By gas chromatographic analysis, the ratio of carbon monoxide to carbon dioxide increased in exhaled breath of 2 mouse strains injected with different doses of Escherichia coli lipopolysaccharide. The ratios correlated with inflammation biomarkers and heme oxygenase-1 expression in blood.
Keywords: breath analysis, biomarker, sepsis, endotoxin, heme oxygenase
Abstract
Background
Analysis of volatile organic chemicals in breath holds promise for noninvasive diagnosis and monitoring of patients, but investigation of this in experimental mouse models has been limited. Of particular interest is endogenous production of carbon monoxide as a biomarker of inflammation and, more particularly, during sepsis.
Methods
Using a nose-only collection procedure for unanesthetized individual adult mice and sensitive gas chromatography of carbon monoxide (CO) and carbon dioxide (CO2) of sampled breath, we investigated the responses of mice to one-time injections with different doses of purified Escherichia coli lipopolysaccharide. Two strains of mice were examined: BALB/c and C3H, including an endotoxin-resistant mutant (HeJ) as well as the wild type (HOuJ).
Results
The CO to CO2 ratio increased in a dose-responsive manner within hours in treated BALC/c mice but not control mice. The CO/CO2 values declined to the range of control mice within 48–72 h after the injection of lipopolysaccharide. Breath CO/CO2 values correlated with systemic inflammation biomarkers in serum and heme oxygenase-1 gene expression in blood. C3H/HOuJ mice, but not the HeJ mice, had similar increases of the CO/CO2 ratio in response to the endotoxin.
Conclusions
Carbon monoxide concentrations in exhaled breath of at least 2 strains of mice increase in response to single injections of endotoxin. The magnitude of increase was similar to what was observed with a bacteremia model. These findings with an experimental model provide a rationale for further studies of normalized CO concentrations in human breath as an informative biomarker for staging and monitoring of sepsis.
Sepsis, a systemic inflammatory response to infection [1], merits attention as an application for breath analysis. Mortality from severe sepsis still exceeds 25% [2], and the nationwide incidence remains steady [3]. As an aid for diagnosis and management of sepsis, several biomarkers are available [4], including procalcitonin [5]; however, these assays require a blood sample, and hours may elapse before results are known. When a patient's status is rapidly changing, and resuscitative and therapeutic maneuvers are under way, real-time data on progression or resolution of the sepsis state are of critical importance [3]. Breath analysis is attractive for this aim, because it is noninvasive and allows for repeated measurement at the bedside, with little biohazard risk to health-care workers [6].
To this end, we applied the analytical capabilities of atmospheric chemistry for detection and quantitation of volatile organic chemicals in the breath to an infection experimental model in mice [7]. With only approximately 0.05% the mass of an adult human, mice pose a technical challenge for breath analysis. But this was achieved with individual, unanesthetized animals and nose-only sampling lasting only a few minutes. This previous study revealed that carbon monoxide (CO) concentrations, normalized for carbon dioxide (CO2) content, in the exhaled breath of infected mice rose with the bacterial burden in the blood and then fell within a few hours of effective antibiotic therapy. CO concentrations in the breath correlated with levels in the blood of heme oxygenase-1, whose enzymatic action on heme is the principal source of endogenously produced CO [8].
Increased heme oxygenase-1 expression in response to sepsis, oxidative stress, and inflammation has been documented in human patients (reviewed in [9]) and in experimental models of infection [10]. But the parameter of CO in the breath had been little studied in this respect in laboratory models. Here we report our findings with a single dose of lipopolysaccharide (LPS) in 2 different strains of immunocompetent mice.
METHODS
Reagents
Escherichia coli 0111:B4 LPS, which had been phenol-extracted and chromatographically purified by gel filtration and then passed through 0.2-µm pore-sized membrane as a 1.0-mg/mL aqueous solution, was from Sigma-Aldrich (catalog L5293). The LPS solution or water was further diluted for use in phosphate-buffered saline (pH 7.4) solution in endotoxin-free Milli-Q (Millipore) water.
