Abstract
Background/Aims
Colonoscopic polypectomy is highly efficient in preventing colorectal cancer, but polyps may not always be completely removed. Improved knowledge of the risk factors for incomplete polyp resection after polypectomy may decrease the cancer risk and additional costs. The aim of this study was to investigate the conditions that can cause incomplete polyp resection (IPR) after colonoscopic polypectomy.
Methods
A total of 12,970 polyps that were removed by colonoscopic polypectomy were investigated. Among them, we identified 228 cases with a positive resection margin and 228 controls with a clear resection margin that were matched for age, gender, and polyp size. We investigated the location, morphology, and histological type of the polyps and evaluated the skills of the endoscopist and assisting nurse.
Results
Multivariate analysis revealed that the polyps, which were located in the proximal part of the colon and rectum, were at significant risk of IPR. Histologically, an advanced polyp and an inexperienced assistant were also independent risk factors for IPR.
Conclusions
Polypectomy should be performed more carefully for polyps suspected to be cancerous and polyps located in the proximal part of the colon or rectum. A systematic training program for inexperienced assistants may be needed to decrease the risk of IPR.
Keywords: Colonic polyps, Colonoscopic polypectomy, Adenomatous polyps, Interval colorectal cancer, Polypectomy
INTRODUCTION
Colorectal adenomas are very common and are detected in about 20% to 40% of screening colonoscopies.1,2 Endoscopic resection of colorectal adenomas with snare polypectomy is commonly performed and prevents death from colorectal cancer.3–5 Sometimes, colonoscopic polypectomy can cause complications, such as perforation, hemorrhage, and postpolypectomy syndrome. However complication rates are acceptable and serious complications seldom develop.6,7
Although colonoscopic polypectomy is highly efficient in preventing colorectal cancer, the risk of interval cancer after polypectomy still exists and one of the main reasons is incomplete polyp resection (IPR).8–10 Little is known about the risk factors that cause IPR after colonoscopic polypectomy. A recent study showed that the incomplete resection rate is affected by the polyp size, sessile serrated histology and the workmanship of the endoscopist.11 However, the study has some problems, such as the small size of the sample and experimenter bias. In addition, we wanted to know whether the histological differences of the resected polyps affected the risk of IPR under the condition of same size of the polyps. Therefore, we decided to conduct this retrospective study. The aim of this study is to evaluate the risk factors for IPR after colonoscopic polypectomy and ultimately to increase the complete polyp resection rate by preventing patient- or procedure-related risk factors.
MATERIALS AND METHODS
1. Patients and study design
A retrospective case-control study was conducted to analyze the risk factors for IPR during colonoscopic polypectomy. We reviewed the records of patients who were referred for colonic polypectomy at Konkuk University Hospital in Seoul, Korea, during a 7-year period from August 2005 to December 2012. For patients with multiple polyps, each of the polyps that were removed by snare polypectomy with submucosal injection was sorted into separate cases. Likewise, for patients with multiple episodes of colonoscopy for polypectomy, each of the polyps was removed by snare polypectomy and also classified into separate cases. We were then able to obtain information about the patient and procedure through a review of the chart, histologic results, and endoscopic images. This study was approved by the Institutional Review Board at our institution. All patients provided written consent to undergo colonoscopic polypectomy and were informed of the risks and potential benefits of the procedures.
2. Selection of cases
To be eligible for inclusion, polyps had to be 5 to 20 mm in diameter, except for Ip polyps. Ip polyps that were from 5 to 25 mm in size were included. To estimate the size of the polyp, we used the diameter measured by the endoscopist during the endoscopy rather than the diameter of the specimen measured by the pathologist. Immediate pinning and fixing of the specimen helped to preserve the tissue size but pinning was not performed routinely after conventional snare polypectomy. Furthermore, the size of the polyp can be measured smaller than its actual size in positive resection margin groups due to the remaining polyp. Therefore, the diameter of the specimen measured by the pathologist was likely to be inaccurate. Depending on the histological result, adenoma and carcinoma were included and others, such as hyperplastic polyp, neuroendocrine tumor, inflammatory polyp, and normal mucosa, were excluded. We additionally excluded polyps resected by other methods including endoscopic submucosal dissection (ESD), endoscopic mucosal resection (EMR)-precutting, hot biopsy, and cold biopsy. If the medical records were inadequate, the polyp was excluded.
