Abstract
Objective
To investigate the characteristics of baseline body fluid content and overnight fluid shifts between non-obstructive sleep apnea (non-OSA) and obstructive sleep apnea (OSA) subjects.
Methods
A case-controlled study was performed between February 2013 and January 2014, with 36 (18 OSA and 18 non-OSA) outpatients enrolled in this study. Polysomnographic parameters and results of body fluid were compared between the two groups.
Results
There were no differences in age, weight, and body mass index (BMI) between groups. Compared with the non-OSA group, OSA group had significantly higher neck circumference (NC) and fluid volume shift in the legs. OSA patients had higher left and right leg fluid indices than non-OSA subjects. There were significant correlations between apnoea-hypopnoea index and baseline fluid indices in both legs as well as the reduction in overnight change in both legs fluid volume. The increase in NC was also significantly correlated with the reduction in overnight change in both legs fluid volume, but not with the change in head and neck fluid volume. There were significant correlations between change in NC and increased fluid shifts in head and neck volume.
Conclusions
OSA patients had a higher baseline fluid content in both legs as compared with non-OSA subjects, which may be the basic factor with regards to fluid shifts in OSA patients. The increase in head and neck fluid shift volume did not directly correlate with the severity of OSA.
Keywords: Obstructive sleep apnea (OSA), fluid shift, body composition analysis
Introduction
Obstructive sleep apnea (OSA) is caused by repetitive occlusion of the upper airway during sleep. A number of factors contribute to upper airway obstruction in OSA patients. The prevalence of OSA increases with increasing body mass index (BMI) and neck circumference (NC) (1,2). However, only one-third of the variability in sleep apnea severity is attributable to these two indices of obesity (3,4). Therefore, factors other than obesity must play a role in the pathogenesis of OSA (5).
Previous studies have shown that fluid accumulates in the legs due to gravity during the daytime, and is redistributed when with supine positioning at night (6). A portion of the shifted fluid accumulates in the neck, and narrows the upper airway, predisposing the patient to OSA (3,7).
Some self-controlled studies have found that the fluid shifts from the legs to the neck existed in healthy subjects (5), OSA patients (7), and heart failure patients (8). However, the characteristics of baseline body fluid content and nocturnal fluid shift between non-OSA and OSA patients remain unclear.
We speculate that OSA patients have more fluid shifts from the legs to the neck as compared with healthy control subjects, which increase the severity of OSA. We designed this study to test our hypothesis. In this study, we measured baseline body fluids distribution and nocturnal fluid shifts by bioelectrical impedance analysis (InBody 720 system, Biospace Co., Ltd., Korea).
Methods
Subjects
This case-controlled study was performed at the Sleep Medicine Center of The First Affiliated Hospital with Nanjing Medical University. Thirty-six outpatients were enrolled in this study between February 2013 and January 2014.
OSA group inclusion criteria were as follows: age from 18 to 70 years, and an apnea-hypopnea index (AHI) ≥5/hour according to the results of polysomnography (PSG). Age and BMI-matched subjects without OSA (AHI <5) were recruited for the control group. Exclusion criteria for both groups were: (I) a history of stroke or clinical signs of peripheral or central nervous system disorders; (II) previously known heart failure, coronary heart disease, or myocardial infarction; (III) chronic obstructive pulmonary disease or history of asthma; (IV) a history of allergy to a local anesthetic; and (V) pregnancy.
This protocol was approved by the Clinical Study Ethics Committee of The First Affiliated Hospital of Nanjing Medical University. All subjects provided written informed consent prior to study participation.
Polysomnography (PSG)
An evaluation of daytime sleepiness based on the Epworth Sleepiness Scale (ESS) score was calculated prior to the sleep study, which was performed by unattended overnight PSG (Embla S4500 System, USA) as described previously (9). PSG was monitored using five electroencephalographic channels (F4-M1, C4-M1, O2-M1, E12-M2, and E2-M2) and a submental electromyogram. Nasal airflow was measured by continuously recording nasal pressure, snoring, pulse oximetry, and body position, as well as chest and abdominal effort. Analyses were performed by two physicians who specialized in sleep medicine, but were not directly involved in this study.