Mouse Experiments
The protocol was approved by the Institutional Animal Care and Utilization Committee of the University of California, Irvine. Eight- to 10-week-old male BALB/cJ, C3H/HeJ (Tlr4Lps-d), and C3H/HeOuJ mice were obtained from the Jackson Laboratory (Bar Harbor, Maine). Mice were housed in isolator cages, kept on a 12-hour light–dark cycle, and provided with autoclaved bedding, water, and food. Solutions in volumes of 250 µL were injected intraperitoneally. During terminal anesthesia, blood, spleens, and lungs were obtained. Spleens were weighed, and lungs fixed in 10% buffered formalin were processed for histopathology at the Comparative Pathology Laboratory at the University of California, Davis. Plasma samples were subjected to bead-based immunoassays at Myriad RBM (Austin, Texas) for the 59 analytes of the RodentMAP version 2.0 panel. Quantitative reverse transcription polymerase chain reaction for the mouse heme oxygenase-1 gene (HMOX1) and β-actin transcripts was carried out as described on total RNA extracted from whole blood [7]. Standards were clones of the targets in a plasmid vector. HMOX1 messenger RNA (mRNA) copies were normalized per 1000 copies of β-actin mRNA.
Collection and Analysis of Exhaled Breath
Breath samples from individual lightly restrained, unanesthetized mice were obtained with a nose-only multiport collection chamber and attached high-efficiency particular air filter and electropolished stainless steel canister as described previously [7, 11, 12]. Mice were first accommodated over 4 days of training to restraint without distress or interference with respiratory effort. Mice were observed throughout the sampling for signs of struggling or discomfort. Connected to the inlet of the chamber by stainless steel flex tubing was a pressurized cylinder of ultrapure air, which was collected at 10 000 feet elevation at the University of California's Crooked Creek White Mountain Research Center. As reported, the collected samples were effectively diluted approximately 10:1 from flushing with the ultrapure air [7]. The duration of the sampling for each mouse was between 3.8 and 4.0 minutes. Blanks for collection periods were taken from room air, which was filtered, constantly kept at 22°C, and in a smoking-free facility. The processing of the breath samples for determination of CO and CO2 by a Hewlett-Packard 5890 gas chromatograph equipped with a flame ionization detector and thermal conductivity detector was carried out as described previously [7]. For CO, accuracy and precision were 1% and 2 parts per billion by volume, respectively; corresponding values for CO2 were 1% and 3 parts per million by volume. CO concentrations were normalized by dividing the total CO in parts per billion by volume by the integer value for the percentage of CO2 content and representing this unitless value by the term “CO/CO2.” Change from the pretreatment breath sample in a subsequent sample for a given mouse was expressed as “ΔCO/CO2”. For means and differences, 95% confidence intervals are given in parentheses. Parametric (t test and analysis of variance) and nonparametric (Kruskal–Wallis) tests for continuous data were 2-tailed and performed with SYSTAT software, version 13.
RESULTS
We first examined selected host responses to the doses of LPS for the experiment. Adult BALB/c mice were injected intraperitoneally with 50 µg or 250 µg of E coli LPS or water at hour 0. Individual mice were bled and euthanized at 4 or 24 hours after injection, and serum samples were analyzed by immunoassays for selected biomarkers of inflammation and innate immunity (Supplementary Table 1). Two mice injected with water alone had interleukin 6 (IL-6) levels of 5.2 pg/mL and ≤3.7 pg/mL after 24 hours; corresponding interleukin 10 (IL-10) levels were below the lower limit of quantitation for the assay. In contrast, at 4 hours after injection, the IL-6 levels were 2350 pg/mL for 50 µg LPS and 10 400 pg/mL for a 250-µg dose; corresponding IL-10 levels were 2040 pg/mL and 4440 pg/mL. At 24 hours the IL-6 level was 253 pg/mL and the IL-10 level was 1030 pg/mL in a mouse that received 250 µg LPS. Other elevated mouse serum proteins, cytokines, or chemokines after the LPS injections included CD40 ligand (CD154), tumor necrosis factor α, macrophage inhibitory protein 1β (CCL4), monocyte chemotactic protein 1 (CCL2), and vascular endothelial growth factor A (Table 1). In a separate experiment under the same conditions, the ratios of HMOX1 transcripts to 1000 actin mRNAs in whole blood were 3.4 in an untreated animal, and 21.3 at 4 hours after a 50-µg dose of LPS, 25.7 at 4 hours after a 250-µg dose, and 25.5 at 24 hours after a 250-µg dose in other individual mice. The magnitude of the elevation of heme oxygenase-1 gene expression in the blood was similar to what was observed with a bacteremia model [7].