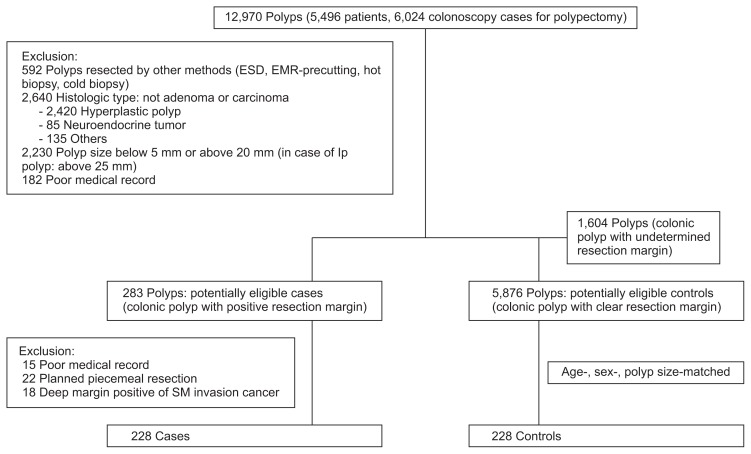
According to the completeness of the polyp resection, each polyp was categorized into the positive margin group, the clear resection margin group, and the undetermined margin group (Fig. 1). Among the polyps, we listed the positive margin group as potentially eligible “case.” The completeness of resection was judged by review of the formal report of the histologic results. When both the lateral and deep margins were free of tumor cells, it was classified into the clear resection margin group. When any of the lateral and deep margins was involved with tumor cells, it was included in the positive margin group. If it was unclear whether resection margin was involved, it was classified into the undetermined margin group.
Fig. 1.
Selection of cases and control subjects. A total of 12,970 polyps were investigated. The polyps were excluded according to the size of the polyp, histologic type, and procedural method used. If the medical records were inadequate, the case was excluded. Finally, we identified 228 cases with a positive resection margin using a validated algorithm and 228 controls with a clear resection margin, which were matched for age, sex, and polyp size. Some cases and controls met more than one exclusion criterion.
ESD, endoscopic submucosal dissection; EMR, endoscopic mucosal resection; SM, submucosa.
We investigated more detailed medical records in each of the “cases” and applied exclusion criteria once again. In addition to the criteria above, polyps with a positive resection margin arising from the deep margin that was involved in the submucosal invasion cancer and margin involvement caused by planed piecemeal resection without adequate pinning and fixing of the resected specimen were excluded.
3. Selection of controls
We selected a “control subject” for each “case” from the clear resection margin group matched for age, sex, and polyp size. When there were several candidates for the “control group” in the population, a randomized number table was used for randomization. The table was generated by the use of Excel 2010 (Microsoft Corp., Redmond, WA, USA) and operated by a researcher who was not involved in the study. The subjects included in the “case group” were not eligible to undergo resampling as “controls.” If the age, sex, and polyp size of the “controls” were not entirely compatible with those of the “cases,” we selected the most similar aged and the same-sex individual who had the same sized polyp.
4. Risk factors for incomplete polyp resection
The probable risk factors for IPR and their categorizations were as follows (Table 1): (1) morphology of the polyp, (2) location of the polyp in the colon, (3) histologic type of the polyp, (4) individual workmanship and work experience of the endoscopist, and (5) individual workmanship and work experience of the assistant. These risk factors were divided into two categories: patient-related risk factors (1 to 3) and procedure-related risk factors (4 to 5).
Table 1.