We used the 2012 standard of the American Academy of Sleep Medicine for scoring sleep apnea events (10): obstructive apnea: complete cessation of airflow with continued paradoxical chest and abdominal excursion for ≥10 s; hypopnea: reduction of airflow >50% baseline lasting ≥10 s and associated with ≥4% desaturation. The AHI was defined as the number of apneas and hypopneas per hour of sleep. A patient with an AHI ≥5/hour was considered to have OSA.
Body fluid analysis
The human body is composed of water, protein, fat, and minerals. These compositions have different bioelectrical impedance. Water and fat content in each part of the body can be detected by bioelectrical impedance analysis. As a user-friendly, safe, simple, and non-invasive technology, bioelectrical impedance analysis can be performed quickly and repeated at short time intervals (11).
Two body fluid analyses were performed using the InBody 720 system (Biospace) for each enrolled subject. The first measurement (pre-sleep, baseline data) was carried out between 21:00 and 22:00 o’clock and the second measurement (post-sleep) was performed when the subject awakened. In order to improve the accuracy of measurement results, fasting was required 3 hours before fluid analyses. The patients were required to empty their bladders, remove their coats and footwear, and stand on the equipment. Subjects were asked to hold the detection handles while breathing calmly. Data would be automatically generated in one minute. The parameters consisted of weight, BMI, NC, left arm fluid index, right arm fluid index, trunk fluid index, left leg fluid index, and right leg fluid index. Fluid shift volume (Δ, mL) for each part of the body was calculated with the formula: (pre-sleep fluid index - post-sleep fluid index) × body weight (kg) ×1,000. Since the fluid index on the head and neck can not be directly detected, we calculated the fluid shift volume by the difference between the other five parts (the total decrease in fluid volume from the legs equals the total increase of fluid volume of the trunk, arms, and head and neck). The formula used for calculation was: Δ left leg + Δ right leg − Δ left arm − Δ right arm − Δ trunk.
Statistical analysis
Statistical analyses were performed using SPSS statistical software (SPSS, Inc., Chicago, IL, USA). Data were expressed as the mean ± standard deviation for normally distributed and as median (interquartile range) for nonnormally distributed, and counts (percentages) for qualitative data. Parameters compared between OSA and non-OSA subjects using the independent t-test for normally distributed data or Mann-Whitney U test for nonnormally distributed data. Pearson correlation analysis was used to analyze the relationship between sleep parameters [AHI, oxygen desaturation index (ODI), mean and minimal SpO2] and body composition parameters (change in NC, baseline fluid index, fluid shift volumes) adjusted by age, gender and BMI.
Results
Each group had 18 subjects (17 male and 1 female). The average age was 47.67±9.99 years in the OSA group and 49.28±7.18 in the non-OSA group. There were no differences in age, weight, and BMI between groups (Table 1).
Table 1. Baseline characteristics between OSA and non-OSA subjects.
| Characteristics | OSA | non-OSA | T | P |
|---|---|---|---|---|
| Male, n (%) | 17 (94.4) | 17 (94.4) | χ2=0.000 | 1 |
| Age (year) | 47.67±9.99 | 49.28±7.18 | −0.556 | 0.582 |
| Weight (kg) | 79.02±6.74 | 76.13±5.92 | 1.367 | 0.181 |
| BMI (kg/m2) | 26.52±1.73 | 26.24±1.78 | 0.49 | 0.628 |
| NC (cm) | 38.42±1.74 | 37.90±1.29 | 1.012 | 0.319 |
| Sleep time (min) | 412.33±37.05 | 411.22±38.53 | 0.088 | 0.93 |
| ESS score | 10.78±4.41 | 4.56±2.55 | 5.18 | <0.001 |
| AHI, h−1 | 42.00±20.19 | 2.06±1.51 | 8.371 | <0.001 |
| ODI, h−1 | 32.61±18.50 | 1.11±1.23 | 7.208 | <0.001 |
| Mean SpO2 (%) | 93.89±2,87 | 97.89±0.68 | −5.760 | <0.001 |
| Minimal SpO2 (%) | 72.94±8.86 | 91.33±3.11 | −8.309 | <0.001 |
OSA, obstructive sleep apnea; BMI, body mass index; NC, neck circumference; ESS, Epworth Sleepiness Scale; AHI, apnea-hypopnea index; ODI, oxygen desaturation index.