Table 1.
Selected Serum Biomarkers of BALB/c Mice Injected With Lipopolysaccharide or Buffer at Hour 0
| LPS Dose, µg | Time, h | CD40 Ligand, ng [0.43] | Interleukin 6, pg [3.8] | Interleukin 10, pg [220] | Tumor Necrosis Factor α, pg [110] | Macrphage Inhibitory Protein 1β, pg [53] | Monocyte Chemotactic Protein 1, pg [8] | Vascular Endothelial Growth Factor A, pg [158] | Haptoglobin, µg [121] |
|---|---|---|---|---|---|---|---|---|---|
| 0 | 24 | 3.5 | 5.2 | ≤219 | ≤109 | 104 | 245 | 644 | 128 |
| 0 | 24 | 1.7 | ≤3.7 | ≤219 | ≤109 | 104 | 210 | 438 | 132 |
| 50 | 4 | 8.7 | 2350 | 2040 | 330 | 48 800 | 6900 | 3720 | 136 |
| 250 | 4 | 10.4 | 10 400 | 4440 | 360 | 59 000 | 6100 | 3400 | 134 |
| 250 | 24 | 21.5 | 253 | 1030 | 160 | 999 | 5480 | 1850 | 170 |
Reported lower limit of quantitation of each analysand at the laboratory (Myriad RBM) for that run is shown in brackets.
Abbreviation: LPS, lipopolysaccharide.
These results were evidence of a systemic inflammatory response to the LPS and suggested that an experimental time frame of approximately 3 days was suitable for a single dose. For the breath analysis experiment itself, male adult BALB/c mice, weighing 22–27 g, were accommodated to the breath collection apparatus. Then breath samples were noninvasively collected on each of 2 days at hours −47 and −23 as a baseline before intraperitoneal injection of LPS at hour 0 in a single dose of 0 (n = 3), 50 (n = 3), or 250 µg (n = 4). Breath samples were collected at hours 4, 10, 25, 30, 49, 73, and 121.
The ΔCO/CO2 values increased over the first 24 after LPS injections and then declined over the next 2 days (Figure 1). Mice given 250 µg had higher and later peak values than did mice with the 50-µg dose, and the decline was slower. A 250-µg dose mouse that had highest change in value from baseline, 816, died before the hour 49 collection. There was trend toward lower CO/CO2 values for the control mice over the course of the experiment, possibly attributable to further accommodation to collection conditions and diminishing stress [9]. For the collections at hours 10, 25, and 30, after control mouse values had already declined from baseline measures, there were significant dose-response differences between groups in absolute CO/CO2 values (Figure 1, inset).
Figure 1.