Baseline Polyp Characteristics
| Characteristic | Case group (n=228) | Control group (n=228) |
|---|---|---|
| Age, yr* | 63.19±11.39 | 63.06±11.17 |
| ≤40 | 10 (4.39) | 10 (4.39) |
| 41–60 | 72 (31.58) | 72 (31.58) |
| 61–80 | 132 (57.89) | 133 (58.33) |
| ≥81 | 14 (6.14) | 13 (5.70) |
| Female sex* | 64 (28.07) | 64 (28.07) |
| Size, mm* | 10±4.33 | 10±4.33 |
| 5–7 | 71 (31.14) | 71 (31.14) |
| 8–9 | 34 (14.91) | 34 (14.91) |
| 10–14 | 76 (33.33) | 76 (33.33) |
| 15–20 | 47 (20.61) | 47 (20.61) |
| Location | ||
| IC valve | 2 | 2 |
| Cecum | 15 (6.58) | 5 (2.19) |
| Ascending colon | 51 (22.37) | 41 (17.98) |
| Hepatic flexure | 12 (5.26) | 15 (6.58) |
| Transverse colon | 24 (10.53) | 49 (21.49) |
| Splenic flexure | 3 (1.32) | 1 |
| Descending colon | 21 (9.21) | 13 (5.70) |
| SD junction | 2 | 3 (1.32) |
| Sigmoid colon | 52 (22.81) | 70 (30.70) |
| RS junction | 8 (3.51) | 4 (1.75) |
| Rectum | 38 (16.67) | 25 (10.96) |
| Morphology | ||
| Ip | 30 (13.16) | 40 (17.54) |
| Isp | 75 (32.89) | 69 (30.26) |
| Is | 94 (41.22) | 95 (41.67) |
| IIa | 8 (3.51) | 11 (4.82) |
| LST-NG | 10 (4.39) | 6 (2.63) |
| LST-G | 11 (4.82) | 7 (3.07) |
| Histologic type | ||
| Tubular | 174 (76.32) | 201 (88.16) |
| Tubulovillous | 23 (10.09) | 16 (7.02) |
| Villous | 4 (1.75) | 0 |
| Tubular with HGD | 11 (4.82) | 11 (4.82) |
| Tubulovillous with HGD | 12 (5.26) | 7 (3.07) |
| Villous with HGD | 1 | 0 |
| Serrated | 9 (3.95) | 4 (1.75) |
| Cancer | 18 (7.89) | 7 (3.07) |
Data are presented as mean±SD or number (%).
Morphological polyp classification was described according to the Paris-Japanese classification.
IC valve, ileocecal valve; SD, sigmoid-descending junction; RS, recto-sigmoid junction; LST-NG, laterally spreading tumor-nongranular type; LST-G, laterally spreading tumor-granular type; HGD, high grade dysplasia.
Indicates matching variable.
The morphologic classification of the polyp was based on the Paris-Japanese classification.12,13 Nonpolypoid type polyps or mixed type polyps larger than 1 cm in diameter were delineated as laterally spreading tumor-granular (LST-G) or -nongranular (LST-NG) and the rest were classified into Ip, Isp, Is, and IIa by the morphology. Location of the polyp in the colon was classified as follows: ileocecal (IC) valve, cecum, ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid-descending (SD) junction, sigmoid colon, recto-sigmoid (RS) junction, and the rectum. The proximal colon was defined as the upper part of the ascending colon, including the IC valve, cecum, and ascending colon. The histologic type of the polyp was divided into tubular adenoma, villous adenoma, tubulovillous adenoma, serrated adenoma, and cancer. Adenoma was subdivided into low grade dysplasia and high grade dysplasia. A histologically advanced polyp was defined as a polyp which had high malignant potential, such as an adenoma with high grade dysplasia, serrated adenoma, or cancer.
Endoscopist was defined as an internal medicine physician who specialized in gastroenterology, and assistant was defined as a registered nurse who worked at the endoscopy center. An endoscopist with over 2 years’ experience and an assistant with over 1 year’s experience were classified as experts. The differences between beginner and expert were analyzed. In addition, an analysis of each endoscopist and assistant, who performed more than 30 polypectomies among all the cases and controls, was undertaken to determine whether individual differences existed. The assistants were assigned on a rotating schedule basis; therefore, endoscopists were randomly matched to their assistants.
5. Endoscopic procedure
A standard colonoscope (CF-H260; Olympus, Tokyo, Japan) was used for the procedure. The colon was cleansed with a 4-L polyethylene glycol electrolyte solution. Midazolam (Dormicum®) for conscious sedation and meperidine (Demerol®) for pain control were selectively given to patients. Polypectomy was performed in a standard fashion by one endoscopist and one assistant. The polyp was removed by oval-shaped snares (Olympus) after saline injection mixed with indigo carmine and epinephrine. There was no additional routine procedure after the polypectomy. However, if it was suspected macroscopically that the polyp had not been removed completely, we performed additional polypectomy by using a snare, biopsy forceps, and/or argon plasma coagulation (APC). If there was a possibility of postpolypectomy hemorrhage, we used hemoclipping, administered an epinephrine injection, and/or APC. The size of the polyp was estimated by the endoscopist during the endoscopy.
