lactate effect on orexin neurons: implications for brain energetics during arousal. J Neurosci 30: 8061–8070, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Patterson CM, Bouret SG, Dunn-Meynell AA, Levin BE. Three weeks of postweaning exercise in DIO rats produces prolonged increases in central leptin sensitivity and signaling. Am J Physiol Regul Integr Comp Physiol 296: R537–R548, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Patterson CM, Bouret SG, park S, Irani BG, Dunn-Meynell AA, Levin BE. Large litter rearing enhances leptin sensitivity and protects selectively bred diet-induced obese (DIO) rats from becoming obese. Endocrinology 151: 4270–4279, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Patterson CM, Bouret SG, Park S, Irani BG, Dunn-Meynell AA, Levin BE. Large litter rearing enhances leptin sensitivity and protects selectively bred diet-induced obese rats from becoming obese. Endocrinology 151: 4270–4279, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Patterson CM, Dunn-Meynell AA, Levin BE. Three weeks of early-onset exercise prolongs obesity resistance in DIO rats after exercise cessation. Am J Physiol Regul Integr Comp Physiol 294: R290–R301, 2008. [DOI] [PubMed] [Google Scholar]
- 395.Patterson LM, Zheng H, Berthoud HR. Vagal afferents innervating the gastrointestinal tract and CCKA-receptor immunoreactivity. Anat Rec 266: 10–20, 2002. [DOI] [PubMed] [Google Scholar]
- 396.Payne PR, Dugdale AA. Mechanisms for the control of body weight. Lancet 8011: 583–568, 1977. [DOI] [PubMed] [Google Scholar]
- 397.Peeters A, Barendregt JJ, Willekens F, Mackenbach JP, Al Mamun A, Bonneux L, Nedcom TNE, Demography Compression of Morbidity Research. Obesity in adulthood and its consequences for life expectancy: a life-table analysis. Ann Internal Med 138: 24–32, 2003. [DOI] [PubMed] [Google Scholar]
- 399.Pelleymounter MA, Cullen MJ, Baker MB, Hecht R, Winters D, Boone T, Collins F. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269: 540–543, 1995. [DOI] [PubMed] [Google Scholar]
- 400.Perello M, Sakata I, Birnbaum S, Chuang JC, Osborne-Lawrence S, Rovinsky SA, Woloszyn J, Yanagisawa M, Lutter M, Zigman JM. Ghrelin increases the rewarding value of high-fat diet in an orexin-dependent manner. Biol Psychiatry 67: 880–886, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.Perello M, Scott MM, Sakata I, Lee CE, Chuang JC, Osborne-Lawrence S, Rovinsky SA, Elmquist JK, Zigman JM. Functional implications of limited leptin receptor and ghrelin receptor coexpression in the brain. J Comp Neurol 520: 281–294, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Petry CJ, Dorling MW, Pawlak DB, Ozanne SE, Hales CN. Diabetes in old male offspring of rat dams fed a reduced protein diet. Int J Exp Diabetes Res 2: 139–143, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 403.Pettitt DJ, Aleck KA, Baird HR, Carraher MJ, Bennett PH, Knowler WC. Congenital susceptibility to NIDDM. Role of intrauterine environment. Diabetes 37: 622–628, 1988. [DOI] [PubMed] [Google Scholar]
- 404.Piao H, Hosoda H, Kangawa K, Murata T, Narita K, Higuchi T. Ghrelin stimulates milk intake by affecting adult type feeding behaviour in postnatal rats. J Neuroendocrinol 20: 330–334, 2008. [DOI] [PubMed] [Google Scholar]
- 405.Pierce A, Xu A. De novo neurogenesis in adult hypothalamus as a compensatory mechanism to regulate energy balance. J Neurosci 30: 723–730, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Pinto S, Roseberry AG, Liu H, Diano S, Shanabrough M, Cai X, Friedman JM, Horvath TL. Rapid rewiring of arcuate nucleus feeding circuits by leptin. Science 304: 110–115, 2004. [DOI] [PubMed] [Google Scholar]
- 407.Plagemann A, Harder T, Brunn M, Harder A, Roepke K, Wittrock-Staar M, Ziska T, Schellong K, Rodekamp E, Melchior K, Dudenhausen JW. Hypothalamic proopiomelanocortin promoter methylation becomes altered by early overfeeding: an epigenetic model of obesity and the metabolic syndrome. J Physiol 587: 4963–4976, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Plagemann A, Harder T, Franke K, Kohlhoff R. Long-term impact of neonatal breast-feeding on body weight and glucose tolerance in children of diabetic mothers. Diabetes Care 25: 16–22, 2002. [DOI] [PubMed] [Google Scholar]
- 409.Plagemann A, Harder T, Janert U, Rake A, Rittel F, Rohde W, Dorner G. Malformations of hypothalamic nuclei in hyperinsulinemic offspring of rats with gestational diabetes. Dev Neurosci 21: 58–67, 1999. [DOI] [PubMed] [Google Scholar]
- 410.Plagemann A, Harder T, Rake A, Janert U, Melchior K, Rohde W, Dorner G. Morphological alterations of hypothalamic nuclei due to intrahypothalamic hyperinsulinism in newborn rats. Int J Dev Neurosci 17: 37–44, 1999. [DOI] [PubMed] [Google Scholar]
- 411.Plagemann A, Harder T, Rake A, Melchior K, Rohde W, Dorner G. Hypothalamic nuclei are malformed in weanling offspring of low protein malnourished rat dams. J Nutr 130: 2582–2589, 2000. [DOI] [PubMed] [Google Scholar]
- 412.Plagemann A, Heidrich I, Gotz F, Rohde W, Dorner G. Lifelong enhanced diabetes susceptibility and obesity after temporary intrahypothalamic hyperinsulinism during brain organization. Exp Clin Endocrinol 99: 91–95, 1992. [DOI] [PubMed] [Google Scholar]
- 413.Plagemann A, Heidrich I, Gotz F, Rohde W, Dorner G. Obesity and enhanced diabetes and cardiovascular risk in adult rats due to early postnatal overfeeding. Exp Clin Endocrinol 99: 154–158, 1992. [DOI] [PubMed] [Google Scholar]