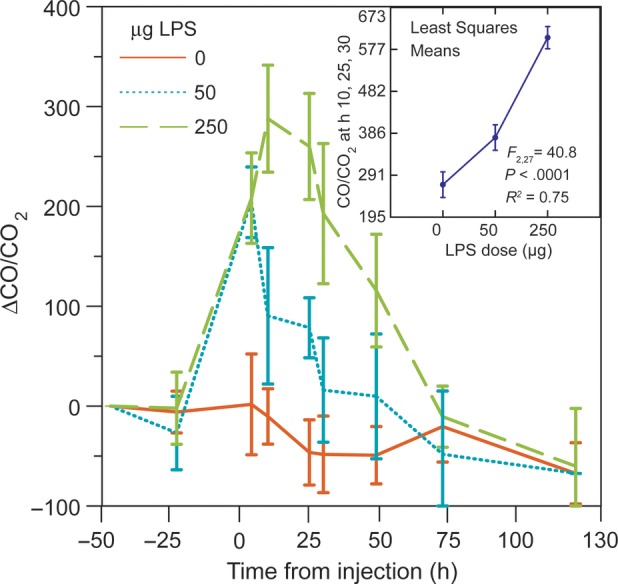
Mean change (Δ) in carbon monoxide (CO) to carbon dioxide (CO2) ratio in separate collections of exhaled breath of individual BALB/c mice injected with 250 µg (n = 4), 50 µg (n = 3), or no (n = 3) Escherichia coli lipopolysaccharide (LPS) at hour 0. The error bars are 95% confidence intervals (CIs). There were no samples at hours 73 and 121 from a mouse that received the 250-µg dose and died after the hour 49 collection. The inset in the upper right corner shows the analysis of variance least square means (with 95% CIs) for combined CO/CO2 values taken at 10, 25, and 30 hours after injection of LPS from the 3 groups of mice. The F statistic and coefficient of determination (R2) are shown.
This experiment confirmed the importance of normalizing CO for the CO2 concentration in samples. With just CO concentrations in the collected samples, no differences between the treated mice and controls would have been noted. For the collections at 10, 25, and 30 h after injection of 250 µg LPS in 4 mice or water in 3 mice, the mean CO concentrations were 108 ((95% confidence interval [CI]) 105–112) for control mice and 110 ((95% [CI]) 107–113) for treated mice (t and Kruskal–Wallis tests, P = .4–.5). But there was a marked difference between these groups in the percentage of CO2 content, with mean values of 0.378 ((95% [CI]) 0.339–0.416) for controls and 0.195 ((95% [CI]) 0.173–0.217) for treated mice (P < .0001). This is accounted for the approximately 2-fold difference between groups in the CO/CO2 ratio: 292 ((95% [CI]) 264–319) for controls and 583 ((95% [CI]) 522–644) for treated mice (P < .0001). Decreased muscular activity and/or metabolism may have accounted for lower CO2 concentration in samples from LPS-treated mice. Injected endotoxin suppressed locomotor activity and food intake in studies of mice [13, 14].
We next examined the specificity of the CO response to endotoxin by studying C3H/HeJ (Tlr4Lps-d) mice, which, as the consequence of a spontaneous mutation in the Toll-like receptor 4 gene, are more resistant to endotoxin. These were compared with mice, which are congenic but wild-type with respect to endotoxin sensitivity. We first assessed the effect on HMOX1 expression in the blood of individual mice at 4, 18, and 42 postinjection of 0, 50, 100, or 250 µg LPS. The responses of the C3H lineage mice were similar to what was observed with BALB/c lineage mice (Figure 2). In the subsequent experiment, C3H/HeOuJ or C3H/HeJ mice received LPS at 10 µg/g of body weight (5 HeJ and 6 HeOuJ mice) or water alone (3 HeOuJ mice). Breaths were collected before the injections and then at 4 hours and 24 hours postinjection, followed by euthanasia. The mean and variances of preinjection body weights of the groups were not significantly different (Table 2).
Figure 2.