The changes of fluid volume between two groups are shown in Table 2. There were no significant differences between groups with regards to changes in weight and BMI (all P>0.05). OSA patients had a significant increase in NC than non-OSA subjects (P<0.01). There were no significant differences between OSA and non-OSA subjects in fluid volume changes in the arms, trunk and head and neck (all P>0.05). As compared with non-OSA subjects, OSA patients had remarkably reduced fluid volume in both legs.
Table 2. Comparisons of fluid volume change between OSA and non-OSA subjects.
| Characteristics | OSA | non-OSA | Z | P |
|---|---|---|---|---|
| Change in weight (kg) | −0.250 (−0.175, −0.325) | −0.200 (−0.200, −0.100) | 1.829 | 0.079 |
| Change in BMI (kg/m2) | −0.010 (0.000, −0.020) | −0.010 (−0.010, −0.000) | 1.148 | 0.279 |
| Change in NC (cm) | +0.600 (0.200, 1.225) | +0.250 (−0.100, 0.325) | −2.825 | 0.004 |
| Δ left arm (mL) | +79.50 (−19.65, 161.55) | +0.000 (−148.50, 78.53) | −0.794 | 0.443 |
| Δ right arm (mL) | +80.25 (−19.45, 155.75) | +67.25 (−78.18, 97.18) | −1.140 | 0.265 |
| Δ trunk (mL) | +79.65 (0.000, 156.00) | +0.000 (−78.20, 96.80) | −1.665 | 0.097 |
| Δ left leg (mL) | −160.50 (−252.83, −77.80) | −80.45 (−181.50, 109.75) | 2.184 | 0.029 |
| Δ right leg (mL) | −272.35 (334.20, −214.23) | −136.50 (−164.50, 0.000) | 3.928 | <0.001 |
| Δ head and neck (mL)# | +241.20 (138.65, 346.90) | +64.70 (−171.75, 302.60) | −1.614 | 0.111 |
OSA, obstructive sleep apnea; BMI, body mass index; NC, neck circumference. Δ fluid shift volume: +, increase fluid volume; −, decrease fluid volume; #, calculation of Δ head and neck: Δ left leg + Δ right leg − Δ left arm − Δ right arm − Δ trunk.
By comparing of the fluid indices between the two groups, we found that OSA patients had higher left and right legs fluid volume indices than that in non-OSA subjects (both P<0.05). Detailed information is shown in Table 3.
Table 3. Comparisons of baseline fluid indices between OSA and non-OSA subjects.
| Baseline fluid indices | OSA | non-OSA | t | P |
|---|---|---|---|---|
| Left arm fluid index | 0.377±0.004 | 0.374±0.004 | 1.902 | 0.066 |
| Right arm fluid index | 0.377±0.004 | 0.375±0.004 | 1.629 | 1.112 |
| Trunk fluid index | 0.377±0.007 | 0.374±0.006 | 1.26 | 0.216 |
| Left leg fluid index | 0.380±0.006 | 0.375±0.006 | 2.118 | 0.042 |
| Right leg fluid index | 0.382±0.007 | 0.377±0.006 | 2.472 | 0.019 |
OSA, obstructive sleep apnea.