- 414.Plagemann A, Roepke K, Harder T, Brunn M, Harder A, Wittrock-Staar M, Ziska T, Schellong K, Rodekamp E, Melchior K, Dudenhausen JW. Epigenetic malprogramming of the insulin receptor promoter due to developmental overfeeding. J Perinatal Med 38: 393–400, 2010. [DOI] [PubMed] [Google Scholar]
- 415.Pocai A, Lam TK, Gutierrez-Juarez R, Obici S, Schwartz GJ, Bryan J, Aguilar-Bryan L, Rossetti L. Hypothalamic K(ATP) channels control hepatic glucose production. Nature 434: 1026–1031, 2005. [DOI] [PubMed] [Google Scholar]
- 416.Polonsky KS, Given BD, Van Cauter E. Twenty-four-hour profiles and pulsatile patterns of insulin secretion in normal and obese subjects. J Clin Invest 81: 442–448, 1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Poulsen P, Vaag AA, Kyvik KO, Moller Jensen D, Beck-Nielsen H. Low birth weight is associated with NIDDM in discordant monozygotic and dizygotic twin pairs. Diabetologia 40: 439–446, 1997. [DOI] [PubMed] [Google Scholar]
- 418.Power KL, Moore CL. Prenatal stress eliminates differential maternal attention to male offspring in Norway rats. Physiol Behav 38: 667–671, 1986. [DOI] [PubMed] [Google Scholar]
- 419.Prado CL, Pugh-Bernard AE, Elghazi L, Sosa-Pineda B, Sussel L. Ghrelin cells replace insulin-producing beta cells in two mouse models of pancreas development. Proc Natl Acad Sci USA 101: 2924–2929, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 420.Prechtl JC, Powley TL. The fiber composition of the abdominal vagus of the rat. Anat Embryol 181: 101–115, 1990. [DOI] [PubMed] [Google Scholar]
- 421.Prentice A, Jebb S. Energy intake/physical activity interactions in the homeostasis of body weight regulation. Nutr Rev 62: S98–104, 2004. [DOI] [PubMed] [Google Scholar]
- 422.Prospective Studies C, Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J, Halsey J, Qizilbash N, Collins R, Peto R. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 373: 1083–1096, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Puro DG, Agardh E. Insulin-mediated regulation of neuronal maturation. Science 225: 1170–1172, 1984. [DOI] [PubMed] [Google Scholar]
- 424.Quek CM, Koh K, Lee J. Parental body mass index: a predictor of childhood obesity? Ann Acad Med Singapore 22: 342–347, 1993. [PubMed] [Google Scholar]
- 425.Ramachandrappa S, Raimondo A, Cali AM, Keogh JM, Henning E, Saeed S, Thompson A, Garg S, Bochukova EG, Brage S, Trowse V, Wheeler E, Sullivan AE, Dattani M, Clayton PE, Datta V, Bruning JB, Wareham NJ, O'Rahilly S, Peet DJ, Barroso I, Whitelaw ML, Farooqi IS. Rare variants in single-minded 1 (SIM1) are associated with severe obesity. J Clin Invest 123: 3042–3050, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 426.Ramnanan CJ, Saraswathi V, Smith MS, Donahue EP, Farmer B, Farmer TD, Neal D, Williams PE, Lautz M, Mari A, Cherrington AD, Edgerton DS. Brain insulin action augments hepatic glycogen synthesis without suppressing glucose production or gluconeogenesis in dogs. J Clin Invest 121: 3713–3723, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Ravelli AC, van der Meulen JH, Michels RP, Osmond C, Barker DJ, Hales CN, Bleker OP. Glucose tolerance in adults after prenatal exposure to famine. Lancet 351: 173–177, 1998. [DOI] [PubMed] [Google Scholar]
- 428.Ravelli GP, Stein ZA, Susser MW. Obesity in young men after famine exposure in utero and early infancy. N EnglJ Med 295: 349–353, 1976. [DOI] [PubMed] [Google Scholar]
- 429.Raybould HE. Gut microbiota, epithelial function and derangements in obesity. J Physiol 590: 441–446, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Reaven GM. Insulin resistance: a chicken that has come to roost. Ann NY Acad Sci 892: 45–57, 1999. [DOI] [PubMed] [Google Scholar]
- 431.Rebuffe-Scrive M, Walsh UA, McEwen B, Rodin J. Effect of chronic stress and exogenous glucocorticoids on regional fat distribution and metabolism. Physiol Behav 52: 583–590, 1992. [DOI] [PubMed] [Google Scholar]
- 432.Recio-Pinto E, Ishii DN. Effects of insulin, insulin-like growth factor-II and nerve growth factor on neurite outgrowth in cultured human neuroblastoma cells. Brain Res 302: 323–334, 1984. [DOI] [PubMed] [Google Scholar]
- 433.Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet 371: 569–578, 2008. [DOI] [PubMed] [Google Scholar]
- 434.Renstrom F, Payne F, Nordstrom A, Brito EC, Rolandsson O, Hallmans G, Barroso I, Nordstrom P, Franks PW, Consortium G. Replication and extension of genome-wide association study results for obesity in 4923 adults from northern Sweden. Hum Mol Genet 18: 1489–1496, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Reusens B, Ozanne SE, Remacle C. Fetal determinants of type 2 diabetes. Curr Drug Targets 8: 935–941, 2007. [DOI] [PubMed] [Google Scholar]
- 436.Reynolds RM, Osmond C, Phillips DI, Godfrey KM. Maternal BMI, parity, and pregnancy weight gain: influences on offspring adiposity in young adulthood. J Clin Endocrinol Metab 95: 5365–5369, 2010. [DOI] [PubMed] [Google Scholar]
- 437.Reznikov AG, Nosenko ND. Early postnatal changes in sexual dimorphism of catecholamine and indoleamine content in the brain of prenatally stressed rats. Neuroscience 70: 547–551, 1996. [DOI] [PubMed] [Google Scholar]