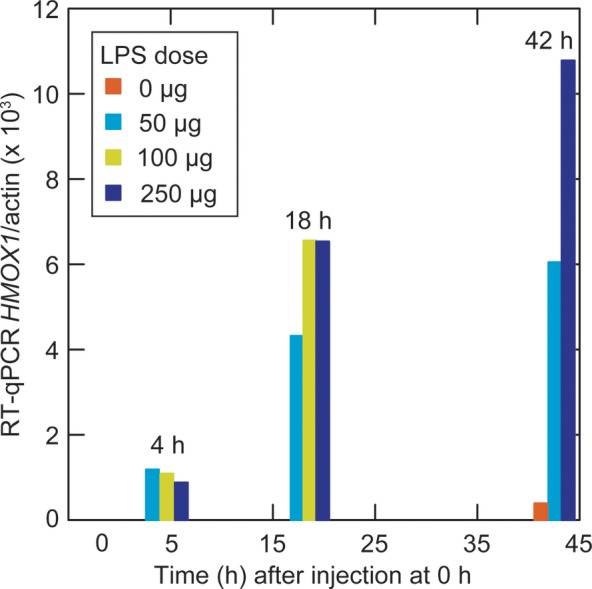
Reverse transcription quantitative polymerase chain reaction (RT-qPCR) of heme oxygenase-1 gene (HMOX1) transcripts in the whole blood of C3H/HeOuJ (wild-type) mice injected with lipopolysaccharide (LPS) at hour 0. The results were normalized per 1000 β-actin transcripts determined by RT-qPCR. Each vertical bar represents a value from an individual mouse at that time of euthanasia and blood collection.
Table 2.
Characteristics of C3H Strain Mice Treated With Lipopolysaccharide at Hour 0
| Group | C3H Strain/Treatment | Body Weight, g, at 0 h | Hematocrit, %, at 24 h | Spleen/Body Weight, %, at 24 h | ΔCO/CO2 at 4 h | ΔCO/CO2 at 24 h |
|---|---|---|---|---|---|---|
| A (n = 3) | HeOuJ/Buffer | 26.4 (25.5–27.4)a | 49 (47–50) | 0.34 (0.29–0.38) | −41 (−61 to −21) | 4 (−41 to 45) |
| B (n = 5) | HeJ/LPS | 25.4 (24.1–26.7) | 48 (48–49) | 0.54 (0.51–0.57) | −24 (−53 to 5) | −44 (−23 to −65) |
| C (n = 6) | HeOuJ/LPS | 25.8 (24.7–26.8) | 50 (49–52) | 0.36 (0.33–0.39) | 60 (51–70) | 120 (85–155) |
| Mean difference, B vs C | 0.5 (−1.3 to 2.3) | 1.9 (0.3–3.6) | −0.18 (−0.14 to −0.22) | 84 (52–116) | 164 (115–213) | |
| P valueb | .57 | .03 | <.0001 | <.0001 | <.0001 |
Abbreviations: ΔCO/CO2, mean change in carbon monoxide to carbon dioxide ratio; HeJ, endotoxin-resistant mutant; HOuJ, wild type; LPS, lipopolysaccharide.
a Mean difference (95% confidence interval).
b Two-tailed t test.