6. Statistical analysis
Continuous variables were summarized as the mean±standard deviation and categorical variables as the frequency (%). We used conditional logistic regression analyses to estimate the odds ratios (ORs) of risk factors that independently cause the IPR. ORs and 95% confidence intervals (CIs) were obtained by using univariate (crude) and multivariate (adjusted) models. All analyses were conducted by using SPSS for Windows versions 19.0 (IBM, Armonk, NY, USA). A p-value of less than 0.05 was considered statistically significant.
RESULTS
1. Characteristics of the polyps and patients
We identified 12,970 polyps (5,496 patients, 6,024 colonoscopy cases for polypectomy) which were removed by colonoscopic polypectomy during the study period (Fig. 1). Among them, 7,763 subjects were included and their polyps were divided into three groups: 283 polyps of the positive margin group, 5,876 polyps of the clear resection margin group, and 1,604 polyps of the undetermined margin group. Out of the positive margin group, 55 polyps were excluded for various reasons. Thus, the final analysis included 228 polyps in each of the “cases” and “controls” groups. The mean ages of the included “cases” and “controls” were 63.19±11.39 years (range, 32 to 95 years) and 63.06±11.17 years (range, 32 to 89 years), respectively (Table 1).
In both of the groups, 64 (28.07%) were females and the mean size of the polyp was 10±4.33 mm (range, 5 to 20 mm). The diameter of the polyp measured 5 to 7 mm in 71 polyps (31.14%), 8 to 9 mm in 34 (14.91%), 10 to 14 mm in 76 (33.33%), and 15 to 20 mm in 47 polyps (20.61%). In all our cases and controls, there was no Ip polyp which measured 21 to 25 mm in size.
In the case and control groups, histologically advanced polyps (adenoma with high grade dysplasia, serrated adenoma, or cancer) were found in 51 polyps (22.37%, mean size 13.37 mm) and 29 polyps (12.72%, mean size 13.38 mm), respectively. In the case group, 68 polyps (29.82%) were located in the IC valve, cecum, and ascending colon, and 46 polyps (20.18%) in the rectum and RS junction. Of all the polyps in the rectum and RS junction, 25 polyps (54.35%) were located in the valve of Houston or in the distal rectum near the anus. In the control group, 48 polyps (21.05%) were located in the IC valve, cecum, and ascending colon, and 29 polyps (12.72%) were found in the rectum and RS junction. Among the polyps in the rectum and RS junction, eight polyps (30.77%) were located near the valve of Houston or the anus.
Thirty polyps (13.16%) were classified as Ip, and 29 polyps (12.72%) were classified as a flat lesion (IIa, LST-G, or LST-NG) in the case group. And, in the control group, the morphology of polyps was classified as Ip in 40 polyps (17.54%) and a flat lesion in 24 polyps (10.53%).
2. Univariate analysis
The mean sizes of the polyps which were removed by the expert, beginner, and each individual were not any different from each other (Table 2). The polyps which were removed by a different assistant did not significantly correlate with the increase of IPR; however, the polyps which were removed by a specific endoscopist had a lower or higher risk of IPR (endoscopist B: OR, 1.875; 95% CI, 1.022 to 3.440; endoscopist F: OR, 0.429; 95% CI, 0.196 to 0.936) (Table 2). The work experience of the endoscopist was not associated with IPR. However, assistants who had less work experience were about two times more likely to incompletely remove the polyp compared with skilled assistants (OR, 2.241; 95% CI, 1.447 to 3.472). Univariate analysis revealed that polyps located in the proximal colon were at significant risk of IPR (OR, 1.625; 95% CI, 1.046 to 2.524) (Table 3). Likewise, polyps located in the rectum and RS junction had a higher risk of IPR (OR, 1.739; 95% CI, 1.041 to 2.905). Histologically advanced polyps were also another risk factor of IPR (OR, 2.158; 95% CI, 1.253 to 3.718). The morphology of the polyp was not associated with the risk of IPR (Ip, p=0.160; LST, p=0.136).