Table 4 showed the correlations between PSG parameters and baseline fluid index. There were significant correlations between AHI and left arm fluid index (r=0.500, P<0.01) and right leg fluid index (r=0.553, P<0.01). There were inverse relationships found between mean and minimal SpO2 and left arm fluid index (r=−0.413, P<0.01; r=0.391, P<0.05) left leg fluid index (r=0.439, P<0.01; r=0.436, P<0.01) and right leg fluid index (r=0.527, P<0.01; r=0.535, P<0.01).
Table 4. Correlations between PSG parameters and baseline fluid indices.
| Variables | Left arm fluid index | Right arm fluid index | Trunk fluid index | Left leg fluid index | Right leg fluid index |
|---|---|---|---|---|---|
| AHI | 0.465** | 0.361* | 0.343* | 0.500** | 0.553** |
| ODI | 0.379* | 0.251 | 0.223 | 0.410** | 0.451** |
| Mean SpO2 | −0.413** | −0.298 | −0.338 | −0.439** | −0.527** |
| Minimal SpO2 | −0.391* | −0.329 | −0.318 | −0.436** | −0.535** |
Note: the values in the table are Pearson correlation coefficient(r) adjusted by age, gender and body mass index; *, P<0.05; **, P<0.01. PSG, polysomnography; AHI, apnea-hypopnea index; ODI, oxygen desaturation index.
Relationships between PSG parameters and change in body composition parameters are shown in Table 5. AHI correlated significantly with an increase in NC (r=0.696, P<0.01), increase in fluid volume in the trunk (r=0.348, P<0.05), and decrease in fluid volume in both legs (r=0.525, P<0.01; r=0.780, P<0.01). However, there was no significant correlation between AHI and change in fluid volume in the head and neck (r=0.333, P>0.05). The significant correlation between the change in NC and the increase fluid shift volume in head and neck was found (r=0.493, P<0.01; Figure 1): the more fluid shift volume in the head and neck, and the greater the increase in NC.
Table 5. Correlations between PSG parameters and changes in body compositions parameters.
| Variables | Change in NC | Δ left arm | Δ right arm | Δ trunk | Δ left leg | Δ right leg | Δ head and neck |
|---|---|---|---|---|---|---|---|
| AHI | 0.696** | 0.263 | 0.307 | 0.348* | −0.525** | −0.780** | 0.333 |
| ODI | 0.634** | 0.188 | 0.266 | 0.342* | −0.490* | −0.690** | 0.324 |
| Mean SpO2 | −0.577** | −0.161 | −0.279 | −0.133 | 0.433* | 0.711** | −0.398* |
| Minimal SpO2 | −0.518** | −0.216 | −0.240 | −0.204 | 0.426** | 0.771** | −0.391* |
Δ fluid shift volume; the values in the table are Pearson correlation coefficient(r) adjusted by age, gender and body mass index; *, P<0.05; **, P<0.01. PSG, polysomnography; NC, neck circumference; AHI, apnea-hypopnea index; ODI, oxygen desaturation index.
Figure 1.
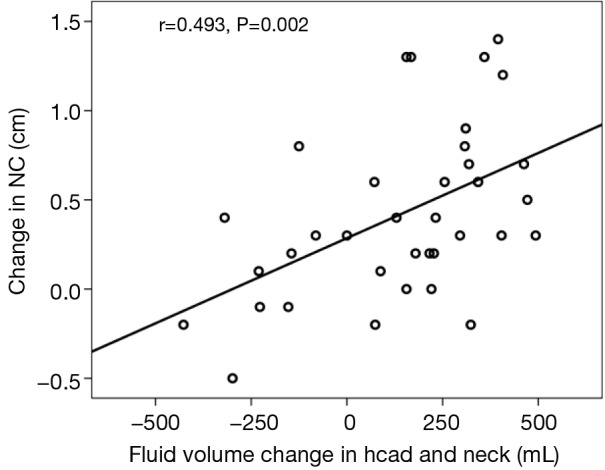
Correlation between overnight change in fluid volume in the head and neck and change in NC. NC, neck circumference.