- 438.Ridaura VK, Faith JJ, Rey FE, Cheng J, Duncan AE, Kau AL, Griffin NW, Lombard V, Henrissat B, Bain JR, Muehlbauer MJ, Ilkayeva O, Semenkovich CF, Funai K, Hayashi DK, Lyle BJ, Martini MC, Ursell LK, Clemente JC, Van Treuren W, Walters WA, Knight R, Newgard CB, Heath AC, Gordon JI. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 341: 124–214, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Rinaman L. Oxytocinergic inputs to the nucleus of the solitary tract and dorsal motor nucleus of the vagus in neonatal rats. J Comp Neurol 399: 101–109, 1998. [DOI] [PubMed] [Google Scholar]
- 440.Rinaman L. Postnatal development of catecholamine inputs to the paraventricular nucleus of the hypothalamus in rats. J Comp Neurol 438: 411–422, 2001. [DOI] [PubMed] [Google Scholar]
- 441.Rinaman L, Levitt P, Card JP. Progressive postnatal assembly of limbic-autonomic circuits revealed by central transneuronal transport of pseudorabies virus. J Neurosci 20: 2731–2741, 2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 442.Rinaman L, Miselis RR. The organization of vagal innervation of rat pancreas using cholera toxin-horseradish peroxidase conjugate. J Auton Nerv Syst 21: 109–125, 1987. [DOI] [PubMed] [Google Scholar]
- 443.Ritter S, Taylor JS. Vagal sensory neurons are required for lipoprivic but not glucoprivic feeding in rats. Am J Physiol Regul Integr Comp Physiol 258: R1395–R1401, 1990. [DOI] [PubMed] [Google Scholar]
- 444.Rodgers AB, Morgan CP, Bronson SL, Revello S, Bale TL. Paternal stress exposure alters sperm microRNA content and reprograms offspring HPA stress axis regulation. J Neurosci 33: 9003–9012, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Roeder LM, Poduslo SE, Tildon JT. Utilization of ketone bodies and glucose by established neural cell lines. J Neurosci Res 8: 671–682, 1982. [DOI] [PubMed] [Google Scholar]
- 446.Romero-Corral A, Somers VK, Sierra-Johnson J, Korenfeld Y, Boarin S, Korinek J, Jensen MD, Parati G, Lopez-Jimenez F. Normal weight obesity: a risk factor for cardiometabolic dysregulation and cardiovascular mortality. Eur Heart J 31: 737–746, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 447.Rorsman P, Braun M. Regulation of insulin secretion in human pancreatic islets. Annu Rev Physiol 75: 155–179, 2013. [DOI] [PubMed] [Google Scholar]
- 448.Rosenbaum M, Leibel RL. Adaptive thermogenesis in humans. Int J Obesity 34Suppl 1: S47–55, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 449.Rosmond R, Dallman MF, Bjorntorp P. Stress-related cortisol secretion in men: relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. J Clin Endocrinol Metab 83: 1853–1859, 1998. [DOI] [PubMed] [Google Scholar]
- 450.Routh VH. Glucosensing neurons in the ventromedial hypothalamic nucleus (VMN) and hypoglycemia-associated autonomic failure (HAAF). Diabetes Metab Res Rev 19: 348–356, 2003. [DOI] [PubMed] [Google Scholar]
- 451.Routh VH, McArdle JJ, Spanswick DC, Levin BE, Ashford MLJ. Insulin modulates the activity of glucose responsive neurons in the ventromedial hypothalamic nucleus (VMN). Abstr Soc Neurosci 23: 577A, 1997. [Google Scholar]
- 452.Routh VH, Murakami DM, Stern JS, Fuller CA, Horwitz BA. Neuronal activity in hypothalamic nuclei of obese and lean Zucker rats. Int J Obesity 14: 879–891, 1990. [PubMed] [Google Scholar]
- 453.Ruderman NB, Carling D, Prentki M, Cacicedo JM. AMPK, insulin resistance, and the metabolic syndrome. J Clin Invest 123: 2764–2772, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Sakata I, Tanaka T, Matsubara M, Yamazaki M, Tani S, Hayashi Y, Kangawa K, Sakai T. Postnatal changes in ghrelin mRNA expression and in ghrelin-producing cells in the rat stomach. J Endocrinol 174: 463–471, 2002. [DOI] [PubMed] [Google Scholar]
- 455.Samuelsson AM, Matthews PA, Argenton M, Christie MR, McConnell JM, Jansen EH, Piersma AH, Ozanne SE, Twinn DF, Remacle C, Rowlerson A, Poston L, Taylor PD. Diet-induced obesity in female mice leads to offspring hyperphagia, adiposity, hypertension, and insulin resistance: a novel murine model of developmental programming. Hypertension 51: 383–392, 2008. [DOI] [PubMed] [Google Scholar]
- 456.Sandholt CH, Hansen T, Pedersen O. Beyond the fourth wave of genome-wide obesity association studies. Nutr Diabetes 2: e37, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Sandovici I, Smith NH, Nitert MD, Ackers-Johnson M, Uribe-Lewis S, Ito Y, Jones RH, Marquez VE, Cairns W, Tadayyon M, O'Neill LP, Murrell A, Ling C, Constancia M, Ozanne SE. Maternal diet and aging alter the epigenetic control of a promoter-enhancer interaction at the Hnf4a gene in rat pancreatic islets. Proc Natl Acad Sci USA 108: 5449–5454, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 458.Saper CB. The Rat Nervous System, edited by Paxinos G. San Diego, CA: Academic, 1995, p. 107–135. [Google Scholar]
- 459.Sarugeri E, Dozio N, Belloni C, Meschi F, Pastore MR, Bonifacio E. Autoimmune responses to the beta cell autoantigen, insulin, and the INS VNTR-IDDM2 locus. Clin Exp Immunol 114: 370–376, 1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Satoh N, Ogawa Y, Katsuura G, Tsuji T, Masuzaki H, Hiraoka J, Okazaki T, Tamaki M, Hayase M, Yoshimasa Y, Nishi S, Hosoda K, Nakao K. Pathophysiological significance of the obese gene product, leptin, in ventromedial hypothalamus (VMH)-lesioned rats: evidence for loss of its satiety effect in VMH-lesioned rats. Endocrinology 138: 947–954, 1997. [DOI] [PubMed] [Google Scholar]
- 461.Sawchenko PE. Toward a new neurobiology of energy balance, appetite, and obesity: the anatomists weigh in. J Comp Neurol 402: 435–441, 1998. [PubMed] [Google Scholar]