Table 2.
Work Experience of the Endoscopist and Assistant and Personal Workmanship: The Relationship of These Factors with Incomplete Polyp Resection
| Individual workmanship and career | Size of polyp, mean | Case group, no. | Control group, no. | p-value | OR (95% CI) |
|---|---|---|---|---|---|
| Endoscopist | |||||
| A | 10.45 | 28 | 20 | 0.220 | 1.471 (0.794–2.723) |
| B | 10.12 | 33 | 19 | 0.042 | 1.875 (1.022–3.440) |
| C | 12.04 | 34 | 33 | 0.893 | 1.037 (0.611–1.759) |
| D | 9.15 | 40 | 57 | 0.058 | 0.646 (0.411–1.014) |
| E | 9.17 | 21 | 14 | 0.168 | 1.636 (0.773–3.465) |
| F | 11.40 | 9 | 21 | 0.033 | 0.429 (0.196–0.936) |
| Expert | 10.17 | 178 | 177 | - | - |
| Beginner | 9.41 | 50 | 51 | 0.913 | 0.976 (0.635–1.501) |
| Assistant | |||||
| A | 10.60 | 24 | 19 | 0.425 | 1.294 (0.687–2.437) |
| B | 9.26 | 13 | 18 | 0.371 | 0.722 (0.354–1.474) |
| C | 9.84 | 24 | 20 | 0.528 | 1.222 (0.656–2.279) |
| D | 10.77 | 28 | 25 | 0.655 | 1.143 (0.636–2.053) |
| E | 9.95 | 20 | 22 | 0.732 | 0.889 (0.453–1.743) |
| F | 8.81 | 20 | 16 | 0.494 | 1.267 (0.644–2.493) |
| G | 9.90 | 20 | 30 | 0.144 | 0.643 (0.356–1.162) |
| Expert | 10.03 | 153 | 189 | - | - |
| Beginner | 9.90 | 75 | 39 | 0.0001 | 2.241 (1.447–3.472) |
OR, odds ratio; CI, confidence interval.
Table 3.
Univariate and Multivariate Conditional Logistic Regression Analysis of the Predictors of an Incomplete Polyp Resection
| Univariate analysis | Multivariate analysis* | |||
|---|---|---|---|---|
|
|
|
|||
| OR (95% CI) | p-value | Adjusted OR (95% CI) | p-value | |
| Morphology of polyp | ||||
| Ip vs non-Ip | 0.667 (0.379–1.174) | 0.160 | - | - |
| LST vs non-LST | 1.800 (0.831–3.899) | 0.136 | - | - |
| Location of polyp in the colon | ||||
| Proximal colon† vs nonproximal colon | 1.625 (1.046–2.524) | 0.031 | 1.973 (1.203–3.237) | 0.007 |
| RS junction and rectum vs the rests | 1.739 (1.041–2.905) | 0.034 | 1.873 (1.058–3.316) | 0.031 |
| Histologic type of polyp | ||||
| Advanced polyp‡ vs nonadvanced polyp | 2.158 (1.253–3.718) | 0.006 | 1.827 (1.021–3.270) | 0.042 |
| Endoscopist | ||||
| B | 1.875 (1.022–3.440) | 0.042 | 1.748 (0.910–3.358) | 0.094 |
| F | 0.429 (0.196–0.936) | 0.033 | 0.525 (0.228–1.206) | 0.129 |
| Career of assistant | ||||
| Beginner vs expert | 2.241 (1.447–3.472) | 0.0001 | 1.994 (1.259–3.157) | 0.003 |
OR, odds ratio; CI, confidence interval; LST, laterally spreading tumor; RS junction, recto-sigmoid junction.
The final regression model included the location of the polyp, histological type of the polyp, career of the assistant, and workmanship of a specific endoscopist (endoscopists B and F). Multivariate analysis was conducted after adjusting for these factors;
Ileocecal valve, cecum, and ascending colon;
Adenoma with high-grade dysplasia, serrated adenoma, and cancer.