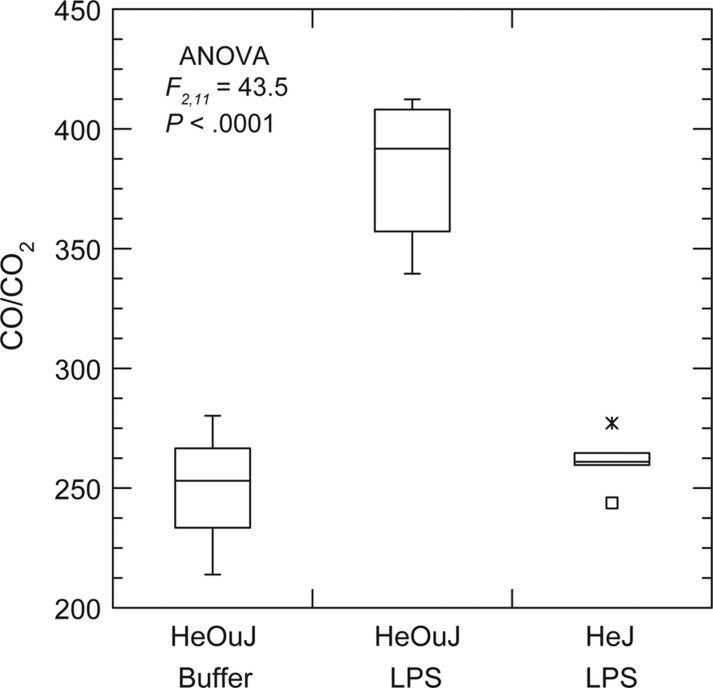
Figure 3 shows the CO/CO2 ratios for the hour 24 collection for the groups. The endotoxin-resistant mice were indistinguishable on this basis from the wild-type mice injected with buffer alone. These groups also significantly differed in ΔCO/CO2 from the hour 0 values at hours 4 and 24 (Table 2). As expected, the wild-type mice injected with LPS had at the highest values for HMOX1 expression in the blood at hour 24 among the groups, but there was also moderate elevation for this parameter among the endotoxin-resistant mice as well. The mean HMOX1 copies were 7.1 ((95% [CI]) 5.1–9.1) for HeOuJ mice treated with water, 21.4 ((95% [CI]) 17.7–25.2) for HeJ mice treated with LPS, and 40.0 ((95% [CI]) 28.8–51.1) for HeOuJ treated with LPS. These results indicated that the effect of LPS on CO production was at least partially mediated through Toll-like receptor signaling and downstream pathways. They also confirmed the correlation between the heme oxygenase-1 activity in the blood and CO production, but also that transcription of HMOX1 may moderately increase in the blood without a concomitant increase in CO in certain backgrounds.
Figure 3.
Carbon monoxide (CO) to carbon dioxide (CO2) ratios in the exhaled breath of C3H/HeOuJ (wild-type) and C3H/HeJ (endotoxin-resistant mutant) strains of mice 24 h after injection with 10 µg lipopolysaccharide (LPS) per gram of body weight or buffer. The graphs are box-whisker plots in which each box indicates the first and third quartiles, and the line inside the box is the median. The 1.5 × interquartile range is indicated by the vertical line (whiskers) bisecting the box, and the value outside this range is indicated by an asterisk. The sample sizes were 3 for HeOuJ mice injected with buffer, 5 for HeJ injected with LPS, and 6 for HeOuJ injected with LPS. The F statistic of the analysis of variance (ANOVA) is shown.
Another possible explanation for increased heme oxygenase-1 activity is hemolysis and resultant heme burden [9]. However, there was no increase in haptoglobin after LPS treatment of the BALB/c mice (Table 1), and the treated HeOuJ mice did not have reduced hematocrit levels or larger spleens compared with treated HeJ mice (Table 2). Finally, histopathologic evaluation of hematoxylin and eosin-stained sections of the lungs showed no difference between HeOuJ mice who had received LPS (n = 3) or water (n = 3), an indication that increased exhaled CO was not attributable to pulmonary pathology [15].
DISCUSSION
A key feature of this experimental model is the collection of breath samples from awake individual mice over a period of a few minutes. The final sample comprises what the mouse has inhaled and exhaled during the period of collection. Because it is the product of several breaths and not a single breath, we need to adjust for the amount of exchange of oxygen and carbon dioxide between the mouse and the environment, that is, respiration, during the time period of collection. The total accounting of respiration over a given period of time depends on basal metabolism, body temperature, and physical activity. The amount of respiration over a given time can be estimated by the difference between the input air and collected sample for either oxygen or carbon dioxide. Unless the organism is moribund, these values should reciprocally correlate over a broad range. A further analysis of data collected from mice with and without bacteremia in our previous study [7] showed that oxygen uptake and carbon dioxide production are highly correlated for the set of both infected and uninfected animals (Supplementary Material). This indicated that CO2 concentrations in the sample are suitable as gauges of respiration during the collection period. Because there is a substantially greater differential for CO2 than for O2 between concentrations in ambient air and exhaled breath, the former parameter is preferable for analytical and interpretation purposes.