Discussion
Although the key pathology of OSA is repetitive collapse of the upper airway during sleep, the cause of upper airway collapse remains unclear. It is well known that obesity, male gender, age, upper airway anatomic abnormalities and genetic factors are the risk factors for OSA. Recent studies have shown that fluid shifts from the legs to the neck may play an important role in upper airway collapse. The researchers found that fluid displacement from the legs by inflation of anti-shock trousers increased NC, narrowed the pharynx, and increased its collapsibility in awake healthy subjects (3,7,12). They speculated that rostral fluid displacement into the neck contributed to the pathogenesis of OSA. The same team then found that nocturnal rostral fluid shifts contribute to the pathogenesis of both OSA and CSA in patients with heart failure, and that the magnitude of the fluid shifts was directly related to the severity of the sleep apnea (5,8).
Our study provided several novel findings on the pathogenesis of OSA, and suggested new approaches to its prevention and therapy. We accurately analyzed fluid shifts in each part of body and found that OSA patients had more fluid shifts from the legs (especially the right leg) to the neck (increasing in NC, P<0.05) than non-OSA subjects (Table 2).
In order to clarify why OSA patients had more fluid shifts than the control subjects, we analyzed baseline fluid index, which indicates the “water content” in each part of the body. We found that OSA patients had a higher baseline fluid index in both left and right legs compared with non-OSA subjects (Table 3). Although White et al. (13) investigated the fluid shift in men with and without OSA, he did not find a difference in leg fluid volume between the two groups. Our results suggest that OSA patients may have mild edema which does not affect normal physiological function in daily life, but lead to more nocturnal fluid shifts. According to this hypothesis, during the day, more fluid accumulates in the intravascular and interstitial spaces of the legs due to gravity, and redistributes rostrally when supine at night, again due to gravity. Some of this fluid may accumulate in the neck, increasing tissue pressure and causing the upper airway to narrow, predisposing to OSA (3,6,7,12).
We also found the direct evidence that an increase in fluid volume in head and neck leads to an increase in NC (Figure 1). AHI significantly correlated with change in NC, but not with fluid volume change in the head and neck (Table 5). Thus, it can be seen that fluid shifts accumulating in the head and neck is not closely related to OSA. The occurrence of OSA depends on where the fluid accumulates. The tendency is for more fluid accumulation in the peripharyngeal tissues, but not the craniofacial subcutaneous soft tissue, and is thus easier to cause upper airway collapse and OSA.
Kasai and Bucca found that diuretic therapy reduced AHI, overnight shifts in leg fluid volume and an increase in NC (14,15). Compression stockings, which prevent daytime leg fluid accumulation and reduce overnight rostral fluid shifts, reduced the AHI in patients with chronic venous insufficiency and OSA (16). Here, we observed that OSA patients had more baseline fluid retention. Considering the present information, we suggest that body composition analysis should be provided for OSA patients, and followed by diuretic therapy with more baseline fluid retention.
There are several potential limitations in this study. First, a previous study documented that fluid displacement into the peripharyngeal soft tissues leads to increased airflow resistance of the pharynx (3,13); however, we did not measure airflow resistance of the pharynx. Second, it is well known that continuous positive airway pressure (CPAP) treatment contributes to the reduction of AHI. However, a recent study documented that the reduction in AHI, as a response to CPAP treatment, correlated with the degree of decrease in NC overnight. Whether the effect of preventing fluid accumulation in the neck is due to preventing fluid shifts from the legs or not remains unclear. Further investigation is needed.
Conclusions
In summary, OSA patients had more baseline fluid content in both legs compared with non-OSA subjects, which may be the basic factor for the increase in fluid shift seen from the legs to the neck during sleep in OSA patients. Since the increase of fluid shift volume in the head and neck did not directly correlate with OSA severity, it has been suggested that the destination of the fluid shifts and accumulation is important in the development of sleep apnea.
Acknowledgements
The authors thank head nurse Xi Dai (Department of Geriatric Medicine) for her assistance in measuring body composition.