- 462.Scallet AC, Olney JW. Components of hypothalamic obesity: bipiperidyl-mustard lesions add hyperphagia to monosodium glutamate-induced hyperinsulinemia. Brain Res 374: 380–384, 1986. [DOI] [PubMed] [Google Scholar]
- 463.Schechter R, Abboud M. Neuronal synthesized insulin roles on neural differentiation within fetal rat neuron cell cultures. Brain Res 127: 41–49, 2001. [DOI] [PubMed] [Google Scholar]
- 464.Schechter R, Yanovitch T, Abboud M, Johnson IG, Gaskins J. Effects of brain endogenous insulin on neurofilament and MAPK in fetal rat neuron cell culture. Brain Res 808: 270–278, 1998. [DOI] [PubMed] [Google Scholar]
- 465.Schwartz GJ. The role of gastrointestinal vagal afferents in the control of food intake: current prospects. Nutrition 16: 866–873, 2000. [DOI] [PubMed] [Google Scholar]
- 466.Schwartz GJ, Moran TH. CCK elicits and modulates vagal afferent activity arising from gastric and duodenal sites. Ann NY Acad Sci 713: 121–128, 1994. [DOI] [PubMed] [Google Scholar]
- 467.Schwartz MW, Marks JL, Sipols AJ, Baskin DG, Woods SC, Kahn SE, Porte D Jr. Central insulin administration reduces neuropeptide Y mRNA expression in the arcuate nucleus of food-deprived lean (Fa/Fa) but not obese (fa/fa) Zucker rats. Endocrinology 128: 2645–2647, 1991. [DOI] [PubMed] [Google Scholar]
- 468.Schwartz MW, Seeley RJ, Campfield LA, Burn P, Baskin DG. Identification of targets of leptin action in rat hypothalamus. J Clin Invest 98: 1101–1106, 1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 469.Sclafani A, Springer D. Dietary obesity in adult rats: similarities to hypothalamic and human obesity syndromes. Physiol Behav 17: 461–471, 1976. [DOI] [PubMed] [Google Scholar]
- 470.Scott MM, Lachey JL, Sternson SM, Lee CE, Elias CF, Friedman JM, Elmquist JK. Leptin targets in the mouse brain. J Comp Neurol 514: 518–532, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 471.Scott MM, Perello M, Chuang JC, Sakata I, Gautron L, Lee CE, Lauzon D, Elmquist JK, Zigman JM. Hindbrain ghrelin receptor signaling is sufficient to maintain fasting glucose. PloS One 7: e44089, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 472.Segar EM, Norris AW, Yao JR, Hu S, Koppenhafer SL, Roghair RD, Segar JL, Scholz TD. Programming of growth, insulin resistance and vascular dysfunction in offspring of late gestation diabetic rats. Clin Sci 117: 129–138, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Shankar K, Harrell A, Liu X, Gilchrist JM, Ronis MJ, Badger TM. Maternal obesity at conception programs obesity in the offspring. Am J Physiol Regul Integr Comp Physiol 294: R528–R538, 2008. [DOI] [PubMed] [Google Scholar]
- 474.Shimizu N, Oomura Y, Sakata T. Modulation of feeding by endogenous sugar acids acting as hunger or satiety factors. Am J Physiol Regul Integr Comp Physiol 246: R542–R550, 1984. [DOI] [PubMed] [Google Scholar]
- 475.Simmons RA, Templeton LJ, Gertz SJ. Intrauterine growth retardation leads to the development of type 2 diabetes in the rat. Diabetes 50: 2279–2286, 2001. [DOI] [PubMed] [Google Scholar]
- 476.Singhal A, Cole TJ, Fewtrell M, Kennedy K, Stephenson T, Elias-Jones A, Lucas A. Promotion of faster weight gain in infants born small for gestational age: is there an adverse effect on later blood pressure? Circulation 115: 213–220, 2007. [DOI] [PubMed] [Google Scholar]
- 477.Singhal A, Kennedy K, Lanigan J, Fewtrell M, Cole TJ, Stephenson T, Elias-Jones A, Weaver LT, Ibhanesebhor S, MacDonald PD, Bindels J, Lucas A. Nutrition in infancy and long-term risk of obesity: evidence from 2 randomized controlled trials. Am J Clin Nutr 92: 1133–1144, 2010. [DOI] [PubMed] [Google Scholar]
- 478.Skibicka KP, Hansson C, Egecioglu E, Dickson SL. Role of ghrelin in food reward: impact of ghrelin on sucrose self-administration and mesolimbic dopamine and acetylcholine receptor gene expression. Addiction Biol 17: 95–107, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Smith GP, Epstein AN. Increased feeding in response to decreased glucose utilization in the rat and monkey. Am J Physiol 217: 1083–1087, 1969. [DOI] [PubMed] [Google Scholar]
- 480.Smith GP, Gibbs J. Cholecystokinin: a putative satiety signal. Pharmacol Biochem Behav 3: 135–138, 1975. [PubMed] [Google Scholar]
- 481.Smith J, Cianflone K, Biron S, Hould FS, Lebel S, Marceau S, Lescelleur O, Biertho L, Simard S, Kral JG, Marceau P. Effects of maternal surgical weight loss in mothers on intergenerational transmission of obesity. J Clin Endocrinol Metab 94: 4275–4283, 2009. [DOI] [PubMed] [Google Scholar]
- 482.Smythe JW, McCormick CM, Meaney MJ. Median eminence corticotrophin-releasing hormone content following prenatal stress and neonatal handling. Brain Res Bull 40: 195–199, 1996. [DOI] [PubMed] [Google Scholar]
- 483.Sohn JW, Elmquist JK, Williams KW. Neuronal circuits that regulate feeding behavior and metabolism. Trends Neurosci 36: 504–512, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 484.Song Z, Levin BE, McArdle JJ, Bakhos N, Routh VH. Convergence of pre- and postsynaptic influences on glucosensing neurons in the ventromedial hypothalamic nucleus (VMN). Diabetes 50: 2673–2681, 2001. [DOI] [PubMed] [Google Scholar]
- 485.Song Z, Routh VH. Differential effects of glucose and lactate on glucosensing neurons in the ventromedial hypothalamic nucleus. Diabetes 54: 15–22, 2005. [DOI] [PubMed] [Google Scholar]