3. Multivariate analysis
Multivariate analysis was done after adjusting for the location of the polyp, histologic type of the polyp, work experience of the assistant, and workmanship of the specific endoscopist (Table 3). The polyps located in the proximal colon (OR, 1.973; 95% CI, 1.203 to 3.237) and the rectum and RS junction (OR, 1.873; 95% CI, 1.058 to 3.316) were at significant risk of IPR, respectively. Histologically advanced polyps (OR, 1.827; 95% CI, 1.021 to 3.270) and polyps removed by assistants who lacked work experience (OR, 1.994; 95% CI, 1.259 to 3.157) were at significant risk of IPR, too (Table 3). There was no association between the workmanship of the specific endoscopist and IPR after adjusting for the variables (endoscopist B, p=0.094; endoscopist F, p=0.129).
DISCUSSION
A previous study revealed that the risk of IPR increased as polyp size increased and sessile serrated histology was a risk factor of IPR.11 However, polyp size and histologic type of the polyp might interact with each other as confounding factors, because a bigger sized polyp is likely to be a histologically advanced polyp.14,15 Therefore, we wanted to know whether the histological differences of the resected polyps affected the risk of IPR under the condition of same size of the polyps. The results of this study suggested that histologically advanced polyps, which included not only serrated adenoma but also cancer and adenoma with high grade dysplasia, were an independent risk factor of IPR. Sessile serrated polyps are similar in shape to hyperplastic polyps, and have a flat morphology and unremarkable color.1,16,17 For that reason, it is difficult to distinguish them from the normal mucosa, and maybe that is why sessile serrated polyps are a risk factor for IPR. Most colorectal cancers are known to arise from adenomatous polyps with dysplasia.14,18 Adenoma surrounding a cancerous lesion tends to be mistaken for normal mucosa because a noticeable cancerous lesion might make it difficult to recognize an adjacent adenoma. This may be a reason why cancer or adenoma with high grade dysplasia is a risk factor for IPR.
Also, location-based differences were observed. The polyps located in the proximal colon and in the rectum and RS junction were at significant risk of IPR. In several previous studies, screening colonoscopies were less effective in decreasing both the incidence and mortality of colorectal cancer in the proximal colon compared with the distal colon.19,20 The reason for this might be not only the difficulty of detection, but also the difficulty of complete resection. Polyps of the cecum or ascending colon occasionally can be hard to remove due to the difficulties in manipulating them, a large amount of stool present in these locations, and deep haustral clefts. We could not predict that the polyps located in the rectum were at significant risk of IPR. The polyp of the rectum has a low missing rate during colonoscopy because the rectal lumen is relatively large.20 However, it is often very difficult to clearly remove polyps when they are located in the valve of Houston or in the distal rectum near the anus. Therefore, we subdivided the location of the rectal polyps through the endoscopic images and found that the polyps of the case group were more frequently located near the valve of Houston or the anus. But, for the polyps which were located near the valve of Houston, we could not subdivide into frontal parts or posterior parts of the valve due to ambiguous images and the sample size was too small to analyze. We expected that Ip polyps would be removed a little more clearly and that there would be more incompletely resected lesions for LST, regardless of the size of the polyp. However, our study showed the morphologic type of the polyp was not a risk factor of IPR.
In a prior study, differences in the rate of IPR among the endoscopists were great, but the researchers of the study overlooked the role of the assistant.11 The current study showed that there was an association between the specific endoscopist and the rate of IPR; however, there was no statistical significance after adjusting for variables. Meanwhile, IPR was irrelevant to the work experience of the endoscopists, but there was a strong correlation between IPR and the work experience of the assistants. Therefore, prerequisite for reducing the risk of IPR was over 1 year’s work experience of the assistant rather than the personal ability of the assistant. Polypectomy is a procedure which is completed by cooperation between the endoscopist and assistant, and the results of this study showed that the role of the assistant was important.