While the endotoxin-treated and untreated mice exhaled similar amounts of CO during the collection in both this and the previous study [7], the endotoxin-treated and infected mice exhaled less CO2 during the same collection than did the control mice. When the differences in respiration between the mice are taken into account by normalizing by CO2 concentration, then the elevated amount of CO in the exhaled breath becomes apparent and significant. An example of a comparable clinical situation is the collection of exhaled breath over a period of time from a patient on a ventilator. Normalization of the CO for CO2 output may be as relevant for potential medical applications as it is for this experimental animal model. If the CO2 concentration was simply a stand-in for CO measurement under all conditions, then there would not be the observed divergences in that relationship for endotoxin-treated mice (Figure 1) or for infected mice [7].
We did not measure in breath the concentration of nitric oxide (NO), another possible biomarker for inflammation. In a study of mice treated with the same LPS and in the same dosage, NO in the exhaled breath rose approximately 30% by 10 hours after injection [16]. In the present and previous study [7], CO/CO2 values were about 200% of the controls’ values at a similar time interval. In comparison to CO, NO is reactive in the presence of oxygen and has a short half-life in the blood [17]. Nitric oxide measurement is suited for assessments of inflammation of the respiratory tract [18], but may be less informative when the source of the gas is in other organs or the blood itself.
The findings extend the experimental model circumstances in which the CO to CO2 ratio in the breath of mice is elevated. They indicate that this is attributable to the host's inflammatory response and not directly to a microbial pathogen. Whether enhanced CO production is for good or ill remains to be determined, but the results encourage further investigation of CO in the breath as a noninvasively sampled analysand that potentially could be measured in real time at the bedside. Numerous handheld CO detectors are available for such purposes as detection of hazardous levels of the gas in the environment or for monitoring adherence to smoking cessation programs. However, these devices are limited in their performances to minimums in the low parts per million range [19], whereas endogenously produced CO yields exhaled breath concentrations in the mid–parts per billion range. In the present study, CO was measured with gas chromatography instrumentation designed for detection of volatile organic chemicals in concentrations down to parts per trillion. For possible medical applications, such instrumentation would be impractical and cost-prohibitive. For this, other technologies such as infrared laser spectroscopy [20] can be adapted for this purpose.
Supplementary Material
Supplementary material is available online at Open Forum Infectious Diseases (http://OpenForumInfectiousDiseases.oxfordjournals.org/).
Notes
Acknowledgments. We thank Pamela Foster for administrative support.
Financial support. This work was supported by the National Institutes of Health (grant number AI-065359).