- 486.Spanswick D, Smith MA, Groppi VE, Logan SD, Ashford ML. Leptin inhibits hypothalamic neurons by activation of ATP- sensitive potassium channels. Nature 390: 521–525, 1997. [DOI] [PubMed] [Google Scholar]
- 487.Spanswick D, Smith MA, Mirshamsi S, Routh VH, Ashford ML. Insulin activates ATP-sensitive K+ channels in hypothalamic neurons of lean, but not obese rats. Nature Neurosci 3: 757–758, 2000. [DOI] [PubMed] [Google Scholar]
- 488.Speakman JR, Levitsky DA, Allison DB, Bray MS, de Castro JM, Clegg DJ, Clapham JC, Dulloo AG, Gruer L, Haw S, Hebebrand J, Hetherington MM, Higgs S, Jebb SA, Loos RJ, Luckman S, Luke A, Mohammed-Ali V, O'Rahilly S, Pereira M, Perusse L, Robinson TN, Rolls B, Symonds ME, Westerterp-Plantenga MS. Set points, settling points and some alternative models: theoretical options to understand how genes and environments combine to regulate body adiposity. Disease Models Mechanisms 4: 733–745, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 489.Stampfer MJ, Maclure KM, Colditz GA, Manson JE, Willett WC. Risk of symptomatic gallstones in women with severe obesity. Am J Clin Nutr 55: 652–658, 1992. [DOI] [PubMed] [Google Scholar]
- 490.Steculorum SM, Bouret SG. Developmental effects of ghrelin. Peptides 32: 2362–2366, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 491.Steculorum SM, Bouret SG. Maternal diabetes compromises the organization of hypothalamic feeding circuits and impairs leptin sensitivity in offspring. Endocrinology 152: 4171–4179, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 492.Stein CJ, Colditz GA. The epidemic of obesity. J Clin Endocrinol Metab 89: 2522–2525, 2004. [DOI] [PubMed] [Google Scholar]
- 493.Stellar E. The physiology of motivation. Psychol Rev 5: 5–22, 1954. [DOI] [PubMed] [Google Scholar]
- 494.Steppan CM, Swick AG. A role for leptin in brain development. Biochem Biophys Res Commun 256: 600–602, 1999. [DOI] [PubMed] [Google Scholar]
- 495.Stettler N, Stallings VA, Troxel AB, Zhao J, Schinnar R, Nelson SE, Ziegler EE, Strom BL. Weight gain in the first week of life and overweight in adulthood: a cohort study of European American subjects fed infant formula. Circulation 111: 1897–1903, 2005. [DOI] [PubMed] [Google Scholar]
- 496.Stevens A, Begum G, Cook A, Connor K, Rumball C, Oliver M, Challis J, Bloomfield F, White A. Epigenetic changes in the hypothalamic proopiomelanocortin and glucocorticoid receptor genes in the ovine fetus after periconceptional undernutrition. Endocrinology 151: 3652–3664, 2010. [DOI] [PubMed] [Google Scholar]
- 497.Stevens J. Obesity and mortality in Africans-Americans. Nutr Rev 58: 346–353, 2000. [DOI] [PubMed] [Google Scholar]
- 498.Stewart CP, Christian P, Schulze KJ, Arguello M, LeClerq SC, Khatry SK, West KP Jr. Low maternal vitamin B-12 status is associated with offspring insulin resistance regardless of antenatal micronutrient supplementation in rural Nepal. J Nutr 141: 1912–1917, 2011. [DOI] [PubMed] [Google Scholar]
- 499.Stommel M, Schoenborn CA. Variations in BMI and prevalence of health risks in diverse racial and ethnic populations. Obesity 18: 1821–1826, 2010. [DOI] [PubMed] [Google Scholar]
- 500.Stratigopoulos G, LeDuc CA, Cremona ML, Chung WK, Leibel RL. Cut-like homeobox 1 (CUX1) regulates expression of the fat mass and obesity-associated and retinitis pigmentosa GTPase regulator-interacting protein-1-like (RPGRIP1L) genes and coordinates leptin receptor signaling. J Biol Chem 286: 2155–2170, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 501.Strazzullo P, D'Elia L, Cairella G, Garbagnati F, Cappuccio FP, Scalfi L. Excess body weight and incidence of stroke: meta-analysis of prospective studies with 2 million participants. Stroke 41: e418–426, 2010. [DOI] [PubMed] [Google Scholar]
- 502.Stunkard AJ, Foch TT, Hrubec Z. A twin study of human obesity. JAMA 256: 51–54, 1986. [PubMed] [Google Scholar]
- 503.Surwit RS, Feinglos MN, Rodin J, Sutherland A, Petro AE, Opara EC, Kuhn CM, Rebuffe-Scrive M. Differential effects of fat and sucrose on the development of obesity and diabetes in C57BL/6J and A/J mice. Metab Clin Exp 44: 645–651, 1995. [DOI] [PubMed] [Google Scholar]
- 505.Surwit RS, Kuhn CM, Cochrane C, McCubbin JA, Feinglos MN. Diet-induced type II diabetes in C57BL/6J mice. Diabetes 37: 1163–1167, 1988. [DOI] [PubMed] [Google Scholar]
- 506.Swanson LW, Sawchenko PE. Hypothalamic integration: organization of the paraventricular and supraoptic nuclei. Annu Rev Neurosci 6: 269–324, 1983. [DOI] [PubMed] [Google Scholar]
- 507.Szczypka MS, Rainey MA, Palmiter RD. Dopamine is required for hyperphagia in Lep(ob/ob) mice. Nature Genet 25: 102–104, 2000. [DOI] [PubMed] [Google Scholar]
- 508.Tamashiro KL, Terrillion CE, Hyun J, Koenig JI, Moran TH. Prenatal stress or high-fat diet increases susceptibility to diet-induced obesity in rat offspring. Diabetes 58: 1116–1125, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 509.Tamura H, Kamegai J, Shimizu T, Ishii S, Sugihara H, Oikawa S. Ghrelin stimulates GH but not food intake in arcuate nucleus ablated rats. Endocrinology 143: 3268–3275, 2002. [DOI] [PubMed] [Google Scholar]
- 510.Tanner GR, Lutas A, Martinez-Francois JR, Yellen G. Single KATP channel opening in response to action potential firing in mouse dentate granule neurons. J Neurosci 31: 8689–8696, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 511.Taricco E, Radaelli T, Nobile de Santis MS, Cetin I. Foetal and placental weights in relation to maternal characteristics in gestational diabetes. Placenta 24: 343–347, 2003. [DOI] [PubMed] [Google Scholar]