If piecemeal resection is performed, pinning of the tissue is needed to evaluate the margin status, but pinning was not performed in a considerable proportion of the piecemeal resection cases. Therefore, we excluded polyps removed by planed piecemeal resection in our study. However, when an en bloc resection was tried and then the remnant polyp was removed by additional polypectomy, we did not exclude this case from the “case group” because the first attempt was regarded as a failure. In our hospital, when the polyp was difficult to remove by en bloc resection, it was removed mainly by EMR with precutting or ESD rather than by piecemeal resection. The polyps removed by EMR with precutting or ESD were also excluded from the cases and controls, because the procedures were regarded as different procedures to snare polypectomy.21
Polyps less than 5 mm in size were generally removed by cold biopsy with forceps, and polyps more than 20 mm in size were removed by using EMR with precutting or ESD method. If the size of the polyp, which is removed without precutting, is bigger than 20 mm in size, the possibility of incomplete resection may increase. Therefore, we included polyps 5 to 20 mm in diameter. But Ip polyps with a diameter between 5 and 25 mm were exceptionally included because they were more easily removed. Actually most polyps above 20 mm have been removed by EMR with precutting or ESD in our unit.
If the polyps belong to the undetermined margin group, it could be due to crush artifact, cautery artifact, and fragmentation of the specimen.22 Smaller polyps can undergo these damages more frequently and this is another reason for matching the size of the polyp between the case and control.
There may be some limitations to this study. First, pathologists are more likely to examine a specimen in detail if the histologic outcome reveals a high risk of cancer. They would cut the specimen into thinner slices and apply stricter standards to assess the margin status. This might be one of the reasons why the case group had more histologically advanced polyps than the control group. Nevertheless, regardless of the reason, it is still important that histologically advanced polyps have a high rate of IPR because the treatment could change completely depending on the histological results.14 The second limitation is as follows. As mentioned earlier, the undetermined margin group was excluded from the study because there was no clear evidence about the resection margin status. However, this can affect the results of the study because many of those polyps might have included polyps in which the resection margin was involved. To minimize such errors, we designed and conducted a case-control study.
In conclusion, this retrospective study indicates that polyps which are located in the proximal part of the colon or rectum were at significant risk of IPR. In addition, histologically advanced polyp and inexperienced assistant were also independent risk factors of IPR. According to our results, polypectomy should be performed more carefully in polyps suspected of having cancer and in polyps located in the proximal part of the colon or rectum. Furthermore, a systematic training program of the polypectomy procedure might be needed to decrease the risk of IPR by inexperienced assistants.
ACKNOWLEDGEMENTS
This paper was written as part of Konkuk University’s research support program for its faculty on sabbatical leave in 2010.
Footnotes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
REFERENCES
- 1.Liang J, Kalady MF, Appau K, Church J. Serrated polyp detection rate during screening colonoscopy. Colorectal Dis. 2012;14:1323–1327. doi: 10.1111/j.1463-1318.2012.03017.x. [DOI] [PubMed] [Google Scholar]
- 2.Dinesen L, Chua TJ, Kaffes AJ. Meta-analysis of narrow-band imaging versus conventional colonoscopy for adenoma detection. Gastrointest Endosc. 2012;75:604–611. doi: 10.1016/j.gie.2011.10.017. [DOI] [PubMed] [Google Scholar]
- 3.Zauber AG, Winawer SJ, O’Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366:687–696. doi: 10.1056/NEJMoa1100370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Winawer SJ, Zauber AG, Ho MN, et al. Prevention of colorectal cancer by colonoscopic polypectomy: The National Polyp Study Workgroup. N Engl J Med. 1993;329:1977–1981. doi: 10.1056/NEJM199312303292701. [DOI] [PubMed] [Google Scholar]