Potential conflicts of interests. All authors: No potential conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Bosmann M, Ward PA. The inflammatory response in sepsis. Trends Immunol. 2013;34:129–36. doi: 10.1016/j.it.2012.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Stevenson EK, Rubenstein AR, Radin GT, et al. Two decades of mortality trends among patients with severe sepsis: a comparative meta-analysis. Crit Care Med. 2013 doi: 10.1097/CCM.0000000000000026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Angus DC, van der Poll T. Severe sepsis and septic shock. New Engl J Med. 2013;369:840–51. doi: 10.1056/NEJMra1208623. [DOI] [PubMed] [Google Scholar]
- 4.Chan T, Gu F. Early diagnosis of sepsis using serum biomarkers. Expert Rev Mol Diagnostics. 2011;11:487–96. doi: 10.1586/erm.11.26. [DOI] [PubMed] [Google Scholar]
- 5.Mitsuma SF, Mansour MK, Dekker JP, et al. Promising new assays and technologies for the diagnosis and management of infectious diseases. Clin Infect Dis. 2013;56:996–1002. doi: 10.1093/cid/cis1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cikach FS, Dweik RA. Cardiovascular biomarkers in exhaled breath. Prog Cardiovas Dis. 2012;55:34–43. doi: 10.1016/j.pcad.2012.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barbour AG, Hirsch CM, Ghalyanchi Langeroudi A, et al. Elevated carbon monoxide in the exhaled breath of mice during a systemic bacterial infection. PLoS One. 2013;8:e69802. doi: 10.1371/journal.pone.0069802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ryter S, Alam J, Choi A. Heme oxygenase-1/carbon monoxide: from basic science to therapeutic applications. Physiol Rev. 2006;86:583–650. doi: 10.1152/physrev.00011.2005. [DOI] [PubMed] [Google Scholar]
- 9.Wegiel B, Hanto DW, Otterbein LE. The social network of carbon monoxide in medicine. Trends Mol Med. 2013;19:3–11. doi: 10.1016/j.molmed.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chung SW, Liu X, Macias AA, et al. Heme oxygenase-1-derived carbon monoxide enhances the host defense response to microbial sepsis in mice. J Clin Invest. 2008;118:239–47. doi: 10.1172/JCI32730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Colman JJ, Swanson AL, Meinardi S, et al. Description of the analysis of a wide range of volatile organic compounds in whole air samples collected during PEM-tropics A and B. Anal Chem. 2001;73:3723–31. doi: 10.1021/ac010027g. [DOI] [PubMed] [Google Scholar]
- 12.Novak BJ, Blake DR, Meinardi S, et al. Exhaled methyl nitrate as a noninvasive marker of hyperglycemia in type 1 diabetes. Proc Natl Acad Sci U S A. 2007;104:15613–8. doi: 10.1073/pnas.0706533104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wolfe RR, Burke JF. Glucose and lactate metabolism in experimental septic shock. Am J Physiol. 1978;235:R219–27. doi: 10.1152/ajpregu.1978.235.5.R219. [DOI] [PubMed] [Google Scholar]
- 14.Kozak W, Conn CA, Kluger MJ. Lipopolysaccharide induces fever and depresses locomotor activity in unrestrained mice. Am J Physiol. 1994;266:R125–35. doi: 10.1152/ajpregu.1994.266.1.R125. [DOI] [PubMed] [Google Scholar]
- 15.Biernacki WA, Kharitonov SA, Barnes PJ. Exhaled carbon monoxide in patients with lower respiratory tract infection. Respir Med. 2001;95:1003–5. doi: 10.1053/rmed.2001.1196. [DOI] [PubMed] [Google Scholar]
- 16.Weicker S, Karachi TA, Scott JA, et al. Noninvasive measurement of exhaled nitric oxide in a spontaneously breathing mouse. Am J Respir Crit Care Med. 2001;163:1113–6. doi: 10.1164/ajrccm.163.5.2007025. [DOI] [PubMed] [Google Scholar]
- 17.Beckman JS, Koppenol WH. Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and the ugly. Am J Physiol. 1996;40:C1424. doi: 10.1152/ajpcell.1996.271.5.C1424. [DOI] [PubMed] [Google Scholar]
- 18.Dweik RA, Boggs PB, Erzurum SC, et al. An official ATS clinical practice guideline: interpretation of exhaled nitric oxide levels (FENO) for clinical applications. Am J Respir Crit Care Med. 2011;184:602–15. doi: 10.1164/rccm.9120-11ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Javors MA, Hatch JP, Lamb RJ. Cut-off levels for breath carbon monoxide as a marker for cigarette smoking. Addiction. 2005;100:159–67. doi: 10.1111/j.1360-0443.2004.00957.x. [DOI] [PubMed] [Google Scholar]
- 20.Shorter JH, Nelson DD, McManus JB, et al. Clinical study of multiple breath biomarkers of asthma and COPD (NO, CO(2), CO and N(2)O) by infrared laser spectroscopy. J Breath Res. 2011;5:037108. doi: 10.1088/1752-7155/5/3/037108. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.