- 512.Taylor PD, McConnell J, Khan IY, Holemans K, Lawrence KM, Asare-Anane H, Persaud SJ, Jones PM, Petrie L, Hanson MA, Poston L. Impaired glucose homeostasis and mitochondrial abnormalities in offspring of rats fed a fat-rich diet in pregnancy. Am J Physiol Regul Integr Comp Physiol 288: R134–R139, 2005. [DOI] [PubMed] [Google Scholar]
- 513.Tennant PW, Rankin J, Bell R. Maternal body mass index and the risk of fetal and infant death: a cohort study from the North of England. Hum Reprod 26: 1501–1511, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 514.Tobi EW, Lumey LH, Talens RP, Kremer D, Putter H, Stein AD, Slagboom PE, Heijmans BT. DNA methylation differences after exposure to prenatal famine are common and timing- and sex-specific. Hum Mol Genet 18: 4046–4053, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 515.Tolle V, Bassant MH, Zizzari P, Poindessous-Jazat F, Tomasetto C, Epelbaum J, Bluet-Pajot MT. Ultradian rhythmicity of ghrelin secretion in relation with GH, feeding behavior, and sleep-wake patterns in rats. Endocrinology 143: 1353–1361, 2002. [DOI] [PubMed] [Google Scholar]
- 516.Tong Q, Ye C, Jones J, Elmquist J, Lowell B. Synaptic release of GABA by AgRP neurons is required for normal regulation of energy balance. Nature Neurosci 11: 998–1000, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 517.Toperoff G, Aran D, Kark JD, Rosenberg M, Dubnikov T, Nissan B, Wainstein J, Friedlander Y, Levy-Lahad E, Glaser B, Hellman A. Genome-wide survey reveals predisposing diabetes type 2-related DNA methylation variations in human peripheral blood. Hum Mol Genet 21: 371–383, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 518.Trabulsi JC, Mennella JA. Diet, sensitive periods in flavour learning, and growth. Int Rev Psychiatry 24: 219–230, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 519.Tsugane S, Sasaki S, Tsubono Y. Under- and overweight impact on mortality among middle-aged Japanese men and women: a 10-y follow-up of JPHC study cohort I. Int J Obes Relat Disorders 26: 529–537, 2002. [DOI] [PubMed] [Google Scholar]
- 520.Turnbaugh PJ, Backhed F, Fulton L, Gordon JI. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe 3: 213–223, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 521.Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444: 1027–1031, 2006. [DOI] [PubMed] [Google Scholar]
- 522.Turnbaugh PJ, Ridaura VK, Faith JJ, Rey FE, Knight R, Gordon JI. The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Science Transl Med 1: 6ra14, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 523.Tzoulaki I, Sovio U, Pillas D, Hartikainen AL, Pouta A, Laitinen J, Tammelin TH, Jarvelin MR, Elliott P. Relation of immediate postnatal growth with obesity and related metabolic risk factors in adulthood: the northern Finland birth cohort 1966 study. Am J Epidemiol 171: 989–998, 2010. [DOI] [PubMed] [Google Scholar]
- 524.Uauy R, Hoffman DR, Peirano P, Birch DG, Birch EE. Essential fatty acids in visual and brain development. Lipids 36: 885–895, 2001. [DOI] [PubMed] [Google Scholar]
- 525.Ueda H, Ikegami H, Kawaguchi Y, Fujisawa T, Yamato E, Shibata M, Ogihara T. Genetic analysis of late-onset type 2 diabetes in a mouse model of human complex trait. Diabetes 48: 1168–1174, 1999. [DOI] [PubMed] [Google Scholar]
- 526.Ungerstedt U. Adipsia and aphagia after 6-hydroxydopamine induced degeneration of the nigro-striatal dopamine system. Acta Physiol Scand Suppl 367: 95–122, 1971. [DOI] [PubMed] [Google Scholar]
- 527.Vaisse C, Halaas JL, Horvath CM, Darnell JE Jr, Stoffel M, Friedman JM. Leptin activation of Stat3 in the hypothalamus of wild-type and ob/ob mice but not db/db mice. Nature Genet 14: 95–97, 1996. [DOI] [PubMed] [Google Scholar]
- 528.Van Assche FA, Holemans K, Aerts L. Long-term consequences for offspring of diabetes during pregnancy. Br Med Bull 60: 173–182, 2001. [DOI] [PubMed] [Google Scholar]
- 529.Van Itallie TB, Beaudoin R, Mayer J. Arteriovenous glucose differences, metabolic hypoglycemia and food intake in man. J Clin Nutr 1: 208–217, 1953. [DOI] [PubMed] [Google Scholar]
- 530.Vannucci SJ, Clark RR, Koehler-Stec E, Li K, Smith CB, Davies P, Maher F, Simpson IA. Glucose transporter expression in brain: relationship to cerebral glucose utilization. Dev Neurosci 20: 369–379, 1998. [DOI] [PubMed] [Google Scholar]
- 531.Vasan RS, Pencina MJ, Cobain M, Freiberg MS, D'Agostino RB. Estimated risks for developing obesity in the Framingham Heart Study. Ann Int Med 143: 473–480, 2005. [DOI] [PubMed] [Google Scholar]
- 532.Vickers MH, Breier BH, Cutfield WS, Hofman PL, Gluckman PD. Fetal origins of hyperphagia, obesity, and hypertension and postnatal amplification by hypercaloric nutrition. Am J Physiol Endocrinol Metab 279: E83–E87, 2000. [DOI] [PubMed] [Google Scholar]
- 533.Vickers MH, Gluckman PD, Coveny AH, Hofman PL, Cutfield WS, Gertler A, Breier BH, Harris M. Neonatal leptin treatment reverses developmental programming. Endocrinology 146: 4211–4216, 2005. [DOI] [PubMed] [Google Scholar]
- 534.Vilberg TR, Keesey RE. Reduced energy expenditure after ventromedial hypothalamic lesions in female rats. Am J Physiol Regul Integr Comp Physiol 247: R183–R188, 1984. [DOI] [PubMed] [Google Scholar]