- 5.Rosa I, Fidalgo P, Soares J, et al. Adenoma incidence decreases under the effect of polypectomy. World J Gastroenterol. 2012;18:1243–1248. doi: 10.3748/wjg.v18.i11.1243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fatima H, Rex DK. Minimizing endoscopic complications: colonoscopic polypectomy. Gastrointest Endosc Clin N Am. 2007;17:145–156. doi: 10.1016/j.giec.2006.10.001. [DOI] [PubMed] [Google Scholar]
- 7.Kim JH, Lee HJ, Ahn JW, et al. Risk factors for delayed post-polypectomy hemorrhage: a case-control study. J Gastroenterol Hepatol. 2013;28:645–649. doi: 10.1111/jgh.12132. [DOI] [PubMed] [Google Scholar]
- 8.Brenner H, Chang-Claude J, Seiler CM, Hoffmeister M. Interval cancers after negative colonoscopy: population-based case-control study. Gut. 2012;61:1576–1582. doi: 10.1136/gutjnl-2011-301531. [DOI] [PubMed] [Google Scholar]
- 9.Huang Y, Gong W, Su B, Zhi F, Liu S, Jiang B. Risk and cause of interval colorectal cancer after colonoscopic polypectomy. Digestion. 2012;86:148–154. doi: 10.1159/000338680. [DOI] [PubMed] [Google Scholar]
- 10.Singh H, Nugent Z, Mahmud SM, Demers AA, Bernstein CN. Predictors of colorectal cancer after negative colonoscopy: a population-based study. Am J Gastroenterol. 2010;105:663–673. doi: 10.1038/ajg.2009.650. [DOI] [PubMed] [Google Scholar]
- 11.Pohl H, Srivastava A, Bensen SP, et al. Incomplete polyp resection during colonoscopy-results of the complete adenoma resection (CARE) study. Gastroenterology. 2013;144:74–80.e1. doi: 10.1053/j.gastro.2012.09.043. [DOI] [PubMed] [Google Scholar]
- 12.Kudo S, Lambert R, Allen JI, et al. Nonpolypoid neoplastic lesions of the colorectal mucosa. Gastrointest Endosc. 2008;68(4 Supp):S3–S47. doi: 10.1016/j.gie.2008.07.052. [DOI] [PubMed] [Google Scholar]
- 13.Endoscopic Classification Review Group. Update on the Paris classification of superficial neoplastic lesions in the digestive tract. Endoscopy. 2005;37:570–578. doi: 10.1055/s-2005-861352. [DOI] [PubMed] [Google Scholar]
- 14.Cunningham D, Atkin W, Lenz HJ, et al. Colorectal cancer. Lancet. 2010;375:1030–1047. doi: 10.1016/S0140-6736(10)60353-4. [DOI] [PubMed] [Google Scholar]
- 15.Ignjatovic A, East JE, Suzuki N, Vance M, Guenther T, Saunders BP. Optical diagnosis of small colorectal polyps at routine colonoscopy (detect InSpect ChAracterise resect and discard; DISCARD trial): a prospective cohort study. Lancet Oncol. 2009;10:1171–1178. doi: 10.1016/S1470-2045(09)70329-8. [DOI] [PubMed] [Google Scholar]
- 16.Leggett B, Whitehall V. Role of the serrated pathway in colorectal cancer pathogenesis. Gastroenterology. 2010;138:2088–2100. doi: 10.1053/j.gastro.2009.12.066. [DOI] [PubMed] [Google Scholar]
- 17.Huang CS, Farraye FA, Yang S, O’Brien MJ. The clinical significance of serrated polyps. Am J Gastroenterol. 2011;106:229–240. doi: 10.1038/ajg.2010.429. [DOI] [PubMed] [Google Scholar]
- 18.Tolliver KA, Rex DK. Colonoscopic polypectomy. Gastroenterol Clin North Am. 2008;37:229–251. doi: 10.1016/j.gtc.2007.12.009. [DOI] [PubMed] [Google Scholar]
- 19.Baxter NN, Goldwasser MA, Paszat LF, Saskin R, Urbach DR, Rabeneck L. Association of colonoscopy and death from colorectal cancer. Ann Intern Med. 2009;150:1–8. doi: 10.7326/0003-4819-150-1-200901060-00306. [DOI] [PubMed] [Google Scholar]
- 20.Brenner H, Hoffmeister M, Arndt V, Stegmaier C, Altenhofen L, Haug U. Protection from right- and left-sided colorectal neoplasms after colonoscopy: population-based study. J Natl Cancer Inst. 2010;102:89–95. doi: 10.1093/jnci/djp436. [DOI] [PubMed] [Google Scholar]
- 21.Fyock CJ, Draganov PV. Colonoscopic polypectomy and associated techniques. World J Gastroenterol. 2010;16:3630–3637. doi: 10.3748/wjg.v16.i29.3630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fry LC, Lazenby AJ, Mikolaenko I, Barranco B, Rickes S, Monkemuller K. Diagnostic quality of: polyps resected by snare polypectomy. Does the type of electrosurgical current used matter? Am J Gastroenterol. 2006;101:2123–2127. doi: 10.1111/j.1572-0241.2006.00696.x. [DOI] [PubMed] [Google Scholar]