Funding: This study was supported by Jiangsu Provincial Department of Education (No. CXLX12_0557) and Jiangsu Provincial Special Program of Medical Science (No. BL2012012).
Disclosure: The authors declare no conflict of interest.
References
- 1.Sakakibara H, Tong M, Matsushita K, et al. Cephalometric abnormalities in non-obese and obese patients with obstructive sleep apnoea. Eur Respir J 1999;13:403-10. [DOI] [PubMed] [Google Scholar]
- 2.Hoffstein V, Szalai JP. Predictive value of clinical features in diagnosing obstructive sleep apnea. Sleep 1993;16:118-22. [PubMed] [Google Scholar]
- 3.Chiu KL, Ryan CM, Shiota S, et al. Fluid shift by lower body positive pressure increases pharyngeal resistance in healthy subjects. Am J Respir Crit Care Med 2006;174:1378-83. [DOI] [PubMed] [Google Scholar]
- 4.Dempsey JA, Skatrud JB, Jacques AJ, et al. Anatomic determinants of sleep-disordered breathing across the spectrum of clinical and nonclinical male subjects. Chest 2002;122:840-51. [DOI] [PubMed] [Google Scholar]
- 5.Redolfi S, Yumino D, Ruttanaumpawan P, et al. Relationship between overnight rostral fluid shift and Obstructive Sleep Apnea in nonobese men. Am J Respir Crit Care Med 2009;179:241-6. [DOI] [PubMed] [Google Scholar]
- 6.White LH, Bradley TD. Role of nocturnal rostral fluid shift in the pathogenesis of obstructive and central sleep apnoea. J Physiol 2013;591:1179-93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Su MC, Chiu KL, Ruttanaumpawan P, et al. Lower body positive pressure increases upper airway collapsibility in healthy subjects. Respir Physiol Neurobiol 2008;161:306-12. [DOI] [PubMed] [Google Scholar]
- 8.Yumino D, Redolfi S, Ruttanaumpawan P, et al. Nocturnal rostral fluid shift: a unifying concept for the pathogenesis of obstructive and central sleep apnea in men with heart failure. Circulation 2010;121:1598-605. [DOI] [PubMed] [Google Scholar]
- 9.Ding N, Ni BQ, Zhang XL, et al. Prevalence and Risk Factors of Sleep Disordered Breathing in Patients with Rheumatic valvular Heart Disease. J Clin Sleep Med 2013;9:781-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Berry RB, Budhiraja R, Gottlieb DJ, et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med 2012;8:597-619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tajiri Y, Kato T, Nakayama H, et al. Reduction of skeletal muscle, especially in lower limbs, in Japanese type 2 diabetic patients with insulin resistance and cardiovascular risk factors. Metab Syndr Relat Disord 2010;8:137-42. [DOI] [PubMed] [Google Scholar]
- 12.Shiota S, Ryan CM, Chiu KL, et al. Alterations in upper airway cross-sectional area in response to lower body positive pressure in healthy subjects. Thorax 2007;62:868-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.White LH, Motwani S, Kasai T, et al. Effect of rostral fluid shift on pharyngeal resistance in men with and without obstructive sleep apnea. Respir Physiol Neurobiol 2014;192:17-22. [DOI] [PubMed] [Google Scholar]
- 14.Kasai T, Bradley TD, Friedman O, et al. Effect of intensified diuretic therapy on overnight rostral fluid shift and obstructive sleep apnoea in patients with uncontrolled hypertension. J Hypertens 2014;32:673-80. [DOI] [PubMed] [Google Scholar]
- 15.Bucca CB, Brussino L, Battisti A, et al. Diuretics in obstructive sleep apnea with diastolic heart failure. Chest 2007;132:440-6. [DOI] [PubMed] [Google Scholar]
- 16.Redolfi S, Arnulf I, Pottier M, et al. Attenuation of obstructive sleep apnea by compression stockings in subjects with venous insufficiency. Am J Respir Crit Care Med 2011;184:1062-6. [DOI] [PubMed] [Google Scholar]