- 535.Vogt MC, Paeger L, Hess S, Steculorum SM, Awazawa M, Hampel B, Neupert S, Nicholls HT, Mauer J, Hausen AC, Predel R, Kloppenburg P, Horvath TL, Bruning JC. Neonatal insulin action impairs hypothalamic neurocircuit formation in response to maternal high-fat feeding. Cell 156: 495–509, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 536.Vohr BR, Boney CM. Gestational diabetes: the forerunner for the development of maternal and childhood obesity and metabolic syndrome? J Maternal Fetal Neonatal Med 21: 149–157, 2008. [DOI] [PubMed] [Google Scholar]
- 537.Vucetic Z, Kimmel J, Totoki K, Hollenbeck E, Reyes TM. Maternal high-fat diet alters methylation and gene expression of dopamine and opioid-related genes. Endocrinology 151: 4756–4764, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 538.Wadden TA, Neiberg RH, Wing RR, Clark JM, Delahanty LM, Hill JO, Krakoff J, Otto A, Ryan DH, Vitolins MZ, Look ARG. Four-year weight losses in the Look AHEAD study: factors associated with long-term success. Obesity 19: 1987–1998, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 539.Waddington CH. Organizers and Genes. Cambridge, UK: Cambridge Univ. Press, 1940. [Google Scholar]
- 540.Wang L, Saint-Pierre DH, Tache Y. Peripheral ghrelin selectively increases Fos expression in neuropeptide Y-synthesizing neurons in mouse hypothalamic arcuate nucleus. Neurosci Lett 325: 47–51, 2002. [DOI] [PubMed] [Google Scholar]
- 541.Wang R, Liu X, Hentges ST, Dunn-Meynell AA, Levin BE, Wang W, Routh VH. The regulation of glucose-excited neurons in the hypothalamic arcuate nucleus by glucose and feeding-relevant peptides. Diabetes 53: 1959–1965, 2004. [DOI] [PubMed] [Google Scholar]
- 542.Warzecha Z, Dembinski A, Ceranowicz P, Dembinski M, Cieszkowski J, Bielanski W, Pawlik WW, Kuwahara A, Kato I. Dual age-dependent effect of ghrelin administration on serum level of insulin-like growth factor-1 and gastric growth in young rats. Eur J Pharmacol 529: 145–150, 2006. [DOI] [PubMed] [Google Scholar]
- 543.Waterland RA, Jirtle RL. Transposable elements: targets for early nutritional effects on epigenetic gene regulation. Mol Cell Biol 23: 5293–5300, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 544.Watts AG. Understanding the neural control of ingestive behaviors: helping to separate cause from effect with dehydration-associated anorexia. Horm Behav 37: 261–283, 2000. [DOI] [PubMed] [Google Scholar]
- 545.Weir GC, Bonner-Weir S. Islet beta cell mass in diabetes and how it relates to function, birth, and death. Ann NY Acad Sci 1281: 92–105, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 546.Welberg LA, Seckl JR, Holmes MC. Prenatal glucocorticoid programming of brain corticosteroid receptors and corticotrophin-releasing hormone: possible implications for behaviour. Neuroscience 104: 71–79, 2001. [DOI] [PubMed] [Google Scholar]
- 547.Wells JC, Haroun D, Levene D, Darch T, Williams JE, Fewtrell MS. Prenatal and postnatal programming of body composition in obese children and adolescents: evidence from anthropometry, DXA and the 4-component model. Int J Obes 35: 534–540, 2011. [DOI] [PubMed] [Google Scholar]
- 548.West DB, Boozer CN, Moody DL, Atkinson RL. Dietary obesity in nine inbred mouse strains. Am J Physiol Regul Integr Comp Physiol 262: R1025–R1032, 1992. [DOI] [PubMed] [Google Scholar]
- 549.West DB, Diaz J, Woods SC. Infant gastrostomy and chronic formula infusion as a technique to overfeed and accelerate weight gain of neonatal rats. J Nutr 112: 1339–1343, 1982. [DOI] [PubMed] [Google Scholar]
- 550.Whitaker KL, Jarvis MJ, Beeken RJ, Boniface D, Wardle J. Comparing maternal and paternal intergenerational transmission of obesity risk in a large population-based sample. Am J Clin Nutr 91: 1560–1567, 2010. [DOI] [PubMed] [Google Scholar]
- 551.Whitaker RC. Predicting preschooler obesity at birth: the role of maternal obesity in early pregnancy. Pediatrics 114: e29–36, 2004. [DOI] [PubMed] [Google Scholar]
- 552.Widdowson EM, McCance RA. The effect of finite periods of undernutrition at different ages on the composition and subsequent development of the rat. Proc R Soc Lond B Biol 158: 329–342, 1963. [DOI] [PubMed] [Google Scholar]
- 553.Widdowson EM, McCance RA. A review: new thoughts on growth. Pediatr Res 9: 154–156, 1975. [DOI] [PubMed] [Google Scholar]
- 554.Willesen MG, Kristensen P, Romer J. Co-localization of growth hormone secretagogue receptor and NPY mRNA in the arcuate nucleus of the rat. Neuroendocrinology 70: 306–316, 1999. [DOI] [PubMed] [Google Scholar]
- 555.Willett WC, Manson JE, Stampfer MJ, Colditz GA, Rosner B, Speizer FE, Hennekens CH. Weight, weight change, and coronary heart disease in women. Risk within the “normal” weight range. JAMA 273: 461–465, 1995. [DOI] [PubMed] [Google Scholar]
- 556.Wilson MR, Hughes SJ. The effect of maternal protein deficiency during pregnancy and lactation on glucose tolerance and pancreatic islet function in adult rat offspring. J Endocrinol 154: 177–185, 1997. [DOI] [PubMed] [Google Scholar]
- 557.Wing RR, Blair E, Marcus M, Epstein LH, Harvey J. Year-long weight loss treatment for obese patients with type II diabetes: does including an intermittent very-low-calorie diet improve outcome? Am J Med 97: 354–362, 1994. [DOI] [PubMed] [Google Scholar]
- 558.Wirtshafter D, Davis JD. Set points, settling points, and the control of body weight. Physiol Behav 19: 75–78, 1977. [DOI] [PubMed] [Google Scholar]
- 559.Wolff GL, Kodell RL, Moore SR, Cooney CA. Maternal epigenetics and methyl supplements affect agouti gene expression in Avy/a mice. FASEB J 12: 949–957, 1998. [PubMed] [Google Scholar]
- 560.Woods SC, Lotter EC, McKay LD, Porte D Jr. Chronic intracerebroventricular infusion of insulin reduces food intake and body weight of baboons. Nature 282: 503–505, 1979. [DOI] [PubMed] [Google Scholar]
- 561.Woods SC, Porte D Jr. Neural control of the endocrine pancreas. Physiol Rev 54: 596–619, 1974. [DOI] [PubMed] [Google Scholar]
- 562.Woods SC, Porte D Jr. The role of insulin as a satiety factor in the central nervous system. Adv Metab Disorders 10: 457–468, 1983. [DOI] [PubMed] [Google Scholar]
- 563.Wren AM, Small CJ, Abbott CR, Dhillo WS, Seal LJ, Cohen MA, Batterham RL, Taheri S, Stanley SA, Ghatei MA, Bloom SR. Ghrelin causes hyperphagia and obesity in rats. Diabetes 50: 2540–2547, 2001. [DOI] [PubMed] [Google Scholar]
- 564.Yajnik CS, Deshmukh US. Maternal nutrition, intrauterine programming and consequential risks in the offspring. Rev Endocr Metab Disorders 9: 203–211, 2008. [DOI] [PubMed] [Google Scholar]
- 565.Yamamoto H, Kishi T, Lee CE, Choi BJ, Fang H, Hollenberg AN, Drucker DJ, Elmquist JK. Glucagon-like peptide-1-responsive catecholamine neurons in the area postrema link peripheral glucagon-like peptide-1 with central autonomic control sites. J Neurosci 23: 2939–2946, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 566.Yamamoto H, Lee CE, Marcus JN, Williams TD, Overton JM, Lopez ME, Hollenberg AN, Baggio L, Saper CB, Drucker DJ, Elmquist JK. Glucagon-like peptide-1 receptor stimulation increases blood pressure and heart rate and activates autonomic regulatory neurons. J Clin Invest 110: 43–52, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 567.Yan X, Huang Y, Zhao JX, Long NM, Uthlaut AB, Zhu MJ, Ford SP, Nathanielsz PW, Du M. Maternal obesity-impaired insulin signaling in sheep and induced lipid accumulation and fibrosis in skeletal muscle of offspring. Biol Reprod 85: 172–178, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 568.Yang BT, Dayeh TA, Kirkpatrick CL, Taneera J, Kumar R, Groop L, Wollheim CB, Nitert MD, Ling C. Insulin promoter DNA methylation correlates negatively with insulin gene expression and positively with HbA(1c) levels in human pancreatic islets. Diabetologia 54: 360–367, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 569.Yeo GS, Farooqi IS, Aminian S, Halsall DJ, Stanhope RG, O'Rahilly S. A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nature Genet 20: 111–112, 1998. [DOI] [PubMed] [Google Scholar]
- 570.Young T, Skatrud J, Peppard PE. Risk factors for obstructive sleep apnea in adults. JAMA 291: 2013–2016, 2004. [DOI] [PubMed] [Google Scholar]
- 571.Younossi ZM, Stepanova M, Afendy M, Fang Y, Younossi Y, Mir H, Srishord M. Changes in the prevalence of the most common causes of chronic liver diseases in the United States from 1988 to 2008. Clin Gastroenterol Hepatol 9: 524–530, 2011. [DOI] [PubMed] [Google Scholar]
- 572.Yura S, Itoh H, Sagawa N, Yamamoto H, Masuzaki H, Nakao K, Kawamura M, Takemura M, Kakui K, Ogawa Y, Fujii S. Role of premature leptin surge in obesity resulting from intrauterine undernutrition. Cell Metab 1: 371–378, 2005. [DOI] [PubMed] [Google Scholar]
- 573.Zahniser NR, Goens MB, Hanaway PJ, Vinych JV. Characterization and regulation of insulin receptors in rat brain. J Neurochem 42: 1354–1362, 1984. [DOI] [PubMed] [Google Scholar]
- 574.Zambrano E, Martinez-Samayoa PM, Rodriguez-Gonzalez GL, Nathanielsz PW. Dietary intervention prior to pregnancy reverses metabolic programming in male offspring of obese rats. J Physiol 588: 1791–1799, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 575.Zhang W, Hu Y, Lin TR, Fan Y, Mulholland MW. Stimulation of neurogenesis in rat nucleus of the solitary tract by ghrelin. Peptides 26: 2280–2288, 2005. [DOI] [PubMed] [Google Scholar]
- 576.Zhang W, Lin TR, Hu Y, Fan Y, Zhao L, Stuenkel EL, Mulholland MW. Ghrelin stimulates neurogenesis in the dorsal motor nucleus of the vagus. J Physiol 559: 729–737, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 577.Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature 372: 425–432, 1994. [DOI] [PubMed] [Google Scholar]
- 578.Zhou L, Podolsky N, Sang Z, Ding Y, Fan X, Tong Q, Lowell B, Levin BE, McCrimmon RJ. The medial amygdalar nucleus: a novel glucose-sensing region that modulates the counterregulatory response to hypoglycemia. Diabetes 59: 2646–2652, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 579.Zhou M, Offer A, Yang G, Smith M, Hui G, Whitlock G, Collins R, Huang Z, Peto R, Chen Z. Body mass index, blood pressure, and mortality from stroke: a nationally representative prospective study of 212,000 Chinese men. Stroke 39: 753–759, 2008. [DOI] [PubMed] [Google Scholar]
- 580.Zigman JM, Jones JE, Lee CE, Saper CB, Elmquist JK. Expression of ghrelin receptor mRNA in the rat and the mouse brain. J Comp Neurol 494: 528–548, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 581.Zobel DP, Andreasen CH, Grarup N, Eiberg H, Sorensen TI, Sandbaek A, Lauritzen T, Borch-Johnsen K, Jorgensen T, Pedersen O, Hansen T. Variants near MC4R are associated with obesity and influence obesity-related quantitative traits in a population of middle-aged people: studies of 14,940 Danes. Diabetes 58: 757–764, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]